Abstract
Plants being sessile are continuously exposed to a wide range of biotic and abiotic stresses that exert adverse effect in their growth and development. Various physiological, biochemical, and molecular machineries are employed by the plants to overcome these stresses. Endophytes are mostly the symbiotic fungi and bacteria that reside inside the plant tissue and stimulate plant growth during stress conditions. Endophyte-mediated plant stress tolerance holds significant role in the analysis of plant-microbe interactions. Although still at its infancy, the endophyte-mediated host stress tolerance including drought, salinity, high-temperature stresses, and pathogenic infection has been well described in the recent times. The molecular mechanism governing the endophyte-mediated stress response includes the induction of plant stress genes and regulation of reactive oxygen species. In the present review, we discuss the evidences for bacterial and fungal endophyte-mediated stress tolerance and associated mechanisms. This information from this review will help the scientific community in the development of suitable biotechnological approaches toward usage of endophyte microbes in the improvement of crop yield under multiple stress conditions.
Access provided by Autonomous University of Puebla. Download reference work entry PDF
Similar content being viewed by others
Keywords
1 Introduction
Endophytes are the group of ancestral endosymbionts, primarily consisting of bacteria, fungi, and actinomycetes that reside for at least a part of their life cycle or throughout life without causing any harm or disease in plants across the arctic to tropical regions of the world [1]. They survive in different parts of the plant including healthy stems, roots, twigs, node, internode, petioles, bark, leaves, fruit, flower, and seeds without exhibiting any infection symptoms or severity in the host tissues [2]. Geological time scale has shown that plant and endophyte interactions have originated as early as the origin of the first group of higher plants on earth [3, 4]. The mutualistic symbiotic behavior of the endophytes and plants is demonstrated from the fact that the host plant gives the shelter and nutrients to endophytes while the later increases the survival chance of the host plant by increasing the tolerance effect to insect herbivory as well as biotic and abiotic stresses [5, 6]. Interaction between plants and microbes can be mostly classified as detrimental or neutral. In most of the cases, the interaction is considered as beneficial, because microbes consume the plants organic product for respiration and metabolism and at the same time help in nutrient recycling and tolerance against various stresses. Beneficial microbes encourage plant growth development and inhibit the plant diseases by enhancing different types of the mechanisms which mainly include production of growth regulators, hormones, and pathogen-inhibiting compounds. In rare cases, the endophytic fungi, bacteria, and viruses have detrimental effect on the host plants [7].
Endophytes have received significant importance in the recent times as they could affect the interactions of plants with their environment and alter the course of their interaction with infecting pathogens [8]. They have the ability to adapt in various adverse environments including nutrient-deficient conditions, heavy pollution, and rigid environmental conditions and produce huge amount of bioactive compounds that are essentially used in pharmaceutical industries [9,10,11]. Endophytic fungi not only inhibit the heat and light-mediated destruction of photosynthetic apparatus, but they also increase the number of photosynthetically active pigments in plants [12, 13]. Bacterial and fungal endophytes facilitate the biological degradation of dead plants which is essential for nutrient recycling and help in phytoremediation of polluted soil and water [14, 15]. Some of the endophytes exhibit a good metal sequestration and chelation system that helps in higher tolerance effect toward the heavy metals [16]. Volatile antibiotics secreted from endophytic fungi inhibit growth of the pathogen through mycofumigation [17, 18]. Endophytes may also increase host fitness and competitive abilities by increasing successful germination and growth rate or enhancing the absorption of nutritional elements by the host [19]. In spite of all these activities, the role of endophytes in crop improvement is still inconspicuous. Recent findings clearly show that endophytes may have an important influence in the regulation of plant growth and stress responses leading to augmentation of productivity. In this chapter, we aim at acquainting the readers with the generation characteristics of endophytes and specifically focus on those facets of endophyte research causing plant improvement through modulation of biotic and abiotic stress responses.
2 Response of Plants to External Stresses
Lack of mobility together with the absence of any specialized immune system in plants brings in numerous challenges for plants in the form of biotic and abiotic stresses. Often, the growth and development of plants gets compromised in overcoming such environmental stresses. With due course of evolution, plants have developed complex regulatory networks, which not only provide an appropriate defense response against specific stresses but also determine the pertinent resource distribution between plant development and defense. Several plants modify their outermost structures such as leaves into trichomes and spines and deposit epidermal layers of lignins, resins, and silica to limit pathogen invasions and discourage herbivory. A diverse repertoire of secondary metabolites further aid in plant defense responses [20].
As a second line of defense, plants can perceive various stress stimuli and can induce local and systemic defense responses [21]. Recognition of pathogens or the pathogen-associated molecular patterns (PAMPs) by the membrane-bound pattern recognizing receptor (PRR) channels activates the plant immune responses through the production of reactive oxygen species (ROS), hypersensitive responses (HR), programmed cell death, tissue reinforcement at the site of infection, and expression of defense-related proteins [21,22,23]. This kind of immune response is often referred to as the PAMP-triggered immunity (PTI). These induced local defense responses consequently lead to the establishment of immune response throughout the plant known as the systemic acquired resistance (SAR) [21]. Having said that, successful pathogen invasions often break down the PTI immunity in plants by secreting several effectors into plant cells. However, the plant resistance genes (R genes), mostly characterized as possessing the nucleotide-binding site (NBS) and leucine-rich repeat (LRR) domains meticulously recognize the pathogen effectors and thus trigger another type of immune response known as effector-triggered immunity (ETI) [22].
Plants possess a complex and intertwined networks of signaling pathways consisting of multiple signal molecules, phytohormones, and protein kinases. The earliest possible signaling event includes the perception of abiotic and biotic stresses through the induction of Ca2+ influx and ROS accumulations in the affected plants [21, 23]. Stress stimuli and presence of secondary signaling molecules like ROS can lead to the accumulation of several phytohormones including abscisic acid (ABA), jasmonic acid (JA), salicylic acid (SA), gibberellins (GA), and ethylene (ET) [24]. Besides, a large number of plant kinases play crucial roles in plant defense signaling and provide a highly complex crosstalk network among all the players involved in defense response. Plant kinases including calcium-dependent protein kinase (CDPK), cyclin-dependent protein kinase (CDK), and mitogen-activated protein kinase (MAPK) have been reported in numerous studies to participate, modulate, and confer defense against many biotic and abiotic stresses in plants [25,26,27,28,29]. Thus, plant response to different stresses in their natural habitat involves the maintenance of balance and fine-tuning of these complex repertoire of defense strategies.
3 Endophytic Symbionts
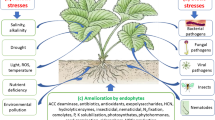
Symbiosis is defined as the mutualistic, parasitic, or commensalic interactions between two organisms for throughout the life or some part of their life [30]. Endophytes are one of the most unexplored and diverse group of organisms that make symbiotic associations with higher life forms and may produce beneficial substances for host [31]. Plants form mutualistic symbioses with a variety of microorganisms including endophytic fungi that live inside the plant and cause no overt symptoms of infection. The microbes enter into the plants through natural pores or wounds created by biotic stress and abiotic stresses and penetrate inside through the secretion of a wide range of cell wall-degrading enzyme such as cellulase, cutinase, protease, pectinase, hemicelluloses, and lignin peroxidase [32] (Fig. 1). The production of ROS and the subsequent activation of multiple cellular mechanisms including programmed cell death, necrosis, and systemic signaling help in maintaining the mutualistic behavior between the plants and the microbes. However, the plants’ response to biotic and abiotic stresses due to the result of fungal, plant, or symbiotic metabolism is still poorly understood [33,34,35]. A fungal endophyte, Neotyphodium coenophialum, present in the Lolium grasses helps in preventing herbivory by producing different types of secondary metabolites [36]. Orchid mycorrhizal fungi provide necessary nutrients critical to symbiotic seed germination [37]. Continued interaction between plant and endophytes may also result in exchange of genetic material leading to sustained synthesis of microbial-derived bioactive compounds from generation after generation in the plant system [38, 39]. Establishment and maintenance of this long-term association between plants and fungus is regulated by a group of stress-activated mitotic kinases (SaKAs). Deletion of SaKA gene(s) often converts the mutualistic interaction into pathogenic action. SaKA mutant fungi often result in developmental defects of plants including stunted growth, loss of apical dominance, and premature senescence [40]. MAP kinases (MAPKs) are another group of enzymes that are expressed during plant-microbe interaction. Activation of MAP kinase pathway only results in the accumulation of ROS which have greater role in the maintenance of abiotic stress tolerance and immunity against the invading pathogen and enhancing endophytic growth [41, 42]. Temperature regulation is another important aspect of symbiotic relationship between endophyte and plants. While both the plants and endophyte can tolerate high temperature up to 65 °C during symbiotic growth, they cannot survive above 40 °C under nonsymbiotic existence [43]. Besides, endophytic symbionts also help in crop improvement through production of antifungal, antibacterial, and nematicides which inhibits the pathogenesis in the crop field. Likewise, siderophore produced by the endophytic fungi helps in uptake of iron in low iron habitats and helps in providing nutrient supplements to the plants [44]. Overall, the symbiotic relationship between plants and microbes especially the fungi is highly significant for growth, development, and productivity of crop plants.
