Abstract
Microbial endophytes are symbionts dwelling within plant tissues without appearance of disease symptoms on host plant and have been recently investigated for their plant growth-promoting properties and their beneficial functions associated with plant responses under abiotic stress conditions. This study focuses on the critical role of endophytic microbes in plant health and their stimulatory different mechanisms to tolerance against abiotic stress in plants. Endophytic microbial community can enhance plant growth through producing secondary active compounds which protect the plant from pathogens such as insect and fungi; also endophytes can produce extracellular enzymes which play critical roles in colonization of endophytes within the plant host. Microbial endophytes have the ability to act as plant growth-promoting agents through producing phytohormones and also enable plants to grow in contaminated soils through breakdown of hazardous compounds. Endophytes manage plant growth under adverse conditions such as salinity, drought, temperature, heavy metal stress, and nutrient stress through different mechanisms. This chapter may introduce new approaches for the use of endophytic inoculants to combat abiotic stresses in agricultural fields, which increases global crop production.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Introduction
Endophytes are microbial communities that currently dwell in the healthy plant tissues such as stems, roots, leaves, and seeds without affecting physiological plant functions and not causing any disease symptoms to the plant tissues. Under normal conditions, endophytes have important roles in host plant growth either by secondary metabolite or nutrient assimilation or by preventing induction of plant disease symptoms by different pathogens. Endophytic microbes including bacteria, actinomycetes, and fungi tend to form a network closely to their host plants and are additionally sheltered from unfavorable climatic and other unwanted change in the environment (Zhao et al. 2011; Passari et al. 2017).
Recent research work suggests that about 300,000 species of plants are present, the unmistakable dominant part of which contains endophytes (Smith et al. 2008). In fact, microbial endophytes especially bacteria and fungi have originated in most plant species that have been investigated. According to Partida-Martínez and Heil (2011), endophyte-free plant is an unusual state to what is typically obtained in nature; a plant without endophytes would be susceptible to environmental stress conditions and lose their ability to resist the pathogens (Timmusk et al. 2011).
The origin of endophytes is still not clear because of the multiplicity of the host’s living environment and the complex association between the endophyte and its host plant. Two hypotheses explaining the origin of endophytes were exogenous and endogenous. It was believed that in the last decade, endophytes are gaged from the chloroplast and mitochondria of the plant, and so it has comparable genetic backgrounds to the host (Wen 2004); this is the endogenous hypothesis, while the latter believes that endophytes arrive from outside of the plant and insert into the host from root wound, induced channels, or surface (Li 2005); this is exogenous hypothesis.
Different parts of plants were used for isolation of microbial endophytes as meristem, scale primordia, resin ducts (Pirttilä et al. 2003), leaf segments with midrib and roots (Hata et al. 2002), leaf blade, stem, petiole, bark, and buds (Pirttilä et al. 2008).
Endophytic fungi insert through the hyphae and enter the kernels in the seeds of plant cells that come below vertical transmission. A variant was detected in horizontal and vertical transmission of the endophyte species invading the host plant cells (Tintjer et al. 2008). The procedures of endophytic microbial growth in plants and methods of propagation were paid more attention to know their role in transmission. The endophytic fungal species transmits horizontally by sexual spores or asexually between different plants in community or a population (Tadych et al. 2014).
The microbial community such as bacteria, algae, fungi, and actinomycetes colonizes the host plant roots (Saharan and Nehra 2011; Prashar et al. 2014). Among microbial population found in the rhizosphere, actinobacteria are considered the second most abundant microorganisms, and they comprise more than 30% of the total microorganisms in the soil (Glick 2014). Endophytes are transmitted between the soil rhizhosphere across the seeds. They spread quickly between endo-rhizosphere through the lateral root junction instigated through microbial phyto-pathogens or nematode (Chi et al. 2005). Also, bacterial endophytes can enter their host plant roots through spaces between root hairs and epidermal cells (Hardoim et al. 2008).
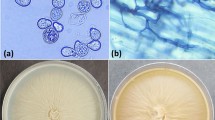
The most common endophytic fungi isolated and identified from numerous plants are Alternaria infectoria, Aspergillus sp., Penicillium sp., Colletotrichum musae, Colletotrichum gloeosporioides, Nigrospora oryzae, Phomopsis sp., Nigrospora sphaerica, Guignardia sp., Cordana musae, Rhizoctonia sp., species of Phialocephala sphaeroides, Xylaria (Wilson et al. 2004), P. chrysogenum Pc_25, A. alternata Aa_27, and Sterile hyphae Sh_26 (Fouda et al. 2015). On the other hand, endophytic P. chrysogenum Pc_25 was mediated biosynthesis of ZnO nanoparticles (Fouda et al. 2019a).
In addition, various endophytic bacterial strains were isolated from economically important plant species. Several of the novel endophytic bacterial species belong to the Arthrobacter spp., Actinobacter spp., Aeromonas spp., Enterobacter spp., Agrobacterium spp., Alcaligenes spp., Bacillus spp., Flavobacterium spp., Azospirillum spp., Azotobacter spp., Pseudomonas spp., Burkholderia spp., Beijerinckia spp., Enterobacter spp., Flavobacterium spp., Erwinia spp., Rhizobium spp., and Serratia spp. were characterized and identified (Gray and Smith 2005). In the last periods, other endophytic actinobacteria such as Streptomyces, Amycolatopsis, Nocardia, Microbispora, Micromonospora, and Streptomyces capillispiralis Ca-1 have been positively isolated from different plant species (Shi et al. 2009; Ruanpanun et al. 2010; Hassan et al. 2018).
6.2 Role of Endophytes in Plant Health
Great effort has been made to study the diversity of endophytic species in plants and their evolutionary biology, ecology, and their roles in defense mechanism against abiotic and biotic stress via production of different metabolites. Endophytic biotechnology can be expended for the efficient production of economically, agriculturally, and industrially significant plants and their crops. The reasonable application for different endophytic species associated with plants can help in improvement of the agricultural products, increasing metabolite productivity in different plants, as well as adjustment tolerance to numerous abiotic and biotic conditions (Wani et al. 2015).
Endophytic species have recently generated important new bioactive substances. It has been suggested that the relationship between different endophytic species and their host plant in the production of a great amount and diversity of biologically active molecules are related together, and this contrasted to epiphytes or soil-related microorganisms (Strobel 2003).
New biotechnology applications for endophytic species such as bioremediation and phytoremediation are gaining considerable impetus (Li et al. 2012a). Endophytes play critical roles in healthy plants through three different mechanisms known as biofertilization, phytostimulation, and biocontrol (Bloemberg and Lugtenberg 2001).
6.2.1 Endophytes as Source for Bioactive and Novel Compounds
Endophytic microbes release specialized biologically active compounds or metabolites without any observable damage to their host tissues (Liarzi et al. 2016). The bioactive compounds synthesized by different endophytic microbes that increase plant resistance against pathogenic microorganisms are too used in the pharmaceutical fields as anticancer, antimicrobial, antiviral, antidiabetic, and other biologically active compounds (Guo et al. 2008). Other biologically active compounds synthesized by endophytic microbes as alkaloids, terpenoids, steroids, peptides, poly-ketones, quinols, flavonoids, phenols, and insecticide azadirachtin are also investigated for their medical, agricultural, and industrial applications (Kusari et al. 2012a; Molina et al. 2012; Zinniel et al. 2002). Numerous of these bioactive compounds showed antioxidant, antimicrobial (antibacterial, antifungal, and antiviral activities), antineoplastic, antiproliferative, anti-leishmanial, cytotoxicity, and fuel production activities (Shankar Naik and Krishnamurthy 2010; Wang and Dai 2011). Examples of antifungal compounds produced by endophytes include cryptocandin, pestaloside, cryptocin, ecomycins, pestalopyrone, and pseudomycins (Yu, et al. 2010).
Naturally, different seasons, locations, environmental conditions, soil, age, and tissue of the host plant, all influence the endophyte biology and thus considerable variants in the synthesis of bioactive metabolites (Strobel and Daisy 2003). Also, cultivation conditions and separation methods can affect the type and variety of metabolites (Gunatilaka 2006). Aly et al. (2011) and Kusari et al. (2012b) reported that sampling, type, and size of the plant tissue used for isolation, composition, and culture conditions for media such as pH, incubation temperature, incubation period, agitation, and culture, all of these factors are controlling the productivity of bioactive compounds in the laboratory.
The productivity of bioactive compounds by endophytic microorganisms can be influenced both genetically and physicochemically (Kharwar et al. 2011). Separation and identification methods of bioactive metabolites from microbes, especially fungal and bacterial endophytic species, are fast growing, as can be detected from numbers of patents, reviews, and original articles available each year in the drug discovery field (Tejesvi and Pirttilä 2011). Fungal endophytic species are a native source for flavonoids, terpenoids, phenols, saponin, alkaloids, carbohydrates tannins, and nematode antagonistic compounds (Liu et al. 2016; Bogner et al. 2017). Endophytic actinomycetes are promising tool for synthesis of bioactive compounds, which can be used as therapeutic agents against different pathogens (Prashith-Kekuda 2016). On the other hand, endophytic bacteria have useful effects as enhancement of nitrogen fixation, phosphate solubilization, production of phytohormones, and reduction of ethylene biosynthesis as response to abiotic (pH, temperature, and osmotic pressure) and biotic (from fungi, bacteria, nematodes, and insects) stresses and have biocontrol activities. Supplementary than 300 endophytic bacteria and actinobacteria belonging to the genera Streptomyces, Rhodococcus, Nocardiopsis, Microbacterium, Brevibacterium, Arthrobacter, Nocardia, Brachybacterium, Tsukamurella, Kocuria, Pseudonocardia, and Nocardioides were isolated from Dracaena cochinchinensis Lour. plant. Of these, 17 endophytic strains have antimicrobial, anthracyclines-producing activities, antifungal properties, and anticancer activities against Hep G2 and MCF-7 as cancer cells (Salam et al. 2017). Endophytic microorganisms have already a bulk for the discovery of biologically active compounds, but still innovative methods are demanded to natural product-based drug discovery.
6.2.2 Extracellular Enzyme Production
Endophytes play an important role in plant health via extracellular enzyme production which have been counted as the most significant and important mechanisms for endophyte colonization in plants. Among enzymes, extracellular enzymes or exoenzymes have industrial importance in different fields such as fermentation process, food, and other biotechnological applications. Microorganisms including fungi and bacteria produce different types of extracellular enzymes, which are oxidoreductases, hydrolases, transferases, and lyases (Traving et al. 2015). Extracellular enzymes breakdown numerous macromolecules such as lignin, sugar-based polymers, proteins, organic phosphate, and carbohydrates to micromolecules and are transported throughout the cells; they are continuously metabolized and help to instruct the host symbiosis process (Strong and Claus 2011; Wingender et al. 1999). Also, extracellular hydro-lyase enzymes increase plant responses to pathogenic infection (Leo et al. 2016).
