Abstract
We studied the altitudinal patterns of plant species richness and examined the effects of geometric constraints, area, and climatic factors on the observed richness patterns along the ridge of the Baekdudaegan Mountains, South Korea. Rapoport’s altitudinal rule was evaluated by examining the relationship between altitudinal range size and midpoint. We also examined the latitudinal effect on species richness. Plant data were collected from 1,100 plots along a 200–1,900 m altitudinal gradient along the ridge of the Baekdudaegan. A total of 802 plant species from 97 families and 342 genera were found. The altitudinal patterns of plant species richness along the ridge of the Baekdudaegan depicted distinctly hump-shaped patterns, although the absolute altitudes of the richness peaks vary somewhat among plant groups. While the mid-domain effect (MDE) was the most powerful explanatory variable in simple regression models, species richness was also associated with climatic factors, especially mean annual precipitation (MAP) and temperature (MAT) in multiple regression models. The relative importance of the MDE and climatic factors were different among plant groups. The MDE was more important for woody plants and for large-ranged species, whereas climatic factors were better predictors for total and herbaceous plants and for small-ranged species. Rapoport’s altitudinal rule and a latitudinal effect on species richness were not supported. Our study suggests that a combined interaction of the MDE and climatic factors influences species richness patterns along the altitudinal gradient of the Baekdudaegan Mountains, South Korea.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding geographical variation in patterns of species richness and distribution has been of interest to ecologists and biogeographers for decades (Rosenzweig 1995; Brown and Lomolino 1998; Colwell and Lees 2000), because this knowledge is important for biodiversity conservation and sustainable use (Grytnes and Vetaas 2002). In recent years, studies on the distribution patterns of species richness across latitudinal and altitudinal gradients and the factors that control these patterns have become very popular approaches in macroecology (Li et al. 2003; Carpenter 2005; Watkins et al. 2006; Grau et al. 2007). In particular, research in mountainous areas has played pivotal roles in understanding the spatial patterns of species richness and their ecological determinants. Altitude is one of the most important factors affecting species richness patterns in mountain ecosystems, because it drives drastic changes in climate (temperature, water availability) as well as overall area. Many studies have documented the altitudinal species richness patterns of plants (Bhattarai and Vetaas 2003, 2006; Oommen and Shanker 2005; Grytnes et al. 2006; Wang et al. 2007), mammals (McCain 2004, 2005, 2007; Rowe 2009), birds (Lee et al. 2004; McCain 2009), and invertebrates (Sanders et al. 2003; Aubry et al. 2005; Beck and Chey 2008; Liew et al. 2010) and different patterns have been observed in different taxa and regions.
Rahbek (1995, 2005) identified three main patterns of altitudinal richness: (1) a monotonic decrease with increasing altitude, (2) a plateau at low altitudes, and (3) a ‘hump-shaped’ distribution with high richness at intermediate altitudes. Although many studies claimed the importance and evidence for Rapoport’s altitudinal rule (Stevens 1992), which has often been used to explain the first two patterns, the hump-shaped pattern is reported to be the most common of these altitudinal patterns. However, the causes of the hump-shaped peak of species richness and the location of the peak are still not fully understood (Kluge et al. 2006).
The many hypotheses that have been proposed to explain the altitudinal patterns of species richness fall into two broad categories, Rapoport’s altitudinal rule and those considering climatic and spatial factors. Rapoport’s altitudinal rule suggests that climates at higher altitudes are more variable, so species at higher altitudes must be able to tolerate a broad range of climatic conditions and therefore have larger altitudinal ranges. Consequently, richness is inflated at low altitudes and species richness decreases with altitude (Stevens 1992). Climatic factors such as temperature and precipitation have been commonly correlated with species richness along altitudinal gradients (Li et al. 2009). Climatic factors can control species distributions directly when they exceed the physiological tolerances of species and affect photosynthetic activity and biological processes directly (Rowe 2009). Several studies have found that the area of altitudinal bands explained a large proportion of the variation in species richness (Bachman et al. 2004; Fu et al. 2004; Kattan and Franco 2004), similar to the well-known species-area relationships. Recent studies suggested that the mid-domain effect (MDE) was highly effective at explaining altitudinal patterns of species richness (McCain 2004, 2009; Kluge et al. 2006). The MDE postulates that geometric constraints on species ranges within a bounded domain will yield a mid-domain peak in richness regardless of ecological factors (Colwell and Hurtt 1994; Colwell and Lees 2000; Colwell et al. 2004b).
Although climatic factors, the MDE, area, and Rapoport’s altitudinal rule are frequently cited in studies of species diversity on mountains, a single factor cannot fully explain the species richness patterns along altitudinal gradients. Recent rigorous comparative studies suggested that the interactions of multiple factors drive altitudinal patterns of species richness in mountain ecosystems (Cardelús et al. 2006; Fu et al. 2006; Kluge et al. 2006; Watkins et al. 2006; McCain 2007, 2009; Rowe 2009).
