Abstract
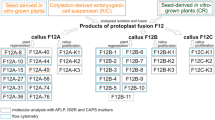
Intergeneric asymmetric somatic hybrids have been obtained by the fusion of metabolically inactivated protoplasts from embryogenic suspension cultures ofFestuca arundinacea (recipient) and protoplasts from a non-morphogenic cell suspension ofLolium multiflorum (donor) irradiated with 10, 25, 50, 100, 250 and 500 Gy of X-rays. Regenerating calli led to the recovery of genotypically and phenotypically different asymmetric somatic hybridFestulolium plants. The genome composition of the asymmetric somatic hybrid clones was characterized by quantitative dot-blot hybridizations using dispersed repetitive DNA sequences specific to tall fescue and Italian ryegrass. Data from dot-blot hybridizations using two cloned Italian ryegrass-specific sequences as probes showed that irradiation favoured a unidirectional elimination of most or part of the donor chromosomes in asymmetric somatic hybrid clones obtained from fusion experiments using donor protoplasts irradiated at doses ≤ 250 Gy. Irradiation of cells of the donor parent with 500 Gy prior to protoplast fusion produced highly asymmetric nuclear hybrids with over 80% elimination of the donor genome as well as clones showing a complete loss of donor chromosomes. Further information on the degree of asymmetry in regenerated hybrid plants was obtained from chromosomal analysis including in situ hybridizations withL. multiflorum-specific repetitive sequences. A Southern blot hybridization analysis using one chloroplast and six mitochondrial-specific probes revealed preferentially recipient-type organelles in asymmetric somatic hybrid clones obtained from fusion experiments with donor protoplasts irradiated with doses higher than 100 Gy. It is concluded that the irradiation of donor cells before fusion at different doses can be used for producing both nuclear hybrids with limited donor DNA elimination or highly asymmetric nuclear hybrid plants in an intergeneric graminaceous combination. For a wide range of radiation doses tested (25–250Gy), the degree of the species-specific genome elimination from the irradiated partner seems not to be dose dependent. A bias towards recipient-type organelles was apparent when extensive donor nuclear genome elimination occurred.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- cpDNA:
-
Chloroplast DNA
- 2, 4-D:
-
2,4-dichlorophenoxyacetic acid
- FDA:
-
fluorescein diacetate
- IOA:
-
iodoacetamide
- mtDNA:
-
mitochondrial DNA
- RFLP:
-
restriction fragment length polymorphism
References
Akagi H, Sakamoto M, Negishi T, Fujimura T (1989) Construction of rice cybrid plants. Mol Gen Genet 215:501–506
Bates GW, Hasenkampf CA, Contolini CL, Piastuch WC (1987) Asymmetric hybridization inNicotiana by fusion of irradiated protoplasts. Theor Appl Genet 74:718–726
Bennett MD, Smith JB, Heslop-Harrison JS (1982) Nuclear DNA amounts in angiosperms. Proc R Soc London Ser B 216:179–199
Creemers-Molenaar J, Hall RD, Krens FA (1992) Asymmetric protoplast fusion aimed at intraspecific transfer of cytoplasmic male sterility (CMS) inLolium perenne L. Theor Appl Genet 84:763–770
Dawson AJ, Jones VP, Leaver CJ (1984) The apocytochrome b gene in maize mitochondria does not contain introns and is preceded by a potential ribosome binding site. EMBO J 3:2107–2113
de Lautour G, Cooper BM (1971) Cold and chemical pretreatments to aid chromosome counts in a grass leaf squash technique incorporating hot pectinase maceration. Stain Technol 46:305–310
Dewey RE, Schuster AM, Levings CS, Timothy DH (1985a) Nucleotide sequence of Fo-ATPase proteolipid (subunit 9) gene of maize mitochondria. Proc Natl Acad Sci USA 82:1015–1019
Dewey RE, Levings CS, Timothy DH (1985b) Nucleotide sequence of ATPase subunit 6 of maize mitochondria. Plant Physiol 79:914–919
