Abstract
The study aimed to assess dry matter dynamics of forests along treeline ecotone concerning environmental variability, and four representative forests, birch, kharsu oak, Himalayan fir and mixed forest along treeline ecotone (3250–3400 m asl) were selected in Kedarnath Wildlife Sanctuary, Western Himalaya. Temperature, precipitation and humidity data were recorded for complete 3 years (2008–2010). Field inventory for assessment of forest structure and dry matter was carried out in a 200 m transect in each forest along treeline using quadrats of different sizes for tree, shrub and herb layers. The biomass and net primary productivity (NPP) of woody vegetation was estimated using region-specific allometric equations and of the herb layer, by harvesting. Litter quantity and decomposition rates were assessed using litter traps and litter bags, respectively. The dry matter budget of treeline ecotone was represented using a compartment model. The mean annual temperature at treeline was recorded 6.6 ± 0.68 °C with warmest month average temperature of 12.6 ± 1.2 °C. The average forest biomass at ecotone was 33.27 ± 16.97 Mg/0.1 ha, ranged between 8.87 Mg/0.1 ha in open and early successional birch forest to 44.98 Mg/0.1 ha in dense canopy mixed forest. NPP ranged from 1.49 to 2.11 Mg/0.1 ha year−1. The birch forest exhibited the highest efficiency in annual dry matter accumulation in the canopy layer among all forests. The leaf litter decomposition rates (% weight loss) was highest for kharsu oak (74%) after 3 years. The study revealed that Western Himalayan treeline exhibit warmer summer and higher amounts of dry matter storage, production and decomposition rates. These treeline forests showed a higher dry matter assimilation rate and so the potential for carbon sequestration under a warming climate.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The alpine treeline ecotone represents the most prominent ecological transition zone in the high mountains. Beyond treeline ecotone, tree species cease to attain their ordinary growth due to unfavorable climatic conditions and only meadows comprising shrubs and herbs species occur. The alpine treeline ecotone exhibit decreased atmospheric pressure, reduced partial pressure of carbon dioxide, increased solar energy flux and increased ultraviolet radiation leading to low primary productivity (Stevens and Fox 1991). During the past few decades, the treeline ecotone has been of considerable ecological interest to researchers owing to their sensitivity to biotic and abiotic factors, particularly climate change (Holtmeier 2009; Körner 2012a). Climate change studies on Himalayan region (IPCC 2007) and particularly on Western Himalaya revealed substantial changes on climatic patterns, glacial dynamics, hydrology and forest ecosystems in recent past (Shekhar et al. 2010; Negi et al. 2012; Tewari et al. 2017) which reflects this region as highly susceptible to climate change impacts. Therefore, systematic ecological studies on diverse aspects of treeline, a sensitive vegetation transition zone, of this understudied region are of vital importance.
Studies conducted at treeline ecotone have described species composition, physiognomies, or climate responses in the Himalayan region (Schickhoff 2005; Li et al. 2008; Rai et al. 2012; Shi and Wu 2013; Gaire et al. 2014). Along treeline, low ambient temperature limits tree growth by preventing the utilization of stored non-structural carbohydrates to biomass (Hoch and Körner 2009). The biomass, net primary productivity and decomposition rates vary with species composition, land-use history, terrain complexity and snow deposition pattern (Speed et al. 2015; Liu et al. 2016). A large amount of carbon also remains on the litter layer at high altitudes due to low decomposition rates (Grafius and Malanson 2015). Increased regeneration of woody species along treeline, densification (Bharti et al. 2012) and upward shift in treeline position (Dubey et al. 2003; Li et al. 2008; Shi et al. 2008; Liang et al. 2011; Gaire et al. 2014; Suwal et al. 2016) has been observed in recent times in the Himalaya.
The carbon sink and source limitations have been of particular interest to the ecologists dealing with impacts of climate change on high altitude forests. At treeline, climate growth limitation by low temperature is more sensitive than the carbon fixation by photosynthesis (Körner 2012b) while carbon loss due to respiration is relatively less under prevalent low temperatures. The study of dry matter production and recycling of treeline ecotone is essential concerning prevailing environmental conditions at intra- and inter-annual scales. In-situ meteorological observations are therefore critical to develop linkages between treeline ecotone vegetation characteristics and environment to predict undesirable consequences of climate alteration.
In this study, we assessed the diversity, biomass, NPP, litterfall, litter decomposition and overall patterns of dry matter dynamics across treeline forests in the Western Himalaya concerning environmental conditions.