In this study, we examined the distribution of terrestrial plants along an altitudinal gradient along the ridge of the Baekdudaegan Mountains (hereinafter referred to as ‘the Baekdudaegan’), South Korea. Using field surveys of plant data, we aimed to (1) explore the altitudinal patterns of species richness for total, woody, and herbaceous plants along the ridge of the Baekdudaegan; (2) evaluate the effects of the MDE, area, and two climatic factors (MAP and MAT) on the altitudinal patterns of plant species richness; (3) statistically evaluate the respective contributions of those spatial and climatic factors; and finally (4) examine the relationship between altitudinal range size and midpoint to test Rapoport’s altitudinal rule, and determine whether species richness patterns are related to latitude.
Methods
Study area
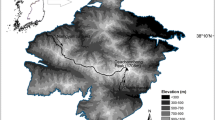
The study transect covered the main ridge of the Baekdudaegan Mountains (35° 15′–38° 22′N, 127° 28′–129° 3′E) in South Korea (Fig. 1). The Baekdudaegan consists of about 487 mountains, hills, and peaks along the Korean peninsula and are a major resource for forest biodiversity (Korea Forest Research Institute 2003). The protected area of the Baekdudaegan was designated in September 2005 by the Korea Forest Service; the total protected area, including the main ridge, covers 2,634 km2 (1,712 km2 core area and 922 km2 buffer zone). The main ridge extends about 650 km from Hyangnobong Peak (1,287 m above sea level, a.s.l) to Mt. Jiri (1,917 m a.s.l) in South Korea. Therefore, one can travel along the ridgelines without crossing any rivers or streams. The altitudinal gradient of the main ridge extends from 200 to 1,909 m a.s.l. based on a digital elevation model (DEM) generated using a mosaic of 1:25,000 topographical maps produced by the National Geographic Information Institute that cover the study area.
Location, topography, and climate diagrams of the study area, the ridge of the Baekdudaegan Mountains in South Korea. Relationships between altitude and a area, b mean annual precipitation (MAP), c mean annual temperature (MAT), and d latitude. Area, MAP, and MAT were calculated for each altitudinal band in an imaginary 100 m-wide transect along the ridge of the Baekdudaegan
The Baekdudaegan in South Korea belongs to a mountain ecoregion and temperate deciduous forest biome (Yim and Kira 1975; Shin 2002). The soil consists of granite, granite gneiss, and highly deformed and recrystallized sedimentary rocks (Shin 2002). Even though the natural environment of the Baekdudaegan is not well known because of insufficient survey data, the Baekdudaegan has many biodiversity hotspots and offers natural habitats for abundant and varied fauna and flora. The Korea Forest Research Institute (2003) reported that a total of 1,477 plant species were distributed along the Baekdudaegan, accounting for 35.2 % of the vascular plant diversity on the Korean peninsula.
The vegetation on the Baekdudaegan can be categorized into 49 communities, including 7 planted communities (e.g., the Larix kaempferi community) and 42 natural vegetation communities (e.g., the Quercus mongolica community). The Korea Forest Research Institute (2003) divided the Baekdudaegan in South Korea into three parts based on characterized plant community groups: (1) the northern part, characterized by Acer komarovii and Betula ermanii, (2) the central part, characterized by Acer pseudosieboldianum and Fraxinus rhynchophylla, and (3) the southern part, characterized by Abies koreana and Fraxinus mandshurica. The vegetation on the Baekdudaegan can also be divided into four major zones along an altitudinal gradient. These altitudinal vegetation zones include (1) temperate (montane) deciduous broad-leaved and pine forest (<550 m a.s.l.) dominated by P. densiflora and Rhus tricocarpha, (2) temperate deciduous broad-leaved and coniferous mixed forest (550–1,100 m a.s.l.) dominated by Q. mongolica, Quercus serrata, Pinus koraiensis, and Abies holophylla, (3) sub-alpine coniferous forest (1,100–1,600 m a.s.l.) dominated by Taxus cuspidata, A. koreana, and Abies nephrolepis, and (4) alpine forest (>1,600 m a.s.l.) dominated by Betula ermanii and Pinus pumila (Yim 1977; Kong 2008).
Plant data
For field sampling, an imaginary 100 m-wide transect was established in a north–south direction along the ridge of the Baekdudaegan, and the ridge was divided into 16 altitudinal bands of 100 m intervals from 200 m a.s.l. to above 1,700 m a.s.l. Although sampling extended to 1,900 m a.s.l., the 1,700 m and higher range was considered to be a single band because only a small number of plots were sampled and few plant species were observed above 1,700 m a.s.l. Plant data were collected within every altitudinal band of this 100 m-wide transect from May 2005 to August 2009. Vegetation sampling was performed to cover the most common and specific physiognomic vegetation types in each 100 m altitudinal band. Data were obtained for a total of 1,100 plots of 400 m2. Within each plot, plants were surveyed according to the method of Braun-Blanquet (1965).
We divided the range of altitude into 100 m bands to examine the relationship between plant species richness and altitude. Plant data from the same altitudinal band were pooled, and the number of species observed in each band was considered to be a measure of richness. All analyses were conducted for each of three plant groups: (1) total, (2) woody and (3) herbaceous plant species. As in Jetz and Rahbek (2002), we also divided the species in each plant group into the halves with largest and smallest ranges, respectively. To evaluate sampling completeness and to control for sampling effort, two distinct methods of analysis were applied to plant data from each altitudinal band (Cardelús et al. 2006). In the first method, a sample-based rarefaction curve (Colwell et al. 2004a) was computed, and we compared richness among each altitudinal bands. In the second method, we computed nonparametric estimates to reduce the bias caused by undersampling using four incidence-based estimators, incidence-based coverage estimator (ICE), Chao2, Jackknife1, and Jackknife2. Nonparametric species richness estimates and sample-based rarefaction curves were computed from incidence data using EstimateS software version 8.2 (Colwell 2009).