4 Endophyte-Mediated Plant Growth
Water, light, and nutrients are integral to the growth, reproduction, and survival of plants under different geographic regions. Often under natural conditions, the plants also face unfavorable and hostile conditions collectively known as abiotic stresses hampering homeostasis and growth. Being sessile, plants are exposed to a broad range of environmental stresses as well as stresses induced by other living and nonliving systems that they cannot escape. Extreme environmental conditions below or above the optimal levels often limit plant growth and development. Drought, low- or high temperature, salt stress and acidic conditions, heavy metal stress, nutrient stress, and starvation are the major abiotic stresses that affect the plants [45]. Biotic stresses include the damage done to plants by bacteria, viruses, fungi, parasites, harmful insects, weeds, and cultivated or native plants. Fortunately, many microorganisms including bacteria, fungi, and protozoa form beneficial or symbiotic association with plants which benefit them not only to overcome various environmental stresses but also support their growth and development. Although extensive studies have been performed in understanding the mutualistic interactions between plants and soil microbes, the interaction between plants and endophytes has only been lately realized. Plant-endophyte interactions have revealed that endophytes help the plants by promoting growth and enhanced defense responses, whereas the plants possess the ability to choose these valuable microbiomes allowing to colonize within its tissues [46, 47]. These endophytes significantly contribute to the regulation of many vital physiological processes and promote the overall plant vigor and growth. For instance, endophytic fungi facilitate the degradation of the cuticular cellulose resulting in improved carbon uptake and promote seed germination [48]. Endophytes serve the growth and development of host plant by (1) assisting in the production of hormones and compounds, (2) aiding in acquiring essential nutrients, and (3) enhancing plant defense against biotic and abiotic stresses.
4.1 Production of Phytohormones and Other Growth-Inducing Compounds
Phytohormones are the regulatory molecules that are essentially involved in the growth, physiological processes, and defense responses in plants [49,50,51]. The colonized endophytes can effectively alter the phytohormonal homoeostasis, thereby accelerating the plant growth [52]. Most of the plant endophytes, especially those associated in plant roots, accelerate the plant growth and development via production of the auxin class hormone indoleacetic acid (IAA). Several physiological processes are influenced by IAA homeostasis including cell differentiation, seed germination, development of vascular tissues, root formation and elongation, vegetative growth, pigmentation, and photosynthesis [53]. Colonization of the plant-growth-promoting bacterial endophytes in Solanum nigrum and Nicotiana attenuata promoted root growth by 1-aminocyclopropane-1-carboxylate (ACC) deaminase activity and elevated production of indole-3-acetic acid (IAA) in the former, whereas only by IAA accumulation in the later, respectively [52]. On the other hand, production of IAA can result in increased colonization efficiency of the bacterial endophytes, thus maximizing the endophyte-mediated plant growth [54]. In Populus trichocarpa, a diazotroph endophytic bacteria Burkholderia vietnamiensis produces IAA required for plant growth [55]. Similarly, Bal et al. [56] identified six endophytic bacteria from rice plants grown in the costal fields which produced IAA and ACC deaminase. Further, inoculation of the isolated bacterial strains into an indica rice cultivar “Naveen” resulted in noticeable plant growth enhancements with elongated roots and increased IAA production and ACC deaminase activity. In japonica rice cultivar Dongjin, the association of endophytic fungi Paecilomyces formosus conferred enhanced plant growth reflected by improved plant height, biomass, and chlorophyll content compared to plants having no endophytic associations [57]. Passari et al. [58] isolated BPSAC6, an endophytic bacterial strain of Bacillus sp. from Clerodendrum colebrookianum which could produce three different phytohormones including IAA, kinetin, and 6-benzyladenine.
Although endophytes can promote the growth kinetics of plants via IAA production, they can also contribute in limiting the production of other phytohormones like GA, JA, and ABA to help the plants overcome various stresses [58, 59]. Often, GA-producing endophytic microorganisms contribute toward improved yield of the host plant. Hamayun et al. [60] isolated an endophytic fungi Phoma herbarum from the salt-stressed soybean plant roots, which showed promising plant growth-promoting characters resulting in increased biomass and elevated production of active GAs including GA1, GA3, GA4, and GA7. Similarly, endophytic fungi Cladosporium sp. isolated from cucumber have plant growth-promoting properties through the production of several active and inactive GAs [60]. Further, in Moringa peregrine, two fungal endophytes Aspergillus caespitosus LK12 and Phoma sp. LK13 isolated from the bark tissues were reported to enhance plant growth by producing bioactive GAs [61]. Besides, endophytes play key roles in regulating the cytokine and ET levels in plants. Ethylene has multitude roles in plants and actively participates in plant defense signaling. The elevated level of endogenous ET during stress response often negatively affects the growth and overall plant development [53, 62]. The endophytes having ACC-deaminase activity catabolize the ET precursor molecule ACC to 2-oxobutanoate and NH3 resulting in a lower level of ET after neutralization of the pathogen attack [63]. The sunflower endophytic bacterial strains SF2, SF3, and SF4 were reported to enhance plant growth under water stress by producing SA [64]. Interestingly, in Arabidopsis and its endophytic fungus Piriformospora indica association, P. indica produces high levels of cytokinins. The trans-zeatin biosynthesis and the cytokinin response 1/Arabidopsis histidine kinase 2 (CRE1/AHK2) receptor combinations are essential for P. indica-mediated growth in Arabidopsis. Arabidopsis mutant lines having impaired CRE1/AHK2 receptor combination or lacking cis-zeatin resulted in reduced cytokinin levels even in colonized roots [65].
Endophytes assist their host plants growth by producing other kinds of growth-simulating chemicals apart from the phytohormones. These chemical entities range from metabolites like sugars and polyamines to volatile compounds. The bacterial endophyte Methylobacterium extorquens and the fungal endophyte Rhodotorula minuta in Pinus sylvestris plants produce adenines and adenine ribosides which serve as potential precursor molecules in cytokine biosynthesis and control the morphological attributes of the plant [66]. In Arabidopsis, exposure to endophytic volatile organic compounds (VOCs) including acetoin and 2, 3-butanediol resulted in enhanced plant growth and better defense responses as compared to the wild relatives [67, 68]. There are perhaps many more chemicals and bioactive compounds synthesized by the endophytes which remains to be discovered and characterized having significant involvement in plant growth development.
4.2 Role of Endophytes in Nutrient Acquisition
Endophytes play crucial roles in nutrient acquisitions in plants from the natural habitat. Many plants lack the natural machinery to acquire some essential nutrients. For instance, nitrogen is a major requirement for plants for its growth and survival. However, most of the plants can’t use the atmospheric nitrogen and heavily rely upon supplies of nitrogenous fertilizers. Others make symbiotic associations with the nitrogen-fixating bacteria, mostly seen in legumes, which help the plants to utilize the atmospheric nitrogen. However, the colonization of endophytes is markedly different than those of rhizobial nitrogen-fixating symbionts. While the rhizobial bacteria colonize within the plant much similar to infections, the endophytes enter into plant roots via root junctions and wounding sites [69]. Plant growth-associated traits provided by different endophytic microorganisms are listed in Table 1. Unlike rhizobial bacteria, endophytes also protect the oxygen-sensitive nitrogenase enzyme by adopting different strategies. Oxygen is present in low concentration in the natural rhizobial conditions, and presence of leg hemoglobin assists to scavenge the traces of free oxygen in the nodules. However, the endophytes create a low oxygen environment condition by adopting various biochemical or physical modifications including exopolysaccharides and interior vesicles to exclude free oxygen and compartmentalize the nitrogenase [70]. Carbohydrates produced through photosynthesis are offered to the endophytic symbionts in return of usable nitrogen. The symbionts convert atmospheric nitrogen to ammonia by the help of nitrogenase enzyme and energy. The most studied associations are the rhizobial and actinorhizal plant-bacterial symbioses where several bacterial root endophytes such as Azoarcus spp., Herbaspirillum spp., and Acetobacter spp. fix the atmospheric nitrogen [71]. Such endophytes converting atmospheric nitrogen into the plant usable ammonia or nitrate form are known as “diazotrophs” and found in important crops including rice, sugarcane, sweet potato, maize, and coffee [69].
