Abstract
Water resources are gradually decreasing and most of water is polluted by different pollutants from household, agricultural and industrial sources. Previously, the main focus of bioremediation treatment technologies was on the use of free microbial cells. Recently, immobilized microbial cells for the bioremediation of pollutants from wastewater have gained immense interest. Microbial biofilms are microbial cells attached on a surface via polymeric substances (extracellular polymeric substances). The main aim of this chapter is to present the role of immobilized microbial cell systems in bioremediation of different pollutants. Microbial cell immobilization methods are being employed to control various recalcitrant pollutants from wastewater, and the tendency of this interesting area is expected to increase in the future. Here, an attempt is made to discuss microbial biofilms and immobilization of microbial cells. Moreover, developments of immobilized cells are highlighted, and different immobilization support materials are discussed in reference to wastewater bioremediation. Also, the applications of immobilized microbial cells in different areas are demonstrated. Finally, brief conclusions and future research outlooks in the area of microbial biofilm cell systems for bioremediation of pollutants are highlighted.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
14.1 Introduction
The increasing pollution in water is of great alarm for the public about recalcitrant hazardous compounds from different sources. It is important to know that environment is contaminated via different pollutants like heavy metals, pesticides, phenolic compounds, dyes, nutrients and organic compounds which pose serious environmental issues (Mohamed et al. 2016; Rodgers-Vieira et al. 2015; Smułek et al. 2015; Bhat et al. 2017).
The majority of wastewater comes from different industries, and these wastewaters are deleterious and pose serious environmental and human health issues. Thus, it is of prime importance to control these pollutants via effective treatment technologies which must be efficient and cheap. Treated water must be recycled back to the same process or reused. Various methods have been tested to treat industrial wastewaters such as chemical, biological and mechanical treatment methods. Biotechnological methods that use microbes for degradation of pollutants in wastewater have been largely used (Zhang et al. 2018). These biological treatment strategies are cheap and in these treatment processes no toxic chemical is added. Also, these methods have the ability to completely degrade pollutants (Sharma 2012). However, using microbes for the removal of pollutants from wastewater, there is also difficulty of separating microbes after treatment of wastewater (Tam et al. 2010).
Treatments of pollutant control methods have mainly two limitations: the microbial cells are difficult to separate after treatment process; their reuse and long-term stability. Thus, immobilized microbial cells via physical or chemical ways can overcome these limitations (Hartmeier 2012; Yiğitoğlu and Temoçin 2010). Immobilized microbial cells are gaining huge significance due to their numerous benefits compared to free cell methods for the treatment of wastewater. Immobilized microbial cells provide high mechanical strength, high mass, reuse, stability and resistance to toxic pollutants (Kadimpati et al. 2013).
Microbial cells can be attached or entrapped on/in different support materials. These support materials can be organic or inorganic or water-insoluble materials. Different immobilization cell systems and support materials were used for various wastewaters (Kadimpati et al. 2013). Whole cell immobilization for wastewater provides simple separation from treated water. Also, whole cell microbial immobilization makes available long-term stability of microbial stability of enzymes and activity of enzymes (Stolarzewicz et al. 2011).
In this chapter, we have comprehensively discussed the role of immobilized microbial cells and focused on immobilization methods and support materials for immobilization of microbial cells. Also, the role of microbial immobilized cell systems in the bioremediation of contaminated wastewater is discussed. Lastly, conclusions and future prospects in biofilm-based bioremediation are highlighted.
14.2 Microbial Immobilization
Microbial immobilization is the attachment or entrapment of microbial cells using support materials. Generally immobilization is the imprisonment or restriction of the movement of a cell (Zhang et al. 2004). Usually immobilization can be used for plant cells, animal cells, microbial cells or enzymes. In recent studies, whole microbial cells have been immobilized on support materials for the control of environmental pollutants. Immobilized microbial cells are of three types which consist of growing, dead and living. Therefore, it is important to select the suitable type of immobilized cells for a particular application (Rahman et al. 2006).
