Summary
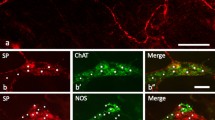
The presence of neurofilament (NF)-like and glial fibrillary acidic protein (GFAP)-like immunoreactivities was studied in sympathetic ganglia of adult rats and guinea pigs during normal conditions and after perturbation. In the superior cervical ganglion (SCG) of normal rats, many ganglion cells and nerve fibers show NF immunoreactivity. Some of these nerve fibers disappear after preganglionic decentralization of SCG; this indicates the presence of a mixture of preand postganglionic NF-positive nerves in the ganglion. Cuts in both preand postganglionic nerves result in a marked increase in GFAP immunoreactivity in SCG, whereas NF immunoreactivity increases in nerve cell bodies after preganglionic cuts. Only a few ganglion cells show NF immunoreactivity in the normal SCG of guinea pig. All intraganglionic NF-positive nerves are of preganglionic origin; decentralization abolishes NF immunoreactivity in these nerve fibers. The inferior mesenteric ganglion, the hypogastric nerves and colonic nerves in guinea pigs contain large numbers of strongly NF-immunoreactive nerve fibers.
When the SCG of adult rat is grafted to the anterior eye chamber of adult rat recipients, both ganglionic cell bodies and nerve fibers, forming on the host iris from the grafted ganglion, are NF-positive. As only the perikarya of these neurons normally exhibit NF immunoreactivity, and the terminal iris arborizations are NF-negative, it appears that the grafting procedure causes NF immunoreactivity to become more widespread in growing SCG neurons.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bignami A, Dahl D (1973) Differentiation of astrocytes in the cerebellar cortex and the pyramidal tracts of the newborn rat. An immunofluorescence study with antibodies to a protein specific to astrocytes. Brain Res 49:393–402
Bignami A, Dahl D (1979) The radial glia of Müller in the rat retina and their response to injury. An immunofluorescence study with antibodies to the glial fibrillary acidic protein. Exp Eye Res 28:63–69
Bignami A, Dahl D, Seiler MW (1980) Neurofilaments in the chick embryo during early development. I. Immunofluorescent study with antisera to neurofilament protein. Dev Neurosci 3:151–161
Björklund H, Dahl D, Haglid K, Rosengren L, Olson L (1983) Astrocytic development in fetal parietal cortex grafted to cerebral and cerebellar cortex of immature rats. Dev Brain Res 9:171–180
Björklund H, Dahl D, Olson L, Seiger Å (1984a) GFA-like immunoreactivity in the iris. Development, distribution and reactive changes following transplantation. J Neurosci 4:978–988
Björklund H, Dahl D, Seiger Å (1984b) Neurofilament and glial fibrillary acidic protein-related immunoreactivity in rodent enteric nervous system. Neuroscience 12:277–287
Björklund H, Bignami A, Dahl D (1985a) Immunohistochemical demonstration of glial fibrillary acidic protein in normal rat Müller glia and retinal astrocytes. Neurosci Lett 54:363–368
Björklund H, Dahl D, Seiger Å (1985b) Immature and mature neurofilament-immunoreactive trigeminal fibers can innervate the iris as studied by intraocular grafting of iris and trigeminal ganglia. Neuroscience 15:841–851
Björklund H, Dalsgaard C-J, Jonsson C-E, Hermansson A (1986) Sensory and autonomic innervation of non-hairy and hairy human skin. An immunohistochemical study. Cell Tissue Res 243:51–57
Coons AH (1958) Fluorescent antibody methods. In: Danielli JF (ed) General cytochemical methods. Academic Press, New York, pp 399–422
Dahl D (1980) Astroglial and axonal proteins in isolated brain filaments. II. Isolation of a 70000-dalton polypeptide from bovine brain filament preparations by immunoaffinity chromatography with antineurofilament antisera. Biochim Biophys Acta 622:9–17
Dahl D (1981) Isolation of neurofilament proteins and of immunologically active neurofilament degradation products from extracts of brain, spinal cord and sciatic nerve. Biochim Biophys Acta 668:299–306
