Conclusions
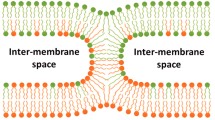
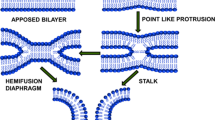
The results reviewed suggest that membrane fusion in diverse biological fusion reactions involves formation of some specific intermediates: stalks and pores. Energy of these intermediates and, consequently, the rate and extent of fusion depend on the propensity of the corresponding monolayers of membranes to bend in the required directions.
Proteins and peptides can control the bending energy of membrane monolayers in a number of ways. Monolayer lipid composition may be altered by different phospholipases [50, 85, 90], flipases and translocases [4, 50]. Proteins and peptides can change monolayer spontaneous curvature or hydrophobic void energy by direct interaction with membrane lipids [20, 32, 111]. Proteins may also provide some barriers for lipid diffusion in the plane of the monolayer [83, 141]. If diffusion of lipids at some specific membrane sites (e.g., in the vicinity of fusion protein) is somehow hindered, the energy of the bent fusion intermediates would reflect the elastic properties of these particular sites rather than the spontaneous curvature of the whole monolayers. Proteins may deform membranes while bringing them locally into close contact. The alteration of the geometric (external) curvature will certainly change the elastic energy of the initial state and, thus affect the energetic barriers of the formation of the intermediates [143]. In addition, the area and the energy of the stalk can be reduced by preliminary bending of the contacting membranes [111]. The possible effects of proteins and polymers on local elastic properties and local shapes of the membranes have been recently analyzed [22, 39, 45, 63]. These studies may provide a good basis for future development of theoretical models of protein-mediated fusion.
Various models for biological fusion have been presented as hypothetical sequences of intermediate conformations of proteins, with membrane lipids just covering the empty spaces between the proteins. Although the results discussed above do not allow us to draw an allexplaining cartoon of the fusion mechanism, they do indicate which properties of membrane lipid bilayers (if modified by fusion proteins) would get these bilayers to fuse. In addition, these data suggest a specific geometry to bent fusion intermediates (stalks and pores) and imply a contribution by lipids to the energy of these intermediates. We think that the synthesis of rapidly developing structural information on fusion proteins with the analysis of the physics of membrane rearrangement may soon yield a real understanding of the fascinating and fundamental phenomenon of membrane fusion.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Almers, W., Tse, F.W. 1990. Neuron 4:813–818
Bentz, J., Ellens, H., Lai, M.Z., Szoka, F.C., Jr. 1985. Proc. Natl. Acad. Sci. USA 82:5742–5745
Bienvenue, A., Sainte-Marie, J. 1994. In: Current Topics in Membranes. D. Hoekstra, editor. pp. 319–354. Academic Press, San Diego
Bogdanov, A., Verhoven, B., Schlegel, R.A., Williamson, P. 1993. Biochem. Soc. Trans. 21:271–275
Breckenridge, L.J., Almers, W. 1987. Nature 328:814–817
Bron, R., Wahlberg, J.M., Garoff, H., Wilschut, J. 1993. EMBO J. 12:693–701
Bullough, P.A., Hughson, F.M., Skehel, J.J., Wiley, D.C. 1994. Nature 371:37–43
Chandler, D.E., Bennett, J.P., Gomperts, B. 1983. J. Ultrastruct. Res. 82:221–232
Chandler, D.E., Heuser, J.E. 1980. J. Cell Biol 86:666–674
Chen, S.Y., Cheng, K.H., Ortalano, D.M. 1990. Chem. Phys. Lipids 53:321–329
Chernomordik, L.V., Chanturiya, A.N., Green, J., Zimmerberg, J. 1994. Biophys. J. 66:A144.
