Abstract
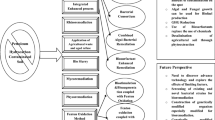
Contamination of soils with oil hydrocarbons is currently an important worldwide issue. Among all the available remediation methods, bioremediation is widely considered to be a cost-effective and environmentally friendly approach. For bioremediation to be effective, the overall rate of intrinsic biodegradation and subsequent removal of hydrocarbons must be accelerated, which can be done through biostimulation and bioaugmentation. A variety of techniques for bioaugmentation and biostimulation have been compiled and summarized in this chapter. The evaluation of the potentials to use such treatments to enhance biodegradation rates in oil hydrocarbon contaminated soil is provided, including some recent works from the author’s laboratory. Factors that may enhance biodegradation of oil hydrocarbons in soil are discussed in detail, and finally some examples of new approaches are presented.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Total Petroleum Hydrocarbon
- Microbial Consortium
- Biodegradation Rate
- Synthetic Surfactant
- Indigenous Microorganism
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Modern societies still continue to rely primarily on the use of petroleum hydrocarbons to cover their energy demands. Despite recent technological advances, accidental spills of crude oil and its refined products occur on a frequent basis during routine operations such as extraction, transportation, storage, refining and distribution (Nikolopoulou et al. 2007). The release of oil hydrocarbons into the environment may pose severe environmental problems due to sustained contamination of air, water and soil (Scherr et al. 2007). Various physical and chemical processes have been employed for effective remediation of oil hydrocarbon contaminated soil (Khan et al. 2004; Malina 2007). However, most of these techniques are expensive, and the byproducts may cause secondary contamination of soil and water, resulting in the need for additional post-treatments (Liang et al. 2009). Moreover, they require continuous monitoring and control for optimum performance. In addition, they do not usually result in a complete destruction of the contaminants (Gouda et al. 2008). Biological methods, such as bioremediation, are considered to be relatively cost-effective and environmentally friendly (Hosokawa et al. 2009). Bioremediation is a treatment method that uses microbiological restoration potentials for decontamination of polluted sites (Scherr et al. 2007). It is a relatively simple practical approach for the complete mineralization of hydrocarbons to carbon dioxide and water under aerobic conditions (Vidali 2001; Sarkar et al. 2005). However, the rate of hydrocarbon biodegradation in soil is affected by several physicochemical properties of the soil and contaminants, as well as biological characteristics of indigenous microorganisms. These include the number and species of microorganisms present, concentrations of hydrocarbons and environmental conditions (pH, temperature, nutrients, oxygen and moisture content) suitable for microbial degradation (Betancur-Galvis et al. 2006; Gouda et al. 2008; Leahy and Colwell 1990; Perfumo et al. 2007; Horel and Schiewer 2009). Two methods are usually considered to increase the activity of microorganisms, thus enhancing the biodegradation rates during bioremediation of soil contaminated with oil hydrocarbons: (a) bioaugmentation through the direct application of selected oil-degraders to the site, and (b) biostimulation involving the application of a proper agent to soil to enhance the activity of indigenous microorganisms (Odokuma and Dickson 2003; Perfumo et al. 2007; Malina and Zawierucha 2007).
Bioaugmentation is a promising and low-cost bioremediation method, in which effective bacterial isolates or microbial consortia capable of degrading oil hydrocarbons are introduced to the contaminated soil. Multiplied indigenous microflora are generally applied in this technique; however, inoculation of soil with exogenous or laboratory-modified bacterial cultures still arouses many reservations (Gentry et al. 2004; Fantroussi and Agathos 2005; Zawierucha and Malina 2006). Sometimes, the application of oil-degrading microorganisms may lead to a failure of bioaugmentation (Vogel 1996; Gentry et al. 2004). This is because the survival and degrading ability of microorganisms introduced to a contaminated site are highly dependent on environmental conditions (Vogel 1996). Thus, in many cases, potentially degrading strains isolated from one site are not necessarily applicable to the other site. Moreover, isolates, including genetically engineered microorganisms, that are efficient oil-degraders under laboratory conditions, are not necessarily effective in situ (Sayler and Ripp 2000). In addition, introducing alien species to soil is not easily acceptable by the public (Hosokawa et al. 2009).
Biostimulation relies on increasing the activity of indigenous bacteria by providing nutrients, oxygen, surfactants or water to the contaminated soil (Coulon and Delille 2003) or modifying the environmental conditions (e.g., temperature, pH, redox potential). It is considered that indigenous bacteria are best adapted to the environment of the treated site (Rahman et al. 2003). Biostimulation, however, does not always work well due to the scarcity of indigenous oil-degraders or very high contaminant concentrations (Ueno et al. 2007).
The major objective of this chapter is to provide various bioremediation strategies based on bioaugmentation and biostimulation for enhancing biodegradation rates of oil hydrocarbon contaminated land.
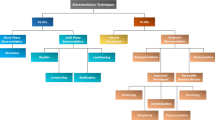
2 Bioaugmentation
Bioaugmentation can be realized in three ways: (a) the enrichment or isolation of indigenous microorganisms from the target site, their subsequent culturing and re-inoculation; (b) isolates or enrichments are not inoculated to the source of the original culture; and (c) with the use of constructed or force-mutated microorganisms (Vogel and Walter 2001).
