Abstract
The productivity of oilseed brassicas is severely affected by aphid pests. Among the different aphid species, turnip/mustard aphid, Lipaphis erysimi (Kaltenbach), is the key pest of oilseed brassicas in Indian subcontinent inflicting 35.4–91.3% losses under different agroclimatic conditions. The development of an aphid-resistant cultivar offers an effective, economic and eco-friendly method of its management which requires the availability of a crossable source of resistance. Brassica plants employ a plethora of biophysical and biochemical defence mechanisms against insects, which range from surface waxes and trichomes to production of toxic biochemicals such as glucosinolates, isothiocyanates, lectins, volatiles, alkaloids, etc. Such resistant plants can be identified by an effective screening protocol, and the gene(s) of interest can be transferred to the desirable agronomic background by conventional breeding or marker-assisted selection. Not much progress has been made in breeding for resistance in brassicas against aphids primarily due to non-availability of resistant source within the crossable germplasm as well as lack of knowledge on its trait genetics. Though some success has been achieved to introgress the gene of interest to a desirable agronomic background, it has complex and elaborate breeding requirements. An alternate strategy to conventional breeding is the use of insect-resistant transgenes through genetic engineering, but this strategy has its own associated issues. Thus, the development of aphid-resistant cultivars requires more research on aphid-plant interactions to identify either an effective aphid resistance gene or a phenomenon that can lead to a new mechanism of resistance.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Introduction
Crop brassicas belong to the family Brassicaceae. It is a major angiosperm family that includes nearly 375 genera and 3200 species (LeCoz and Ducombs 2006). Members of this family provide major sources of oilseeds, vegetables and condiments. Canola (Brassica napus); Indian mustard (B. juncea); B. rapa ssp. oleifera, viz., toria and brown sarson; and Abyssinian mustard (B. carinata) account for almost 13% of the vegetable oil supplies of the world. Besides its economic importance, Brassicaceae are of special significance in the study of insect-plant interactions as all members produce glucosinolates, which have a great influence on such relationships. Further, the genome of the closely related Arabidopsis thaliana has been sequenced, which can provide ready access to genetic and genomic resources (Hegedus and Erlandson 2012). A. thaliana is ideal as a model system for the study of insect-plant interactions at genetic and molecular level (Mitchell-Olds 2001). This chapter focuses on various aspects of breeding for resistance to mustard aphid in rapeseed-mustard. We also discuss various aspects of aphid biology, host-pest interactions and factors associated with resistance responses of the host.
6.2 The Aphid Complex of Brassicas
Aphids are global pests. Despite forming a small insect group, they inflict serious damage to agricultural crops (Remaudière and Remaudière 1997; Dedryver et al. 2010). They belong to family Aphididae and comprise approximately 5000 species (Smith and Chuang 2014), of which nearly 100 are very damaging for crop plants (Blackman and Eastop 2000, 2007). The main aphids infesting brassica crops are cabbage aphid [Brevicoryne brassicae (L.)], turnip/mustard aphid [Lipaphis erysimi (Kaltenbach)/Lipaphis pseudobrassicae (Davis)], shallot aphid [Myzus ascalonicus Doncaster], peach-potato aphid [Myzus persicae (Sulzer)], potato aphid [Macrosiphum euphorbiae (Thomas)], corn root aphid [Aphis maidiradicis Forbes] and root-feeding aphid species, namely, cabbage root aphid/poplar petiole gall aphid [Pemphigus populitransversus Riley] and bean root aphid [Smynthurodes betae Westwood] (Blackman and Eastop 2000). B. brassicae, a native to Europe and worldwide in distribution, is a major pest on vegetable brassicas in most European countries with strong yield reducing impacts. It is a brassica specialist insect that feeds on phloem sap of its host plants (Cole 1997). Though a primary pest of vegetable brassicas, it also infests other species in genus Brassica (Cole 1994a, b, 1997; Kift et al. 2000). L. erysimi is a native to eastern Asia (Blackman and Eastop 2000). It is the most serious pest of oilseed brassica, especially in India and other subtropical regions of the world. It may cause 10–90% productivity losses, depending upon the agroclimatic conditions, intensity of population development and crop growth stage (Singh and Sachan 1994; Ahuja et al. 2009). L. erysimi is also a vector of ten non-persistent plant viruses like cabbage black ring spot and mosaic diseases of cauliflower, radish and turnip (Blackman and Eastop 1984; Rana 2005). It is a brassica specialist and can develop only on brassicaceous plants. Generally, B. rapa and B. juncea are better hosts than other Brassica species (Rana 2005).
Peach-potato aphid, Myzus persicae, is a generalist pest with a host range of more than 400 plant species (Quaglia et al. 1993). It is a major vector of more than 100 plant viruses including potato virus Y and potato leaf roll virus and various mosaic viruses, including western yellows (Ponsen 1972; Eskanderi et al. 1979; Bwye et al. 1997). It is cosmopolitan, polyphagous and an efficient vector of plant viruses. It possesses wide genetic variation for colour, life cycle, host plant relationships and mechanisms of insecticide resistance. Although many consider it to have originated from China, the native place of its primary host Prunus persica, others believe it to be a native of Europe (Blackman and Eastop 2007).
6.3 Aphid Biology
Aphids are the specialized phloem sap feeders. Their ability to rapidly exploit the ephemeral habitats makes them serious pests. High reproductive potential and dispersal capacities add to their wide adaptability (Dedryver et al. 2010). Aphids exhibit parthenogenetic viviparity – a process that limits the need for males to fertilize females and obviate egg stage from the life cycle. Thus, aphids reproduce clonally and give birth to young ones. Embryonic development of an aphid begins before its mother’s birth leading to telescoping of generations. These attributes permit aphids to efficiently exploit the periods of rapid plant growth, conserve energy and allow rapid generation turnover. Nymphs of certain aphid species can reach maturity in as little as 5 days (Goggin 2007). Parthenogenesis sets them apart from other Hemiptera and has a great influence on their biology. Many species of aphids also exhibit alternation of generations. Evolution of alternating hermaphrodite generations with a series of parthenogenetic, all-female generations dates back to Triassic period (Blackman and Eastop 2007). Coupled with viviparity, this reduces the development period and permits rapid multiplication of aphids. Further, to conserve energy for maximizing their reproduction and survival, aphid colonies exhibit wing dimorphism to produce highly fecund wingless (apterae) morphs or less prolific winged (alate) progeny that can disperse to new host plants depending on the resource availability. All these strategies contribute to aphids’ success and their abundance in temperate zones. An enormous propagation rate precipitates abnormally high population under favourable conditions (Goggin 2007).