Methods
Study area
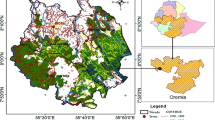
The study was conducted at Tungnath area in Kedarnath Wildlife Sanctuary (lat: 30° 29′ N–30° 30′ N, long: 79° 12′ E–79° 13′ E) in Uttarakhand. It represents treeline ecotone between 3250 and 3400 m asl in the Greater Himalayan range (Fig. 1). Topographically, the mountain ridges were east–west oriented having a predominance of south–west facing slopes. Geologically, the weathering bedrocks that provide the bulk of the loose material in these mountains are crystalline and metamorphic having sedimentary deposits. The soil was sandy loam in texture, with a high proportion of sand and silt, and generally acidic (pH 4–5). The mean annual temperature at the treeline (3300 m) was 6.65 ± 0.68 °C, while daily temperature ranged between − 8.9 and 25.6 °C. The mean temperature of the warmest month was 12.6 ± 1.2 °C (July). Annual precipitation was 2410 ± 432 mm, of which about 90% was received during June–September. The area remains under snow-cover for 85 ± 24 days in a year. Heavy snowfall and episodic severe hailstorms during November–April is a characteristic feature of the area.
The vegetation in the study area includes subalpine oak forest, fir forest, birch-Rhododendron forest and alpine scrub (Champion and Seth 1968). The dominant species at subalpine-treeline ecotones are birch (Betula utilis), Himalayan fir (hereafter fir, Abies spectabilis), kharsu oak (Quercus semecarpifolia) and mixed stands of Abies-Quercus-Betula. The tree layer was comprised of Betula utilis, Sorbus foliolosa and Abies spectabilis in the cool moist slopes whereas Q. semecarpifolia and Rhododendron arboreum on warm slopes. The shrub layer formed by Rhododendron campanulatum, Viburnum grandiflorum, Salix denticulata and Lonicera myrtillus of which R. campanulatum forms Krummholz vegetation. Along the forest edges Rhododendron anthopogon and R. lepidotum represented the moist alpine scrub vegetation. The herbaceous vegetation along the treeline formed by a variety of herbs and graminoid species. Danthonia cachemyriana is dominant in graminoids whereas herbaceous vegetation was formed by one or more species of Ranunculus, Bistorta, Polygonum, Potentilla, Geum, Primula, Cyananthus, Geranium etc. distributed along suitable micro-habitats. Study area forms habitat of endangered fauna species viz., Musk deer and Himalayan tahr. The Krummholz of Rhododendron campanulatum is a habitat of Himalayan monal pheasant. The area visited by local pastoral communities for livestock grazing during the summers.
Field sampling design
Four forest types (birch, fir, kharsu and mixed forests) were identified and selected based on species composition (Fig. 2). A 200 m belt transect in each forest type along treeline ecotone was identified in the different forest types. Ten quadrats of 10 × 10 m were laid at a fixed distance for tree sampling. A 5 × 5 m centre plot within each 10 × 10 m plot was laid out for the shrub layer. Besides, 25 plots of 1 × 1 m were randomly laid out in 200 m belt transect for the herb layer in each forest type (Fig. 3). The individuals with a circumference at breast height (cbh) > 20 cm and > 3 m height were considered as tree and cbh measured to estimate the basal cover. Individuals with < 20 cm girth were considered as tree saplings. For the shrub layer, all the individuals were counted and individuals with > 5 cm circumference at ground level were measured. All individuals of the herbs and tussocks of the grasses was counted. Tree diversity, density, basal area, and importance value index (IVI) were computed using standard methods.
Forest biomass estimation
The tree and shrub layer biomass was estimated using allometric equations developed for the subalpine-treeline zones of Western Himalaya (Singh et al. 1992; Singh and Singh 1992; Adhikari et al. 1995; Garkoti and Singh 1995) (Supplementary Table S1). These equations are based on a linear relationship between cbh and biomass of different components (bole, bark, branch, twig, foliage, stump root, lateral root and fine root) of individual tree species. These allometric equations are used to compute biomass of all the components of the species. For herb and seedling layer biomass estimation, ten 50 × 50 cm quadrats were harvested at monthly interval from April to September, and peak biomass was added to total forest biomass. The biomass of tree, sapling, seedling, shrub and herb was summed to get the total forest biomass. The results are presented in the units of sampled area and also computed to standard scales (per hectare) for system analysis and discussion and comparison with other studies.