Different extracellular enzymes such as β-1,3-glucanase, protease, lipase, and chitinase associated with endophytic microbes lyse the cell walls of pathogenic bacteria and fungi and hence can be used as biocontrol agents (Fouda et al. 2015, Wang et al. 2014).
On the other hand, improvement in endophytic microbial growth within host plant tissues and then reduction in the pathogens are influenced by production of various enzymes such as xylanases, cellulases, pectinases, lipases, proteases, phosphatases, amylase, and glucosidases (Kannan et al. 2015; Pereira et al. 2016; Khan et al. 2016; Ayob and Simarani 2016).
Chathurdevi and Gowrie (2016) reported that the endophytic fungi isolated from medicinal plants can support plant growth to overcome the adverse conditions through producing different extracellular enzymes. Also, approximately 50 endophytic fungal strains having amylase, laccase, cellulase, pectinase, lipase, and protease were isolated and identified according to Sunitha et al. (2013). In addition, bacterial endophytes have been investigated to produce 1-aminocyclopropane-1-carboxylate (ACC) deaminase, amylase, cellulases, esterase, pectinase, protease, lipase, asparaginase, phytase, and xylanase (Carrim et al. 2006; Gupta et al. 2013; Fouda et al. 2015; Akinsanya et al. 2016). Vijayalakshmi et al. (2016) isolated bacterial endophytes from medicinally significant plants producing various extracellular enzymes as cellulase, amylase, and protease.
6.2.3 Plant Growth-Promoting Activity
Endophytic microbes play a critical role in plants’ adaptation to stress conditions and varying environments which can limit their development and growth. To contract with extreme environments, plants may form network with microorganisms in symbiotic relationship, which confer helpful effects on evolution to both partners and appropriateness (Rodriguez et al. 2009). Extreme environment conditions such as low water and nutrient availability, high radiation, strong winds, and low temperatures affect plant survival and organization (Convey 2011).
A practical strategy to reducing stress without cooperating plant growth is the use of plant growth-promoting microbe relationship. Widespread varieties of metabolite substances produced by endophytic fungi are used to improve plant growth (Waqas et al. 2015).
Endophytic microbes enhance plant growth through their ability to synthesize enzymes and various bioactive metabolites. Endophytic microorganisms, especially fungi such as Sebacina vermifera, Piriformospora indica and numerous species of Colletotrichum and Penicillium, are distinguished to have better plant growth-promoting effects under unfavorable conditions (Waller et al. 2005; Redman et al. 2011; Hamilton and Bauerle 2012). Plant growth-promoting microbes (PGPM) associated with many species of plants tend to have useful effects such as intensified plant growth and decreased sensitivity to diseases instigated by plant pathogenic viruses, fungi, bacteria, and nematodes. Main activities of PGPM are accompanied with plant hormone synthesis such as indole-3-acetic acid (IAA), cytokinins, gibberellins, siderophores, phosphate solubilization, nutrient uptake, and antagonism to phytopathogens. Also, PGPM can induce chemical or physical changes related to plant protection, a process signified as induced systemic resistance (ISR). PGPM have developed to be beneficial for plants constantly under many abiotic stresses. Several reports confirmed the critical roles of plant growth-promoting fungi (PGPF) in increasing tolerance against different stresses such as heat, drought, salinity, cold, and heavy metals (Khan et al. 2012). On the other hand, stresses like salinity and drought induce osmotic stress, which is conveyed through abscisic acid (ABA)-independent or ABA-dependent pathways (Cao et al. 2014), and low levels of ABA productions were achieved under fungal action (Jahromi et al. 2008; Khan et al. 2014). Miransari (2012) reported that plants needed low attempt to synthesize ABA and hence protect cell progress under stress, as the water equilibrium in plant was achieved by treatment with endophytic Penicillium spp.
Plant growth-promoting bacteria (PGPB) are proficient to enhance plant growth through independent or linked mechanisms for maintainable agriculture (Compant et al. 2010; Palacios et al. 2014). PGPB showed different responses against numerous stresses in plants (Kim et al. 2012), fighting against plant pathogens (Raaijmakers et al. 2009) and supplementary in the recovery of damaged cells or degraded constituents (de Bashan et al. 2012). Colonization of host plant tissues by endophytic bacterial species has been reported by Yang et al. (2016) and Tang et al. (2017), and its capability to promote growth, fix nitrogen, and repress phytopathogens with induced systemic resistance (ISR) of this pathogen was reported by Pieterse et al. (2014), Puri et al. (2016), and Padda et al. (2016). Endophytic actinobacteria can improve plant growth via one or more plant growth-promoting mechanisms including nitrogen fixation, solubilization of inorganic nutrients, excretion of phytohormones, and siderophores (Dudeja et al. 2012).
Indoleacetic acid (IAA) is fundamental plant growth hormone used for the development and growth of shoot and root cells of plants; many microorganisms including plant growth-promoting rhizobacteria (PGPR) produce IAA (Hassan 2017). Soil microorganisms such as bacteria and fungi synthesize plant growth-promoting compounds such as gibberellins and IAA (Radhakrishnan et al. 2013; Limtong et al. 2014).
Several reports proved that endophytic actinobacteria synthesize plant growth regulators such as auxins, cytokinins, gibberellins (gibberellic acid), and IAA in vitro (Ghodhbane-Gtari et al. 2010; Fouda et al. 2019b).
Siderophores are small compounds with high-affinity iron chelators (soluble Fe3+-binding agents) synthesized by microbes such as fungi, bacteria, and actinobacteria growing under low iron stress. Several endophytic microbes have been explored to synthesize siderophores, with a molecular weight ranging between 400 and 1500 daltons (Kannahi and Senbagam 2014). There are four types of siderophores synthesized by bacteria, viz., catecholate, salicylate, hydroxamate, and carboxylate. Endophytic actinobacteria including the genera Pseudonocardia, Streptomyces, Nocardia, Actinopolyspora, Micromonospora, Salinispora, Actinomadura, and Kibdelosporangium are recognized as siderophore producers (Gangwar et al. 2011; Kannahi and Senbagam 2014; Bhosale and Kadam 2015). Endophytic actinobacteria synthesize siderophores as an extramechanism, which act as plant growth regulators and in defense against pathogens (Rungin et al. 2012).
Moreover salicylic acid (SA), as a phytohormone, is a significant plant hormone concerned in many processes such as root initiation, seed germination, floral induction, stomatal closure, and increased tolerance of plant to abiotic and biotic stresses. Bacterial endophytes synthesize SA, which enhance the growth of plant seedlings under water stress and reduce the growth of plant pathogens such as fungi (Klessig et al. 2016).
6.2.4 Biocontrol Agents
Endophytic microorganisms are defined as a functional biocontrol agent, instead of chemical control. Endophytic fungi play a critical role in controlling insect herbivores not only in grasses but also in conifers (Parker 1995). Tefera and vidal (2009) reported that Beauveria bassiana, an endophytic fungi known as an entomopathogen, was used to control the borer insects in sorghum. Also, acute rotting caused by fungal pathogens in tomato fruits can be achieved during storage and shelf life. Different bacterial endophytic strains such as Bacillus subtilis isolated from Speranskia tuberculata (Bge.) Baill has an antagonistic effect in vitro against the pathogen Botrytis cinerea, which cause rotting of tomato fruits during storage (Wang et al. 2009). New endophytes, such as Burkholderia pyrrocinia JK-SH007 and Bacillus cepacia, were used in biocontrol study against poplar canker (Ren et al. 2011). New approaches in biocontrol studies induced genetically engineered gene expression into an endophytic microorganism to synthesize anti-pest proteins like lectins for insect control.
On the other hand, endophytic strains such as Chaetomium globosum YY-11 isolated from rape seedlings, Enterobacter sp., and Bacillus subtilis isolated from seedlings of rice were utilized for the expression of Pinellia ternate agglutinin (PtA) gene (Zhao et al. 2010). The previous endophytic recombinant fungal and bacterial strains which express the PtA gene were used successfully to control sap-sucking pests in numerous crop seedlings. Similarly, in another study, Enterobacter cloacae, as a recombinant endophytic bacterial strain expressing the PtA gene, was proved as a bio-insecticidal agent against the white backed plant hopper Sogatella furcifera (Zhang et al. 2011). The recombinant endophytic strains which easily dwell within several plants can be used as a new strategy to control different plant pests through expression of different anti-pest proteins. Also, Hassan et al. (2018) reported that copper nanoparticles synthesized using the endophyte Streptomyces capillispiralis Ca-1 have the ability to biocontrol Culex pipiens (Mosquito) and Musca domestica (housefly). On the other hand, copper oxide nanoparticles synthesized by two endophytic actinomycetes, Streptomyces zaomyceticus Oc-5 and Streptomyces pseudogriseolus Acv-11, isolated from Oxalis corniculate L. plant have antimicrobial activity against four phytopathogenic fungi, namely, Phoma destructiva, Fusarium oxysporum, Alternaria alternata, and Curvularia lunata (Hassan et al. 2019).
6.2.5 Bioremediation/Biodegradation Activity
Endophytic microorganisms have a powerful ability to enhance plant growth in contaminated soil through breakdown of hazard compounds. Bioremediation is defined as elimination of pollutants and hazardous wastes from contaminated environments by breakdown of these wastes using biological processes. This is due to the major microbial diversity. Mastretta et al. (2009) reported the ability of Nicotiana tabacum plants in bioremediation through inoculation of their seeds with endophytes. They have showed enhanced plant biomass production under Cadmium (Cd) as heavy metal stress, and the Cd concentration in plant tissue was higher compared to noninoculated plants. These results proved the useful effects of endophytic inoculated seeds on accumulation and assimilation of heavy metals.
To discover the role of endophytic microbes in the breakdown of contaminants such as plastics, different endophytic fungal strains were subjected to growth in agar and broth media containing polyester polyurethane (PUR) as a sole carbon source (Russell et al. 2011). Among fungal endophytic strains, two Pestalotiopsis microspora isolates have the ability to use PUR as the sole carbon source under aerobic and anaerobic conditions and using serine hydrolase enzyme for the degradation of PUR.
