Abstract
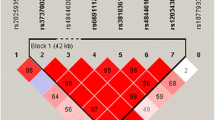
Increasing evidence suggests that inflammation is one pathophysiological mechanism in Alzheimer’s disease (AD). Recent studies have identified an association between the poly (ADP-ribose) polymerase 1 (PARP1) gene and AD. This gene encodes a protein that is involved in many biological functions, including DNA repair and chromatin remodeling, and is a mediator of inflammation. Therefore, we performed a targeted genetic association analysis to investigate the relationship between the PARP1 polymorphisms and brain microglial activity as indexed by [11C]PBR28 positron emission tomography (PET). Participants were 26 non-Hispanic Caucasians in the Indiana Memory and Aging Study (IMAS). PET data were intensity-normalized by injected dose/total body weight. Average [11C]PBR28 standardized uptake values (SUV) from 6 bilateral regions of interest (thalamus, frontal, parietal, temporal, and cingulate cortices, and whole brain gray matter) were used as endophenotypes. Single nucleotide polymorphisms (SNPs) with 20% minor allele frequency that were within +/- 20 kb of the PARP1 gene were included in the analyses. Gene-level association analyses were performed using a dominant genetic model with translocator protein (18-kDa) (TSPO) genotype, age at PET scan, and gender as covariates. Analyses were performed with and without APOE ε4 status as a covariate. Associations with [11C]PBR28 SUVs from thalamus and cingulate were significant at corrected p<0.014 and <0.065, respectively. Subsequent multi-marker analysis with cingulate [11C]PBR28 SUV showed that individuals with the “C” allele at rs6677172 and “A” allele at rs61835377 had higher [11C]PBR28 SUV than individuals without these alleles (corrected P<0.03), and individuals with the “G” allele at rs6677172 and “G” allele at rs61835377 displayed the opposite trend (corrected P<0.065). A previous study with the same cohort showed an inverse relationship between [11C]PBR28 SUV and brain atrophy at a follow-up visit, suggesting possible protective effect of microglial activity against cortical atrophy. Interestingly, all 6 AD and 2 of 3 LMCI participants in the current analysis had one or more copies of the “GG” allele combination, associated with lower cingulate [11C]PBR28 SUV, suggesting that this gene variant warrants further investigation.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
- Positron Emission Tomography
- Mild Cognitive Impairment
- Standardize Uptake Value
- Positron Emission Tomography Data
- Allele Combination
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Hickman, S.E., Allison, E.K., El Khoury, J.: Microglial dysfunction and defective beta-amyloid clearance pathways in aging Alzheimer’s disease mice. J. Neurosci. 28, 8354–8360 (2008)
Jones, L., Holmans, P.A., Hamshere, M.L., Harold, D., Moskvina, V., Ivanov, D., Pocklington, A., Abraham, R., Hollingworth, P., Sims, R., Gerrish, A., et al.: Genetic evidence implicates the immune system and cholesterol metabolism in the aetiology of Alzheimer’s disease. PLoS One 5, e13950 (2010)
Lambert, J.C., Grenier-Boley, B., Chouraki, V., Heath, S., Zelenika, D., Fievet, N., Hannequin, D., Pasquier, F., Hanon, O., Brice, A., Epelbaum, J., Berr, C., Dartigues, J.F., Tzourio, C., Campion, D., Lathrop, M., Amouyel, P.: Implication of the immune system in Alzheimer’s disease: evidence from genome-wide pathway analysis. J. Alzheimers Dis. 20, 1107–1118 (2010)
Zhang, R., Miller, R.G., Madison, C., Jin, X., Honrada, R., Harris, W., Katz, J., Forshew, D.A., McGrath, M.S.: Systemic immune system alterations in early stages of Alzheimer’s disease. J. Neuroimmunol. 256, 38–42 (2013)
Harold, D., Abraham, R., Hollingworth, P., Sims, R., Gerrish, A., Hamshere, M.L., Pahwa, J.S., Moskvina, V., Dowzell, K., Williams, A., Jones, N., Thomas, C., et al.: Genome-wide association study identifies variants at CLU and PICALM associated with Alzheimer’s disease. Nat. Genet. 41, 1088–1093 (2009)
Hollingworth, P., Harold, D., Sims, R., Gerrish, A., Lambert, J.C., Carrasquillo, M.M., Abraham, R., Hamshere, M.L., Pahwa, J.S., Moskvina, V., et al.: Common variants at ABCA7, MS4A6A/MS4A4E, EPHA1, CD33 and CD2AP are associated with Alzheimer’s disease. Nat. Genet. 43, 429–435 (2011)
Lambert, J.C., Heath, S., Even, G., Campion, D., Sleegers, K., Hiltunen, M., Combarros, O., Zelenika, D., Bullido, M.J., Tavernier, B., Letenneur, L., Bettens, K., et al.: Genome-wide association study identifies variants at CLU and CR1 associated with Alzheimer’s disease. Nat. Genet. 41, 1094–1099 (2009)