Microbial immobilized cells are more stable compared to immobilized enzymes. In this system, it is not necessary to extract enzymes from microbial cells. When using enzymes, they are prone to less stability in harsh conditions; moreover, enzyme systems’ unnecessary reactions take place (Stolarzewicz et al. 2011). It is important to know that the area of whole cell immobilization is different from health sciences to food industries. Immobilized microbes on support materials can be reused in fresh bioprocess reactions for treatment of wastewater or production of a variety of products. Reuse of immobilized cells can reduce cost of the production or treatment process (Mrudula and Shyam 2012; Ohta et al. 1994).
14.3 Support Materials for Immobilization
For microbial cell immobilization, it is important to select a suitable support material. Support materials must meet the following norms (Zacheus et al. 2000):
-
(a)
Immobilization support materials must have long shelf life.
-
(b)
Support materials must be non-biodegradable and must be nonhazardous.
-
(c)
Materials must be cheap and easily available.
-
(d)
These support materials must be easily separated from cells.
-
(e)
Support materials must have high chemical and mechanical stability.
-
(f)
These materials can be sterilizable.
-
(g)
Materials must be suitable for regeneration.
It is vital to know that the choice of support material for anoxic biomass immobilization can greatly affect the efficacy of a bioreactor or fermenter. Microbial cells attached on the surface depend on the support material which directly affects the number of microbes attached to it. Support materials are mainly classified in two main sets: organic and inorganic support materials (Lu and Toy 2009). Organic support materials are used such as dextran, celluloses, and agarose, while inorganic materials are porous glass, activated charcoal, clay, etc. (Lu and Toy 2009).
Organic support materials are available in large variety compared to inorganic support materials. Also, organic immobilization materials can be acquired with desired porosity. These materials are sensitive to pH, while inorganic support materials are resistant to chemicals, pH and microbial degradation. These support materials are also more feasible in scale-up process (Ispas et al. 2009; Magner 2013).
Also, organic support materials can be classified into synthetic and natural polymers. Different synthetic polymers such as polyvinyl, resins, acrylamide and polyurethane are also employed for microbial immobilization. Few examples of organic natural materials are agar, agarose and carrageenan (Hartmann 2005). In most of the studies, alginate polymers were used due to various benefits like they are environment-friendly and nontoxic to humans. Also, they are cheap and obtainable in huge amount. Moreover, immobilization in alginate also avoids changes in physiological condition (Buque et al. 2002).
14.4 Immobilization Methods
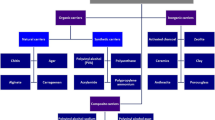
Recently, there is more focus on using immobilization methods for bioremediation of wastewater. Different types of immobilization methods have been employed for immobilization. Among these methods most important are encapsulation, adsorption, binding on surface and entrapment (Kourkoutas et al. 2004). This method is also elaborated briefly in Fig. 14.1.
Different immobilization methods (Bayat et al. 2015)
14.4.1 Adsorption
This method is simple and quick. This method is reversible. Adsorption is the most commonly used method for bioremediation of wastewater via immobilization technique. Adsorption can be defined as immobilization of enzymes or microbes via their physical interaction with surface of support materials. There is no need for addition of chemical additives. Adsorption method is cheap and environment-friendly. Adsorption completely is achieved via formation of weak bonds (hydrogen, ionic and van der waals forces, etc.). Also, these interactions are not strong and unstable; hence there is possibility of leakage in environment (Hou et al. 2014).
14.4.2 Covalent Binding
This method of immobilization is reversible due to covalent bond formation with the support material and microbial cell when a cross-linking material is present. This method is mostly used for immobilization of enzymes. In rare cases covalent binding method is applied owing to the toxicity which causes cell death. Covalent binding method is different from electrostatic binding (Groboillot et al. 1994).