The endophytic associations between Gluconacetobacter diazotrophicus and sugarcane and pines are the well-studied symbiotic associations where the endophyte helps the host plant in nitrogen fixation [71]. In sugarcane, G. diazotrophicus facilitate a microaerobic environment to protect nitrogenase enzyme by metabolizing sucrose at an extreme rate to fix atmospheric nitrogen. This phenomenon is known as the respiratory protection, which is peculiar only to endophytes than the rhizobial symbionts [72]. Apart from G. diazotrophicus, endophytes from other genera have also been discovered to be associated with the cash crop sugarcane functioning in nitrogen fixation process. Mixture of endophytic bacterial inoculation including Burkholderia, Azospirillum, and Herbaspirillum with Gluconacetobacter into sugarcanes resulted in enhanced nitrogen fixation efficiency [73]. The resultant nitrogen fixation in sugarcane was found to double the amount as compared to inoculation of a single bacterial strain. The abundance of B. vietnamiensis endophytes in sugarcane was reported to fix nitrogen by producing nitrogenase enzyme and reducing acetylene, an indirect assay for nitrogen fixation [74].
In rice, 13 nitrogen-fixating endophytes from the Burkholderia genus were confirmed positive for nifD gene and acetylene reduction assay (ARA) [75]. GUS assay revealed that B. vietnamiensis strain MGK3, an endophyte showing highest ARA activity, entered via the root tips and lateral root junctions and colonized in the intercellular spaces of the root cortex. In maize, B. unamae possessing nif gene cluster and acetylene reductase activity colonizes in roots and stems [76]. Apart from Burkholderia, endophytes from Rhizobium, Rhanella, Pantoea, Pseudomonas, Azospirillum, and Herbaspirillum genera were also reported to be found in maize which showed potential biological nitrogen fixation capabilities [77]. In another major crop wheat, Klebsiella pneumoniae was found to be an entophyte aiding wheat plants to fix atmospheric nitrogen [78]. In addition to these crops, many diazotrophic endophytes have been identified from plants like poplar, sweet potato, coffee, and cottonwood. Poplar endophytic bacteria Paenibacillus spp. strain P22 have been reported to help its host via nitrogen fixation and contributing toward overall increase in total nitrogen pool [79]. Another study reported that poplar and willow trees harbor numerous endophytes having putative nitrogen-fixating roles, mainly from genera of Burkholderia, Acinetobacter, Enterobacter, Pseudomonas, Herbaspirillum, Rahnella, and Sphingomonas [80]. Nitrogen fixation is a vital physiological process in plants that contributes to the overall vigor of the plant, especially for plants growing in low soil-nitrogen concentration areas. Thus, the endophyte-mediated nitrogen fixation serves as a crucial and highly beneficial function for the host plants resulting in proper plant growth and development.
Apart from nitrogen, iron act in many beneficial ways in plant physiology. Several plant endophytes are reported to play role in maintaining the iron homeostasis in plants. Siderophores, produced by the endophytes, are the high-affinity iron-chelating compounds to help in iron uptake [81]. A comparative genomic study of the bacterial endophytes by Mitter et al. [82] revealed that endophytes deficient of the siderophore biosynthesis genes possess additional clusters of membrane receptor encoding genes to facilitate iron transport across the cell membrane. In addition, siderophores help in building and maintaining the mutualistic relationship in some plants and their endophytes. Production of siderophores by Epichloë festucae was found to be an essential criterion to maintain the symbiotic relationship with the host grass species Lolium perenne [81].
5 Endophyte-Mediated Abiotic Stress Tolerance
Plant growth and development is often compromised by the onset of several environmental stresses as plants prioritize resistance over growth. In this scenario, the endophytes living inside the host plants come in great support in overcoming the challenges. Although endophytes are very short-lived as compared to their host, their shorter life span helps in their rapid evolution in aiding the host toward tackling the diversities. The capacity of different endophytes providing resistance against these environmental stresses are exploited in modern sustainable agriculture [83]. In a plant-endophyte association, the later adopts and employs various strategies to mitigate the abiotic stresses that come from the natural habitat of the host (Fig. 2). Mechanisms like production of growth-promoting hormones and other compounds and nitrogen fixations by plant endophytes, which have been already discussed in this chapter, are actually two major ways of counteracting the deficiency of nutrients in the habitat. Additionally, some endophyte can go a step ahead in providing critical protection to host plants against harsh environmental conditions not only by producing the growth-promoting compounds or nutrient acquisition but integrating key genetic elements into the host plant genome which leads to production of stress-responsive molecules in the plant [84].
Endophytes aid in plant stress resistance either by activating the host stress responses or by producing chemical metabolites that act as anti-stress compounds to mitigate the stress effects [85]. For instance, in cucumber, Penicillium and Phoma spp. not only confer resistance against hyper-sodium toxicity and polyethylene glycol (PEG)-mediated osmotic stress and drought but also positively regulate the growth parameters including plant biomass and nutrition acquisitions [86]. In addition, endophytes are reported to be involved in controlling the gene expressions in host plants with respect to the abiotic stresses [48, 87]. Plants like rice and tomato with beneficial endophytes can survive in water-deficit habitats and show better growth kinetics as compared to the plants lacking these endophytes. The common drought tolerance mechanisms provided by the endophytes include formation of thick cuticles, accumulation of more solutes in plant tissues, and reduction in stomatal transpiration [85]. Removal of Neotyphodium coenophialum endophytes from the tall fescue grass caused water intake in plant, nutrient acquisition, and photosynthesis [88]. In rice, the class 2 fungal endophytes originally classified by Rodriguez et al. [89] were reported to confer drought and salinity tolerance in two commercial cultivars originally intolerant to both drought and salinity [90]. Further, the same cultivars exhibited endophyte-mediated improved growth and biomass accumulation, enhanced yield, and 20–30% reduction in water consumption. Vahid et al. [91] reported the involvement of fungal endophyte Neotyphodium spp. in conferring drought resistance in Lolium perenne plants reflected by improved growth parameters in terms of higher biomass, plant height, and tiller numbers as compared to the non-infected plants. More recently, bacterial endophytes have been reported as controlling the expression of plant root vacuolar proton pumps [92]. The study reported that bacterial endophyte-mediated manifestation of H+-PPase genes resulted in upregulated expression of the root vacuolar proton pump conferring drought tolerance in Capsicum annuum.
Apart from poor irrigation or water-deficit driven drought stress, variation in the salt concentration of the soil is often considered a major threat to global agriculture. High salinity in soil often result in severe crop damage, compromised yield, and at times plant death. Plants possess many complex mechanisms to cope with soil salinity. Regulation of salinity by the mutualistic in planta dwellers, the endophytes, has been crucial in rescuing and overcoming such adversities. For instance, Piriformospora indica, a fungal endophyte isolated from the rhizosphere of Prosopis juliflora and Ziziphus nummularia, help its host to adapt the hostile dessert habitat [93]. Inoculation of P. indica into barley has resulted in enhanced resistance to biotic stresses and improved salt tolerance [94]. Further, Baltruschat et al. [95] reported that colonization by P. indica in the roots of the salt-sensitive barley cultivar “Ingrid” resulted in increased plant biomass and decreased salinity-induced lipid peroxidation, fatty acid desaturation, and metabolic heat efflux in the leaves. Additionally, endophyte-inoculated barley plants displayed elevated ascorbic acid accumulations and escalated activity of the antioxidant enzymes under salt stress. Inoculation of systemic class 2 fungal endophytes in tomato plants conferred salt tolerance in the infected plants [96]. Moreover, the infected plants exhibited higher plant biomass, improved water-use efficiency, and enhanced photosynthetic abilities than the uninfected tomato plants under salinity conditions. More recently, Aspergillus flavus CHS1 isolated from Chenopodium album has been reported to have high salt tolerance characteristics [97]. In the in planta CHS1-soybean interaction study, inoculation of CHS1 resulted in improved plant growth and mitigated the imposed salinity stress by subsiding ABA and JA synthesis. Moreover, activity of the antioxidant enzymes including superoxide dismutase (SOD), catalase (CAT), peroxidase, and polyphenol oxidase was found to be upregulated in compared to endophyte-free soybean plants under salt stress.