Bioaugmentation that uses indigenous microorganisms to the sites (soil and water) to be remediated is defined as autochthonous bioaugmentation (Ueno et al. 2007). Isolated single strains or enriched cultures, which can be obtained “before” or “after” the contamination of the target sites, are inoculated into the sites to be remediated (Hosokawa et al. 2009). The use of indigenous microorganisms with adapted biochemical potentials was proved to be one of the most powerful tools for bioaugmentation (Devinny and Chang 2000). Bento et al. (2005) noted that the addition of a bacterial consortium previously isolated from the Long Beach soil degraded 73–75% of the light (C12–C23) and heavy (C23–C40) fractions of total petroleum hydrocarbons (TPH) present in the soil, while only 46–49% removal was obtained as a result of intrinsic biodegradation. Next, in field-scale microcosms, Gouda et al. (2008) observed that about 86% of kerosene was degraded upon bioaugmentation of clay with indigenous Pseudomonas sp. CK for 20 days, but only 80% in the case of intrinsic biodegradation of the natural microflora. Introducing naturally developed microbial consortia may be more effective in comparison with single strains isolated and applied as pure cultures (Mrozik and Piotrowska-Seget 2009). This is in agreement with the results of Mancera-Lopez et al. (2008), who noted that in the bioaugmented systems three indigenous fungi strains (Rhizopus sp., Penicillium funiculosum and Aspergillus sydowii) removed 47, 45 and 40% of TPH, respectively, and these were even higher by 29–36% with respect to the pure fungi strains. Thus, the most effective bioaugmentation performance may be approached by the use of multiplied indigenous microorganisms to increase their abundance in soil (Malina and Zawierucha 2007). In the study of Wu et al. (2008) microcosms were set up with a PAHs contaminated soil using bioaugmentation with indigenous filamentous fungus, Monilinia sp. W5-2. After 30 days of treatment, bioaugmented microcosms resulted in a 35% decrease in the total PAHs, while the control microcosms showed only a 3% decrease. Bioaugmented microcosms also revealed about 70 and 72% decreases in benzo[a]pyrene and anthracene, respectively, while the values for the control microcosms were much lower.
Our respirometry studies conducted to determine the effect of bioaugmentation for enhancing biodegradation in soils contaminated with oil hydrocarbons at a former military airport showed the highest biodegradation rates (estimated from the O2 uptake and CO2 production rates) for samples to which the bacterial inoculum containing 4.8 × 1015 CFU/ml was added (Zawierucha and Malina 2006). Enhanced biodegradation rates were in this case four times higher than intrinsic biodegradation rates. Moreover, when the indigenous bacterial consortium was applied, the increase of biodegradation rate was about 22–46% higher compared to the exogenous bacterial consortium. This could be explained by the autochthonous adaptation that allows microorganisms to be physiologically compatible with their habitat, as compared to transient autochthonous organisms that do not occupy a functional niche (Atlas and Bartha 1998). Ueno et al. (2007) also noticed that bioaugmentation capacity of isolated bacterial species in soil microcosms contaminated with diesel oil was much higher than that of exogenous P. aeruginosa strain WatG. Therefore, it will be more practical to apply bioaugmentation with bacteria isolated from the soil that is to be cleaned-up (Hosokawa et al. 2009).
Native populations present in contaminated sites are certainly adapted to the climatic, physicochemical and nutrient conditions. However, these communities frequently do not include species with the enzymatic abilities needed to allow bioremediation to start and/or to proceed at increased rates, thus resulting in long-time processes. Application to contaminated soils of exogenous microorganism with proven hydrocarbon-degrading abilities may solve this problem. Possible sources to obtain exogenous microorganisms are remediated or contaminated sites, commercial suppliers and genetic engineering (Diaz-Ramirez et al. 2008).
Biodegradation studies of 4CA in two soil types (loam soil-NN and sandy clay loam soil-CM) using exogenous 4CA-degrading Klebsiella sp. CA17 for bioaugmentation were carried out by Tongarun et al. (2008). Biodegradation of 4CA in soil-NN microcosms was substantially enhanced by bioaugmentation with 4CA-degrading Klebseilla sp. CA17. Compared to that of intrinsic biodegradation (40% of 4CA degradation), the total 4CA biodegradation at the end of bioaugmentation treatment period finally reached 70 ± 4%. In the case of soil-CM microcosms, total 4CA degradation was 44%, as compared to rather poor (10%) intrinsic biodegradation at the end of the treatment period. Moreover, bioaugmentation of a 4CA-degrading culture was successful in soil-NN, where the bioaugmented culture survived and maintained its population size, with a gradual increase, throughout the entire treatment, while it was eventually outnumbered by indigenous microorganisms in soil-CM. These results may indicate that the degree of 4CA biodegradation in soil microcosms depends not only on the site characteristics, for example, soil properties, but also on the characteristics of the indigenous microbial population, and the stability of population density of the bioaugmented culture, which definitely affected the efficiency of biodegradation.
The choice of chronically contaminated areas or the sites where land farming was applied for remediation for the screening of exogenous bacterial strains potentially useful in bioaugmentation seems to be an appropriate approach. Ruberto et al. (2003) observed that in soil with B-2-2, a psychrotolerant hydrocarbon-degrading Acinetobacter strain previously isolated from a chronically polluted river degraded 75% of hydrocarbons, whereas autochthonous bacterial communities were able to degrade important fractions of the gas oil only by 35%. Next, Jacques et al. (2008) evaluated the capacity of an exogenous microbial consortium (five bacteria: Mycobacterium fortuitum, Bacillus cereus, Microbacterium sp., Gordonia polyisoprenivorans, Microbacteriaceae bacterium – naphthalene-utilizing bacterium; and a fungus identified as Fusarium oxysporum) obtained from a petrochemical site treated by means of land farming, to degrade and mineralize anthracene, phenanthrene and pyrene present in soil at different concentrations. They noted that the microbial consortium mineralized on average 98% of the three PAHs present at different concentrations in the soil after 70 days. On the contrary, the autochthonous soil microbial population showed no substantial mineralization of the PAHs. Moreover, bacterial and fungal isolates from the consortium, when inoculated separately to the soil, were less effective in anthracene mineralization compared to that of the consortium. These results may indicate synergistic promotion of PAHs mineralization by mixtures of the monoculture isolates (the microbial consortium). Individual microorganisms can mineralize only a limited range of substrates; so assemblages of mixed populations with overall broad enzymatic capacities are required to increase the rate and extent of petroleum biodegradation (Farinazleen et al. 2004; Heinaru et al. 2005). Enhanced biodegradation of spiked anthracene (ANT), pyrene (PYR) and benzo[a]pyrene (B[a]P) in soil was also studied by Hamdi et al. (2007). In this case, bioaugmentation was carried out by mixing the previously treated aged PAH-contaminated soil containing PYR- and B[a]P-degraders with the experimental PAH-spiked soil. In the control samples, the PAH removal was the lowest revealing ANT, PYR and B[a]P dissipation rates of 63, 33 and 35%, respectively, after 120 days. In turn, in bioaugmented samples, the final degradation rates were higher than those observed in nonamended PAH-spiked soils, and they were above 96% for ANT and PYR and 60% for B[a]P. Therefore, the bioaugmentation with exogenous bacteria can be recommended in the case of more recalcitrant chemicals, or when the local microbial population is insufficient or inadequate (Mariano et al. 2007).