14.4.3 Entrapment in Porous Matrix
Entrapment of microbial cells is widely used for pollutants treatment. Once microbes are entrapped, they can move inside the entrapped support material. Depending on the support materials, it can reduce the leakage of materials across the support material. Also, it can reduce the passage of nutrients. It is found that those microbes which stay near the surface have high activity and metabolic process, while other microbes have less activity (Bleve et al. 2011). There are numerous advantages of entrapment method such as it is environmentally friendly, cheap and nontoxic (Wojcieszyńska et al. 2012). This method helps in protection of microbes from the harsh environmental conditions. It is vital to understand that pore sizes of the support entrapment material must be smaller than the microbes. If these support materials have large pore size, there is possibility of leakage (Bleve et al. 2011).
14.4.4 Encapsulation
This method is quite similar to the method of entrapment. It is irreversible method of immobilization of microbial cells. In this method particles are isolated from external environment. The major benefit of this method is the protection of cells from toxic or extreme conditions. The protective barrier around the microorganism allows the passage of nutrients. This technique is cheap and fast. In addition to its benefits, there are also some limitations which hinder the large-scale application of this technique. Among the major limitations is injury can occur to the encapsulation material. Due to the limitations discussed here, this method is not widely used for bioremediation of wastewater. Pore size is also important, and if immobilization support material leaks, it can decrease the loading, and hence, it affects the efficiency of bioremediation process (Klein et al. 2012).
14.5 Role of Microbial Biofilm Cell Systems in Bioremediation of Wastewater
The increasing water pollution poses serious environmental concerns. It is important to find new ways to control water pollution. Recently, microbial biofilm-based strategies for the control of water pollution are increasing to cater this issue. In this section of the chapter, we will focus on the bioremediation of different wastewaters via immobilized microbial cells.
14.5.1 Bioremediation of Heavy Metals
Most of the industrial wastewaters contain hazardous metals such as copper, lead, cadmium, etc. (Jencarova and Luptakova 2017). These wastewaters produce free radicals which can cause serious environmental hazards and concerns (Gumpu et al. 2015). Hence, it is of prime important to treat heavy metals containing wastewater. Several methods have been tested for the treatment of heavy metal wastewater, but most of these methods are expensive and have various limitations. In few methods, several adsorbents are used but mostly are not efficient. Also their efficiency is usually augmented by increasing the surface area of the adsorbents (Ahmed et al. 2015). In one of the study reported, Penicillium citrinum was entrapped sodium alginate matrix. Sodium alginate beads containing Penicillium citrinum were produced, and beads were used for bioremediation of Cu(II) removal. It was observed that immobilized beads containing microbe remove Cu(II) up to 84.5%. However, for free cells it was 82.4%. From these results, it was exhibited that immobilized microbial cells are more efficient in removal of tested metal. Table 14.1 also demonstrates other methods for bioremediation of heavy metals using immobilized microbes. In another study, under batch conditions, 95% metal removal was achieved using sulphate-reducing bacteria in sodium alginate. A continuous removal strategy was also employed in which 99% of Cu(II) and 95.8% of Zn(II) were achieved (Kiran et al. 2018).
14.5.2 Bioremediation of Refractory Organic Wastewater
Bioremediation of phenolic- or aniline-based compounds is difficult to achieve (Luan et al. 2017). Most of the refractory compounds such as aniline or phenolic compounds are not degraded efficiently through present conventional treatment methods (Cesaro et al. 2013). It is owing to the long time needed for the microbial cells to grow and stay in the reaction process. However, immobilized microbial cells provide higher mass of cells and also microbial cells are more stable. In one of the study, Bacillus sp. SAS19 for phenol degradation was immobilized on porous carbonaceous gels. In this study it was exhibited that immobilized bacteria were more efficient in degradation of phenolics. It was found that immobilized bacteria can degrade phenol (1600 mg/L) up to 100% in 24 h (Ke et al. 2018). Lu and Toy (2009) also reported application of Phanerochaete chrysosporium . This fungus was immobilized on wood chips. Immobilized fungus was applied for biodegradation of phenolic compounds from coking wastewater. Immobilized fungus exhibited 87.05% degradation of phenol which was higher compared to non-attached fungus. The optimal removal of phenol was 84% in 3 days.