Unlike many recent studies, we did not use interpolated species richness modified from actual distribution records. The underlying reason for interpolation is that gaps in altitudinal distribution are caused by undersampling (Kluge et al. 2006). However, several studies have reported three problems with interpolation (Grytnes and Vetaas 2002; Diniz-Filho et al. 2003; Kluge et al. 2006). First, it disrupts the crucial control of sampling area and intensity as species are added that were not in fact present in the plots. Second, interpolation might artificially increase richness to a higher degree at intermediate altitudes, because gaps are filled only between the lower and upper range limits; this basically assumes that no individuals of a species have been missed beyond the observed range limits, but that individuals have been missed at sampling points within the range limits. Third, species richness at nearby altitudes is more similar than at distant altitudes, and the resulting spatial autocorrelation inflates Type I errors. The spurious effects of autocorrelation increase when using interpolated distribution data. However, many studies on altitudinal richness patterns used interpolated data, and comparisons of such studies with our non-interpolated results might be difficult. Therefore, we also calculated the interpolated richness for total, woody and herbaceous plants (Table 1). Empirical and interpolated richness patterns showed the same pattern along the altitudinal gradient and were strongly correlated (total, R 2 = 0.92; woody, R 2 = 0.90; herbaceous, R 2 = 0.92; P < 0.001 in all cases). Thus, we presented results based only on the empirical richness values without interpolation in this study. Plant species checklists for each altitudinal band are listed in Supplementary Material 1.
Spatial and climatic factors
The two spatial factors used in this study were the MDE and area. The MDE null model was used to test the influence of geometric constraints on the spatial patterns of species richness along an altitudinal gradient. We used a novel, discrete MDE model based on Colwell and Hurtt’s (1994) continuous Model 2 that does not necessitate the use of interpolated ranges (Dunn et al. 2006; Fu et al. 2006). We avoided interpolation because it tends to inflate richness estimates in the middle of the domain more than at the limits of the domain, which might spuriously amplify or even create mid-domain peaks in richness (Grytnes and Vetaas 2002; Zapata et al. 2003). RangeModel software version 5 (Colwell 2006) was used for simulation. The simulation process was repeated 5,000 times, and we used expected mean richness and its 95 % confidence intervals to assess the effects of geometric constraints on the altitudinal gradient of species richness for each plant group. To test species–area relationships, we calculated the area of each altitudinal band in the 100 m-wide transect along the ridge of the Baekdudaegan. Calculations were performed using a Digital Elevation Model (DEM) with 3D analysis in ArcGIS.
Two climatic factors, including mean annual precipitation (MAP) and temperature (MAT), were also investigated with respect to species richness. We used digital climate maps produced by the Korea Meteorological Administration and National Center of Agrometeorology to extract the meteorological parameters for each altitudinal band. MAP data were 1981–2009 and MAT data were from 1971 to 2008. The spatial resolution of the raster data was 270 m for MAP and 30 m for MAT. MAP and MAT were calculated for each altitudinal band in the 100 m-wide transect along the ridge of the Baekdudaegan.
To test the effects of individual factors (the MDE, area, MAP, and MAT) on altitudinal patterns of plant species richness, we performed simple ordinary least squares (OLS) regression of empirical species richness for each plant group. Multiple OLS regressions were also computed to explore multivariate explanations for altitudinal patterns of plant species richness. In addition to OLS regressions, we computed simple and multiple conditional autoregressive (CAR) models. Recent studies have recommended autoregressive models, because they avoid inflation of type I errors and invalid parameter estimates due to spatial autocorrelation, and they identify the predictive power of the hypotheses for explaining geographic richness patterns of different taxa (Jetz and Rahbek 2002; Fu et al. 2006; Marini et al. 2010). OLS and CAR models were performed with SAM version 4.0 (Rangel et al. 2010).
Rapoport’s altitudinal rule
To examine the relationship between the altitudinal ranges of plant species and altitude, we calculated the altitudinal range size of a species as the difference between its maximum and minimum altitudes. To overcome statistical non-independence of spatial data, we used the ‘midpoint method’ as a measure of central tendency (Rohde et al. 1993), and the altitudinal midpoint was calculated as the mean of maximum and minimum altitudes at which the species was recorded. The relationship between altitudinal range size and midpoint was evaluated using simple regression analysis as well as second-order polynomial models. When the relationship between both variables is positive, Rapoport’s altitudinal rule is predicted.
Latitudinal effect
Although we presumed that the altitude was the main gradient for determining plant species richness patterns along the Baekdudaegan ridge, we could not rule out a latitudinal effect on species richness, because of the long latitudinal range of the study area (almost 3°). Thus, we divided the range of latitude into seven 0.5° bands to examine the relationship between species richness and latitude. Plant data from the same latitudinal band were pooled, and the number of species in each band was considered to be a measure of richness. Simple regression analysis was used to evaluate the relationship between the two variables. A statistically significant relationship between the two variables would indicate the existence of a latitudinal effect.
