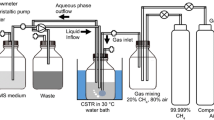
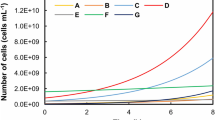
Abstract
Aerobic methanotrophs are metabolically unique bacteria that are able to utilize methane as a sole source of energy. The selective approach to enrich methanotrophs therefore employs mineral media with methane added as a growth substrate. The composition of a mineral medium and the incubation conditions strongly determine the outcome of the enrichment procedure. Adjusting medium composition to mirror the conditions of a natural habitat is a key to isolating environmentally relevant organisms. Methane-oxidizing enrichments typically contain many non-methanotrophic bacteria growing on by-products of methanotrophs or on medium contaminants, so obtaining isolated cultures is a time-consuming process that involves repeated series of plating on solid media, dilution–extinction in liquid media, and other purification procedures. Purity tests represent an important component of the isolation procedure. Final steps include registration of growth dynamics on methane and molecular identification of isolates.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Söhngen NL (1906) Über Bakterien, welche Methan als Kohlenstoffnahrung and Energiequelle gebrauchen. Zentralbl Bakteriol Parasitik Abt I 15:513–517
Stein LY, Roy R, Dunfield PF (2012) Aerobic methanotrophy and nitrification: processes and connections. In: Battista J et al (eds) Encyclopedia of life sciences. Wiley, Chichester, www.els.net
Nazaries L, Murrell JC, Millard P, Baggs L, Singh BK (2013) Methane, microbes and models: fundamental understanding of the soil methane cycle for future predictions. Environ Microbiol 15:2395–2417
Op den Camp HJM, Islam T, Stott MB, Harhangi HR, Hynes A, Schouten S, Jetten MSM, Birkeland N-K, Pol A, Dunfield PF (2009) Environmental, genomic and taxonomic perspectives on methanotrophic Verrucomicrobia. Environ Microbiol Rep 1:293–306
Sharp CE, Smirnova AV, Graham JM, Stott MB, Khadka R, Moore TR, Grasby SE, Strack M, Dunfield PF (2014) Distribution and diversity of Verrucomicrobia methanotrophs in geothermal and acidic environments. Environ Microbiol 16:1867–1878
Ettwig KF, Butler MK, Le Paslier D, Pelletier E, Mangenot S, Kuypers MM, Schreiber F, Dutilh BE, Zedelius J, de Beer D, Gloerich J, Wessels HJ, van Alen T, Luesken F, Wu ML, van de Pas-Schoonen KT, Op den Camp HJ, Janssen-Megens EM, Francoijs KJ, Stunnenberg H, Weissenbach J, Jetten MS, Strous M (2010) Nitrite-driven anaerobic methane oxidation by oxygenic bacteria. Nature 464:543–548
Whittenbury R, Phillips KC, Wilkinson JF (1970) Enrichment, isolation and some properties of methane-utilizing bacteria. J Gen Microbiol 61:205–218
Dedysh SN, Panikov NS, Tiedje JM (1998) Acidophilic methanotrophic communities from Sphagnum peat bogs. Appl Environ Microbiol 64:922–929
Dedysh SN (2009) Exploring methanotroph diversity in acidic northern wetlands: molecular and cultivation-based studies. Microbiology 78:655–669
Kip N, Ouyang W, van Winden J, Raghoebarsing A, van Niftrik L, Pol A, Pan Y, Bodrossy L, van Donselaar EG, Reichart G-J, Jetten MSM, Sinninghe Damsté JS, Op den Camp HJM (2011) Detection, isolation and characterization of acidophilic methanotrophs from Sphagnum mosses. Appl Environ Microbiol 77:5643–5654
Khmelenina VN, Kalyuzhnaya MG, Starostina NG, Suzina NE, Trotsenko YA (1997) Isolation and characterization of halotolerant alkaliphilic methanotrophic bacteria from Tuva soda lakes. Curr Microbiol 35:257–261
Heyer J, Berger U, Hardt M, Dunfield PF (2005) Methylohalobius crimeensis gen. nov., sp. nov., a moderately halophilic, methanotrophic bacterium isolated from hypersaline lakes of Crimea. Int J Syst Evol Microbiol 55:1817–1826
Sorokin DY, Jones BE, Kuenen GJ (2000) An obligately methylotrophic, methane-oxidizing Methylomicrobium species from highly alkaline environment. Extremophiles 4:145–155
Bowman J (2006) The methanotrophs – the families Methylococcaceae and Methylocystaceae. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E (eds) The prokaryotes: a handbook on the biology of bacteria, 3rd ed, vol 5. Springer, New York, рp 266–289
Heyer J, Galchenko V, Dunfield P (2002) Molecular phylogeny of type II methane-oxidizing bacteria isolated from various environments. Microbiology 148:2831–2846
Stott BM, Crowe MA, Mountain BW, Smirnova AV, Hou S, Alam M, Dunfield PF (2008) isolation of novel bacteria, including a candidate division, from geothermal soils in New Zealand. Environ Microbiol 10:2030–2041