6.3.1 Aphid Life Cycles
Most of the aphid species display relatively complicated life cycles, and each of these life cycles has morphs which specialize in reproduction, dispersal and survival under adverse conditions. Based on how aphids utilize their host plants, life cycle can be of two types: heteroecious or host alternating and monoecious/autoecious or non-host alternating. Heteroecious aphids live on one plant species (primary host) in winter and migrate to another taxonomically unrelated plant species (secondary host) in summer and again migrate to primary host in autumn. On the primary host plant, eggs are laid by females after mating with males. However, on the secondary host plant, they reproduce parthenogenetically. Aphids that interrupt parthenogenetic reproduction with sexual reproduction are termed as holocyclic. In contrast to host-alternating aphids, non-host-alternating aphids remain either on the same or closely related host species throughout the year. They complete both sexual and parthenogenetic life cycle on the same host species. There are also species which do not produce eggs and are known as anholocyclic. Some species can live both holocyclic and anholocyclic lives, simultaneously across wide geographies (Bhatia et al. 2011). However, monoecy and heteroecy can coexist rarely (Williams and Dixon 2007). The presence of both sexual and asexual life cycle ensures that aphids take advantage of both parthenogenesis and genetic recombination that help them to evolve.
Lipaphis erysimi is a holocyclic species with a chromosome number of 2n = 10 (Blackman and Eastop 2000). Although it produces parthenogenetically in warmer climates, a holocyclic reproduction has been reported in western Honshu, Japan, on cruciferous crops (B. rapa, Raphanus sativus) (Kawada and Murai 1979). A chromosome number of 2n = 8 and differing in karyotype from holocyclic populations have been reported from Northern Europe. Most anholocyclic parthenogenetic populations have 2n = 9, probably derived from eight chromosomes through dissociation of one autosome to produce a small, unpaired element. Though sexual morphs have been reported from North India, populations were mostly anholocyclic (Blackman and Eastop 2007).
Brevicoryne brassicae is a monoecious species that exhibits holocyclic life cycle with parthenogenetic reproduction in warmer climates as well as during warmer periods of temperate climates. However, with the fall in temperature during autumn, males are also produced (Blackman and Eastop 1984), which mate with the females to produce eggs for overwintering. As per Hines and Hutchison (2013), about 15 overlapping generations are passed in a crop season in the United States.
Myzus persicae exhibits holocyclic life cycle, and it overwinters as egg stage on its primary host (peach and related trees). In the subsequent spring or summer season, fundatrix/fundress (the winged stem mother) returns as alate emigrants to secondary host plants and multiplies to apterous and alate viviparae (Moran 1992; Bhatia et al. 2011). The wingless female then gives birth to young ones by parthenogenesis and multiplies at a very fast rate. This results in large aphid populations on different crop plants. When the temperature starts falling late in the season, some of the apterous viviparae turn into apterous oviparae and alate viviparae into alate males. These males and females start sexual reproduction and lay eggs on the primary host plant (Stern 1995). At the end of winters, females (stem mothers) hatch from the eggs the next spring season and start reproducing parthenogenetically (Bhatia et al. 2011).
6.3.2 Aphid-Host Plant Interactions
Aphids are specialized phloem sap feeders which insert their needle-like stylets in the plant tissue avoiding/counteracting the different plant defences. They withdraw large quantities of phloem sap while keeping the phloem cells alive. In contrast to the insects with biting and chewing mouthparts which tear the host tissues, aphids penetrate their stylets between epidermal and parenchymal cells to finally reach sieve tubes with slight physical damage to the plants, which is hardly perceived by the host plant (Bhatia et al. 2011). The long and flexible stylets move through intercellular spaces in the apoplasm of the cell wall (Giordanengo et al. 2010), although stylets also make intracellular punctures to probe the internal chemistry of a cell (Zust and Agrawal 2016). The high pressure within sieve tubes helps in passive feeding (Bhatia et al. 2011). During the stylet penetration and feeding, aphids produce two types of saliva. The first type is dense and proteinaceous (including phenoloxidases, peroxidases, pectinases, β-glucosidases) that forms an intercellular tunnelled path around the stylet in the form of sheath (Felton and Eichenseer 1999; Zust and Agrawal 2016). In addition to proteins, this gelling saliva also contains phospholipids and conjugated carbohydrates (Urbanska et al. 1998; Miles 1999; Cherqui and Tjallingii 2000; Sharma et al. 2014). This stylet sheath forms a physical barrier and protects the feeding site from plant’s immune response (Will et al. 2012, 2013). When the stylets encounter active flow of phloem sap, the feeding aphid releases digestive enzymes in the vascular tissue in the form of second type of ‘watery’ saliva. The injection of watery saliva (E1) prevents the coagulation of proteins in plant sieve tubes, and during feeding the watery (E2) saliva gets mixed with the ingested sap which prevents clogging of proteins inside the capillary food canal in the insect stylets (Bhatia et al. 2011; Sharma et al. 2014; Zust and Agrawal 2016). Though the actual biochemical mode of action that inhibits protein coagulation is unknown, the calcium-binding proteins of aphid saliva are reported to interact with the calcium of plant tissues. This results in suppression of calcium-dependent occlusion of sieve tubes and subsequent delayed plant response (Will et al. 2007, 2009, 2013). This mechanism of feeding is more specialized and precise, which helps the aphid to avoid different allelochemicals and indigestible compounds found in other plant tissues (Schoonhoven et al. 2007). In addition to this, aphid saliva also contains non-enzymatic reducing compounds, which in the presence of oxidizing enzymes inactivate different defence-related compounds produced by plants in response to the insect attack (Miles 1999).
There are commonalities of events during initial plant reaction to insect feeding or pathogen infection. These include protein phosphorylation, calcium influx, membrane depolarization and release of reactive oxygen species (ROS), such as hydrogen peroxide (Garcia-Brugger et al. 2006). These lead to activation of phytohormone-dependent pathways. In response to infestation/infection, different phytohormone-dependent pathways are activated. Ethylene (ET) and jasmonate (JA) pathways are activated by different necrotrophic pathogens (Thomma et al. 2001) and grazing insects (Maffei et al. 2007), while salicylate (SA)-dependent responses are induced by biotrophic pathogens (Thomma et al. 2001). These responses lead to the production of various defence-related proteins and secondary metabolites with antixenotic or antibiotic properties. In the event of infestation by aphids, a SA-dependent response was seemingly activated. In contrast, JA-dependent genes were repressed (Zhu-Salzman et al. 2004; Thompson and Goggin 2006; Gao et al. 2007; Walling 2008). All these responses lead to the manipulation of the plant metabolism to ensure compatible aphid-plant interactions.
