Abstract
To understand the role of a geographical region in the in situ conservation of the genetic diversity of any crop, it is necessary to analyze the current conservation status of the crop and any genetic changes that have occurred within the last few decades in the region. Lima bean (Phaseolus lunatus L.) is an important crop in the Mayan agriculture of the Yucatan Peninsula, Mexico, its Mesoamerican center of diversity. In this region, 3 of the 21 landraces dominate 71.24 % of the cultivated area, and 12 are rare landraces grown in only 6.29 % of the area. This chapter analyzes the risk of the genetic erosion in Lima bean landraces from the Yucatan Peninsula using molecular markers, with the objective of generating data to develop comprehensive in situ conservation programs for the crop. Molecular analyses showed that the many landraces that are planted by only a few peasants contained higher levels of genetic diversity compared with the three most abundant landraces. Also, they showed that the landraces planted in 1979 have higher levels of genetic diversity than those planted in 2007 and that, over the last 30 years, the genetic make-up of this crop has shifted. If current trends in the cultivation of the Lima bean landraces continue, many will no longer be planted within two to three generations, contributing to further genetic erosion. The establishment of evidence-based programs for the in situ conservation of Lima bean landraces is urgently needed in this center of genetic diversity.
Note: Copyright of Tables and Figs. (8.3, 8.4, 8.5, 8.6) used in this Chapter belong to Springer Science + Business Media Dordrecht.
Original papers were published by Genetic Resources and Crop Evolution journal: November 2008, Volume 55, Issue 7, pp 1065–1077.
DOI:10.1007/s10722-008-9314-1 http://springerlink.bibliotecabuap.elogim.com/article/10.1007/s10722-008-9314-1).February 2012, Volume 59, Issue 2, pp 191–204.
DOI:10.1007/s10722-011-9675-8 http://springerlink.bibliotecabuap.elogim.com/article/10.1007/s10722-011-9675-8).
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- ISSR markers
- Landraces
- Loss of genetic diversity
- SSR markers
- Traditional mayan agriculture
- Yucatan peninsula
8.1 Introduction
Mexico forms part of the Mesoamerican center of domestication (Vavilov 1926). The ecological, productive, and cultural conditions of the traditional agroecosystems in Mexico have helped to conserve a large number of domesticated species. These conditions have also maintained these species as part of a dynamic scenario for the development of new crops and the evolution of species, processes that favor high levels of variation and genetic contact with wild relatives (Hernández-Xolocotzi 1973). Genetic erosion, the loss, or reduction of genetic diversity between and within populations of the same species over time (Jarvis et al. 2000), is a significant issue affecting diversity in crop domestication areas as Mexico: (1) concentrate the highest genetic diversity; (2) traditional growers conserve ancestral landraces (i.e., the local populations of cultivated species generated by traditional farmers), along with the knowledge and cultural practices that created this diversity; and (3) the presence of wild–crop introgression (Bellon and Taylor 1993; Brush 1991). Most often resulting from agricultural, economic, and social changes (FAO 1996), genetic erosion in domesticated species has been evaluated at the level of landrace (Hammer and Laghetti 2005; Tsegaye and Berg 2006) because it constitutes the primary available genetic pool for hybridization and genetic improvement programs (Harlan and De Wet 1971). Several decades ago, Frankel and Bennett (1970) noted the importance of genetic erosion within traditional agricultural systems on the word. They stated that many genetic reservoirs for crop plants were rapidly disappearing, and the detailed five principles: (1) diversity in crops exists because of adaptation by localized populations; (2) traditional agriculture that continues in centers of diversity maintains high, stable diversity; (3) modern agricultural technology, including modern varieties, is a recent phenomenon and leads to instability; (4) competition between local and introduced varieties results in displacement of local varieties; and (5) displacement of local varieties reduces the genetic variability of the entire crop gene pool.
In Mexico, the milpa is the most important traditional agricultural system. It is an ancestral Mesoamerican dry land farming system based on human energy by which vegetation is cyclically slashed and burned to plant a group of basic crops. In the milpa, after 2–4 years of cultivation (depending on soil fertility), the land is allowed to rest for 5–15 years before a new cycle is begun (Hernández-Xolocotzi 1992; Pérez-Toro 1945). The conservation of patches of vegetation that are cyclically cultivated is, in turn, the mainstay of the milpa’s productivity, as it assures the recovery of soil fertility and maintains the habitat for a large part of the plant genetic resources integrated into the milpa agroforestry production system (Colunga-GarcíaMarín and May-Pat 1993; Hernández-Xolocotzi 1992). The three principal crops of the milpa are corn (Zea mays L.), beans (P. vulgaris L., P. coccineus L., P. polyanthus Greenman, P. lunatus L.), and squash [Cucurbita moschata (Duch) Duch ex Poir; C. argyrosperma Huber]. Alongside these basic crops, many other secondary species are cultivated, such as chilli peppers (Capsicum spp.), batata (Ipomoea batatas L.; sweet potato), tomato (Solanum esculentum L.), and cassava (Manihot sculenta Crantz). Within any of these crops, a great intraspecific diversity is reflected in the existence of a large number of landraces. It is the case of the Lima bean (P. lunatus).