Mitigation of drought and salt stresses are usually associated with the accumulation and subsequent scavenging of reactive oxygen species (ROS) [98]. Although reduction in plant ROS levels can be aided by antioxidant compounds like glutathione, ascorbate, and tocopherol, the major scavengers of ROS include SOD, CAT, dehydroascorbate reductases (DHAR), monodehydroascorbate reductases (MDHAR), glutathione reductases (GR), and ascorbate or thiol-dependent peroxidases (APX) [99]. Accumulation of ROS in the plant cell can be toxic, leading to oxidative damage to proteins, lipids, and DNA. Having said that, ROS often act as an early event in the plant stress response signaling cascades [100]. Although production and accumulation of ROS can often be seen in plant-pathogen interactions, relatively low amount of ROS is also needed in the development of a beneficial plant-endophyte association. For example, the production of hydrogen peroxides or superoxides by Epichloë festucae is preconditioned by a mutualistic association between the fungal endophyte and the host Lolium perenne grass [34]. While low concentration of ROS is beneficial for the plant growth and signaling, elevated ROS accumulation may cause detrimental effects. Endophytes dwelling inside the plants help their hosts in managing ROS concentrations and thus protecting them from the detrimental effects of ROS. Exposure to high salt conditions caused ROS accumulations in tomato, panic grass, and dunegrass without any endophytes, whereas the endophyte-containing plants didn’t show ROS accumulations [101]. Interestingly, some endophytes produce ROS to check the colonization levels in plants and maintain mutualism, while others reduce the ROS levels to nullify the abiotic stress effects on their hosts [34, 43]. Besides, salinity and drought both can disturb the osmotic homeostasis in a plant causing imbalance in solute and ion concentrations within a plant cell. Osmotic stress stimuli can be perceived by the plants as both primary and secondary signals where accumulation of solutes, water loss, and ion influx/efflux belong to the primary signals and accumulation of phytohormones, generation of nitric oxide and phospholipids, and liberation of ROS belong to the secondary signals. In addition, change in the habitat temperature and soil contaminations with heavy metals has arisen as potential abiotic stresses for the plants. While the plant and its intrinsic machinery fail at times to provide protection against high temperature conditions, the colonizing endophytes rescue their host from these hostilities. For example, the fungal endophyte Curvularia protuberate in Dichanthelium lanuginosum grass helps the host to survive and tolerate soil temperatures as high as 65 °C [12]. Another Ascomycetes endophyte isolated from durum wheat (Triticum turgidum) conferred significant tolerance to drought and heat in the inoculated plants [102]. Interestingly, the endophyte-free seeds produced from the Ascomycetes-inoculated wheat plants under drought and heat stress showed increased germination rate than the non-inoculated wheat plant seeds [102]. Similarly, several plant endophytes, especially the root-associated endophytes, have been reported to support their host in heavy metal tolerance and mitigate metal toxicity. Several strains of Pestalotiopsis spp. isolated from the mangrove palm species Nypa fruticans conferred significant tolerance against heavy metals including copper (Cu), zinc (Zn), lead (Pp), and chromium (Cr) [103]. In vitro studies revealed that 1 out of the 93 endophytic isolates from the nipa palm could resist heavy metal contaminations to a level as high as 1000 ppm. Exophiala pisciphila, a root-associated fungal endophyte in maize, was reported to provide tolerance against soil cadmium (Cd) toxicity [104]. Upon subjection of Cd stress, the E. pisciphila-inoculated plants exhibited upregulated expression of maize genes responsible for metal uptake, translocation and chelation, increased Cd accumulation on maize cell walls, conversion of Cd to lesser toxic forms, and upregulated activities of antioxidants and antioxidant enzymes. Moreover, the plants containing endophytes displayed improved growth parameters with a significant decrease in Cd-induced phototoxicity levels as compared to the maize plants lacking the endophytes. Penicillium funiculosum LHL06 has been reported to secret gibberellin that alleviated metal toxicity and reprogramed the growth of soybean plants under Cu and Cd stress [105]. Similarly, endophytes like Pseudomonas spp. and Gigaspora spp. can improve plant resistance against metal toxicity by altering the endogenous ethylene levels [106]. More recently, Mukherjee et al. [107] reported that inoculation with Kocuria spp. and Enterobacter spp. isolated from Lantana camara into a surrogate host Solanum nigrum resulted in enhanced arsenic (As) tolerance and phytoremediation. Under an applied As consortium, endophyte-inoculated S. nigrum plants exhibited improved photosynthesis, root-to-shoot As transport, increased As bioaccumulation, and improved plant growth. Elevated ROS levels were also observed in response to As stress, however no ROS-mediated toxicities were experienced by the plants as the inoculated plants exhibited enhanced antioxidant levels. Thus, endophytes efficiently and effectively serve their host plants in overcoming various abiotic stresses there by maintaining a homeostasis in between plant growth and defense response.
6 Endophyte-Mediated Plant Defense Response
Even with the intrinsic and well-evolved defense mechanisms, plants at times require additional sophisticated defense strategies assisted by their microbial alliances to defend and rescue themselves from an array of biotic stresses. During the interaction between plant and endophytes, initially the infection of endophytes triggers the plant defense similar to of a pathogen infection, but subsequently, the endophytes escape these defense and colonize in the host plants [108] (Fig. 2). However, the defense responses induced by the colonization of endophytes in plants act as a priming effect and provide enhanced resistance against other phytopathogen. This phenomenon in plants conferred by the endophytes is known as induced systemic resistance (ISR) and commonly seen in bacteria-plant endophytic associations [108, 109]. In potato plants, inoculation of endophytes from genera Pseudomonas and Methylobacterium resulted in improved resistance against the necrotrophic pathogen Pectobacterium atrosepticum via ISR [110]. However, the extent of resistance conferred by colonization of Methylobacterium bacteria was inversely proportional to the amount of inoculum used. The induction of ISR and consecutive-enhanced pathogen defense in host plants have been reported in response to the colonization of Pseudomonas and Bacillus spp. bacteria [111]. Bacillus amyloliquefaciens, a bacterial endophyte isolated from corn plants, exhibited in vitro antifungal activities against multiple phytopathogens including Aspergillus flavus, Colletotrichum gloeosporioides, and Fusarium moniliforme. Further, the pretreatment of B. amyloliquefaciens to corn seedlings induced the expression of defense-related genes against pathogen infection as compared to the non-inoculated controls [112].
While bacterial endophytes are the masters of manipulating the plant defense and mimicking a priming defense effect against phytopathogen via ISR, the fungal endophytes commonly don’t exhibit ISR-mediated defense responses in their hosts [113, 114]. Conversely, the fungal endophytes produce growth-inhibiting chemical compounds against the invading pathogens and attacking herbivores to protect their host plants. These chemicals include phenols, terpenoids, flavonoids, alkaloids, quinols, steroids, polyketones, and peptides [71]. Clavicipitaceous fungi of several grass species produce alkaloids which provide defense against herbivory. For instance, indole diterpenoids, also known as lolitrems, are neurotoxic and produced in endophyte-infected grass, which are responsible for intoxication of cattle grazing on them [71]. Endophytic actinomycetes are extensively characterized for producing antimicrobial compound including munumbicins, kakadumycinx, and coronamycin [71]. In Kandelia candel, the endophytic Streptomyces sp. HKI0595 produces multicyclic indolosesquiterpene which has antibacterial activity [115]. In orchid plants, spoxazomicins A to C having antitrypanosomal activity were produced by the endophyte Streptosporangium oxazolinicum K07-0450T [116]. These compounds having different bioactive functions are exploited in clinical or agricultural purposes, whereas their exact roles in plant-microbe interactions are still under investigation [117]. Furthermore, the horizontal transfer of endophytes and their establishment results in enhanced biotic stress tolerance capacities by production of antibacterial, antiviral, antifungal, and insecticidal compounds [118]. However, enhancing the host immunity is not supported by all horizontally transmitted endophytes often due to the lack of opportunity for interaction with plant pathogens; hence, their role in host protection is yet to be characterized. For example, colonization of plant pathogens in the leaves of cacao plants doesn’t always result in disease, rather at times it acts as harmless or beneficial endophytes [2, 119]. Moreover, the endophytic-mediated production of antimicrobial compounds can be induced by the presence of a phytopathogen [120].
Apart from production of antimicrobial compounds in their hosts, endophytes code for several defense-related proteins and enzymes that further enhance the host defense responses. In rice, the endophytic bacteria Paenibacillus polymyxa confer defense against the rice false smut pathogen Ustilaginoidea oryzae by exhibiting antagonistic activity and β-1,3-1,4-glucanase production [121]. In wheat, bacterial endophyte colonizations support the host to overcome infection by Fusarium graminearum [122]. Similarly, endophyte colonization of varied bacterial genera in Zea mays, Arachis hypogaea, and Cucurbita pepo conferred resistance to fungal phytopathogens by producing compounds of antifungal nature [123,124,125]. In Fraxinus plants, the endophytes Pantoea agglomerans, Staphylococcus succinus, and Aerococcus viridans produce several antibiotics that help to protect the host from bacterial infections [126]. Recently, inhibition of infection in tomato plants by the fungal pathogen Phytophthora infestans was achieved by the inoculation of endophytic Phoma eupatorii isolate 8082. The inhibition of P. infestans was found to be conferred by the enhanced endophyte-mediated production of anthocyanins [127]. Cosme et al. [128] reported that tolerance to root herbivory in plants can be aided by root colonizing fungal endophytes by manipulating the JA and GA hormonal signaling. In addition to plant defense, endophyte-produced secondary metabolites are involved in mechanisms of genetic regulations, signaling, and the establishment of symbiosis and even influence host secondary metabolism [129, 130]. Inoculation of Methylobacterium spp. into strawberry plants resulted in the biosynthesis of flavor compounds, such as furanones [131]. Another report confirming the regulation of host metabolism by the endophytes reported the discovery of bacterial endophytes and gene transcripts in the vascular tissues of strawberry, the locations where the furanone biosynthesis is carried out in plants [132]. Likewise, inoculation of a fungal endophyte Paraphaeosphaeria sp. in the bilberry plant resulted in upregulated biosynthesis and significant accumulation of phenolic acids, flavan-3-ols, and oligomeric proanthocyanidins [133]. Overall, the colonization and establishment of endophytes in plants helps in boosting the host immunity by employing one or more of the above discussed strategies.