Dahl D, Bignami A (1976) Immunogenic properties of the glial fibrillary acidic protein. Brain Res 116:150–157
Dahl D, Bignami A (1977) Preparation of antisera to neurofilament protein from chicken brain and human sciatic nerve. J Comp Neurol 176:645–658
Dahl D, Bignami A, Bich NT, Chi NH (1981) Immunohistochemical localization of the 150 K neurofilament protein in the rat and the rabbit. J Comp Neurol 195:659–666
Dahl D, Chi NH, Miles LE, Nguyen BT, Bignami A (1982) Glial fibrillary acidic (GFA) protein in Schwann cells: Fact or artifact. J Histochem Cytochem 30:912–918
Dalsgaard C-J, Elfvin L-G (1982) Structural studies on the connectivity of the inferior mesenteric ganglion of the guinea pig. J Auton Nerv Syst 5:265–277
Dalsgaard C-J, Björklund H, Jonsson C-E, Hermansson A, Dahl D (1984) Distribution of neurofilament-immunoreactive nerve fibers in human digital skin. Histochemistry 81:111–114
Eng LF, DeArmond SJ (1981) Glial fibrillary acidic (GFA) protein immunocytochemistry in development and neuropathology. In: Vidio EA, Fedoroff S (eds) Glial and neuronal cell biology. Alan R. Liss, New York, pp 65–79
Falck B (1962) Observations on the possibilities of the cellular localization of monoamines by a fluorescence method. Acta Physiol Scand 56 [Suppl] 197:1–25
Hoffman PN, Lasek RJ (1975) The slow component of axonal transport. Identification of major structural polypeptides of the axon and their generality among mammalian neurons. J Cell Biol 66:351–366
Jessen KR, Mirsky R (1980) Glial cells in the enteric nervous system contain glial fibrillary acidic protein. Nature 268:736–737
Jessen KR, Thorpe R, Mirsky R (1984) Molecular identity, distribution and heterogeneity of glial fibrillary acidic protein: An immunoblotting and immunohistochemical study of Schwann cells, satellite cells, enteric glia and astrocytes. J Neurocytol 13:187–200
Lazarides E (1980) Intermediate filaments as mechanical integrators of cellular space. Nature 283:249–256
Malmfors T (1965) Studies on adrenergic nerves. The use of rat and mouse iris for direct observations on their physiology and pharmacology at cellular and subcellular levels. Acta Physiol Scand Suppl 248:1–93
Olson L, Seiger Å, Strömberg l (1983) Intraocular transplantation in rodents. A detailed account of the procedure and examples of its use in neurobiology with special reference to brain tissue grafting. In: Fedoroff S, Hertz L (eds) Advances in cellular neurobiology, vol 4, Academic Press, New York, pp 407–442
Schlaepfer WW (1977) Immunological and ultrastructural studies of neurofilaments isolated from rat peripheral nerve. J Cell Biol 74:226–240
Schlaepfer WW, Lynch RG (1977) Immunofluorescence studies of neurofilaments in the rat and human peripheral nervous system. J Cell Biol 74:241–250
Seiger Å, Dahl D, Ayer-LeLievre Ch, Björklund H (1984) Appearance and distribution of neurofilament immunoreactivity in iris nerves. J Comp Neurol 223:457–470
Shaw G, Osborn M, Weber K (1981) Arrangement of neurofilaments, microtubules and microfilament-associated proteins in cultured dorsal root ganglia cells. Eur J Cell Biol 24:20–27
Strömberg I, Björklund H, Dahl D, Jonsson G, Sundström E, Olson L (1986) Astrocyte responses to dopaminergic denervations by 6-hydroxydopamine and l-methy1–4-phenyl-1,2,3,6-tetrahydropyridine as evidenced by glial fibrillary acidic protein immunohistochemistry. Brain Res Bull 17:225–236
Yen S-H, Fields KL (1981) Antibodies to neurofilament, glial filament and fibroblast intermediate filament proteins bind to different cell types of the nervous system. J Cell Biol 88:115–126
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Elfvin, L.G., Björklund, H., Dahl, D. et al. Neurofilament-like and glial fibrillary acidic protein-like immunoreactivities in rat and guinea-pig sympathetic ganglia in situ and after perturbation. Cell Tissue Res. 250, 79–86 (1987). https://doi.org/10.1007/BF00214657
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00214657