14.5.3 Bioremediation of Industrial Dyes
Different industries utilize dyes for different purposes including textile, leather, pharmaceutical, etc. After these processes, a huge amount of wastewater containing these dyes is discharged into the water bodies which pose different environmental concerns. Also, these dyes cause mutation and deleterious health concerns. Thus, it is important to treat these dyes containing wastewater. Many studies are performed to cater this issue and control dye wastewater before its discharge into the water streams. Different microorganisms are tested for dye treatment under different conditions in batch and continuous mode. Most common microbes are funguses which are more efficient in removal and degradation of dyes from wastewater (Couto 2009). In one of the study, Brevibacillus parabrevis was immobilized on coconut shell biochar. It was exhibited that under optimum conditions and with inoculum of 3 ml, removal of 95.7% of Congo red dye was achieved after 6 days (Talha et al. 2018). Hameed and Ismail (2018) found that using immobilized mix cells, reactive red dye (10 mg/L) was completely decolourized within 30 h under anaerobic conditions. For other studies, Table 14.1 shows different methods of bioremediation of dye wastewater.
14.5.4 Bioremediation of Nitrogen and Phosphorus
Nutrients are vital for the growth of microorganisms, but their increase in water bodies causes eutrophication (Tang et al. 2017).
Hence, it is necessary to treat water containing excessive nutrients before discharging it into rivers and lakes. Various techniques have been used for the control of nutrients (e.g. nitrogen and phosphorus). These conventional processes are adsorption, membrane processes, chemical precipitation, biological processes, etc. (Kumar et al. 2018). Due to the tremendous benefits of immobilized microbial cell technology, various studies have utilized immobilized cells for the bioremediation of nitrogen and phosphorus removal from wastewater. Shi et al. (2007) tested two green microalgae (Chlorella vulgaris and Scenedesmus rubescens) for phosphorus and nitrogen removal. Microalgae were immobilized via twin-layer system. It was exhibited that both algae tested removed nitrate from wastewater. When they have used secondary wastewater, both algae removed nitrate and phosphate to less than 10% in 9 days. Table 14.1 illustrates different immobilized microbial cells to control nutrients from wastewater.
14.6 Conclusions
It is exhibited from the studies that immobilized microbial cells have great potential for bioremediation of wastewater. In many studies it was found that immobilized cells are efficient compared to free cells for bioremediation of polluted water. Immobilized microbial cell systems provide numerous benefits compared to free cells for treatment of wastewater. In various studies, it was reported that immobilized cells have longer stability, lower cost, and higher degrading ability. Also, immobilized cells increase tolerance to harsh conditions which also makes them more suitable compared to free cells.
14.7 Future Prospects
Cloning of genes for biosurfactant synthesis and chemotactic ability of Genetically Engineered Microbes (GEMs) can further enhance the biodegradative capability of modified microbes. Nevertheless, the release and use of GEM in the nature and transmission is under much debate and controversial. However, the majority of organisms usually have other disabling mutations that will not permit the microbes to grow outside a given environment. Reengineering of secreted proteins in biofilm matrix is also an area for further development in the field of bioremediation. Bioremediation studies to test the effectiveness of biofilm under conditions similar to those encountered in natural environment still remain few. Many issues remain unclear such as the correlation between biofilm microstructure and biodegradation process, long-term behaviour of biofilms exposed to fluctuations in pollutant concentrations and the detail of the correlation between soil composition and biofilm behaviour. Solutions to these issues will provide a predictive and quantitative model for bioremediation using biofilm-based methods, so to improve this large scale of application of this environmentally sustainable technology.