7 Conclusions
The sessile nature of the plants makes them to associate with diverse groups of symbiotic microbes exhibiting significant role in plant development and protection. Plants constitute an ecological niche within its inner space for the assemblage and sustenance of multiple microbial symbionts. It appears that a majority of these mutualistic microbes affect in the way plants network with their environment. Myriads of studies have shown that endophytes confer stress tolerance, survivality, and improved growth of the host plant species under extreme environmental conditions including salinity, extreme temperature, drought, and environmental toxicity [101, 134]. Additionally, recent reports also demonstrated that endophytes have good potential for application in plant improvement and disease control [135]. Therefore, it is collectively accepted that plants survive and flourish in the biotic and abiotically stressed environments for the reason that the endophytic symbionts have simultaneously evolved and essentially involved in their adaptation to stressed ecosystems [136].
Multiple endophytes penetrate and infect plants without any symptoms. Further, a few of them are inseparable from the plant system and vertically transmitted from one generation to another [137]. This suggests that endophytes provide a vast reservoir of heritable DNA providing genetic flexibility toward adaptation of the plants to changing environments. The usage of such endophytes in a mechanized agricultural system will be significant in the development of novel germplasms leading to rapid agricultural productions. Besides, the ability of certain endophytes to infect multiple hosts suggests that they could be transferred to native plants and agricultural crops to improve their capabilities in tolerating multiple types of environmental stresses brought about by ecological changes [101]. Although several intellectual property rights and patents have been granted on different aspects of endophytes [138], the commercial applications of endophytic microbes are still at its infancy. It is essential that a large majority of these endophytes are subjected to experimental trials and tests to evaluate their efficiency under natural conditions. Both the plant host and the endophytes have to deal with the natural environment for their survival. As such, a broader understanding and acknowledgment of the symbiotic interaction with the microbes is essential for optimum plant growth and development.
Abbreviations
- ABA:
-
Abscisic acid
- ACC:
-
1-Aminocyclopropane-1-carboxylate
- AHK2:
-
Arabidopsis histidine kinase 2
- APX:
-
Ascorbate-dependent peroxidases
- CDK:
-
Cyclin-dependent protein kinase
- CDPK:
-
Calcium-dependent protein kinase
- CRE1:
-
Cytokinin response 1
- DHAR:
-
Dehydroascorbate reductases
- GR:
-
Glutathione reductases
- HR:
-
Hypersensitive responses
- IAA:
-
Indole acetic acid
- IAA:
-
Indole-3-acetic acid
- ISR:
-
Induced systemic resistance
- JA:
-
Jasmonic acid
- MAPK:
-
Mitogen-activated protein kinase
- MDHAR:
-
Monodehydroascorbate reductases
- PAMP:
-
Pathogen-associated molecular patterns
- PRR:
-
Pattern recognizing receptors
- ROS:
-
Reactive oxygen species
- SA:
-
Salicylic acid
- SAKA:
-
Stress-activated mitotic kinase
- SAR:
-
Systemic acquired resistance
- SOD:
-
Superoxide dismutase
- VOC:
-
Volatile organic compounds
References
Kaul S, Sharma T, Dhar MK (2016) “Omics” tools for better understanding the plant–endophyte interactions. Front Plant Sci 7:9
Arnold AE (2007) Understanding the diversity of foliar endophytic fungi: progress, challenges, and frontiers. Fungal Biol Rev 21:51–66
Redecker D, Kodner R, Graham LE (2000) Glomalean fungi from the Ordovician. Science 289:1920–1921
Krings M, Taylor TN, Hass H, Kerp H, Dotzler N, Hermsen EJ (2007) Fungal endophytes in a 400-million-yr-old land plant: infection pathways, spatial distribution, and host responses. New Phytol 174:648–657
Higginbotham SJ, Arnold AE, Ibañez A, Spadafora C, Coley PD, Kursar TA (2013) Bioactivity of fungal endophytes as a function of endophyte taxonomy and the taxonomy and distribution of their host plants. PLoS One 8:73192
Rho H, Hsieh M, Kandel SL, Cantillo J, Doty SL, Kim SH (2018) Do endophytes promote growth of host plants under stress? A meta-analysis on plant stress mitigation by endophytes. Microb Ecol 75:407–418
Berg G (2009) Plant–microbe interactions promoting plant growth and health: perspectives for controlled use of microorganisms in agriculture. Appl Microbiol Biotechnol 84:11–18
Wiewiora B, Zurek G, Panka D (2015) Is the vertical transmission of Neotyphodium lolii in perennial ryegrass the only possible way to the spread of endophytes? PLoS One 10:0117231
Anand P, Isar J, Saran S, Saxena RK (2006) Bioaccumulation of copper by Trichoderma viride. Bioresour Technol 97:1018–1025
Aly AH, Debbab A, Kjer J, Proksch P (2010) Fungal endophytes from higher plants: a prolific source of phytochemicals and other bioactive natural products. Fungal Divers 4:1–16
Kaul S, Gupta S, Ahmed M, Dhar MK (2012) Endophytic fungi from medicinal plants: a treasure hunt for bioactive metabolites. Phytochem Rev 11:487–505
Redman RS, Sheehan KB, Stout RG, Rodriguez RJ, Henson JM (2002) Thermotolerance generated by plant/fungal symbiosis. Science 298:1581
Rozpądek P, Wężowicz K, Nosek M, Wazny R, Tokarz K, Lembicz M, Miszalski Z, Turnau K (2015) The fungal endophyte Epichloë typhina improves photosynthesis efficiency of its host orchard grass (Dactylis glomerata). Planta 242:1025–1035
Strobel GA (2002) Rainforest endophytes and bioactive products. Crit Rev Biotechnol 22:315–333
Newman LA, Reynolds CM (2005) Bacteria and phytoremediation: new uses for endophytic bacteria in plants. Trends Biotechnol 23:6–8
Deng Z, Cao L (2017) Fungal endophytes and their interactions with plants in phytoremediation: a review. Chemosphere 168:1100–1106
Zhi-Lin Y, Yi-Cun C, Bai-Ge X, Chu-Long Z (2012) Current perspectives on the volatile-producing fungal endophytes. Crit Rev Biotechnol 32:363–373
Reddy BN, Hindumathi A (2017) Potential of microbial volatile organic compounds for crop protection against phytopathogenic fungi. In: Choudhary D, Sharma A, Agarwal P, Varma A, Tuteja N (eds) Volatiles and food security. Springer, Singapore
Smith SE, Gianinazzi-Pearson V (1988) Physiological interactions between symbionts in vesicular-arbuscular mycorrhizal plants. Annu Rev Plant Physiol Plant Mol Biol 39:221–244
Kabera JN, Semana E, Mussa AR, He X (2014) Plant secondary metabolites: biosynthesis, classification, function and pharmacological properties. J Pharm Pharmacol 2:377–392
Hilleary R, Gilroy S (2018) Systemic signaling in response to wounding and pathogens. Curr Opin Plant Biol 43:57–62
Jones JD, Dangl JL (2006) The plant immune system. Nature 444:323
Bittner N, Trauer-Kizilelma U, Hilker M (2017) Early plant defence against insect attack: involvement of reactive oxygen species in plant responses to insect egg deposition. Planta 245:993–1007
Gulyani V, Kushwaha HR, Kumar P (2018) Role of phytohormones in plant defense: signalling and cross talk. In: Singh A, Singh IK (eds) Molecular aspects of plant-pathogen interaction. Springer Nature, Singapore, pp 159–184
Boudsocq M, Sheen J (2013) CDPKs in immune and stress signaling. Trends Plant Sci 18:30–40
Zhang L, Du L, Poovaiah BW (2014) Calcium signaling and biotic defense responses in plants. Plant Signal Behav 9:973818