8.2 Lima Bean (Phaseolus Lunatus L., Fabaceae)
Lima bean (Phaseolus lunatus L.) is one of five domesticated species of the genus Phaseolus that has evolved in the neotropics for at least 6000 years (Kaplan and Lynch 1999). It is, after common bean (P. vulgaris), the second most important commercial species of Phaseolus beans around the world (Baudoin et al. 2004). Its primary genetic pool has wild (P. lunatus var. silvester) and domesticated (P. lunatus var. lunatus) forms (Baudet 1977). Recent studies using cpDNA and ITS polymorphisms (Motta-Aldana et al. 2010; Serrano-Serrano et al. 2010, 2012; Andueza-Noh et al. 2013) have indicated that the organization of the genetic diversity of P. lunatus comprised three major gene pools: Andean (A), Mesoamerican I (MI), and Mesoamerican II (MII), all containing both wild and domesticated populations. Three cultigroups (cv-gr) are recognized in the domesticated forms (Baudet 1977): (1) Potato, with small, round seeds; (2) Sieva, with medium-sized, kidney-shaped seeds; and (3) Big Lima, with large, flat seeds. The Potato and Sieva cultigroups represent the MI and MII groups, and the Big Lima represents the Andean. The domestication area of the Andean gene pool has been located between Ecuador and northern Peru (Motta-Aldana et al. 2010; Serrano-Serrano et al. 2012). Evidence generated recently indicates a domestication event for MI in western Mexico (Motta-Aldana et al. 2010; Serrano-Serrano et al. 2012; Andueza-Noh et al. 2013). Although domestication of MII has yet to be defined, recent evidence suggests that, if present, it will be located in the region between Guatemala and Costa Rica (Andueza-Noh et al. 2013).
8.2.1 Lima Bean in the Yucatan Peninsula, Mexico
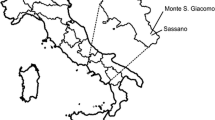
The Lima bean landraces, called Ibes in Mayan, represent the fourth main crop for the Maya of the Yucatan Peninsula, the region with the highest morphological variation of landraces in all Mexico (Ballesteros 1999). This crop is planted into the milpa (Fig. 8.1a), principally and, less frequently, in home gardens (Fig. 8.1b). At present, there are four geographic areas in the Yucatan Peninsula where the milpa continues to be the most important economic activity (Fig. 8.2). These areas correspond to four of the 13 cultural-geographic zones established by Adams and Culbert (1977) for the origin of the Maya lowland civilization: (1) northeastern Campeche (NECAMP), in “Los Chenes” zone, (2) southern Yucatan (SYUC), in the “Puuc” zone, (3) southeastern Yucatan (SEYUC), located within the “Northern Plains” zone, and (4) central eastern Quintana Roo (CEQROO), within the “Río Bec” zone (Fig. 8.2).
Four agricultural regions where the Mayan milpa is an important traditional agricultural system. SYUC southern Yucatan, NECAMP northeastern Campeche, SEYUC southeastern Yucatan, CEQROO central eastern Quintana Roo. Also, this Figure shows the three wild-weedy-domesticated complexes (X-Bilincok, Xocen and X-Hazil) of Lima bean studied by Dzul-Tejero (2011)
Using morphophenological and ethnobotanical data, Martínez-Castillo et al. (2004) found 25 landraces of Lima bean with Potato, Sieva and intermediate forms of both cultigroups and characterized their relative abundance based on the percentage of cultivated area and the number of farmers that plant each landrace. Using a sample of 160 traditional Mayan farmers from the four agricultural zones before mentioned, these authors found that, of 25 landraces planted, three accounted for 71.24 % of the cultivated area. Most of the remaining 22 landraces were rare, meaning each accounted for less than 2 % of the cultivated area; in many cases, they were grown by a single farmer. This situation indicates that the crop is at serious risk for genetic erosion, which is increased by three factors: (1) environmental factors such as drought and hurricanes, which have led to loss of seed; (2) intensification of the traditional Mayan agriculture, which displaces cultivation of these landraces; and (3) increasing rural population and socioeconomic changes have led to migration of Mayan growers to tourist centers, with consequent abandonment of agricultural activity and changes in the traditional Mayan diet (Cuanalo and Arias 1997; Ku-Naal 1995; Reyes and Aguilar 1992).
8.3 Genetic Erosion in Lima Bean from the Yucatan Peninsula: Evidence from ISSR Molecular Markers
Since Zietkiewicz et al. (1994) invented the Inter-Simple Sequence Repeats (ISSR) technique, it has proven to be a rapid, simple and inexpensive way to assess genetic structure and diversity (Culley et al. 2007; González et al. 2005), to analyze genetic relationships among cultivars (Prevost and Wilkinson 1999; Martins et al. 2003), and to study evolutionary processes (Galván et al. 2003). The ISSR technique allows the detection of polymorphism without previous knowledge of DNA sequences. It amplifies DNA using the polymerase chain reaction (PCR) and a single primer composed of a microsatellite (SSR) sequence, anchored at the 3′ or 5′ end by two to four arbitrary, often degenerate, nucleotides (Zietkiewicz et al. 1994). The ISSR is a dominant marker so that the heterozygote cannot be directly distinguished from the dominant homozygote phenotype (band) at individual loci and consequently, the estimation of allele frequencies using ISSR markers presents some statistical difficulties (Lynch and Milligan 1994). These difficulties have been resolved using appropriate estimators for the analysis of dominant markers such as the Shannon diversity index (I) (Shannon and Weaver 1949), percentage of polymorphic loci (%P), Nei’s genetic diversity index (h) considering the Taylor expansion (Lynch and Milligan 1994) and with new methods as average heterozygosity (H) using the Bayesian approach proposed by Zhivotovsky (1999).
