Abstract
Treeline ecotone, though studied the world over because of its sensitivity to changing climate, has received limited attention in the Himalaya. It is in this backdrop that an extensive study in the Daksum-Sinthan Top area of Kashmir Himalaya, India, was carried out to document the taxonomic, life-form and phylogenetic diversity of plant assemblages at the treeline ecotone in relation to elevation and aspect. A total of 235 species belonging to 168 genera and 71 families were recorded in the ecotone. Only 26% of species were common between the north-facing and south-facing aspects, and a decline in the total number of species with elevation was the general trend. Herbs were predominant at all the elevations on both aspects. Sørensen’s dissimilarity across the elevations and aspects was low and the turnover component (βsim) was the major contributor to the overall dissimilarity. Phylogenetic overdispersion was noticed at lower elevations and phylogenetic clustering was prevalent at higher elevations on both aspects. Diffuse-type treeline-form was more common on the north-facing slope and tree-island type on the south-facing slope. The dominant treeline species on the north-facing slope was Betula utilis, and Pinus wallichiana on the south-facing slope.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
9.1 Introduction
Treeline ecotone, a vegetation zone between closed-canopy forests and alpine meadows (Holtmeier 2000; Holtmeier et al. 2003), is dynamic (Nagy and Grabherr 2009; Camarero et al. 2017), species-rich (Wielgolaski et al. 2017) and sensitive to environmental conditions (Nagy and Grabherr 2009; Camarero et al. 2017). Its dynamic nature is due to climatic factors, such as the growing season temperature (Körner 2012) and precipitation (Kharuk et al. 2010; Liang et al. 2014), and non-climatic factors, such as nutrient limitation (Li et al. 2008), biotic interactions (Hofgaard et al. 2010; Speed et al. 2011; Shrestha et al. 2015) and land-use change (Schickhoff 2005). Globally, treeline ecotones vary from abrupt lines to extended zones of increasingly small, stunted and/or dispersed trees (Bader et al. 2021). Spatial treeline patterns also differ greatly reflecting the underlying ecological processes that produce such patterns, and Holtmeier (2009) distinguished four types of treeline form, namely abrupt forest limit, transition zone (ecotone), ‘true krummholz belt’ and gradual transition from high-stemmed forest to crippled trees of the same species. ‘True krummholz’ was used for genetic krummholz, i.e. species that can grow only as shrubs, but now krummholz usually refers to environmentally induced stunted and crippled trees and genetic krummholz are referred to as shrubs (Bader et al. 2021). Subsequently, four treeline forms – diffuse, abrupt, island and krummholz—were recognized by Harsch and Bader (2011) with growth limitation being dominant only in the most common diffuse treeline, and dieback and seedling mortality in other treeline forms. Recently, Bader et al. (2021) proposed a framework of 12 forms at the hillslope scale based on the spatial pattern of tree cover as seen from above and changes in tree stature and physiognomy. The response of these treeline forms to climate change varies (Harsch and Bader 2011; Schickhoff et al. 2015) with diffuse treeline responding frequently to growing season warming and growth limitation is also higher in comparison to other treeline forms (Harsch and Bader 2011). Treeline shift in response to climate change has been a major focus of scientific investigation globally for more than a century (Bader et al. 2007), and a meta-analysis of 166 treeline sites revealed advancement in treeline at 52% of sites since 1900, recession in only 1% of sites and no change in treeline position at 47% sites (Harsch et al. 2009).
Himalaya, owing to its complexity, dynamic nature and greater sensitivity towards changing environmental conditions (Telwala et al. 2013; Aryal et al. 2014; Anup and Ghimire 2015), is likely to undergo changes, and more so in the treeline ecotone (Xu et al. 2009; Mishra and Mainali 2017) with serious consequences for invaluable goods and services that these ecosystems provide to the dependent humans. Pastoralism, wood logging, fuelwood gathering, excessive grazing and overexploitation of economically important species (Schickhoff 2002, 2005; Miehe et al. 2015) are the other factors that affect species composition, community structure (Lee et al. 2019; Ali et al. 2020), species distribution range (Pacifici et al. 2015; Kosanic et al. 2018) and more common upward shift in the species ranges in response to global climate warming (Chen et al. 2011; Lenoir and Svenning 2015). Many studies have been carried out in the Himalaya that deal with the taxonomic composition of vascular plants (Bharti et al. 2012; Pandita et al. 2019; Singh et al. 2019), phytosociology (Bürzle et al. 2017), dry matter dynamics (Rai et al. 2020), community structure and species regeneration (Shrestha et al. 2007; Gairola et al. 2008; Ghimire et al. 2010; Wong et al. 2010; Pandey et al. 2018) and community structure (Adhikari et al. 2012), but these studies do not focus specifically on treeline ecotone and, in particular, the influence of aspect and elevation on taxonomic, functional and phylogenetic diversity (PD) has not been studied.
Taking note of the above-mentioned ecological significance and lack of detailed ecological studies in the Himalaya, a comprehensive study was undertaken in a typical Kashmir Himalayan treeline ecotone to assess the complementary and interrelated components of taxonomic (species richness), functional (life-form) and phylogenetic diversity, and how these multiple measures of diversity vary in relation to elevation and aspect. While the documentation of species richness is the first essential step in biodiversity studies, functional diversity quantified based on species traits not only offers valuable insights into the functional uniqueness, redundancy and complementarity of communities but also provides clues to the resistance or resilience of communities. Likewise, phylogenetic diversity is a measure of the evolutionary relationships among the taxa, and species assemblage with greater phylogenetic diversity may provide cushion against long-term environmental change. We also examined how elevation and aspect influence α- and β-diversity patterns in the treeline ecotone. Such detailed studies are expected to expand our understanding of the structural organization of the treeline ecotone and could provide valuable insights into the response of communities to changing climate and other anthropogenic factors that are necessary for better management of ecosystems and ecotones.
9.2 Methodology
9.2.1 Study Area
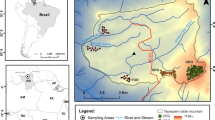
This study was carried out in the Daksum-Sinthan Top area of Kashmir Himalaya, India (Fig. 9.1). Daksum lies within the geographical coordinates of 33°36′43″ N and 75°26′6″ E and is located at an altitude of 2438 m above sea level (masl). Sinthan Top lies within the geographical coordinates of 33°34′ N and 75°30′ E and a road (NH1B highway) traverses this top that connects Kashmir Valley with Kishtwar, which lies in Jammu province of the Union Territory and is at 12,500 ft. (3800 m) above sea level. Physiographically, the area is hilly with lush mixed coniferous forests and grassy meadows. The average annual precipitation is about 1000 mm, with November as the driest month (31 mm rainfall), and the average annual air temperature is about 12.8 °C, with the highest temperature of about 20 °C recorded in July (Nanda et al. 2018; Fayaz et al. 2019).
9.2.2 Vegetation Sampling
Systematic sampling using the less biased quadrat method (Bellhouse 2005; Bhatta et al. 2012) was used to assess plant species composition in the treeline ecotone along the two elevation transects on the north-facing and south-facing aspects. Treeline ecotone in this study refers to the transition zone between the timberline to the treeless alpine vegetation (Fig. 9.2).
The treeline ecotone on the north-facing slope extended from 3200 to 3600 masl while on the south-facing slope it extended from 3300 to 3600 masl. The treeline ecotone on the north-facing and south-facing aspects was divided into elevation bands that were 100 m apart and hence we had five elevation bands on the north-facing slope and four bands on the south-facing slope. Three plots of 50 × 50 m2 area were established in each elevational band and in each of these 3 plots, 10 quadrats of 10 × 10 m2 area each were laid for sampling trees, 20 quadrats of 5 × 5 m2 area each for shrub sampling and 40 quadrats of 1 × 1 m2 area each for herbs were laid randomly during the growing season. Thus, in each elevational band, 30 quadrats were laid for tree sampling, 60 quadrats for the sampling of shrubs and 120 quadrats for herbs. In all, 1890 quadrats (270 for tress, 540 for shrubs and 1080 for herbs) were used for sampling vegetation in the treeline ecotone. During the sampling, if a rock of cover >10% was encountered along the transect or at places where the slope was inclined over 30°, the distance between the adjacent plots was increased by 10–20 m (Mosley et al. 1989; Gómez-Díaz et al. 2017; Bhatta et al. 2018).
