Abstract
This chapter provides a comprehensive overview of the potential application of microbial fuel cell (MFC) technology for the bioremediation of sewage sludge. The abundance of sewage sludge and its associated contaminants presents a significant challenge to environmental sustainability, necessitating the development of innovative approaches for its treatment. The implementation of MFC technology offers a promising alternative approach that can simultaneously provide bioremediation and power generation benefits. The chapter highlights current research and studies on the utilization of MFC for sewage sludge treatment, including an overview of the mechanisms involved in the adaptation of the technology to address environmental issues associated with sewage sludge pollution. The chapter also discusses parameters for enhancing MFC performance, such as the combination of inoculum, substrate pretreatment, sludge concentration, and the effect of nitrate and sulfate. The earliest applications of MFC technology for sludge treatment are also discussed, including the configuration of the system, the use of sludge as a substrate, and the adjustment of pH to suit the system. Early MFC research also focused on nutrient recovery. Overall, the chapter aims to provide a comprehensive understanding of the potential contribution of MFC technology to sustainable wastewater management. By utilizing MFC technology for the bioremediation of sewage sludge, researchers can develop innovative solutions for addressing environmental challenges, thereby enhancing environmental sustainability.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Sewage sludge may be attributed to solid, semi-solid or liquid residue generated as the bulk of the residual material removed during the wastewater treatment process from society-based liquid wastes following aerobic biological treatment, with the potential to contain unfavourable trace pollutant contaminants. Periodic removal has been imperative in preventing excessive biomass concentration in the system or possible pass through rivers and other surface waters. As such, developed countries have outlined regulations for the safety of the public and the environment via each of the following methods [1].
-
1.
Application to the land as soil conditioner, or fertilizer in agricultural use
-
2.
Disposal to the sea
-
3.
Disposal on land at a surface disposal site
-
4.
Placing on municipal solid waste landfill unit
-
5.
Application as ‘bio-soil’, production for sale in marketplace, composting, land reclamation, etc.
-
6.
Incineration
-
7.
Sludge to energy.
Two main strategies for sewage sludge management involve disposal or reuse, such as for agricultural and for landscaping purposes [2]. However, even with reuse options available, there remain restrictions in place prior to application. With respect to current legislation, characterization, ecotoxicology and waste management routes, treatment and disposal remain the most popular approach for application. To assist in decision making, tools such as ‘end-of-waste criteria’ and ‘Life Cycle Assessment’ has been invaluable in proper assessment of the probable environmental, technical and economical evaluation between different systems. Sewage sludge treatment at wastewater treatment plant (WWTP) is seen in Fig. 1 [3].
Sewage sludge treatment processes at wastewater treatment plant. Reprinted with permission from [3] (CCBY)
2 Abundance of Sewage Sludge
Sewage sludge usually represents 1–2% of the treated wastewater [3]. Despite these low proportions, current production and subsequent abundance of sewage sludge cannot be understated. In excess of 10 million tonnes of dry solids (DS) of sewage sludge were produced in 26 EU Member States as of 2008 [4], while as per UN-Habitat’s statistics, the existing wastewater treatment plants (WWTPs) in the United States, for example, produce over 6.5 million tonnes of dry solids annually; while it is estimated to be around 2.0 and 3.0 Mt per annum produced in Japan and, respectively. Such figures are figures are naturally anticipated to further increase as applications of WWTP in developing countries continue to grow [5]. Table 1 exemplifies the production and disposal methods of selected countries as of 2012.
3 Characteristics of Sewage Sludge
In general, sewage sludge is a heterogenous mix of microorganisms, undigested organics such as paper, plant residues, oils, faecal material, inorganic materials and moisture [6]. Depending on stabilization processes, dewatered sewage sludge (dry) may contain an average of 50–70% organic matter, 30–50% mineral components (including 1–4% incorganic carbon), 3.4–4.0% N, 0.5–2.5% P and significant amount of additional nutrients [7,8,9]. Organic matter present within sewage sludge mineralizes quickly owing to the small content of lignin or cellulose, with following rapid degradation potentially generating a peak in the nitrate and pollutant concentration in soil.
Presence of large concentrations of nitrogen and phosphorus makes it suitable for application as fertilizer for plants [7]. However, presence of numerous contaminants, both inorganic (including heavy metals) and organic (such as polycyclic aromatic hydrocarbons (PAHs), polychlori-nated biphenyls (PCBs), adsorbable organohalogens (AOX)), surfactants, pesticides, hormones, pharmaceuticals, nanoparticles and several others [10]. In addition to this, several pathogenic species of living organisms such as bacteria, viruses and protozoa as well as other parasitic organisms present a health hazard for humans, animals and plants [11, 12]. With appropriate sludge treatment (such as lime treatment), the number of pathogenic and parasitic organisms within sludge may be sufficiently reduced prior to application into land, thereby mitigating the health risks present [13, 14]. Different types of sewage sludge and their inherent characteristics are seen in Table 2.
4 Case for Energy Recovery
The finiteness of fossil-based energy resources continues to be a concern as globalization and localization of technologies and manufacturing capacities are coupled with a high speed of energy consumption growth [15]. Globally, rising energy consumption has been noticeable at an increasingly rapid rate, within a 10-year period, from 8,588.9 million tonnes oil equivalent (Mtoe) in 1995 to 13,147.3 Mtoe in 2015, with fossil fuels providing approximately 86.0% of the global total energy needs [16]. In BP Statistical Review of World Energy (2020), China was highlighted as the dominant country pushing global energy markets with renewables recording the biggest percentage growth yet in primary energy, with levels of power generation exceeding nuclear energy for the first time. On a more localized perspective, wastewater treatment technologies suffer from drawbacks such as energy demand, large amount of residual generation and low effectiveness in catching energy potential from wastewater [17]. Usage of inexpensive, renewable energy from sludge is expected to become more essential as the cost of sewage treatment continues to rise [15].