Chernomordik, L.V., Kozlov, M.M., Leikin, S.L., Markin, V.S., Chizmadzhaev Yu, A. 1986. Dokl Akad Nauk SSSR 288:1009–1013
Chernomordik, L.V., Leikina, E.A., Zimmerberg, J. 1995. Virology 69:3049–3058
Chernomordik, L.V., Melikyan, G.B., Abidor, I.G., Markin, V.S., Chizmadzhev, Y.A. 1985. Biochim. Biophys. Acta 812:643–655
Chernomordik, L.V., Melikyan, G.B., Chizmadzhev, Y.A. 1987. Biochim. Biophys. Acta 906:309–352
Chernomordik, L.V., Vogel, S.S., Sokoloff, A., Onaran, H.O., Leikina, E.A., Zimmerberg, J. 1993. FEBS. Lett. 318:71–76
Cohen, F.S., Akabas, M.H., Zimmerberg, J., Finkelstein, A. 1984. J. Cell Biol. 98:1054–1062
Conti, C., Mastromarino, P., Orsi, N. 1991. Comp. Immunol. Microbiol. Infect. Dis. 14:303–313
Creutz, C.E. 1981. J. Cell Biol. 91:247–256
Cullis, P.R., Tilcock, C.P., Hope, M.J. 1991. In: Membrane Fusion. J. Wilschut, D. Hoekstra, editors. pp. 35–64. Marcel Dekker, The Netherlands
Curran, M.J., Cohen, F.S., Chandler, D.E., Munson, P.J., Zim-merberg, J. 1993. J. Membrane Biol. 133:61–75
Dan, N., Berman, A., Pincus, P., Safran, S.A. 1994. J. Phys. 4:1713–1725
Devaux, P.F. 1992. Annu. Rev. Biophys. Biomol. Struct. 21:417–439
Duzgunes, N., Straubinger, R.M., Baldwin, P.A., Friend, D.S., Papahadjopoulos, D. 1985. Biochemistry 24:3091–3098
Duzgunes, N., Wilschut, J., Fraley, R., Papahadjopoulos, D. 1981. Biochim. Biophys. Acta 642:182–195
Elhai, J., Scandella, C.J. 1983. Exp. Cell. Res. 148:63–71
Ellens, H., Bentz, J., Szoka, F.C. 1986. Biochemistry 25:285–294
Epand, R.M. 1985. Biochemistry 24:7092–7095
Epand, R.M. 1986. Biosci. Rep. 6:647–653
Epand, R.M., Bottega, R. 1987. Biochemistry 26:1820–1825
Epand, R.M., Cheetham, J.J., Epand, R.F., Yeagle, P.L., Richardson, C.D., Rockwell, A. Degrado, W.F. 1992. Biopolymers 32:309–314
Epand, R.M., Epand, R.F. 1994. Biochem. Biophys. Res. Commun. 202:1420–1425
Epand, R.M., Epand, R.F., Ahmed, N., Chen, R. 1991. Chem. Phys. Lipids 57:75–80
Finkelstein, A., Zimmerberg, J., Cohen, F.S. 1986. Annu. Rev. Physiol. 48:163–174
Gawrisch, K., Parsegian, V.A., Hajduk, D.A., Tate, M.W., Gruner, S.M., Fuller, N.L., Rand, R.P. 1992. Biochemistry 31:2856–2864
Gelbart, W.M., Roux, D., Ben-Shaul, A. 1994. Modern Ideas and Problems in Amphiphilic Sciences. Springer; Berlin.