Results
Altitudinal richness patterns of plant species
A total of 802 plant species belonging to 97 families and 342 genera were recorded from 1,100 plots along the altitudinal gradient of the Baekdudaegan ridge. More than half of these species were herbaceous (69 %; 62 families, 249 genera, and 554 species), while woody species account for 31 % (47 families, 99 genera, and 248 species) (Table 1 and Supplementary Material 1).
Interpolated richness and nonparametric estimators (ICE, Chao2, Jackknife1, and Jackknife2) were similar to the patterns of empirical richness, but the interpolated and nonparametric numbers of species were somewhat higher in each altitudinal band (Table 1 and Supplementary Material 2). Woody and herbaceous plant species had different contributions to total species richness patterns. There was a higher correlation coefficient between total and herbaceous plant species than total and woody plant species (total vs woody, R 2 = 0.80, P < 0.001; total vs herbaceous, R 2 = 0.97, P < 0.001). Overall species richness of total, woody and herbaceous plants showed hump-shaped patterns with maximum richness between 800 and 1,100 m a.s.l. (Fig. 2). The peak in overall woody species richness was at lower altitudes than that of total or herbaceous overall species richness. When plant species were partitioned into two range-size categories, large-ranged and small-ranged species contributed to overall species richness patterns with different magnitudes (Fig. 2). There were higher correlation coefficients between overall and large-ranged species richness (total, R 2 = 0.95; woody, R 2 = 0.97; herbaceous, R 2 = 0.97; P < 0.001 in all cases) than between overall and small-ranged species richness (total, R 2 = 0.81; woody, R 2 = 0.73; herbaceous, R 2 = 0.87; P < 0.001 in all cases). The MDE null model showed deviation of the empirical species richness from simulated richness (Fig. 2). The analysis revealed that more than 40 % of the data points fell outside the 95 % confidence interval of the MDE null model for overall species richness (50, 44 and 81 % for total, woody, and herbaceous plants, respectively). These large deviations were also observed for large-ranged (38, 38 and 63 % for total, woody and herbaceous plants, respectively) and small-ranged (56, 31 and 63 % for total, woody and herbaceous plants, respectively) species richness. However, the relationship between empirical and predicted richness was correlated significantly for all plant groups (Table 2).
Empirical species richness, predicted richness (computed from 5,000 randomizations), and the 95 % confidence intervals for the predicted mid-domain effect (MDE) richness as a function of altitude for total (a–c), woody (d–f), and herbaceous (g–i) plant species and for overall (a, d, g), large-ranged (b, e, h), and small-ranged (c, f, i) species
Although the species accumulation curves (sample-based rarefaction curves) for all altitudinal bands failed to reach asymptotes, these curves indicated clear mid-altitudinal peaks between 800 and 1,300 m for total, woody, and herbaceous plants (Fig. 3 and Supplementary Material 3).
Richness patterns with spatial and climatic factors
The simple OLS and CAR models yielded similar results for all plant groups. Based on simple linear regressions, total, woody, and herbaceous species richness were correlated strongly with the MDE and area. Furthermore, the explanatory powers of the MDE and area were less for small-ranged species than for large-ranged species and overall (Table 2).
The multiple OLS and CAR models also showed similar results across all datasets except for large-ranged total species, although the absolute magnitudes of the parameters varied somewhat between the two models (Table 3). The multiple regression models with all factors (model A) included the MDE, MAP, and MAT and explained more than 80 % of the variation in overall species richness of total, woody, and herbaceous plants. Area was the weakest predictor for explaining the variation in overall richness. A second model (model B) excluded the MDE but included area, MAP, and MAT and explained more than 80 % of the variation in overall species richness of total, woody, and herbaceous plants. Although there were several exceptions, the explanatory powers of climatic factors (MAP and MAT) were higher for total and herbaceous plant species, while the MDE was more important for woody plant species. When the species data were divided into large- and small-ranged species, the effects of the MDE and climatic factors in multiple regression models showed different patterns between the two different range-size groups. The explanatory powers of the MDE for large-ranged species were higher than for small-ranged species, whereas MAP and MAT were better predictors than the MDE for small-ranged species.
Rapoport’s altitudinal rule
The simple linear regression analysis between altitudinal range size and midpoint showed no significant relationships for any plant group. A second-order polynomial regression model showed maximum altitudinal ranges at intermediate altitudes, with minimum range sizes at extreme altitudes. The explanatory powers of simple regressions were lower than those of quadratic polynomial regressions (Fig. 4). These analyses do not support Rapoport’s altitudinal rule.
Latitudinal effect
Simple linear regression analysis between species richness and latitude showed no significant relationships for any plant group (Fig. 5). Furthermore, the relationships between mean latitudinal range size of plant species and latitude were also not significant (Supplementary Material 4). These analyses do not support the latitudinal effect on plant species richness patterns along the ridge of the Baekdudaegan.