Pol A, Barends TRM, Dietl A, Khadem AF, Eygensteyn J, Jetten MSM, Op den Camp HJM (2013) Rare earth metals are essential for methanotrophic life in volcanic mudpots. Environ Microbiol 16:255–264
Lidstrom ME (1988) Isolation and characterization of marine methanotrophs. Antonie Van Leeuwenhoek 54:189–199
Dunfield PF, Khmelenina VN, Suzina NE, Trotsenko YA, Dedysh SN (2003) Methylocella silvestris sp. nov. a novel methanotrophic bacterium isolated from an acidic forest cambisol. Int J Syst Evol Microbiol 53:1231–1239
Bothe HM, Møller-Jensen K, Mergel A, Larsen J, Jørgensen C, Bothe H, Jørgensen L (2002) Heterotrophic bacteria growing in association with Methylococcus capsulatus (Bath) in a single cell protein production process. Appl Microbiol Biotechnol 59:33–39
Kalyuzhnaya MG, Yang S, Rozova ON, Smalley NE, Clubb J, Lamb A, Nagana Gowda GA, Raftery D, Fu Y, Bringel F, Vuilleumier S, Beck DAC, Trotsenko YA, Khmelenina VN, Lidstrom ME (2013) Highly efficient methane biocatalysis revealed in a methanotrophic bacterium. Nat Commun 4:2785. doi:10.1038/ncomms3785
Wilkinson T, Topiwala HH, Hamer G (1974) Interactions in a mixed bacterial population growing on methane in continuous culture. Biotechnol Bioeng 16:41–59
Bussmann I, Rahalkar M, Schink B (2006) Cultivation of methanotrophic bacteria in opposing gradients of methane and oxygen. FEMS Microbiol Ecol 56:331–344
Koch IH, Gich F, Dunfield PF, Overmann J (2008) Edaphobacter modestus gen. nov., sp. nov., and Edaphobacter aggregans sp. nov., two novel acidobacteria isolated from alpine and forest soils. Int J Syst Evol Microbiol 58:1114–1122
Dedysh SN, Kulichevskaya IS, Serkebaeva YM, Mityaeva MA, Sorokin VV, Suzina NE, Rijpstra WI, Damste JS (2012) Bryocella elongata gen. nov., sp. nov., a novel member of subdivision 1 of the Acidobacteria isolated from a methanotrophic enrichment culture and emended description of Edaphobacter aggregans Koch et al. 2008. Int J Syst Evol Microbiol 62:654–664
Linton JD, Buckee JC (1977) Interactions in a methane-utilizing mixed bacterial culture in a chemostat. J Gen Microbiol 101:219–225
McDonald IR, Bodrossy L, Chen Y, Murrell JC (2008) Molecular ecology techniques for the study of aerobic methanotrophs. Appl Environ Microbiol 74:1305–1315
Sharp CE, Stott MB, Dunfield PF (2012) Detection of autotrophic verrucomicrobial methanotrophs in a geothermal environment using stable isotope probing. Front Microbiol 3:303. doi:10.3389/fmicb. 2012.00303
Auman AJ, Stolyar S, Costello AM, Lidstrom ME (2000) Molecular characterization of methanotrophic isolates from freshwater lake sediment. Appl Environ Microbiol 66:5259–5266
Svenning MM, Wartiainen I, Hestnes AG, Binnerup SJ (2003) Isolation of methane oxidizing bacteria from soil by use of a soil substrate membrane system. FEMS Microbiol Ecol 44:347–354
Amaral JA, Knowles R (1995) Growth of methanotrophs in methane and oxygen counter gradients. FEMS Microbiol Lett 126:215–220
Hoefman S, van der Ha D, Boon N, Vandamme P, de Vos P, Heylen K (2014) Customized media based on miniaturized screening improve growth rate and cell yield of methane-oxidizing bacteria of the genus Methylomonas. Antonie Van Leeuwenhoek 105:353–366
Dedysh SN, Dunfield PF (2011) Facultative and obligate methanotrophs: how to identify and differentiate them. In: Rosenzweig AC, Ragsdale SW (eds) Methods in enzymology, vol 495. Academic, Burlington, pp 31–44
Trotsenko YA, Murrell JC (2008) Metabolic aspects of aerobic obligate methanotrophy. In: Laskin AI, Sariaslani S, Gadd GM (eds) Advances in applied microbiology, vol 63. Elsevier, pp 183–229
Khadem AF, Pol A, Wieczorek A, Mohammadi SS, Francoijs K-J, Stunnenberg HG, Jetten MSM, Op den Camp HJM (2011) Autotrophic methanotrophy in Verrucomicrobia: Methylacidiphilum fumariolicum SolV uses the Calvin-Benson-Bassham cycle for carbon dioxide fixation. J Bacteriol 193:4438–4446
Vorob’ev AV, Dedysh SN (2008) Inadequacy of enrichment culture technique for assessing the structure of methanotrophic communities in peat soil. Microbiology 77:504–507
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Berlin Heidelberg
About this protocol
Cite this protocol
Dedysh, S.N., Dunfield, P.F. (2014). Cultivation of Methanotrophs. In: McGenity, T., Timmis, K., Nogales , B. (eds) Hydrocarbon and Lipid Microbiology Protocols. Springer Protocols Handbooks. Springer, Berlin, Heidelberg. https://doi.org/10.1007/8623_2014_14
Download citation
DOI: https://doi.org/10.1007/8623_2014_14
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-45178-6
Online ISBN: 978-3-662-45179-3
eBook Packages: Springer Protocols