Nanda S, Nayak S, Joshi RK (2014) Molecular cloning and expression analysis of four turmeric MAP kinase genes in response to abiotic stresses and phytohormones. Biol Plant 58:479–490
Zhu Y, Schluttenhoffer CM, Wang P, Fu F, Thimmapuram J, Zhu JK, Lee SY, Yun DJ, Mengiste T (2014) CYCLIN-DEPENDENT KINASE8 differentially regulates plant immunity to fungal pathogens through kinase-dependent and-independent functions in Arabidopsis. Plant Cell 26:4149–4170
Nanda S, Rout E, Joshi RK (2016) Curcuma longa mitogen-activated protein kinase 6 (ClMPK6) stimulates the defense response pathway and enhances the resistance to necrotrophic fungal infection. Plant Mol Biol Report 34:886–898
Bais HP, Weir TL, Perry LG, Gilroy S, Vivanco JM (2006) The role of root exudates in rhizosphere interactions with plants and other organisms. Annu Rev Plant Biol 57:233–266
Shiomi HF, Silva HSA, Melo ISD, Nunes FV, Bettiol W (2006) Bioprospecting endophytic bacteria for biological control of coffee leaf rust. Sci Agric 63:32–39
de Vries RP, Visser J (2001) Aspergillus enzymes involved in degradation of plant cell wall polysaccharides. Microbiol Mol Biol Rev 65:497–522
Tanaka A, Tapper BA, Popay A, Parker EJ, Scott B (2005) A symbiosis expressed non-ribosomal peptide synthetase from a mutualistic fungal endophyte of perennial ryegrass confers protection to the symbiotum from insect herbivory. Mol Microbiol 57:1036–1050
Tanaka A, Christensen MJ, Takemoto D, Park P, Scott B (2006) Reactive oxygen species play a role in regulating a fungus–perennial ryegrass mutualistic interaction. Plant Cell 18:1052–1066
Tanaka A, Takemoto D, Hyon GS, Park P, Scott B (2008) NoxA activation by the small GTPase RacA is required to maintain a mutualistic symbiotic association between Epichloë festucae and perennial ryegrass. Mol Microbiol 65:1165–1178
Brosi GB, McCulley RL, Bush LP, Nelson JA, Classen AT, Norby RJ (2011) Effects of multiple climate change factors on the tall fescue–fungal endophyte symbiosis: infection frequency and tissue chemistry. New Phytol 189:797–805
Dearnaley JD (2007) Further advances in orchid mycorrhizal research. Mycorrhiza 17:475–486
Puri SC, Nazir A, Chawla R, Arora R, Riyaz-ul-Hasan S, Amna T, Ahmed B, Verma V, Singh S, Sagar R, Sharma A (2006) The endophytic fungus Trametes hirsuta as a novel alternative source of podophyllotoxin and related aryl tetralinlignans. J Biotechnol 122:494–510
Wang Y, Dai CC (2011) Endophytes: a potential resource for biosynthesis, biotransformation, and biodegradation. Ann Microbiol 61:207–215
Eaton C, Cox M, Ambrose B, Becker M, Hesse U, Schardl C, Scott B (2010) Disruption of signaling in a fungal-grass symbiosis leads to pathogenesis. Plant Physiol 153(4):1780–1794
Eaton CJ, Jourdain I, Foster SJ, Hyams JS, Scott B (2008) Functional analysis of a fungal endophyte stress-activated MAP kinase. Curr Genet 53:163–174
Eaton CJ, Cox MP, Scott B (2011) What triggers grass endophytes to switch from mutualism to pathogenism? Plant Sci 180:190–195
Rodriguez RJ, Henson J, Van Volkenburgh E, Hoy M, Wright L, Beckwith F, Kim YO, Redman RS (2008) Stress tolerance in plants via habitat-adapted symbiosis. ISME J 2:404
Bartholdy BA, Berreck M, Haselwandter K (2001) Hydroxamate siderophore synthesis by Phialocephala fortinii, a typical dark septate fungal root endophyte. Biol Met 14:33–42
Chaves MM, Oliveira MM (2004) Mechanisms underlying plant resilience to water deficits: prospects for water-saving agriculture. J Exp Bot 55:2365–2384
Marasco R, Rolli E, Ettoumi B, Vigani G, Mapelli F, Borin S (2012) A drought resistance-promoting microbiome is selected by root system under desert farming. PLoS One 7:48479
Shahzad R, Waqas M, Khan AL, Al-Hosni K, Kang SM, Seo CW, Lee IJ (2018) Indole acetic acid production and plant growth promoting potential of bacterial endophytes isolated from rice (Oryza sativa L.) seeds. Acta Biol Hung 68:175–186
Jerry B (1994) A role of endophytic fungi in regulating nutrients and energy in plants within a desert ecosystem. In: International symposium and workshop on desertification in developed countries
Erb M, Meldau S, Howe GA (2012) Role of phytohormones in insect-specific plant reactions. Trends Plant Sci 17:250–259
Iqbal N, Khan NA, Ferrante A, Trivellini A, Francini A, Khan MIR (2017) Ethylene role in plant growth, development and senescence: interaction with other phytohormones. Front Plant Sci 8:475
Egamberdieva D, Wirth SJ, Alqarawi AA, Abd Allah EF, Hashem A (2017) Phytohormones and beneficial microbes: essential components for plants to balance stress and fitness. Front Microbiol 8:2104
Long HH, Schmidt DD, Baldwin IT (2008) Native bacterial endophytes promote host growth in a species-specific manner; phytohormone manipulations do not result in common growth responses. PLoS One 3:2702
Ahmad M, Kibret M (2013) Mechanism and applications of plant growth promoting rhizobacteria: current perspective. J King Saud Univ Sci 26:1–20
Suzuki S, He Y, Oyaizu H (2003) Indole-3-acetic acid production in Pseudomonas fluorescens HP72 and its association with suppression of creeping bent grass brown patch. Curr Microbiol 47:138–143
Xin G, Zhang G, Kang JW, Staley JT, Doty SL (2009) A diazotrophic, indole-3-acetic acid-producing endophyte from wild cottonwood. Biol Fertil Soils 45:669–674
Bal HB, Nayak L, Das S, Adhya TK (2013) Isolation of ACC deaminase PGPR from rice rhizosphere and evaluating their plant growth promoting activity under salt stress. Plant Soil 366:93–105
Waqas M, Khan AL, Shahzad R, Ullah I, Khan AR, Lee IJ (2015) Mutualistic fungal endophytes produce phytohormones and organic acids that promote japonica rice plant growth under prolonged heat stress. J Zhejiang Univ Sci B 16:1011–1018
Passari AK, Mishra VK, Leo VV, Gupta VK, Singh BP (2016) Phytohormone production endowed with antagonistic potential and plant growth promoting abilities of culturable endophytic bacteria isolated from Clerodendrum colebrookianum Walp. Microbiol Res 193:57–73
Vandenbussche F, Fierro AC, Wiedemann G, Reski R, Van Der Straeten D (2007) Evolutionary conservation of plant gibberellin signalling pathway components. BMC Plant Biol 7:65
Hamayun M, Khan SA, Khan AL (2010) Gibberellin production and plant growth promotion from pure cultures of Cladosporium sp. MH-6 isolated from cucumber (Cucumis sativus L.). Mycologia 102:989–995
Khan AL, Waqas M, Hussain J, Al-Harrasi A, Lee IJ (2014) Fungal endophyte Penicillium janthinellum LK5 can reduce cadmium toxicity in Solanum lycopersicum (Sitiens and Rhe). Biol Fertil Soils 50:75–85
Li W, Ma M, Feng Y, Li H, Ma Y, Li M, An F, Guo H (2015) EIN2-directed translational regulation of ethylene signaling in Arabidopsis. Cell 163:670–683
Arshad M, Saleem M, Hussain S (2007) Perspectives of bacterial ACC deaminase in phytoremediation. Trends Biotechnol 25:356–362
Forchetti G, Masciarelli O, Izaguirre MJ, Alemano S, Alvarez D, Abdala G (2010) Endophytic bacteria improve seedling growth of sunflower under water stress, produce salicylic acid, and inhibit growth of pathogenic fungi. Curr Microbiol 61:485–493
Vadassery J, Ritter C, Venus Y, Camehl I, Varma A, Shahollari B, Novák O, Strnad M, Ludwig-Müller J, Oelmüller R (2008) The role of auxins and cytokinins in the mutualistic interaction between Arabidopsis and Piriformospora indica. Mol Plant Microbe Interact 21:1371–1383
Pirttilä AM, Joensuu P, Pospiech H, Jalonen J, Hohtola A (2004) Bud endophytes of Scots pine produce adenine derivatives and other compounds that affect morphology and mitigate browning of callus cultures. Physiol Plant 121:305–312
