Abstract
Methane is a potent greenhouse gas that is produced in many sectors. Agriculture and, more specifically, livestock contribute to this phenomenon. Methane is produced as a result of fermentation in the rumen of dairy cows with a significant amount of gas being released in the atmosphere via the mouth of ruminants. The total intake is the main factor influencing methane production followed by digestibility, fat, and the amount of fibre in the diet. Many strategies exist to reduce methane emissions such as chemicals, essential oils, and the red macroalgae in the diet of dairy cows. The majority of these strategies are either expensive or not feasible to use in a long-term period of time since the microbes in the rumen will adapt to this change. There is a wide range of methods and tools to measure methane emissions both in vitro and in vivo. The respiration chamber is the golden method to measure and quantify the fluxes (methane emissions) in dairy cows. In some cases where measurements of methane are impossible, vitro techniques together with modelling approaches could be used to predict methane emissions. Empirical and mechanistic modelling is a technique widely used to predict methane emissions. In this case by knowing some feed and animal characteristics methane could be reliably estimated.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Methane
- Livestock
- Fermentation
- Rumen
- In vitro and In vivo
- Respiration chamber
- Empirical and mechanistic modelling
10.1 Methane Gas
Water vapour is the number one contributor to greenhouse gas (GHG) effect followed by carbon dioxide (CO2) and methane (CH4) (Kiehl and Trenberth 1997). Methane is a compound with relatively high energy combustion of 55.5 MJ/kg (Crutzen 1995) that contributes to about 20% of total anthropogenic GHG emissions as shown by Lassey (2007). Methane has a very short turn-over time of about 10 years in the atmosphere as compared to CO2, but it can trap the heat 20 times greater than CO2, playing a key part in global climate change. The concentration of CH4 gas has been rising rapidly in the atmosphere over the past decade compared to three centuries ago; it has raised over 2.5-fold (Lassey 2007). Emissions of CH4 lead to increased ground-level ozone, with significant damage to public health and agriculture (Howarth 2019), giving an estimated social cost of 2700 USD per ton of CH4 (Shindell 2015).
10.2 Sources of Methane Emissions
There are many sources that CH4 originates from; it can be from wetlands, rice paddies, termites, agriculture, and burning biomass (Immig 1996). The rice paddies have been shown to be an important contributor with annual emissions reported to be around 115 teragrams (Tg) per year (Thorpe 2009). The agriculture sector contributes to about 10–12% of the total global anthropogenic GHG emissions (McAllister et al. 2011) with livestock sector (enteric fermentation) contributing the most within the agricultural sector of around 37% of total anthropogenic CH4 emissions. Other reports claim that CH4 emissions from the livestock sector is about 51% of the total agricultural CH4 emissions and that the contribution of rice paddies and livestock is rather similar, 100 and 110 Tg/year, respectively (Moss et al. 2000).
There is a high demand by the Intergovernmental Panel on Climate Change (IPCC) to evaluate the number of gases produced and to develop methods and strategies to reduce the emissions of GHG within a time frame (Ahmed 2017; Moss et al. 2000). Within the European countries, livestock, mainly the enteric fermentation, has been reported to be the leading CH4 producer within the agriculture sector (Moss et al. 2000).
Within the European Union (EU-27) and based on data that was obtained in 2003, Lesschen et al. (2011) reported that dairy cow and beef cattle contributed to the most GHG emissions (Fig. 10.1).
Greenhouse gas emissions from the livestock production in the EU-27. (Source: Lesschen et al. 2011)
As shown in Fig. 10.1, the enteric fermentation from dairy cow and beef cattle contributes the most to the GHG emissions followed by the N2O soil emission and manure management.
Recently published data based on radioactive carbon (C14) content in CH4 indicates that anthropogenic emissions of CH4 in recent decades have been higher than previously estimated (Petrenko et al. 2017). Satellite data (Howarth 2019) suggest that the increased global CH4 emissions in the period 2005–2015 were mostly due to increased extraction of shale gas and that the natural gas and oil industry contributes twice as much CH4 emissions as animal agriculture.