Another bioaugmentation approach involves the use of genetically engineered microorganisms (GEMs) with increased capacity to degrade and tolerate toxic compounds. Mutations and horizontal gene transfer using molecular biology are employed to improve the microbial degradation activity (Rodrigues et al. 2006). The aim of the application of genetically modified bacterial strains is to enhance the ability of newly generated strains to degrade a broader range of xenobiotics, and to increase the degradation effectiveness in comparison with “wild” (natural) strains (Mrozik and Piotrowska-Seget 2009). Filonov et al. (2005) studied the effectiveness of a genetically tagged, plasmid-containing, naphthalene-degrading strain Pseudomonas putida KT2442 (pNF142: TnMod-OTc) in the experimental soil contaminated with naphthalene. They noted that the concentration of naphthalene in the experimental soil block, into which laboratory naphthalene-degrading strain KT2442 was introduced, decreased from 2.0 to 0.2 mg/g, whereas in the control soil block, which contained only indigenous naphthalene degraders, the concentration decreased only to 0.6 mg/g over the same time period (20 days). Moreover, 20 days after introducing the strain KT2442, the number of bacterial cells increased from 105 to 6 × 106 CFU/g of soil, amounting to 90% of the total population of naphthalene degraders. Mishra et al. (2004) also noted that the TPH level in the microcosm soil bioaugmented with a recombinant Acinetobacter baumannii pJES strain was reduced by 39.6% at the end of a 90 days treatment, while in the untreated soil this reduction was only by 6.9%. Next, Massa et al. (2009) compared two bacterial strains, the natural isolate Arthrobacter sp. FG1 and the engineered strain Pseudomonas putida PaW340/pDH5, for their efficiency in degrading 4-chlorobenzoic acid (4-CBA) in a slurry phase system. The recombinant strain was obtained by cloning the Arthrobacter sp. FG1 dehalogenase encoding genes in P. putida PaW340. The 4-CBA-grown engineered strain appeared to be significantly more efficient in the 4-CBA degradation than the “wild” Arthrobacter in soil slurries regardless of the presence or absence of indigenous bacteria, which did not affect biodegradation. On the other hand, Lima et al. (2009) examined the efficacy of bioaugmentation with rifampicin-resistant mutant of Pseudomonas sp. ADP for bioremediation of an atrazine-contaminated land. They observed that, for a more moderate level of soil contamination (ca. 7 g of atrazine per g of soil), bioaugmentation using one single inoculation with P. ADP could be sufficient for successful treatment. The atrazine removal was of 99% after 8 days of the treatment. Therefore, the use of GEMs has been suggested to improve or accelerate the remediation of sites polluted with xenobiotics (Filonov et al. 2005; Lima et al. 2009).
The novel approach, the so-called immobilized bioaugmentation based on delivering microbial cultures in an immobilized form, is applied to achieve more complete and/or more rapid degradation of hydrocarbons in soil. Such treatment offers the protection of inoculated microorganisms from sub-optimal environmental conditions (improper pH, presence of toxic substances, etc.), and it reduces their competition with indigenous microflora. Moreover, immobilization increases the biological stability of cells, including plasmids (Cunningham et al. 2004). For immobilization, both natural and synthetic materials are used. The first group includes dextran, agar, agarose, alginate, chitosan polyacrylamides, and κ-carrageenan, while the second includes poly(carbamoyl)sulphonate, polyacrylamide and polyvinyl alcohol (Mrozik and Piotrowska-Seget 2009). Immobilized matrix may also act as a bulking agent in contaminated soil, facilitating the transfer of oxygen crucial for rapid hydrocarbon mineralization (Cunningham et al. 2004; Liang et al. 2009). Results obtained by Oh et al. (2003) indicate that the inoculum immobilized on diatomaceous earth could be very effective for retaining microbial cells in association with the sand contaminated with crude oil. Cunningham et al. (2004) examined the potential of immobilized hydrocarbon-degrading microorganisms for the cleanup of diesel-contaminated soil, and compared it with the liquid-culture bioaugmentation. Using polyvinyl alcohol (PVA) cryogelation as an entrapment technique, they noted that bioaugmentation with a liquid enrichment culture reduced by 36.7% of oil and grease contents after 32 days of treatment, while the immobilized system resulted in the 48.1% reduction. Moreover, the reductions of diesel in these bioaugmentation systems were about 25.3–36.7% higher as compared to the non-amended (control) treatment pile. Next, Liang et al. (2009) explored the role of bio-carriers, such as activated carbon and zeolites, in immobilizing indigenous hydrocarbon-degrading bacteria and enhancing biodegradation in crude oil contaminated soils. They observed high microbial colonization of both zeolites and activated carbon. Microbial biomass reached concentrations of 1010 cells/g of activated carbon and 106 cells/g of zeolites, indicating that the first carrier was better for the enrichment of bacteria. Total microbial and dehydrogenase activity were 12 and 3 times higher, respectively, in activated carbon than in zeolite. Moreover, the activated carbon bio-carrier enhanced the biodegradation of crude oil resulting in the removal of 48.9%, in comparison to the intrinsic biodegradation (13.0%), and liquid-culture bioaugmentation (free-living bacteria) – 37.4%. According to the authors, the bio-carrier improved the mass transfer of oxygen and nutrients, as well as the water-holding capacity of the soil, which were the limiting factors for biodegradation of crude oil.