Dudits D, Maroy E, Praznovsky T, Olah Z, Gyorgyey J, Cella R (1987) Transfer of resistant traits from carrot into tobacco by asymmetric somatic hybridization: regeneration of fertile plants. Proc Natl Acad Sci USA 84:8434–8438
Famelaer I, Gleba YY, Sidorov VA, Kaleda VA, Parokonny AS, Boryshuk NV, Cherep NN, Negrutiu I, Jacobs M (1989) Intrageneric asymmetric hybrids betweenNicotiana plumbaginifolia andNicotiana sylvestris obtained by ‘gamma-fusion’. Plant Sci 61:105–117
Feinberg AP, Vogelstein B (1983) A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 132:6–13
Fox TD, Leaver CJ (1981) TheZea mays mitochondrial gene coding for cytochrome oxidase subunit II has an intervening sequence and does not contain TGA codons. Cell 26:315–323
Gleba YY, Sytnik KM (1984) Protoplast fusion. Genetic engineering in higher plants. R Shoeman (ed). Springer, Berlin Heidelberg New York
Gleba YY, Hinnisdaels S, Sidorov VA, Kaleda VA, Parokonny AS, Boryshuk NV, Cherup NN, Negrutiu I, Jacobs M (1988) Intergeneric asymmetric hybrids betweenNicotiana plumbaginifolia andAtropa belladonna obtained by ‘gamma-fusion’. Theor Appl Genet 76:760–766
Gupta PP, Schieder O, Gupta M (1984) Intergeneric nuclear gene transfer between somatically and sexually incompatible plants through asymmetric protoplast fusion. Mol Gen Genet 197:30–35
Hayashi Y, Kyozuka J, Shimamoto K (1988) Hybrids of rice (Oryza Sativa L.) and wildOryza species obtained by cell fusion. Mol Gen Genet 246:6–10
Hutchinson J, Rees H, Seal AG (1979) An assay of the activity of supplementary DNA inLolium. Heredity 43:411–421
Imamura J, Saul MW, Potrykus I (1987) X-ray irradiation promoted asymmetric somatic hybridisation and molecular analysis of the products. Theor Appl Genet 74:445–450
Isaac PG, Jones VP, Leaver CJ (1985a) The maize cytochrome c oxidase subunit I gene: sequence, expression and rearrangement in cytoplasmic male-sterile plants. EMBO J 4:1617–1623
Isaac PG, Brennicke A, Dunbar SM, Leaver CJ (1985b) The mitochondrial genome of fertile maize (Zea mays L.) contains two copies of the gene encoding theα-subunit of the F1-ATPase. Curr Genet 10:321–328
Itoh K, Iwabuchi M, Shimamoto K (1991) In situ hybridization with species-specific DNA probes gives evidence for asymmetric nature ofBrassica hybrids obtained by X-ray fusion. Theor Appl Genet 81:356–362
Kyozuka J, Hayashi Y, Shimamoto K (1987) High frequency plant regeneration from rice protoplasts by novel nurse culture methods. Mol Gen Genet 206:408–413
Kyozuka J, Kaneda T, Shimamoto K (1989) Production of cytoplasmic male-sterile rice (Oryza sativa L.) by cell fusion. Bio/Technology 7:1171–1174
Lichtenstein C, Draper J (1985) Genetic engineering of plants. In: Glover DM (ed) DNA cloning, vol II. IRL Press, Oxford Washington, pp 67–119
Melzer JM, O'Connell MA (1990) Molecular analysis of the extent of asymmetry in two asymmetric somatic hybrids of tomato. Theor Appl Genet 79:193–200
Müller AJ, Grafe R (1978) Isolation and characterization of cell lines ofNicotina tabacum lacking nitrate reductase. Mol Gen Genet 161:67–76
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Ozias-Atkins P, Ferl RJ, Vasil IK (1986) Somatic hybridization in the gramineae:Pennisetum americanum (L.) K. Schum. (pearl millet)+Panicum maximum Jacq. (Guinea grass). Mol Gen Genet 203:365–370
Parokonny AS, Kenton AY, Gleba YY, Bennett MD (1992) Genome reorganization inNicotiana asymmetric somatic hybrids analysed by in situ hybridization. Plant J 2:863–874
Perez-Vicente R, Petris L, Osusky M, Potrykus I, Spangenberg G (1992) Molecular and cytogenetic characterization of repetitive DNA sequences fromLolium andFestuca: applications in the analysis ofFestulolium hybrids. Theor Appl Genet 84:145–154
Piastuch WC, Bates GW (1990) Chromosomal analysis ofNicotiana asymmetric somatic hybrids by dot blotting and in situ hybridization. Mol Gen Genet 222:97–103