10.3 Methane in Ruminants
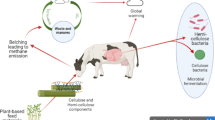
Methane is produced in the rumen of ruminants with a minor contribution from the hindgut as a result of food digestion and fermentation. The majority (95%) of CH4 gas is produced during the enteric fermentation and is lost to the atmosphere via belching, whereas the remaining 5% is emitted through the rectal (Fig. 10.2).
The food eaten by dairy cows (mainly silage and concentrates) is then fermented in the rumen by the help of microorganisms. A result of this fermentation is hydrogen (H2) gas which then needs to be absorbed in order to make this fermentation pathway happening all time. There are specific microorganisms in the rumen belonging to the domain of Archaea (Methanobrevibacter spp.) which uses the H2 to produce CH4 gas. One of the dominant species of methanogenic bacteria living in the rumen is Methanobacterium ruminantium (Miller et al. 1986). The phenomenon of CH4 emission starts around 4 weeks after birth in dairy cows when the rumen is almost shaped, and solid particles are kept in the rumen (Johnson and Johnson 1995). Methanogenic bacteria are mainly in both the liquid and solid phases in the rumen (Morgavi et al. 2010). The food entering the rumen (stomach) of a cow is first digested by microorganisms that contain mainly bacteria, protozoa, and fungi. The simple monomers produced by primary microorganisms are then used by both primary and secondary fermenters to produce end products such as volatile fatty acids (VFA), CO2, and H2 (McAllister et al. 1996). In the final step of fermentation, the H2 that is produced in previous steps is then used together with CO2 to produce CH4 gas by methanogens in the rumen (Eq. 10.1).
Methane emission from dairy cows depends on many factors, such as type of feed, the amount of feed intake, quality of the feed, and digestibility. Grass contains energy; this energy is called gross energy (GE) and once eaten by dairy cows a part of this energy is lost as CH4 gas. Depending on the factors mentioned above, CH4 emission as a proportion of GE varies between 2% and 12% of GE intake (Johnson and Johnson 1995).
10.4 Factors Affecting Methane Emission
There are many factors influencing CH4 emission in dairy cows. The main element is dry matter intake. In addition to intake, diet digestibility, amount of fat and fibre in the diet has effects on CH4 emission in dairy cows (Ramin and Huhtanen 2013).
There are some feed characteristics influencing CH4 emission in dairy cows as there is a close relationship between rumen-fermented organic matter and CH4 emission (Ramin and Huhtanen 2013). Diets that contain high amounts of digestible fibre will increase the digestibility in the diet resulting in higher emissions of CH4. The forage to concentrate ratio in the diet also affects CH4 emission, for example, feeding high concentrate proportions (above 90%) in the diet of feedlot beef cattle can reduce CH4 significantly (Johnson and Johnson 1995). Moss et al. (1995) showed that CH4 as a proportion of GE increased more when the concentrate was increased in the diet of sheep fed on a low level of intake. The effect of fat in the diet is another factor influencing CH4 emission (Grainger and Beauchemin 2011). There are some theories behind the effect of fat on CH4 emissions: (1) unsaturated fatty acids are bio-hydrogenated in the rumen, a process that utilizes H2, (2) inclusion of fat in the diet simply reduces the supply of carbohydrates resulting in less fermentable substrates, and (3) inclusion of fat in the diet favours the pathway of propionic acid production (H2 sink) in the rumen (Ramin and Huhtanen 2013).
10.5 Factors Inhibiting CH4 Emissions
To date, there are many strategies to reduce CH4 emission in dairy cows, ranging from chemicals to algae. Some show direct effects on methanogenic bacteria and some act by interrupting the last step in the biochemical process of producing CH4 in the rumen. For the chemicals, the efficient methane inhibitor identified is 3-nitrooxypropanol (3NOP). The 3NOP has proven to be the most effective inhibitor without showing any adverse effect on milk production (Hristov et al. 2015). The amount of 3NOP needed to reduce enteric methane from cows is very small, 80 mg per kg of DM intake showed reductions up to 30% of methane production from high producing dairy cows (Hristov et al. 2015). In addition, other chemicals have been reported in the literature decreasing CH4 emissions, such as 2-nitroethanol and bromoform (Chagas et al. 2019; Zhang and Yang 2011).