On the basis of the plant material and data collected by Martínez-Castillo et al. (2004, 2008) used 90 ISSR loci (Table 8.1) to analyze the genetic diversity of 21 Lima bean landraces of differing relative abundances: (a) three abundant landraces, each grown on more than 16 % of the total cultivated area and planted by 10–33 producers in four agricultural zones; (b) six common landraces, each grown on 3–5 % of the cultivated area and by 5–14 producers; and (c) 12 rare landraces, each planted on less than 2 % of the total area and grown by 1–4 farmers (Fig. 8.3, Table 8.2). These authors found that the Yucatan Peninsula has high levels of hgenetic diversity (h = 0.28) in comparison with others studies. Using alloenzymes, Maquet et al. (1997) reported an = 0.26 for the P. lunatus base collection of the Germplasm Bank of the International Center for Tropical Agriculture (CIAT-Colombia) and they stated that this is a significant level and higher than reported for other plants that, like P. lunatus, are mixed-mating or short-lived perennial species (h = 0.12) (Hamrick et al. 1991). Using RAPD markers, Nienhuis et al. (1995) found a lower genetic diversity for domesticated Mesoamerican forms (h = 0.11). Also, using AFLP molecular markers, Castiñeiras et al. (2007) found a lower genetic diversity in landraces planted in Cuban home gardens (h = 0.119). Compared with all these studies, results from Martínez-Castillo et al. (2008) could be reflecting the high genetic diversity maintained by Mayan farmers in the milpa of the Yucatan Peninsula, México (Table 8.3).
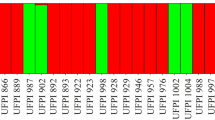
Groups of Lima bean landraces analyzed. Line A (abundant landraces): Mulición, Sac, Putsica-sutsuy; line B (common landraces): Bacalar, Nuk, Chak-saac, Mejen, Chak-petch, Balche; line C (rare landraces): Box-petch, Balam-pach, Tsisibal, Kan, Chak-mejen, Madzakitam; line D (rare landraces): Pool-santo, Tabaco, Box-uolis, Chak-uolis, Chak-chí, Chocolate. Landraces are named from left to right. Culti-groups: P (cv-gr Potato), S (cv-gr Sieva), I (intermediate forms between Potato and Sieva) (Reproduced from Martínez-Castillo et al. 2008)
Within the domesticated gene pool from the Yucatan Peninsula, Martínez-Castillo et al. (2008) found that the common group of landraces had the highest genetic diversity (except for %P), although the differences between the three groups (abundant, common, and rare landraces groups) considered were not statistically significant (Table 8.3). The rare landraces group had genetic diversity values (h and I) slightly lower than the common landraces group, but higher for %P (Table 8.3), probably because nine of the 12 rare landraces were represented by only one accession (Table 8.2), whereas all the common landraces were represented by at least five accessions. The minimal abundance of the rare landraces is the main factor that most increases their risk for genetic erosion since it can lead to their local extinction. During a travel made in 2007 to collect germplasm of Lima bean landraces, a farmer from southeastern of the Yucatan reported that he had lost his seed of Pool-santo and Chak-chí landraces in the 2006 agricultural cycle due to a lack of rain. In another case, a farmer from Central-east of Quintana Roo stopped planting the Chocolate and Tabaco landraces in 2005 because he became sick that year and did not cultivate his milpa. This farmer was the only one who had these two rare landraces and these have not been collected again until now. Two factors that could reduce the risk of genetic erosion in some of the rare landraces are dark seed color and their mixed management by Mayan farmers. Both aspects favor the entrance of wild alleles through formation of wild–weedy–domesticated complexes and the generation of weedy forms (Martínez-Castillo et al. 2004). A special case in the use of seed mixtures is the Bacalar landrace, which has become a kind of “genetic dump” as it contain seeds similar to many different landraces, such as Mejen, Nuk, and Pool-santo, including weedy forms (Fig. 8.4).
Weedy forms found into a Bacalar seed lot from central-east Quintana Roo (Reproduced from Martínez-Castillo et al. 2008)
Martínez-Castillo et al. (2008) showed that the abundant landraces group had the lowest values of genetic diversity among the three groups for all estimators, except for H that equaled the value for the rare landrace group (Table 8.3). These low values could reflect a germplasm selection influenced by external market demands. Martínez-Castillo et al. (2004) reported that one of the main selection criteria for the three most abundant landraces (Mulición, Sac, and Putsica-sutsuy) is production of seed for sale; thus, Mayan farmers currently tend to plant white-seeded landraces (Mulición, Sac, Mejen, Nuk). This tendency leads to selection against weedy forms that are produced from crosses between landraces and the wild populations surrounding the milpas, consequently limiting introgression of wild alleles and increasing the risk of genetic erosion. In relation to the dominant Lima bean landraces, Debouck (1979) collected at least 10 different landraces in northeastern Campeche in 1979, but currently only three have been observed, and these are dominated by Mulición and Sac. Informal interviews with Mayan growers suggest that this loss of landraces is associated with the introduction of mechanized agriculture and monoculture of improved varieties of corn. Recent field observations indicate that even the planting of abundant Lima bean landraces such as Mulición and Sac is decreasing in response to low prices. A similar case is happening in southern Yucatan, where the Mejen landrace has been replacing the other landraces with seeds that are not white (Martínez-Castillo et al. 2004). Recently, the area sown with Mejen has decreased because of low market demand. Even though in the 2004 study, Mejen was considered as a landrace, evidence suggests that it could be an improved variety introduced approximately 25 years ago: (1) it was not found by Debouck in 1979, (2) it is a variety planted as a monocrop (a rarity in traditional Mayan agriculture) and is not associated with maize as are all the other landraces, and (3) it is a variety with a very short production cycle that depends on a lot of water, a limited resource in the Yucatan Peninsula. This decrease in the number and density of planted populations may mean that a new genetic bottleneck is soon to come for the abundant landraces.