Discussion
In this study, we explored altitudinal patterns of plant species richness at a regional scale using a large primary data set. Previous studies showing altitudinal patterns of species richness can be divided into two main groups of spatial scales: local and regional (Romdal and Grytnes 2007). Many previous studies aimed to explain mechanisms at very large regional scales, such as country and continent scales (Jetz and Rahbek 2002; Grau et al. 2007; Wang et al. 2007; McCain 2009; Rowe 2009; Alexander et al. 2011) using secondary distribution data from the literature associated with the study areas. The studies at regional scales cover larger areas and larger fractions of the total biota because they combine data from numerous collecting efforts (Karger et al. 2011). Basically, regional-scale studies, including our study, presume that the large-scale study area is a closed and massive mountain system from a macroecological perspective. Therefore, they focus on the altitudinal effect and consider that the effect of latitude on species richness is either not significant or extremely small (Marini et al. 2010). Indeed, in our study, plant species richness did not show significant relationships with latitude, nor was the relationship between mean latitudinal range size and latitude significant for any plant group. These results support the assumption that latitude is not a significant gradient for plant species richness patterns, at least along the ridge of the Baekdudaegan. Moreover, most studies on the relationship between species richness and latitude have been performed for wide latitudinal ranges of more than 10° at the continent or hemisphere scales (Stevens 1989; Buckley et al. 2003; Cruz et al. 2005).
The altitudinal patterns of species richness and their underlying causes have long been controversial issues in ecology and biogeography (Wang et al. 2007). Many studies have suggested that species richness peaks at intermediate altitudinal zones (Colwell and Lees 2000; Sanders 2002; McCain 2004), and some studies indicated that species richness decreases monotonically with increasing altitude (Stevens 1992). Our study also showed that plant species richness peaked at intermediate altitudes along the ridge of the Baekdudaegan, even though the absolute altitudes of the richness peaks varied somewhat among the three plant groups (total, woody, and herbaceous) and two range-size groups (large- and small-ranged species). At the most general level, our study adds to the growing evidence that plant species richness exhibits a strong hump-shaped altitudinal pattern in mountainous areas. This hump-shaped distribution has been explained as reflecting an optimal combination of spatial factors, such as the MDE and area (Wang et al. 2007), and climatic factors, including water and temperature (Bhattarai and Vetaas 2003; Kluge et al. 2006). The relative influence of each of these determinants of richness may vary among altitudinal gradients and taxa. Below we discuss how the MDE, area, climatic factors, and Rapoport’s altitudinal rule may influence altitudinal patterns of plant species richness along the ridge of the Baekdudaegan.
Spatial factors: the MDE and area
Although the empirical species richness deviated greatly from the MDE null model, the MDE was the most powerful factor in the simple linear regression and accounted for a significantly large proportion of the altitudinal patterns of total, woody, and herbaceous species richness in our study. In a recent work, Acharya et al. (2011) reported that, like other plant groups (Carpenter 2005; Kluge et al. 2006; Grau et al. 2007; Ah-Peng et al. 2012), tree species richness in the eastern Himalayas deviated greatly from that predicted by the MDE null model. The large deviation may be due to a large proportion of singletons and doubletons, i.e., species present in only one or two samples, respectively (Ah-Peng et al. 2012). The degree of deviation may also suggest that other factors (ecological, historical, and evolutionary) could explain the observed distribution pattern (Acharya et al. 2011). In our study, total and herbaceous plants had more singleton and doubleton species than woody plants (36, 27, and 41 % for total, woody, and herbaceous plants, respectively). Therefore, we suggest that these singletons and doubletons are likely to result in larger deviations for total and herbaceous plants than for woody plants.
Many studies have documented that the MDE is an important factor influencing species richness patterns along altitudinal gradients (Colwell and Lees 2000; Bachman et al. 2004; McCain 2004; Oommen and Shanker 2005; Cardelús et al. 2006; Watkins et al. 2006). The MDE presumes that hard boundaries cause more overlap of species ranges toward the center of a bounded geographical domain (Colwell et al. 2004b) and predicts a hump-shaped richness pattern with maximum richness at the intermediate altitudes of the mountain ecosystem (McCain 2004). Along the ridge of the Baekdudaegan, woody plant richness was correlated more strongly with the MDE using multiple regression models than were total or herbaceous plant richness. Also, the explanatory power of the MDE was stronger for large-ranged species than for small-ranged species in each of the three plant groups (total, woody and herbaceous species). Our results suggested that the MDE is a robust predictor of woody species richness and is effective in explaining altitudinal woody plant richness patterns. These results also confirmed the prediction of the MDE hypothesis that small-ranged species will show less of an MDE peak than large-ranged species (Jetz and Rahbek 2002; Colwell et al. 2004b; Cardelús et al. 2006; Dunn et al. 2006; Fu et al. 2006; Kluge et al. 2006; Watkins et al. 2006).
Area is a crucial factor determining altitudinal species richness patterns and can have both indirect and direct effects on species richness (Connor and McCoy 1979; Rahbek 1995). In our study, area was an important predictor of total, woody, and herbaceous plant species richness in simple regression models. However, area was a weak predictor of richness patterns in multiple regression models with the MDE from for all plant groups except for small-ranged woody species. This apparent contradiction may be due to the special relationship between the MDE and area. Because the MDE and area are highly correlated (R 2 > 0.55, P < 0.001 in all cases), the area effect may be substituted by the MDE in the multiple regression models. Therefore, we suspect that the effect of area was masked by the strength of the MDE, at least for the plant species in this study.