We constructed species accumulation curves using the accumresult function of the BiodiversityR package in R (version 3.6.2; R Core Team, 2019) with 1000 permutations and random method to evaluate sampling adequacy separately for trees, shrubs and herbs (Fig. 9.3), because of differing size and number of sampling units used for the three life-form categories.
9.2.3 Data Analysis
Vegetation data were analysed for taxonomic and life-form composition, and phytosociological characteristics such as frequency, density and abundance were computed following Mandal and Joshi (2014). While presence–absence data were recorded for all the species, density and abundance were recorded for trees and shrubs only. Non-metric multidimensional scaling (NMDS), based on presence–absence of species in the sampling units, was performed to visualize the differences in species composition between north- and south-facing slopes, and analysis of similarity (ANOSIM) based on Hellinger distance with 999 permutations was used to test whether the overall compositional difference between north- and south-facing slopes was significant (Yang et al. 2020). All analyses were performed using vegan package in R.
Species richness was estimated from replicate samples laid across elevation bands on the two aspects. Since the total number of species observed is almost always an underestimate of the total number of species in the assemblage (Colwell and Elsensohn 2014), we used several incidence-based non-parametric estimators (Chao, Jack1, Jack2, Bootstrap) to correct this bias using EstimateS (9.1.0; Colwell 2013). Standard errors for all the estimators (except Jackknife2 for which it is not available as yet) were also computed. The performance of each non-parametric estimator was evaluated by quantifying bias, precision and accuracy (Walther and Moore 2005) at 25% and 50% sampling effort following Walther and Morand (1998) and Walther and Martin (2001) who suggested that the performance of species richness estimators should be evaluated at ‘intermediate’ sampling effort when the observed species richness has not reached the asymptote and is still increasing. Scaled performance measures, namely scaled mean error (SME) for bias, coefficient of variation (CV) for precision and scaled mean square error (SMSE) were used. Estimators that have smaller bias, higher precision and reduced SMSE were considered more accurate than others (Branco et al. 2018). To assess the changes in the richness and abundance of trees and shrubs with elevation, we computed Simpson’ dominance, Shannon’s diversity and Pielou’s evenness (J-evenness) indices for each elevation band on the two aspects in the treeline ecotone.
Beta-diversity (β-diversity) was calculated on the basis of presence–absence data, which was then partitioned into turnover and nestedness components using function beta.pair in R package betapart. In all, three matrices were produced based on pairwise comparisons of all elevation bands: the Sørensen dissimilarity index (βsor) that expresses the total compositional variation with values ranging between 0 and 1, the Simpson dissimilarity index matrix (βsim) expressing compositional changes due to species turnover, and βsor minus βsim giving the resultant nestedness component (βsne) of β-diversity.
β-diversity data (βsor, βsim, βsne) were further processed to distinguish between (1) along-elevation beta-diversity accounting for pairs starting from the lowest elevation to all other elevations, and (2) stepwise beta-diversity including pairs from one elevation to the next neighbouring one (Fontana et al. 2020). The effect of elevation on both stepwise and along-gradient beta-diversity was tested with a linear mixed-effects model using the ordinary least squares regression (OLS) method in the Past software 4.02 (Hammer et al. 2001).
We constructed a phylogenetic tree for all the vascular plants using the V. PhyloMaker (Jin and Qian 2019) in the R platform. This package contains a megaphylogeny, which was a combination of GBOTB (i.e., GenBank taxa with a backbone provided by Open Tree of Life version 9.1) for seed plants (Smith and Brown 2018) and the clades in (Zanne et al. 2014) phylogeny. For the genera and species in our dataset that are absent from the megaphylogeny, we added them to their respective genera (in the case of species) and families (in the case of genera) using the Phylomatic and BLADJ approaches (Webb et al. 2008) implemented in V. PhyloMaker (scenario 3; Jin and Qian 2019). V. PhyloMaker sets branch lengths of added taxa of a family by placing the nodes evenly between dated nodes and tips within the family, and adds a missing genus at the mid-point of its family branch and a missing species at the mid-point of its genus branch.
Five metrics were computed to document the phylogenetic diversity. These included: phylogenetic diversity (PD) (Faith 1992), which is the sum of evolutionary history in millions of years; mean pairwise distance (MPD) (Webb 2000), which is one of the most popular measures for computing the phylogenetic distance between a given group of species; mean nearest taxon distance (MNTD) (Webb et al. 2002), which is the average distance between an individual and the most closely related (non-conspecific) individual; net relatedness index (NRI); and nearest taxon index (NTI). NRI and NTI compare the phylogenetic diversity in the dataset to a randomly generated null model, from the regional species pool, revealing either phylogenetic overdispersion or evenness (co-occurring species more distantly related than expected by chance) or phylogenetic clustering (species more closely related than expected by chance). A positive NRI/NTI indicates that co-occurring species are more phylogenetically related (phylogenetic clustering) than expected by chance, whereas the negative value indicates that species are more distantly related (phylogenetic overdispersion) than expected by chance (Webb et al. 2002). All the phylogenetic indices were obtained with the R package picante (Kembel et al. 2010) (Table 9.1).
9.3 Results
9.3.1 Taxonomic and Life-Form Diversity
A total of 235 species belonging to 168 genera and 71 families were recorded at the treeline ecotone. Angiosperms with 164 species (116 genera and 40 families) were predominant and dicots were more numerous with 150 species (108 genera and 35 families) than monocots, which were represented by 14 species (8 genera and 5 families). Gymnosperms were represented by 4 species belonging to 4 genera and 2 families. Pteridophytes included 24 species (13 genera and 9 families) and bryophytes were represented by 43 species belonging to 35 genera and 20 families (Table 9.2).
Dominant families at the ecotone among angiosperms were Asteraceae, which included 27 species, followed by Lamiaceae (12 spp.), Rosaceae, Poaceae and Caryophyllaceae each with 9 species, Polygonaceae (7 spp.), Ranunculaceae and Boraginaceae (6 spp.) and Crassulaceae and Gentianaceae (5 spp.). In gymnosperms, Pinaceae was the dominant family. In the case of pteridophytes, Dryopteridaceae and Athyriaceae were the dominant families, which included 7 and 5 species respectively. In bryophytes, Pottiaceae was the dominant family with 7 species followed by Polytrichaceae and Dicranaceae (6 spp.).
Among the life-form groups, herbs were predominant with 176 species belonging to 117 genera and 39 families followed by mosses, which included 42 species belonging to 34 genera and 19 species. Other life-form groups, such as trees, shrubs and liverworts, were not well represented in the ecotone (Table 9.2).
9.3.2 Effect of Aspect on Taxonomic and Life-Form Diversity
Aspect had a distinct influence on species richness as 132 species were recorded on the north-facing slope as against 164 species on the south-facing slope (Table 9.3). In particular, angiosperms and bryophytes were more on the south-facing slope than on the north-facing slope. In respect of life-forms, herbs and mosses were more on the south-facing slope compared to the north-facing slope (Table 9.3).
Non-metric multidimensional scaling (Fig. 9.4) showed that species composition on the north- and south-facing slopes were strongly separated in ordination space with no overlap (stress value = 0.028).
The analysis of similarity (ANOSIM; Fig. 9.5) showed significant compositional difference between the aspects (R = 0.994, p = 0.007).