Forest litter fall and standing litter mass estimation
To assess the litterfall patterns, ten wooden litter traps (50 cm × 50 cm × 15 cm size) were placed at different locations viz., under the canopy and in between the canopies of the trees in each forest. The fallen litter was collected monthly from May to October and in April of the subsequent year. The litter was categorized into five components: fresh leaf, wood, reproductive, moss and lichens and miscellaneous litter. The litter samples were oven-dried at 60 °C till constant weight to obtain dry litter mass from direct fall.
The litter accumulated on the forest floor was collected within three 50 × 50 cm plots and categorized into fresh leaf (intact leaves), wood, partially decomposed (with identifiable leaves of different species), more decomposed (unidentifiable) and miscellaneous litter (reproductive parts, epiphytes, mosses and bark) following Adhikari et al. (1995). The litter was oven-dried at 60 °C to obtain dry litter mass. Heavy snow cover limited the litter collection during winter months.
Net primary productivity estimation
Trees of different girth classes were marked for recording girth increment in each forest type at 1.37 m height above the ground level and measured for two consecutive years (B1) and (B2). The circumference of shrub species was estimated at ground level. The biomass of different components (aboveground: bole, bark, branch, twig and foliage; belowground: stump root, lateral root and fine root) was computed using the allometric equations. The net change in biomass (B = B2− B1) yielded annual biomass accumulation. The annual litterfall (e.g., leaf, twig, branches) were added to respective components. The tree and shrub fine root (< 5 mm in diameter) production rate were estimated at monthly intervals using soil cores. In each forest type, three steel core of 10 cm diameter were used to dig the soil up to 30 cm depth (Gower et al. 1992) and fine roots were extracted. For the herbaceous layer, the peak biomass (above and belowground) corresponds to the net herb layer production. The sum of the net production values of tree, shrub and herb layers yielded the total net primary productivity of different forest types. For the system analysis, vegetation layers of each forest were divided into three components viz, tree (including saplings), shrub and herbs (including seedlings).
Litter decomposition and turnover rates
The litter bag method was used to study litter decomposition and mineralization process (Witkamp and Olsen 1963). The mature and senesced leaves were plucked from the significant tree and shrub branches. About 120 nylon bags of 10 × 10 cm size (with 1 mm mesh size) containing 5 g air-dried mature and senesced leaf litter were placed randomly at different forest sites for each species. Three replicates of litterbags were retrieved monthly for consecutive 3 years except for winter months. The degradation rates of leaf litter and nutrient dynamics for N, P, K, Na, Ca and Mg in the decomposing leaves and soil at different depths were analysed in the laboratory. The turnover rate and turnover time of the litter on the forest floor were estimated following Olson (1963).
Meteorological data
The ambient air and soil temperature data was recorded through automatic HOBO® data loggers (Onset Inc., USA) at hourly intervals and daily readings averaged to get the mean daily temperature. The soil temperature was recorded at 10 cm depth. Humidity was measured using hygrometer and rainfall through rain gauge stationed near treeline (3300 m asl.).
Results
Treeline structure, diversity and dry biomass patterns
A total of eight tree species and 14 shrub species were recorded across four forest types. The tree species richness across the forest types varied from three (birch) to five (mixed forest) and shrub species richness ranged between 5 (fir) and 12 (birch). Total basal area (TBA, m2 0.1 ha−1) was found between 0.64 (birch) to (5.5) fir and tree densities (individuals 0.1 ha−1) between 34 and 78. Importance value index (IVI) computation revealed single tree species dominance in treeline forests except for mixed forest. Sapling density (individuals 0.1 ha−1) was between 1 (fir) to 110 (birch) and similar trends for shrub density in these forests.
The tree layer biomass [Megagram (Mg)] for the sampled area (0.1 ha) in birch, fir, kharsu and mixed forest estimated 3.2, 36.5, 31.4 and 38.7, respectively. Dominant species contributed 73–96% to total tree layer biomass except in the mixed forest. The shrub layer biomass ranged between 0.14 and 0.30 Mg/25 m2. The herb layer biomass was fractional of the total forest biomass. The total forest biomass (Mg 0.1 ha−1) of treeline forests was 8.8, 44.9, 34.8 and 44.5 for birch, fir, kharsu and mixed forests, respectively.
The annual average standing litter mass in the treeline forests was between 0.61 and 0.82 kg m−2 and seasonally it varied across the forest types. The highest litter mass was recorded in the rainy season for birch, kharsu and mixed forest (0.74, 1.02, 0.69 kg m−2, respectively) and during summer for fir forest (0.82 kg m−2). A significant proportion of litter was under partially and more decomposed categories throughout the year. The annual turnover rate of litter on the forest floor was 31%, 40%, 48% and 52% for the birch, fir, kharsu and mixed forests with a turnover time of 3.2, 2.5, 2.1 and 1.9 years, respectively.