Climatic factors: MAP and MAT
Two climatic factors, MAP and MAT, were correlated only weakly with species richness patterns along the altitudinal gradients in simple regression models. However, in the multiple regression models, the two climatic factors contributed strongly to empirical richness patterns, especially for total and herbaceous plants, along the altitudinal gradients. Climate is an obvious factor controlling species distribution and richness in many areas, especially for vascular plants (Fang and Lechowicz 2006). Along the ridge of Baekdudaegan, the combined effects of precipitation and temperature may limit species richness at both extremes of the gradient, but in different ways: at lower altitudes by a reduction of humidity through high temperatures and at higher altitudes by low temperatures (Kluge et al. 2006; ZenHua et al. 2007). We concluded that an optimal range of temperature and precipitation exists at intermediate altitudes along the ridge of the Baekdudaegan and that favorable climate conditions at intermediate altitudes may lead to higher total and herbaceous plant species richness, because optimal climatic conditions lead to maximum productive energy available in the mountain ecosystem (Bhattarai et al. 2004; Kluge et al. 2006; ZenHua et al. 2007). Furthermore, McCain (2007) proposed a climatic model and advocated that appropriate climate conditions (e.g., precipitation and temperature) at the intermediate altitudes might contribute to the hump-shaped patterns. The intermediate altitudes may provide the best combinations of heat and water for plant growth and consequently allow a rate of higher resource use and the co-existence of more species (McCain 2007). Furthermore, when the species data were divided into large and small-ranged species, the relative contributions of the two climatic factors in multiple regression models were higher for small-ranged than large-ranged species. Fu et al. (2006) found that climatic factors were more important than spatial factors for small-ranged species.
Rapoport’s altitudinal rule
Our results did not support Rapoport’s altitudinal rule (Fig. 3). Because the plant species along the ridge of the Baekdudaegan that occurred at the lowest and highest mean altitudes tended to have smaller altitudinal ranges, and because only species at intermediate altitudes had large altitudinal ranges, an alternative and complementary explanation for the empirical pattern seen is the random placement of species’ altitudinal ranges along an altitudinal gradient, like the MDE. In any case, species ranges result from complex interactions among many factors, such as physiological traits, the complex evolutionary history of speciation and dispersal, and constraints resulting from continent shape (Webb and Gaston 2003). No general trends appear to exist for Rapoport’s altitudinal rule for all biological organisms, suggesting that the factors determining range size are complex and remain poorly understood (Grau et al. 2007).
Conclusion
Along the ridge of the Baekdudaegan, plant species richness had distinctly hump-shaped patterns along the altitudinal gradient, even though the absolute altitudes of the richness peaks varied somewhat among total, woody, and herbaceous plants. The MDE and climatic factors had good explanatory power and were the main potential factors determining the richness patterns of plant species in the regression analyses. The MDE was the most important explanatory factor for woody plants and for large-ranged species, whereas the combined interaction of the climatic factors was important predictors for total and herbaceous plants and for small-ranged species. Furthermore, Rapoport’s altitudinal rule and latitudinal effect were not supported for any plant groups. Even though the MDE and climatic factors were the primary drivers of the best simple and multiple regression models, discrimination among these factors was not possible through a simple comparison of the regression coefficients, because both the MDE and climatic factors were correlated strongly with plant species richness. Many studies have reported that altitudinal species richness patterns can be influenced by a series of climatic, spatial, and historical factors (Grytnes and Vetaas 2002; Rahbek 2005; Cardelús et al. 2006; Li et al. 2009; Acharya et al. 2011). In this study, we evaluated the relationships among spatial and climatic factors and the altitudinal richness patterns of plant species. However, historical evolutionary factors were not considered. Further study on the influence of evolutionary history, including historical contingency and niche conservatism, might help researchers gain a better understanding of the factors controlling the altitudinal distribution of plant communities from a macroecological perspective.