Naj, A.C., Jun, G., Beecham, G.W., Wang, L.S., Vardarajan, B.N., Buros, J., Gallins, P.J., Buxbaum, J.D., Jarvik, G.P., Crane, P.K., Larson, E.B., Bird, T.D., et al.: Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer’s disease. Nat. Genet. 43, 436–441 (2011)
Kauppinen, T.M., Suh, S.W., Higashi, Y., Berman, A.E., Escartin, C., Won, S.J., Wang, C., Cho, S.H., Gan, L., Swanson, R.A.: Poly(ADP-ribose)polymerase-1 modulates microglial responses to amyloid beta. J. Neuroinflammation 8, 152 (2011)
Liu, H.P., Lin, W.Y., Wu, B.T., Liu, S.H., Wang, W.F., Tsai, C.H., Lee, C.C., Tsai, F.J.: Evaluation of the poly(ADP-ribose) polymerase-1 gene variants in Alzheimer’s disease. J. Clin. Lab. Anal. 24, 182–186 (2010)
Nho, K., Corneveaux, J.J., Kim, S., Lin, H., Risacher, S.L., Shen, L., Swaminathan, S., Ramanan, V.K., Liu, Y., Foroud, T., Inlow, M.H., Siniard, A.L., et al.: Whole-exome sequencing and imaging genetics identify functional variants for rate of change in hippocampal volume in mild cognitive impairment. Mol. Psychiatry (2013), doi:10.1038/mp.2013.24
Strosznajder, J.B., Czapski, G.A., Adamczyk, A., Strosznajder, R.P.: Poly(ADP-ribose) polymerase-1 in amyloid beta toxicity and Alzheimer’s disease. Mol. Neurobiol. 46, 78–84 (2012)
Gehrmann, J., Matsumoto, Y., Kreutzberg, G.W.: Microglia: intrinsic immuneffector cell of the brain. Brain Res. Brain Res. Rev. 20, 269–287 (1995)
Kreisl, W.C., Fujita, M., Fujimura, Y., Kimura, N., Jenko, K.J., Kannan, P., Hong, J., Morse, C.L., Zoghbi, S.S., Gladding, R.L., Jacobson, S., Oh, U., Pike, V.W., Innis, R.B.: Comparison of [(11)C]-(R)-PK 11195 and [(11)C]PBR28, two radioligands for translocator protein (18 kDa) in human and monkey: Implications for positron emission tomographic imaging of this inflammation biomarker. Neuroimage 49, 2924–2932 (2010)
Risacher, S.L., Wudunn, D., Pepin, S.M., MaGee, T.R., McDonald, B.C., Flashman, L.A., Wishart, H.A., Pixley, H.S., Rabin, L.A., Pare, N., Englert, J.J., Schwartz, E., Curtain, J.R., West, J.D., O’Neill, D.P., Santulli, R.B., Newman, R.W., Saykin, A.J.: Visual contrast sensitivity in Alzheimer’s disease, mild cognitive impairment, and older adults with cognitive complaints. Neurobiol. Aging 34, 1133–1144 (2013)
Saykin, A.J., Wishart, H.A., Rabin, L.A., Santulli, R.B., Flashman, L.A., West, J.D., McHugh, T.L., Mamourian, A.C.: Older adults with cognitive complaints show brain atrophy similar to that of amnestic MCI. Neurology 67, 834–842 (2006)
Owen, D.R., Yeo, A.J., Gunn, R.N., Song, K., Wadsworth, G., Lewis, A., Rhodes, C., Pulford, D.J., Bennacef, I., Parker, C.A., St Jean, P.L., Cardon, L.R., Mooser, V.E., Matthews, P.M., Rabiner, E.A., Rubio, J.P.: An 18-kDa translocator protein (TSPO) polymorphism explains differences in binding affinity of the PET radioligand PBR28. J. Cereb. Blood Flow. Metab. 32, 1–5 (2012)
Yoder, K.K., Nho, K., Risacher, S.L., Kim, S., Shen, L., Saykin, A.J.: Influence of TSPO genotype on [11C]PBR28 standardized uptake values. Journal of Nuclear Medicine (2013), doi:10.2967/jnumed.112.118885
Purcell, S., Neale, B., Todd-Brown, K., Thomas, L., Ferreira, M.A., Bender, D., Maller, J., Sklar, P., de Bakker, P.I., Daly, M.J., Sham, P.C.: PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007)
Shen, L., Kim, S., Risacher, S.L., Nho, K., Swaminathan, S., West, J.D., Foroud, T., Pankratz, N., Moore, J.H., Sloan, C.D., Huentelman, M.J., Craig, D.W., Dechairo, B.M., Potkin, S.G., Jack Jr., C.R., Weiner, M.W., Saykin, A.J.: Whole genome association study of brain-wide imaging phenotypes for identifying quantitative trait loci in MCI and AD: A study of the ADNI cohort. Neuroimage 53, 1051–1063 (2010)
Risacher, S.L., Kim, S., Yoder, K.K., Shen, L., West, J.D., McDonald, B.C., Wang, Y., Nho, K., Tallman, E., Hutchins, G.D., Fletcher, J.W., Ghetti, B., Gao, S., Farlow, M.R., Saykin, A.J.: Relationship of microglial activation measured by [11C]PBR28 PET, atrophy on MRI, and plasma biomarkers in individuals with and at-risk for Alzheimer’s disease. In: Alzheimer’s Association International Conference 2013 (Abstract number: 39417) (2013)
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer International Publishing Switzerland
About this paper
Cite this paper
Kim, S. et al. (2013). PARP1 Gene Variation and Microglial Activity on [11C]PBR28 PET in Older Adults at Risk for Alzheimer’s Disease. In: Shen, L., Liu, T., Yap, PT., Huang, H., Shen, D., Westin, CF. (eds) Multimodal Brain Image Analysis. MBIA 2013. Lecture Notes in Computer Science, vol 8159. Springer, Cham. https://doi.org/10.1007/978-3-319-02126-3_15
Download citation
DOI: https://doi.org/10.1007/978-3-319-02126-3_15
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-02125-6
Online ISBN: 978-3-319-02126-3
eBook Packages: Computer ScienceComputer Science (R0)