Analysis of similarity (ANOSIM) plot showing dissimilarity between and within the two aspects. The bold horizontal bar in the box indicates median; bottom of the box indicates 25th percentile; top of the box indicates 75th percentile; and whiskers extend to the most extreme data point. Data points falling outside the range (o) are plotted as outliers
9.3.3 Effect of Elevation on Taxonomic and Life-Form Diversity
Taxonomic composition and life-form diversity in different elevation bands on the two aspects are presented in Figs. 9.6 and 9.7. The overall trend was that the species number of all the taxonomic groups declined with elevation on both aspects (Fig. 9.6). Angiosperms were the predominant elements at all the elevations on the two aspects and their number ranged from 58 at the lowest elevation to 48 at the highest elevation in the treeline ecotone on the north-facing slope. Likewise, the number of angiosperms on the south-facing slope declined from 65 species at the lowest elevation to 61 species at the highest elevation (Fig. 9.6). Another notable feature was that pteridophytes were more common on the north-facing slope than the south-facing slope. In contrast, bryophytes and gymnosperms were more represented on the south-facing slope than the north-facing slope.
In life-form groups, a declining trend in the number of species with elevation was also noticed on both aspects (Fig. 9.7). Herbs were predominant floristic elements on both the aspects and at all the elevations. The number of herbs declined from 68 species at the lowest treeline ecotone elevation to 55 species almost at the upper limit of treeline ecotone on the north-facing slope. However, the number of herb species on the south-facing slope declined from 66 to 46 in the adjacent elevation bands (Fig. 9.7).
Only 61 (26%) species occurred on both aspects and 174 (74%) species occurred in only one of the two aspects (Table 9.4). Angiosperms, particularly dicots, among the taxonomic groups and herbs and mosses among the life-form groups included most of the exclusive species.
9.3.4 Species Richness
The observed number of species belonging to various life-form groups based on sampling in different elevation bands and the estimated number of species using incidence-based non-parametric estimators are presented in Table 9.5. A perusal of the data reveals that estimated values of species richness based on all the estimators were more or less similar to the observed species richness on the north-facing slope. On the contrary, the richness estimators predicted higher values for herbs in comparison to observed richness across all elevations (Table 9.5).
Species richness estimated using various non-parametric indices across the entire ecotone on both the slopes is presented in Figs. 9.8 and 9.9. On the north-facing slope, species accumulation curves reached saturation with our sampling effort (Fig. 9.8). Bootstrap richness estimators approached the asymptote earlier than other estimators in respect of trees and shrubs while for herbs all the estimators reached asymptote almost at the same level of sampling effort. Jack1 and Jack2 for trees and shrubs had an erratic behaviour at low sampling effort, but all the estimators tended to stabilize with increasing sampling effort. On the south-facing slope also, almost all the indices reached asymptote with our sampling effort for trees, shrubs and herbs (Fig. 9.9).
The species richness estimators had a different performance computed on the basis of bias, precision and accuracy (Tables 9.6 and 9.7). Invariably, Jack2 performed better across the groups and slopes and even at a low sampling effort.
9.3.5 Species Diversity
Species diversity estimated for each elevation band on the two slopes (Fig. 9.10) revealed that Simpson’s index ranged from 0.72 in the lowest elevation band to 0.26 in the highest elevation band of the north-facing slope. Simpson’s index on the south-facing slope ranged from 0.69 to 0.46 indicating a relatively modest decline with elevation and thus comparatively more uniform pattern across elevations. Shannon’s index ranged from 1.44 to 0.44 on the north-facing slope and 1.29 to 0.88 on the south-facing slope. On both the slopes, the Shannon diversity index was lowest in the uppermost elevation band of the ecotones, but higher values were recorded in different elevation bands on the two slopes. J-evenness values also showed a pattern similar to other indices (Fig. 9.10) with values ranging from 0.93 to 0.40 on the north-facing slope and 0.93 to 0.55 on the south-facing slope. Thus, the plant assemblages had even distribution of individuals among the species at lower elevations than in the higher elevations.
Analysis of variance (ANOVA; Table 9.8) revealed that elevation and aspect had a significant effect on the three diversity indices but the significant interactive effects of elevation and aspect were noticed only for Simpson’s and Shannon’s indices.
9.3.6 β-Diversity
Data presented in Fig. 9.11a and b reveal that Sørensen’s dissimilarity on both the slopes is low and the turnover component (βsim) was the major contributor to the overall dissimilarity. Cluster analysis based on turnover (βsim) revealed that the highest elevation band (3600 masl) in the treeline ecotone on the north-facing slope (Fig. 9.11c) was most dissimilar while as on the south-facing slope, the first two elevation bands (3300 and 3400 masl) represented one dissimilar pair and elevation bands at 3500 and 3600 masl represented the other dissimilar pair. Cluster analysis performed on the basis of nestedness (βsne) revealed that the basal elevation band (3200 masl) was most dissimilar to the rest of the elevation bands on the north-facing slope (Fig. 9.11e) but on the south-facing slope two dissimilarity clusters were obtained (Fig. 9.11f). When turnover (βsim) and nestedness (βsne) components of β-diversity were computed for pairs starting from the lowest elevation to all other elevations, and for pairs from one elevation to the next neighbouring elevation (Fig. 9.12), it again revealed that turnover contributed to the dissimilarity between the elevation bands.
Compositional dissimilarities across elevations and aspect. (a and b) β-diversity, turnover and nestedness on the north-facing slope (a) and south-facing slope (b). Pairwise average clustering of elevation bands based on βsim dissimilarity on north-facing slope (c) and south-facing slope (d). Pairwise average clustering of elevation bands based on βsne dissimilarity on north-facing slope (e) and south-facing slope (f)
Total β-diversity and turnover increased significantly with elevation (p-value = 0.05 or <0.05), but nestedness decreased insignificantly along the north-facing slope (Table 9.9, Fig. 9.13). Likewise, stepwise total β-diversity and turnover also increased at higher elevations but the increase was not statistically significant. Stepwise nestedness decreased significantly at higher elevation steps (p-value = 0.05) along the north-facing slope. On the south-facing slope, the total β-diversity did not show a significant increase but its turnover component showed a significant increase with elevation (p < 0.05). The stepwise total β-diversity and stepwise turnover also increased while stepwise nestedness decrease was not significant at higher elevations on the south-facing slope (Table 9.9, Fig. 9.14).
9.3.7 Phylogenetic Diversity
Phylogenetic diversity computed in terms of PD, MPD and MNTD, and NRI and NTI (Fig. 9.15) varied both in relation to elevation and aspect. Faith’s phylogenetic diversity (FD) decreased with elevation on the north-facing slope but did not show such a declining trend with elevation on the south-facing slope. Such a pattern was also recorded for mean pairwise distance (MPD). The mean nearest taxon distance (MNTD) index increased initially with elevation on both the north-facing and south-facing slopes but thereafter declined (Fig. 9.15).
Further analysis of the phylogenetic pattern using the net relatedness index (NRI) revealed an increase from a negative value of −0.57 at 3200 masl to the highest value of 2.20 at 3400 masl followed by comparatively lower values at higher elevations on the north-facing slope. NRI on the south-facing slope showed an initial decline at 3400 masl to −0.64 but it subsequently increased with elevation reaching a maximum value of 2.19 at the highest elevation of treeline ecotone. Nearest taxon index (NTI), on the north-facing slope, showed a pattern that was almost reverse of NRI (Fig. 9.15) but it almost followed the pattern similar to NRI on the south-facing slope.
Based on the above-mentioned indices, phylogenetic diversity was compared between the two aspects (Fig. 9.16). Analysis of variance revealed that MNTD (F = 43, p = 0.0003) and NTI (F = 7.26, p = 0.031) were significantly different between the two aspects and the rest of the indices did vary significantly between the aspects. The phylogenetic distribution of vascular plants on the two slopes is shown in Fig. 9.17a and b.
9.3.8 Treeline-Form
Two types of treeline forms were observed in the study area: diffuse type and tree-island type. The diffuse type was more common on the north-facing slope and tree-island type was more common on the south-facing slope (Fig. 9.18). The dominant treeline species on the north-facing slope was Betula utilis and on the south-facing slope, the treeline species was Pinus wallichiana. These treeline species were associated with thickets of shrubs, such as Rhododendron campanulatum and Juniperus squamata.