Evaluation of sludge as a source for energy recovery involves consideration of its respective compositions, of which the energy content of sludge is inherent in the volatile solids, which is subdivided into two components: readily degradable (50% in primary sludge and 25% in WAS) and not readily degradable (30% in primary sludge and 55% in WAS) [1]. Analysis has revealed the energy contained within wastewater sludge is substantial, reaching as much as 3–10 times higher than the energy required for wastewater treatment itself [18]. As per the sludge characteristics mentioned prior, considerations must be taken when deciding if sewage sludge is applicable to be salvaged for energy recovery, along with the approach to be considered.
As a conventional approach, sewage sludge is often incinerated with the exhaust gases and ashes treated, albeit its complexity and costs make it applicable only in large plants. Another established approach involves biogas production through anaerobic digestion of sewage sludge. Here, organic matter in sludge is converted by bacteria into a mixture of methane (60–60%), carbon dioxide (35–40%) and trace gases, with the resultant biogas directly applied in combined heat and power (CHP) systems for utilization in sewage treatment plants. This approach also reduces the solid content of sludge by up to 30%, further reducing the associated energy costs involved in transportation [15]. Low sludge yield may also prove to be advantageous given its accounts for approximately 25–65% of total plant operating costs [19].
5 Necessity for Bioremediation
With the potential untapped resources present in sewage sludge, many countries have recognized the value of sludge by-product for substrate in fertilization of agricultural lands as well as remediation of polluted areas. However, the potential harms must also be recognized as sewage sludge applications on agricultural land might lead to the dispersal of numerous unwanted constituents on soils possibly used for food production. Such undesirable contaminants (potentially toxic elements (PTE) such as metals, trace organic compounds (TrOC), and pathogenic organisms) may pose sanitary and environmental risks. Processes to reduce pathogens involve composting, autothermal thermophilic aerobic digestion, alkaline stabilization, pasteurization, incineration, thermal drying and wet oxidation while approaches to remove heavy metal presence include activated sludge, aerated lagoon and facultative pond to mention a few, each with differing importance in terms of implementation and operation [20].
6 Microbial Fuel Cells
As the search for renewable energy gains pace, microbial fuel cells (MFC) have emerged as a rapidly expanding field of science and technology which associate biological catalytic redox activity with abiotic electrochemical reactions and physics. By definition, MFCs form a branch of biological fuel cells, where electroactive microbes are utilized within devices, for the degradation of organics and subsequent conversion from chemical to electrical energy, by means of electrochemical reactions [21]. MFCs have been recognized for its versatility in providing direct power, as well as feedstock treatment, nutrient recovery and sensing for continuous parallel observation of processed substrates [22].
7 Mechanism of MFC
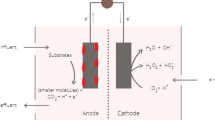
At its basic form, MFCs are made up of an anodic and a cathodic compartment separated by the presence of an ion exchange membrane (IEM), which functions in preventing the migration of electrolytes between chambers [23]. Separation of environmental conditions allows for maximization of potential differences between both anodic and cathodic electrodes, thereby optimizing power generation [24]. Organic matter is supplemented to the anode compartment to act as fuel once oxidized as substrate by anaerobic microorganisms, liberating electrons (e−) and protons (H+) [25]. In the cathode compartment, electrons are released to the cathode electrode surface and combined with protons and oxygen via reduction to form water [26]. Figure 2 outlines the basic configuration of an MFC.
Basic configuration of microbial fuel cells. Reprinted with permission [27] (CCBY)
8 Advantages of MFC
As an alternative power generator, microbial fuel cells are advantageous in comparison to conventional fuel cells by having a breadth of materials, substrates and system architectures applicable to bacteria for bioenergy production, albeit at relatively low power levels [28]. MFCs also have the potential as sustainable long-term power applications though potential issues in health and safety following bacterial usage must also be addressed prior [29].
9 MFC in Wastewater Treatment
10 Integration of MFC into Wastewater Treatment
Integration of MFC reactors into existing wastewater treatment plant trains can be done as a replacement to the existing biological treatment unit, such as an activated sludge reactor, for accomplishing carbon oxygen demand (COD) removal, electricity production and reduction in sludge production relative to those of an aerobic process such as activated sludge. However, a secondary process would be required to further remove COD to levels acceptable for discharge, as the current generation is minimal once the COD is less than ∼100 − 150 mg/L. [33]. While the activated sludge process is currently commonly applied for the treatment of domestic and industrial wastewater, there remains inherent limitations within such as high energy consumption and waste sludge production [34]. An adaptable and sustainable technology capable of simultaneous wastewater treatment and resource recovery is therefore favoured [35].
11 Advantages of MFC Application in Wastewater Treatment
MFCs can prove advantageous for wastewater treatment by (1) direct conversion of chemical energy within substrates to electricity, (2) relatively lower amount of activated sludge production compared to other methods, (3) environmentally friendly, (4) additional gas treatment not required and (5) aeration made unnecessary. MFC application also allows for use of various types of wastewaters [18]. Direct conversion into electricity by MFC bypasses the need for separation, purification and conversion of the energy products which along with the mild operating conditions suggest as to a relatively more environmentally friendly approach [36].
Simple substrates such as sucrose, glucose and protein to undefined and complex substrates such as wastewater from domestic and municipal, brewery, dairy, pharmaceutical, food processing, agro-processing livestock, petroleum and paper recycling industry may be utilized with MFC technology. Operation of MFC differs from conventional bioremediation as organic compounds are converted to H2O and CO2 under aerobic conditions [37]. As the example with high strength wastewater, with the integration of MFC and bioremediation, synergy between both processes may result in enhanced energy and resource recovery with higher pollutant removal [38]. In certain cases, sludge disposal rate and treatment efficiency attained with MFC technology is comparable and even surpassed conventional technologies [39].
12 Proposed Reactions from Breakdown of Chemical Composition of Sludge
Utilization of sewage sludge as inoculum in MFCs introduces a mixed culture of microorganisms for substrate consumption and bioelectricity generation. As bioelectricity is generated with a transfer of electrons, relating the phenomenon towards the understanding of metabolism of microorganisms (in aerobic and anaerobic conditions) is considered essential.