The successful soil bioaugmentation requires the knowledge of not only types and contents of contaminants but also microbial strains or their consortia that are suitable for biodegradation. The selection of a culture appropriate for bioaugmentation should take into consideration the following features of microorganisms: fast growth rate, ease of cultivation, resistance to high contaminant concentrations and ability to survive in a wide range of environmental conditions (Mrozik and Piotrowska-Seget 2009). Moreover, for designing an optimum bioaugmentation method, it is necessary to evaluate the fractions of bioavailable contaminants to determine the required concentrations of a degrading inoculum to be added (Zawierucha and Malina 2006).
3 Biostimulation
Biostimulation is a technique that relies on increasing the activity of the indigenous bacteria by adjusting the factors that may limit their activity, mainly oxygen and nutrients. The main aim of biostimulation is to provide bacterial communities with a favorable environment, in which they can effectively degrade contaminants (Mohan et al. 2006; Ueno et al. 2007).
3.1 Biostimulation by Oxygen Supply
Bioremediation of hydrocarbon contaminated soils under aerobic conditions is preferable to improve degradation yields, given that the most common microbial degraders are aerobic (Menendez-Vega et al. 2007). Oxygen is supplied to soil to stimulate microbial activity and enhance aerobic biodegradation rates in the case when O2 is considered as a limiting factor. Commonly used oxygen supply techniques may include tilling, forced aeration and chemical methods (Atlas 1991; Brown and Crosbie 1994; Riser-Roberts 1998). Tilling is recommended as a physical method to accelerate biodegradation during land farming, but it is effective only for top soils. Forced aeration techniques, including injection of aerated water, air or pure oxygen, are commonly used for enhancing biodegradation in soils and groundwater contaminated with petroleum hydrocarbons (Brown and Crosbie 1994; Riser-Roberts 1998). Chemical methods involve addition of alternative oxygen sources, such as oxygen-releasing compounds ORC®, or agents such as potassium permanganate (KMnO4), hydrogen peroxide (H2O2) and ozone (O3) (Riser-Roberts 1998).
In our study, we tested the effectiveness of diverse sources of oxygen (aerated water, aqueous solutions of H2O2 and KMnO4) to enhance biodegradation of oil hydrocarbons in soil at a former military airport (Malina and Zawierucha 2007). Based on respirometric tests the highest CO2 production rates (71–97% higher compared to a control) were achieved when the aqueous solution of KMnO4 in concentration of 20 g/L was applied. On the other hand, on average, only 15% increase of CO2 production rates was observed when the aqueous solutions of H2O2 were used, whereas aerated water did not cause any improvement of the biodegradation rates (addition of aerated water resulted in a decrease of CO2 production rates as compared to a control). Most probably, the amount of added water led to excessive soil moisture that could reduce, in fact, the air-filled porosity and, consequently the oxygen contents in soil (Malina 1999).
Potassium permanganate is known to readily oxidize alkene carbon–carbon double bonds (Wolfe and Ingold 1981; Walton et al. 1992). Brown et al. (2003) observed the concentration reduction of benzo[a]pyrene (72.1%), pyrene (64.2%), phenanthrene (56.2%) and anthracene (53.8%) in soil, after 30 min of oxidation using 160 mM KMnO4. They suggested that permanganate oxidation could be applied in remediation technology for soils contaminated with oil hydrocarbons. While hydrocarbons are not completely mineralized by permanganate oxidation reactions, their structure is altered by polar functional groups providing vast improvements in aqueous solubility, increased bioavailability for microorganisms, thus biodegradation enhancement.
Hydrogen peroxide is known in environmental applications as a chemical oxidizing agent, disinfectant and source of oxygen (Hamby 1996; Olexsey and Parker 2006; Goi et al. 2006). Aerobic biodegradation of hydrocarbons in soil can benefit from the presence of oxygen released during the H2O2 decomposition (Sturman et al 1995). On the other hand, it can be toxic to microorganisms (Riser-Roberts 1998) as high contents of H2O2 (100–200 mg/l) can inhibit bacterial metabolism (Huling et al. 1990). However, Tsai and Kao (2009) noted that 43 and 47% of TPH were removed from soil using 15 and 30% aqueous solution of H2O2, which corresponded to the H2O2 concentrations of 150 and 300 mg/L, respectively, after 40 h of treatment, while the TPH removal using 1% aqueous solutions of H2O2 was only of 1.1%. These results indicate that the TPH oxidation can be enhanced by higher H2O2 concentration. Moreover, Goi et al. (2006) noted that the efficiency of H2O2 application was strongly dependent on the soil matrix. Treatment of shale and transformer oils adsorbed on peat (a model of organic-rich soil) resulted in lower degree of oil removal, and required more H2O2 than the treatment of oil in sand matrix representing the mineral part of soil.
Aerated water can be an effective O2 carrier for aerobic biodegradation of oil hydrocarbons in soil, and it may facilitate the transport of substrates to bacterial cells (Malina 2007). For example, Liu et al. (2001) found a 50% increase of phenanthrene biodegradation when increasing the soil water content to 200% of the soil field capacity. But on the other hand, at higher water contents, near or over saturation, all the pores are filled with water, which limits the oxygen transfer that determines the activity of aerobic microorganisms, thus biodegradation of contaminants may be hampered (Ramirez et al. 2009), and which could actually be the case in our experiment (Malina and Zawierucha 2007).