9.4 Discussion
A total of 235 species belonging to 168 genera and 71 families of various taxonomic and life-form groups were recorded at the treeline ecotone during this study, which is higher in comparison to many previous studies. For example, Junyan et al. (2014) reported 218 species, 164 genera and 67 families in the tropical coniferous broadleaved forest ecotone in China; Shrestha and Vetaas (2009) reported 84 species of vascular plants from 37 families and 55 genera in an ecotone in arid Trans-Himalayan Landscape of Nepal; Bürzle et al. (2017) reported 103 species of vascular plant species in treeline ecotone in Rolwaling Himal, Nepal; and Gaire et al. (2010) recorded just 30 plant species in the treeline ecotone of Langtang National Park, Central Nepal. Rai et al. (2012) reported 58–75 species in different communities in the Kedarnath Wildlife Sanctuary. Pandita and Dutt (2017) observed that herbaceous species in the northwest Himalayan ecotone range from 26 to 42 species. Mir et al. (2017) reported 48 and 54 plant species from Sonamarg and Gulmarg areas respectively of Kashmir Himalaya. However, a higher number of species (253 species) compared to the present study was reported by Singh et al. (2019) at the semi-disturbed treeline ecotone in Bhaderwah, Jammu and Kashmir. Such differences in species composition of treeline ecotones at regional and smaller spatial scales have been reported previously as well (Wielgolaski et al. 2017) and attributed to differences in the biotic and abiotic factors, such as human activities, soil conditions (Callaghan et al. 2004; Vittoz et al. 2010), temperature and precipitation (including snow cover) (Kullman 1995, 2003; Mathisen et al. 2014; Schwörer et al. 2014), micro-topography and disturbances (Rai et al. 2012), habitat heterogeneity (Junyan et al. 2014) and evolutionary processes (Schilthuizen 2000).
Among the life-forms, herbs were predominant in the treeline ecotone on both the slopes. A multitude of factors could be responsible for high herb richness, such as wide range of life-history adaptations that allow the herbs to persist and flourish (Whigham 2004), forest stand characteristics (Petersson et al. 2019) like spaced-out trees in the treeline ecotone that increase light availability (Dormann et al. 2020), soil physical and chemical properties (Hulshof and Spasojevic 2020), topography (Tardella et al. 2019) and topography-related microclimatic conditions (Macek et al. 2019). In addition to these present-day conditions, historical processes like climate fluctuations over millions of years (glacial events) could be also responsible for the present structure of treeline ecotone in Kashmir Himalaya with a very less number of tree species. Post-glacial low recolonization has been attributed to dispersal limitations and a majority of European tree species are reported to be filling less than 50% of their potentially climatically suitable range (Svenning and Skov 2004).
In this study, we also noticed that the number of species in the treeline ecotone was higher compared to the alpine meadow but lower than the sub-alpine closed-canopy forest (unpublished personal data). Such an observation is consistent with studies that have found species diversity at ecotones to be intermediate between the two bounded communities (Harper 1995; Turton and Duff 1992; Mészáros 1990; Meiners et al. 2000). Thus, the enhanced diversity at ecotones as reported in many studies (Shmida and Wilson 1985; Wolf 1993; Kernaghan and Harper 2001) is not a universal property but depends on the attributes of an ecotone in question. Further, it was also observed that a small fraction of species was ecotonal in nature with 16 (12.12%) and 14 (8.83%) species in the north-facing and south-facing slopes respectively growing exclusively in the ecotone. Most of the species in the treeline ecotone were either from the sub-alpine closed-canopy forest below the treeline ecotone or from the alpine meadow above the treeline ecotone, which indicates additive blending, instead of ecotonal specialization (Pandita and Dutt 2017; Pandita et al. 2019), is responsible for the species composition in the treeline ecotone. The two aspects did not differ significantly in terms of the number of species (F = 0.209, p = 0.653) but the species composition was different, with only 61 (26%) species being shared between the two aspects. Such a variation could be attributed to the modification of the local environment by aspect, slope and other topographic elements. Previous studies have also reported a significant influence of aspect on vegetation distribution (Moeslund et al. 2013; Hamid et al. 2020a, 2020b; Yang et al. 2020). This study also brought out that the south-facing (drier) slope in the ecotone was relatively species-rich compared to the north-facing (wetter) slope, which could be due to micro-relief spatial heterogeneity and favourable temperature on the south-facing slope. While the role of soil temperature in species compositional differences between aspects has been invoked by Dearborn and Danby (2017) and Hamid et al. (2020a; b), the influence of micro-relief heterogeneity in species richness has been established by Leutner et al. (2012). In fact, a higher number of mosses on the drier slope as observed in this study was also reported by Leutner et al. (2012) who attributed it to heterogeneity caused by rocks and boulders, which we also noticed during sampling in the study area. Species diversity computed as Simpson’s diversity decreased at the highest elevations on both aspects. Thus, it indicates that species diversity is relatively higher at lower elevations and lower at higher elevations. Lower diversity at higher elevations has been reported by other workers as well (Ahmad et al. 2020) and has been attributed to hard climatic conditions in the higher elevations (Lee et al. 2013; Gómez-Díaz et al. 2017). Evenness, measured as the relative abundance of different species in different elevation bands, revealed more or less similar pattern on the two slopes with higher values of evenness in the mid-elevation bands of the treeline ecotone and low evenness in the higher elevations. Our results are in contrast to the findings of Dar and Sundarapandian (2016) who observed an increase in evenness with elevation in Kashmir Himalaya. A decrease in Shannon index with elevation as recorded in this study is supported by similar observations of other workers (Gracia et al. 2007; Zhang et al. 2016). However, an increase in Shannon index with elevation has been also reported (Cui and Zheng 2016) with the highest in middle elevations (Kessler 2001; Grytnes and Vetaas 2002; Zhang et al. 2013). Such differences in species richness and diversity with elevation have been attributed to variation in climate, spatial habitat heterogeneity and anthropogenic factors (Cui and Zheng 2016). Mean β-diversity based on all possible pairs of comparisons of the sampled elevation bands did not vary much between the aspects, and turnover was the major contributor to the dissimilarity between the elevation bands irrespective of the aspect, which points towards the role of historical events, geographical isolation, habitat specialization, and environmental filtering and dispersal processes in shaping the plant assemblages (Baselga 2010; Pinto-Ledezma et al. 2018) and not due to patterns of colonization and extinction (Fontana et al. 2020). Along-elevation and stepwise β-diversity (Fig. 9.12) further reveals that species turnover increased with elevation and the increase was more distinct on the north-facing slope while nestedness decreased with elevation. Fontana et al. (2020) also reported an increase in turnover and a decrease in nestedness for vascular plants with elevation. Our results indicate that species sorting by environmental filtering and dispersal limitations seems to prevail, leading to higher species turnover and thereby reducing the probability of species-poor plant assemblages being a subset of species richness assemblages (nestedness) in the ecotone. On the contrary, Hamid et al. (2020a, b) reported that nestedness-resultant dissimilarity contributed more to β-diversity among the summits than the species turnover. Such variations could be because of differences in the elevation ranges considered in these studies for the study of β-diversity and its components.
Faith’s phylogenetic diversity index (FD) declined with elevation on the north-facing slope but not on the south-facing slope (Fig. 9.15). We also noticed a significant positive correlation between PD and species richness on both north-facing (r = +0.995, p < 0.001) and south-facing (r = +0.953, p = 0.047) slopes, which is consistent with the findings of Cheng et al. (2018) and Zhu et al. (2019). It is quite likely that species richness may be the dominant factor driving the phylogenetic diversity across elevations and aspect. Our data also revealed higher mean pairwise distance (MPD) values than the mean nearest taxon distance (MNTD) index (Fig. 9.15). It is expected because MPD is the average of all pairwise species and MNTD is the average of only the nearest neighbour distances and such observations have been made also by Cadotte et al. (2010) and Kellar et al. (2015). MPD was higher at lower elevations in the ecotone and then declined sharply with elevation on the north-facing slope but the decline was steady on the south-facing slope (Fig. 9.15). Higher values of MPD indicate the presence of distantly related species co-occurring in the lower elevations of treeline ecotone particularly on the north-facing slope where it declined with elevation. On the south-facing slope, MPD did not show a sharp decline with elevation (Fig. 9.15). With respect to MNTD, the pattern was different between the two aspects and on the north-facing slope, it increased with elevation and then declined. A small mid-elevation increase in MNTD was also noticed on the south-facing slope. It indicates that mid-elevations in the ecotone have witnessed recent diversification compared to lower elevations where diversification might have taken place in the past. It implies that there are more closely related terminal species at higher elevations compared to lower elevations, which has also been reported by Zhang et al. (2021). Net relatedness index (NRI) and nearest taxon index (NTI) revealed phylogenetic overdispersion at lower elevations on both aspects but phylogenetic clustering at higher elevations, particularly on the south-facing slope. Such a pattern has also been reported by Manish and Pandit (2018) in the Himalaya, and ecological filtering at higher elevations could be responsible for phylogenetic clustering while interspecific competition may be determining phylogenetic overdispersion at low and mid-elevations of the treeline ecotone.