Aerobic bacteria prevent the transfer of electrons from organic compounds to electron acceptors such as O2, while on the other hand, anaerobic bacteria in the absence of O2 utilize alternative electron acceptors such as nitrate and solid electrodes. Reduction of electrons occurs on the extracellular wall where the cytochromes reduce extracellular electrons from the substrate, upon which energy in the form of ATP can be produced for bacterial growth and reproduction.
In the respiration pathway of microorganisms, the metabolism involved utilizes nicotinamide adenine dinucleotide (NADH) as the energy source. By relating the biological standard potential (EOJ’[V]) to NADH and with O2 as an electron acceptor, a theoretical potential difference may be constructed, as seen in Eqs. 1, 2 and 3 [40].
In the absence of electron acceptor, electrons from the microbes are transferred directedly to the surface of the anode, which may prove complicated as several surfaces are incapable to do so.
Electricity is generated with the use of substrates as fuel for microbes in the anode chamber, upon which electrons released are passed to the cathode to reduce the electron acceptors (O2). Protons from the anode chamber are then transferred through a membrane to the anolyte for reduction. Hence, the substrate of choice must be considered in determining the electricity output from the MFC. An example is as given by Das and Mangwani [40] on the proposed consumption of acetate and sucrose where present, as seen in Eqs. 4, 5 and 6, 7, respectively.
13 Application of MFC for Bioremediation and Electricity Generation
Substrates are significant in any biological processes as carbon and energy source. Key towards determining the efficiency and economic viability of converting organic wastes as fuel to bioenergy are the characteristics, chemical composition and concentrations of the material to be used [41]. In the process of wastewater treatment and subsequent sludge stabilization, a significant amount of energy and high cost is required. However, this pales in comparison to the energy content within wastewater which is approximately 9 times higher than the energy applied for treatment [42]. Given the abundance of energy contained within sludge following treatment, it has been argued that MFCs are essential in the extraction of energy in organics for possible reduction of energy demand and expenses in a wastewater treatment plant [43]. Table 3 outlines several selected applications of sewage sludge as substrate in MFCs for bioremediation and electricity generation.
14 Earliest Sludge Treatment Using MFC Technology
Application of sludge in MFC began with work by Dentel et al. [44], which represents the earliest attempt at the conversion of organic matters in digested sludge to electricity under ambient temperature, atmospheric pressure and neutral pH upon which a maximum electrical current of approximately 0.065 mA and a maximum voltage of 0.517 V, while concluding that current is restricted to degree of degradation of organic matter. 16s rDNA analysis identified the presence of Geobacter as a natural constituent of microbial population in the anaerobic sludge utilized albeit not directly involved in electricity generation, by the lack of biofilm formed. Instead, it was suggested that more than one or more redox mediator may be involved in electron cycling between the anode and the solution, carried out by strains such as Aeromonas, Geovibrio, Clostridium and Desulfotomacum reducens, albeit none were positively identified in this study.
14.1 MFC Configuration
Another study by Hu [43] introduced the application of a baffle for mixing, in a membraneless, single chamber MFC with anaerobic sludge as fuel. Such a design was constructed to reduce mixing in the vicinity of the cathode and attain thick biofilm formation (>1 mm) on the cathode upon the addition of anaerobic/biomass sludge, thereby contribute to maintaining anaerobic conditions inside the reactor by minimizing oxygen diffusion through the cathode. Initial recorded electricity generated was low at 0.3 mW/m2 under endogenous decay conditions, which suggests anaerobic sludge to be difficult for conversion into electricity. Maximum power of 161 mW/m2 instead was only attained and attributed to glucose degradation as fuel. The baffle-chamber membraneless MFC was concluded to be effective in restricting fluid mixing within the anode chamber, with inoculation of the anaerobic sludge at a final concentration of approximately 4000 mg CODL−1 promoted thick (>1 mm, via COD analysis) biofilm formation on the cathode. Presence of strict anaerobic conditions allows for fuel (glucose) retention that would have been consumed aerobically for prolong electricity generation.
14.2 Sludge as Substrate and Inoculum
Work by Zhihua Liu et al. [45] focused on the application of surplus sludge as both inoculum and substrate in a single chamber floating-cathode MFC for electricity production. Maximum voltage of 440.7 mV was obtained with external resistance of 1000 Ω [44]. A periodic time of MFC was divided into 4 phases as seen in Fig. 3, beginning with a rapid fall phase due to the mass decomposition of biodegradation matter such as volatile fatty acid.
Electricity generation over time in steady state from surplus sludge-based MFC. Phase I: rapid fall phase; Phase II: stationary phase; Phase III: fall phase; Phase IV: stationary phase of low voltage. Reprinted with permission [44] (CCBY)
Following this, SCOD concentration of substrate stabilizes during the stationary phase and upon reaching stationary phase of low voltage, the SCOD concentration was found to be high still, a finding contrary to that argued by Dentel et al. [44]. Across the four phases, output voltage range of 150–300 mV persisted for 107 h in stationary phase. Recorded maximum power density was 220.7 mWm−2 and internal resistance of 368.13 Ω in the MFC.
In another application of sewage sludge as both inoculum and substrate, a study conducted by Jiang et al. [31] constructed a two-chambered MFC with potassium ferricyanide as electron acceptor. Here, stable electrical power production during operation for 250 h was demonstrated along with TCOD removal by 46.4% from the initial TCOD of 10,850 mg/L. Several parameters such as substrate concentration, cathode catholyte concentration and anodic pH were also tested to determine their effects on bioremediation and electrical output upon which although the aforementioned parameters did not affect significantly, power production was found to be closely related to soluble chemical oxygen demand (SCOD) of sludge. Recognizing immobilization of organic matter within sewage sludge constitutes organic matter hydrolysis as the rate-limiting step [49], further ultrasonic pre-treatment of sludge applied also managed to increase TCOD removal rate albeit without additional significant enhancement on power output.