Forest NPP, litter fall and decomposition patterns
NPP of treeline forests was 1.9 ± 0.24 Mg/0.1 ha year−1 which was lowest in birch and highest in mixed stands. The tree layer NPP was 0.8–1.5 Mg/0.1 ha year−1. A significant proportion of tree aboveground NPP was formed by foliage (34–51%). The fine roots contributed significantly (77–86%) in total belowground production. The sapling layer production was minimum in fir (0.02 Mg/0.1 ha year−1) and maximum in birch forest (0.17 Mg/0.1 ha year−1). The shrub layer production across the forests was 0.45–0.80 Mg/25 m2 year−1 and R. campanulatum contributed 15.7–99.5% in shrub layer productivity of all the forests whereas Viburnum grandiflorum dominated in birch forest (67.3%). The herb layer production (kg m−2 year−1) varied from 0.23 to 0.34, of which the aboveground part was accounted for 46.5–83.8%.
The biomass accumulation ratio (BAR, the proportion of aboveground biomass to net annual production) expresses accumulation of persistent and particularly woody tissues in terrestrial forests (Whittaker 1975). In the treeline forests, BAR were 5.8, 25.9, 18.1 and 21.1 for birch, fir, kharsu and mixed forests. The lowest BAR in birch forest depicts the young growth with high efficiency of biomass accumulation as compared to fir, kharsu and mixed forests.
The annual litterfall was 2.2 ± 0.2 kg m−2 year−1 having the highest in kharsu (0.75) and lowest in birch forests (0.28). The seasonal pattern of the litterfall was highly variable across the forest types. In birch forest peak wood litterfall was recorded during winter (October to April) which contributed 44% to the annual litterfall followed by leaf fall (33.5%). Leaf fall was maximum during rainy season (July–September) for birch and during summer for kharsu and mixed forests. In mixed forest leaf litterfall was about 50% of the total litterfall. The proportion of leaf and wood fall in fir forest was 43.4% and 29.6%, respectively, where peak leaf and wood fall was recorded during winter months.
Leaf litter decomposition rate was highest for Q. semecarpifolia (24.5%) and lowest for R. arboreum (3.8%) after first 4 months of leaf fall. Decomposition in foliage litter of A. spectabilis was accelerated after 4 months and reached 26.2% after 8 months. In other species highest rate of degradation (11–33%) was observed after 8 months of incubation. The 50% degradation in the leaf litter of B. utilis, R. arboreum, A. spectabilis, Q. semecarpifolia and R. campanulatum occurred after 30, 28, 27, 25 and 24 months. After nearly 3 years, the rate of degradation (% day−1) was 0.07% (after 942 days), 0.06% (823 days), 0.07% (943 days), 0.07 (976 days) and 0.08% (884 days) for these species, respectively (Fig. 4).
The rate of nutrient fluxes (release and accumulation) varied significantly across the species. The initial concentration of N in the leaf litter ranged between 0.5% (R. arboreum) to 1.7% (B. utilis). During decomposition, accumulation of N and P on the leaf litter was more than the loss of these nutrients whereas K showed maximum release and negligible accumulation in any stage of decomposition in all the species. Except small amount of releases, N accumulation reached up to 47% higher of the initial concentration in R. campanulatum. No release of P was recorded in the leaf litter of A. spectabilis and R. arboreum at any stage of decomposition and it reached up to 2.4 times higher concentration at later stages. K showed continuous release in all the species after initial accumulation in some species. The initial level of Ca was between 0.87 and 1.88% and it showed least changes in strength during any stages of decomposition but few higher peaks in B. utilis and A. spectabilis observed. Initially, Mg concentration was ranged between 0.09 and 0.15% and its high accumulation (3.7 fold) was found in B. utilis in later stages. The most top release of Mg was recorded in both the species of Rhododendron. All the species showed accumulation of Na which was highest for A. spectabilis with 71% higher, followed by B. utilis (46%). The release of Na was highest in Q. semecarpifolia. In both the Rhododendron species high accumulation and no release of Na was observed. The patterns of nutrient dynamics with litter decomposition are presented in Fig. 5.