Glick, B.S., Rothman, J.E. 1987. Nature 326:309–312
Golan, D.E., Furlong, S.T., Brown, C.S., Caulfield, J.P. 1988. Biochemistry 27:2661–2667
Goulian, M., Bruinsma, R., Pincus, P. 1993. Europhys. Lett. 22:145–150
Gruner, S.M. 1985. Proc. Natl. Acad. Sci. USA 82:3665–3669
Gruner, S.M., Cullis, P.R., Hope, M.J., Tilcock, C.P. 1985. Annu. Rev. Biophys. Biophys. Chem. 14:211–238
Harbich, W., Servuss, R.M., Helfrich, W. 1978. Z. Naturforsch. 33a:1013–1017
Helfrich, W. 1973. Z. Naturforsch. 28c:693–703
Helfrich, W. 1978. Z. Naturforsch. 33a:305–315
Helfrich, W., Kozlov, M.M. 1994. J. Phys. 4:1427–1438
Helm, C.A., Israelachvili, J.N., McGuiggan, P.M. 1989. Science 246:919–922
Helm, C.A., Israelachvili, J.N., McGuiggan, P.M. 1992. Biochemistry 31:1194–1805
Herrmann, A., Clague, M.J., Blumenthal, R. 1993. Membr. Biochem. 10:3–15
Herrmann, A., Clague, M.J., Puri, A., Morris, S.J., Blumenthal, R., Grimaldi, S. 1990. Biochemistry 29:4054–4058
Hinkovska-Galchev, V., Srivastava, P.N. 1992. Mol. Reprod. Dev. 33:281–286
Hoekstra, D., Kok, J.W. 1989. Biosci. Rep. 9:273–305
Hope, M.J., Cullis, P.R. 1981. Biochim. Biophys. Acta 640:82–90
Hui, S.W., Sen, A. 1989. Proc. Natl. Acad. Sci. USA 86:5825–5829
Hui, S.W., Stewart, T.P., Boni, L.T., Yeagle, P.L. 1981. Science 212:921–923
Israelachvili, J.N., Mitchell, D.J., Ninham, B.W. 1976. J. Chem. Soc., Faraday Trans. 2 72:1525–1568
Kelsey, D.R., Flanagan, T.D., Young, J.E., Yeagle, P.L. 1991. Virology 182:690–702
Kemble, G.W., Danieli, T., White, J.M. 1994. Cell 76:383–391
Kielian, M.C., Helenius, A. 1984. J. Virol. 52:281–283
Kirk, G.L., Gruner, S.M., Stein, S.L. 1984. Biochemistry 23:1093–1102
Klausner, R.D., Bhalla, D.K., Dragsten, P., Hoover, R.L., Karnovsky, M.J. 1980. Proc. Natl. Acad. Sci. USA 77:437–441
Knoll, G., Braun, C., Plattner, H. 1991. J. Cell Biol. 113:1295–1304
Koynova, R., Caffrey, M. 1994. Chem. Phys. Lipids 69:1–34
Kozlov, M.M., Helfrich, W. 1993. Langmuir 9:2761–2763
Kozlov, MM, Leikin, S., Rand, R.P. 1994. Biophys. J. 67:1603–1611
Kozlov, M.M., Leikin, S.L., Chernomordik, L.V., Markin, V.S., Chizmadzhev, Y.A. 1989. Eur. Biophys. J. 17:121–129
Kozlov, M.M., Leikin, S.L., Markin, V.S. 1989. J. Chem. Soc. Faraday Trans. 2 85:277–292
Kozlov, M.M., Markin, V.S. 1983. Biofizika 28:255–261
Kozlov, M.M., Winterhalter, M. 1991. J. Phys. 1:1085–1100
Kozlov, M.M., Winterhalter, M. 1991. J. Phys. 1:1077–1084
Kumar, V. 1991. Proc. Natl. Acad. Sci. USA 88:444–448
Kumar, V.V., Malewicz, B., Baumann, W.J. 1989. Biophys. J. 55:789–792
Leckband, D.E., Helm, C.A., Israelachvili, J. 1993. Biochemistry 32:1127–1140
Leikin, S.L., Kozlov, M.M., Chernomordik, L.V., Markin, V.S., Chizmadzhev, Y.A. 1987. J. Theor. Biol. 129:411–425
Lentz, B.R. 1994. Chem. Phys. Lipids 73:91–106
Lentz, B.R., McIntyre, G.F., Parks, D.J., Yates, J.C., Massenburg, D. 1992. Biochemistry 31:2643–2653
Liberman, E.A., Nenashev, V.A. 1972. Biofizika 17:1017–1023
Llanos, M.N., Morales, P., Riffo, M.S. 1993. J. Exp. Zool. 267:209–216
Luzatti, V. 1968. In: Biological Membranes. D. Chapman, editor. pp. 71–123. Academic Press, New York.