Another study by Nandy et al. [49] utilized activated sludge as inoculum and monitored for remediation with the application of Fe2O3 coating on carbon electrodes as an alternative. Cell C labelling MFC with coated anode and cathode (30.18 mW.m−2) exhibited 78% higher power output in comparison to those in Cell A with non-coated carbon electrodes (6.72 mW.m−2), albeit still lower than that of Cell B, containing Fe2O3 coated carbon anode with Pt coated carbon cathode (73.16 mW.m−2). Microbial community analysis on both the biofilm formed and liquid electrolyte for all cells highlighted the presence of bacterial Peptostreptococcaceae as dominant in Cell A, but shifted through Cells B and C to reduced Firmicutes population, and enhanced Gammaproteobacteria and methanogens. COD removal efficiency was recorded at about 60% for cell B, 51% for cell C and 39% for cell A.
14.3 Parameter Adjustment (pH)
Work carried out by Yuan et al. [46] elucidated as to the effects of pH on electricity production from sludge-based MFC. Here, an insert-type air-cathode microbial fuel cell was constructed as pH adjustment to alkaline pH was conducted to enhance sludge substrate bioavailability and suppression of methane production. Maximum power density of 73 ± 5 mWm−2 attained from sludge at pH 10, which was higher than that of 33 ± 3 mWm−2 at pH 8 and 4 ± 0.5 mWm−2 at pH 6. Soluble chemical oxygen demand (SCOD) which was attributed to protein and carbohydrate content meanwhile increased with increasing pH as the alkaline condition proved favourable for enhancing the substrate bioavailability of sludge by assisting in the dissolution of insoluble organics into a soluble form. TCOD reduction from 13,167 to 6280 mg L−1 at pH 10.0, representing a 53% TCOD removal efficiency, was also higher than those at pH 8.0 and 6.0 (42% and 20% TCOD removal, respectively). Finally, increased coulombic efficiency from increased pH was attributed to complete methane production suppression under such conditions, thereby suggesting alkaline pH to be a successful approach for the enhancement of electricity from MFC.
14.4 Nutrient Recovery
Another example of MFC application involves the recovery of value-added elements from sewage sludge, using MFC. Work by Fischer et al. [47] focused on the recovery of orthophosphate, of which phosphor, an essential element sought globally for agricultural and industrial purposes may be extracted. Here, MFC has proven advantageous for application in ambient temperatures while serving as a power source delivering not only electrons, but also the needed protons to reduce electrochemically insoluble FePO4. Liberated electrons are able to reduce the iron cations and charges are then replaced by protons, which results in orthophosphate (H3PO4, \({\text{H}}_{2} {\text{PO}}_{4}^{ - }\), \({\text{HPO}}_{4}^{2 - }\), and \({\text{PO}}_{4}^{3 - }\)) motility into the aqueous supernatant solution.
15 Long-Term Application
As a substrate to MFC, another study by Ge et al. [48] sought to investigate the long-term performance of sludge treatment by application of two MFCs, each with primary sludge and digested sludge respectively, over a period approaching 500 days. In preliminary Phase I, MFC fed with primary sludge managed to remove 69.8 ± 24.1% of total chemical oxygen demand (TCOD) and 68.4 ± 17.9% of volatile suspended solids (VSS); while the MFC with digested sludge achieved 36.2 ± 24.4% of TCOD and 46.1 ± 19.2% of VSS reduction, respectively. Further application in Phase II as a two-stage system recorded TCOD removal by 60% and VSS removal by 70% from the primary sludge. Recorded energy obtained from primary sludge MFC and digested sludge MFC were up to 1.43 kWh/m3 from a primary sludge or 1.8 kWh/m3, respectively. As energy analysis revealed electrical energy generated from MFC was minimal in proportion of total energy compared to energy released as methane gas, MFCs were suggested as a polishing step to effluent from anaerobic digesters, as opposed to energy recovery from primary sludge.
16 Pretreatment Method Prior MFC Usage
Application of pre-treatment methods on sludge substrate was also conducted with the aim of enhancing dissolved organic concentrations [41, 50]. Work by Jiang et al. [31] mentioned prior applied sonication of sewage sludge [31] and in work by Abourached et al. [41], fermentation of sewage sludge was applied as pre-treatment within a novel cloth electrode assembly MFC. Maximum power density of 1200 mW/m2 was achieved after a fermentation time of 96 h. At the time, the recorded power density was an enhancement by 275% over MFCs reported prior. Even then, given known energy efficiency of MFCs treating sludge is only at 11.5%, MFCs represent an exciting modification to existing wastewater treatment infrastructure. Table 3 shows the application of MFC for bioremediation and electricity generation.
17 Sludge as Inoculum
Applications of sludge in MFC studies involve several perspectives as seen in Table 4. A popular approach is to utilize sludge as mixed culture inoculum within the anode compartment. MFCs inoculated with mixed cultures have been argued to produce significantly greater power densities than those of pure cultures [51]. Within anaerobic sludge applied as mixed culture inoculum, the simultaneous presence of electrophiles/anodophiles and groups utilizing natural mediators within the same chamber allows for wider substrate utilization [29]. Table 4 details several strains of note identified present within inoculum sludge. As an undefined inoculum, recycled activated sludge point of an aeration tank from a local municipal wastewater treatment plant was used by Liang et al. [52] in efforts to elucidate the composition and distribution of internal resistance from three separate MFC configurations. Another study by Zhidan Liu et al. [53] used mesophilic anaerobic sludge to focus on identifying the effect of operational performance and electrical response of a mediator-less MFC fed with either acetate as carbon-rich substrate or protein-rich synthetic wastewater in fed batch mode.
18 Bioremediation of Heavy Metals
For this application, it is worth noting that metal ions present in wastewater do not biodegrade into harmless end products, thereby necessitating special approaches for treatment. With several metal ions having high redox potentials, it may therefore be advantageous for reduction and precipitation in MFC [56]. If implemented properly, MFCs can therefore serve not only as remediation of wastewater but along with concomitant metal recovery too [57].