System analysis of dry matter dynamics
The patterns of dry matter distribution and flow in various vegetation layers of four treeline forests are represented in the compartment model (Fig. 6). Solar radiation, the source of energy for autotrophic systems, was to the tune of 5.04 kWh m−2 day−1 as derived from Surface Meteorology and Solar Energy, NASA (http://eosweb.larc.nasa.gov/sse/). The proportional contribution of tree layer to the total forest biomass ranged between 35.6% (birch forest) to 90.4% (kharsu forest). Shrub layer had highest proportional contribution to the total biomass in birch forest (53.3%) where herb layer also had the highest proportion (6.1%) among all forests. The shrub layer and herb layer contributed lowest to the NPP in kharsu (3.5%) and fir forest (0.9%), respectively. Being a canopy layer, the maximum contribution to the NPP was due to trees, which were 54.6, 65.6, 68.5 and 71.9% in birch, fir, kharsu and mixed forest, respectively. The herbaceous layer had a second significant proportion to the NPP in birch, kharsu and mixed forests (22.8, 12.8 and 13.1%, respectively), whereas shrub layer was a second major contributor to fir forest biomass (18.3%). In the aboveground production of tree layer, the foliage had the highest contribution for fir, kharsu and mixed forests (65.6, 68.5 and 71.9%, respectively), whereas in a birch forest twig contributed the most (36.9%). The annual dry matter return through the litter formation were 6.6, 8.0, 9.3 and 10.5 Mg ha−1 in birch, fir, kharsu and mixed forests, respectively. In the birch forest, the herb layer had the highest proportion (43.9%) in the annual return of the biomass to the soil, whereas in other forests tree layer apportioned the maximum viz., 59.1% in fir, 80.6% in kharsu and 67.2% in mixed forests. The annual restitution of net biomass production by the tree layer ranged from 18 to 40%, by shrub layer 1.6–12% and herb layer 8–19%. The turnover rate (Mg ha−1 year−1) of the forest floor litter due to decomposition were 2.1, 3.2, 4.5 and 5.5 for birch, fir, kharsu and mixed forests, respectively. In an annual cycle of litter degradation and decay, 4.5–4.9 Mg ha−1 litter was carried over to the next year in these forests.
Compartment model of dry matter distribution in the forests at treeline in western Himalaya. Rounded rectangles represent compartments for standing state of dry matter, arrows represent net annual flux rate. Circles represents total solar insolation incident, rectangle represents total net production (kg ha−1 year−1) and heptagon represents total disappearance (kg ha−1 year−1). Compartment values area in kg ha−1 and flow rates kg ha−1 year−1 (a birch, b fir, c kharsu oak, and d mixed)
Seasonality in herb layer and fine root productivity
Perennial and thick rhizome bearing herb species viz., Bistorta, Selinum, Tanacetum, as well as tall forbs (Polygonum, Hackelia, Impatiens, etc.) contributed significantly to the herb layer density and biomass at treeline. Due to winter snow cover, aboveground biomass production initiates after the snowmelt in April–May months. Perennial belowground parts constitutes a substantial proportion of the live biomass. The average monthly aboveground herb layer productivity (kg m−2) ranged from 0.03 (April) to 0.18 (September). Peak herb layer productivity across the forest types was varied between 0.11 and 0.29 kg m−2. The peak aboveground biomass coincided with soil and air temperatures (Fig. 7). The productivity (kg m−2) was highest in September for birch (0.29), kharsu (0.15) and mixed (0.19) forests whereas it has peaked during June in fir forest. Monthly root productivity across the forest types ranged between 0.05 and 0.48 kg m−2 and the maximum was in the kharsu forest. The peak root productivity was in June to September for different forest types (Fig. 8).
Seasonality in litter fall patterns
Litterfall patterns across the months varied across the forest types due to differences in the phenological cycle of the species. The peak leaf falls in kharsu forest was in May coinciding the rise in the air and soil temperature whereas in case of birch forest it was with drop in temperature at the end of growing season during September–October. Peak woodfall was occurred during the winter and monsoon months (Fig. 9).
Discussion
The mean annual ambient temperature at the treeline position in the study area was recorded at 6.6 °C. This is within the global treeline limit of 6–8 °C (Körner and Paulsen 2004). However, the warmest month temperature was recorded at 12.6 ± 1.2 °C, which is nearly 2 °C higher as compared to global treelines (10 °C). Similar observations were also made in Central Himalaya (Müller et al. 2016). The higher summer temperature of the growing season may lead to higher biomass accumulation. In general, the dominant forests in the study area stored a significantly high amount of standing dry matter except to the birch forest because of its sparse growth. Studies carried at treeline with similar elevations and temperature conditions have reported a lower quantity of biomass (Zhang et al. 2013; Rai et al. 2018). Higher biomass (521 ± 115 Mg ha−1) similar elevation range (3100–3400 m) is reported for oak treeline in western Himalaya (Sharma et al. 2018).