3.2 Biostimulation by Nutrients Supply
Additional nutrients (mainly nitrogen and phosphorus) introduced into contaminated soil in the form of organic and/or inorganic fertilizers may enhance intrinsic biodegradation of petroleum hydrocarbons by improving the C:N:P ratio (Sarkar et al. 2005). In theory, approximately 150 mg of nitrogen and 30 mg of phosphorus are utilized in the conversion of 1 g of hydrocarbon to cell materials (Rosenberg and Ron 1996). Based on this, the optimal C:N:P mole-ratio recommended for enhancing hydrocarbon removal is 100:10:1 (Malina 1999). However, as the soil environment is very complex and heterogeneous, the effectiveness of nutrient sources tends to be affected by the soil physicochemical properties (Malina 2007). We compared various sources of nutrients (N, P) with different C:N:P ratios for enhancing biodegradation of oil hydrocarbons in soil (Zawierucha et al 2008). The highest enhanced biodegradation rates (2–26 times higher than intrinsic biodegradation rates) were observed at the C:N:P ratio of 100:10:5. Moreover, the best results were achieved when the combination of (NH4)2SO4 and Na2HPO4 was used, as the enhanced biodegradation rates were 120–1,556% higher compared to the intrinsic biodegradation rates. Ubochi et al. (2006) examined the potential of biostimulation treatment options in oil contaminated soil with different contents of the NPK fertilizer in soil. They noted that the application of 60 g NPK fertilizer was the best treatment option with the removal of 50.5% of crude oil while in the control (no nutrient addition) the removal was only of 29.5%. However, the effectiveness of biostimulation depends not only on the proper C:N:P ratio but also on the type of soil. Aspray et al. (2008) observed that for the sandy gravel and silty clay soils contaminated with petroleum hydrocarbons, both O2 consumption and CO2 production showed enhanced microbial activity when amended with NH4NO3, whereas, these results differed for the sandy loam soil. In this soil amended with nitrogen, inhibition of respiration was observed. Moreover, the form of nutrients (especially nitrogen) supply plays a role in effective fertilization (Chaillan et al. 2006). The amendment with nitrogen (particularly using inorganic fertilizers) can have no effect or, when applied at high concentrations, even deleterious effects (Bento et al. 2005; Walworth et al. 2007). Inorganic nitrogen fertilizers composed of nitrate and ammonium salts increase the salt concentration of soil pore water, lowering the soil osmotic potential and, thus inhibiting the microbial activity (Walworth et al. 2007). In addition, Sarkar et al. (2005) found that the microbial population in the fertilizer-amended soils dropped appreciably, suggesting a toxic effect due to fertilizer-induced acidity and/or NH3 overdosing. Therefore, the fertilizers must be precisely dosed taking the local environmental conditions into consideration. The effectiveness of hydrocarbon biodegradation is not proportional to the nutrient concentrations and over-fertilization may inhibit decomposition of less biodegradable compounds (Chaillan et al. 2006). It is also recommended that the C:N:P ratio should be calculated on the basis of the concentration of saturated hydrocarbons, degradation of which is most sensitive to the level of nutrients in soil (Chaineau et al. 2005).
3.3 Biostimulation by Surfactants Supply
A critical aspect of bioremediation of oil-contaminated soils is the availability of contaminants for microorganisms limited by their water solubility (Menendez-Vega et al. 2007). This problem can be solved using natural and synthetic surfactants (Lai et al. 2009). Hydrocarbon-degrading microorganisms produce a variety of surface-active natural agents (the so-called biosurfactants) that improve bioavailability. However, synthetic surfactants may still be required when the contaminants are highly hydrophobic, and/or firmly sorbed in clay particles or soil organic matter (Menendez-Vega et al. 2007). Surfactants contain both hydrophobic and hydrophilic fractions and are useful in reducing the interfacial tension between hydrocarbons and soil water, thereby improving the water solubility of hydrophobic substances (Urum et al. 2006; Zawierucha et al. 2007). Consequently, surfactants increase bioavailability of hydrocarbons to microorganisms and in turn their biodegradation (Lee et al. 2005). In our study on the effect of surfactant (Tween 80) on biodegradation of oil hydrocarbon and microbial activity in soil, the highest O2 consumption and CO2 production rates, as well as dehydrogenase activities were observed at the surfactant dose of 1% (v/v) (Zawierucha et al. 2007). In this case, the O2 consumption and CO2 production rates were 115 and 49% higher, respectively, while the dehydrogenase activity increased 98%, as compared to a control (no addition of the surfactant). These results indicate that surfactants can improve biodegradation effectiveness in the soil contaminated with oil hydrocarbons, which was also postulated by Bento et al. (2005), Rous et al. (1994) and Xie (2003). The potential application of biosurfactants, surfactin (SF) and rhamnolipid (RL), for enhanced diesel biodegradation was investigated by Whang et al. (2009). They observed that, compared to the control treatment (no bio-surfactants added), application of RL or SF resulted in diesel emulsification, and therefore enhanced biodegradation. Lai et al. (2009) compared the effectiveness of biosurfactants with that of synthetic surfactants in heavily oil-polluted soil. They found that biosurfactants exhibited much higher TPH removal efficiency than the synthetic ones. By using rhamnolipids, surfactin, Tween 80, and Triton X-100 in the concentration of 0.2% (w/w), the TPH removal for the soil contaminated with 3,000 mg TPH/kg dry soil was of 23, 14, 6, and 4%, respectively, and it increased to 63, 62, 40, and 35%, respectively, for the soil contaminated with 9,000 mg TPH/kg dry soil. Moreover, the biosurfactants-enhanced TPH removal efficiency did not vary significantly with the contact time. These results indicate the superior performance of biosurfactants over synthetic surfactants in terms of mobilization of oil pollutants from the contaminated soil, and thus confirm their use as biostimulation agents for bioremediation. Biosurfactants are preferred to chemical surfactants, as they have lower toxicity and shorter persistence in the environment (Nievas et al. 2008). The potential advantages of biosurfactants for enhancing bioremediation of hydrocarbon contaminated soils also include their unusual structural diversity that may lead to unique properties, as well as their biodegradability. Moreover, biosurfactants could easily be produced from renewable resources via microbial fermentation, making it an additional advantage over chemically synthetic surfactants (Mulligan 2005).
4 Conclusions
Technologies employing biological treatments are developing worldwide over the last decade, and are viewed as ready-made approaches for bioremediation of petroleum contaminated sites. This chapter presented the potential of oil hydrocarbons biodegradation enhancement in contaminated soils by applying various bioaugmentation and biostimulation methods. Based on our extensive review of literature and practical experience, bioremediation is not a panacea for all problems associated with oil hydrocarbon contaminated soil. The successful bioremediation requires the strategies tailored for the site-specific environmental parameters of both contaminated soils and contaminants. The key parameters to select the appropriate bioremediation strategy includes number and activity of microorganisms; types, concentrations and bioavailability of contaminants; oxygen and nutrients supply, and characteristics of soil. In the case of bioaugmentation, an additional database needs to be established, containing abilities of microorganisms to degrade oil hydrocarbons, together with their proliferation in the respective ecosystems, as well as their cellular resistance to xenobiotics and adaptation potentials to environmental conditions.