6.3.3 Aphid Endosymbionts
The phloem sap is a highly unbalanced diet composed principally of sugars and amino acids with high C:N content. The most of the amino acids are present at very low concentrations. Despite their nutritionally poor diet, aphids exhibit high growth and reproduction rates. Since aphids directly feed on the sugars and amino acids, their assimilation efficiency is very high. In addition, essential amino acids required by their growth and development are synthesized by symbiotic bacteria present in their body. These include primary (obligate) symbionts and secondary (facultative) symbionts. Buchnera aphidicola (gamma-3 proteobacteria, Escherichia coli, is also a member of this group) is the most common vertically transmitted primary symbiont present in most aphid species (Munson et al. 1991; Oliver et al. 2010). Some species of aphids also bear other bacteria, i.e. ‘secondary symbionts’. These include several species of gamma-proteobacteria such as Serratia symbiotica, Regiella insecticola and Hamiltonella defensa (Chen et al. 1996; Chen and Purcell 1997; Fukatsu et al. 2000, 2001; Darby et al. 2001; Sandstrom et al. 2001; Haynes et al. 2003; Russell et al. 2003; Moran et al. 2005; Oliver et al. 2010). B. aphidicola is a coccoid hosted in the cytoplasm of specialized cells called mycetocytes/bacteriocytes in the haemocoel of insect. These endosymbionts upgrade the aphid diet by converting non-essential amino acids to essential amino acids. The evolution of symbiotic relationship with endosymbionts has enabled aphids to exploit new ecological niches, i.e. to feed on the plant phloem sap which is otherwise the nutritionally poor diet.
6.4 Plant Defence Responses Against Insects
Brassicas possess an array of defence mechanisms against different biotic stressors including insect herbivores. These include surface waxes, trichomes, plant secondary metabolites and different volatiles, which provide varying degree of protection against insects feeding on them. Such defence mechanisms can be constitutive or inducible and direct or indirect defences. The constitutive defences comprise physical and chemical barriers that exist before insect attack (preformed/innate defences). These may be the ancient defences involving different plant receptors that recognize microbial cell surface molecules, signal transduction pathways that induce transcription of defence-associated genes and antimicrobial effectors, cationic peptides and proteins (Boman 1995; Borregaard et al. 2000; Thomma et al. 2002 as cited from Ahuja et al. 2009). In contrast, inducible defences are induced following invasion of an insect herbivore. This kind of defence is particularly important when the defence is bioenergetically expensive and insect attack is frequent and unpredictable (Haukioja 1999). The defences that show their effect on the herbivore through synthesis of toxins are called direct defences, while the defences that affect herbivores through the attraction of natural enemies of insects are called indirect defences (Dicke 1999). Brassica plants release different volatile compounds to attract natural enemies of insects that feed on them. This release of volatile organic compounds is construed as a ‘cry or call’ for help by the plant from herbivore predators. The different defence components of brassica plants are discussed in the following subsections.
6.4.1 Biophysical Defences
Many morphological and anatomical characters may influence the suitability of a plant as host to the insect (Southwood 1986). These characters may include epicuticular wax, trichomes, depth of vascular bundles, etc. The epicuticular wax is the first site of interaction between insect and its host plant, and hence, its chemical composition is critical for an insect to feed, probe or oviposit on a plant. The waxes are complex mixtures of very-long-chain lipids substituted with primary alcohols, aldehydes, fatty acids and alkyl esters, all of which primarily occur with even-numbered chain lengths and hydrocarbons, secondary alcohols and ketones with predominance of odd chain lengths (Walton 1990). Waxiness has been found to hinder L. erysimi from reaching the undersurface of leaves, where it normally feeds during the vegetative plant stage (Åhman 1990). However, Lamb et al. (1993) reported that elevation of leaf wax did not improve the resistance of B. napus or B. oleracea (kale and collard) to L. erysimi. The neonate larvae of diamondback moth, Plutella xylostella L., have been shown to spend more time walking at a faster pace on waxy line of cabbage compared to that on non-waxy one (Eigenbrode et al. 1991). The young larvae of mustard beetle, Phaedon cochleariae (Fab.), find it difficult to climb the heavily waxed culm of cabbage on waxy cultivars and failed to reach their feeding site, while they easily walked on the non-waxy cultivars (Stork 1980). Although waxy trait is responsible for resistance to insect pests, glossiness is not a preferred trait in vegetables. Increased resistance to P. xylostella was observed in B. oleracea and B. rapa genotypes having glossy leaves (Ulmer et al. 2002). A significant increase in the feeding by flea beetle, Phyllotreta cruciferae (Goeze), was observed after removal of epicuticular wax from leaves of B. napus and B. oleracea particularly from the area where wax was removed (Bodnaryk 1992) and most difference in feeding preference was explained by the presence of leaf wax. Reifenrath et al. (2005) observed an increase in P. cochleariae activity after removal of leaf wax, suggesting that wax occludes stimulatory signals such as glucosinolates, and they suggested that the resistance was primarily antixenosis. The importance of waxes on leaf surface has received increased attention in the recent years due to their association with polar compounds like glucosinolates, the key host recognition signals for specialist insects (Badenes-Pérez et al. 2010; Städler and Reifenrath 2009). Badenes-Pérez et al. (2010) reported the presence of glucosinolates on leaf surface of three Barbarea species but not on the surface of test B. napus genotype. The leaf surface wax has been reported to affect even the third trophic level. The aphids’ parasitoid host recognition behaviour is influenced by aphid cuticular waxes which in turn are related to the plant surface waxes (Muratori et al. 2006).
Trichomes may also influence leaf herbivory by insects. The trichomes are small, sometimes branched, hair-like structures that are produced from cells of aerial epidermis, produced by most plant species (Werker 2000). Glandular trichomes produce secondary metabolites (e.g. flavonoids, alkaloids, terpenoids) which can either repel or trap insects or can be poisonous (Duffey 1986). The trichome producing morphotype of Arabidopsis lyrata was reported to be less damaged by insect herbivores than the glabrous form (Loe et al. 2007). The non-glandular trichomes, unlike glandular trichomes, do not produce secondary metabolites but mainly function as structural defence against small herbivores by interfering with insect movement on the plant surface (Southwood 1986). The insects feeding on trichome-bearing plants show poor weight gain due to poor nutritive value of cellulose-rich trichomes resulting in increased mortality. B. nigra lines having high number of trichomes supported less growth of Pieris rapae (L.) and increased mortality of P. cruciferae (Traw and Dawson 2002). Agrawal (1999) reported an increase in trichome density after insect damage in Raphanus raphanistrum. Similarly, Traw (2002) reported an increase in the trichome density as well as glucosinolate level after feeding by P. rapae in black mustard. Trichome-bearing pods of Sinapis alba were reported to be resistant to flea beetle, while glabrous pods of cultivated Brassica species are readily attacked (Lamb 1980).
Expression of A. thaliana myb-like transcriptor factor, GLABRA3 (GL3) in B. napus, resulted in the production of a dense coat of trichomes on the adaxial leaf surface (Gruber et al. 2006), and P. xylostella larvae had difficulty in feeding on these lines and grew slower (Adamson et al. 2008). Despite their negative effects on herbivore insects, trichomes may have their effect at the third trophic level. For example, trichomes on the leaves of trichome-bearing line of Arabidopsis thaliana affected the movement of aphid predator, Episyrphus balteatus (De Geer), and resulted in reduced performance (Wietsma 2010). Further, trichomes play an important role in the acceptance of host plants for oviposition (Sadeghi 2002), and there was comparatively less oviposition on A. thaliana line having higher trichome density (Wietsma 2010).