The sapling density was low (1–26 individuals/0.1 ha) fir, oak and mixed forest while higher in the birch forests (110) owing to sparse growth. Among all shrub species Rhododendron campanulatum contributed the maximum to the total biomass of the treeline forests. Profuse growth of herbs including tall forbs (Bistorta, Selinum, Tanacetum, Polygonum, Hackelia, Impatiens, etc.), indicated suitable soil nutrients and moisture conditions. Increased herb layer production in open canopy conditions along the treeline as compared to dense forests is also reported from China in similar circumstances (Deng et al. 2006). The fine root production is high due to their high temporal turnover owing to a short life span and it recorded more soils rich in nutrients mainly N (Supplementary Table S2).
The biomass accumulation efficiency of trees is an essential functional character that exhibits the ability of the forests to accumulate persistent tissues annually. The biomass accumulation ratio in forests was in the range of 10–50 (Whittaker 1966), where lower values reflect higher accumulation efficiencies. Birch, an early successional, deciduous species accumulated biomass more efficiently as compared to other forests. The initial response of birch in terms of an extended growing period is reported based on phenological studies (Rai et al. 2013) and studies said that future climate warming would be suitable for the expansion of birch forests in the Himalayan region (Hamid et al. 2019).
Treeline zone is known to have slow decomposition rates and high accumulation of soil organic matter on the forest floor (Prichard et al. 2000) and it plays an essential role in maintaining the nutrient balance of the soil. In the present study, a significant proportion of dry matter was recorded on the forest floor with high variability in nutrient concentration across species which likely to influenced the decomposition rates and turnover of the dry matter. The colonization of litter by decomposers (fungi, bacteria and nematodes) primarily regulates the litter degradation and release of nutrients to the soil (Tian et al. 1998; Jeong et al. 2015). A sharp rise in litter degradation rates was recorded during the summer months. Gelifluction and heavy monsoon fall is a unique feature of the subalpine-alpine region of the Himalaya which maintains the higher soil moisture, a condition favorable for the growth of decomposers. These conditions regulate and enhance the litter turnover rates in the Himalayan region as compared to high altitude treelines across the globe (Pinos et al. 2017).
The seasonality was observed to influence production as well as the decomposition processes of this transition zone. However, the marked changes were found in the herbs and fine root production. Except for fir forest, herb layer productivity was peaked in August–September whereas fine root production was more or less similar from June to September in all forests. The peak litter decomposition was also recorded during this period making higher availability of the nutrients for growth of herbs and fine roots. Open canopy conditions along ecotone favored the growth of herb layers which compensated low tree layer productivity along the treeline.
The system analysis demonstrated the flow and transfer of dry matter in the treeline forests. The treeline ecotone is represented by both old-growth and early successional forests. However, old-growth fir and oak forests contributed mostly to the dry matter storage and production of ecotone while the first successional birch forest, owing to their sparse growth contribute less to overall dry matter storage but showed higher efficiency in assimilation. Old-growth mixed forests contribute significantly to the forest floor dry mass and the dynamics vary with the composition and ages of forest (Norden et al. 2015; Schilling et al. 2016; Souza et al. 2019). This analysis depicts the potential in old-growth and climax forests for stability and equilibrium in dry matter during the assimilation potential of dry matter in early successional species in future scenarios.
Conclusion
The present study inventoried structural and functional characteristics of alpine treeline ecotone vegetation of Western Himalaya with simultaneous observations on local climatology. The study observed that treeline ecotone experiences higher summer temperatures and exhibit higher efficiencies in dry matter production and nutrient cycling as compared to other treelines of the globe. The mature stands of hardwood species showed potential for higher storage and production of the dry matter while primary colonizer softwood forest showed rapid growth and higher efficiency in assimilation hence, higher ability in dry matter dynamics.