Although both bioaugmentation and biostimulation alone appeared to be effective in enhancing intrinsic biodegradation of oil hydrocarbons in soil, the simultaneous action of these techniques seems to improve the biodegradation rates more efficiently.
References
Aspray T, Gluszek A, Carvalho D (2008) Effect of nitrogen amendment on respiration and respiratory quotient (RQ) in three hydrocarbon contaminated soils of different type. Chemosphere 72:947–951
Atlas RM (1991) Microbial hydrocarbon degradation-bioremediation of oil spills. J Chem Tech Biotechnol 52:149–156
Atlas RM, Bartha R (1998) Microbial ecology: fundamentals and applications. Benjamin/Cummings, Menlo Park, CA
Bento FM, Camargo FAO, Okeke BC, Frankenberger WT (2005) Comparative bioremediation of soils contaminated with diesel oil by natural attenuation, biostimulation and bioaugmentation. Bioresour Technol 96:1049–1055
Betancur-Galvis LA, Alvarez-Bernal D, Ramos-Valdivia AC, Dendooven L (2006) Bioremediation of polycyclic aromatic hydrocarbon-contaminated saline-alkaline soils of the former Lake Texcoco. Chemosphere 62:1749–1760
Brown RA, Crosbie JR (1994) Oxygen sources for in situ bioremediation. In: Flathman PE, Jerger DE, Jurgen HE (eds) Bioremediation: field experience. CRC, LLC Boca Raton, FL, pp 311–332
Brown GS, Barton LL, Thomson BM (2003) Permanganate oxidation of sorbed polycyclic aromatic hydrocarbons. Waste Manage 23:737–740
Chaillan F, Chaineau CH, Point V, Saliot A, Oudot J (2006) Factors inhibiting bioremediation of soil contaminated with weathered oils and drill cuttings. Environ Pollut 144:255–265
Chaineau CH, Rougeux G, Yepremian C, Oudot J (2005) Effects of nutrient concentration on the biodegradation of crude oil and associated microbial populations in the soil. Soil Biol Biochem 37:1490–1497
Coulon F, Delille D (2003) Effects of biostimulation on growth of indigenous bacteria in sub-Antarctic soil contaminated with oil hydrocarbons. Oil Gas Sci Technol 58:469–479
Cunningham CJ, Ivshina IB, Lozinsky VI, Kuyukina MS, Philp JC (2004) Bioremediation of diesel-contaminated soil by microorganisms immobilised in polyvinyl alcohol. Int Biodeterior Biodegradation 54:167–174
Devinny J, Chang SH (2000) Bioaugmentation for soil bioremediation. In: Wise DL, Trantolo D (eds) Bioremediation of contaminated soils. Dekker, New York, pp 465–488
Diaz-Ramirez IJ, Escalante-Espinosa E, Favela-Torres E, Gutierrez-Rojas M, Ramirez-Saad H (2008) Design of bacterial defined mixed cultures for biodegradation of specific crude oil fractions, using population dynamics analysis by DGGE. Int Biodeterior Biodegradation 62:21–30
Fantroussi SE, Agathos SN (2005) Is bioaugmentation a feasible strategy for pollutant removal and site remediation? Curr Opin Microbiol 8:268–275
Farinazleen MG, Raja Noor ZA, Abu BS, Mahiran B (2004) Biodegradation of hydrocarbons in soil by microbial consortium. Int Biodeterior Biodegradation 54:61–67
Filonov AE, Akhmetov LI, Puntus IF, Esikova TZ, Gafarov AB, IzmalkovaTY SSL, Kosheleva IA, Boronin AM (2005) The construction and monitoring of genetically tagged, plasmid-containing, naphthalene degrading strains in soil. Microbiology 74:453–458
Gentry TJ, Rensing C, Pepper IL (2004) New approaches for bioaugmentation as a remediation technology. Crit Rev Environ Sci Tech 34:447–494
Goi A, Kulik N, Trapido M (2006) Combined chemical and biological treatment of oil contaminated soil. Chemosphere 63:1754–1763
Gouda MK, Omar SH, Nour Eldin HM, Checkroud ZA (2008) Bioremediation of kerosene II: a case study in contaminated clay (Laboratory and field: scale microcosms). World J Microbiol Biotechnol 24:1451–1460
Hamby DM (1996) Site remediation techniques supporting environmental restoration activities: a review. Sci Total Environ 191:203–224
Hamdi H, Benzarti S, Manusadzianas L, Aoyama I, Jedidi N (2007) Bioaugmentation and biostimulation effects on PAH dissipation and soil ecotoxicity under controlled conditions. Soil Biol Biochem 39:1926–1935
Heinaru E, Merimaa M, Viggor S, Lehiste M, Leito I, Truu J, Heinaru A (2005) Biodegradation efficiency of functionally important populations selected for bioaugmentation in phenol- and oil-polluted area. FEMS Microbiol Ecol 51:363–373
Horel A, Schiewer S (2009) Investigation of the physical and chemical parameters affecting biodegradation of diesel and synthetic diesel fuel contaminating Alaskan soils. Cold Reg Sci Technol 58:113–119
Hosokawa R, Nagai M, Morikawa M, Okuyama H (2009) Autochthonous bioaugmentation and its possible application to oil spills. World J Microbiol Biotechnol 25:1519–1528
Huling SG, Bledsoe BE, White MV (1990) Enhanced bioremediation utilizing hydrogen peroxide as a supplemental source of oxygen: a laboratory and field study. US EPA RS Kerr Environ Res Lab, Ada, OK, Rep EPA/600/2-90/006. http://nepis.epa.gov/Exe/ZyPURL.cgi?Dockey=20007M9S.txt
Jacques RJS, Okeke BC, Bento FM, Teixeira AS, Peralba MCR, Camargo FAO (2008) Microbial consortium bioaugmentation of a polycyclic aromatic hydrocarbons contaminated soil. Bioresour Technol 99:2637–2643
Khan FI, Husain T, Hejazi R (2004) An overview and analysis of site remediation technologies. J Environ Manage 71:95–122