8.4 Genetic Erosion in Lima Bean from the Yucatan Peninsula: Temporal Analysis Using SSR Molecular Markers
The microsatellite (SSR-Simple Sequence Repeat) marker, a codominant marker that is highly polymorphic and discriminating and distributed throughout the genome (Tautz and Renz 1984), have proven useful in studies of genetic structure and diversity in cultivated plants (Martínez-Castillo et al. 2006, 2007; Zhou et al. 2006). Using nine SSR polymorphic loci (Table 8.4), Martínez-Castillo et al. (2011) temporally analyzed genetic erosion in Lima bean landraces from northeastern Campeche (NECAMP) (Fig. 8.2). These authors analyzed material collected during two different years: (1) seeds of 23 accessions that were collected in 1979 by Dr. Debouck in Nohalal town (in northeastern Campeche) were obtained from the Germplasm Bank of the Centro Internacional de Agricultura Tropical (CIAT-Colombia) (Fig. 8.5a), and (2) seeds of 21 accessions were collected directly in 2007 by Dr. Martínez-Castillo in Nohalal and three adjacent Mayan towns: Chunyaxnic, X-Bilincock, and Yaax-haltun (Fig. 8.5b).
a Accessions of Lima bean analyzed from Nohalal-1979. b Accessions of Lima bean analyzed from 2007. In Fig. 8.5b, letter after the accession represent the town where that accession was collected: a Yaax-haltun, b Chunyaxnic, c X-Bilincok, d Nohalal (Reproduced from Martínez-Castillo et al. 2011)
First, Martínez-Castillo et al. (2011) analyzed genetic erosion in the same Mayan town (Nohalal) for the two collected years (1979 vs 2007). The authors found that all the genetic diversity estimators gave higher values for 1979 (Table 8.5). These results can be a consequence of the number and kind of landraces collected during each year. In 1979, Dr. Debouck collected 10 landraces at least, whereas in 2007 only three landraces were collected, and many of the accessions were seeds with white testa (Fig. 8.5b). At present, white seed landraces (Sac, Mulición and Mejen) dominate Lima bean production in the Yucatan Peninsula (Martínez-Castillo et al. 2004). Secondly, the authors analyzed the genetic erosion between the accessions collected in 1979 in Nohalal compared with all accessions collected in 2007 in Nohalal and the three neighboring towns. Values of %P and the number of alleles per locus (N a) between the two collected years did not differ, meaning that the allelic richness is similar in both years. However, the number of effective alleles (N e) and h, whose values were higher in the accessions collected in 1979 (Table 8.5), did differ for the two groups, indicating that the genetic diversity was greater in 1979. Importantly, including accessions collected in the three other Mayan towns generated a decrease in the differences observed between the genetic diversity estimators, in relation to the previous analysis (Nohalal-1979 vs Nohalal-2007) (Table 8.5). This decrease could be due to the fact that the germplasm collected in 2007 in the three towns contained accessions of other landraces that were not found in Nohalal in 2007. These landraces may have high levels of genetic diversity that could compensate for the lower levels of genetic diversity present in the landraces collected in 2007 in Nohalal. These landraces are no longer planted in Nohalal, even though the four Mayan towns are neighbors. The main reason for this could be the agricultural intensification present in Nohalal over the last 30 years. The Nohalal Mayan farmers have better soils that could be used to plant improved varieties of maize and other species with high commercial value. In the case of the Lima bean landraces, only those with commercial value (e.g., Sac and Mulición) are considered for planting in these areas.
To understand better the genetic erosion process in the landraces from northeastern Campeche between 1979 and 2007, Martínez-Castillo et al. (2011) also analyzed the genetic differentiation among the landraces between these two collected years. An analysis of molecular variance (AMOVA) (Excoffier et al. 1992) revealed that a great proportion (82.2 %) of the total variation can be explained by differentiation among the two temporal groups of accessions (1979 vs all-2007), with only 12.9 % of the total among accessions within years. To confirm these results, the authors analyzed the genetic relationships among the all accessions. An Unweighted Pair Group Method with Arithmetic mean (UPGMA) analysis (Fig. 8.6) indicated a grouping of the accessions in accordance with the years of collection, with high bootstrap values supporting each group. This result indicated that the genetic erosion is not only quantitative as discussed earlier, but also qualitative. Thus, over the last 30 years, there has been a shift in the genetic make-up of this crop in NECAMP. This result is also similar to other studies that have indicated that plant breeding can generate a qualitative change in the genetic diversity of crops (Khlestkina et al. 2004; Le Clerc et al. 2005; Mantegazza et al. 2008; Xiu-Quiang et al. 2007).
Dendrogram (UPGMA) based on Nei’s genetic distance (1978) of 44 accessions analyzed using 9 SSR loci. The numbers at the nodes are the proportion of similar replicates supporting each node (Reproduced from Martínez-Castillo et al. 2011)
Finaly, Martínez-Castillo et al. (2011) used the Bottleneck program (Luikart and Cornuet 1997) to look for a possible bottleneck event between 1979 and 2007, first for each year of collection (accessions from 1979 in Nohalal, accessions collected in 2007 in Nohalal, and accessions collected in 2007 in Nohalal, and the other three adjacent towns). For this analysis, Wilcoxon sign-rank tests were not significant, indicating there is no excess in gene diversity under any of three mutation models (Stepwise Mutation Model [SMM], Two-Phased Model [TPM], Infinite Allele Model [IAM]) considered in the Bottleneck program and thus no bottleneck event in these three gene pools. Only the Lima bean accessions collected in 1979 in Nohalal yielded values close to α = 0.05, indicating a possible bottleneck event. It is important to remember that the accessions collected in Nohalal in 1979 were obtained from the CIAT gene bank where the rejuvenation process of the seed alone can lead to genetic erosion. Even considering this factor, these accessions had higher genetic diversity than did all the accessions collected in 2007 (h = 0.18 vs h = 0.05, respectively).
