Abstract
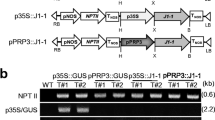
The anionic peroxidase genes of tomato, tap1 and tap2, are induced by wounding in tomato fruits and by elicitor treatment in cell suspension cultures. These homologous genes code for anionic peroxidases that are postulated to cause polymerization of the phenolic residues into wall polymers in wound-healing and pathogen-infected tissues. An expression construct containing the entire TAP1 gene with its 5′ and 3′ flanking sequences was introduced into tobacco by Agrobacterium tumefaciens-mediated gene transfer. Also, constructs containing the 5′ upstream regions of tap1 and tap2 including sequences coding for their respective putative leader peptides fused translationally to the β-glucuronidase (GUS) reporter gene were made and introduced into tobacco. Northern blot analysis of transcripts from wound-healing leaf tissues of transformants containing tap1 showed that the introduced gene was being transcribed in the heterologous host. The induction of tap1 transcripts in the wound-healing transgenic tobacco tissues was observed by 48 h and increased over time period of 84 h. Wounding also led to expression of GUS in tap1/GUS and tap2/GUS transformants and GUS activity was localized to the wound site. Activation of the tap1 and tap2 promoters in wound-healing transgenic tobacco tissues showed a GUS expression profile that correlated with the postulated role for anionic peroxidases in phenolic polymerization in suberizing tissues. Inoculation of tap1/GUS and tap2/GUS transformant leaves with fungal conidia from Fusarium solani f. sp. pisi caused expression of GUS in locally inoculated regions, and GUS expression increased over a period of four days.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bevan MW: Binary Agrobacterium vectors for plant transformation. Nucl Acids Res 12: 8711–8721 (1984).
Borchert R. Time course and spatial distribution of phenylalanine ammonia-lyase and peroxidase activity in wounded potato tuber tissue. Plant Physiol 62: 789–793 (1978).
Chomczynski P, Sacchi N: Single-step method of RNA isolation by acid guanidium thiocyanate-phenol-chloroform extraction. Anal Biochem 162: 156–159 (1987).
Cottle W, Kolattukudy PE: Biosynthesis, deposition, and partial characterization of potato suberin phenolics. Plant Physiol 69: 393–399 (1982).
Dean BB, Kolattukudy PE: Synthesis of suberin during wound-healing in jade leaves, tomato fruit, and bean pods. Plant Physiol 58: 411–416 (1976).
Espelie KE, Franceschi VR, Kolattukudy PE: Immunocytochemical localization and time course of appearance of an anionic peroxidase associated with suberization in wound-healing potato tuber tissue. Plant Physiol 81: 487–492 (1986).
Everdeen DS, Kiefer S, Willard JJ, Muldoon EP, Dey PM, Li XB, Lamport DTA: Enzymatic cross-linkage of monomeric extensin precursors in vitro. Plant Physiol 87: 616–621 (1988).
Fourney RM, Miyakoshi J, DayIII RS, Paterson MC: Northern blotting: efficient RNA staining and transfer. Focus 10: 5–7 (1988).
Gaspar T, Penel CL, Thorpe T, Greppin H: Peroxidases 1970–1980. A Survey of their Biochemical and Physiological Roles in Higher Plants, pp. 89–112. (Université de Genève, Genève) (1982).
Gillikin JW, Graham JS: Purification and developmental analysis of the major anionic peroxidase from the seed coat of Glycine max. Plant Physiol 96: 214–220 (1991).
Graham MY, Graham TL: Rapid accumulation of anionic peroxidase and phenolic polymers in soybean cotyledon tissues following treatment with Phytophthora megasperma f. sp. glycinea wall glucan. Plant Physiol 97: 1145–1155 (1991).
Gross GG: Biosynthesis of lignin and related monomers. In: Loewus FA, Runeckles VC (eds) Recent Advances in Phytochemistry vol. 11: pp. 141–184 (1977).
Hahlbrock K, Griseback H: Enzymic controls in the biosynthesis of lignin and flavonoids. Annu Rev Plant Physiol 30: 105–130 (1979).
Horsch RB, Fry J, Hoffmann N, Neidermeyer J, Rogers SG, Fraley RT: Leaf disc transformation. In: Gelvin SB, Schilperoort RA, Verma DPS (eds) Plant Molecular Biology Manual, pp. A5/1-A5/9. Kluwer Academic Publishers, Dordrecht (1988).
Iturriaga G, Jefferson RA, Bevan MW: Endoplasmic reticulum targeting and glycosylation of hybrid proteins in transgenic tobacco. Plant Cell 1: 381–390 (1989).
Jefferson RA: Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol Biol Rep 5: 387–405 (1987).
Kolattukudy PE: Biopolyester membranes of plants: cutin and suberin. Science 208: 990–1000 (1980).
Kolattukudy PE: Biochemistry and function of cutin and suberin. Can J Bot 62: 2918–2933 (1984).
Kolattukudy PE: Lipid-derived defensive polymers and waxes and their role in plant-microbe interaction. In: Stumpf PK, Conn EE (eds) The Biochemistry of Plants, vol. 9, pp. 291–314. Academic Press, New York (1987).
Kolattukudy PE, Mohan R, Bajar MA, Sherf BA: Plant peroxidase gene expression and function. In: Boyer S (ed) Plant Oxygenases, Peroxidases and Oxidases. Biochemical Society Transactions, vol. 20, pp. 333–337 (1992).