Two types of treeline forms, namely diffuse type and tree-island type, were common in the study area (Fig. 9.18). The diffuse type was more common on the north-facing slope and the tree-island type on the south-facing slope. Diffuse type gets formed due to slow density change with elevation in the treeline species (Bader et al. 2021) with mortality within the ecotone coupled with environmental heterogeneity or stochasticity and/or seed-limited colonization. Tree-island type treeline form is also a consequence of mortality within the ecotone, environmental heterogeneity, clustered seed dispersal, clonal reproduction and microclimate alterations due to wind or snow distribution, abrasion etc. (Bader et al. 2021). Others (Schickhoff et al. 2016; Harsch and Bader 2011; Körner 2012) have also reported diffuse, abrupt, island and krummholz treeline forms in different parts of Himalaya with the diffuse form being more vulnerable to climate change (Harsch and Bader 2011). In this study, we noticed that the treeline species differ on the two aspects. On the north-facing aspect, Betula utilis (tree) and Rhododendron campanulatum (shrub) were the dominant treeline species. On the contrary, the dominant treeline species on the south-facing slope was Pinus wallichiana. As many as 58 treeline species belonging to 14 genera and 8 families have been reported by Singh et al. (2020). The most common treeline species in the Himalaya belong to genera such as Abies, Betula, Picea (Table 9.10).
9.5 Concluding Remarks
While compiling this chapter, it became apparent that many knowledge gaps exist about community structure, species composition and turnover, and ecological and evolutionary diversification of species in the treeline ecotones across the Himalaya. Thus, it is high time that detailed and holistic studies are undertaken related to species diversity with a focus not only on vascular plants but also on other plant groups, animals and even so far neglected microbes as well and all other interacting factors at varied spatial scales. Particularly interesting would be the studies explicating the role of mutualists (mycorrhizas and dark septate hyphae etc.) in the growth of certain treeline species and their interaction under various climate change scenarios. Equally valuable would be to discern the relative role of inorganic and organic forms of nutrients, particularly nitrogen, in the growth and development of species in the treeline ecotone. It is also necessary to establish sites for long-term monitoring of treeline ecotone across Himalaya for the study of temporal changes in the structural organization and functional integrity of treeline ecotone essentially in relation to climate change and other anthropogenic pressures. Well-planned long-term spatial and temporal studies are likely to open new vistas in our understanding of the seasonal, annual and more long-term changes in the treeline ecotone and its response to changing environment, which is of pivotal importance in formulating effective strategies for conservation of such fragile ecosystems in the Himalaya for larger and longer benefit to the dependent and marginalized human population.
References
Adhikari BS, Kumar R (2020) Effect of snowmelt regime on phenology of herbaceous species at and around treeline in Western Himalaya, India. Not Sci Biol 12(4):901–919
Adhikari BS, Rawat GS, Bargal K (2012) Community structure along timberline ecotone in relation to micro-topography and disturbances in Western Himalaya. Not Sci Biol 4(2):41–52
Ahmad M, Uniyal SK, Batish DR, Singh HP, Jaryan V, Rathee S, Sharma P, Kohli RK (2020) Patterns of plant communities along vertical gradient in Dhauladhar Mountains in lesser Himalayas in North-Western India. Sci Total Environ 716:136919
Ali A, Sanaei A, Li M, Nalivan OA, Ahmadaali K, Pour MJ, Valipour A, Karami J, Aminpour M, Kaboli H (2020) Impacts of climatic and edaphic factors on the diversity, structure and biomass of species-poor and structurally-complex forests. Sci Total Environ 706:135719
Anup K, Ghimire A (2015) High-altitude plants in era of climate change: a case of Nepal Himalayas. In: Climate change impacts on high-altitude ecosystems. Springer, Cham, pp 177–187
Aryal A, Brunton D, Raubenheimer D (2014) Impact of climate change on human-wildlife-ecosystem interactions in the trans-Himalaya region of Nepal. Theor Appl Climatol 115(3):517–529
Bader MY, Rietkerk M, Bregt AK (2007) Vegetation structure and temperature regimes of tropical alpine treelines. Arct Antarct Alp Res 39(3):353–364
Bader MY, Llambí LD, Case BS, Buckley HL, Toivonen JM, Camarero JJ, Cairns DM, Brown CD, Wiegand T, Resler LM (2021) A global framework for linking alpine-treeline ecotone patterns to underlying processes. Ecography 44(2):265–292
Baker B, Moseley R (2007) Advancing treeline and retreating glaciers: implications for conservation in Yunnan, PR China. Arct Antarct Alp Res 39(2):200–209
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19(1):134–143
Baselga A, Orme CDL (2012) Betapart: an R package for the study of beta diversity. Methods Ecol Evol 3(5):808–812
Batllori E, Blanco-Moreno J, Ninot J, Gutiérrez E, Carrillo E (2009) Vegetation patterns at the alpine treeline ecotone: the influence of tree cover on abrupt change in species composition of alpine communities. J Veg Sci 20(5):814–825
Bellhouse D (2005) Systematic sampling methods. Encyclopedia Biostat. https://doi.org/10.1002/9781118445112.stat05723
Bharti RR, Adhikari BS, Rawat GS (2012) Assessing vegetation changes in timberline ecotone of Nanda Devi National Park, Uttarakhand. Int J Appl Earth Obs Geoinf 18:472–479
Bhatta KP, Chaudhary RP, Vetaas OR (2012) A comparison of systematic versus stratified-random sampling design for gradient analyses: a case study in subalpine Himalaya. Nepal Phytocoenologia 42(3–4):191–202
Bhatta KP, Grytnes JA, Vetaas OR (2018) Scale sensitivity of the relationship between alpha and gamma diversity along an alpine elevation gradient in Central Nepal. J Biogeogr 45(4):804–814
Branco M, Figueiras FG, Cermeño P (2018) Assessing the efficiency of non-parametric estimators of species richness for marine microplankton. J Plankton Res 40(3):230–243
Burnham KP, Overton WS (1979) Robust estimation of population size when capture probabilities vary among animals. Ecology 60(5):927–936
Bürzle B, Schickhoff U, Schwab N, Oldeland J, Müller M, Böhner J, Chaudhary RP, Scholten T, Dickoré WB (2017) Phytosociology and ecology of treeline ecotone vegetation in RolwalingHimal, Nepal. Phytocoenologia 47(2):197–220
Cadotte MW, Jonathan Davies T, Regetz J, Kembel SW, Cleland E, Oakley TH (2010) Phylogenetic diversity metrics for ecological communities: integrating species richness, abundance and evolutionary history. Ecol Lett 13(1):96–105
Callaghan TV, Björn LO, Chernov Y, Chapin T, Christensen TR, Huntley B, Ims RA, Johansson M, Jolly D, Jonasson S (2004) Biodiversity, distributions and adaptations of Arctic species in the context of environmental change. AMBIO: A J Human Environ 33(7):404–417
Camarero JJ, Linares JC, García-Cervigón AI, Batllori E, Martínez I, Gutiérrez E (2017) Back to the future: the responses of alpine treelines to climate warming are constrained by the current ecotone structure. Ecosystems 20(4):683–700
