Abstract
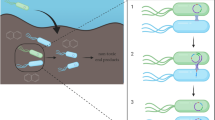
A genetically tagged, plasmid-containing, naphthalene-degrading strain, Pseudomonas putida KT2442(pNF142:: TnMod-OTc), has been constructed. The presence of the gfp gene (which codes for green fluorescent protein) and the kanamycin and rifampicin resistance genes in the chromosome of this strain allows the strain’s fate in model soil systems to be monitored, whereas a minitransposon, inserted into naphthalene biodegradation plasmid pNF142 and containing the tetracycline resistance gene, makes it possible to follow the horizontal transfer of this plasmid between various bacteria. Plasmid pNF142::TnMod-OTc is stable in strain P. putida KT2442 under nonselective conditions. The maximal specific growth rate of this strain on naphthalene is found to be higher than that of the natural host of plasmid pNF142. When introduced into a model soil system, the genetically tagged strain is stable and competitive for 40 days. The transfer of labeled plasmid pNF142::TnMod-OTc to natural soil bacteria, predominantly fluorescent pseudomonads, has been detected.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
Cerniglia, C.E., Biodegradation of Polycyclic Aromatic Hydrocarbons, Curr. Opin. Biotechnol., 1993, vol. 4, pp. 331–338.
Pieper, D.H. and Reineke, W., Engineering Bacteria for Bioremediation, Curr. Opin. Biotechnol., 2000, vol. 11, pp. 262–270.
Ramos-Gonzalez, M.-I., Duque, E., and Ramos, J.L., Conjugational Transfer of Recombinant DNA in Cultures and in Soils: Host Range of Pseudomonas putida TOL Plasmids, Appl. Environ. Microbiol., 1991, vol. 57, no.10, pp. 3020–3027.
Stuart-Keil, K.G., Hohnstock, A.M., Drees, K.P., Herrick, J.B., and Madsen, E.L., Plasmids Responsible for Horizontal Transfer of Naphthalene Catabolism Genes between Bacteria at a Coal Tar-Contaminated Site Are Homologous to pDTG1 from Pseudomonas putida NCIB 9816-4, Appl. Environ. Microbiol., 1998, vol. 64, no.10, pp. 3633–3640.
Sayler, G.S., Hooper, S.W., Layton, A.C., and Henry, King, J.M., Catabolic Plasmids of Environmental and Ecological Significance, Microbiol. Ecol., 1990, vol. 19, pp. 1–20.
Boronin, A.M., Diversity of Pseudomonas Plasmids: To What Extent?, FEMS Microbiol. Lett., 1992, vol. 79, nos.1–3, pp. 461–468.
Stotzky, G. and Babich, H., Survival of, and Genetic Transfer by Genetically Engineered Bacteria in Natural Environments, Adv. Appl. Microbiol., 1986, vol. 31, pp. 93–137.
Top, E., Springael, D., and Boon, N., Catabolic Mobile Genetic Elements and Their Potential Use in Bioaugmentation of Polluted Soils and Waters, FEMS Microbiol. Ecol., 2002, vol. 42, pp. 199–208.
Dennis, J.J. and Zylstra, G.J., Plasposons: Modular Self-Cloning Minitransposon Derivatives for Rapid Genetic Analysis of Gram-Negative Bacterial Genomes, Appl. Environ. Microbiol., 1998, vol. 64, no.7, pp. 2710–2715.
Bagdasarian, M., Lurz, R., Ruchert, B., Franklin, F.C.H., Bagdasarian, M.M., Frey, J., and Timmis, K.N., Specific-Purpose Plasmid Cloning Vectors: II. Broad-Host-Range, High-Copy-Number, RSF1010-Derived Vectors for Gene Cloning in Pseudomonas, Gene, 1981, vol. 16, nos.1–3, pp. 237–247.