8.5 Genetic Diversity in Lima Bean from the Yucatan Peninsula Compared with the Highland Mayan Subarea
Mayan culture is one of the most important of Mesoamerica. The Mayan geographical region has been divided into two subareas: lowlands and highlands (Ruz 1981; Sharer 1999). The Yucatan Peninsula is part of the Mayan lowlands; Chiapas state (Mexico), and Guatemala (except the Peten area) are part of the Mayan highlands. Whereas the genetic diversity of Lima bean planted in the Mayan lowlands is welldocumented (Martínez-Castillo et al. 2004, 2008, 2011), until 2012 there was no molecular data for Lima bean from the Mayan highlands. Considering this, using 73 ISSR loci, Camacho-Pérez (2012) analyzed the genetic diversity in the entire Mayan region and in each subarea (23 accessions from each subarea). Using a Bayesian approach (Zhivotovsky 1999), Camacho-Pérez found high levels of genetic diversity in the Mayan area (H = 0.45), with higher levels in the lowlands (H = 0.44) than in the highlands (H = 0.36). This author also found a high genetic differentiation between the two subareas (AMOVA = 65 % of total variation between groups) and a grouping pattern based on the presence of the two subareas. These results confirmed the importance of the Yucatan Peninsula as a center of genetic diversity for the Lima bean landraces.
8.6 Gene Flow and Genetic Introgression: Factors that Counter Genetic Erosion in Lima Bean in the Yucatan Peninsula
In the Yucatan Peninsula, inter-landrace gene flow and natural introgression of wild alleles may prevent the genetic erosion of Lima bean landraces. Martínez-Castillo et al. (2004) reported the planting of up to seven landraces in a single milpa and existence of a wide variety of hybrid seeds. Using eight SSR markers, Martínez-Castillo (2005) observed very high gene flow levels between landraces in each of the agricultural regions of the Yucatan Peninsula. On the other hand, Martínez-Castillo et al. (2006) reported high genetic diversity levels in the wild populations of Lima bean from this region, and Martínez-Castillo et al. (2007) documented wild–domesticated gene flow and weedy forms derived from this flow (Fig. 8.7). Ethnobotanical observations showed that these weedy forms generated by introgression are being incorporated by Mayan farmers into their cultivated gene pools (Fig. 8.4). It is considered that the natural wild–domesticated introgression has played a vital role in the evolution of domesticated species and continues to be an important factor in increasing genetic diversity in modern crops (Arnold 1992; Harlan 1965; Jarvis and Hodgkin 1999; Slatkin 1987).
Considering before mentioned, and the important role that traditional farmers have played in this microevolutionary process, Dzul-Tejero (2011) used 11 SSR loci to assess levels of introgression in three wild–weedy–domesticated complexes of Lima bean from three Mayan milpas of the Yucatan Peninsula (Fig. 8.3) analyzes its impact on the genetic diversity of this crop. A test of assignment of individuals using the STRUCTURE program (Pritchard et al. 2000) indicated that the complex with the lowest level of introgression was one where the farmer actively selected against wild plants and introgressed seed (Fig. 8.8). This complex was Xocen located in southern Yucatan-SEYUC- (Fig. 8.3). By contrast, the complex with the highest level of introgression was one where the farmer had been consciously selecting a weedy morphotype for 15 years and had already incorporated into his diet (Fig. 8.8). This complex was X-Hazil located in Central-east Quintana Roo -CEQROO- (Fig. 8.3). Dzul-Tejero (2011) also found that the genetic diversity was higher in the complex with the higher level of introgression. These results confirmed the importance of genetic introgression to maintain and increase the levels of genetic diversity in crops.
8.7 Conclusions
Studying the genetic erosion in Lima bean landraces in the Yucatan Peninsula is important because this region is likely playing an important role in the in situ conservation of the Mesoamerican gene pool of this crop (Ballesteros 1999; Camacho-Pérez 2012; Martínez-Castillo et al. 2004). Martínez-Castillo et al. (2008) showed that many landraces of Lima bean are at higher risk of genetic erosion because, with few farmers planted these landraces and with moderate genetic diversity, they represent the greatest loss of unique alleles if these landraces go to local extinction. On the other hand, the abundant landraces have the lowest genetic diversity levels and are thus at great risk of genetic erosion due to selection criteria imposed by an external market, too. Although the high gene flow levels observed in the domesticated gene pool (Martínez-Castillo 2005) and the existence of genetic introgression between domesticated and wild populations (Dzul-Tejero 2011) could be limiting the genetic erosion of the Lima bean landraces, it is important to consider that the landraces are not just a group of unrelated alleles. Instead, each is a package of alleles selected during centuries by traditional Mayan farmers to cope with different environmental restrictions. The loss of landraces could mean a great loss in the genetic diversity of this crop and in the production and surviving strategies of the Mayan farmers of this part of Mexico.
The temporal analysis done by Martínez-Castillo et al. (2011) confirmed the high risk of genetic erosion faced by Lima bean from the Yucatan Peninsula, indicating that a smaller number of landraces planted means also a minor level of genetic diversity present in the crop. Indeed, molecular data showed the existence of a decrease in the genetic diversity in between 1979 and 2007 as well as a great shift in the allelic composition. This genetic shift might possibly be a consequence of the introduction of improved varieties of P. lunatus or by changes in the Mayan criteria selection of germplasm or both. In the same way as in the Mayan town of Nohalal, the landraces may be disappearing in many other Mayan towns from this part of Mexico. This loss is not only a consequence of factors associated with agricultural intensification or the incorporation of the Mayan farmers into nonlocal market system. A series of environmental, socioeconomic and cultural factors are participating, too. For example, the Dean hurricane in 2007 in the south central state Quintana Roo caused seed loss of many cultivated species, among them, many rare landraces of P. lunatus. At present, several of these landraces have not been collected by our research group.