Before reaching the sieve tubes for feeding, aphid stylets had to pass through different cell layers such as the epidermis, endodermis, cortex and pericycle. The plants with densely packed cell layers may pose hindrance to the stylets and, hence, may be less preferred (Henning 1966). Moderate resistance to aphids in B. carinata, B. alba and Eruca sativa has been attributed to this factor (Malik 1981). The depth of sieve tubes is an important factor in resistance of a plant to aphids. Aphids must have long stylets to feed on plant tissues with deeply localized vascular bundles (Gibson 1972). Further, such aphids will require more energy to probe deep into the plant tissue, while aphids with short stylets will starve and die (Berlinski 1965).
6.4.2 Biochemical Defences
6.4.2.1 Glucosinolates and Myrosinase-Glucosinolate System
Glucosinolates (GSLs) of brassica plants are a class of secondary metabolites. These amino acid-derived, secondary plant products containing β-D-thioglucose and sulphonated oxime moieties are found almost exclusively in the order Capparales (Halkier and Gershenzon 2006). They are a large group of naturally occurring, non-volatile, sulphur-containing, organic anionic compounds and are reported to be present in 16 plant families (Fahey et al. 2001). GSLs include approximately 140 naturally occurring thioglucosides that mainly differ in their R-group substitutes (Fenwick et al. 1983), and 30 of these are present in Brassica species (Bellostas et al. 2007). Although the glucosinolates may confer resistance to insects which feed on them, their breakdown products released after myrosinase hydrolysis can be more toxic. Myrosinase (thioglucoside glucohydrolase, EC 3.2.3.1) catalyses the cleavage of glucosinolates to produce an aglycone moiety (thiohydroxamate-O-sulfonate), glucose and sulphate. The aglycone moiety, being unstable, rearranges to form isothiocyanates (ITCs), thiocyanates, nitriles, amines, oxazolidine-thiones and epithionitriles depending upon the glucosinolate being hydrolysed and the reaction conditions (Rask et al. 2000; Sadasivam and Thayumanavan 2003). The concentration of glucosinolates varies widely depending upon different species, plant parts and agronomic and climatic conditions (Font et al. 2005; Tripathi and Mishra 2007). A drastic decline in the concentration of glucosinolates (mainly aliphatic ones) occurs in B. napus seeds during the first 7 days of imbibition, while de novo synthesis of indolyl glucosinolates and an aromatic glucosinolate (gluconasturtin) takes place concomitantly. Gluconasturtin is not initially present in the seed. During the subsequent growth period, some more glucosinolates also accumulate (Clossais-Besnard and Larher 1991). On the other hand, glucosinolates occur in low concentrations in the fully expanded leaves (Porter et al. 1991). With the start of the reproductive phase of plant, i.e. during flowering, there is a reduction in the concentration of glucosinolates in vegetative plant parts as well as in inflorescence, which otherwise has relatively large amounts of glucosinolates. In contrast to this, during maturation of seeds, glucosinolate synthesis occurs in siliques which are then transported to the seeds through pod shells (Rask et al. 2000). The levels of glucosinolates can also be influenced by environmental conditions. An increase in the concentration of glucosinolates occurs in brassica plants under drought conditions (Bouchereau et al. 1996; Jensen et al. 1996). However, there is no consistent relationship between glucosinolate concentration and water stress since increased levels of glucosinolates are also observed in plants grown under moist conditions compared to those grown in dry soil (Louda and Mole 1991). In intact plant tissues, glucosinolates and myrosinase are housed separately and individually where these are inactive thus preventing self-toxicity (Jones and Vogt 2001). This intracellular localization of myrosinase has been widely investigated. Lüthy and Matile (1984) propounded ‘the mustard oil bomb hypothesis’ for this organization. As per this hypothesis, glucosinolates are present in the myrosin grains (vacuoles) of myrosin cells, while the myrosinase is associated with the membranes in the cytoplasm. However, later studies proved that glucosinolates (Kelly et al. 1998) are present in vacuoles of different types of cells, while myrosinases are localized in the myrosin cells (Thangstad et al. 1991; Höglund et al. 1992; Kissen et al. 2009) scattered across the plant tissues. Myrosin cells carry myrosin grains (Bones et al. 1991; Kissen et al. 2009), forming a continuous reticular system or myrosin body (Andreasson et al. 2001; Ahuja et al. 2009). Tissue damage caused by insect feeding brings glucosinolates and myrosinase together, precipitating immediate release of glucosinolate-breakdown products (Bones and Rossiter 2006). Such defensive responses (or ‘mustard oil bomb’) play multiple roles in plant-insect interactions (Rask et al. 2000; Kissen et al. 2009). These defend the plants against the attacks by generalist feeders (Rask et al. 2000) but at the same time expose them to attack by specialist feeders (Renwick 2002; Bjorkman et al. 2011). Glucosinolates are feeding and oviposition stimulants for more than 25 insect species of the orders Coleoptera, Lepidoptera and Diptera (Hopkins et al. 2009). As a consequence of coevolution, insects like B. brassicae and L. erysimi (both crucifer specialists) can sequester glucosinolates from host plant to protect themselves from predators. These insects can synthesize their own thioglucosidase endogenously, which is spatially separated in the insect body from sequestered glucosinolates in their non-flight muscles. When an insect is crushed or fed upon by a predator, thioglucosidase hydrolyses the sequestered glucosinolates (glucosinolate concentration in haemolymph is normally 15–20 times more than those in the leaf tissue) to produce toxic products (Bridges et al. 2002; Rossiter et al. 2003). These products taste badly and also release volatiles to alarm other insects in the colony. In comparison, the generalist aphid, M. persicae, excretes glucosinolates in its honeydew (Hopkins et al. 2009). Another example of coevolution is the production of a glucosinolate sulfatase enzyme (GSS) by the diamondback moth, P. xylostella (specialist) (Ratzka et al. 2002), and desert locust, Schistocerca gregaria (Forskål) (generalist) (Falk and Gershenson 2007). GSS desulphonates glucosinolates to produce desulphoglucosinolates which are not amenable to hydrolysis by myrosinase. Thus, the production of toxic isothiocyanates is prevented. This enables the insects to feed on glucosinolate-rich plants (Ratzka et al. 2002; Falk and Gershenson 2007). In contrast, P. rapae is able to manipulate glucosinolate hydrolysis reaction in such a way that instead of toxic isothiocyanates, less toxic nitriles are formed (Wittstock et al. 2004). Glucosinolates are also known to stimulate larval feeding and oviposition by adults of the large white butterfly, Pieris brassicae (L.), and small white butterfly, P. rapae (Renwick et al. 1992; Smallegange et al. 2007). These also stimulate oviposition by P. xylostella (Renwick et al. 2006). Many insects such as B. brassicae (Nottingham et al. 1991) and P. xylostella (Renwick et al. 2006) carry receptor neurons that can detect isothiocyanates to find host location.
