Summary
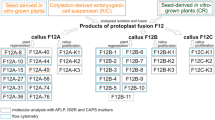
Intergeneric symmetric and asymmetric somatic hybrids have been obtained by fusion of metabolically inactivated protoplasts from embryogenic suspension cultures of tall fescue (Festuca arundinacea Schreb.) and unirradiated or 10–500 Gy-irradiated protoplasts from non-morphogenic cell suspensions of Italian ryegrass (Lolium multiflorum Lam.). Genotypically and phenotypically different somatic hybrid Festulolium mature flowering plants were regenerated.
Species-specific sequences from F. arundinacea and L. multiflorum being dispersed and evenly-represented in the corresponding genomes were isolated and used for the molecular characterization of the nuclear make-up of the intergeneric, somatic Festulolium plants recovered. The irradiation of Italian ryegrass protoplasts with ≤250 Gy X-rays prior to fusogenic treatment favoured the unidirectional elimination of most or part of the donor chromosomes. Irradiation of L. multiflorum protoplasts with 500 Gy produced highly asymmetric (over 80% donor genome elimination) nuclear hybrids and clones showing a complete loss of donor chromosomes.
The RFLP analysis of the organellar composition in symmetric and asymmetric tall fescue (+) Italian ryegrass regenerants confirmed their somatic hybrid character and revealed a bias towards recipient-type organelles when extensive donor nuclear genome elimination had occurred.
Approaches aimed at improving persistence of ryegrasses based on asymmetric somatic hybridization with largely sexually-incompatible grass species (F. rubra and Alopecurus pratensis), and at transferring the cytoplasmic male sterility trait by intra- and inter-specific hybridization in L. multiflorum and L. perenne, have been undertaken.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- cpDNA:
-
chloroplast DNA
- CMS:
-
cytoplasmic male sterility
- 2,4-D:
-
2,4-dichlorophenoxy-acetic acid
- IOA:
-
iodoacetamide
- mtDNA:
-
mitochondrial DNA
- RFLP:
-
restriction fragment length polymorphism
References
Akagi H., M. Sakamoto, T. Negishi & T. Fujimura, 1989. Construction of rice cybrid plants. Mol. Gen. Genet. 215: 501–506.
Creemers-Molenaar J., R.D. Hall & F.A. Krens, 1992. Asymmetric protoplast fusion aimed at intraspecific transfer of cytoplasmic male sterility (CMS) in Lolium perenne L. Theor. Appl. Genet. 84: 763–770.
Crowder L.V., 1953. Interspecific and intergeneric hybrids of Festuca and Lolium. J. Hered. 44: 195–203.
de Lautour G. & B.M. Cooper, 1971. Cold and chemical pretreatments to aid chromosome counts in a grass leaf squash technique incorporating hot pectinase maceration. Stain. Technol. 46: 305–310.
Eizenga G.C. & R.C. Buckner, 1986. Cytological and isozyme evaluation of tall fescue x Italian ryegrass hybrids. Plant Breeding 97: 340–344.
Feinberg A.P. & B. Vogelstein, 1983. A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity. Anal. Biochem. 132: 6–13.
Gleba Y.Y., S. Hinnisdaels, V.A. Sidorov, V.A. Kaleda, A.S. Parokonny, N.V. Boryshuk, N.N. Cherup, I. Negrutiu & M. Jacobs, 1988. Intergeneric asymmetric hybrids between Nicotiana plumbaginifolia and Atropa belladonna obtained by ‘gamma-fusion’. Theor. Appl. Genet. 76: 760–766.
Hayashi Y., J. Kyozuka & K. Shimamoto, 1988. Hybrids of rice (Oryza sativa L.) and wild Oryza species obtained by cell fusion. Mol. Gen. Genet. 246: 6–10.
Kyozuka J., Y. Hayashi & K. Shimamoto, 1987. High frequency plant regeneration from rice protoplasts by novel nurse culture methods. Mol. Gen. Genet. 206: 408–413.
Kyozuka J., T. Kaneda & K. Shimamoto, 1989. Production of cytoplasmic male sterile rice (Oryza sativa L.) by cell fusion. Bio/Technology 7: 1171–1174.
Lichtenstein C. & J. Draper, 1985. Genetic engineering of plants p. 67–119. In: D.M. Glover (Ed). DNA Cloning, Vol. II. IRL Press, Oxford Washington.
Müller A.J. & R. Grafe, 1978. Isolation and characterization of cell lines of Nicotiana tabacum lacking nitrate reductase. Mol. Gen. Genet. 161: 67–76.
Murashige T. & F. Skoog, 1962. A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15: 473–497.
Perez-Vicente R., L. Petris, M. Osusky, I. Potrykus & G. Spangenberg, 1992. Molecular and cytogenetic characterization of repetitive DNA sequences from Lolium and Festuca: applications in the analysis of Festulolium hybrids. Theor. Appl. Genet. 84: 145–154.