References
Adhikari BS, Rawat YS, Singh SP (1995) Structure and function of high altitude forests of Central Himalaya I. Dry matter dynamics. Ann Bot 75:237–248
Bharti RR, Adhikari BS, Rawat GS (2012) Assessing vegetation changes in timberline ecotone of Nanda Devi National Park, Uttarakhand. Int J Appl Earth Obs Geoinf 18:472–479
Champion HG, Seth SK (1968) A revised survey of the forest types of India. Manager of Publications. Govt. of India, New Delhi
Deng K-M, Shi P-L, Yang Z-L (2006) Biomass allocation and net primary productivities at treeline ecotone on the Changbai Mountains, Northeast China. J Nat Resour 21:942–948
Dubey B, Yadav RR, Singh J, Chaturvedi R (2003) Upward shift of Himalayan pine in Western Himalaya, India. Curr Sci 85:1135–1136
Gaire NP, Koirala M, Bhuju DR, Borgaonkar HP (2014) Treeline dynamics with climate change at the central Nepal Himalaya. Clim Past 10:1277–1290
Garkoti SC, Singh SP (1995) Variation in net primary productivity and biomass of forests in the high mountains of Central Himalaya. J Veg Sci 6:3–28
Gower S, Vogt TKA, Grier CC (1992) Carbon dynamics of rocky mountain douglas-fir: influence of water and nutrient availability. Ecol Monogr 62:43–65
Grafius DR, Malanson GP (2015) Biomass distributions in dwarf tree, krummholz, and tundra vegetation in the alpine treeline ecotone. Phys Geogr 36:337–352
Hamid M, Khuroo AA, Charles B, Ahmad R, Singh CP, Aravind NA (2019) Impact of climate change on the distribution range and niche dynamics of Himalayan birch, a typical treeline species in Himalayas. Biodivers Conserv 28:2345–2370. https://doi.org/10.1007/s10531-018-1641-8
Hoch G, Körner C (2009) Growth and carbon relations of tree line forming conifers at constant vs. variable low temperatures. J Ecol 97:57–66
Holtmeier F-K (2009) Mountain timberlines: ecology. Patchiness and dynamics. Springer, Amsterdam
IPCC (2007) Climate change 2007: synthesis report. Contribution of working groups I, II and III to the fourth assessment report of the intergovernmental panel on climate change. IPCC, Geneva
Jeong J, Lee IK, Lim JH, Kim C (2015) Nutrient dynamics by decomposing leaf litter at the Guemsan (Mt.) long-term ecological research site, Korea. For Sci Technol 11:97–103
Körner C (2012a) Treelines will be understood once the functional difference between a tree and a shrub is. Ambio 41:197–206
Körner C (2012b) Alpine treelines: functional ecology of the global high elevation tree limits. Springer, New York
Körner C, Paulsen J (2004) A world-wide study of high altitude treeline temperatures. J Biogeogr 31:713–732
Li MH, Xiao WF, Wang SG, Gen-Wei C, Cherubini P, Cai X-H, Liu X-L, Wang X-D, Zhu W-Z (2008) Mobile carbohydrates in Himalayan treeline trees I. Evidence for carbon gain limitation but not for growth limitation. Tree Physiol 28:1287–1296
Liang E, Wang Y, Eckstein D, Luo T (2011) Little change in the fir tree-line position on the southeastern Tibetan Plateau after 200 years of warming. New Phytol 190:760–769
Liu Y, Chen Y, Zhang J, Yang W, Peng Z, He X, Deng C, He R (2016) Changes in foliar litter decomposition of woody plants with elevation across an alpine forest-tundra ecotone in eastern Tibet Plateau. Plant Ecol 217:495–504
Müller M, Schickhoff U, Scholten T, Drollinger S, Böhner J, Chaudhary RP (2016) How do soil properties affect alpine treelines? General principles in a global perspective and novel findings from Rolwaling Himal, Nepal. Prog Phys Geogr 40:135–160
Negi GCS, Samal PK, Kuniyal JC, Kothyari BP, Sharma RK, Dhyani PP (2012) Impact of climate change on the western Himalayan mountain ecosystems: an overview. Trop Ecol 53:345–356
Norden N, Angarita HA, Bongers F, Martínez-Ramos M, La Cerda IG, Van Breugel M, Lebrija-Trejos E, Meave JA, Vandermeer J, Williamson GB, Finegan B, Mesquita R, Chazdon RL (2015) Successional dynamics in Neotropical forests are as uncertain as they are predictable. PNAS 112:803–818
Olson JS (1963) Energy storage and balance of producers and decomposers in ecological systems. Ecology 44:322–331
Pinos J, Studholme A, Carabajo A, Gracia C (2017) Leaf litterfall and decomposition of Polylepis reticulata in the treeline of the Ecuadorian Andes. Mt Res Dev 37:87–96
Prichard SJ, Peterson DL, Hammer RD (2000) Carbon distribution in subalpine forests and meadows of the Olympic Mountains, Washington. Soil Sci Soc Am J 64:1834–1845