Regarding dietary strategies with the potential to mitigate CH4 emission, the rapeseed oil added to a grass silage-based diet reduced ruminal CH4 emissions from lactating cows as reported by Bayat et al. (2018), where the decrease in CH4 was explained by reductions in DM intake and the dilution effect on fermentable organic matter. Franco et al. (2017) in an in vitro study replaced soybean meal by dried distiller’s grain in grass silage-based diet, and the authors reported a decrease in predicted in vivo CH4 production, which was related to a shift in the ruminal fermentation pattern to decreased acetate and butyrate production and increased propionate production. Further, the use of oats in the diet has also been shown as a potential strategy to reduce CH4 emission, and a recent study conducted by Fant et al. (2020) showed that predicted in vivo CH4 emission was 8.5% lower for a diet that used oats compared to barley.
Several studies have recently reported the potential of essential oils to reduce enteric CH4 production, primarily in vitro and short-term trials. The most common essential oils reported in the literature as methane mitigate strategies are derived from thyme, oregano, horseradish, rhubarb, frangula, and highlighting garlic, cinnamon, and its derivatives (Benchaar and Greathead 2011). However, the authors draw attention to the need for in vivo investigation to propose whether these ingredients/additives can be used successfully to inhibit rumen methanogenesis, without depressing feed intake, digestibility, and animal productivity.
Recently, the red macroalgae Asparagopsis taxiformis (AT) has shown promising effects on reducing CH4 emission from dairy cows. In vivo (Stefenoni et al. 2019) and in vitro (Chagas et al. 2019) studies showed a decrease of 80% on CH4 emission by adding 0.5% of AT on a dry matter basis and inhibition of CH4 by adding 0.5% of AT on organic matter basis, respectively. Previous in vitro studies also had reported the potential to mitigate methane emission to adding AT in ruminants diets (Machado et al. 2014, 2016).
One major problem with additives used in the diet is the excess of H2 gas in the rumen if there is no other sink to uptake the H2 production (Fig. 10.3).
This cartoon shows the side effects of dietary CH4 inhibitors. (Reprinted from: An Introduction to Rumen Studies by J.W. Czerkawski, page 172. Copyright © (1986))
10.6 Methods and Models for Measuring or Predicting CH4 Emission
There are many tools and models in the literature to predict CH4 emission. Respiration chamber is the most accurate method of measuring CH4 emission in dairy cows (Johnson and Johnson 1995). The animal is basically allocated in a chamber for 2–3 days in which all exhaled breath is measured including CH4. The technique is laborious with high construction costs. The alternative to in vivo techniques measuring CH4 emissions, in vitro methods, is also used. In the in vitro method, a small sample size (1 g) is incubated in fermentation units in which buffered rumen fluid is added. The fermentation takes place in an anaerobic condition at the same temperature of the rumen (39 °C). The unit is then gently shacked for about 48 h.
The in vitro gas production system’s main advantage is that it provides a large number of data points, which allow accurate estimates of CH4 emissions. In another hand, this system has some limitations compared with in vivo studies (e.g. no absorption of VFA over time and the intake dynamics).
Recently, Ramin and Huhtanen (2012) developed the application of an in vitro method so CH4 emission could be predicted in vivo by applying the data obtained from the in vitro in a rumen model. The method allows estimation of CH4 emissions every 20 min of incubating a sample up to 48 h. Figure 10.4 shows the curve of CH4 emission over a 48 h incubation time for a diet consisting of silage. One main advantage of in vitro systems is that it allows digestion kinetics to be evaluated from feeds and that the method could be used as a screening tool for assessing different CH4 inhibitors.
In vitro method (a) and methane emission (b) over a 48 h incubation period from a silage-based diet using the model as described by Ramin and Huhtanen (2012)
Danielsson et al. (2017) evaluated the in vitro system developed by Ramin and Huhtanen (2012) by formulating 49 diets used in 13 in vivo studies in which CH4 emission was measured by the respiration chamber. The results indicated that the in vitro system predicted in vivo CH4 emissions very well with a high R2 = 0.96. However, the values obtained (mean 399 L/d) also showed a slight underestimation compared to the observed (mean 418 L/d) in vivo CH4 emissions (Fig. 10.5).