6.2.6 Induced Systemic Resistance (ISR)
Endophytic microorganisms increase plant resistance to pathogen through induce defense mechanisms, so-called induced systematic resistance (ISR) (Zamioudis and Pieterse 2012). At an initial stage, there is increasing evidence that interactions between endophytic microorganisms and their hosts stimulate immune response in host plants; this is similar to those happening against pathogens; later on, endophytic microorganisms colonize plants through escaping from defense responses as occurring in the bacterial genera Bacillus and Pseudomonas (Kloepper and Ryu 2006). Different bacterial factors such as antibiotics, salicylic acid, N-acyl-homoserine lactones, siderophores, jasmonic acid, lipopolysaccharides, and volatiles (e.g., acetoin) are responsible for induction of ISR (Bordiec et al. 2011). The defense mechanisms and protections of plants against herbivorous insects and pathogens were related to ISR. Although several endophytic bacteria have increased ISR via salicylic acid induction, ethylene (ET) and jasmonic acid (JA) as plant hormones have important regulatory roles in signaling pathways implicated in ISR induction (Pieterse et al. 2012). The endophytic bacterium Pseudomonas fluorescens 89B-61 was the first reported, explaining the ISR induction to protect cucumber plants against cucumber anthracnose (Kloepper and Ryu 2006). The resistance of potato plant against the pathogen Pectobacterium atrosepticum was increased in the presence of the endophyte Methylobacterium sp. IMBG290. The resistance manner was related to changes in composition of the native endophytic community. Changes in endophytic community were linked to disease resistance, which means the endophytic community has a critical role in disease repressions (Ardanov et al. 2011). Also, endophytic fungi have been involved in protection mechanisms via ISR induction but less than endophytic bacteria (Bae et al. 2011). The potentiality of endophytic fungi in producing metabolites has inhibitory activities against herbivores, and plant pathogens were recorded. These metabolites comprise steroids, alkaloids, peptides, terpenoids, flavonoids, polyketones, phenols, chlorinated compounds, and quinols (Higginbotham et al. 2013; Tejesvi et al. 2013). On the other hand, metabolites having antibacterial, antiviral, insecticidal, and antifungal activities were reported by fungal endophytes, which transmitted horizontally, forming local disease in their hosts (Gunatilaka 2006; Tejesvi et al. 2011).
6.3 Alleviation of Abiotic Stresses via Microbial Endophytes
Plant growth and development are restricted by different extreme conditions which include environmental stresses as well as stresses caused by living communities.
Plants can tolerate abiotic stress by two mechanisms: (i) plants can avoid negative effects of stress via activation of response systems directly after exposure to stress (Meena et al. 2017), and (ii) biochemical compounds are synthesized by endophytes and act as anti-stress agents (Schulz et al. 2002). The up- and downregulation for some stress-inducible genes in pepper plant were reduced after inoculation with the endophyte Arthrobacter sp. and Bacillus sp. when compared with gene expression in uninoculated plants. Assimilation of nutrients, such as magnesium, potassium, and calcium, plant biomass, growth parameters, and decreased sodium toxicity were significantly increased in cucumber plants under sodium chloride and drought stress after inoculation with Phoma glomerata and Penicillium sp. when compared with uninoculated plants (Waqas et al. 2012). Bailey et al. (2006) revealed that Trichoderma sp. isolated from Theobroma cacao increases tolerance in cacao plant against abiotic stress especially drought via gene expression change.
The resistance of tissue cultured Kalmia latifolia L. to drought stress was increased after seedling inoculation with Streptomyces padanus AOK-30 as endophytic actinomycetes as reported by Hasegawa et al. (2004).
Bae et al. (2009) reported that sugars and amino acids showed significant increase in endophyte-colonized plants due to drought stress. Increase in sugar and amino acid production (as indicators for increased osmolytic activity) is due to intricate symbiotic relationship in plants possessing a drought-tolerant phenotype (Shinozaki and Yamaguchi-Shinozaki 2007). Significantly producing biomass is one response to drought, temperature, and salt stress in endophyte-colonized plants than their non-colonized one (Redman et al. 2011). Zhang and Nan (2010) revealed that increased seedling growth as drought response was due to higher antioxidant activity. Also, Zhang and Nan (2007) showed that increase in biomass, proline concentrations, and relative water content as a result of endophyte colonization under low water conditions was investigated. Inoculation of wheat with Burkholderia phytofirmans PsJN increased CO2 assimilation, photosynthetic rate, water use efficiency, and chlorophyll content under drought conditions (Naveed et al. 2014).
The following are examples for abiotic stresses that have a negative effect on plant growth and development‚ and alleviate via endophytes.
6.3.1 Drought Stress
Drought is one of the most important abiotic stresses that suppress plant growth, development, and productivity. Plants undergo drought conditions through either limiting water supply to the roots or very high transpiration (Anjum et al., 2011). It has been concluded that diurnal water stress normally occurs in most plant species during noon and afternoon hours in temperate climates, even though the soil water contents are normal. This temporary drought stress has a negative impact on the growth rate (Granier and Tardieu 1999). Drought causes reduced germination rates, membrane loss of its integrity, repression of photosynthesis, and increase in the productivity of reactive oxygen species (Greenberg et al. 2008). Furthermore, elevated drought and salinity were the main causes of osmotic stress to plants. While drought leads to osmotic stress, salinity shows both ionic or ion-toxicity, and osmotic stress impacts cells (Zhu 2002). The shoot system symptoms of osmotic stress caused by salinity interfere with that of drought stress including stunted growth and leaf senescence (Munns 2002).
Plants harboring endophytes (rice, tomato, dune grass, and panic grass) consumed significantly less water and had enhanced biomass than nonsymbiotic plants. Increased accumulation of solutes in tissues of endophyte-associated plants comparable with noninfected plants, or because of thicker cuticle formation, or by decreased leaf conductance and a slower transpiration stream may explain the drought tolerance phenomenon (Malinowski and Beleskey 2000). The ability of plant to tolerate water stress may be related to morphological and genetic adaptation and biochemical responses. However, the central response to water deficits is the increase in the biosynthesis of plant hormone ABA and/or reduction in ABA breakdown (Bray 2002). In plants suffering from drought, it is supposed that ABA behaves like the signal that manages the plant’s resistance to water deficit, principally by controlling water loss and stomata closure (Zhang and Outlaw 2001). Also, other evidence proposes that ABA has a role in root branching, enhancing the plant water absorption capacity (De Smet et al. 2006).
ABA was defined using full scan mass spectrometry as a by-product of chemically enhanced growth cultures of Azospirillum brasilense Sp 245. Adding NaCl to the culture medium led to increased bacterial ABA production, and ABA levels were improved in Arabidopsis thaliana seedlings inoculated with Azospirillum brasilense Sp 245 (Cohen et al. 2008).
6.3.2 Salinity Stress
Soil salinization happens when water-soluble salts accumulate in the soil to a level that affected environmental health, agricultural production, and economics. In the first stages, salinity has a negative impact on the metabolism of soil organisms and hence decreases soil productivity, but in advanced stages, it destroys all vegetation and other organisms living in the soil, consequently transforming fertile and productive land into barren and desertified lands (Jones et al. 2012). A saline soil is known to have an electrical conductivity (EC) of the saturation extract (ECe) in the root zone more than 4 dS m−1 (nearly 40 mM NaCl) at 25 °C with exchangeable sodium of 15%. The yield of most crop plants is decreased at this ECe, and many crops showed reduced yield at lower ECes (Jamil et al., 2011).
It is a key factor contributing to reduced productivity of cultivated soils. Although accurate estimation is difficult, the salinized soil area is increased, and this phenomenon is particularly dense in irrigated soils. It is estimated that about 20% (45 million hectares) of irrigated land, which produces one-third of global food, is affected by salinity (Shrivastava and Kumar 2015). Soil salinity impacts an estimated one million hectares in the European Union, particularly in the Mediterranean countries, a major cause of desertification. In Spain, about 3% of irrigated land (3.5 million hectares) is severely affected, significantly reducing their agricultural potential, while another 15% are at high risk (Stolte et al. 2015). In the Mediterranean area, soil alkalization associated with land degradation may deteriorate at increasing rates in the coming decades due to the expected increase in irrigated regions and the increasing deficiency of good-quality water (Bowyer et al. 2009).
6.3.2.1 Effect of Soil Salinity on Plants
Salinity significantly affects agricultural crops, which reduces agricultural output and affects the physical and chemical properties of the soil and environmental balance of the region, as well as low economical findings and soil corrosions (Hu and Schmidhalter 2002).
Complex interactions led to salinity effects comprising biochemical, physiological, and morphological processes including vegetative growth, water uptake, seed germination, enzyme activity, seedling growth, protein synthesis, and mitosis of DNA and RNA (Akbarimoghaddam et al. 2011). It has deep impact on reproductive development by stamen filament elongation and inhibiting microsporogenesis, ovule abortion, and senescence of fertilized embryos and enhanced cell death in tissue types. Since many salts are also plant nutrients, increased salt concentrations in the soil can disturb the plant nutritional balance or interfere with the absorption of some nutrients (N, Ca, K, P, Fe, and Zn) leading to nutrient deficiency. Because P ions precipitate with Ca ions, P uptake is significantly reduced by soil salinity (Bano and Fatima 2009).
While K+ has a main role in biochemical reactions, acting as a cofactor for various enzymes and in protein synthesis, high concentrations of K+ mediates binding of tRNA to ribosomes. However, soil salinity leads to ion toxicity resulting from replacing K+ by Na+ in such reactions. Furthermore, Cl− and Na+ induced conformational modifications in proteins (Zhu 2002). Soil salinity imposes osmotic stress, which leads to loss of turgidity, cell dehydration, and, finally, death of cells. Osmotic stress and ion toxicity lead to metabolic imbalance, which in turn causes oxidative stress (Ashraf 2004).
Photosynthesis is negatively affected by salinity of the soil through reducing photosystem II capacity, chlorophyll content, leaf area, and stomatal conductance (Netondo et al. 2004). Moreover, salinity may impede the supply of hormones or photosynthetic assimilates to growing tissue (Ashraf 2004). The cell cycle is transiently arrested by salinity stress which causes reduction in the activity and expression of cyclins that result in fewer cells in the meristem, consequently limiting growth. In addition, the posttranslational inhibition during salinity stress causes reduction in the activity of cyclin-dependent kinase (Seckin et al. 2009).
6.3.2.2 Salinity Stress Alleviation by Microbial Endophytes
Salinity problem threatens more than 20% of agricultural soil (Zhu 2000), and by 2050, about 50% of important agricultural land will be affected by salinity stress (Munns and Tester 2008). Endophytic microbes can enhance growth properties and modulate metabolism and phytohormone signaling. In addition, endophytic microbes improve adaptation to abiotic and biotic stress. Endophytes represent a particular concern for improved crop adaptation to stress as they are relatively protected from the harsh soil environment under high salt, drought, or other stress conditions (Sturz et al. 2000).
The following are the major advantages of endophytes to minimize salinity impacts on plants.
Plant Antioxidant Status
Reactive oxygen species in plants are formed on the onset of salt and osmotic stress. Scavenging enzymes such as ascorbate peroxidase, catalase, and superoxide dismutase inhibit oxidation of DNA, membrane proteins, and lipids. Microorganisms use similar methods to deal with oxidative stress. Hamilton and colleagues in 2012 reported the fungal endophyte mediation of reactive oxygen species in plants (Hamilton et al. 2012). Previous studies have suggested the relationship between tolerance of plants to salt stress and the alleviation of antioxidant enzymes (Sekmen et al. 2007). Scavinging enzymes for ROS include glutathione reductases (GR), superoxide dismutases (SOD), catalases (CAT), dehydroascorbate reductases (DHAR), ascorbate or thiol-dependent peroxidases (APX), and mono-dehydroascorbate reductases (MDHAR), in addition to tocopherol and glutathione (Rouhier et al. 2008). These enzymes involved in the removal of ROS either directly (APX, SOD, CAT) or indirectly via regeneration of glutathione and ascorbate in the cell. On constant, when the nonsymbiotic plants Leymus mollis (dunegrass)subjected to 500 mmol l−1 NaCl solution becomes severely wilted, desiccated within 7 days and ultimately dead after 14 days (Rodriguez et al. 2008). Plants infected with Fusarium culmorum did not show the symptoms of wilt until it was subjected to 500 mmol l−1 NaCl for 14 days. The endophyte Piriformospora indica induces salt tolerance by improving the antioxidant status of barley (Baltruschat et al. 2008).