Buxdorf et al. (2013) experimented with Arabidopsis thaliana mutants having varying levels of glucosinolates and glucosinolate-breakdown products to study the effects of these phytochemicals on phytopathogenic fungi. It was observed that Alternaria brassicicola was more strongly affected by aliphatic glucosinolates and isothiocyanates as decomposition products. B. cinerea also induced glucosinolate accumulation at a level higher than that by A. brassicicola. For A. brassicicola, the type of glucosinolate-breakdown product was more important than the type of glucosinolate from which that product was derived. For example, the sensitivity of the Ler background and the sensitivity gained in Col-0 plants expressing epithiospecifier protein depended upon the type of breakdown products, both of which accumulate simple nitrile and epithionitriles, but not isothiocyanates. Correlations between identical compounds in different plant tissues permit (co-)regulation of their biosynthesis or emission. The glucosinolate content seemed positively correlated in leaves and other tissues indicating independent regulation of emission (Sotelo et al. 2014; Gupta et al. 2015). However, none of the leaf or flower volatiles was positively associated with gluconapin, glucobrassicanapin or the sum of all glucosinolates in either leaves or flowers. The lack of consistent positive correlations between VOCs and major defence compounds may indicate that plants avoid eavesdropping by specialist herbivores to locate their host plants. Negative correlations may indicate chemical trade-offs for synthesis of the secondary metabolites.
Although glucosinolates play a defensive role in plants against herbivorous insects, there have been concerns regarding increased insect susceptibility of canola cultivars with exceptionally low level of these compounds. Such concerns may be far-fetched since low glucosinolate levels in such cultivars are confined mainly to the seeds (Milford et al. 1989). Also, high and low glucosinolate cultivars did not differ in their susceptibility to pod midge (Dasineura brassicae) (Åhman 1982). Extensive studies in India with both B. napus and B. juncea canola have shown no reasons to believe that canola quality cultivars were more susceptible than their non-canola counterparts. In fact, the inheritance mechanism of glucosinolates in B. juncea seemed to be different in leaves and seeds. Major QTLs accounting for a large variation in seeds or leaves were not co-localized (Gupta et al. 2015). Though there are no supporting references, low glucosinolate plants may be less attractive to specialist insects for which these compounds serve as attractants and feeding stimuli (Gabrys and Tjallingii 2002; Mewis et al. 2002). This is supported by the work of Giamoustaris and Mithen (1995) who reported that increase in the content of glucosinolates in B. napus resulted in increased feeding damage by the specialist insects, flea beetles [Psylliodes chrysocephala (L.)] and greater incidence of small white butterfly (P. rapae), while the damage by generalist pests, i.e. pigeons and slugs, was reduced. Further, glucosinolate-rich flower tissues are preferred more by P. brassicae and sustain higher growth compared to leaf tissues (Smallegange et al. 2007) indicating the selective role of glucosinolates to elicit feeding in this specialist insect and the adaptation of the insect to use these compounds to its advantage.
6.4.2.2 Phytoalexins and Phytoanticipins
Phytoalexins are antimicrobial secondary metabolites produced de novo by plants in response to biotic or abiotic stresses (Bailey and Mansfield 1982; Pedras and Yaya 2010), while phytoanticipins are constitutive defences already present in the plant irrespective of the stress. Plant secondary metabolites can be phytoalexins in one plant species and phytoanticipins in the other.
Polyphenolics – phenolic acids, flavonoids and lignans, terpenoids, phytosterols and alkaloids – have been associated with plant defences. Phenolics, especially the condensed tannins, are feeding deterrents to several pests on B. napus (Meisner and Mitchell 1984; Muir et al. 1999). These act by inactivating digestive enzymes (Nguz et al. 1998) or through antibiotic effects (Duffey and Stout 1996). A sinapic acid – precursor of sinapine – has been found to deter the oviposition by Delia radicum (L.) on cauliflower plants (Jones et al. 1988). Flavonoids show both stimulatory and deterrent effects on insects feeding on brassica plants. Quercetin and kaempferol from Armoracia rusticana stimulated feeding by Phyllotreta armoraciae (Koch) (Nielsen et al. 1979) and P. xylostella (van Loon et al. 2002). In contrast, isorhamnetin-3-sophoroside-7-glucoside and kaempferol 3,7-diglucoside found in B. napus were deterrent to Mamestra configurata (Walker), at levels higher than those found in vegetative tissues (Onyilagha et al. 2004). The phytosterols, strophanthidin and strophantidol, found in Cheiranthus and Erysimum species, exhibited feeding deterrent action against flea beetle species, Phyllotreta undulata (Kutschera), Phyllotreta tetrastigma (Comolli) and P. cochleariae (Nielsen 1978). Camalexin-deficient A. thaliana mutants showed greater susceptibility to the cabbage aphid, B. brassicae (Kusnierczyk et al. 2008), suggesting the role of camalexin in insect resistance.
6.4.2.3 Volatile Compounds
Volatile compounds are associated with plant-insect communication, plant-pathogen communication and plant-plant communication (Baldwin et al. 2002). These volatiles can be monoterpenes, sesquiterpenes, indole or ‘green leafy volatiles’ (Tumlinson et al. 1999). The hydrolysis of glucosinolates leads to the production of volatile thiocyanates, isothiocyanates and nitriles. Cabbage seed weevils, Ceutorhynchus assimilis (Paykull), are attracted to 3-butenyl and 4-pentenyl isothiocyanate in B. napus, but not to 2-phenylethyl isothiocyanate (Bartlet et al. 1993). Similarly, cabbage root fly, Delia brassicae L., was attracted to 4-methylthio-3-butenyl isothiocyanate and 1-cyano-4-methylthio-3-butene produced after glucosinolate hydrolysis in Raphanus sativus (Ellis et al. 1980). Though different herbivore insects use these volatile compounds as cues to locate their hosts, these also serve as a means of indirect defence against the herbivores. Plants release volatiles following insect attack to attract natural enemies that keep a check on the herbivore insect population. Volatile z-jasmone not only repels L. erysimi but also attracts its parasitoids on brassica plants (Birkett et al. 2000). Blande et al. (2007) have reported the attraction of the aphid parasitoid, Diaeretiella rapae (M’Intosh) towards semiochemicals produced by turnip plants after feeding by L. erysimi (specialist) and M. persicae (generalist). Pope et al. (2008) studied the orientation response of cabbage aphid, B. brassicae, and its parasitioid, D. rapae, to alkenyl glucosinolate hydrolysis products. The electroantennogram responses indicated peripheral odour perception in D. rapae females to all the 3-butenylglucosinolate hydrolysis products.