One little-studied factor in the genetic erosion of crops is a change in the food preferences of rural populations. For Lima bean in the Yucatan Peninsula, such preference takes three forms: (1) young adults and children do not eat them, (2) only the elderly plant many of the rare landraces for their own use, and (3) cowpea [Vigna unguiculata (L.) Walpers, locally known as x-pelon], introduced to the region from Africa in the twentieth century, has been replacing P. lunatus. Because the process of reintroducing a crop plant is a long one, Esquivel and Hammer (1988) in a study in Cuba proposed maintaining Lima bean landraces as part of the traditional horticultural system. In several Mayan towns of the Yucatan Peninsula, some landraces are planted into home gardens (Fig. 8.2), including the X-Konan jonal landrace (keeper of the house in Mayan, because this landrace can be planted in either home gardens or the milpa). However, such dual-planting of a landrace is not a common agricultural practice. On the other hand, loss of landraces is also apparently linked to different generations of human populations. Mayan farmers that plant a large variety of rare landraces are elderly; their death almost surely means the loss of these landraces.
8.8 Prospects
If the data about relative abundance reported by Martínez-Castillo et al. (2004) reflect the current condition of the domesticated Lima bean pool in the Yucatan Peninsula, then this species is at very high risk of genetic erosion since this region is one of its main centers of genetic diversity in Mesoamerica (Ballesteros et al. 1999; Camacho-Pérez 2012; Martínez-Castillo et al. 2004). If current trends continue in the region, many Lima bean landraces may cease to be grown into the milpa in two to three generations. To prevent the loss of this valuable germplasm, in situ conservation programs are needed to implement (1) an emergency collecting effort to save all landraces ex situ, as a backup for in situ conservation activities, (2) in situ conservation of landraces and their alleles, (3) conservation of wild–weedy–domesticated complexes that allow introgression of wild alleles into landraces, and (4) reintroduction of rare landraces and programs to promote their planting and acceptance among young Mayan producers and their families. To do this, areas need to be selected that favor in situ conservation while considering the natural, economic, social and cultural factors that contribute to this conservation. In the case of the Yucatan Peninsula, we consider that the corridor SEYUC–CEQROO is a good area for this in situ conservation, not only for P. lunatus, but also for the many other domesticated plants of the Mayan milpa of this region of Mexico. Our research group is presently collecting the landraces and wild populations of P. lunatus in the Yucatan Peninsula, to create a core collection representative of this important crop in the traditional Mayan agriculture of the Yucatan of Mexico.
References
Adams REW, Culbert TP (1977) The origins of civilization in the Maya lowlands. In: Adams REW (ed) The origins of Maya civilization. University of New Mexico, Albuquerque, pp 3–34
Andueza-Noh RH, Serrano-Serrano ML, Chacón MI, Sánchez del-Pino I, Camacho-Pérez L, Coello-Coello J, Mijangos-Cortés JO, Debouck DG, Martínez-Castillo J (2013) Multiple domestications of the Mesoamerican gene pool of Lima bean (Phaseolus lunatus L.): evidence from chloroplast DNA sequences. Genet Resour Crop Evol 60:1069–1086
Arnold ML (1992) Natural hybridization as an evolutionary process. Annu Rev Ecol Syst 23:237–261
Ballesteros GA (1999) Contribuciones al conocimiento del frijol Lima (Phaseolus lunatus L.) en América Tropical. Ph. D thesis. Colegio de Posgraduados. Montecillos, Estado de México, México
Baudet JC (1977) The taxonomic status of the cultivated types of lima bean (Phaseolus lunatus L.). Trop Grain Legume 7:29–30
Baudoin JP, Rocha O, Degreef J, Maquet A, Guarino L (2004) Ecogeography, demography, diversity and conservation of phaseolus lunatus L. in the Central Valley of Costa Rica. Systematic and ecogeographic studies on crop genepools. Internacional Plant Genetic Resources Institute, Rome, Italy. 94 p
Bellón MR, Taylor JE (1993) Farmer soil taxonomy and technology adoption. Econ Develop Cult Change 41:764–786
Brush S (1991) A farmer-based approach to conservation crop germplasm. Econ Bot 45:153–165
Camacho-Pérez L (2012) Diversidad, Estructura y relaciones genéticas del frijol Lima (Phaseolus lunatus L. var. lunatus) en el área maya. Master thesis. Centro de Investigación Científica de Yucatán, Mérida, México. 66 p
Castiñeiras L, Guzmán FA, Duque MC, Shagarodsky T, Cristóbal R, De Vicente MC (2007) AFLPs and morphological diversity of Phaseolus lunatus L. in Cuban home gardens: approaches to recovering the lost ex situ collection. Biodivers Conserv. doi:10.1007/s10531-006-9025-x
Colunga-GarcíaMarín P, May-Pat F (1993) Agave studies in Yucatán, Mexico I. Past and present germplasm diversity and uses. Econ Bot 47:312–327