Lagrimini LM, Rothstein S: Tissue specificity of tobacco peroxidase isozymes and their induction by wounding and tobacco mosaic virus infection. Plant Physiol 84: 438–442 (1987).
Lagrimini LM, Burkhart W, Moyer M, Rothstein S: Molecular cloning of complementary DNA encoding the lignin-forming peroxidase from tobacco: Molecular analysis and tissue specific expression. Proc Natl Acad Sci USA 84: 7542–7546 (1987).
Lawton MA, Lamb C: Transcriptional activation of plant defense genes by fungal elicitor, wounding, and infection. Mol Cell Biol 7: 335–341 (1987).
Logemann J, Lipphardt S, Lorz H, Hauser I, Willimitzer L, Schell J: 5′ upstream sequences from the wun1 gene are responsible for gene activation by wounding in transgenic plants. Plant Cell 1: 151–158 (1989).
Macheix JJ, Fleuriet A, Quessada MP: Involvement of phenols and peroxidases in wound healing and grafting. In: Greppin H, Panel C, Gaspar Th (eds) Molecular and Physiological Aspects of Plant Peroxidases Plants, pp. 267–286. Université de Genève, Genève (1986).
Maniatis T, Fritsch FE, Sambrook J: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY (1982).
Mohan R, Kolattukudy PE: Differential activation of expression of a suberization-associated anionic peroxidase in near-isogenic resistant and susceptible tomato lines by elicitors of Verticillium albo-atrum. Plant Physiol 921: 276–280 (1992).
Pearce RB, Rutherford J: A wound-associated suberized barrier to the spread of decay in the sapwood of oak (Quercus robur L.). Physiol Plant Path 19: 359–369 (1981).
Plunkett G, Senear DF, Zuroske G, Ryan CA: Proteinase inhibitors I and II from leaves of wounded tomato plants: purification and properties. Arc Biochem Biophys 213: 463–472 (1982).
Rasmussen SK, Welinder KG, Hejgaard J: cDNA cloning, characterization and expression of an endosperm-specific barley peroxidase. Plant Mol Biol 16: 317–327 (1991).
Rebmann G, Hertig C, Bull J, Mauch F, Dudler R: Cloning and sequencing of cDNAs encoding a pathogen-induced putative peroxidase of wheat (Triticum aestivum L.). Plant Mol Biol 16: 329–331 (1991).
Rhodes MJC, Wooltorton LSC: Biochemistry of Wounded Plant Tissues. In: Kahl G (ed) The Biochemistry of Phenolic Compounds in Wounded Plant Storage Tissues, pp. 243–286. Walter de Gruyter, Berlin (1978).
Robb J, Lee SW, Mohan R, Kolattukudy PE: Chemical characterization of stress-induced vascular coating. Plant Physiol 97: 528–536 (1991).
Roberts E, Kolattukudy PE: Molecular cloning, nucleotide sequence and abscisic acid induction of a suberization-associated highly anionic peroxidase. Mol Gen Genet 217: 223–231 (1989).
Roberts E, Kutchan T, Kolattukudy PE: Cloning and sequencing of cDNA for a highly anionic peroxidase from potato and the induction of its mRNA in suberizing potato tubers and tomato fruits. Plant Mol Biol 11: 15–26 (1988).
Roby D, Broglie K, Cressman R, Biddle P, Chet I, Broglie R. Activation of a bean chitinase promoter in transgenic tobacco plants by phytopathogenic fungi. Plant Cell 2: 999–1007 (1990).
Rogers SO, Benedisch AJ: Extraction of DNA from plant tissues. In: Gelvin SB, Schilperoort RA, Verma DPS (eds) Plant Molecular Biology Manual, pp. A6/1-A6/10. Kluwer Academic Publishers, Dordrecht (1988).
Showalter AM, Varner JE: Plant hydroxyproline-rich glycoproteins. In: Marcus A (ed) The Biochemistry of Plants: A Comprehensive Treatise, vol. 11. Molecular Biology, pp. 485–520. Academic Press, New York (1989).
Siebertz B, Logemann J, Willmitzer L, Schell J: Cis-analysis of the wound-inducible promoter wun1 in transgenic tobacco plants and histochemical localization of its expression. Plant Cell 1: 961–968 (1989).
Stanford AC, Northcote DH, Bevan MW: Spatial and temporal patterns of transcription of a wound-induced gene in potato. EMBO J 9: 593–603 (1990).
Ye XS, Pan SQ, Kuc J: Association of pathogenesis-related proteins and activities of peroxidase, β-1,3-glucanase and chitinase with systemic induced resistance to blue mold of tobacco but not to systemic tobacco mosaic virus. Physiol Mol Plant Path 36: 523–531 (1990).
Zenk MH: Recent work on cinnamoyl CoA derivatives. In: Swain T, Harborne J, Van Sumere C (eds) Recent Advances in Phytochemistry, vol. 12, pp. 139–176 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mohan, R., Bajar, A.M. & Kolattukudy, P.E. Induction of a tomato anionic peroxidase gene (tap1) by wounding in transgenic tobacco and activation of tap1/GUS and tap2/GUS chimeric gene fusions in transgenic tobacco by wounding and pathogen attack. Plant Mol Biol 21, 341–354 (1993). https://doi.org/10.1007/BF00019949
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00019949