References
Ahmed YM, Al-Mamun A, Al Khatib MFR, Jameel AT, AlSaadi MAHAR (2015) Efficient lead sorption from wastewater by carbon nanofibers. Environ Chem Lett 13(3):341–346
Bayat Z, Hassanshahian M, Cappello S (2015) Immobilization of microbes for bioremediation of crude oil polluted environments: a mini review. Open Microbiol J 9:48
Bhat RA, Shafiq-ur-Rehman, Mehmood MA, Dervash MA, Mushtaq N, Bhat JIA, Dar GH (2017) Current status of nutrient load in Dal Lake of Kashmir Himalaya. J Pharma Phytochem 6(6):165–169
Bleve G, Lezzi C, Chiriatti M, D’Ostuni I, Tristezza M, Di Venere D, Sergio L, Mita G, Grieco F (2011) Selection of non-conventional yeasts and their use in immobilized form for the bioremediation of olive oil mill wastewaters. Bioresour Technol 102(2):982–989
Buque EM, Chin-Joe I, Straathof AJ, Jongejan JA, Heijnen JJ (2002) Immobilization affects the rate and enantioselectivity of 3-oxo ester reduction by baker’s yeast. Enzym Microb Technol 31(5):656–664
Cesaro A, Naddeo V, Belgiorno V (2013) Wastewater treatment by combination of advanced oxidation processes and conventional biological systems. J Bioremed Biodegrad 4(8):1–8
Couto SR (2009) Dye removal by immobilised fungi. Biotechnol Adv 27(3):227–235
Ereqat SI, Abdelkader AA, Nasereddin AF, Al-Jawabreh AO, Zaid TM, Letnik I, Abdeen ZA (2018) Isolation and characterization of phenol degrading bacterium strain Bacillus thuringiensis J20 from olive waste in Palestine. J Environ Sci Health A 53(1):39–45
Erkaya IA, Arica MY, Akbulut A, Bayramoglu G (2014) Biosorption of uranium (VI) by free and entrapped Chlamydomonas reinhardtii: kinetic, equilibrium and thermodynamic studies. J Radioanal Nucl Chem 299(3):1993–2003
Eroglu E, Agarwal V, Bradshaw M, Chen X, Smith SM, Raston CL, Iyer KS (2012) Nitrate removal from liquid effluents using microalgae immobilized on chitosan nanofiber mats. Green Chem 14(10):2682–2685
Groboillot A, Boadi D, Poncelet D, Neufeld R (1994) Immobilization of cells for application in the food industry. Crit Rev Biotechnol 14(2):75–107
Gumpu MB, Sethuraman S, Krishnan UM, Rayappan JBB (2015) A review on detection of heavy metal ions in water–an electrochemical approach. Sensors Actuators B Chem 213:515–533
Gupta A, Balomajumder C (2015) Simultaneous removal of Cr (VI) and phenol from binary solution using Bacillus sp. immobilized onto tea waste biomass. J Water Process Eng 6:1–10
Hameed BB, Ismail ZZ (2018) Decolorization, biodegradation and detoxification of reactive red azo dye using non-adapted immobilized mixed cells. Biochem Eng J 137:71–77
Hartmann M (2005) Ordered mesoporous materials for bioadsorption and biocatalysis. Chem Mater 17(18):4577–4593
Hartmeier W (2012) Immobilized biocatalysts: an introduction. Springer Science & Business Media, Berlin
Hou J, Dong G, Ye Y, Chen V (2014) Laccase immobilization on titania nanoparticles and titania-functionalized membranes. J Membr Sci 452:229–240
Ismail ZZ, Khudhair HA (2015) Recycling of immobilized cells for aerobic biodegradation of phenol in a fluidized bed bioreactor. Syst Cyber Informat 13(5):81–86
Ispas C, Sokolov I, Andreescu S (2009) Enzyme-functionalized mesoporous silica for bioanalytical applications. Anal Bioanal Chem 393(2):543–554
Jencarova J, Luptakova A (2017) The application of biogenically created sorbent for metal ions elimination. Inżynieria Mineralna 18