ACC Deaminase
Although endophytic microbes might produce ACC deaminase enzyme and do not benefit from it, the enzyme has a role in promoting plant growth and enhances plant stress tolerance through cleaving ethylene, which acts as a precursor for the synthesis of ACC (Glick 2014). ACC deaminase can reduce plant ethylene levels by cleaving its precursor ACC (1-aminocyclopropane-1-carboxylate) to 2-oxobutanoate and ammonia, inhibiting ethylene signaling (Glick et al. 1998). Ethylene is a significant plant hormone that contributes in seed germination, in response to several stresses, and it is the main regulator for bacterial colonization of plant tissues (Iniguez et al. 2005). Ethylene accumulation in plants as a stress response is commonly detrimental to plant health and growth (Czarny et al. 2006). In addition to stress relief, ACC deaminase enzyme supports bacterial endophyte colonization of the plant. Burkholderia phytofirmans PsJN lost the capacity of root elongation in canola plant seedlings when the gene of ACC deaminase was inactivated (sun et al. 2009). A previous study on cut flowers reported the ability of endophytic bacteria to colonize shoot. Moreover, the ACC deaminase enzyme delayed flower senescence (Ali et al. 2012).
Phytohormone Production
Endophytes capable of promoting plant growth considerably produce auxins, principally indole-3-acetic acid (IAA) (Witzel et al. 2012). Auxins act against ethylene and play a major role in promotion of root development and growth. So, endophytic management of auxin production might be a significant tool in awarding salt tolerance in halophytic plants. IAA production was found in (i) species of Serratia, Bacillus, Vibrio, Brevundimonas, Oceanobacillus Exiguobacterium, Staphylococcus, and Halobacillus isolated from four samples of halotolerant plants grown in coastal sandbank of China (Bian et al. 2011) and (ii) salinity-tolerant rhizobacteria (Halomonas sp., Arthrobacter sp., Pseudomonas mendocina, Bacillus pumilus, and Nitrinicola lacisaponensis) originating from extremely saline habitats (Tiwari et al. 2011). It was suggested that IAA, one of the auxins, increases the efficiency of colonization (Suzuki et al. 2003), probably via interference with the host defense system (Navarro et al. 2006), and the production of such compounds or other related compounds might be a significant property for colonization of plant by endophytes. The halophytic plant Prosopis strombulifera also produced ABA, gibberellins, and IAA (Piccoli et al. 2011). ABA is a vital hormone for plant development and growth, and plants increase their ABA levels in stressed conditions. The main role of ABA is to regulate water balance of plant and tolerance of osmotic stress (Tuteja 2007). Wheat plants growing in saline soil showed increased fitness when inoculated with rhizobacteria having IAA producing and salt-tolerant capacity (Tiwari et al. 2011). The function of phytohormones for enhancing salt tolerance has not been analyzed for root fungi either mycorrhizal or endophytic (Rupple et al. 2013).
Nitrogen Fixation
Benefits of endophytes include pathogen suppression, phytohormone production, nutrient supply, and nitrogen fixation; these mechanisms also contribute to the mitigating effects of endophytes when the host plant faces unfavorable ecological conditions (Rupple et al. 2013). Various root endophytes could fix nitrogen (e.g., Azoarcus spp., Acetobacter diazotrophicus, and Herbaspirillum spp.). Nitrogen fixation improves host plant fitness, mostly in poor nitrogen environments. Even if fixed nitrogen in single species is found in a low amount, it should be clarified whether fixed nitrogen is intended for the microbial demands and/or host plant demands. The endophytic strain Paenibacillus P22 isolated from poplar trees conferred the fixed nitrogen to the pool of total nitrogen of host plant, as well as induced changes in plant metabolism (Hardoim et al. 2015).
Compatible Solutes
Sequestration of Na+ and Cl− ions in the vacuole of plant cell causes osmotic pressure. To balance this pressure, metabolically compatible organic solutes must be accumulated (even at elevated concentrations) in organelles and cytosol. Sucrose, glycine betaine, and proline are the most prevalent accumulated solutes (Munns and Tester 2008).
Cumulating organic solutes consider a vital mechanism to counter osmotic pressure, and this was also found in halophytic plants (Flowers and Colmer 2008); proline amino acid was the topic of research to understand the increased salt tolerance in plants colonized with endophytes. Nevertheless, mycorrhizal fungi gave variable results and suggested that accumulation of proline is generally considered as the effect, but it is not the reason for salinity tolerance (Ruiz-Lozano et al. 2012). Osmosis can also be regulated by betaines and sugars. Increased levels of sugars and betaines in mycorrhizal plants suggested that they have a role in salinity tolerance (Manchanda and Garg 2011). The endophyte Pseudomonas pseudoalcaligenes showed an improvement in salinity tolerance of rice by stimulating the accumulation of glycine betaine-like compounds in high concentrations (Jha et al. 2011).
6.3.3 Temperature Stress
Extreme temperature adversely affects plant growth, and high temperature leads to significant damage to cellular proteins that are widely denaturated and aggregated, leading to cell death. On the other hand, low temperature causes impaired metabolism due to inhibition of enzyme reactions, interactions among macromolecules, changes in protein structure, and modulating the membrane properties (Andreas et al. 2012).
Only few reports have the detrimental effects of extreme temperatures, which are often related to water limitation. In this regard, Burkholderia phytofirmans enhance resistance of plants grown at low temperatures (Ait Barka et al. 2006). The grass Dichanthelium lanuginosum was able to survive, although soil temperatures ranged from 38 °C to 65 °C in the Yellowstone National Park due to Curvularia protuberata and its thermal tolerance mycovirus Curvularia (CThTV) (Redman et al. 2002). The fungal endophytes have increased wheat tolerance to temperature regarding grain weight and seed germination of the second generation (Hubbard et al. 2014).
High temperature, precipitation, and latitude can interact and influence the endophyte composition in plants. For example, in sweet root (Osmorhiza depauperata), the endophytes Sinorhizobium meliloti and Agrobacterium tumefaciens were more abundant in locations with higher precipitation and annual temperature, while Paenibacillus strains were more common at sites with lower precipitation and higher latitudes (Li et al. 2012b).
Matsouri et al. (2010) reported that enhanced tolerance of endophyte-colonized plants to temperature and salt stress arise from alterations in ratios of oxidized-to-reduced forms of ascorbate and glutathione as well as lipid peroxidation. Endophytes enhance the adaptation of plant with chilling temperatures. This results in reduced cellular damage, increased photosynthetic activity, and accumulation of various metabolites related to cold stress such as phenolic compounds, proline, and starch. Endophytes also have a positive effect on the metabolic balance, which also reduces the impact of drought stress on wheat growth in reduced watering conditions (Naveed et al. 2014).
6.3.4 Heavy Metal Stress
Toxicity by heavy metals is one of the most important abiotic stresses that cause the loss of about 25–80% of various cultivated crops. In acidic soils, low crop productivity and reduced soil fertility are principally due to manganese and aluminum toxicities along with nutrient deficiencies (K, Mg, P, and Ca) (Singh et al. 2011). Heavy metals are very toxic to roots of cultivated plants and cause poor development of the root system (Singh et al. 2011). Heavy metal toxicity has become a serious problem that restricts crop productivity in acidic soils, in addition to overlapping with many physiological and biochemical processes including nutrient uptake, protein and nitrogen metabolism, photosynthesis, and respiration (Zhang et al. 2009).
It is recognized that bacterial endophytes participate in immobilization and mobilization of the metal cations, which affect availability of cations to plants (Pandey et al. 2016). In Cd-stressed soil, the dark septate endophyte (DSE) Exophiala pisciphila associated with Zea mays root showed improved activity of the antioxidant enzymes (Wang et al. 2016). Three major genes contributing to detoxification, transport, and uptake of Cd have been identified as PCS and MTP upregulation and ZIP downregulation when plants were inoculated with DSE and subsequently exposed to high concentrations of Cd. Changes in the content of 1-aminocyclopropane-1-carboxylate (ACC) by Gigaspora and Pseudomonas can change heavy metal tolerance directly by manipulating levels of plant ethylene (Friesen et al. 2011).
6.3.5 Nutrient Stress
Light, mineral nutrients, carbon, and water are essential prerequisites of plants for development, reproduction, and optimal growth. Starvation and nutrient stress are important abiotic stresses that harm plants (Chaves and Oliveira 2004).
Endophytes can provide their host with micronutrients and macronutrients. Bacteria that have nitrogen-fixing capacity can metabolize root exudates of plants and, in turn, supply nitrogen for the synthesis of plant amino acids. Endophytes can promote growth of plant by gibberellins (GAs), phosphate solubilization, cytokinins, IAA, and production of siderophore and supply essential vitamins to the plant (Jha et al. 2011). Choi et al. (2008) have reported that solubilization of phosphate in wheat and rice was mediated by gibberellic acid produced by Pseudomonas sp. The uptake of mineral nutrients (particularly Zn) in wheat plant has been improved by Azotobacter chroococcum and Piriformospora indica (Abadi and Sepehri 2015). Studies proved the function of endophytes in biological degradation of litter of the host plants. Endophytes initially colonize plants, facilitating the action of saprophytic microbes by antagonism, in that way increasing decomposition of litter (Terekhova and Semenova 2005). Another study explained the ability of all endophytes to decompose the organic components including cellulose lignin and hemicelluloses that facilitate nutrient cycling (He et al. 2012).
6.4 Conclusion
About 300,000 species of plants in the world harbor one or more endophytes. Each endophyte has its own function that helps to improve plant growth and protect it from diverse biotic and abiotic stresses. This benefit does not involve host specificity, so we can use endophytes as inoculants to alleviate abiotic stresses arising from changeful environmental conditions. With increasing interest on environmental protection, food security, and sustainable agriculture, exploiting useful endophytes is urgent. Endophytes may also be a good tool for enhancement of yield and quality of the plant products by producing various kinds of pioneer biologically active metabolites which may be able to positively regulate plant physiological disorders. They can protect plants from pathogens and remediate toxic residues of insecticides, herbicides, and various heavy metals. In addition, it has quick responses in stimulation of immune defense of the host.
Endophytes can be used as alternative strategies to plants that adapted to many stresses like drought, salinity, temperature, nutrient stress, and heavy metals. Further studies of endophytes will provide a better understanding of their relationship with host plant and maximize its utilization as promoters of plant growth as well as its ability to protect the plant from many harmful factors.