Cuanalo de la Cerda HE, Arias LM (1997) Cultural and economic factors that affect farmer decision-making in Yucatan, Mexico. In: Jarvis DI, Hodgkin T (eds) Strengthening the scientific basis of in situ conservation of agricultural biodiversity on-farm. Options for data collecting and analysis, IPGRI, Rome, p 14
Culley TM, Sbita SJ, Wick A (2007) Population genetic effects of urban habitat fragmentation in the perennial herb Viola pubescens (Violaceae) using ISSR Markers. Ann Bot 100:91–100
Debouck DG (1979) Proyecto de recolección de germoplasma de Phaseolus en México. CIAT-INIA, Centro Internacional de Agricultura Tropical (CIAT), Colombia
Dzul-Tejero F (2011) Introgresión genética silvestre-domesticado del Ib (Phaseolus lunatus L.) en la agricultura Maya de la península de Yucatán, México. Master thesis. Centro de Investigación Científica de Yucatán, Mérida, Yucatán, México
Esquivel H, Hammer K (1988) The “conuco”- an important refuge of Cuban plant genetic resources. Kulturpflanze 36:451–463
Excoffier L, Smouse P, Quattro J (1992) Analysis of molecular variance inferred from metric distance among DNA haplotypes: applications to human mitochondrial DNA restriction data. Genetics 131:479–491
FAO (1996) The State of the World’s plant genetic resources: diversity and erosion. Third world resurgence. Farmers’ rights and the battle for agrobiodiversity. Issue No. 72/73 KDN PP6738/1/96. An excerpt from the report on the State of the World’s plant genetic resources prepared by the FAO secretariat for the international technical conference on plant genetic resources at Leipzig, Germany, 17–23 June 1996
Frankel OH, Bennett E (1970) Genetic resources in plants-their exploration and conservation. IBP Handbook No. 11. Blackwell Scientific Publications, Oxford
Galván MZ, Bornet B, Balatti PA, Branchard M (2003) Inter-simple sequence repeat (ISSR) markers as a tool for the assessment of both genetic diversity and gene pool origin in common bean (Phaseolus vulgaris L.). Euphytica 132:297–301. 94:597–602
González A, Wong A, Delgado-Salinas A, Papa R, Gepts P (2005) Assessment of inter simple sequence repeat markers to differentiate sympatric wild and domesticated populations of common bean. Crop Sci 45:606–615
Hammer K, Laghetti G (2005) Genetic erosion—examples from Italy. Genet Resour Crop Evol 52:629–634
Hamrick JL, Godt MJW, Murawski DA, Loveless MD (1991) Correlations between species traits and allozyme diversity: implications for conservation biology. In: Falk DA, Holsinger KE (eds) Genetics and conservation of rare plants. Oxford University Press, New York, pp 75–86
Harlan JR (1965) The possible role of weedy races in the evolution of cultivated plants. Euphytica 14:173–176
Harlan JR, de Wit JMJ (1971) Toward a rational classification of cultivated plants. Taxon 20:509–517
Hernández-Xolocotzi E (1973) Genetic resources of primitive varieties of Mesoamerica: Zea spp., Phaseolus spp., Capsicum spp., and Cucurbita spp. In: Survey of crop genetic resources in their centers of diversity. FAO, Roma, pp 76–115
Hernández-Xolocotzi E (1992) Racionalidad tecnológica del sistema de producción agrícola de roza-tumba-quema en Yucatán. In: Zizumbo-Villarreal D, Ramussen Ch, Arias-Reyes LM, Terán S (eds) La modernización de la milpa en Yucatán: utopía o realidad. CICY-DANIDA, Mérida, pp 187–194
Jarvis DI, Hodgkin T (1999) Wild relatives and crop cultivars: detecting natural introgression and farmer selection of new genetic combinations in agroecosystems. Mol Ecol 8:S159–S173
Jarvis DI, Myer L, Klemick H, Guarino L, Smale M, Brown AHD (2000) A training guide for in situ conservation on-farm. Version 1. International Plant Genetic Resources Institute, Rome, Italy
Kaplan L, Lynch T (1999) Phaseolus (Fabaceae) in Archaeology: AMS radio-carbon dates and their significance for pre-Colombian agriculture. Econ Bot 53:261–272
Ku-Naal R (1995) Cambios técnicos en la milpa bajo roza-tumba-quema en Yaxcabá, Yucatán. In: Hernández XE, Bello BE, Levy TS (eds) La milpa en Yucatán: Un sistema de producción agrícola tradicional. Colegio de Postgraduados, México, pp 401–418
Khlestkina EK, Huang XQ, Quenum FBJ, Chebotar S, Röder MS, Börner A (2004) Genetic diversity in cultivated plants—loss or stability? Theor Appl Genet 108:1466–1472
Le Clerc V, Bazante F, Baril C, Guiard J, Zhang D (2005) Assessing temporal change in genetic diversity of maize varieties using microsatellite markers. Theor Appl Genet 110:294–302
Luikart G, Cornuet JM (1997) Empirical evaluation of a test for identifying recently bottlenecked populations from allele frequency data. Conserv Biol 12:228–237
Lynch M, Milligan BG (1994) Analysis of population genetic structure with RAPD markers. Mol Ecol 3:91–99
Mantegazza R, Biloni M, Grassi F, Basso B, Lu BR, Cai XX, Sala F, Spada A (2008) Temporal trends of variation in Italian rice germplasm over the past two centuries revealed by AFLP and SSR markers. Crop Sci 48:1832–1840
Maquet A, Zoro Bi I, Delvaux M, Wathelet B, Baudoin JP (1997) Genetic structure of a Lima bean base collection using allozyme markers. Theor Appl Genet 95:980–991
Martínez-Castillo J (2005). Diversidad intraespecífica de Phaseolus lunatus L. e intensificación de la agricultura tradicional en la Península de Yucatán, México. Ph. D. thesis. Centro de Investigación Científica de Yucatán, A. C., Mérida, México