Relationship between predicted in vivo CH4 emission by the in vitro technique and observed CH4 emission in vivo (L/d; n = 49), with fixed and mixed model regression analysis. (Source: Danielsson et al. 2017)
Models are developed from data sets that consist of animal and dietary characteristics. The most widely used models to predict CH4 emissions are the empirical models. However, models can be categorized into two main groups: empirical models (e.g. Ellis et al. 2007; Ramin and Huhtanen 2013; Niu et al. 2018) or dynamic mechanistic models (Huhtanen et al. 2015).
Empirical models relate CH4 emissions to the total amount of intake and dietary composition (Ramin and Huhtanen 2013). The empirical models developed by Ramin and Huhtanen (2013) use a data set in which no additive study is used. It is also advisable to use a mixed model regression analysis so that random study effect will be taken into account (St-Pierre 2001) when developing models predicting CH4 emission. The model predicting CH4 as a proportion of GE developed by Ramin and Huhtanen (2013) takes into account total dry matter intake per kg of body weight (DMIBW), organic matter digestibility estimated at the maintenance level of feeding (OMDm), and dietary concentrations of neutral detergent fibre (NDF), non-fibre carbohydrates (NFC), and ether extract (EE).
And the equation predicting total CH4 emission (litres per day) developed by Ramin and Huhtanen (2013) was closely related to total DMI, and further adding other variables improved the model:
Mechanistic models are a little bit more complicated as compared to empirical models. Mechanistic models simulate CH4 emissions using mathematical formulas and descriptions of ruminal fermentation biochemistry, making it a great tool for understanding the mechanisms and factors influencing CH4 emissions in the rumen. Karoline is a dynamic, deterministic, and mechanistic simulation model of a lactating dairy cow developed by Danfær et al. (2006). The sub-model predicting CH4 emission was further developed by Huhtanen et al. (2015). A recent evaluation of the Karoline model using a data set developed from studies that respiration chamber was used to measure CH4 emission suggested that the model has a potential to predict CH4 emissions accurately and precisely as shown in Fig. 10.6 (Ramin and Huhtanen 2015). Furthermore, evalution of CH4 at whole farm scale is need of time (Ahmed et al. 2020).
Relationship between predicted (using the Karoline model) and observed CH4 emissions (L/d) (n = 184) with fixed and mixed model regression analysis. (Source: Ramin and Huhtanen 2015)
Table 10.1 summarizes some empirical and mechanistic models developed in the literature. The empirical model developed by Ramin and Huhtanen (2013) predicted CH4 emission better than other models as observed by a smaller root mean square error (RMSE). The mechanistic model Karoline also showed better predictions of CH4 emission (Table 10.1) compared to the mechanistic model evaluated by Mills et al. (2001).
There are many equations developed in the literature predicting CH4 production. Equations are basically developed from larger data sets in which intake and dietary factors are gathered. Since dry matter intake is the driving force in predicting CH4 production, often all equations require this parameter for predicting CH4 production. Table 10.2 summarizes some equations predicting CH4 production in dairy cattle.
10.7 Conclusion
Methane is emitted from ruminants as a result of fermentation in rumen. There are many strategies to inhibit CH4 emissions from ruminants. Most strategies reducing CH4 emission require adaptation of the inhibitor used in the rumen and that the rechannelling of H2 remains unclear in the rumen upon using any inhibitor. There are both in vitro and in vivo methods to measure CH4 emission from dairy cows. Empirical and mechanistic models such as the Karoline model usually predicts CH4 emission reliably in which they could be used by national inventories and advisory services for predicting CH4 emission.