To this aim lies the works of Li et al. [58] which utilized sewage sludge inoculum as a means of disposing Cr6+ from real electroplating wastewater with simultaneous electricity generation, using a dual chamber MFC. Chromium removal and power density were optimum at pH 2 and application of graphite paper as cathode electrode. Maximum power density of 1600 mW/m2 was generated at a columbic efficiency of 12% at initial Cr6+ concentration of 204 ppm, with a further rise in Cr6+ capable of enhancing power density. Removal efficiency was at 99.5% Cr6+ and 66.2% total Cr through the reduction of Cr2O72− to Cr2O3 precipitating on the surface of cathode electrode.
Another example involves the removal of silver ions from wastewaters, as demonstrated by Choi and Cui [59]. Using sludge as inoculum, following 8 h reaction, efficiency of silver metal recovery was recorded to be as high as 99.91–98.26% across initial concentration ranges of 50 ppm to 200 ppm. Energy output was calculated to be at a rate of 69.9 kg of silver per KWh of energy output, with maximum power attained of approximately 4.25 W/m2, maximum voltage of 0.749 V, maximum current density of 5.67 A/m2 and a fill factor of 0.626 was achieved at 1000 ppm initial silver ion concentration.
In another study, Cu2+ was targeted for recovery with excess sludge as the anodic substrate within a MFC and CuSO4 solution as the catholyte. Here, Cu2+ was proven to be a viable cathodic electron acceptor, with a stable voltage output of 0.478 V and a maximum power density of 536 mW/m3 obtained at external resistance of 1000 Ω and Cu2+ concentration of 6400 mg/L. For Cu2+ removal from wastewater, 97.8% removal efficiency was achieved following 288 h of operation with no external resistance and initial Cu2+ concentration of 1000 mg/L. Final products attained were dependent on cathodic reducibility, with Cu2+ primarily deposited as Cu20 and a small part as Cu4(OH)6SO4. Excess sludge in particular was deemed crucial for supporting long-term operations, with the acclimation stage of exoelectrogenic bacteria on the anode key in determining MFC performance and cathodic reduction of Cu2+.
19 Improvement Parameter for Enhancing MFC Performance
As an inoculum, numerous efforts have since been conducted for the improvement of sludge inoculum to this purpose.
19.1 Combination of Inoculums
Sun et al. [60] investigated the effect of different combinations of sludge inoculums consisting of aerobic sludge (AES), anaerobic sludge (ANS) and wetland sediment (WLS) towards overall MFC performance in wastewater treatment. To this, it was found that the application of multiple sludge inoculums was advantageous whereby MFC inoculated with ANS + WLS produced maximal power density of 373 mW/m2 with a low internal resistance of 38 Ω. A similar observation was made with combinations of ANS + AES and ANS + AES + WLS in comparison to single sludge inoculation, with the former exhibiting the highest Coulombic efficiency. COD removal of more than 92% was recorded irrespective of the membranes and inoculums applied.
19.2 Comparison of Inoculums
In another study by Baranitharan et al. [61] a developed control inoculum (CI) was compared to that of anaerobic sludge as inoculum for use in MFC with palm oil mill effluent (POME). CI consisted of predominant microorganisms such as Pseudomonas aeruginosa, Azospira oryzae, Acetobacter peroxydans and Solimonas variicoloris isolated from palm oil anaerobic sludge in combination with a biofilm of MFC anode operated prior with anaerobic sludge. Maximum power density of MFC utilizing CI was found to be twice higher and with enhanced maximum Coulombic efficiency, albeit noticeable lower COD removal of about 32%, possibly due to the absence of necessary fermentative microorganisms within the CI for POME utilization.
19.3 Substrate Pre-Treatment
Xie et al. [62] have argued for pre-treatment of parent inoculum in anode chamber of MFC as a means of selective enrichment of a specific group of bacteria. Understanding the composition of bacterial associations to be dependent on substrate (wastewater) composition and of the symbiotic relationships within a given population, application of low-frequency ultrasonication for a short duration was proposed as stimulant, by promoting enzyme activity, cell growth and cell membrane permeability. Under stressful conditions such as high temperatures, extreme acidity and alkalinity, methanogenic bacteria without the capability of forming protective spores and with lower growth rate than hydrogen producing and electrogenic bacteria are at a disadvantage, thereby less likely to survive and thus leave the latter as dominant species in mixed culture of anaerobic sludge [63, 64]. In another study conducted by More and Ghangrekar [64], a low-frequency ultrasound pre-treatment was applied to anaerobic sludge inoculum to be used in MFC based on synthetic wastewater. Maximum power density was subsequently achieved following ultrasonication at 40 kHz, 120 W for 5 min, which was 2.5 times higher than that of untreated inoculum sludge along with enhanced COD removal by 14%.
19.4 Sludge Concentrations
Another study conducted by Khan et al. [65] applied sludge of different concentrations up to 200 ml/L to study the resultant effects on current generation and COD removal. With constant 100 Ω resistance applied, highest current generated of 314 µA was from MFC batch utilizing 200 ml/L of drainage sludge after 35 h with concomitant highest degree of COD removal of up to 78%. Under these conditions, the maximum power density was 15.12 mW/m2 with current density at 97.34 mA/m2. This was further tested by Amin et al. [66] as electricity generation by a membraneless MFC operating in continuous mode was modified in several parameters, including sludge concentration. Sludge feed rate of 150 mL/L showed best current generation, with a maximum value of 250 µA after 24 h of operation, albeit with less stability than that of lower concentrations. The results thereby conclude that for a given fixed amount of substrate and salt, current generation can be expected to increase with the increase in sludge concentration, attributed to higher amounts of electricity generating microorganism.
19.5 Effect of Nitrate and Sulphate
In replicating concentrations of elements commonly found in industrial wastewater for application with MFCs, Seo et al. [35] sought to study the effect of adding nitrate and sulphate independently towards the bacterial community and performance of a membraneless, single chamber microbial fuel cell. With anaerobic sludge from sewage treatment plant used as inoculum, bacteria of the genus Clostridium, Petrimonas and Rhodocyclaceae were found in abundance prior to addition.