Markin, V.S., Kozlov, M.M., Borovjagin, V.L. 1984. Gen. Physiol. Biophys. 3:361–377
Martin, I., Dubois, M.C., Saermark, T., Epand, R.M., Ruysschaert, J.M. 1993. FEBS. Lett. 333:325–330
Mayorga, L.S., Colombo, M.I., Lennartz, M., Brown, E.J., Rahman, K.H., Weiss, R., Lennon, P.J., Stahl, P.D. 1993. Proc. Natl. Acad. Sci. USA 90:10255–10259
Meers, P., Hong, K., Papahadjopoulos, D. 1988. Biochemistry 27:6784–6794
Melikyan, G.B., Niles, W.D., Cohen, F.S. 1993. J. Gen. Physiol. 102:1151–1170
Melikyan, G.B., Niles, W.D., Peeples, M.E., Cohen, F.S. 1993. J. Gen. Physiol. 102:1131–1149
Monck, J.R., Fernandez, J.M. 1992. J. Cell Biol. 119:1395–1404
Monck, J.R., Oberhauser, A.F., Alvarez de Toledo, G., Fernandez, J.M. 1991. Biophys. J. 59:39–47
Moreau, P., Morre, D.J. 1991. J. Biol. Chem. 266:4329–4333
Mutz, M., Helfrich, W. 1990. J. Phys. 51:991–1002
Nanavati, C., Markin, V.S., Oberhauser, A.F., Fernandez, J.M. 1992. Biophys. J. 63:1118–1132
Neher, E. 1974. Biochim. Biophys. Acta 373:327–336
Nieva, J.L., Bron, R., Corver, J., Wilschut, J. 1994. EMBO. J. 13:2797–2804
Nieva, J.L., Goni, F.M., Alonso, A. 1993. Biochemistry 32:1054–1058
Nussbaum, O., Rott, R., Loyter, A. 1992. J. Gen. Virol. 72:2831–2837
Oberhauser, A.F., Fernandez, J.M. 1993. Biophys. J. 65:1844–1852
Oberhauser, A.F., Monck, J.R., Fernandez, J.M. 1992. Biophys. J. 61:800–809
Ohki, S. 1984. J. Membrane Biol. 77:265–275
Ornberg, R.L., Reese, T.S. 1981. J. Cell Biol 90:40–54
Paiement, J., Lavoie, C., Gavino, G.R., Gavino, V.C. 1994. Biochim. Biophys. Acta 1190:199–212
Parsegian, V.A., Ninham, B.W. 1971. J. Colloid Interface Sci. 37:332–341
Perin, M.S., MacDonald, R.C. 1989. J. Membrane Biol. 109:221–232
Pinto da Silva, P., Nogueira, M.L. 1977. J. Cell. Biol. 73:161–181
Poole, A.R., Howell, J.I., Lucy, J.A. 1970. Nature 227:810–814
Rand, R.P., Fuller, N.L. 1994. Biophys. J. 66:2127–2138
Rand, R.P., Parsegian, V.A. 1989. Biochim. Biophys. Acta 988:351–376
Rilfors, L., Lindblom, G., Wieslander, A., Christiansson, A. 1984. In: Membrane Fluidity. M. Kates, L.A. Manson, editors. pp. 205–245. Plenum, New York
Roos, D.S., Duchala, C.S., Stephensen, C.B., Holmes, K.V., Choppin, P.W. 1990. Virology 175:345–357
Rosenberg, J., Duzgunes, N., Kayalar, C. 1983. Biochim. Biophys. Acta 735:173–180
Rothman, J.E. 1994. Nature 372:55–63
Sarkar, D.P., Morris, S.J., Eidelman, O., Zimmerberg, J., Blumenthal, R. 1989. J. Cell Biol. 109:113–122
Schudt, C., Pette, D. 1976. Cytobiologie 13:74–84
Seddon, J., Templer, R. 1993. Phil Trans. R. Soc. Lond. A 344:377–401
Siegel, D.P. 1986. Biophys. J. 49:1171–1183
Siegel, D.P. 1993. Biophys. J. 65:2124–2140
Siegel, D.P., Green, W.J., Talmon, Y. 1994. Biophys. J. 66:402–414
Sjolund, M., Lindblom, G., Rilfors, L., Arvidson, G. 1987. Biophys. J. 52:145–153
Song, L.Y., Ahkong, Q.F., Georgescauld, D., Lucy, J.A. 1991. Biochim. Biophys. Acta 1065:54–62
Spruce, A.E., Breckenridge, L.J., Lee, A.K., Almers, W. 1990. Neuron 4:643–654