Sambrook, J., E.F. Fritsch & T. Maniatis, 1989. Molecular cloning — a laboratory manual. Second edition. Cold Spring Harbor Laboratory Press. CSH.
Sanger F., A.R. Coulson, B.G. Barrel, A.J.H. Smith & B.A. Roe, 1980. Cloning in single-stranded bacteriophage as an aid to rapid DNA sequencing. J. Mol. Biol. 143: 161–178.
Saul M.W. & I. Potrykus, 1984. Species-specific repetitive DNA used to identify interspecific somatic hybrids. Plant Cell Rep. 3: 65–67.
Shillito R.D., J. Paszkowski, M. Müller & I. Potrykus, 1983. Agarose plating and a bead type culture technique enable and stimulate development of protoplast-derived colonies in a number of plant species. Plant Cell Rep. 2: 244–247.
Spangenberg G., M.P. Vallés, Z.Y. Wang, P. Montavon, J. Nagel & I. Potrykus, 1994a. Asymmetric somatic hybridization between tall fescue (Festuca arundinacea Schreb.) and irradiated Italian ryegrass (Lolium multiflorum Lam.) protoplasts. Theor. Appl. Genet. 88: 509–519.
Spangenberg G., Z.Y. Wang, J. Nagel & I. Potrykus, 1994b. Protoplast culture and generation of transgenic plants in red fescue (Festuca rubra L.). Plant Sci. 97: 83–94.
Tabaeizadeh Z., R.J. Ferl & I.K. Vasil, 1986. Somatic hybridization in the Gramineae: Saccharum officinarum L. (Sugarcane) + Pennisetum americanum (L.) K. Schum. (Pearl millet). Proc. Natl. Acad. Sci. USA 83: 5616–5619.
Takamizo T. & G. Spangenberg, 1994. Somatic hybridization in Festuca and Lolium. p. 112–131. In: Y.P.S. Bajaj (Ed). Biotechnology in Agriculture and Forestry, Vol. 27: Somatic hybridization in crop improvement. Springer Verlag, Berlin, Heidelberg.
Takamizo T., G. Spangenberg, K. Suginobu & I. Potrykus, 1991. Intergeneric somatic hybridization in Gramineae: somatic hybrid plants between tall fescue (Festuca arundinacea Schreb.) and Italian ryegrass (Lolium multiflorum Lam.). Mol. Gen. Genet. 231: 1–6.
Takamizo T., K. Suginobu & R. Ohsugi, 1990. Plant regeneration from suspension culture derived protoplasts of tall fescue (Festuca arundinacea Schreb.) of a single genotype. Plant Sci. 72: 125–131.
Terada R., J. Kyozuka, S. Nishibayashi & K. Shimamoto, 1987. Plantlet regeneration from somatic hybrids of rice (Oryza sativa L.) and barnyard grass (Echinochloa oryzicola Vasing). Mol. Gen. Genet. 210: 39–43.
Vasil V., R.J. Ferl & I.K. Vasil, 1988. Somatic hybridization in the Gramineae: Triticum monococcum L. (Einkorn) + Pennisetum americanum (L.) K. Schum. (Pearl millet). J. Plant. Physiol. 132: 160–163.
Wang Z.Y., T. Takamizo, V.A. Iglesias, M. Osusky, J. Nagel, I. Potrykus & G. Spangenberg, 1992. Transgenic plants of tall fescue (Festuca arundinacea Schreb.) obtained by direct gene transfer to protoplasts. Bio/Technology 10: 691–696.
Wang Z.Y., M.P. Vallés, P. Montavon, I. Potrykus & G. Spangenberg, 1993a. Fertile plant regeneration from protoplasts of meadow fescue (Festuca pratensis Huds.). Plant Cell Rep. 12: 95–100.
Wang Z.Y., J. Nagel, I. Potrykus & G. Spangenberg, 1993b. Plants from cell suspension-derived protoplasts in Lolium species. Plant Sci. 94: 179–193.
Yamashita Y., R. Terada, S. Nishibayashi & K. Shimamoto, 1989. Asymmetric somatic hybrids of Brassica: partial transfer of B. campestris genome into B. oleracea by cell fusion. Theor. Appl. Genet. 77: 189–194.
Yang Z.Q., T. Shikanai, K. Mori & Y. Yamada, 1989. Plant regeneration from cytoplasmic hybrids of rice (Oryza sativa L.). Theor. Appl. Genet. 77: 305–310.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Spangenberg, G., Wang, Z.Y., Legris, G. et al. Intergeneric symmetric and asymmetric somatic hybridization in Festuca and Lolium . Euphytica 85, 235–245 (1995). https://doi.org/10.1007/BF00023952
Issue Date:
DOI: https://doi.org/10.1007/BF00023952