Nitrate addition resulted in consistent Clostridium proportions, albeit with lowered Petrimonas (20 → 6%) and concomitant increase by Rhodocyclaceae (6.2 → 17.1%) and Castellaniella (0.4 → 4.1%) on the anode surface. On the cathode surface, nitrate addition resulted in Rhodocyclaceae (25.5%) being most abundant, followed by Clostridium (10.4%), Castellaniella (7.3%) and Petrimonas (1.5%). Known denitrifying ability of Rhodocyclaceae suggests for both anodic and cathodic denitrification function in MFC with its relative larger presence on the cathode biofilm as opposed to the anode and other competing denitrifying bacteria to be most suitable. Castellaniella instead assists in autotrophic denitrification of MFC by alleviating build-up of intermediates such as nitrite.
Addition of sulphate instead resulted in increase of Clostridium (23.1 → 43.2%) and decrease of Petrimonas (20.0 → 5.5%) proportions on the anode surface. A different proportion was noticeable on the cathode as Desulfovibrio (32.9%) increased significantly to abundance, followed by Clostridium (12.9%), Petrimonas (5.0%) and Thiobacillus (2.4%). Desulfovibrio and Thiobacillus function simultaneously as sulphate reducer and sulphide oxidizer respectively, which may explain the lower COD removal efficiencies as compared to that achieved following nitrate addition. Overall, nitrate addition resulted in higher electrical performances and higher nitrate removal efficiency (93%) while the opposite occurred following sulphate addition with concomitant lower sulphate removal efficiency (17.6%).
References
Zsirai I (2011) Sewage sludge as renewable energy. J Residuals Sci Technol 8(4):165–179. https://doi.org/10.13140/RG.2.1.1939.0483
Grobelak A, Czerwińska K, Murtaś A (2019) General considerations on sludge disposal, industrial and municipal sludge. In: Industrial and municipal sludge: emerging concerns and scope for resource recovery. Butterworth-Heinemann, Oxford, pp 135–153. https://doi.org/10.1016/B978-0-12-815907-1.00007-6
Kacprzak M et al (2017) Sewage sludge disposal strategies for sustainable development. Environ Res 156:39–46. https://doi.org/10.1016/j.envres.2017.03.010
Guangyin Z, Youcai Z (2017) Sewage sludge generation and characteristics. In: Pollution control and resource recovery: sewage sludge. Butterworth-Heinemann, Oxford, pp 1–11. https://doi.org/10.1016/B978-0-12-811639-5.00001-2
RPA, WRC, and milieu (2008) Environmental, economic and social impacts of the use of sewage sludge on land final report Part II: report on options and impacts, p 158
Rorat A, Courtois P, Vandenbulcke F, Lemiere S (2019) Sanitary and environmental aspects of sewage sludge management. In: Industrial and municipal sludge: emerging concerns and scope for resource recovery. Butterworth-Heinemann, Oxford, pp 155–180. https://doi.org/10.1016/B978-0-12-815907-1.00008-8
Tyagi VK, Lo SL (2013) Sludge: a waste or renewable source for energy and resources recovery? Renew Sustain Energ Rev 25(71):708–728. https://doi.org/10.1016/j.rser.2013.05.029
Fytili D, Zabaniotou A (2008) Utilization of sewage sludge in EU application of old and new methods—a review. Renew Sustain Energy Rev 12(1):116–140
Samolada MC, Zabaniotou AA (2014) Comparative assessment of municipal sewage sludge incineration, gasification and pyrolysis for a sustainable sludge-to-energy management in Greece. Waste Manage 34(2):411–420. https://doi.org/10.1016/J.WASMAN.2013.11.003
Siebielska I (2014) Comparison of changes in selected polycyclic aromatic hydrocarbons concentrations during the composting and anaerobic digestion processes of municipal waste and sewage sludge mixtures. Water Sci Technol J Int Assoc Water Pollut Res 70(10):1617–1624. https://doi.org/10.2166/WST.2014.417
Fijalkowski KL, Kacprzak MJ, Rorat A (2014) Occurrence changes of Escherichia coli (including O157:H7 serotype) in wastewater and sewage sludge by quantitation method of (EMA) real time—PCR. New pub: Balaban 52(19–21):3965–3972. https://doi.org/10.1080/19443994.2014.887499
Kacprzak M, Stan´czyk E, Stan´czyk-Mazanek S, (2003) Changes in the structure of fungal communities of soil treated with sewage sludge. Biol Fertil Soils 38:89–95. https://doi.org/10.1007/s00374-003-0633-2
Farzadkia M, Bazrafshan E (2014) Lime stabilization of waste activated sludge. Health Scope 3:1–5
Olszewski JM, Lozano N, Haines C, Rice CP, Ramirez M, Torrents A (2013) The effect of liming on antibacterial and hormone levels in wastewater biosolids. J Environ Sci Health. Part A, Toxic/Hazard Subst Environ Eng 48(8):862–870. https://doi.org/10.1080/10934529.2013.761488
Dong K (2020) How renewable energy consumption lower global CO2 emissions? evidence from countries with different income levels, vol 2017, pp 1665–1698. https://doi.org/10.1111/twec.12898.