Ryu CM (2004) Bacterial volatiles induce systemic resistance in Arabidopsis. Plant Physiol 134:1017–1026
Ryu CM, Hu CH, Locy RD, Kloepper JW (2005) Study of mechanisms for plant growth promotion elicited by rhizobacteria in Arabidopsis thaliana. Plant Soil 268:285–292
Doty SL (2011) Nitrogen-fixing endophytic bacteria for improved plant growth. In: Bacteria in agrobiology: plant growth responses. Springer, Berlin/Heidelberg, pp 183–199
Gallon JR (1992) Reconciling the incompatible – N2 fixation and O2. New Phytol 122:571–609
Hardoim PR, Van Overbeek LS, Berg G, Pirttilä AM, Compant S, Campisano A, Döring M, Sessitsch A (2015) The hidden world within plants: ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol Mol Biol Rev 79:293–320
Flores-Encarnación M, Contreras-Zentella M, Soto-Urzua L, Aguilar-Gutiérrez GR, Baca BE, Escamilla-Marván JE (1999) The respiratory system and diazotrophic activity of Acetobacter diazotrophicus Pal 5. J Bacteriol 181:6987–6995
Oliveira ALM, Urquiaga S, Döbereiner J, Baldani JI (2002) The effect of inoculating endophytic N2-fixing bacteria on micropropagated sugarcane plants. Plant Soil 242:205–215
Govindarajan M, Balandreau J, Muthukumarasamy R, Revathi G, Lakshminarasimhan C (2006) Improved yield of micropropagated sugarcane following inoculation by endophytic Burkholderia vietnamiensis. Plant Soil 280:239–252
Govindarajan M, Balandreau J, Kwon SW, Weon HY, Lakshminarasimhan C (2008) Effects of the inoculation of Burkholderia vietnamensis and related endophytic diazotrophic bacteria on grain yield of rice. Microb Ecol 55:21–37
Caballero-Mellado J, Martinez-Aguilar L, Paredes-Valdez G, Estrada-de los Santos P (2004) Burkholderia unamae sp. nov., an N2-fixing rhizospheric and endophytic species. Int J Syst Evol Microbiol 54:1165–1172
Montanez A, Abreu C, Gill PR, Hardarson G, Sicardi M (2009) Biological nitrogen fixation in maize (Zea mays L.) by 15N isotope dilution and identification of associated culturable diazotrophs. Biol Fertil Soils 45:253–263
Iniguez L, Dong Y, Triplett EW (2004) Nitrogen fixation in wheat provided by Klebsiella pneumoniae 342. Mol Plant Microbe Interact 17:1078–1085
Scherling C, Ulrich K, Ewald D, Weckwerth W (2009) A metabolic signature of the beneficial interaction of the endophyte Paenibacillus sp. isolate and in vitro-grown poplar plants revealed by metabolomics. Mol Plant Microbe Interact 22(8):1032–1037. https://doi.org/10.1094/MPMI-22-8-1032
Doty SL, Oakley B, Xin G, Kang JW, Singleton G, Khan Z, Vajzovic A, Staley JT (2009) Diazotrophic endophytes of native black cottonwood and willow. Symbiosis 47:23–33
Johnson LJ, Koulman A, Christensen M, Lane GA, Fraser K, Forester N, Johnson RD, Bryan GT, Rasmussen S (2013) An extracellular siderophore is required to maintain the mutualistic interaction of Epichloë festucae with Lolium perenne. PLoS Pathog 9:1003332
Mitter B, Petric A, Shin MW, Chain PS, Hauberg-Lotte L, Reinhold-Hurek B, Nowak J, Sessitsch A (2013) Comparative genome analysis of Burkholderia phytofirmans PsJN reveals a wide spectrum of endophytic lifestyles based on interaction strategies with host plants. Front Plant Sci 4:120
Sturz AV, Nowak J (2000) Endophytic communities of rhizobacteria and the strategies required to create yield enhancing associations with crops. Appl Soil Ecol 15:183–190
Choudhury DK (2012) Microbial rescue to plant under habitat-imposed abiotic and biotic stresses. Appl Microbiol Biotechnol 96:1137–1155
Lata R, Chowdhury S, Gond SK, White JF (2018) Induction of abiotic stress tolerance in plants by endophytic microbes. Lett Appl Microbiol 66:268–276
Waqas M, Khan AL, Kamran M, Hamayun M, Kang SM, Kim YH, Lee IJ (2012) Endophytic fungi produce gibberellins and indoleacetic acid and promotes host-plant growth during stress. Molecules 17:10754–10773
Naya L, Ladrera R, Ramos J (2007) The response of carbon metabolism and antioxidant defenses of alfalfa nodules to drought stress and to the subsequent recovery of plants. Plant Physiol 144:1104–1114
Bacon CW (1993) Abiotic stress tolerances (moisture, nutrients) and photosynthesis in endophyte-infected tall fescue. Agric Ecosyst Environ 44:123–141
Rodriguez RJ, White JF Jr, Arnold AE, Redman ARA (2009) Fungal endophytes: diversity and functional roles. New Phytol 182:314–330
Redman RS, Kim YO, Woodward CJ, Greer C, Espino L, Doty SL, Rodriguez RJ (2011) Increased fitness of rice plants to abiotic stress via habitat adapted symbiosis: a strategy for mitigating impacts of climate change. PLoS One 6:14823
Vahid J, Mohamad RB, Islam M, Farrokh D (2015) The effect of endophytic fungi in drought resistance of Lolium perenne in Iran (Isfahan) condition. Adv Stud Biol 7:245–257
Vigani G, Rolli E, Marasco R (2018) Root bacterial endophytes confer drought resistance and enhance expression and activity of a vacuolar H+ -pumping pyrophosphatase in pepper plants. Environ Microbiol. https://doi.org/10.1111/1462-2920.14272
Verma A, Verma S, Sahay N, Butehorn B, Franken P (1998) Piriformospora indica, a cultivable plant-growth-promoting root endophyte. Appl Environ Microbiol 65:2741–2744
Waller F, Achatz B, Baltruschat H, Fodor J, Becker K, Fischer M, Heier T, Hückelhoven R, Neumann C, von Wettstein D, Franken P (2005) The endophytic fungus Piriformospora indica reprograms barley to salt-stress tolerance, disease resistance, and higher yield. Proc Natl Acad Sci USA 102:13386–13391
Baltruschat H, Fodor J, Harrach BD, Niemczyk E, Barna B, Gullner G, Janeczko A, Kogel KH, Schäfer P, Schwarczinger I, Zuccaro A (2008) Salt tolerance of barley induced by the root endophyte Piriformospora indica is associated with a strong increase in antioxidants. New Phytol 180:501–510
Azad K, Kaminskyj S (2016) A fungal endophyte strategy for mitigating the effect of salt and drought stress on plant growth. Symbiosis 68:73–78
Lubna SA, Muhammad H, Humaira G, Lee IJ, Anwar H (2018) Aspergillus niger CSR3 regulates plant endogenous hormones and secondary metabolites by producing gibberellins and indoleacetic acid. J Plant Interact 13(1):100–111. https://doi.org/10.1080/17429145.2018.1436199
Sekmen HA, Türkan I, Takio S (2007) Differential responses of antioxidative enzymes and lipid peroxidation to salt stress in salt-tolerant Plantago maritima and salt-sensitive Plantago media. Physiol Plant 131:399–411
Rouhier N, San Koh C, Gelhaye E, Corbier C, Favier F, Didierjean C, Jacquot JP (2008) Redox based anti-oxidant systems in plants: biochemical and structural analyses. Biochim Biophys Acta 1780:1249–1260
Noctor G, Reichheld JP, Foyer CH (2017) ROS-related redox regulation and signaling in plants. Semin Cell Dev Biol 80:3–12
Singh LP, Gill SS, Tuteja N (2011) Unraveling the role of fungal symbionts in plant abiotic stress tolerance. Plant Signal Behav 6:175–191
Hubbard M, Germida JJ, Vujanovic V (2014) Fungal endophytes enhance wheat heat and drought tolerance in terms of grain yield and second-generation seed viability. J Appl Microbiol 116:109–122
Choo J, Sabri NBM, Tan D, Mujahid A, Müller M (2015) Heavy metal resistant endophytic fungi isolated from Nypa fruticans in Kuching Wetland National Park. Ocean Sci J 50: 445–453
Wang JL, Li T, Liu GY, Smith JM, Zhao ZW (2016) Unraveling the role of dark septate endophyte (DSE) colonizing maize (Zea mays) under cadmium stress: physiological, cytological and genic aspects. Sci Rep 6:22028
Khan A, Lee IJ (2013) Endophytic Penicillium funiculosum LHL06 secretes gibberellin that reprograms Glycine max L. growth during copper stress. BMC Plant Biol 13:86