Lai CC, Huang YC, Wei YH, Chang JS (2009) Biosurfactant-enhanced removal of total petroleum hydrocarbons from contaminated soil. J Hazard Mater 167:609–614
Leahy JG, Colwell RR (1990) Microbial degradation of hydrocarbons in the environment. Microbiol Rev 54:305–315
Lee M, Kim MK, Kwon M, Park BD, Kim MH, Goodfellow M, Lee S (2005) Effect of the synthesized mycolic acid on the biodegradation of diesel oil by Gordonia nitida strain LE31. J Biosci Bioeng 100:429–436
Liang Y, Zhang X, Dai D, Li G (2009) Porous biocarrier-enhanced biodegradation of crude oil contaminated soil. Int Biodeterior Biodegradation 63:80–87
Lima D, Viana P, Andre S, Chelinho S, Costa C, Ribeiro R, Sousa JP, Fialho AM, Viegas CA (2009) Evaluating a bioremediation tool for atrazine contaminated soils in open soil microcosms: the effectiveness of bioaugmentation and biostimulation approaches. Chemosphere 74:187–192
Liu B, Banks M, Schwab P (2001) Effects of soil water content on biodegradation of phenanthrene in a mixture of organic contaminants. Soil Sediment Contam 10:633–658
Malina G (1999) The bioventing of unsaturated zone contaminated with oil compounds. Monograph 66, Wydawnictwo Politechniki Częstochowskiej, Częstochowa
Malina G (2007) Risk reduction of soil and groundwater at contaminated areas. Monograph 132, Wydawnictwo Politechniki Częstochowskiej, Częstochowa
Malina G, Zawierucha I (2007) Potential of bioaugmentation and biostimulation for enhancing intrinsic biodegradation in oil hydrocarbon-contaminated soil. Biorem J 11:141–147
Mancera-Lopez ME, Esparza-Garcia F, Chavez-Gomez B, Rodriguez-Vazquez R, Saucedo-Castaneda G, Barrera-Cortes J (2008) Bioremediation of an aged hydrocarbon-contaminated soil by a combined system of biostimulation-bioaugmentation with filamentous fungi. Int Biodeterior Biodegradation 61:151–160
Mariano AP, Kataoka AP, Angelis D, Bonotto DM (2007) Laboratory study on the bioremediation of diesel oil contaminated soil from a petrol station. Braz J Microbiol 38:346–353
Massa V, Infantino A, Radice F, Orlandi V, Tavecchio F, Giudiuci R, Conti F, Urbini G, Guardo D, Barbieri P (2009) Efficiency of natural and engineered bacterial strains in the degradation of 4-chlorobenzoic acid in soil slurry. Int Biodeterior Biodegradation 63:112–125
Menendez-Vega D, Gallego JLR, Pelaez AI, de Cordoba GF, Moreno J, Munoz D, Sanchez J (2007) Engineered in situ bioremediation of soil and groundwater polluted with weathered hydrocarbons. Eur J Soil Biol 43:310–321
Mishra S, Sarma PM, Lal B (2004) Crude oil degradation efficiency of a recombinant Acinetobacter baumannii strain and its survival in crude oil-contaminated soil microcosm. FEMS Microbiol Lett 235:323–331
Mohan SV, Kisa T, Ohkuma T, Kanaly RA, Shimizu Y (2006) Bioremediation technologies for treatment of PAH-contaminated soil and strategies to enhance process efficiency. Rev Environ Sci Biotechnol 5:347–374
Mrozik A, Piotrowska-Seget Z (2009) Bioaugmentation as a strategy for cleaning up of soils contaminated with aromatic compounds. Microbiol Res. doi:10.1016/j.micres.2009.08.001 Microbiol Res 165:363–375
Mulligan CN (2005) Environmental applications for biosurfactants. Environ Pollut 133:183–198
Nievas ML, Commendatore MG, Esteves JL, Bucala V (2008) Biodegradation pattern of hydrocarbons from a fuel oil-type complex residue by an emulsifier-producing microbial consortium. J Hazard Mater 154:96–104
Nikolopoulou M, Pasadakis N, Kalogerakis N (2007) Enhanced bioremediation of crude oil utilizing lipophilic fertilizers. Desalination 211:286–295
Odokuma LO, Dickson AA (2003) Bioremediation of a crude oil polluted tropical rain forest soil. Global J Environ Sci 2:29–40
Oh YS, Sim DS, Kim SJ (2003) Effectiveness of bioremediation on oil-contaminated sand in intertidal zone. J Microbiol Biotechnol 13:437–443
Olexsey RA, Parker RA (2006) Current and future in situ treatment techniques for the remediation of hazardous substances in soil, sediments, and groundwater. In: Twardowska I, Allen HE, Häggblom MM, Stefaniak S (eds) Viable methods of soil and water pollution: monitoring, protection and remediation. Earth and Environmental Sciences, 69, pp 211–219
Perfumo A, Banat IM, Marchant R, Vezzulli L (2007) Thermally enhanced approaches for bioremediation of hydrocarbon-contaminated soils. Chemosphere 66:179–184
Rahman KSM, Rahman TJ, Kourkoutas Y, Petsas I, Marchant R, Banat IM (2003) Enhanced bioremediation of n-alkane in petroleum sludge using bacterial consortium amended with rhamnolipid and micronutrients. Bioresour Technol 90:159–168
Ramirez ME, Zapien B, Zegarra HG, Rojas NG, Fernandez LC (2009) Assessment of hydrocarbon biodegradability in clayed and weathered polluted soils. Int Biodeterior Biodegradation 63:347–353
Riser-Roberts E (1998) Remediation of petroleum contaminated soil: biological, physical, and chemical processes. CRC, LLC Boca Raton, Florida
Rodrigues JLM, Kachel A, Aiello MR, Quensen JF, Maltseva OV, Tsoi TV, Tiedje JM (2006) Degradation of Aroclor 1242 dechlorination products in sediments by Burkholderia xenovorans LB400(ohb) and Rhodococcus sp. strain RHA1(fcb). Appl Environ Microbiol 72:2476–2482