6.4.2.4 Lectins
Lectins are found across a range of plant, microbial and animal tissues (Nachbar and Oppenheim 1980; Komath et al. 2006; Michiels et al. 2010; Vandenborre et al. 2011). These are the proteins which selectively bind with carbohydrate moieties of glycoproteins that are located on animal cell surface. Lectins incorporated in artificial diets have been shown to reduce performance of several insect pests (Murdock et al. 1990; Powell et al. 1993; Sauvion et al. 2004a; Vandenborre et al. 2011). Although the actual mechanism of insecticidal action is not clearly known, these are not adequately metabolized by digestive enzymes. These can be lethal due to their affinity to epithelial cells in the insect gut (Vasconcelos and Oliveira 2004). They can bind with gut proteins (e.g. glycosylated proteins) with high affinity (Macedo et al. 2004; Sauvion et al. 2004b). Since, lectins interact with mono- and oligosaccharides, the insecticidal activity may involve a specific carbohydrate-lectin interaction with glycoconjugates on the surface of digestive tract epithelial cells (Macedo et al. 2004), precipitating nausea, vomiting and diarrhoea. They may also cause membrane disruption of epithelial cell microvilli of insects fed upon diet containing lectins (Hart et al. 1988). Lectins show biological activity against a range of sap-sucking insects (Foissac et al. 2000; Powell 2001). Brassica fruticulosa – a wild relative of cultivated brassicas – appeared to possess resistance against the cabbage aphid, B. brassicae (Cole 1994a, b; Ellis and Farrell 1995; Ellis et al. 2000) as well as to L. erysimi (Kumar et al. 2011). A high concentration of lectins appeared responsible for the resistance. Feeding preference/choice tests have shown that L. erysimi had maximum feeding preference for B. rapa ssp. brown sarson cv. BSH 1. Least preference was reported for B. fruticulosa. The antixenosis to feeding in B. fruticulosa has been reported earlier for cabbage aphid, B. brassicae. Monitoring of feeding behaviour of this species by electrical penetration graph (EPG) revealed a significant reduction in the duration of passive phloem uptake on B. fruticulosa compared to the susceptible B. oleracea var. capitata cv. ‘Offenham Compacta’. There was either quick withdrawal of stylets from sieve elements or disrupted phloem uptake (Cole 1994a).
6.5 Host Resistance Against Aphids
Brassica plants are among the oldest cultivated plants known to humans with documented records dating back to ca. 1500 BC (Raymer 2002). The domestication of brassica plants resulted in the narrowing of their genetic base. The breeding efforts in brassica plants were largely focused on high yield and desirable quality traits such as low glucosinolates and erucic acid content, and little attention was paid by plant breeders to maintain adequate level of insect and/or disease resistance. All this led to loss of genes employed by their ancestors to ward off insect herbivores. It may be possible to remobilize lost defensive genes which requires the screening of a large brassica germplasm for resistance against insects which further requires a quick and efficient screening methodology.
6.5.1 Screening Methodology
Many attempts have been made to identify sources of resistance in primary gene pool of crop Brassica species (Brar and Sandhu 1978; Amjad and Peters 1992; Sekhon and Åhman 1992; Bhadoria et al. 1995; Saxena et al. 1995). The literature on the screening techniques for aphid resistance has been reviewed extensively by Bakhetia and Bindra (1977). Available screening techniques are summarized in this section.
6.5.1.1 Screening at Seedling Stage
Screening at seedling stage is always desirable since screening at adult plant stage is often laborious and time consuming. However, no serious attempt has been made to correlate seedling stage resistance with the adult plant resistance. Bakhetia and Bindra (1977) have tried to develop seedling screening methodology which is compatible with adult plant evaluation which is based on the seedling mortality at a defined aphid population level. Population levels of 11, 20, 20 and 30 wingless aphids and 1 ml and 3 ml aphids (1 ml = about 600 nymphs + wingless adults) per plant appeared optimal for resistance screening at cotyledonary, 2-leaf, 4-leaf, 6-leaf, flower bud initiation and flowering stages, respectively (Sekhon and Åhman 1992). The results obtained at all the test stages were comparable when screening was conducted under optimum level of aphid population per plant. The effect on the survival and fecundity was also similar at all the stages studied. Despite its advantages, this screening technique is not widely used for brassica germplasm screening against aphids.
6.5.1.2 Screening at Adult Plant Stage
Adult plant screening is the most widely used method for screening against aphids. Though it is laborious and time consuming, it reflects the resistance shown by plants under actual field conditions. It is based on the different injury symptoms manifested upon aphid feeding such as yellowing, curling, crinkling of leaves, drying of flower buds and flowers and shrivelling of developing pods. Different workers have adopted different grading systems, but the one published by Bakhetia and Sandhu (1973) is generally adopted for screening at adult plant stage. A major limitation of this method is the failure to account for different phenologies of the test genotypes. Late flowering genotypes are sometimes misclassified as resistant as flowering initiations in late genotypes may coincide with season end high temperatures, which are invariably less than congenial for aphid infestation.
Different injury grades are given to the test entries on the basis of degree of insect damage.
Aphid infestation index (AII) | Description |
|---|---|
0 | Free from aphid infestation. Even if a single wingless aphid is present, the plant is considered infested. Plants showing excellent growth |
1 | Normal growth, no curling or yellowing of the leaves, except only a few aphids along with little or no symptoms of injury. Good flowering or pod setting on almost all the branches |
2 | Average growth, curling and yellowing of a few leaves. Average flowering and pod setting on all the branches |
3 | Growth below average, curling and yellowing of the leaves on some branches. Plants showing some stunting, poor flowering and little pod setting |
4 | Very poor growth, heavy curling and the yellowing of leaves, stunting of plants, little or no flowering and only a few pods forming. Heavy aphid colonies on plants |
5 | Heavy stunting of plants, curling, crinkling and yellowing of almost all the leaves. No flowering and pod formation. Plants full of aphids |
A specific injury grade is given to every observed plant, and the aphid infestation index (AII) is worked out by multiplying the number of plants falling under each grade with the respective grade number. The AII is calculated at pre-flowering, flowering and pod formation stages as
where a, b, c, d, e and f are the number of plants falling under each injury grade.
The different test entries are classified into different resistance categories based on the AII as
Aphid infestation index (AII) | Reaction |
|---|---|
0.00–1.50 | Resistant |
1.51–2.50 | Moderately resistant |
2.51–3.50 | Susceptible |
> 3.50 | Highly susceptible |
6.5.1.3 Other Screening Methods
Only limited attempts have been made to develop a screening technique based on the biology of mustard aphid, despite its significance in identifying sources of resistance. According to Bakhetia and Bindra (1977), it is possible to develop such a criterion for screening since nymphal survival, fecundity, longevity and reproduction are similar at all the plant growth stages. Singh et al. (1965) and Malik (1981) have also reported fecundity to be inversely related to resistance. Aphid population at a particular stage and an increase in the population during a given time interval can also be used in germplasm screening (Bakhetia and Sekhon 1989). More recently, Kloth et al. (2015) have demonstrated the use of automated video tracking for phenotyping of plants for resistance to aphids. Though this method can be used to screen a large number of accessions at a time, it has the limitation that it uses the leaf discs instead of intact plants and, hence, does not reflect the actual resistance exhibited by plants. The resistance effect was partially lost in the leaf discs. However, this limitation can be overcome by the use of electrical penetration graphs (EPG) (Tjallingii 1988; Trebicki et al. 2012) which uses the intact leaf instead of leaf disc, but this technique has its own high equipment cost limitation.