Jimenez-Perez M, Sanchez-Castillo P, Romera O, Fernandez-Moreno D, Pérez-Martınez C (2004) Growth and nutrient removal in free and immobilized planktonic green algae isolated from pig manure. Enzym Microb Technol 34(5):392–398
Kadimpati KK, Mondithoka KP, Bheemaraju S, Challa VRM (2013) Entrapment of marine microalga, Isochrysis galbana, for biosorption of Cr (III) from aqueous solution: isotherms and spectroscopic characterization. Appl Water Sci 3(1):85–92
Ke Q, Zhang Y, Wu X, Su X, Wang Y, Lin H, Mei R, Zhang Y, Hashmi MZ, Chen C (2018) Sustainable biodegradation of phenol by immobilized Bacillus sp. SAS19 with porous carbonaceous gels as carriers. J Environ Manag 222:185–189
Kiran MG, Pakshirajan K, Das G (2018) Heavy metal removal from aqueous solution using sodium alginate immobilized sulfate reducing bacteria: mechanism and process optimization. J Environ Manag 218:486–496
Klein S, Avrahami R, Zussman E, Beliavski M, Tarre S, Green M (2012) Encapsulation of Pseudomonas sp. ADP cells in electrospun microtubes for atrazine bioremediation. J Ind Microbiol Biotechnol 39(11):1605–1613
Kourkoutas Y, Bekatorou A, Banat IM, Marchant R, Koutinas A (2004) Immobilization technologies and support materials suitable in alcohol beverages production: a review. Food Microbiol 21(4):377–397
Kumar TP, Mandlimath TR, Sangeetha P, Revathi S, Kumar SA (2018) Nanoscale materials as sorbents for nitrate and phosphate removal from water. Environ Chem Lett 16(2):389–400
Lertsutthiwong P, Boonpuak D, Pungrasmi W, Powtongsook S (2013) Immobilization of nitrite oxidizing bacteria using biopolymeric chitosan media. J Environ Sci 25(2):262–267
Lu J, Toy PH (2009) Organic polymer supports for synthesis and for reagent and catalyst immobilization. Chem Rev 109(2):815–838
Luan M, Jing G, Piao Y, Liu D, Jin L (2017) Treatment of refractory organic pollutants in industrial wastewater by wet air oxidation. Arab J Chem 10:S769–S776
Magner E (2013) Immobilisation of enzymes on mesoporous silicate materials. Chem Soc Rev 42(15):6213–6222
Mehta SK, Gaur JP (2001) Removal of Ni and Cu from single and binary metal solutions by free and immobilized Chlorella vulgaris. Eur J Protistol 37(3):261–271
Mohamed A, El-Sayed R, Osman T, Toprak M, Muhammed M, Uheida A (2016) Composite nanofibers for highly efficient photocatalytic degradation of organic dyes from contaminated water. Environ Res 145:18–25
Mrudula S, Shyam N (2012) Immobilization of Bacillus megaterium MTCC 2444 by Ca-alginate entrapment method for enhanced alkaline protease production. Braz Arch Biol Technol 55(1):135–144
Ogugbue CJ, Morad N, Sawidis T, Oranusi NA (2012) Decolorization and partial mineralization of a polyazo dye by Bacillus firmus immobilized within tubular polymeric gel. 3. Biotech 2(1):67–78
Ohta T, Ogbonna J, Tanaka H, Yajima M (1994) Development of a fermentation method using immobilized cells under unsterile conditions. 2. Ethanol and L-lactic acid production without heat and filter sterilization. Appl Microbiol Biotechnol 42(2–3):246–250
Podder M, Majumder C (2015) Bacteria immobilization on neem leaves/MnFe2O4 composite surface for removal of As (III) and As (V) from wastewater. Arab J Chem. https://doi.org/10.1016/j.arabjc.2015.08.025
Rahman RNZA, Ghazali FM, Salleh AB, Basri M (2006) Biodegradation of hydrocarbon contamination by immobilized bacterial cells. J Microbiol 44(3):354–359
Rodgers-Vieira EA, Zhang Z, Adrion AC, Gold A, Aitken MD (2015) Identification of anthraquinone-degrading bacteria in soil contaminated with polycyclic aromatic hydrocarbons. Appl Environ Microbiol 81(11):3775–3781