Chao A (1984) Nonparametric estimation of the number of classes in a population. Scand J Stat. 11:265–270
Chen I-C, Hill JK, Ohlemüller R, Roy DB, Thomas CD (2011) Rapid range shifts of species associated with high levels of climate warming. Science 333(6045):1024–1026
Cheng X-L, Yuan L-X, Nizamani MM, Zhu Z-X, Friedman CR, Wang H-F (2018) Taxonomic and phylogenetic diversity of vascular plants at Ma’anling volcano urban park in tropical Haikou, China: Reponses to soil properties. PLoS One 13(6):e0198517
Chhetri PK, Cairns DM (2015) Contemporary and historic population structure of Abies spectabilis at treeline in Barun valley, eastern Nepal Himalaya. J Mt Sci 12(3):558–570
Chhetri PK, Bista R, Shrestha KB (2020) How does the stand structure of treeline-forming species shape the treeline ecotone in different regions of the Nepal Himalayas? J Mt Sci 17(10):2354–2368
Colwell RK (2006) EstimateS: statistical estimation of species richness and shared species from simples, version 8.0. http://purl oclc org/estimates
Colwell R (2013) EstimateS: statistical estimation of species richness and shared species from samples. Version 9.1.0, User's Guide and Application http://purl.oclc.org/estimates
Colwell RK, Elsensohn JE (2014) EstimateS turns 20: statistical estimation of species richness and shared species from samples, with non-parametric extrapolation. Ecography 37(6):609–613
Cui W, Zheng X-X (2016) Spatial heterogeneity in tree diversity and forest structure of evergreen broadleaf forests in southern China along an altitudinal gradient. Forests 7(10):216
Dar JA, Sundarapandian S (2016) Patterns of plant diversity in seven temperate forest types of Western Himalaya, India. J Asia-Pac Biodivers 9(3):280–292
Dearborn KD, Danby RK (2017) Aspect and slope influence plant community composition more than elevation across forest–tundra ecotones in subarctic Canada. J Veg Sci 28(3):595–604
Dormann CF, Bagnara M, Boch S, Hinderling J, Schäfer D, Schall P, Hartig F (2020) Plant species richness increases with light availability, but not variability, in temperate forests understorey. BMC Ecol 20:43. https://doi.org/10.1186/s12898-020-00311-9
Faith DP (1992) Conservation evaluation and phylogenetic diversity. Biol Conserv 61(1):1–10
Fayaz M, Jain AK, Bhat MH, Kumar A (2019) Ethnobotanical survey of Daksum forest range of Anantnag District, Jammu and Kashmir, India. J Herbs Spices Med Plants 25(1):55–67
Fontana V, Guariento E, Hilpold A, Niedrist G, Steinwandter M, Spitale D, Nascimbene J, Tappeiner U, Seeber J (2020) Species richness and beta diversity patterns of multiple taxa along an elevational gradient in pastured grasslands in the European Alps. Sci Rep 10(1):1–11
Gaire N, Dhakal Y, Lekhak H, Bhuju D, Shah S (2010) Vegetation dynamics in treeline ecotone of Langtang National Park, Central Nepal. Nepal J Sci Technol 11:107–114
Gairola S, Rawal R, Todaria N (2008) Forest vegetation patterns along an altitudinal gradient in sub-alpine zone of west Himalaya, India. Afr J Plant Sci 2(6):042–048
Ghimire B, Mainali KP, Lekhak HD, Chaudhary RP, Ghimeray AK (2010) Regeneration of Pinus wallichiana AB Jackson in a trans-Himalayan dry valley of north-Central Nepal. Himal J Sci 6(8):19–26
Gómez-Díaz JA, Krömer T, Carvajal-Hernández CI, Gerold G, Heitkamp F (2017) Richness and distribution of herbaceous angiosperms along gradients of elevation and forest disturbance in Central Veracruz, Mexico. Bot Sci 95(2):307–328
Gracia M, Montané F, Piqué J, Retana J (2007) Overstory structure and topographic gradients determining diversity and abundance of understory shrub species in temperate forests in Central Pyrenees (NE Spain). For Ecol Manag 242(2–3):391–397
Grytnes JA, Vetaas OR (2002) Species richness and altitude: a comparison between null models and interpolated plant species richness along the Himalayan altitudinal gradient, Nepal. Am Nat 159(3):294–304
Hamid M, Khuroo AA, Malik AH, Ahmad R, Singh CP (2020a) Assessment of alpine summit flora in Kashmir Himalaya and its implications for long-term monitoring of climate change impacts. J Mt Sci 17(8):1974–1988
Hamid M, Khuroo AA, Malik AH, Ahmad R, Singh CP, Dolezal J, Haq SM (2020b) Early evidence of shifts in alpine summit vegetation: a case study from Kashmir Himalaya. Front Plant Sci 11:421
Hammer Ø, Harper DA, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4(1):9
Harper KA (1995) Effect of expanding clones of Gaylussacia baccata (black huckleberry) on species composition in sandplain grassland on Nantucket Island, Massachusetts. Bull Torrey Bot Club 122:124–133
Harsch MA, Bader MY (2011) Treeline form–a potential key to understanding treeline dynamics. Glob Ecol Biogeogr 20(4):582–596
Harsch MA, Hulme PE, McGlone MS, Duncan RP (2009) Are treelines advancing? A global meta-analysis of treeline response to climate warming. Ecol Lett 12(10):1040–1049
He Z, Zhao W, Zhang L, Liu H (2013) Response of tree recruitment to climatic variability in the alpine treeline ecotone of the Qilian Mountains, northwestern China. For Sci 59(1):118–126
Hofgaard A, Løkken JO, Dalen L, Hytteborn H (2010) Comparing warming and grazing effects on birch growth in an alpine environment–a 10-year experiment. Plant Ecol Divers 3(1):19–27
Holtmeier F (2000) Die Höhengrenze der Gebirgswälder (ArbeitenausdemInstitut für Landschaftsökologie 8). WestfälischeWilhelms-Universität, Münster
Holtmeier F-K (2009) Mountain timberlines: ecology, patchiness, and dynamics, vol 36. Springer, Cham
Holtmeier F-K, Broll G, Müterthies A, Anschlag K (2003) Regeneration of trees in the treeline ecotone: northern Finnish Lapland. Fennia 181(2):103–128
Hulshof CM, Spasojevic MJ (2020) The edaphic control of plant diversity. Glob Ecol Biogeogr 29(10):1634–1165
Jin Y, Qian H (2019) V. PhyloMaker: an R package that can generate very large phylogenies for vascular plants. Ecography 42(8):1353–1359
Junyan Z, Kewu C, Runguo Z, Yi D (2014) Changes in floristic composition, community structure and species diversity across a tropical coniferous-broadleaved forest ecotone. Trop Conserv Sci 7(1):126–144
Kellar PR, Ahrendsen DL, Aust SK, Jones AR, Pires JC (2015) Biodiversity comparison among phylogenetic diversity metrics and between three north American prairies. Appl Plant Sci 3(7):1400108
Kembel S, Cowan P, Helmus M, Cornwell W, Morlon H, Ackerly D, Blomberg S, Webb C (2010) Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26:1463–1464
Kernaghan G, Harper K (2001) Community structure of ectomycorrhizal fungi across an alpine/subalpine ecotone. Ecography 24(2):181–188
Kessler M (2001) Patterns of diversity and range size of selected plant groups along an elevational transect in the Bolivian Andes. Biodivers Conserv 10(11):1897–1921
Kharuk VI, Im ST, Dvinskaya ML, Ranson KJ (2010) Climate-induced mountain tree-line evolution in southern Siberia. Scand J For Res 25(5):446–454
Körner C (2012) Alpine treelines: functional ecology of the global high elevation tree limits. Springer, Cham
Körner C, Paulsen J (2004) A world-wide study of high altitude treeline temperatures. J Biogeogr 31(5):713–732
Kosanic A, Anderson K, Harrison S, Turkington T, Bennie J (2018) Changes in the geographical distribution of plant species and climatic variables on the West Cornwall peninsula (south West UK). PLoS One 13(2):e0191021