Martínez-Castillo J, Zizumbo-Villarreal D, Perales-Rivera H, Colunga-GarcíaMarín P (2004) Intraspecific diversity and morpho-phenological variation in Phaseolus lunatus L. from the Yucatan Peninsula, Mexico. Econ Bot 58(3):354–380
Martínez-Castillo J, Zizumbo-Villarreal Z, Gepts P, Delgado-Valerio P, Colunga-GarcíaMarín P (2006) Structure and genetic diversity of wild populations of Lima Bean (Phaseolus lunatus L.) from the Yucatan Peninsula, Mexico. Crop Sci 46:1071–1080
Martínez-Castillo J, Zizumbo-Villarreal D, Gepts P, Colunga-GarcíaMarín P (2007) Gene flow and genetic structure in the wild-weedy-domesticated complex of Lima bean (Phaseolus lunatus L.) in its Mesoamerican center of domestication and diversity. Crop Sci 47:58–66
Martínez-Castillo J, Colunga-GarcíaMarín P, Zizumbo-Villarreal D (2008) Genetic erosion and in situ conservation of Lima bean (Phaseolus lunatus L.) landraces in its Mesoamerican diversity center. Genet Resour Crop Evol 55:1065–1077
Martínez-Castillo J, Camacho-Pérez L, Coello-Coello J, Andueza-Noh RH (2011) Wholesale replacement of Lima bean (Phaseolus lunatus L.) landraces over the last 30 years in northeastern Campeche, Mexico. Genet Resour Crop Evol 59:191–204
Martins M, Tenreiro R, Oliveira MM (2003) Genetic relatedness of Portuguese almond cultivars assessed by RAPD and ISSR markers. Plant Cell Rep 22:71–78
Motta-Aldana J, Serrano-Serrano ML, Torres HJ, Villamizar CG, Debouck DG, Chacón MI (2010) Multiple origins of Lima bean landraces in the Americas: evidence from chloroplast and nuclear DNA polymorphisms. Crop Sci 50:1773–1787
Nienhuis J, Tivang J, Skroch P, dos Santos JB (1995) Genetic relationships among cultivars and landraces of Lima bean (Phaseolus lunatus L.) as measured by RAPD markers. J Am Soc Hortic Sci 120(2):300–306
Pérez-Toro A (1945) La agricultura milpera de los mayas de Yucatán. In: Enciclopedia yucatanense, vol. VI, ediciones del Gobierno de Yucatán, México
Pritchard J, Stephens M, Donelly P (2000) Inference of population structure usin multilocus genotype data. Genetics 155:945–959
Prevost A, Wilkinson M (1999) A new system of comparing PCR primers applied to ISSR fingerprinting of potato cultivars. Theor Appl Genet 98:107–112
Reyes GD, Aguilar CG (1992) Intensificación de la milpa en Yucatán. In: Zizumbo-Villarreal D, Ramussen Ch, Arias-Reyes LM, Terán S (eds) La modernización de la milpa en Yucatán: utopía o realidad. CICY-DANIDA, Mérida, pp 347–358
Ruz LA (1981) El pueblo maya. Salvat Mexicana de Ediciones, México
Serrano-Serrano ML, Hernandez-Torres J, Castillo-Villamizar G, Debouck DG, Chacón MI (2010) Gene pools in wild Lima bean (Phaseolus lunatus L.) from the Americas: evidences for an Andean origin and past migrations. Mol Phylogen Evol 54:76–87
Serrano-Serrano ML, Andueza-Noh RH, Martínez-Castillo J, Debouck DG, Chacón MI (2012) Evolution and domestication of Lima bean (Phaseolus lunatus L.) in Mexico: evidence from ribosomal DNA. Crop Sci 52:1698–1712
Shannon CE, Weaver W (1949) The mathematical theory of communication. University of Illinois Press, Urbana
Sharer R (1999) La civilización maya. Fondo de Cultura Económica, México
Slatkin M (1987) Gene flow and the geographic structure of natural populations. Science 236:787–792
Tautz D, Renz M (1984) Simple sequences are ubiquitous repetitive components of eukaryote genomes. Nucleid Acids Res 12:4127–4137
Tsegaye B, Berg T (2006) Genetic erosion of Ethiopian tetraploid wheat landraces in Eastern Shewa, Central Ethiopia. Genet Resour Crop Evol. doi:10.1007/s10722-006-0016-2
Vavilov NI (1926) Centers of origin of cultivated plants. Bull Appl Bot Genet Plant Breed 16:248
Xiu-Qiang H, Wolf M, Ganal MW, Orford S, Koebner RMD, Röder MS (2007) Did modern plant breeding lead to genetic erosion in European winter wheat varieties? Crop Sci 47:343–349
Zhou WJ, Zhang GQ, Tuvesson S, Dayteg C, Gertsson B (2006) Genetic survey of Chinese and Swedish oilseed rape (Brassica napus L.) by simple sequence repeats (SSRs). Genet Resour Crop Evol 53:443–447
Zhivotovsky LA (1999) Estimating population structure in diploids with multilocus dominant DNA markers. Mol Ecol 8:907–913
Zietkiewicz E, Rafalski A, Labuda D (1994) Genome finger-printing by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 20:176–183
Acknowledgments
This research was done in the Molecular Markers Laboratory of the Department of Natural Resources-CICY. The authors thank Julian Coello Coello for laboratory assistance.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Martínez-Castillo, J., May-Pat, F., Camacho-Pérez, L., Andueza-Noh, R.H., Dzul-Tejero, F. (2016). Genetic Erosion and In Situ Conservation of Lima Bean (Phaseolus Lunatus L.) Landraces in Mesoamerican Diversity Center. In: Ahuja, M., Jain, S. (eds) Genetic Diversity and Erosion in Plants. Sustainable Development and Biodiversity, vol 8. Springer, Cham. https://doi.org/10.1007/978-3-319-25954-3_8
Download citation
DOI: https://doi.org/10.1007/978-3-319-25954-3_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-25953-6
Online ISBN: 978-3-319-25954-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)