References
Abadi VAJM, Sepehri M (2015) Effect of Piriformospora indica and Azotobacter chroococcum on mitigation of zinc deficiency stress in wheat (Triticum aestivum L). Symbiosis 69:9–19
Ait Barka E, Nowak J, Clément C (2006) Enhancement of chilling resistance of inoculated grapevine plantlets with a plant growth-promoting rhizobacterium, Burkholderia phytofirmans strain PsJN. Appl Environ Microbiol 72:7246–7252
Akbarimoghaddam H, Galavi M, Ghanbari A, Panjehkeh N (2011) Salinity effects on seed germination and seedling growth of bread wheat cultivars. Trakia J Sci 9(1):43–50
Akinsanya MA, Ting A, Goh JK, Lim SP (2016) Biodiversity, enzymatic and antimicrobial activities of bacterial endophytes in selected local medicinal plants. J Biomed Pharm Res 16:5(1)
Ali S, Charles TC, Glick BR (2012) Delay of flower senescence by bacterial endophytes expressing 1-aminocyclopropane-1-carboxylate deaminase. J Appl Microbiol 113:1139–1144. https://doi.org/10.1111/j.1365-2672.2012.05409.x
Aly AH, Debbab A, Proksch P (2011) Fungal endophytes: unique plant inhabitants with great promises. Appl J Microbiol Biotechnol 90:1829–1845
Andreas T, Christophe C, Essaïd AB (2012) Physiological and molecular changes in plants at low temperatures. Planta 235:1091–1105. https://doi.org/10.1007/s00425-012-1641-y
Anjum SA, Wang LC, Farooq M, Hussain M, Xue LL, Zou CM (2011) Brassinolide application improves the drought tolerance in maize through modulation of enzymatic antioxidants and leaf gas exchange. J Agron Crop Sci 197:177–185. https://doi.org/10.1111/j.1439-037X.2010.00459.x
Ardanov P, Ovcharenko L, Zaets I, Kozyrovska N, Pirttilä AM (2011) Endophytic bacteria enhancing growth and disease resistance of potato (Solanum tuberosum L.). Biol Control 56:43–49. https://doi.org/10.1016/j.biocontrol.2010.09.014
Ashraf M (2004) Some important physiological selection criteria for salt tolerance in plants. Flora 199:361–376
Ayob FW, Simarani K (2016) Endophytic filamentous fungi from a Catharanthus roseus: identification and its hydrolytic enzymes. Saudi Pharm J 24:273 278
Bae H, Sicher RC, Kim MS, Kim SH, Strem MD, Melnick RL, Bailey BA (2009) The beneficial endophyte Trichoderma hamatum isolate DIS 219b promotes growth and delays the onset of the drought response in Theobroma cacao. J Exp Bot 60(11):3279–3295
Bae H, Roberts DP, Lim H-S, Strem MD, Park S-C, Ryu C-M, Melnick RL, Bailey BA (2011) Endophytic Trichoderma isolates from tropical environments delay disease onset and induce resistance against Phytophthora capsici in hot pepper using multiple mechanisms. Mol Plant-Microbe Interact 24:336–351. https://doi.org/10.1094/MPMI-09-10-0221
Bailey BA, Bae H, Strem MD, Roberts DP, Thomas SE, Crozier J, Samuels GJ, Choi IY, Holmes KA (2006) Fungal and plant gene expression during the colonization of cacao seedlings by endophytic isolates of four Trichoderma species. Planta 224:1449–1464
Baltruschat H, Fodor J, Harrach BD, Niemczyk E, Barna B, Gullner G, Janeczko A, Kogel KH, Schäfer P, Schwarczinger I, Zuccaro A, Schäfer P, Schwarczinger I, Zuccaro A, Skoczowski A (2008) Salt tolerance of barley induced by the root endophyte Piriformospora indica is associated with a strong increase in antioxidants. New Phytol 180:501–510
Bano A, Fatima M (2009) Salt tolerance in Zea mays (L.) following inoculation with Rhizobium and Pseudomonas. Biol Fertil Soils 45:405–413
Bhosale HJ, Kadam TA (2015) Generic diversity and a comparative account on plant growth promoting characteristics of actinobacteria in roots and rhizosphere of Saccharum officinarum. Int J Curr Microbiol App Sci 4:230–244
Bian G, Zhang Y, Qin S, Xing K, Xie H, Jiang J (2011) Isolation and biodiversity of heavy metal tolerant endophytic bacteria from halotolerant plant species located in coastal shoal of Nantong. Acta Microbiol Sin 51(11):1538–1547
Bloemberg GV, Lugtenberg BJ (2001) Molecular basis of plant growth promotion and biocontrol by rhizobacteria. Curr Opin Plant Biol 4(4):343–350
Bogner CW, Kamdem RST, Sichtermann G, Mattheaus C, Heolscher D, Popp J, Proksch P, Grundler FMW, Schouten A (2017) Bioactive secondary metabolites with multiple activities from a fungal endophyte. Microb Biotechnol 10:175–188. https://doi.org/10.1111/1751-7915.12467
Bordiec S, Paquis S, Lacroix H, Dhondt S, Ait Barka E, Kauffmann S, Jeandet P, Mazeyrat-Gourbeyre F, Clement C, Baillieul F, Dorey S (2011) Comparative analysis of defence responses induced by the endo- phytic plant growth-promoting rhizobacterium Burkholderia phytofirmans strain PsJN and the non-host bacterium Pseudomonas syringae pv. pisi in grapevine cell suspensions. J Exp Bot 62:595–603. https://doi.org/10.1093/jxb/erq291
Bowyer C, Withana S, Fenn I, Bassi S, Lewis M, Cooper T, Benito P, Mudgal S (2009) Land Degradation and Desertification Policy Department Economic and Scientific Policy IP/A/ENVI/ST/2008-23. European Parliament, Brussels
Bray EA (2002) Abscisic acid regulation of gene expression during water-deficit stress in the era of the Arabidopsis genome. Plant Cell Environ 25:153–161
Cao MJ, Wang Z, Zhao Q, Mao JL, Speiser A, Wirtz M, Hell R, Zhu JK, Xiang CB (2014) Sulfate availability affects ABA levels and germination response to ABA and salt stress in Arabidopsis thaliana. Plant J 77:604–615
Carrim AJ, Barbosa EC, Vieira JD (2006) Enzymatic activity of endophytic bacterial isolates of Jacaranda decurrens Cham (Carobinha-do-campo). Braz Arch Biol Technol 49:353–359
Chathurdevi G, Gowrie SU (2016) Endophytic fungi isolated from medicinal plant—a source of potential bioactive metabolites. Int J Curr Pharm Res 8:50–56
Chaves MM, Oliveira MM (2004) Mechanisms underlying plant resilience to water deficits: prospects for water-saving agriculture. J Exp Bot 55:2365–2384
Chi F, Shen S, Cheng H, Jing Y, Yanni Y, Dazzo F (2005) Ascending migration of endophytic rhizobia, from roots to leaves, inside rice plants and assessment of benefits to rice growth physiology. Appl Environ Microbiol 71:7271–7278
Choi O, Kim J, Kim JG, Jeong Y, Moon JS, Park CS, Hwang I (2008) Pyrroloquinoline quinone is a plant growth promotion factor produced by Pseudomonas fluorescens B16. Plant Physiol 146:657–668
Cohen AC, Bottini R, Piccoli P (2008) Azospirillum brasilense Sp 245 produces ABA in chemically-defined culture medium and increases ABA content in Arabidopsis plants. Plant Growth Regul 54:97–103
Compant S, Clément C, Sessitsch A (2010) Plant growth-promoting bacteria in the rhizo- and endosphere of plants: their role, colonization, mechanisms involved and prospects for utilization. Soil Biol Biochem 42:669–678. https://doi.org/10.1016/j.soilbio.2009.11.024
Convey P (2011) Antarctic terrestrial biodiversity in a changing world. Polar Biol 34:1629e1641
Czarny JC, Grichko VP, Glick BR (2006) Genetic modulation of ethylene biosynthesis and signaling in plants. Biotechnol Adv 24(4):410–419. https://doi.org/10.1016/j.biotechadv.2006.01.003
De Bashan LE, Hernandez JP, Bashan Y (2012) The potential contribution of plant growth-promoting bacteria to reduce environmental degradation- a comprehensive evaluation. Appl Soil Ecol 61:171–189. https://doi.org/10.1016/j.apsoil.2011.09.003
De Smet I, Zhang H, Inzé D, Beeckman T (2006) A novel role for abscisic acid emerges from underground. Trends Plant Sci 11:434–439
Dudeja SS, Giri R, Saini R, Suneja-Madan P, Kothe E (2012) Interaction of endophytic microbes with legumes. J Basic Microbiol 52:248–260
Flowers TJ, Colmer TD (2008) Salinity tolerance in halophytes. New Phytol 179(4):945–963. https://doi.org/10.1111/j.1469-8137.2008.02531.x
Fouda AH, Hassan SE, Eid AM, Ewais EE (2015) Biotechnological applications of fungal endophytes associated with medicinal plant Asclepias sinaica (Bioss.). Ann Agric Sci 60:95–104
Fouda A, Abdel-Maksoud G, Abdel-Rahman MA, Salem SS, Hassan SE, El-Sadany MA (2019a) Eco-friendly approach utilizing green synthesized nanoparticles for paper conservation against microbes involved in biodeterioration of archaeological manuscript. Int Biodeterior Biodegrad 142:160–169
Fouda A, Hassan SE, Eid AM, El-Din Ewais E (2019b) The interaction between plants and bacterial endophytes under salinity stress. In: Jha S (ed) Endophytes and secondary metabolites. Springer, Cham, pp 1–18
Friesen M, Porter SS, Stark SC, von Wettberg EJ, Sachs JL, Martinez Romero E (2011) Microbially mediated plant functional traits. Annu Rev Ecol Evol Syst 42:23–46
Gangwar M, Dogra S, Sharma N (2011) Antagonistic bioactivity of endophytic actinobacteria isolated from medicinal plants. J Adv Lab Res Biol 2(4):1–4
Ghodhbane-Gtari F, Essoussi I, Chattaoui M, Chouaia B, Jaouani A, Daffonchio D, Boudabous A, Gtari M (2010) Isolation and characterization of non-Frankia actinobacteria from root nodules of Alnus glutinosa, Casuarina glauca and Elaeagnus angustifolia. Symbiosis 50:51–57
Glick BR (2014) Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol Res 169(1):30–39
Glick BR, Penrose DM, Li JP (1998) A model for the lowering of plant ethylene concentrations by plant growth-promoting bacteria. J Theor Biol 190:63–68. https://doi.org/10.1006/jtbi.1997.0532
Granier C, Tardieu F (1999) Water deficit and spatial pattern of leaf development. Variability in responses can be stimulated using a simple model of leaf development. Plant Physiol 119:609–619
Gray EJ, Smith DL (2005) Intracellular and extracellular PGPR: commonalities and distinctions in the plant–bacterium signaling processes. Soil Biol Biochem 37:395–412