References
Acharya BK, Chettri B, Bijayan L (2011) Distribution patterns of trees along an elevation gradient Eastern Himalaya, India. Acta Oecol 37:329–336
Ah-Peng C, Wilding N, Kluge J, Descamps-Julien B, Bardat J, Chuah-Petiot M, Strasberg D, Hedderson TAJ (2012) Bryophyte diversity and range size distribution along two altitudinal gradients: continent versus island. Acta Oecol 42:58–65
Alexander JM, Kueffer C, Daehler CC, Edwards PJ, Pauchard A, Seipel T, Consortium M (2011) Assembly of nonnative floras along elevational gradients explained by directional ecological filtering. Proc Natl Acad Sci USA 108:656–661
Aubry S, Magnin F, Bonnet V, Preece RC (2005) Multi-scale altitudinal patterns in species richness of land snail communities in south-eastern France. J Biogeogr 32:985–998
Bachman S, Baker WJ, Brummitt N, Dransfield J, Moat J (2004) Elevational gradients, area, and tropical island diversity: an example from the palms of New Guinea. Ecography 27:299–310
Beck J, Chey VK (2008) Explaining the elevational diversity pattern of geometrid moths from Borneo: a test of five hypotheses. J Biogeogr 35:1452–1464
Bhattarai KR, Vetaas OR (2003) Variation in plant species richness of different life forms along a subtropical elevational gradient in the Himalayas, east Nepal. Global Ecol Biogeogr 12:327–340
Bhattarai KR, Vetaas OR (2006) Can Rapoport’s rule explain tree species richness along the Himalayan elevational gradient, Nepal? Divers Distrib 12:373–378
Bhattarai KR, Vetaas OR, Grytnes JA (2004) Fern species richness along a Central Himalayan elevational gradient. Nepal. J Biogeogr 31:389–400
Braun-Blanquet J (1965) Plant sociology. Hafner, New York
Brown JH, Lomolino MV (1998) Biogeography, 2nd edn. Sinauer, Sunderland
Buckley HL, Miller TE, Ellison AM, Gotelli NJ (2003) Reverse latitudinal trends in species richness of pitcher-plant food webs. Ecol Lett 6:825–829
Cardelús CL, Colwell RK, Watkins JE (2006) Vascular epiphyte distribution patterns: explaining the mid-elevation richness peak. J Ecol 94:144–156
Carpenter C (2005) The environmental control of plant species density on a Himalayan elevation gradient. J Biogeogr 32:999–1018
Colwell RK (2006) RangeModel: a Monte Carlo simulation tool for assessing geometric constraints on species richness. Version 5. User’s Guide and application published at: http://viceroy.eeb.uconn.edu/rangemodel
Colwell RK (2009) Estimate S: Statistical estimation of species richness and shared species from samples. Version 8.2. User’s Guide and application published at: http://purl.oclc.org/estimates
Colwell RK, Hurtt GC (1994) Nonbiological gradients in species richness and a spurious Rapport effect. Am Nat 144:570–595
Colwell RK, Lees DC (2000) The mid-domain effect: geometric constraints on the geography of species richness. Trends Ecol Evol 15:70–76
Colwell RK, Mao CX, Chang J (2004a) Interpolating, extrapolating and comparing incidence-based species accumulation curves. Ecology 85:2717–2727
Colwell RK, Rahbek C, Gotelli NJ (2004b) The mid-domain effect and species richness patterns: what have we learned so far? Am Nat 163:E1–E23
Connor EF, McCoy ED (1979) The statistics and biology of the species-area relationship. Am Nat 113:791–833
Cruz FB, Fitzgerald LA, Espinoza RE, Schulte JA (2005) The importance of phylogenetic scale in tests of Bergmann’s and Rapoport’s rules: lessons from a clade of South American lizards. J Evol Biol 18:1559–1574
Diniz-Filho JAE, Bini LM, Hawkins BA (2003) Spatial autocorrelation and red herrings in geographical ecology. Global Ecol Biogeogr 12:53–64
Dunn RR, Colwell RK, Nilsson C (2006) The river domain: why are there more species halfway up the river? Ecography 29:251–259
Fang JY, Lechowicz MJ (2006) Climatic limits for the present distribution of beech (Fagus L.) species in the world. J Biogeogr 33:1804–1819
Fu C, Wu J, Wang X, Lei G, Chen J (2004) Patterns of diversity, altitudinal range and body size among freshwater fishes in the Yangtze River basin, China. Global Ecol Biogeogr 13:543–552
Fu C, Hua X, Li J, Chang Z, Pu Z, Chen J (2006) Elevational patterns of frog species richness and endemic richness in the Hengduan Mountains, China: geometric constraints, area and climate effects. Ecography 29:919–927
Grau O, Grytnes JA, Birks HJB (2007) A comparison of altitudinal species richness patterns of bryophytes with other plant groups in Nepal, Central Himalaya. J Biogeogr 34:1907–1915
Grytnes JA, Vetaas OR (2002) Species richness and altitude: a comparison between null models and interpolated plant species richness along the Himalayan altitudinal gradient. Nepal. Am Nat 159:294–304
Grytnes JA, Heegaard E, Ihlen PG (2006) Species richness of vascular plants, bryophytes, and lichens along an altitudinal gradient in western Norway. Acta Oecol 29:241–246
Jetz W, Rahbek C (2002) Geographic range size and determinants of avian species richness. Science 297:1548–1551
Karger DN, Kluge J, Krömer T, Hemp A, Lehnert M, Kessler M (2011) The effect of area on local and regional elevational patterns of species richness. J Biogeogr 38:1177–1185
Kattan GH, Franco P (2004) Bird diversity along elevational gradients in the Andes of Colombia: area and mass effects. Global Ecol Biogeogr 13:451–458