BP, “BP Statistical Review of World Energy 2020,” 2020
NACWA (2010) Renewable energy resources: banking on biosolids, p 23
Gude VG (2018) Wastewater treatment in microbial fuel cells—an overview. J Clean Prod 122:287–307. https://doi.org/10.1016/j.jclepro.2016.02.022
Katuri KP, Scott K, Head IM, Picioreanu C, Curtis TP (2011) Microbial fuel cells meet with external resistance. Bioresour Technol 102(3):2758–2766. https://doi.org/10.1016/j.biortech.2010.10.147
Andreoli CV, von Sperling M, Fernandes F (2007) Sludge treatment and disposal, vol 1, IWA Publishing. https://doi.org/10.2166/9781780402130
Santoro C, Arbizzani C, Erable B, Ieropoulos I (2017) Microbial fuel cells: from fundamentals to applications: a review. J Power Sources 356:225–244. https://doi.org/10.1016/j.jpowsour.2017.03.109
Gajda I, Greenman J, Ieropoulos IA (2018) Recent advancements in real-world microbial fuel cell applications. Curr Opin Electrochem 11:78–83. https://doi.org/10.1016/j.coelec.2018.09.006
Ho NAD, Babel S, Sombatmankhong K (2018) Bio-electrochemical system for recovery of silver coupled with power generation and wastewater treatment from silver (I) diammine complex. J Water Process Eng 23:186–194. https://doi.org/10.1016/j.jwpe.2018.04.001
Fornero JJ, Rosenbaum M, Angenent T (2010) Electric power generation from municipal, food, and animal wastewaters using microbial fuel cells. Electroanalysis 22(7–8):832–843. https://doi.org/10.1002/elan.200980011
Omeroglu S, Sanin FD (2016) Bioelectricity generation from wastewater sludge using microbial fuel cells: a critical review. Clean Soil Air Water 44(9999):1–9. https://doi.org/10.1002/clen.201500829
Ucar D, Zhang Y, Angelidaki I, Franks AE (2007) An overview of electron acceptors in microbial fuel cells. Front Microbiol 8:643. https://doi.org/10.3389/fmicb.2017.00643
Logan BE (ed) (2008). Wiley, Hoboken, NJ
Du Z, Li H, Gu T (2007) A state of the art review on microbial fuel cells: a promising technology for wastewater treatment and bioenergy. 25:464–482. https://doi.org/10.1016/j.biotechadv.2007.05.004
Ahn Y, Zhang F, Logan BE (2014) Air humidity and water pressure effects on the performance of air-cathode microbial fuel cell cathodes. J Power Sour 247:655–659. https://doi.org/10.1016/j.jpowsour.2013.08.084
Feng C, Huang L, Yu H, Yi X, Wei C (2015) Simultaneous phenol removal, nitrification and denitrification using microbial fuel cell technology. Water Res 76:160–170. https://doi.org/10.1016/J.WATRES.2015.03.001
Jiang J, Zhao Q, Zhang J, Zhang G, Lee D (2009) Electricity generation from bio-treatment of sewage sludge with microbial fuel cell. Bioresour Technol 100(23):5808–5812. https://doi.org/10.1016/j.biortech.2009.06.076
Logan BE, Wallack MJ, Kim K, He W, Feng Y, Saikaly PE (2015) Assessment of microbial fuel cell configurations and power densities. Environ Sci Technol Lett 2:206–214. https://doi.org/10.1021/acs.estlett.5b00180
Fazal U et al (2021) Microbial fuel cell: study of bioresource potential of dairy effluent and Associated Process Limitations. Research Square, pp 1–17
Shamsudin NA, Sabri MNIM, Tajarudin HA, Mokhtar AMA, Makhtar MMZ (2022) The future promising alternative renewable energy from microbial fuel cell BT. In: Yaser AZ, Tajarudin HA, Embrandiri A (eds) Waste management, processing and valorisation. Springer, Singapore, pp 235–263. https://doi.org/10.1007/978-981-16-7653-6_13
Seo Y, Kang H, Chang S, Lee Y, Cho K (2017) Toxic/hazardous substances and environmental engineering effects of nitrate and sulfate on the performance and bacterial community structure of membrane- less single-chamber air-cathode microbial fuel cells. J Environ Sci Health, Part A 53(1):13–24. https://doi.org/10.1080/10934529.2017.1366242
Rajasulochana P, Preethy V (2016) Comparison on efficiency of various techniques in treatment of waste and sewage water—a comprehensive review. Resource-efficient Technol 2(4):175–184. https://doi.org/10.1016/J.REFFIT.2016.09.004
Vu HT, Min B (2019) Integration of submersible microbial fuel cell in anaerobic digestion for enhanced production of methane and current at varying glucose levels. Int J Hydrogen Energy 44(14):7574–7582. https://doi.org/10.1016/J.IJHYDENE.2019.01.091
Wang Z, Mei X, Ma J, Wu Z (2012) Recent advances in microbial fuel cells integrated with sludge treatment. Chem Eng Technol 35(10):1733–1743. https://doi.org/10.1002/ceat.201200132
Munoz-Cupa C, Hu Y, Xu C, Bassi A (2021) An overview of microbial fuel cell usage in wastewater treatment, resource recovery and energy production. Sci Total Environ 754:142429. https://doi.org/10.1016/J.SCITOTENV.2020.142429
Das S, Mangwani N (2010) Recent developments in microbial fuel cells. J Sci Ind Res 69:727–731
Abourached C, Lesnik KL, Liu H (2014) Enhanced power generation and energy conversion of sewage sludge by CEA—microbial fuel cells. Bioresour Technol 166:229–234. https://doi.org/10.1016/j.biortech.2014.05.027
Ting CH, Lee DJ (2007) Production of hydrogen and methane from wastewater sludge using anaerobic fermentation. Int J Hydrogen Energy 32(6):677–682. https://doi.org/10.1016/J.IJHYDENE.2006.06.063
Hu Z (2008) Electricity generation by a baffle-chamber membraneless microbial fuel cell. J Power Sour 179(1):27–33. https://doi.org/10.1016/j.jpowsour.2007.12.094
Dentel SK, Strogen B, Chiu P (2004) Direct generation of electricity from sludges and other liquid wastes. Water Sci Technol 50(9):161–168. https://doi.org/10.2166/wst.2004.0561
Liu Z et al (2009) Production of electricity from surplus sludge using a single chamber floating-cathode microbial fuel cell. Water Sci Technol 60(9):2399–2404. https://doi.org/10.2166/wst.2009.313