Spruce, A.E., Iwata, A., White, J.M., Almers, W. 1989. Nature 342:555–558
Stamatatos, L., Duzgunes, N. 1993. J. Gen. Virol. 74:1043–1054
Stegmann, T. 1993. J. Biol. Chem. 268:26886–26892
Stegmann, T., Doms, R.W., Helenius, A. 1989. Annu. Rev. Biophys. Biophys. Chem. 18:187–211
Tse, F.W., Iwata, A., Almers, W. 1993. J. Cell Biol. 121:543–552
Verkleij, A.J., Leunissen-Bijvelt, J., de Kruijff, B., Hope, M., Cullis, P.R. 1984. Ciba Found. Symp. 103:45–59
Vogel, S.S., Chernomordik, L.V., Zimmerberg, J. 1992. J. Biol. Chem. 267:25640–25643
Vogel, S.S., Delaney, K., Zimmerberg, J. 1991. Ann. N.Y. Acad. Sci. 635:35–44
Vogel, S.S., Leikina, E.A., Chernomordik, L.V. 1993. J. Biol. Chem. 268:25764–25768
Voyta, J.C., Slakey, L.L., Westhead, E.W. 1978. Biochem. Biophys. Res. Commun. 80:413–417
Walter, A., Yeagle, P.L., Siegel, D.P. 1994. Biophys. J. 66:366–376
Weaver, J.C., Mintzer, R.A. 1981. Phys. Lett. 86A:57–59
Weiss, C.D., White, J.M. 1993. J. Virol. 67:7060–7066
Weltzien, H.U. 1979. Biochim. Biophys. Acta 559:259–287
Westhead, E.W. 1987. Ann. N.Y. Acad. Sci. 493:92–100
White, J., Helenius, A. 1980. Proc. Natl. Acad. Sci. USA 77:3273–3277
White, J.M. 1992. Science 258:917–924
Wilschut, J. 1991. In: Membrane Fusion. J. Wilschut, D. Hoekstra, editors. pp. 89–126. Marcel Dekker, New York
Wilson, I.A., Skehel, J.J., Wiley, D.C. 1981. Nature 289:366–373
Yeagle, P.L. 1989. Faseb. J. 3:1833–1842
Yeagle, P.L., Smith, F.T., Young, I.E., Flanagan, T.D. 1994. Biochemistry 33:1820–1827
Yeagle, P.L., Young, J., Hui, S.W., Epand, R.M. 1992. Biochemistry 31:3177–3183
Zachowski, A., Henry, J.P., Devaux, P.F. 1989. Nature 340:75–76
Zimmerberg, J. 1993. Methods Enzymol. 221:99–112
Zimmerberg, J., Blumenthal, R., Curran, M., Sarkar, D., Morris, S. 1994. J. Cell Biology. 127:1885–1894
Zimmerberg, J., Curran, M., Cohen, F.S. 1991. Ann. N.Y. Acad. Sci. 635:307–317
Zimmerberg, J., Curran, M., Cohen, F.S., Brodwick, M. 1987. Proc. Natl. Acad. Sci. USA 84:1585–1589
Zimmerberg, J., Vogel, S.S., Chernomordik, L.V. 1993. Annu. Rev. Biophys. Biomol. Struct. 22:433–466
Author information
Authors and Affiliations
Additional information
Special thanks to Drs. G. Melikyan, S. Leikin and P. Blank for their valuable input and critical readings of the manuscript; and to Dr. G. Melikyan for kindly providing his previously unpublished results, presented in Fig. 2. Figure 3 was kindly provided by Dr. S. Hui. We would also like to thank Dr. D. Siegel for very helpful discussions. We are grateful to Drs. R. Holz and J. Wilschut for allowing us to cite their unpublished results. Finally, Lynn Kelly provided invaluable help with preparation of the manuscript.
Rights and permissions
About this article
Cite this article
Chernomordik, L., Kozlov, M.M. & Zimmerberg, J. Lipids in biological membrane fusion. J. Membarin Biol. 146, 1–14 (1995). https://doi.org/10.1007/BF00232676
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00232676