6.5.2 Breeding for Aphid Resistance
Three different mechanisms are responsible for imparting insect resistance to plants: antixenosis, antibiosis and tolerance. Antixenosis is rarely effective under no-choice conditions since insects can learn to feed on the less preferred plant. In contrast, antibiosis puts a selection pressure on the insects, and there is always a risk of development of insect biotypes, a danger not applicable to tolerance. Tolerance imparts least pressure on the insect to adapt. A sustainable resistance results from amalgamation of all three mechanisms (Smith 1989).
Different breeding methods have been used to develop resistant cultivars. These include intervarietal hybridization, induced mutagenesis or autotetraploidy. B. napus strains and colchicine-induced tetraploid toria (B. rapa) appeared more resistant to mustard aphid in contrast to diploids (Rajan 1961; Singh et al. 1965; Jarvis 1970; Gill and Bakhetia 1985; Kalra et al. 1987), and the resistance was attributed to be due to antibiosis; however, these were cytogenetically unstable. Many workers have also attempted to artificially synthesize alloploids of B. napus (Prakash and Raut 1983) and B. rapa x Eruca sativa (Agnihotri et al. 1990 as cited from Sekhon and Åhman 1992), but these were not resistant to the aphids.
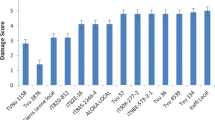
In the past, Lammerink (1968) attempted to develop cabbage aphid-resistant variety of rape after selection in the F3 generation of the cross (Broad Leaf Essex rape x Colder Swede) x giant rape. He also attempted recurrent selection in the crosses involving purple top white Globe and Sjodin turnip for breeding mustard aphid-resistant variety. Recently Kumar et al. (2011) reported wild B. fruticulosa (Plate 6.1) to be resistant to mustard aphid and described attempts at the introgression of resistance gene(s) from B. fruticulosa to B. juncea. B. fruticulosa have been previously reported to possess resistance against the cabbage aphid, B. brassicae (Cole 1994a, b, Ellis and Farrel 1995, Ellis et al. 2000). Study of feeding behaviour of B. brassicae electronically by electrical penetration graph (EPG) showed a large reduction in the duration of passive phloem uptake from B. fruticulosa compared to B. oleracea var. capitata cv. ‘Offenham Compacta’. There was either quick withdrawal of stylets from sieve elements or disrupted phloem uptake (Cole 1994a). Ellis and Farrel (1995) concluded that resistance of B. fruticulosa was due to high levels of both antixenosis and antibiosis. The resistance in B. fruticulosa due to antibiosis against D. radicum has also been reported by Jenson et al. (2002). Rorippa indica is another wild crucifer which is resistant to mustard aphid, and the genes conferring resistance have been recently identified by Bandopadhyay et al. (2013). Sarkar et al. (2016) have cloned, purified and characterized a novel R. indica defensin (RiD) which is toxic to L. erysimi. This aphid resistance trait can also be successfully introgressed to the cultivated backgrounds as demonstrated by somatic hybrids and their backcross progenies (Mandal 2003; Dutta 2007).
In addition to this, different workers have attempted to induce mutations in B. juncea for aphid resistance through chemical (Srinivasachar and Verma 1971) and physical mutagens (Srinivasachar and Malik 1972; Labana 1976), but all these efforts did not yield any result.
6.5.3 Genetic Engineering for Aphid Resistance
An alternative strategy to conventional breeding is the transgenic technology. For phloem-feeding insects, the different strategies can be employed such as expression of protease inhibitors, RNA interference (RNAi), antimicrobial peptides and repellents.
The Cauliflower mosaic virus (CaMV) 35S promoter is used to control transgene expression in many transgenic plants (Will and Vilcinskas 2013) which regulates the expression of a β-glucuronidase (GUS) reporter gene for the expression of dsRNA to protect the plants against the coleopterans (Baum et al. 2007) and aphids (Pitino et al. 2011).
The phloem-specific promoters can be used for phloem-specific expression of defence-related compounds against aphids. This would lead to more targeted expression of defence-related compounds with little/no exposure to the nontarget insects. This would also limit GM-associated bioenergetics investment of plant by avoiding the expression of defence-related compounds in plant tissues in the absence of pest attack. The SUC2 promoter that regulates the AtSUC2 sucrose-H+ symporter gene is restricted to the plant phloem which produces aphid toxic proteins. This green florescent protein is transferred through the sieve elements where aphids actually feed (Imlau et al. 1999). Protease inhibitors (PIs) can also be used to confer resistance in plants against different insects including aphids by genetic engineering. These small peptides/proteins reduce or inhibit the activity of proteases required for digestion of proteins. They have been shown to be toxic to a number of pests belonging to order Lepidoptera, Coleoptera and Orthoptera (Boulter et al. 1989). Their potential as insecticidal proteins has also been explored in aphids. PIs ingested with phloem sap may disrupt the digestion of proteins in aphid gut and hence can interfere with normal amino acid assimilation leading to the reduction in growth and subsequent pest damage. The expression of trypsin inhibitors and other PI-like chymotrypsin inhibitors has already been achieved in the phloem of transgenic plants (Dannenhoffer et al. 2001; Kehr 2006). The cysteine protease inhibitor of barley, HvCPI-6, inhibited the performance of M. persicae and Acyrthosiphon pisum (Harris) in artificial diet (Carrillo et al. 2011). Similarly, cysteine protease inhibitors, oryzacystatin I (OC I), inhibited the growth of M. persicae, A. gossypii and A. pisum (Rahbé et al. 2003). A reduction in adult weight, fecundity and biomass of M. persicae fed on transgenic B. napus expressing (OC I) was observed in comparison with those fed on control plants. PIs were also shown to defend white cabbage cultivars and A. thaliana against B. brassicae (Broekgaarden et al. 2008). PIs, thus, show detrimental effects against aphids, and their use in aphid management, therefore, appears to be an effective strategy for pest management.
Lectins are another class of proteins that have toxic effects on aphids and have the potential to be used for aphid control through genetic engineering. These are the proteins that selectively bind carbohydrates and the carbohydrate moieties of glycoproteins and can be poisonous. Lectins have been reported to show biological activity against a wide range of insects, especially the sap-sucking insects (Foissac et al. 2000; Powell 2001). Genes encoding wheat germ agglutinin from Triticum spp. (Kanrar et al. 2002), ACA from Allium cepa (Hossain et al. 2006), fusion ASAL from A. sativum and ACA from A. cepa (Hossain et al. 2006) have been introduced into Indian mustard, B. juncea, that provide protection against the mustard aphid, L. erysimi. These transgenic plants showed significant toxic effect against L. erysimi as evidenced by bioassays under controlled conditions.