Rai ID, Adhikari BS, Rawat GS, Bargali K (2012) Community structure along timberline ecotone in relation to micro-topography and disturbances in Western Himalaya. Not Sci Biol 4:41–52
Rai ID, Bharti RR, Adhikari BS, Rawat GS (2013) Structure and functioning of timberline vegetation in the Western Himalaya: a case study. In: Ning W, Rawat GS, Joshi S, Ismail M, Sharma E (eds) High-altitude rangelands and their interfaces in the hindu Kush Himalayas. ICIMOD, Nepal, pp 91–107
Rai S, Pandey A, Badola HK (2018) Biomass and carbon stock estimation across the timberline of Khangchendzonga National Park, Eastern Himalaya, India. Taiwania 63:311–320
Schickhoff U (2005) The upper timberline in the Himalayas, Hindu Kush and Karakorum: a review of geographical and ecological aspects. In: Broll G, Keplin B (eds) Mountain ecosystems. Studies in treeline ecology. Springer, Berlin, pp 275–354
Schilling EM, Waring BG, Schilling JS, Powers JS (2016) Forest composition modifies litter dynamics and decomposition in regenerating tropical dry forest. Oecologia 182:287–297
Sharma CM, Tiwari OP, Rana YS, Krishan R, Mishra KA (2018) Elevational behaviour on dominance—diversity, regeneration, biomass and carbon storage in ridge forests of Garhwal Himalaya, India. For Ecol Manag 424:105–120
Shekhar MS, Chand H, Kumar S, Srinivasan K, Ganju A (2010) Climate-change studies in the western Himalaya. Ann Glaciol 51:105–112
Shi P, Wu N (2013) The timberline ecotone in the Himalayan region: an ecological review. In: Ning W, Rawat GS, Joshi S, Ismail M, Sharma E (eds) High-altitude rangelands and their interfaces in the hindu Kush Himalayas. ICIMOD, Nepal, pp 108–116
Shi P, Körner C, Hoch G (2008) A test of the growth-limitation theory for alpine treeline formation in evergreen and deciduous taxa of the eastern Himalayas. Funct Ecol 22:213–220
Singh JS, Singh SP (1992) Forest of Himalaya. Gyanodaya Prakashan, Nanital
Singh SP, Singh RP, Rawat YS (1992) Patterns of soil and vegetation and factors determining their forms and hydrologic cycle in Nanda Devi Biosphere Reserve. Final technical report. Govt. of India, New Delhi
Souza SR, Veloso MD, Espírito-Santo MM, Silva JO, Sánchez-Azofeifa A, e Brito BG, Fernandes GW (2019) Litterfall dynamics along a successional gradient in a Brazilian tropical dry forest. For Ecosyst 6:35. https://doi.org/10.1186/s40663-019-0194-y
Speed JDM, Martinsen V, Hester AJ, Holand Ø, Mulder J, Mysterud A, Austrheim G (2015) Continuous and discontinuous variation in ecosystem carbon stocks with elevation across a treeline ecotone. Biogeosciences 12:1615–1627
Stevens GC, Fox JF (1991) The causes of treeline. Annu Rev Ecol Evol Syst 22:177–191
Suwal MK, Shrestha KB, Shakya LGR, Shrestha K, Bhuju DR, Vetaas OR (2016) Land-use change under a warming climate facilitated upslope expansion of Himalayan silver fir (Abies spectabilis (D. Don) Spach). Plant Ecol 217:993–1002
Tewari VP, Verma RK, von Gadow K (2017) Climate change effects in the Western Himalayan ecosystems of India: evidence and strategies. For Ecosyst 4:13
Tian X, Takeda H, Ando T (1998) Dynamics and carbon and nutrients in Abies needles and Betula leaf litters during the two-year decomposition period in a subalpine coniferous forest. Appl For Sci 7:43–48
Whittaker RH (1966) Forest dimensions and production in the Great Smoky Mountains. Ecology 47:103–121
Whittaker RH (1975) Community and ecosystems, 2nd edn. MacMillan, New York
Witkamp M, Olson JS (1963) Breakdown of confined and nonconfined oak litter. Oikos 14:138–147
Zhang Y, Gu F, Liu S, Liu Y, Li C (2013) Variations of carbon stock with forest types in subalpine region of southwestern China. For Ecol Manag 300:88–89
Acknowledgements
The authors are thankful to the Director, Wildlife Institute of India, Dehradun for providing necessary facilities and encouragements, and Uttarakhand Forest Department for permission to carry out fieldwork.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rai, I.D., Padalia, H., Singh, G. et al. Vegetation dry matter dynamics along treeline ecotone in Western Himalaya, India. Trop Ecol 61, 116–127 (2020). https://doi.org/10.1007/s42965-020-00067-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42965-020-00067-9