References
Ahmed M (2017) Greenhouse gas emissions and climate variability: an overview. In: Ahmed M, Stockle CO (eds) Quantification of climate variability, adaptation and mitigation for agricultural sustainability. Springer International Publishing, Cham, pp 1–26. https://doi.org/10.1007/978-3-319-32059-5_1
Ahmed M, Ahmad S, Waldrip HM, Ramin M, Raza MA (2020) Whole farm modeling: a systems approach to understanding and managing livestock for greenhouse gas mitigation, economic viability and environmental quality. In: Waldrip H, Pagliari P, He Z (eds) Animal manure. https://doi.org/10.2134/asaspecpub67.c25
Axelsson J (1949) The amount of produced methane energy in the European metabolic experiments with adult cattle. Ann R Agric Coll Sweden 16:404–418
Bayat AR, Tapio I, Vilkki J et al (2018) Plant oil supplements reduce methane emissions and improve milk fatty acid composition in dairy cows fed grass silage-based diets without affecting milk yield. J Dairy Sci 101:1136–1151
Bell M, Eckard R, Moate PJ, Yan T (2016) Modelling the effect of diet composition on enteric methane emissions across sheep, beef cattle and dairy cows. Animals 54:1–16
Benchaar C, Greathead H (2011) Essential oils and opportunities to mitigate enteric methane emissions from ruminants. Anim Feed Sci Technol 166:338–355
Chagas et al (2019) In vitro evaluation of different dietary methane mitigation strategies. In: Udén P, Eriksson T, Spörndly R et al (eds) 10th Nordic feed science conference, Uppsala July 2019. SLU Repro, Uppsala, p 62
Crutzen PJ (1995) Ruminant physiology: digestion, metabolism, growth and reproduction. In: Engelhardt WV et al (eds) The role of Methane in atmospheric chemistry and climate. Ventura, pp 291–313
Czerkawski JW (1986) An introduction to rumen studies. Robert Maxwell MC, Oxford
Danfaer A, Huhtanen P, Udén P (2006) The Nordic dairy cow model, Karoline-description. In: Kebreab E, Dijkstra J, Bannink A et al (eds) Nutrient digestion and utilization in farm animals. Modeling approaches. CABI publishing, Wallingford, p 383
Danielsson R, Ramin M, Bertilsson J et al (2017) Evaluation of an in vitro system for predicting methane production in vivo. J Dairy Sci 100:8881–8894
Ellis JL, Kebreab E, Odongo NE et al (2007) Prediction of methane production from dairy and beef cattle. J Dairy Sci 90:3456–3466
Fant P, Ramin M, Jaakkola S et al (2020) Effects of different barley and oat varieties on methane production, digestibility and fermentation pattern in vitro. J Dairy Sci In press
Franco MO et al (2017) In vitro evaluation of agro-industrial by-products replacing soybean meal in two different basal diets for ruminants. In: Udén P, Eriksson T, Spörndly R et al (eds) 8th Nordic feed science conference, Uppsala July 2017. SLU Repro, Uppsala, p 170613
Grainger C, Beauchemin KA (2011) Can enteric methane emissions from ruminants be lowered without lowering their production? Anim Feed Sci Technol 166-167:308–320
Howarth RW (2019) Ideas and perspectives: is shale gas a major driver of recent increase in global atmospheric methane? Biogeosciences 16:3033–3046
Hristov AN, Oh J, Giallongo F et al (2015) An inhibitor persistently decreased enteric methane emission from dairy cows with no negative effect on milk production. Proc Natl Acad Sci U S A 112:10663–10668
Huhtanen P, Ramin M, Udén P (2015) Nordic dairy cow model Karoline in predicting methane emissions: 1. Model description and sensitivity analysis. Livest Sci 178:71–80
Immig I (1996) The rumen and hindgut as source of ruminant methanogenesis. Environ Monit Assess 42:57–72
Jentsch W, Schweigel M, Weissbach F, Scholze H, Pitroff W, Derno M (2007) Methane production in cattle calculated by the nutrient composition of the diet. Arch Anim Nutr 61:10–19
Johnson KA, Johnson DE (1995) Methane emissions from cattle. J Anim Sci 73:2483–2492
Kiehl JT, Trenberth KE (1997) Earth’s annual global mean energy budget. Bull Am Meteorol 78:197–208
