Summary
Glial fibrillary acidic protein has been firmly established as the predominant component of astrocyte intermediate filaments. It has also been detected immunohistochemically in the glial cells of the enteric nervous system and some Schwann cells in the P.N.S. The molecular identity of this GFAP immunoreactivity in the P.N.S. has so far not been investigated. This study compares GFAP in the C.N.S. and P.N.S. of adult rats both immunochemically and immunohistochemically.
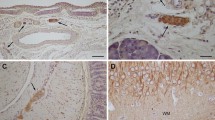
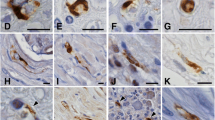
Using SDS polyacrylamide gel electrophoresis combined with immunoblotting, and a polyclonal antiserum to brain GFAP, we show that the peripheral GFAP immunoreactivity resides in a polypeptide with a molecular weight of 49 kd, which is identical to that of rat brain GFAP. Furthermore, we find that this GFAP reactivity can be detected immunohistochemically in Schwann cells in a wide variety of nerves in the P.N.S. and in some satellite cells in both sensory and sympathetic ganglia, in addition to enteric glia. The pattern of distribution of GFAP filaments in Schwann cells suggests that, in the nerves surveyed, they may be expressed by most or all non-myelin forming Schwann cells but not by myelin-forming Schwann cells. We also show, using a monoclonal antibody to GFAP (anti-GFAP-3) in both immunohistochemical and immunoblotting studies, that the GFAP found in most peripheral glia is not identical to that of astrocytes since it lacks an antigenic determinant, defined by this monoclonal antibody, which is present in astrocytes. An exception to this finding is seen in the myenteric plexuses where immunohistochemically detectable GFAP is found in some, but not all, of the enteric glia, using the monoclonal antibody.
Thus, the results suggest that GFA polypeptides may be a heterogeneous group, that share some common determinants and a common molecular weight, and show a widespread and complex distribution in the glia of both the C.N.S. and P.N.S.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Albrechtsen, M., Von Gerstenberg, A. C. &Bock, E. (1983) Mouse monoclonal antibodies reacting with human brain glial fibrillary acidic protein.Journal of Neurochemistry (in press).
Anderton, B. H. (1981) Intermediate filaments: a family of homologous structures.Journal of Muscle Research and Cell Motility 2, 141–66.
Barber, P. C. &Lindsay, R. M. (1982) Schwann cells of the olfactory nerves contain glial fibrillary acidic protein and resemble astrocytes.Neuroscience 7, 3077–99.
Bignami, A., Eng, L. F., Dahl, D. &Uyeda, C. T. (1972) Localization of the glial fibrillary acidic protein in astrocytes by immunofluorescence.Brain Research 43, 429–35.
Coggeshall, R. E., Appelbaum, M. L., Fazen, M., Stubbs, T. B. III &Sykes, M. T. (1975) Unmyelinated axons in human ventral roots, a possible explanation for the failure of dorsal rhizotomy to relieve pain.Brain 98, 157–66.
Dahl, D., Chi, N. H., Miles, L. E., Nguyen, B. T. &Bignami, A. (1982) Glial fibrillaiy acidic (GFA) protein in Schwann cells: fact or artifact.Journal of Histochemistry and Cytochemistry 30, 912–18.
Davison, P. F. &Jones, R. N. (1981) Filament proteins in central, cranial and peripheral mammalian nerves.Journal of Cell Biology.88, 67–72.
Dyck, P. J. &Hopkins, A. P. (1972) Electron microscopic observations on degeneration and regeneration of unmyelinated fibres.Brain 95, 223–34.
Jessen, K. R. &Mirsky, R. (1980) Glial cells in the enteric nervous system contain glial fibrillary acidic protein.Nature 286, 736–37.
Jessen, K. R. &Mirsky, R. (1983) Astrocyte-lrke glia in the peripheral nervous system: an immunohistochemical study of enteric glia.Journal of Neuroscience 3 (in press).
Jessen, K. R., Saffrey, M. J. &Burnstock, G. (1983) The enteric nervous system in tissue culture I. Cell types and their interactions in expiants of the myenteric and submucous plexuses from guinea pig, rabbit and rat.Brain Research 262, 17–35.
Laemmli, U. K. &Favre, M. (1973) Maturation of the head of bacteriophage T4.1.DNA packaging events.Journal of Molecular Biology 80, 575–99.
Langford, L. A. &Coggeshall, R. E. (1981) Branching of sensory axons in the peripheral nerve of rat.Journal of Comparative Neurology 203, 745–50.
Lazarides, E. (1980) Intermediate filaments as mechanical integrators of cellular space.Nature 283, 249–56.
Lee, L. D. &Baden, H. P. (1976) Organization of the polypeptide chains in mammalian keratin.Nature 264, 377–79.
Raff, M. C., Fields, K. L., Hakamori, S.-I., Mirsky, R., Pruss, R. M. &Winter, J. (1979) Cell-type-specific markers for distinguishing and studying neurons and the major classes of glial cells in culture.Brain Research 174, 283–308.
Schachner, M., Hedley-Whyte, E. T., Hsu, D. W., Schoonmaker, G. &Bignami, A. (1977) Ultrastructural localisation of glial fibrillary acidic protein in mouse cerebellum by immunoperoxidase labelling.Journal of Cell Biology 75, 67–73.
Thorpe, R., Delacourte, A., Ayers, M., Bullock, C. &Anderton, B. H. (1979) Polypeptides of isolated brain 10 nm filaments and their association with polymerized tubulin.Biochemical Journal 181, 275–84.
Towbin, H., Staehlin, T. &Gordon, J. (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications.Proceedings of the National Academy of Sciences USA 76, 4350–54.
Yen, S. &Fields, K. L. (1981) Antibodies to neurofilament, glial filament and fibroblast intermediate filament proteins bind to different cell types in the nervous system.Journal of Cell Biology 88, 115–26.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jessen, K.R., Thorpe, R. & Mirsky, R. Molecular identity, distribution and heterogeneity of glial fibrillary acidic protein: an immunoblotting and immunohistochemical study of Schwann cells, satellite cells, enteric glia and astrocytes. J Neurocytol 13, 187–200 (1984). https://doi.org/10.1007/BF01148114
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01148114