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning — a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
Shillito RD, Paszkowski J, Müller M, Potrykus I (1983) Agarose plating and bead-type culture technique enable and simulate development of protoplast-derived colonies in a number of plant species. Plant Cell Rep 2:244–247
Tabaeizadeh Z, Ferl RJ, Vasil IK (1986) Somatic hybridization in the Gramineae:Saccharum officinarum L. (Sugarcane)+Pennisetum americanum (L.) K. Schum. (Pearl millet). Proc Natl Acad Sci USA 83:5616–5619
Takamizo T, Suginobu K, Ohsugi R (1990) Plant regeneration from suspension culture-derived protoplasts of tall fescue (Featuca arundinacea Schreb.) of a single genotype. Plant Sci 72:125–131
Takamizo T, Spangenberg G, Suginobu K, Potrykus I (1991) Intergeneric somatic hybridization in Gramineae: somatic hybrid plants between tall fescue (Festuca arundinacea Schreb.) and Italian ryegrass (Lolium multiflorum Lam.). Mol Gen Genet 231:1–6
Terada R, Kyozuka J, Nishibayashi S, Shimamoto K (1987) Plantlet regeneration from somatic hybrids of rice (Oryza sativa L.) and barnyard grass (Echinochloa oryzicola Vasing). Mol Gen Genet 210:39–43
Vallés MP, Wang ZY, Montavon P, Potrykus I, Spangenberg G (1993) Analysis of genetic stability of plants regenerated from suspension cultures and protoplasts of meadow fescue (Festuca pratensis Huds.). Plant Cell Rep 12:101–106
Vasil V, Ferl RJ, Vasil IK (1988) Somatic hybridization in the Gramineae:Triticum monococcum L. (Einkorn)+Pennisetum americanum (L.) K. Schum. (Pearl millet). J Plant Physiol 132:160–163
Wang ZY, Takamizo T, Iglesias VA, Osusky M, Nagel J, Potrykus I, Spangenberg G (1992) Transgenic plants of tall fescue (Festuca arundinacea Schreb.) obtained by direct gene transfer to protoplasts. Bio/Technology 10:691–696
Wang ZY, Vallés MP, Montavon P, Potrykus I, Spangenberg G (1993) Fertile plant regeneration from protoplasts of meadow fescue (Festuca pratensis Huds.). Plant Cell Rep 12:95–100
Wijbrandi J, Wolters AMA, Koornneef M (1990a) Asymmetric somatic hybrids betweenLycopersicon esculentum and irradiatedLycopersicon peruvianum. 2. Analysis with marker genes. Theor Appl Genet 80:665–672
Wijbrandi J, Zabel P, Koornneef M (1990b) Restriction fragment length polymorphism analysis of somatic hybrids betweenLycopersicon esculenium and irradiatedL. peruvianum: evidence for limited donor genome elimination and extensive chromosome rearrangements. Mol Gen Genet 222:270–277
Wolters AMA, Schoenmakers HCH, van der Meulen-Muisers JJM, van der Knaap E, Derks FHM, Koornneef M, Zelcer A (1991) Limited DNA elimination from the irradiated potato parent in fusion products of albinoLycopersicon esculenium andSolanum tuberosum. Theor Appl Genet 83:225–232
Yang ZQ, Shikanai T, Yamada Y (1988) Asymmetric hybridization between male-sterile (CMS) and fertile rice (Oryza sativa L.) protoplasts. Theor Appl Genet 76:801–808
Yang ZQ, Shikanai T, Mori K, Yamada Y (1989) Plant regeneration from cytoplasmic hybrids of rice (Oryzya sativa L.). Theor Appl Genet 77:305–310
Yamashita Y, Terada R, Nishibayashi S, Shimamoto K (1989) Asymmetric somatic hybrids ofBrassica: partial transfer ofB. campestris genome intoB. oleracea by cell fusion. Theor Appl Genet 77:189–194
Author information
Authors and Affiliations
Additional information
Communicated by M. Koorneef
Rights and permissions
About this article
Cite this article
Spangenberg, G., Vallés, M.P., Wang, Z.Y. et al. Asymmetric somatic hybridization between tall fescue (Festuca arundinacea Schreb.) and irradiated Italian ryegrass (Lolium multiflorum Lam.) protoplasts. Theoret. Appl. Genetics 88, 509–519 (1994). https://doi.org/10.1007/BF01240911
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01240911