Lassey KR (2007) Livestock methane emission: from the individual grazing animal through national inventories to the global methane cycle. Agric For Meteorol 142:120–132
Lesschen JP, van den Berg M, Westhoek HJ et al (2011) Greenhouse gas emission profiles of European livestock sectors. Anim Feed Sci Technol 166-167:16–28
Machado L, Magnusson M, Paul NA et al (2014) Effects of marine and freshwater macroalgae on in vitro total gas and methane production. PLoS One. https://doi.org/10.1371/journal.pone.0085289
Machado L, Magnusson M, Paul NA et al (2016) Identification of bioactives from the red seaweed Asparagopsis taxiformis that promote antimethanogenic activity in vitro. J Appl Phycol 28:3117–3126
McAllister TA, Okine EK, Mathison GW et al (1996) Dietary, environmental and microbiological aspects of methane production in ruminants. Can J Anim Sci 76:31–243
McAllister TA, Beauchemin KA, McGinn SM et al (2011) Greenhouse gases in animal agriculture. Finding a balance between food production and emissions. Anim Feed Sci Technol 166-167:1–6
Miller TL, Wolin MJ, Zhao H et al (1986) Characteristics of methanogens isolated from bovine rumen. Appl Environ Microbiol 51:201–202
Mills JAN, Dijkstra J, Bannink A et al (2001) A mechanistic model of whole-tract digestion and methanogenesis in the lactating dairy cow: model development, evaluation, and application. J Anim Sci 79:1584–1597
Morgavi DP, Forano E, Martin C et al (2010) Microbial ecosystem and methanogenesis in ruminants. Animal 4:1024–1036
Moss AR, Givens DI, Garnsworthy PC (1995) The effect of supplementing grass silage with barley on digestibility, in sacco degradability, rumen fermentation and methane production in sheep at two levels of intake. Anim Feed Sci Technol 55:9–33
Moss AR, Jouany JP, Newbold J (2000) Methane production by ruminants: its contribution to global warming. Ann Zootech 49:231–253
Niu M, Kebreab E, Hristov AN et al (2018) Enteric methane production, yield and intensity prediction models of various complexity levels using a global database. Glob Chang Biol 24:3368–3389
Petrenko VV, Smith AM, Schaefer H et al (2017) Minimal geological methane emissions during the Younger Dryas–Preboreal abrupt warming event. Nature 548:443–446
Ramin M, Huhtanen P (2012) Development of an in vitro method for determination of methane production kinetics using a fully automated in vitro gas system – a modelling approach. Anim Feed Sci Technol 174:190–200
Ramin M, Huhtanen P (2013) Development of equations for predicting methane emissions from ruminants. J Dairy Sci 96:2476–2493
Ramin M, Huhtanen P (2015) Nordic dairy cow model Karoline in predicting methane emissions: 2. Model evaluation. Livest Sci 178:81–93
Shindell D (2015) The social cost of atmospheric release. Clim Chang 130:313–326
Stefenoni H, Räisänen S, Melgar A et al (2019) Dose-response effect of the macroalga Asparagopsis taxiformis on enteric methane emission in lactating dairy cows. Paper presented at the American Dairy Science Association, Ohio, 23–26 June 2019
St-Pierre NR (2001) Integrating quantitative findings from multiple studies using mixed model methodology. J Dairy Sci 84:741–755
Thorpe A (2009) Enteric fermentation and ruminant eructation: the role (and control?) of methane in the climate change debate. Clim Chang 93:407–431
Zhang DF, Yang HJ (2011) In vitro ruminal methanogenesis of a hay-rich substrate in response to different combination supplements of nitrocompounds; pyromellitic diimide and 2-bromoethanesulphonate. Anim Feed Sci Technol 163:20–23
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Ramin, M., Chagas, J.C., Krizsan, S.J. (2020). Methane Production in Dairy Cows, Inhibition, Measurement, and Predicting Models. In: Ahmed, M. (eds) Systems Modeling. Springer, Singapore. https://doi.org/10.1007/978-981-15-4728-7_10
Download citation
DOI: https://doi.org/10.1007/978-981-15-4728-7_10
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-4727-0
Online ISBN: 978-981-15-4728-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)