Kullman L (1995) Holocene tree-limit and climate history from the Scandes Mountains, Sweden. Ecology 76(8):2490–2502
Kullman L (2003) Recent reversal of Neoglacial climate cooling trend in the Swedish Scandes as evidenced by mountain birch tree-limit rise. Glob Planet Chang 36(1–2):77–88
Lee C-B, Chun J-H, Song H-K, Cho H-J (2013) Altitudinal patterns of plant species richness on the Baekdudaegan Mountains, South Korea: mid-domain effect, area, climate, and Rapoport’s rule. Ecol Res 28(1):67–79
Lee CK, Laughlin DC, Bottos EM, Caruso T, Joy K, Barrett JE, Brabyn L, Nielsen UN, Adams BJ, Wall DH (2019) Biotic interactions are an unexpected yet critical control on the complexity of an abiotically driven polar ecosystem. Comm Biol 2(1):1–10
Lenoir J, Svenning JC (2015) Climate-related range shifts–a global multidimensional synthesis and new research directions. Ecography 38(1):15–28
Leutner BF, Steinbauer MJ, Müller CM, Früh AJ, Irl S, Jentsch A, Beierkuhnlein C (2012) Mosses like it rough—growth form specific responses of mosses, herbaceous and woody plants to micro-relief heterogeneity. Diversity 4(1):59–73
Li MH, Xiao WF, Shi P, Wang SG, Zhong YD, Liu XL, Wang XD, Cai XH, Shi ZM (2008) Nitrogen and carbon source–sink relationships in trees at the Himalayan treelines compared with lower elevations. Plant Cell Environ 31(10):1377–1387
Liang E, Dawadi B, Pederson N, Eckstein D (2014) Is the growth of birch at the upper timberline in the Himalayas limited by moisture or by temperature? Ecology 95(9):2453–2465
Macek M, Kopecký M, Wild J (2019) Maximum air temperature controlled by landscape topography affects plant species composition in temperate forests. Landsc Ecol 34(11):2541–2556
Mandal G, Joshi SP (2014) Analysis of vegetation dynamics and phytodiversity from three dry deciduous forests of Doon Valley, Western Himalaya, India. J Asia-Pac Biodivers 7(3):292–304
Manish K, Pandit MK (2018) Phylogenetic diversity, structure and diversification patterns of endemic plants along the elevational gradient in the eastern Himalaya. Plant Ecol Divers 11(4):501–513
Mathisen IE, Mikheeva A, Tutubalina OV, Aune S, Hofgaard A (2014) Fifty years of tree line change in the K hibiny M ountains, R ussia: advantages of combined remote sensing and dendroecological approaches. Appl Veg Sci 17(1):6–16
Meiners S, Handel S, Pickett S (2000) Tree seedling establishment under insect herbivory: edge effects and inter-annual variation. Plant Ecol 151(2):161–170
Mészáros I (1990) Spatial changes in herb layer in a beech forest/clear cut area ecotone from northern Hungary. Spatial processes in plant communities Academia, Prague, pp 59–71
Miehe G, Gravendeel B, Kluge J, Kuss P, Long D, Opgenoorth L, Pendry C, Rajbhandari K, Rajbhandary S, Ree R (2015) Flora. Royal Botanic Garden Edinburgh, Edinburgh
Mir NA, Masood T, Sofi P, Husain M, Rather T (2017) Life form spectrum of vegetation in Betula dominant tree stands along the available altitudinal gradient in north western Himalayas of Kashmir. J Pharmacogn Phytochem 6(4):267–272
Mishra NB, Mainali KP (2017) Greening and browning of the Himalaya: spatial patterns and the role of climatic change and human drivers. Sci Total Environ 587:326–339
Moeslund JE, Arge L, Bøcher PK, Dalgaard T, Svenning JC (2013) Topography as a driver of local terrestrial vascular plant diversity patterns. Nord J Bot 31(2):129–144
Mosley JC, Bunting SC, Hironaka M (1989) Quadrat and sample sizes for frequency sampling mountain meadow vegetation. Great Basin Nat 49:241–248
Nagy L, Grabherr G (2009) The biology of alpine habitats. Oxford University Press on Demand, Oxford
Nanda SA, Reshi ZA, Ul-haq M, Lone A, Mir SA (2018) Taxonomic and functional plant diversity patterns along an elevational gradient through treeline ecotone in Kashmir. Trop Ecol 59(2):211–224
Pacifici M, Foden WB, Visconti P, Watson JE, Butchart SH, Kovacs KM, Scheffers BR, Hole DG, Martin TG, Akçakaya HR (2015) Assessing species vulnerability to climate change. Nat Clim Chang 5(3):215–224
Pandey A, Badola HK, Rai S, Singh SP (2018) Timberline structure and woody taxa regeneration towards treeline along latitudinal gradients in Khangchendzonga National Park, Eastern Himalaya. PLoS One 13(11):e0207762
Pandita S, Dutt HC (2017) Herbaceous species in structuring the ecotone between forest-grassland in lesser stratum of north-west (NW) Himalaya. NeBIO 8:287â
Pandita S, Kumar V, Dutt HC (2019) Environmental variables Vis-a-Vis distribution of herbaceous tracheophytes on northern sub-slopes in Western Himalayan ecotone. Ecol Process 8(1):1–9
Petersson L, Holmström E, Lindbladh M, Felton A (2019) Tree species impact on understory vegetation: vascular plant communities of scots pine and Norway spruce managed stands in northern Europe. For Ecol Manag 448:330–345
Pielou F (1975) Ecological diversity. wileyIntescieco, New York
Pinto-Ledezma JN, Larkin DJ, Cavender-Bares J (2018) Patterns of beta diversity of vascular plants and their correspondence with biome boundaries across North America. Front Ecol Evol 6:194
Pradhan UC, Lachungpa ST (1990) Sikkim-Himalayan Rhododendrons. Primulaceae Books
Rai ID, Adhikari BS, Rawat GS (2012) Floral diversity along sub-alpine and alpine ecosystems in Tungnath area of Kedarnath wildlife sanctuary, Uttarakhand. Indian For 138(10):927–940
Rai I, Bharti R, Adhikari B, Rawat G (2013) Structure and functioning of timberline vegetation in the western Himalaya: a case study. In: Wu N, Rawat GS, Joshi S, Ismail M, Sharma E (eds) High-altitude rangelands and their interfaces in the Hindu Kush Himalayas. International Centre for Integrated Mountain Development, Nepal, p 91107
Rai ID, Padalia H, Singh G, Adhikari BS, Rawat GS (2020) Vegetation dry matter dynamics along treeline ecotone in Western Himalaya, India. Trop Ecol 61(1):116–127
Schickhoff U (2002) Die Degradierung der GebirgswälderNordpakistans. Faktoren, Prozesse und Wirkungszusammenhänge in einemregionalen Mensch-Umwelt-System ErdwissForsch 41
Schickhoff U (2005) The upper timberline in the Himalayas, Hindu Kush and Karakorum: a review of geographical and ecological aspects. In: Mountain ecosystems. Springer, Cham, pp 275–354
Schickhoff U, Bobrowski M, Böhner J, Bürzle B, Chaudhary R, Gerlitz L, Heyken H, Lange J, Müller M, Scholten T (2015) Do Himalayan treelines respond to recent climate change? An evaluation of sensitivity indicators. Earth Syst Dynam 6(1):245–265
Schickhoff U, Bobrowski M, Böhner J, Bürzle B, Chaudhary RP, Gerlitz L, Lange J, Müller M, Scholten T, Schwab N (2016) Climate change and treeline dynamics in the Himalaya. In: Climate change, glacier response, and vegetation dynamics in the Himalaya. Springer, Cham, pp 271–306
Schilthuizen M (2000) Ecotone: speciation-prone. Trends Ecol Evol 15(4):130–131
Schwab N, Kaczka RJ, Janecka K, Böhner J, Chaudhary RP, Scholten T, Schickhoff U (2018) Climate change-induced shift of tree growth sensitivity at a central Himalayan treeline ecotone. Forests 9(5):267
Schwörer C, Henne PD, Tinner W (2014) A model-data comparison of Holocene timberline changes in the Swiss Alps reveals past and future drivers of mountain forest dynamics. Glob Chang Biol 20(5):1512–1526