Greenberg BM, Huang XD, Gerwing P, Yu XM, Chang P, Wu SS, Gerhardt K, Nykamp J, Lu X, Glick B (2008) Phytoremediation of salt impacted soils: greenhouse and the field trials of plant growth promoting rhizobacteria (PGPR) to improve plant growth and salt phyto-accumulation. In: Proceeding of the 33rd AMOP technical seminar on environmental contamination and response. Environment Canada, Ottawa, pp 627–637
Gunatilaka AAL (2006) Natural products from plant-associated micro-organisms: distribution, structural diversity, bioactivity, and implications of their occurrence. J Nat Prod 69:509–526. https://doi.org/10.1021/np058128
Guo B, Wang Y, Sun X, Tang K (2008) Bioactive natural products from endophytes: a review. Prikl Biokhim Mikrobiol 44:153–158
Gupta V, Trivedi N, Kumar M, Reddy CR, Jha B (2013) Purification and characterization of exo-b-agarase from an endophytic marine bacterium and its catalytic potential in bioconversion of red algal cell wall polysaccharides into galactans. Biomass Bioenergy 28:290–298
Hamilton CE, Bauerle TL (2012) A new currency for mutualism? Fungal endophytes alter antioxidant activity in hosts responding to drought. Fungal Divers 54:39–49
Hamilton CE, Gundel PE, Helander M, Saikkonen K (2012) Endophytic mediation of reactive oxygen species and antioxidant activity in plants: a review. Fungal Divers 54(1):1–10. https://doi.org/10.1007/s13225-012-0158-9
Hardoim PR, Overbeek LS, Elsas JD (2008) Properties of bacterial endophytes and their proposed role in plant growth. Trends Microbiol 16:463–471. https://doi.org/10.1016/j.tim.2008.07.008
Hardoim PR, van Overbeek LS, Berg G, Pirttilä AM, Compant S, Campisano A, Döring M, Sessitsch A (2015) The hidden world within plants: ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol Mol Biol Rev 79(3):293–320. https://doi.org/10.1128/MMBR.00050-14
Hasegawa S, Meguro A, Nishimura T, Kunoh H (2004) Drought tolerance of tissue- cultured seedlings of mountain laurel (Kalmia latifolia L.) induced by an Endophytic actinomycete. Actinomycetologica 18:43–47
Hassan SE (2017) Plant growth-promoting activities for bacterial and fungal endophytes isolated from medicinal plant of Teucrium polium L. J Adv Res 8(6):687–695
Hassan SE, Salem SS, Fouda A, Awad MA, El-Gamal MS, Abdo AM (2018) New approach for antimicrobial activity and bio-control of various pathogens by biosynthesized copper nanoparticles using endophytic actinomycetes. J Radiat Res Appl Sci 11:262–270
Hassan SE, Fouda A, Radwan AA, Salem SS, Barghoth MG, Awad MA, El-Gamal MS, Abdo AM (2019) Endophytic actinomycetes Streptomyces spp. mediated biosynthesis of copper oxide nanoparticles as a promising tool for biotechnological applications. J Biol Inorg Chem 24(3): 377–393
Hata K, Atari R, Sone K (2002) Isolation of endophytic fungi from leaves of Pasania edulis and their within-leaf distributions. Mycoscience 43(5):369–373
He X, Han G, Lin Y, Tian X, Xiang C, Tian Q, Wang F, He Z (2012) Diversity and decomposition potential of endophytes in leaves of a Cinnamomum camphora plantation in China. Ecol Res 27:273–284
Higginbotham SJ, Arnold AE, Ibañez A, Spadafora C, Coley PD, Kursar TA (2013) Bioactivity of fungal endophytes as a function of endophyte taxonomy and the taxonomy and distribution of their host plants. PLoS One 8:e73192. https://doi.org/10.1371/journal.pone.0073192
Hu Y, Schmidhalter U (2002) Limitation of salt stress to plant growth. In: Hock B, Elstner CF (eds) Plant Toxicology. Marcel Dekker Inc, New York, pp 91–224
Hubbard M, Germida JJ, Vujanovic V (2014) Fungal endophytes enhance wheat heat and drought tolerance in terms of grain yield and second-generation seed viability. J Appl Microbiol 116:109–122
Iniguez AL, Dong YM, Carter HD, Ahmer BMM, Stone JM, Triplett EW (2005) Regulation of enteric endophytic bacterial colonization by plant defenses. Mol Plant-Microbe Interact 18:169–178. https://doi.org/10.1094/MPMI-18-0169
Jahromi F, Aroca R, Porcel R, Ruiz-Lozano JM (2008) Influence of salinity on the in vitro development of Glomus intraradices and on the in vivo physiological and molecular responses of mycorrhizal lettuce plants. Microb Ecol 55:45–53
Jamil A, Riaz S, Ashraf M, Foolad MR (2011) Gene expression profiling of plants under salt stress. Crit Rev Plant Sci 30(5):435–458
Jha Y, Subramanian RB, Patel S (2011) Combination of endophytic and rhizospheric plant growth promoting rhizobacteria in Oryza sativa shows higher accumulation of osmoprotectant against saline stress. Acta Physiol Plant 33:797–802. https://doi.org/10.1007/s11738-010-0604-9
Jones A, Panagos P, Barcelo S, Bouraoui F, Bosco C, Dewitte O, Gardi C, Hervás J, Hiederer R, Jeffery S (2012) The state of soil in Europe -a contribution of the JRC to the European Environment Agency’s Environment State and Outlook R- SOER 2010
Kannahi M, Senbagam N (2014) Studies on siderophore production by microbial isolates obtained from rhizosphere soil and its antibacterial activity. J Chem Pharm Res 6(4):1142–1145
Kannan R, Damodaran T, Umamaheswari S (2015) Sodicity tolerant polyembryonic mango root stock plants: a putative role of endophytic bacteria. Afr J Biotechnol 14:350–359
Khan AL, Hamayun M, Hussain J, Kang SM, Lee IJ (2012) The newly isolated endophytic fungus Paraconiothyrium sp. LK1 produces ascotoxin. Molecules 17:1103–1112
Khan AL, Waqas M, Hussain J, Al-Harrasi A, Lee IJ (2014) Fungal endophyte Penicillium janthinellum LK5 can reduce cadmium toxicity in Solanum lycopersicum (Sitiens and Rhe). Biol Fertil Soils 50:75–85
Khan AL, Al-Harrasi A, Al-Rawahi A, Al-Farsi Z, Al-Mamari A, Waqas M, Asaf S, Elyassi A, Mabood F, Shin JH, Lee IJ (2016) Endophytic fungi from frankincense tree improves host growth and produces extracellular enzymes and indole acetic acid. PLoS One 11:e0158207
Kharwar RN, Mishra A, Gond SK, Stierle A, Stierle D (2011) Anticancer compounds derived from fungal endophytes: their importance and future challenges. Nat Prod Rep 28:1208–1228
Kim YC, Glick BR, Bashan Y, Ryu CM (2012) Enhancement of plant drought tolerance by microbes. In: Aroca R (ed) Plant responses to drought stress: from morphological to molecular features. Springer, Heidelberg, pp 383–413. https://doi.org/10.1007/978-3-642-32653-0_15
Klessig DF, Tian M, Choi HW (2016) Multiple targets of salicylic acid and its derivatives in plants and animals. Front Immunol 7:206. https://doi.org/10.3389/fimmu.2016.00206
Kloepper JW, Ryu CM (2006) Bacterial endophytes as elicitors of induced systemic resistance. In: Schulz BJE, Boyle CJC, Sieber TN (eds) Microbial root endophytes. Springer, Berlin, pp 33–52. https://doi.org/10.1007/3-540-33526-9_3
Kusari S, Verma VC, Lamshoeft M, Spiteller M (2012a) An endophytic fungus from Azadirachta indica A. Juss. That produces azadirachtin. World J Microbiol Biotechnol 28:1287–1294
Kusari S, Hertweck C, Spiteller M (2012b) Chemical ecology of endophytic fungi: origin of secondary metabolites. Chem Biol 19:792–798
Leo VV, Passari AK, Joshi JB, Mishra VK, Uthandi S, Ramesh N, Gupta VK, Saikia R, Sonawane VC, Singh BP (2016) A novel triculture system (CC3) for simultaneous enzyme production and hydrolysis of common grasses through submerged fermentation. Front Microbiol 7. https://doi.org/10.3389/fmicb.2016.00447
Li WK (2005) Endophytes and Natural Medicines. Chin J Nat Med 3(4):193–199
Li HY, Wei DQ, Shen M, Zhou ZP (2012a) Endophytes and their role in phytoremediation. Fungal Divers 54:11–18
Li L, Sinkko H, Montonen L, Wei G, Lindstrom K, Rasanen LA (2012b) Biogeography of symbiotic and other endophytic bacteria isolated from medicinal Glycyrrhiza species in China. FEMS Microbiol Ecol 79:46–68
Liarzi O, Bucki P, Braun Miyara S, Ezra D (2016) Bioactive volatiles from an endophytic Daldinia cf. concentrica isolate affect the viability of the plant parasitic nematode Meloidogyne javanica. PLoS One 11:e0168437. https://doi.org/10.1371/journal.pone.0168437
Limtong S, Kaewwichian R, Yongmanitchai W, Kawasaki H (2014) Diversity of culturable yeasts in phylloplane of sugarcane in Thailand and their capability to produce indole-3-acetic acid. World J Microbiol Biotechnol 30(6):1785–1796
Liu G, Lai D, Liu ZQ, Zhou L, Liu LZ (2016) Identification of nematicidal constituents of Notopterygium incisum rhizomes against Bursaphelenchus xylophilus and Meloidogyne incognita. Molecules 21:1276. https://doi.org/10.3390/molecules21101276
Malinowski CP, Beleskey DP (2000) Adaptations of endophyte-infected cool-season grasses to environmental stresses: mechanisms of drought and mineral stress tolerance. Crop Sci 40:923–940
Manchanda G, Garg N (2011) Alleviation of salt-induced ionic, osmotic and oxidative stresses in Cajanus cajan nodules by AM fungi inoculation. Plant Biosyt 145(1):88–97. https://doi.org/10.1080/11263504.2010.539851
Mastretta C, Taghavi S, van der Lelie D et al (2009) Endophytic bacteria from seeds of Nicotiana tabacum can reduce cadmium phytotoxicity. Int J Phytoremediation 11(3):251–267
Matsouri F, Björkman T, Harman GE (2010) Seed treatment with Trichoderma harzianum alleviates biotic, abiotic, and physiological stresses in germinating seeds and seedlings. Biol Control 100:1213–1221
Meena KK, Sorty AM, Bitla UM, Choudhary K, Gupta P, Pareek A, Singh DP, Prabha R, Sahu PK, Gupta VK, Singh HB, Krishanani KK, Minhas PS (2017) Abiotic stress responses and microbe-mediated mitigation in plants: the omics strategies. Front Plant Sci 8:172. https://doi.org/10.3389/fpls.2017.00172