Friesen ML, Porter SS, Stark SC, von Wettberg EJ, Sachs JL, Martinez-Romero E (2011) Microbially mediated plant functional traits. Annu Rev Ecol Evol Syst 42(1):23–46
Mukherjee G, Saha C, Naskar N, Mukherjee A, Mukherjee A, Lahiri S, Majumdar AL, Seal A (2018) An endophyteic bacterial consortium modulates multiple strategies to improve arsenic phytoremediation efficacy in Solanum nigrum. Sci Rep 8:6979
Zamioudis C, Pieterse CMJ (2012) Modulation of host immunity by beneficial microbes. Mol Plant Microbe Interact 25:139–150
Robert-Seilaniantz A, Grant M, Jones JDG (2011) Hormone crosstalk in plant disease and defense: more than just jasmonate-salicylate antagonism. Annu Rev Phytopathol 49:317–343
Ardanov P, Sessitsch A, Haggman H, Kozyrovska N, Pirttila AM (2012) Methylobacterium-induced endophyte community changes correspond with protection of plants against pathogen attack. PLoS One 7(10):e46802. https://doi.org/10.1371/journal.pone.0046802
Chanway C (1998) Bacterial endophytes: ecological and practical implications. In: 7th international congress on plant pathology, Edinburgh, 9–16 Aug 1998
Gond SK, Bergen MS, Torres MS, White JF, Kharwar RN (2015) Effect of bacterial endophyte on expression of defense genes in Indian popcorn against Fusarium moniliforme. Symbiosis 66:133–140
Blodgett JT, Eyles A, Bonello P (2007) Organ-dependent induction of systemic resistance and systemic susceptibility in Pinus nigra inoculated with Sphaeropsis sapinea and Diplodia scrobiculata. Tree Physiol 27:511–517
Bae H, Roberts DP, Lim HS, Strem MD, Park SC, Ryu CM, Melnick RL, Bailey BA (2011) Endophytic Trichoderma isolates from tropical environments delay disease onset and induce resistance against Phytophthora capsici in hot pepper using multiple mechanisms. Mol Plant Microbe Interact 24:336–351
Ding L, Maier A, Fiebig HH, Lin WH, Peschel G, Hertweck C (2011) Kandenols A-E, Eudesmenes from an endophytic Streptomyces sp. of the mangrove tree Kandelia candel. J Nat Prod 75:2223–2227
Inahashi Y, Matsumoto A, Omura S, Takahashi Y (2011) Streptosporangium oxazolinicum sp. nov., a novel endophytic ectinomycete producing new antitrypanosomal antibiotics, spoxazomicins. J Antibiot 64:297–302
Brader G, Compant S, Mitter B, Trognitz F, Sessitsch A (2014) Metabolic potential of endophytic bacteria. Curr Opin Biotechnol 27:30–37
Tejesvi MV, Kajula M, Mattila S, Pirttilä AM (2011) Bioactivity and genetic diversity of endophytic fungi in Rhododendron tomentosum H. Fungal Divers 47:97–107
Arnold AE, Mejía LC, Kyllo D, Rojas EI, Maynard Z, Robbins N, Herre EA (2003) Fungal endophytes limit pathogen damage in a tropical tree. Proc Natl Acad Sci USA 100: 15649–15654
Combès A, Ndoye I, Bance C, Bruzaud J, Djediat C, Dupont J, Nay B, Prado S (2012) Chemical communication between the endophyte fungi Paraconiothyrium variabile and the Phytopathogen Fusarium oxysporum. PLoS One 7:47313
Grady EN, MacDonald J, Liu L, Richman A, Yuan ZC (2016) Current knowledge and perspectives of Paenibacillus: a review. Microb Cell Fact 15:203
Díaz Herrera S, Grossi C, Zawoznik M, Groppa MD (2016) Wheat seeds harbour bacterial endophytes with potential as plant growth promoters and biocontrol agents of Fusarium graminearum. Microbiol Res 186–187:37–43
Rijavec T, Lapanje A, Dermastia M, Rupnik M (2007) Isolation of bacterial endophytes from germinated maize kernels. Can J Microbiol 53:802–808
Sobolev VS, Orner VA, Arias RS (2013) Distribution of bacterial endophytes in peanut seeds obtained from axenic and control plant material under field conditions. Plant Soil 371:367–376
Fürnkranz M, Lukesch B, Muller H, Huss H, Grube M, Berg G (2012) Microbial diversity inside pumpkins: microhabitat specific communities display a high antagonistic potential against phytopathogens. Microb Ecol 63:418–428
Donnarumma F, Capuana M, Vettori C, Petrini G, Giannini R, Indorato C, Mastromei G (2011) Isolation and characterisation of bacterial colonies from seeds and in vitro cultures of Fraxinus spp. from Italian sites. Plant Biol 13:169–176
de Vries S, von Dahlen JK, Schnake A, Ginschel S, Schulz B, Rose LE (2018) Broad-spectrum inhibition of Phytophthora infestans fungal endophytes. FEMS Microbiol Ecol 94:1–15
Cosme M, Lu J, Erb M, Stout MJ, Franken P, Wurst S (2016) A fungal endophyte helps plants to tolerate root herbivory through changes in gibberellin and jasmonate signaling. New Phytol 211:1065–1076
Schulz B, Boyle C (2005) The endophytic continuum. Mycol Res 109:661–686
Zhang HW, Song YC, Tan RX (2006) Biology and chemistry of endophytes. Nat Prod Rep 23:753–771
Verginer M, Siegmund B, Cardinale M, Muller H, Choi Y, Miguez CB, Leitner E, Berg G (2010) Monitoring the plant epiphyte Methylobacterium extorquens DSM 21961 by real time PCR and its influence on the strawberry flavour. FEMS Microbiol Ecol 74:136–145
Nasopoulou C, Pohjanen J, Koskimaki JJ, Zabetakis I, Pirttila AM (2014) Localization of strawberry (Fragaria x ananassa) and Methylobacterium extorquens genes of strawberry flavour biosynthesis in strawberry tissue by in situ hybridization. J Plant Physiol 171: 1099–1105
Koskimaki JJ, Hokkanen J, Jaakola L, Suorsa M, Tolonen A, Mattila S, Pirttila AM (2009) Flavonoid biosynthesis and degradation play a role in early defence responses of bilberry (Vaccinium myrtillus) against biotic stress. Eur J Plant Pathol 125:629. https://doi.org/10.1007/s10658-009-9511-6
Meena KK, Sorty AM, Bitla UM, Choudhary K, Gupta P, Pareek A, Singh DP, Prabha R, Sahu PK, Gupta VK, Singh HB, Krishanani KK, Minhas PS (2017) Abiotic stress responses and microbe-mediated mitigation in plants: the omics strategies. Front Plant Sci 8:172
Muthukumar A, Venkatesh A (2017) Exploitation of fungal and endophytic bacteria for the management of leaf blight of ribbon plant. J Plant Pathol Microbiol 4:209
Barrow JR, Lucero ME, Reyes-Vera I, Havstad KM (2008) Do symbiotic microbes have a role in plant evolution, performance and response to stress? Commun Integr Biol 1:69–73
Rodriguez RJ, Redman RS, Henson JM (2004) The role of fungal symbioses in the adaptation of plants to high stress environments. Mitig Adapt Strateg Glob Chang 9:261. https://doi.org/10.1023/B:MITI.0000029922.31110.97
Gokhale M, Gupta D, Gupta U, Faraz R, Sandhu SS (2017) Patents on endophytic fungi. Recent Pat Biotechnol 11(2):120–140
Acknowledgments
SN is thankful for the award of a Postdoctoral Fellowship (PDF) from the Chinese Academy of Agricultural Sciences, Peoples Republic of China. BM is thankful for the award of institutional Doctoral Fellowship from Siksha O Anusandhan University, Bhubaneswar, India. The authors are thankful to the Head of the Center of Biotechnology, Siksha O Anusandhan University, and the Head of the PG Dept. of Biotechnology, Rama Devi Women’s University, for their guidance and support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this entry
Cite this entry
Nanda, S., Mohanty, B., Joshi, R.K. (2019). Endophyte-Mediated Host Stress Tolerance as a Means for Crop Improvement. In: Jha, S. (eds) Endophytes and Secondary Metabolites. Reference Series in Phytochemistry. Springer, Cham. https://doi.org/10.1007/978-3-319-90484-9_28
Download citation
DOI: https://doi.org/10.1007/978-3-319-90484-9_28
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-90483-2
Online ISBN: 978-3-319-90484-9
eBook Packages: Chemistry and Materials ScienceReference Module Physical and Materials ScienceReference Module Chemistry, Materials and Physics