Shannon C, Weaver W (1949) rep. 1998, The mathematical theory of communication (Urbana. University of Illinois Press). Foreword by Richard E. Blahut and Bruce Hajek
Shi P, Körner C, Hoch G (2006) End of season carbon supply status of woody species near the treeline in western China. Basic Appl Ecol 7(4):370–377
Shi P, Körner C, Hoch G (2008) A test of the growth-limitation theory for alpine tree line formation in evergreen and deciduous taxa of the eastern Himalayas. Funct Ecol 22(2):213–220
Shmida A, Wilson MV (1985) Biological determinants of species diversity. J Biogeogr 12(1):1–20
Shrestha KB, Vetaas OR (2009) The forest ecotone effect on species richness in an arid trans-Himalayan landscape of Nepal. Folia Geobot 44(3):247–262
Shrestha BB, Ghimire B, Lekhak HD, Jha PK (2007) Regeneration of treeline birch (Betula utilis D. Don) forest in a trans-Himalayan dry valley in Central Nepal. Mt Res Dev 27(3):259–267
Shrestha KB, Hofgaard A, Vandvik V (2015) Recent treeline dynamics are similar between dry and Mesic areas of Nepal, central Himalaya. J Plant Ecol 8(4):347–358
Simpson E (1949) Measurement of diversity. Nature 163:688. Simpson688163Nature1949
Singh SP (2018) Research on Indian Himalayan treeline ecotone: an overview. Trop Ecol 59(2):163–176
Singh D, Sharma A, Sharma N (2019) Composition, richness and floristic diversity along an elevational gradient in a semi-disturbed treeline ecotone, Bhaderwah, Jammu and Kashmir. J Appl Nat Sci 11(1):23–34
Singh SP, Gumber S, Singh RD, Singh G (2020) How many tree species are in the Himalayan treelines and how are they distributed? Trop Ecol 61(3):317–327
Smith SA, Brown JW (2018) Constructing a broadly inclusive seed plant phylogeny. Am J Bot 105(3):302–314
Smith EP, van Belle G (1984) Nonparametric estimation of species richness. Biometrics. 40:119–129
Speed JD, Austrheim G, Hester AJ, Mysterud A (2011) Growth limitation of mountain birch caused by sheep browsing at the altitudinal treeline. For Ecol Manag 261(7):1344–1352
Svenning JC, Skov F (2004) Limited filling of the potential range in European tree species. Ecol Lett 7(7):565–573
Tardella FM, Postiglione N, Bricca A, Cutini M, Catorci A (2019) Altitude and aspect filter the herb layer functional structure of sub-Mediterranean forests. Phytocoenologia 49(2):185–198
Telwala Y, Brook BW, Manish K, Pandit MK (2013) Climate-induced elevational range shifts and increase in plant species richness in a Himalayan biodiversity epicentre. PLoS One 8(2):e57103
Turton S, Duff G (1992) Light environments and floristic composition across an open forest-rainforest boundary in northeastern Queensland. Aust J Ecol 17(4):415–423
Vittoz P, Camenisch M, Mayor R, Miserere L, Vust M, Theurillat J-P (2010) Subalpine-nival gradient of species richness for vascular plants, bryophytes and lichens in the Swiss inner Alps. Bot Helv 120(2):139–149
Walther BA, Martin JL (2001) Species richness estimation of bird communities: how to control for sampling effort? Ibis 143(4):413–419
Walther BA, Moore JL (2005) The concepts of bias, precision and accuracy, and their use in testing the performance of species richness estimators, with a literature review of estimator performance. Ecography 28(6):815–829
Walther B, Morand S (1998) Comparative performance of species richness estimation methods. Parasitology 116(4):395–405
Wang T, Zhang QB, Ma K (2006) Treeline dynamics in relation to climatic variability in the central Tianshan Mountains, northwestern China. Glob Ecol Biogeogr 15(4):406–415
Webb CO (2000) Exploring the phylogenetic structure of ecological communities: an example for rain forest trees. Am Nat 156(2):145–155
Webb CO, Ackerly DD, McPeek MA, Donoghue MJ (2002) Phylogenies and community ecology. Annu Rev Ecol Syst 33(1):475–505
Webb CO, Ackerly DD, Kembel SW (2008) Phylocom: software for the analysis of phylogenetic community structure and trait evolution. Bioinformatics 24(18):2098–2100
Whigham DF (2004) Ecology of woodland herbs in temperate deciduous forests. Annu Rev Ecol Evol Syst 35:583–621
Wielgolaski FE, Hofgaard A, Holtmeier F-K (2017) Sensitivity to environmental change of the treeline ecotone and its associated biodiversity in European mountains. Clim Res 73(1–2):151–166
Wolf JH (1993) Diversity patterns and biomass of epiphytic bryophytes and lichens along an altitudinal gradient in the northern Andes. Ann Mo Bot Gard 80:928–960
Wong MH-G, Duan C-Q, Long Y-C, Luo Y, Xie G-Q (2010) How will the distribution and size of subalpine Abies georgei forest respond to climate change? A study in Northwest Yunnan, China. Phys Geogr 31(4):319–335
Xu J, Grumbine RE, Shrestha A, Eriksson M, Yang X, Wang Y, Wilkes A (2009) The melting Himalayas: cascading effects of climate change on water, biodiversity, and livelihoods. Conserv Biol 23(3):520–530
Yang J, El-Kassaby YA, Guan W (2020) The effect of slope aspect on vegetation attributes in a mountainous dry valley, Southwest China. Sci Rep 10(1):1–11
Zanne AE, Tank DC, Cornwell WK, Eastman JM, Smith SA, FitzJohn RG et al (2014) Three keys to the radiation of angiosperms into freezing environments. Nature 506(7486):89–92
Zhang B, Liang C, He H, Zhang X (2013) Variations in soil microbial communities and residues along an altitude gradient on the northern slope of Changbai Mountain, China. PLoS One 8(6):e66184
Zhang C, Liu G, Xue S, Wang G (2016) Soil bacterial community dynamics reflect changes in plant community and soil properties during the secondary succession of abandoned farmland in the loess plateau. Soil Biol Biochem 97:40–49
Zhang R, Zhang Z, Shang K, Zhao M, Kong J, Wang X, Wang Y, Song H, Zhang O, Lv X (2021) A taxonomic and phylogenetic perspective on plant community assembly along an elevational gradient in subtropical forests. J Plant Ecol 14(4):702–716
Zhu Z-X, Nizamani MM, Sahu SK, Kunasingam A, Wang H-F (2019) Tree abundance, richness, and phylogenetic diversity along an elevation gradient in the tropical forest of Diaoluo Mountain in Hainan, China. Acta Oecol 101:103481
Zong S, Wu Z, Xu J, Li M, Gao X, He H, Du H, Wang L (2014) Current and potential tree locations in tree line ecotone of Changbai Mountains, Northeast China: the controlling effects of topography. PLoS One 9(8):e106114
Acknowledgements
Thanks are due to Prof. S.P. Singh, Chairman, Central Himalayan Environment Association, Nanital, for his valuable intellectual inputs. Financial support under the Timberline project supported by the Ministry of Environment, Forest and Climate change (New Delhi) through G.B. Pant National Institute of Himalayan Environment and Sustainable Development (Almora), Uttarakhand, is gratefully acknowledged. We are thankful to the Head, Department of Botany, for facilities and encouragement.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Nanda, S.A., Reshi, Z.A. (2023). Patterns of Plant Taxonomic, Life-Form and Phylogenetic Diversity at a Treeline Ecotone in Northwestern Himalaya: Role of Aspect and Elevation. In: Singh, S.P., Reshi, Z.A., Joshi, R. (eds) Ecology of Himalayan Treeline Ecotone. Springer, Singapore. https://doi.org/10.1007/978-981-19-4476-5_9
Download citation
DOI: https://doi.org/10.1007/978-981-19-4476-5_9
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-4475-8
Online ISBN: 978-981-19-4476-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)