Miransari M (2012) Role of phytohormone signaling during stress. In: Ahmad P, Prasad MNV (eds) Environmental adaptations and stress tolerance of plants in the era of climate change. Springer, New York, p 381_393
Molina G, Pimentel MR, Bertucci TCP, Pastore GM (2012) Application of fungal endophytes in biotechnological processes. Chem Eng Trans 27:289–294
Munns R (2002) Comparative physiology of salt and water stress. Plant Cell Environ 25:239–250. https://doi.org/10.1046/j.0016-8025.2001.00808.x
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681
Navarro L, Dunoyer P, Jay F, Arnold B, Dharmasiri N, Estelle M, Voinnet O, Jones JDG (2006) A plant miRNA contributes to antibacterial resistance by repressing auxin signaling. Science 312:436–439. https://doi.org/10.1126/science.1126088
Naveed M, Hussain MB, Zahir ZA, Mitter B, Sessitsch A (2014) Drought stress amelioration in wheat through inoculation with Burkholderia phytofirmans strain PsJN. Plant Growth Regul 73:121–131. https://doi.org/10.1007/s10725-013-9874-8
Netondo GW, Onyango JC, Beck E (2004) Sorghum and salinity: II. Gas exchange and chlorophyll fluorescence of sorghum under salt stress. Crop Sci 44:806–811
Padda KP, Puri A, Chanway CP (2016) Plant growth promotion and nitrogen fixation in canola by an endophytic strain of Paenibacillus polymyxa and its GFP-tagged derivative in a long-term study. Botany 94:1209–1217. https://doi.org/10.1139/cjb-2016-0075
Palacios OA, Bashan Y, de-Bashan LE (2014) Proven and potential involvement of vitamins in interactions of plants with plant growth promoting bacteria-an overview. Biol Fertil Soils 50:415–432. https://doi.org/10.1007/s00374-013-0894-3
Pandey PK, Singh S, Singh AK, Samanta R, Yadav RNS, Singh MC (2016) Inside the plant: Bacterial endophytes and abiotic stress alleviation. J Appl Nat Sci 8(4):1899–1904
Parker MA (1995) Plant fitness variation caused by different mutualist genotypes. Ecology 76(5):1525–1535
Partida-Martínez LP, Heil M (2011) The microbe-free plant: fact or artifact? Front Plant Sci 29(2):100. https://doi.org/10.3389/fpls.2011.00100.eCollection
Passari AK, Mishra VK, Singh G, Singh P, Kumar B, Gupta VK, Sarma RK, Saikia R, Donovan AO, Singh BP (2017) Insights into the functionality of endophytic actinobacteria with a focus on their biosynthetic potential and secondary metabolites production. Sci Rep 7:11809. https://doi.org/10.1038/s41598-017-12235-4
Pereira SIA, Monteiro C, Vega AL, Castro PML (2016) Endophytic culturable bacteria colonizing Lavandula dentate L. plants: isolation, characterization and evaluation of their plant growth-promoting activities. Ecol Eng 87:91–96
Piccoli P, Travaglia C, Cohen A, Sosa L, Cornejo P, Masuelli R, Bottini R (2011) An endophytic bacterium isolated from roots of the halophyte Prosopis strombulifera produces ABA, IAA, gibberellins A(1) and A(3) and jasmonic acid in chemically-defined culture medium. Plant Growth Regul 64(2):207–210. https://doi.org/10.1007/s10725-010-9536-z
Pieterse CM, Van der Does D, Zamioudis C, Leon-Reyes A, Van Wees SC (2012) Hormonal modulation of plant immunity. Annu Rev Cell Dev Biol 28:489–521. https://doi.org/10.1146/annurev-cellbio-092910-154055
Pieterse CM, Zamioudis C, Berendsen RL, Weller DM, Van Wees SC, Bakker PA (2014) Induced systemic resistance by beneficial microbes. Annu Rev Phytopathol 52:347–375
Pirttilä AM, Pospiech H, Laukkanen H, Myllyla R, Hohtola A (2003) Two endophytic fungi in different tissues of Scots pine buds (Pinus sylvestris L.). Microb Ecol 45(1):53–62
Pirttilä AM, Podolich O, Koskimäki JJ, Hohtola E, Hohtola A (2008) Role of origin and endophyte infection in browning of bud-derived tissue cultures of Scots pine (Pinus sylvestris L.). Plant Cell Tissue Organ Cult 95(1):47–55
Prashar P, Kapoor N, Sachdeva S (2014) Rhizosphere: its structure, bacterial diversity and significance. Rev Environ Sci Biotechnol 13(1):63–77
Prashith-Kekuda TR (2016) Isolation, characterization and antimicrobial potential of endophytic actinomycetes. Int J Curr Microbiol App Sci 5:100–116. https://doi.org/10.20546/ijcmas.2016.507.008
Puri A, Padda KP, Chanway CP (2016) Evidence of nitrogen fixation and growth promotion in canola (Brassica napus L.) by an endophytic diazotroph Paenibacillus polymyxa P2b-2R. Biol Fertil Soils 52:119–125. https://doi.org/10.1007/s00374-015-1051-y
Raaijmakers J, Paulitz TC, Steinberg C, Alabouvette C, Moënne-Loccoz Y (2009) The rhizosphere: a playground and battlefield for soil borne pathogens and beneficial microorganisms. Plant Soil 321:341–361. https://doi.org/10.1007/s11104-008-9568-6
Radhakrishnan R, shim k-b, lee b-w, Hwang c-d, Pae S-B, Park C-H, Kim S-U, Lee C-K, In YB (2013) IAA-producing Penicillium sp. NICS01 triggers plant growth and suppresses Fusarium sp.-induced oxidative stress in sesame (Sesamum indicum L.). J Microbiol Biotechnol 23:856–863
Redman RS, Sheehan KB, Stout RG, Rodriguez RJ, Henson JM (2002) Thermotolerance generated by plant/fungal symbiosis. Science 298:1581–1581
Redman RS, Kim YO, Woodward CJ, Greer C, Espino L, Doty SL, Rodriguez RJ (2011) Increased fitness of rice plants to abiotic stress via habitat adapted symbiosis: a strategy for mitigating impacts of climate change. PLoS One 6:e14823
Ren JH, Ye JR, Liu H, Xu XL, Wu XQ (2011) Isolation and characterization of a new Burkholderia pyrrocinia strain JK SH007 as a potential biocontrol agent. World J Microbiol Biotechnol 27(9):2203–2215
Rodriguez RJ, Henson J, Van Volkenburgh E, Hoy M, Wright L, Beckwith F, Kim YO, Redman RS (2008) Stress tolerance in plants via habitat-adapted symbiosis. ISME 2:404–416
Rodriguez RJ, White JFJR, Arnold AE, Redman RS (2009) Fungal endophytes: diversity and functional roles-Tansley review. New Phytol 182:314–330
Rouhier N, San Koh C, Gelhaye E, Corbier C, Favier F, Didierjean C, Jacquot JP (2008) Redox based anti- oxidant systems in plants: biochemical and structural analyses. Biochim Biophys Acta 1780:1249–1260
Ruanpanun P, Tangchitsomkid N, Hyde KD, Lumyong S (2010) Actinomycetes and fungi isolated from plant-parasitic nematode infested soils: screening of the effective biocontrol potential, indole-3-acetic acid and siderophore production. World J Microbiol Biotechnol 26:1569–1578
Ruiz-Lozano JM, Porcel R, Azcon C, Aroca R (2012) Regulation by arbuscular mycorrhizae of the integrated physiological response to salinity in plants: new challenges in physiological and molecular studies. J Exp Bot 63(11):4033–4044. https://doi.org/10.1093/jxb/ers126
Rungin S, Indanand C, Suttiviriya P, Kruasuwan W, Jaemsaeng R, Thamchaipenet A (2012) Plant growth enhancing effects by a siderophore producing endophytic streptomycete isolated from a Thai jasmine rice plant (Oryza sativa L. cv. KDML105). Antonie Van Leeuwenhoek 102:463–472
Rupple S, Franken P, Witzel K (2013) Properties of the halophyte microbiome and their implications for plant salt tolerance. Funct Plant Biol 40:940–951. https://doi.org/10.1071/FP12355
Russell JR, Huang J, Anand P, Kucera K, Sandoval AG, Dantzler KW, Hickman D, Jee J, Kimovec FM, Koppstein D, Marks DH, Mittermiller PA, Núñez SJ, Santiago M, Townes MA, Vishnevetsky M, Williams NE, Vargas MP, Boulanger LA, Bascom-Slack C, Strobel SA (2011) Biodegradation of polyester polyurethane by endophytic fungi. Appl Environ Microbiol 77(17):6076–6084
Saharan BS, Nehra V (2011) Plant growth promoting rhizobacteria: a critical review. Life Sci Med Res 21(1):30–37
Salam N, Khieu T, Liu M, Vu T, Chu-Ky S, Quach N, Phi Q, Rao MPN, Fontana A et al (2017) Endophytic actinobacteria associated with Dracaena cochinchinensis Lour.: isolation, diversity, and their cytotoxic activities. Biomed Res Int 2017:1. https://doi.org/10.1155/2017/1308563. (Article ID 1308563
Schulz B, Boyle C, Draeger S, R€ommert AK, Krohn K (2002) Endophytic fungi: a source of novel biologically active secondary metabolites. Mycol Res 109:996–1004
Seckin B, Sekmen AH, Turkan I (2009) An enhancing effect of exogenous mannitol on the antioxidant enzyme activities in roots of wheat under salt stress. J Plant Growth Regul 28:12–20
Sekmen AH, Turkan I, Takio S (2007) Differential responses of antioxidative enzymes and lipid peroxidation to salt stress in salt-tolerant Plantago maritima and salt- sensitive Plantago media. Physiol Plant 131:399–411
Shankar Naik B, Krishnamurthy YL (2010) Endophytes: the real untapped high energy biofuel resource. Curr Sci 98(7):883
Shi Y, Lou K, Li C (2009) Promotion of plant growth by phytohormone-producing endophytic microbes of sugar beet. Biol Fertil Soils 45:645–653
Shinozaki K, Yamaguchi-Shinozaki K (2007) Gene networks involved in drought stress response and tolerance. J Exp Bot 58:221–227
Shrivastava P, Kumar R (2015) Soil salinity: a serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J Biol Sci 22:123–131
Singh D, Singh NP, Chauhan SK, Singh P (2011) Developing aluminium tolerant crop plants using biotechnological tools. Curr Sci 100(12):1807–1814
Smith SA, Tank DC, Boulanger LA, Bascom-Slack CA, Eisenman K, Kingery D, Babbs B, Fenn K, Greene JS, Hann BD, Keehner J, Kelley-Swift EG, Kembaiyan V, Lee SJ, Li P, Light DY, Lin EH, Ma C, Moore E, Schorn MA, Vekhter D, Nunez PV, Strobel GA, Donoghue MJ, Strobel SA (2008) Bioactive endophytes warrant intensified exploration and conservation. PLoS One 3(8):e3052
Stolte J, Tesfai M, Øygarden L, Kværnø S, Keizer J, Verheijen F, Panagos P, Ballabio C, Hessel R (2015) Soil threats in Europe: status, methods, drivers and effects on ecosystem services. A review report, deliverable 2.1 of the RECARE Project, vol EUR 27607. Office for Official Publications of the European Community, Luxembourg, pp 69–78