Kluge J, Kessler M, Dunn RR (2006) What drives elevational patterns of diversity? A test of geometric constraints, climate and species pool effects for pteridophytes on an elevational gradient in Costa Rica. Global Ecol Biogeogr 15:358–371
Kong WS (2008) Biogeography of Korea Plants, 2nd edn (in Korean). GeoBook, Seoul
Korea Forest Research Institute (2003) Ecological aspects of Baekdu Mountains in Korea and delineation of their management and conservation area (in Korean). Korea Forest Research Institute [report no. 198], Seoul
Lee PF, Ding TS, Hsu FH, Geng S (2004) Breeding bird species richness in Taiwan: distribution on gradients of elevation, primary productivity and urbanization. J Biogeogr 31:307–314
Li JS, Song YL, Zeng ZG (2003) Elevational gradients of small mammal diversity on the northern slopes of Mt. Qilian, China. Global Ecol Biogeogr 12:449–460
Li J, He Q, Hua X, Zhou J, Xu H, Chen J, Fu C (2009) Climate and history explain the species richness peak at mid-elevation for Schizothorax fishes (Cypriniformes: Cyprinidae) distributed in the Tibetan Plateau and its adjacent regions. Global Ecol Biogeogr 18:264–272
Liew TS, Schilthuizen M, Lakim M (2010) The determinants of land snail diversity along a tropical elevational gradient: insularity, geometry and niches. J Biogeogr 37:1071–1078
Marini L, Bona E, Kunin WE, Gaston KJ (2010) Exploring anthropogenic and natural processes shaping fern species richness along elevational gradient. J Biogeogr 38:78–88
McCain CM (2004) The mid-domain effect applied to elevational gradients: species richness of small mammals in Costa Rica. J Biogeogr 31:19–31
McCain CM (2005) Elevational gradients in diversity of small mammals. Ecology 86:366–372
McCain CM (2007) Area and mammalian elevational diversity. Ecology 88:76–86
McCain CM (2009) Global analysis of bird elevational diversity. Global Ecol Biogeogr 18:346–360
Oommen MA, Shanker K (2005) Elevational species richness patterns emerge from multiple local mechanisms in Himalayan woody plants. Ecology 86:3039–3047
Rahbek C (1995) The elevational gradient of species richness: a uniform pattern? Ecography 18:200–205
Rahbek C (2005) The role of spatial scale and the perception of large-scale species-richness patterns. Ecol Lett 8:224–239
Rangel TF, Diniz-Filho JAF, Bini LM (2010) SAM: a comprehensive application for spatial analysis in macroecology. Ecography 33:46–50
Rohde K, Heap M, Heap D (1993) Rapoport’s rule does not apply to marine teleosts and cannot explain latitudinal gradients in species richness. Am Nat 142:1–16
Romdal TS, Grytnes JA (2007) An indirect area effect on elevational species richness patterns. Ecography 30:440–448
Rosenzweig ML (1995) Species diversity in space and time. Cambridge University Press, Cambridge
Rowe RJ (2009) Environmental and geometric drivers of small mammal diversity along elevational gradients in Utah. Ecography 32:411–422
Sanders NJ (2002) Elevational gradients in ant species richness: area, geometry, and Rapoport’s rule. Ecography 25:25–32
Sanders NJ, Moss J, Wagner D (2003) Patterns of ant species richness along elevational gradients in an arid ecosystem. Global Ecol Biogeogr 12:93–102
Shin JH (2002) Ecosystem geography of Korea. In: Lee DW, Jin V, Choe JC, Son YW, Yoo SJ, Lee HY, Hong SK, Ihm BS (eds) Ecology of Korea. Bumwoo, Seoul, pp 19–46
Stevens GC (1989) The latitudinal gradient in geographical range: how so many species coexist in the tropics. Am Nat 133:240–256
Stevens GC (1992) The elevational gradient in altitudinal range: an extension of Rapoport’s latitudinal rule to altitude. Am Nat 140:893–911
Wang Z, Tang Z, Fang J (2007) Altitudinal patterns of seed plant richness in the Gaoligong Mountains, south-east Tibet, China. Divers Distrib 13:845–854
Watkins JE, Cardelús C, Colwell RK, Moran RC (2006) Species richness and distribution of ferns along an elevational gradient in Costa Rica. Am J Bot 93:73–83
Webb TJ, Gaston KJ (2003) On the heritability of geographic range sizes. Am Nat 161:553–566
Yim YJ (1977) Distribution of forest vegetation and climate in the Korean Peninsula: IV. zonal distribution of forest vegetation in relation to thermal climate. Jpn J Ecol 27:269–278
Yim YJ, Kira T (1975) Distribution of forest vegetation and climate in the Korean Peninsula: I. distribution of some indices of thermal climate. Jpn J Ecol 25:77–88
Zapata FA, Gaston KJ, Chown SL (2003) Mid-domain models of species richness gradients: assumptions, methods and evidence. J Anim Ecol 72:677–690
ZenHua S, ShengJin P, XiaoKun O (2007) Rapid assessment and explanation of tree species abundance along the elevation gradient in Gaoligong Mountains, Yunnan, China. Chin Sci Bull 52:225–231
Acknowledgments
We thank Mr. Keun-Wook Lee and Mr. Sang-Hyouk Seo for invaluable help during the fieldwork and data analysis in this study. Thanks are also due to Dr. Joon-Hwan Shin and Dr. Sang-Gon Park for their support and encouragement. This paper forms a part of the “Korea Big Tree Project” funded by the Korea Green Promotion Agency, Korea Forest Service.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Lee, CB., Chun, JH., Song, HK. et al. Altitudinal patterns of plant species richness on the Baekdudaegan Mountains, South Korea: mid-domain effect, area, climate, and Rapoport’s rule. Ecol Res 28, 67–79 (2013). https://doi.org/10.1007/s11284-012-1001-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-012-1001-1