Rosenberg E, Ron EZ (1996) Bioremediation of petroleum contamination. In: Crawford RL, Crawford DL (eds) Bioremediation: principles and applications. Cambridge University Press, Cambridge, pp 100–124
Rous JD, Sabatini DA, Suflita JM, Harwell JH (1994) Influence of surfactants on microbial degradation of organic compounds. Crit Rev Environ Sci Tech 24:325–370
Ruberto L, Vazquez SC, Cormack WPM (2003) Effectiveness of the natural bacterial flora, biostimulation and bioaugmentation on the bioremediation of a hydrocarbon contaminated Antarctic soil. Int Biodeterior Biodegradation 52:115–125
Sarkar D, Ferguson M, Datta R, Birnbaum S (2005) Bioremediation of petroleum hydrocarbons in contaminated soils: comparison of biosolids addition, carbon supplementation and monitored natural attenuation. Environ Pollut 136:187–195
Sayler GS, Ripp S (2000) Field application of genetically engineered microorganisms for bioremediation processes. Curr Opin Biotechnol 11:286–289
Scherr K, Aichberger H, Braun R, Loibner AP (2007) Influence of soil fractions on microbial degradation behaviour of mineral hydrocarbons. Eur J Soil Biol 43:341–350
Sturman PJ, Stewart PS, Cunningham AB, Bouwer EJ, Wolfram JH (1995) Engineering scale-up of in situ bioremediation processes: a review. J Contam Hydrol 19:171–203
Tongarun R, Luepromchai E, Vangnai AS (2008) Natural attenuation, biostimulation and bioaugmentation in 4-chloroaniline-contaminated soil. Curr Microbiol 56:182–188
Tsai TT, Kao CM (2009) Treatment of petroleum-hydrocarbon contaminated soils using hydrogen peroxide oxidation catalyzed by waste basic oxygen furnace slag. J Hazard Mater 170:466–472
Ubochi KC, Ibekwe VI, Ezeji EU (2006) Effect of inorganic fertilizer on microbial utilization of hydrocarbons on oil contaminated soil. Afr J Biotechnol 5:1584–1587
Ueno A, Ito Y, Yumoto I, Okuyama H (2007) Isolation and characterization of bacteria from soil contaminated with diesel oil and the possible use of these in autochthonous bioaugmentation. World J Microbiol Biotechnol 23:1739–1745
Urum K, Grigson S, Pekdemir T, McMenamy S (2006) A comparison of the efficiency of different surfactants for removal of crude oil from contaminated soils. Chemosphere 62:1403–1410
Vidali M (2001) Bioremediation: an overview. Pure Appl Chem 73:1163–1172
Vogel TM (1996) Bioaugmentation as a soil bioremediation approach. Curr Opin Biotechnol 7:311–316
Vogel TM, Walter MV (2001) Bioaugmentation. In: Hurst CJ, Crawford RL, Garland JL, Lipson DA, Mills AL (eds) Manual of environmental microbiology. American Society for Microbiology, Washington, DC, pp 952–959
Walton J, Labine P, Reidies A (1992) The chemistry of permanganate in degradative oxidations. In: Eckenfelder WW, Bowers AR, Roth JA (eds) Chemical oxidation technologies for the nineties. Technomic Publishing Company, New York, pp 205–221
Walworth J, Pond A, Snape I, Rayner J, Ferguson S, Harvey P (2007) Nitrogen requirements for maximizing petroleum bioremediation in a sub-Antarctic soil. Cold Reg Sci Technol 48:84–91
Whang LM, Liu PWG, Ma CC, Cheng SS (2009) Application of rhamnolipid and surfactin for enhanced diesel biodegradation: effects of pH and ammonium addition. J Hazard Mater 164:1045–1050
Wolfe S, Ingold CF (1981) Oxidation of olefins by potassium permanganate, mechanism of α-ketol formation. J Am Chem Soc 103:938–939
Wu Y, Luo Y, Zou D, Ni J, Liu W, Teng Y, Li Z (2008) Bioremediation of polycyclic aromatic hydrocarbons contaminated soil with Monilinia sp.: degradation and microbial community analysis. Biodegradation 19:247–257
Xie W (2003) Effect of bioventing remediation by surfactant on in-situ oil-contaminated soil. Taiwan, China (M.Sc. Dissertation. NYUST) http://ethesys.yuntech.edu.tw/ETD-db/ETD-search/view_etd?URN=etd-0823104-163027
Zawierucha I, Malina G (2006) Bioaugmentation as a method of biodegradation enhancement in oil hydrocarbons contaminated soil. Ecohydrol Hydrobiol 6:163–169
Zawierucha I, Szewczyk A, Malina G (2007) Surfactant-enhanced natural attenuation of oil hydrocarbons in soil: the effect of surfactant dose on biodegradation and dehydrogenases activity. In: Goliński P, Zabawa S (eds) Reclamation and revitalization of demoted areas. PZITS, Poznań, pp 237–246
Zawierucha I, Szewczyk A, Malina G (2008) Biostimulation with nutrients as a method of biodegradation enhancement in oil hydrocarbons contaminated soil. In: Malina G (ed) Reclamation and revitalization of demoted areas. PZITS, Poznań, pp 99–110
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Zawierucha, I., Malina, G. (2011). Bioremediation of Contaminated Soils: Effects of Bioaugmentation and Biostimulation on Enhancing Biodegradation of Oil Hydrocarbons. In: Singh, A., Parmar, N., Kuhad, R. (eds) Bioaugmentation, Biostimulation and Biocontrol. Soil Biology, vol 108. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-19769-7_8
Download citation
DOI: https://doi.org/10.1007/978-3-642-19769-7_8
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-19768-0
Online ISBN: 978-3-642-19769-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)