Strobel GA (2003) Endophytes as sources of bioactive products. Microbes Infect 5:535–544
Strobel G, Daisy B (2003) Bioprospecting for microbial endophytes and their natural products. Microbiol Mol Biol Rev 67:491–502
Strong PJ, Claus H (2011) Laccase: A review of its past and its future in bioremediation. Crit Rev Environ Sci Technol 41(4):373
Sturz AV, Christie BR, Nowak J (2000) Bacterial endophytes: potential role in developing sustainable systems of crop production. CRC Crit Rev Plant Sci 19:1–30
Sun Y, Cheng Z, Glick BR (2009) The presence of a 1-aminocyclopro- pane-1-carboxylate (ACC) deaminase deletion mutation alters the physiology of the endophytic plant growth-promoting bacterium Burkholderia phytofirmans PsJN. FEMS Microbiol Lett 296:131–136. https://doi.org/10.1111/j.1574-6968.2009.01625.x
Sunitha VH, Devi DN, Srinivas C (2013) Extracellular enzymatic activity of endophytic fungal strains isolated from medicinal plants. WJAS 9:1–9
Suzuki S, He YX, Oyaizu H (2003) Indole-3-acetic acid production in Pseudomonas fluorescens HP72 and its association with suppression of creeping bentgrass brown patch. Curr Microbiol 47:138–143. https://doi.org/10.1007/s00284-002-3968-2
Tadych M, Bergen M, White JF (2014) Epichloë spp. associated with grasses: new insights on life cycles, dissemination and evolution. Mycologia 106(2):181–201
Tang Q, Puri A, Padda KP, Chanway CP (2017) Biological nitrogen fixation and plant growth promotion of lodgepole pine by an endophytic diazotroph Paenibacillus polymyxa and its GFP-tagged derivative. Botany 95:611–619. https://doi.org/10.1139/cjb-2016-0300
Tefera T, Vidal S (2009) Effect of inoculation method and plant growth medium on endophytic colonization of sorghum by the entomopathogenic fungus Beauveria bassiana. BioControl 54(5):663–669
Tejesvi MV, Pirttilä AM (2011) Potential of tree endophytes as sources for new drug compounds. In: Pirttilä AM, Frank AC (eds) Endophytes of forest trees: biology and applications. Springer, New York, pp 295–312
Tejesvi MV, Kajula M, Mattila S, Pirttilä AM (2011) Bioactivity and genetic diversity of endophytic fungi in Rhododendron tomentosum Harmaja. Fungal Divers 47:97–107. https://doi.org/10.1007/s13225-010-0087-4
Tejesvi MV, Segura DR, Schnorr KM, Sandvang D, Mattila S, Olsen PB, Neve S, Kruse T, Kristensen HH, Pirttilä AM (2013) An antimicrobial peptide from endophytic Fusarium tricinctum of Rhododendron tomentosum Harmaja. Fungal Divers 60:153–159. https://doi.org/10.1007/s13225-013-0227-8
Terekhova VA, Semenova TA (2005) The structure of micromycete communities and their synecologic interactions with basidiomycetes during plant debris decomposition. Microbiology 74:91–96
Timmusk S, Paalme V, Pavlicek T, Bergquist J, Vangala A, Danilas T, Nevo E (2011) Bacterial distribution in the rhizosphere of wild barley under contrasting microclimates. PLoS One 6:e17968
Tintjer T, Leuchtmann A, Clay K (2008) Variation in horizontal and vertical transmission of the endophyte Epichloë elymi infecting the grass Elymus hystrix. New Phytol 179(1):236–246
Tiwari S, Singh P, Tiwari R, Meena KK, Yandigeri M, Singh DP, Arora DK (2011) Salt tolerant rhizobacteria mediated induced tolerance in wheat (Triticum aestivum) and chemical diversity in rhizosphere enhance plant growth. Biol Fertil Soils 47(8):907–916. https://doi.org/10.1007/s00374-011-0598-5
Traving SJ, Thygesen UH, Riemann L, Stedmon CA (2015) A model of extracellular enzymes in free-living microbes: which strategy pays off? Appl Environ Microbiol 81:7385–7393
Tuteja N (2007) Abscisic acid and abiotic stress signaling. Plant Signal Behav 2(3):135–138. https://doi.org/10.4161/psb.2.3.4156
Vijayalakshmi R, Kairunnisa K, Sivvaswamy SN, Dharan SS, Natarajan S (2016) Enzyme production and antimicrobial activity of endophytic bacteria isolated from medicinal plants. Indian J Sci Technol 9(14):23–32
Waller F, Achatz B, Baltruschat H, Fodor J, Becker K, Fischer M, Heier T, Hückelhoven R, Neumann C, von Wettstein D, Franken P, Kogel KH (2005) The endophytic fungus Piriformospora indica reprograms barley to salt stress tolerance, disease resistance, and higher yield. Proc Natl Acad Sci U S A 102:13386–13391
Wang Y, Dai CC (2011) Endophytes: a potential source for biosynthesis transformation and biodegradation. Ann Microbiol 61:207–215
Wang S, Hu T, Jiao Y, Wei J, Cao K (2009) Isolation and characterization of Bacillus subtilis EB-28, an endophytic bacterium strain displaying biocontrol activity against Botrytis cinereal Pers. Front Agric China 3(3):247–252
Wang M, Xing Y, Wang J, Xu Y, Wang G (2014) The role of the chi1 gene from the endophytic bacteria Serratia proteamaculans 336x in the biological control of wheat take-all. Can J Microbiol 60(8):533–540
Wang JL, Li T, Liu GY, Smith JM, Zhao ZW (2016) Unravelling the role of dark septate endophyte (DSE) colonizing maize (Zea mays) under cadmium stress: physiological, cytological and genic aspects. Sci Rep 6:22028
Wani ZA, Ashraf N, Mohiuddin T, Riyaz-Ul-Hassan S (2015) Plant-endophyte symbiosis, an ecological perspective. Appl Microbiol Biotechnol 99:2955–2965
Waqas M, Khan AL, Kamran M, Hamayun M, Kang SM, Kim YH, Lee IJ (2012) Endophytic fungi produce gibberellins and indoleacetic acid and promotes host-plant growth during stress. Molecules 17:10754–10773
Waqas M, Khan AL, Hamayun M, Shahzad R, Kang SM, Kim JG et al (2015) Endophytic fungi promote plant growth and mitigate the adverse effects of stem rot: an example of Penicillium citrinum and Aspergillus terreus. J Plant Interact 10:280_287
Wen CY (2004) Recent advances and issues on the endophyte. Chinese J Ecol 23(2):86–91
Wilson BJ, Addy HD, Tsuneda A, Hambleton S, Currah RS (2004) Phialocephala sphaeroides sp. nov., a new species among the dark septate endophytes from a boreal wetland in Canada. Can J Bot 82:607–617. https://doi.org/10.1139/b04-030
Wingender J, Neu TR, Flemming HC (1999) Microbial extracellular polymeric substances: characterization, structure and function. Springer, Berlin/Heidelberg. https://doi.org/10.1007/978-3-642-60147-7
Witzel K, Gwinn-Giglio M, Nadendla S, Shefchek K, Ruppel S (2012) Genome sequence of Enterobacter radicincitans DSM16656T, a plant growth-promoting endophyte. J Bacteriol 194(19):5469. https://doi.org/10.1128/JB.01193-12
Yang H, Puri A, Padda KP, Chanway CP (2016) Effects of Paenibacillus polymyxa inoculation and different soil nitrogen treatments on lodgepole pine seedling growth. Can J For Res 46:816–821. https://doi.org/10.1139/cjfr-2015-0456
Yu H, Zhang L, Li L, Zheng C, Guo L, Li W, Sun P, Qin L (2010) Recent developments and future prospects of antimicrobial metabolites produced endophytes. Microbiol Res 165:437–449
Zamioudis C, Pieterse CMJ (2012) Modulation of host immunity by beneficial microbes. Mol Plant-Microbe Interact 25:139–150. https://doi.org/10.1094/MPMI-06-11-0179
Zhang YP, Nan ZB (2007) Growth and anti-oxidative systems changes in Elymus dahuricus is affected by Neotyphodium endophyte under contrasting water availability. J Agron Crop Sci 193:377–386
Zhang YP, Nan ZB (2010) Germination and seedling anti-oxidative enzymes of endophyte-infected populations of Elymus dahuricus under osmotic stress. Seed Sci Technol 38:522–527
Zhang SQ, Outlaw WH Jr (2001) Abscisic acid introduced into the transpiration stream accumulates in the guard-cell apoplast and causes stomatal closure. Plant Cell Environ 24:1045–1054
Zhang F, Zhang H, Wang G, Xu L, Shen Z (2009) Cadmium-induced accumulation of hydrogen peroxide in the leaf apoplast of Phaseolus aureus and Vicia sativa and the roles of different antioxidant enzymes. J Hazard Mater 168:76–84
Zhang X, Li J, Qi G, Wen K, Lu L, Zhao X (2011) Insecticidal effect of recombinant endophytic bacterium containing Pinellia ternata agglutinin against white backed plant hopper, Sogatella furcifera. Crop Prot 30(11):1478–1484
Zhao X, Qi G, Zhang X, Lan N, Ma X (2010) Controlling sapsucking insect pests with recombinant endophytes expressing plant lectin. Nat Precedings 21:21
Zhao K, Penttinen P, Guan T, Xiao J, Chen Q, Xu J et al (2011) The diversity and anti-microbial activity of endophytic actinomycetes isolated from medicinal plants in Panxi Plateau China. Curr Microbiol 62:182_190
Zhu JK (2000) Over expression of a delta-pyrroline-5- carboxylate synthetase gene and analysis of tolerance to water and salt stress in transgenic rice. Trends Plant Sci 6:66–72
Zhu JK (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53:247–273
Zinniel DK, Lambrecht P, Harris NB, Feng Z, Kuczmarski D, Higley P (2002) Isolation and characterization of endophytic colonizing bacteria from agronomic crops and prairie plants. Appl Environ Microbiol 68:2198–2208. https://doi.org/10.1128/AEM.68.5.2198-2208.2002
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Eid, A.M., Salim, S.S., Hassan, S.ED., Ismail, M.A., Fouda, A. (2019). Role of Endophytes in Plant Health and Abiotic Stress Management. In: Kumar, V., Prasad, R., Kumar, M., Choudhary, D. (eds) Microbiome in Plant Health and Disease. Springer, Singapore. https://doi.org/10.1007/978-981-13-8495-0_6
Download citation
DOI: https://doi.org/10.1007/978-981-13-8495-0_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-8494-3
Online ISBN: 978-981-13-8495-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)