Sharma S (2012) Bioremediation: features, strategies and applications. Asian J Pharm Life Sci 2231:4423
Shi J, Podola B, Melkonian M (2007) Removal of nitrogen and phosphorus from wastewater using microalgae immobilized on twin layers: an experimental study. J Appl Phycol 19(5):417–423
Smułek W, Zdarta A, Guzik U, Dudzińska-Bajorek B, Kaczorek E (2015) Rahnella sp. strain EK12: cell surface properties and diesel oil biodegradation after long-term contact with natural surfactants and diesel oil. Microbiol Res 176:38–47
Stolarzewicz I, Białecka-Florjańczyk E, Majewska E, Krzyczkowska J (2011) Immobilization of yeast on polymeric supports. Chem Biochem Eng Q 25(1):135–144
Suganya K, Revathi K (2016) Decolorization of reactive dyes by immobilized bacterial cells from textile effluents. Int J Curr Microbiol Appl Sci 5:528–532
Talha MA, Goswami M, Giri B, Sharma A, Rai B, Singh R (2018) Bioremediation of Congo red dye in immobilized batch and continuous packed bed bioreactor by Brevibacillus parabrevis using coconut shell bio-char. Bioresour Technol 252:37–43
Tam N, Chan M, Wong Y, Popov V, Itoh H, Mander U (2010) Removal and biodegradation of polycyclic aromatic hydrocarbons by immobilized microalgal beads. WIT Trans Ecol Environ 140:391–402
Tang C-J, Duan C-S, Yu C, Song Y-X, Chai L-Y, Xiao R, Wei Z, Min X-B (2017) Removal of nitrogen from wastewaters by anaerobic ammonium oxidation (ANAMMOX) using granules in upflow reactors. Environ Chem Lett 15(2):311–328
Upendar G, Dutta S, Chakraborty J, Bhattacharyya P (2016) Removal of methylene blue dye using immobilized bacillus subtilis in batch & column reactor. Mater Today Proc 3(10):3467–3472
Wang W, Ding Y, Wang Y, Song X, Ambrose RF, Ullman JL, Winfrey BK, Wang J, Gong J (2016) Treatment of rich ammonia nitrogen wastewater with polyvinyl alcohol immobilized nitrifier biofortified constructed wetlands. Ecol Eng 94:7–11
Wojcieszyńska D, Hupert-Kocurek K, Jankowska A, Guzik U (2012) Properties of catechol 2, 3-dioxygenase from crude extract of Stenotrophomonas maltophilia strain KB2 immobilized in calcium alginate hydrogels. Biochem Eng J 66:1–7
Yiğitoğlu M, Temoçin Z (2010) Immobilization of Candida rugosa lipase on glutaraldehyde-activated polyester fiber and its application for hydrolysis of some vegetable oils. J Mol Catal B Enzym 66(1–2):130–135
Zacheus OM, Iivanainen EK, Nissinen TK, Lehtola MJ, Martikainen PJ (2000) Bacterial biofilm formation on polyvinyl chloride, polyethylene and stainless steel exposed to ozonated water. Water Res 34(1):63–70
Zhang Y-Q, Tao M-L, Shen W-D, Zhou Y-Z, Ding Y, Ma Y, Zhou W-L (2004) Immobilization of L-asparaginase on the microparticles of the natural silk sericin protein and its characters. Biomaterials 25(17):3751–3759
Zhang W, Grimi N, Jaffrin MY, Ding L, Tang B, Zhang Z (2018) Optimization of RDM-UF for alfalfa wastewater treatment using RSM. Environ Sci Pollut Res 25(2):1439–1447
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Siddiqui, M.F. et al. (2020). Microbial Biofilm Cell Systems for Remediation of Wastewaters. In: Hakeem, K., Bhat, R., Qadri, H. (eds) Bioremediation and Biotechnology. Springer, Cham. https://doi.org/10.1007/978-3-030-35691-0_14
Download citation
DOI: https://doi.org/10.1007/978-3-030-35691-0_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-35690-3
Online ISBN: 978-3-030-35691-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)