Yuan Y, Chen Q, Zhou S, Zhuang L, Hu P (2011) Improved electricity production from sewage sludge under alkaline conditions in an insert-type air-cathode microbial fuel cell. J Chem Technol Biotechnol 87(1):80–86. https://doi.org/10.1002/jctb.2686
Fischer F, Bastian C, Happe M, Mabillard E, Schmidt N (2011) Microbial fuel cell enables phosphate recovery from digested sewage sludge as struvite. Bioresour Technol 102(10):5824–5830. https://doi.org/10.1016/j.biortech.2011.02.089
Ge Z, Zhang F, Grimaud J, Hurst J, He Z (2013) Long-term investigation of microbial fuel cells treating primary sludge or digested sludge. Bioresour Technol 136:509–514. https://doi.org/10.1016/j.biortech.2013.03.016
Nandy A, Sharma M, Venkatesan SV, Taylor N, Gieg L, Thangadurai V (2019) Comparative evaluation of coated and non-coated carbon electrodes in a microbial fuel cell for treatment of municipal sludge. Energies 12(6):1034. https://doi.org/10.3390/en12061034
Mohd Zaini Makhtar M, Tajarudin HA, Samsudin MDM, Vadivelu VM, Shoparwe NF, ‘Izzah Zainuddin N (2021) Membrane-less microbial fuel cell: Monte Carlo simulation and sensitivity analysis for COD removal in dewatered sludge. AIP Adv 11(6):65016. https://doi.org/10.1063/5.0039014
Pant D, Van Bogaert G, Diels L, Vanbroekhoven K (2010) A review of the substrates used in microbial fuel cells (MFCs) for sustainable energy production. Bioresour Technol 101(6):1533–1543. https://doi.org/10.1016/j.biortech.2009.10.017
Liang P, Huang X, Fan MZ, Cao XX, Wang C (2007) Composition and distribution of internal resistance in three types of microbial fuel cells. Appl Microbiol Biotechnol 77(3):551–558. https://doi.org/10.1007/s00253-007-1193-4
Liu Z, Liu J, Zhang S, Su Z (2009) Study of operational performance and electrical response on mediator-less microbial fuel cells fed with carbon-and protein-rich substrates. Biochem Eng J 45(3):185–191. https://doi.org/10.1016/j.bej.2009.03.011
Chabert N, Amin Ali O, Achouak W (2015) All ecosystems potentially host electrogenic bacteria. Bioelectrochem 106:88–96. https://doi.org/10.1016/j.bioelechem.2015.07.004
Logan BE, Regan JM (2006) Electricity-producing bacterial communities in microbial fuel cells. Trends in Microbiol 14(12):512–518. https://doi.org/10.1016/j.tim.2006.10.003
Wang H, Ren ZJ (2014) Bioelectrochemical metal recovery from wastewater: a review. Water Res 66:219–232. https://doi.org/10.1016/J.WATRES.2014.08.013
Mathuriya AS, Yakhmi JV (2014) Microbial fuel cells to recover heavy metals. Environ Chem Lett 12(4):483–494. https://doi.org/10.1007/S10311-014-0474-2/FIGURES/3
Li Z, Zhang X, Lei L (December 2008) Electricity production during the treatment of real electroplating wastewater containing Cr6+ using microbial fuel cell. Process Biochem 43(12):1352–1358. https://doi.org/10.1016/J.PROCBIO.2008.08.005
Choi C, Cui Y (March 2012) Recovery of silver from wastewater coupled with power generation using a microbial fuel cell. Biores Technol 107:522–525. https://doi.org/10.1016/J.BIORTECH.2011.12.058
Sun J, Hu Y, Bi Z, Cao Y (2009) Improved performance of air-cathode single-chamber microbial fuel cell for wastewater treatment using microfiltration membranes and multiple sludge inoculation. J Power Sour 187(2):471–479. https://doi.org/10.1016/j.jpowsour.2008.11.022
Baranitharan E et al (2015) Enhanced power generation using controlled inoculum from palm oil mill effluent fed microbial fuel cell. Fuel 143:72–79. https://doi.org/10.1016/j.fuel.2014.11.030
Xie B, Liu H, Yan Y (2009) Improvement of the activity of anaerobic sludge by low-intensity ultrasound. J Environ Manag. https://doi.org/10.1016/j.jenvman.2007.09.004
Zhu H, Béland M (2006) Evaluation of alternative methods of preparing hydrogen producing seeds from digested wastewater sludge. Int J Hydrogen Energy 14(31):1980–1988. https://doi.org/10.1016/J.IJHYDENE.2006.01.019
More TT, Ghangrekar MM (2010) Improving performance of microbial fuel cell with ultrasonication pre-treatment of mixed anaerobic inoculum sludge. Bioresour Technol 101(2):562–567. https://doi.org/10.1016/j.biortech.2009.08.045
Khan MR, Amin MSA, Sarker S, Ferdaus K (2012) Design and fabrication of membrane less microbial fuel cell (ML-MFC) using food industries wastewater for power generation. J Chem Eng 27(2):55–59
Amin MSA, Haque T, Tarannum R, Khan MR (2014) Wastewater treatment and electricity generation by membrane less microbial fuel. Int J Environ Eng 6(3):314. https://doi.org/10.1504/ijee.2014.064306
Palanisamy G, Jung HY, Sadhasivam T, Kurkuri MD, Kim SC, Roh SH (2019) A comprehensive review on microbial fuel cell technologies: processes, utilization, and advanced developments in electrodes and membranes. J Clean Prod 221:598–621. https://doi.org/10.1016/j.jclepro.2019.02.172
Vavilin VA, Rytov SV, Lokshina LY (1996) A description of hydrolysis kinetics in anaerobic degradation of particulate organic matter. Bioresour Technol 56(2–3):229–237. https://doi.org/10.1016/0960-8524(96)00034-X
Acknowledgements
The authors would like to thank the Universiti Sains Malaysia for the financial support of this study via APEX Era grant (1001/PTEKIND/881004). The authors have declared no conflict of interest for the manuscript.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Mohamad Sobri, M.F., Mohd Zaini Makhtar, M. (2023). Application of Microbial Fuel Cell for Bioremediation of Sewage Sludge. In: Mohd Zaini Makhtar, M., Shukor, H., Yaser, A.Z. (eds) Microbial Fuel Cell (MFC) Applications for Sludge Valorization. Green Energy and Technology. Springer, Singapore. https://doi.org/10.1007/978-981-99-1083-0_5
Download citation
DOI: https://doi.org/10.1007/978-981-99-1083-0_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-99-1082-3
Online ISBN: 978-981-99-1083-0
eBook Packages: EnergyEnergy (R0)