Dunn, N.W. and Gunsalus, I.C., Transmissible Plasmid Coding for Early Enzymes of Naphthalene Oxidation in Pseudomonas putida, J. Bacteriol., 1973, vol. 114, pp. 974–979.
Carhart, G. and Hegeman, G., Improved Method of Selection for Mutants of Pseudomonas putida, Appl. Microbiol., 1975, vol. 30, no.6, p. 1046.
King, O.E., Ward, W., and Raney, D.E., Two Simple Media for Demonstration of Pyocyanin and Fluorescin, J. Lab. Clin. Methods, 1954, vol. 44, no.2, pp. 301–307.
Evans, C.G.T., Herbert, D., and Tempest, D.B., The Continuous Cultivation of Microorganisms: 2. Construction of a Chemostat, Methods Microbiol, 1970, vol. 2, pp. 277–327.
Sambrook, J., Fritsch, E.F., and Maniatis, T., Molecular Cloning: A Laboratory Manual, New York: Cold Spring Harbor Lab., 1989.
Birnboim, H.C. and Doly, J.A., A Rapid Alkaline Extraction Procedure for Screening Recombinant Plasmids DNA, Nucleic Acids Res., 1979, vol. 7, no.6, p. 1513.
James, F. and Roos, M., MINUIT, a System for Function Minimization and Analysis of the Parameters Errors and Correlation, Comput. Phys. Commun., 1975, vol. 10, pp. 343–367.
Filonov, A.E. and Boronin, A.M., Plasmid Stability and the Competitiveness of Plasmid-containing and Plasmid-less Strains during Continuous Cultivation, Antibiot. Khimioter., 1990, vol. 35, no.5, pp. 46–50.
Gorb, T.E. and Tovkach, F.I., Method for Studying the Horizontal Transfer of Plasmids in Erwinia carotovora, Mikrobiol. Zh. (Kiev), 2002, vol. 64, no.3, pp. 20–26.
De Rore, H., Demolder, K., de Wilde, K., Top, E., Houwen, F., and Verstraete, W., Transfer of the Catabolic Plasmid RP4::Tn4377 to Indigenous Soil Bacteria and Its Effect on Respiration and Biphenyl Breakdown, FEMS Microbiol. Ecol., 1994, vol. 15, pp. 71–78.
Newby, D.T., Gentry, T.J., and Pepper, I.L., Comparison of 2.4-Dichlorophenoxyacetic Acid Degradation and Plasmid Transfer in Soil Resulting from Bioaugmentation with Two Different pJP4 Donors, Appl. Environ. Microbiol., 2000, vol. 66, no.8, pp. 3399–3407.
Brokamp, A.F. and Schmidt, R.J., Survival of Alcali-genes xylosoxidans Degrading 2.2-Dichloropropionate and Horizontal Transfer of Its Halidohydrolase Gene in a Soil Microcosm, Curr. Microbiol., 1991, vol. 22, pp. 299–306.
Herrick, J.B., Stuart-Keil, K.G., Ghiorse, W.C., and Madsen, E.L., Natural Horizontal Transfer of a Naphthalene Dioxygenase Gene between Bacteria Native to a Coal Tar-Contaminated Field Site, Appl. Environ. Microbiol., 1997, vol. 63, no.6, pp. 2330–2337.
Author information
Authors and Affiliations
Corresponding author
Additional information
__________
Translated from Mikrobiologiya, Vol. 74, No. 4, 2005, pp. 526–532.
Original Russian Text Copyright © 2005 by Filonov, Akhmetov, Puntus, Esikova, Gafarov, Izmalkova, Sokolov, Kosheleva, Boronin.
Rights and permissions
About this article
Cite this article
Filonov, A.E., Akhmetov, L.I., Puntus, I.F. et al. The Construction and Monitoring of Genetically Tagged, Plasmid-Containing, Naphthalene-Degrading Strains in Soil. Microbiology 74, 453–458 (2005). https://doi.org/10.1007/s11021-005-0088-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11021-005-0088-6