Another method of aphid control through transgenic technology is the RNA interference (RNAi), which involves gene suppression at the level of RNA and involves post-translational RNA-mediated gene silencing. The transgenic plants that delivered dsRNA to aphids resulted in inhibition of Rack1 (located in the gut) and C002 (located in the salivary gland) proteins in peach-potato aphid, M. persicae (Pitino et al. 2011). The transformed plants of tobacco and A. thaliana resulted in reduction in fecundity of aphids with up to 60%t silencing in feeding aphids. Although salivary proteins (Mutti et al. 2006, 2008) and gut proteins (Shakesby et al. 2009) are the most promising RNAi targets for insects with piercing and sucking mouthparts such as aphids, the other targets may include transporters in the bacteriocyte plasma membrane required for nutrients’ transport between aphids and their endosymbiont, Buchnera aphidicola.
6.6 The Way Forward
Plant resistance to aphids has great potential in managing populations of these important insect pests. Earlier efforts by plant breeders have focused on host plant resistance as a single component of pest management, and hence, greater emphasis was laid on screening for virtual immunity to aphids. Such extremely high level of resistance can result from very high level of toxic (to aphids) substance in the plant, which has many disadvantages such as continuous selection pressure on the insect population to develop resistant biotypes, possible side effects on natural enemies as well as yield drag. Thus, for sustainable pest management, partial resistance to insects has the potential for the future. Such partially resistant cultivars can be integrated with other methods of pest management, which is the main feature of IPM. The effective IPM strategy against aphids infesting rapeseed-mustard could not be developed due to a lack of resistant variety. This is primarily because of lack of in-depth knowledge about the mechanism of resistance. Though transgenics conferring resistance to aphids have been developed, their efficacy in reducing aphid populations had been evaluated under controlled environments, and field testing of such transgenics is still awaited.
In addition to the inherently or transgenically expressed toxins in plants, other methods to reduce aphid populations on plants can also be developed. Since aphids utilize many secondary plant compounds especially volatiles in host plant recognition, plants can be genetically manipulated to alter their volatile profile, and limited success has been achieved under laboratory conditions (Beale et al. 2006; Schnee et al. 2006). It is a well-known fact that aphids reproduce at exceptionally high rate. A single mother aphid can produce 5.9 billion offspring in 6 weeks (Dixon 2005). Thus, disrupting the host recognition process of a mother aphid can significantly reduce the offspring population. However, this is a theoretical concept, and there is no report highlighting the validity of this strategy. Another potential area of research is the genetic manipulation of induced resistance in plants which is influenced by jasmonic acid (JA), salicylic acid (SA) and ethylene. The associated signalling pathways can be altered genetically to enhance the innate plant resistance level.
An effective and sustainable aphid management requires the adoption of integrated pest management (IPM) strategy. Since host plant resistance forms the core of any IPM programme, there is no effective IPM programme against aphids infesting brassica crops due to the lack of resistant crop cultivars. Rather than complete resistance to aphids, it is the partial resistance that has greater potential for the future, to maintain sustainability of pest management systems.
References
Adamson JB, Soroka J, Holowachuk J (2008) Feeding and oviposition of diamondback moth (Plutella xylostella) on modified “Hairy” canola, Honours Undergraduate Dissertation, University of Saskatchewan, Saskatoon
Agnihotri A, Gupta V, Lakshmikumaran MS, Shivanna KR, Prakash S, Jagannathan V (1990) Production of Eruca-Brassica hybrid by embryo rescue. Plant Breed 104:281–289
Agrawal AA (1999) Induced responses to herbivory in wild radish: effects on several herbivores and plant fitness. Ecology 80:1713–1723
Åhman I (1982) A comparison between high and low glucosinolate cultivars of summer oilseed rape (Brassica napus L.) with regard to their levels of infestation by the brassica pod midge (Dasineura brassicae Winn.). Z Angew Entomo l 94:103–109
Åhman I (1990) Plant surface characteristics and movements of two Brassica-feeding aphids, Lipaphis erysimi and Brevicoryne Brassicae. In: Symposia Biologica Hungaria No. 39. Publishing house of Hungarian Academy of Sciences, Budapest, pp 119–125
Ahuja I, Rohloff J, Bones AM (2009) Defence mechanisms of brassicaceae: implications for plant-insect interactions and potential for integrated pest management-A review. Agron Sustain Dev 30(2):311–348. doi:10.1051/agro/2009025
Amjad MD, Peters C (1992) Survival, development and reproduction of turnip aphids (Homoptera: Aphididae) on oilseeds Brassica. J Econ Entomol 85:2003–2007
Andreasson E, Jorgensen LB, Höglund AS, Rask L, Meijer J (2001) Different myrosinase and idioblast distribution in Arabidopsis and Brassica napus. Plant Physiol 127:1750–1763
Badenes-Pérez FR, Reichelt M, Gershenzon J, Heckel DG (2010) Phylloplane location of glucosinolates in Barbarea spp., and misleading assessment of host suitability by a specialist herbivore. New Phytol 189(2):549–556. doi:10.1111/j.1469-8137.2010.03486.x
Bailey JA, Mansfield JW (1982) Phytoalexins. Blackie and Son, Glasgow
Bakhetia DRC, Bindra OS (1977) Screening techniques for aphid resistance in Brassica crops. SABRAO J 9:91–107
Bakhetia DRC, Sandhu RS (1973) Differential response of Brassica species/varieties to the aphid, Lipaphis erysimi (Kalt.) infestation. J Res Punjab Agric Univ 10:272–279
Bakhetia DRC, Sekhon BS (1989) Insect pests and their management in rapeseed-mustard. J Oilseeds Res 6:269–299
Baldwin IT, Kessler A, Halitschke R (2002) Volatile signaling in plant-plant-herbivore interactions: what is real? Curr Opinion Plant Biol 5:351–354
Bandopadhyay L, Basu D, Sikdar SR (2013) Identification of genes involved in wild crucifer Rorippa indica resistance response on mustard aphid Lipaphis erysimi challenge. PLoS One 8(9), e73632. doi:10.1371/journal.pone.0073632
Bartlet E, Blight MM, Hick AJ, Williams IH (1993) The responses of the cabbage seed weevil (Ceutorhynchus assimilis) to the odour of oilseed rape (Brassica napus) and to some volatile isothiocyanates. Entomol Exp Appl 68:295–302
Baum JA, Bogaert T, Clinton W, Heck GR, Feldmann P, Ilagan O, Johnson S, Plaetinck G, Munyikwa T, Pleau M, Vaughn T, Roberts J (2007) Control of coleopteran insect pests through RNA interference. Nat Biotechnol 25:1322–1326
Beale MH, Birkett MA, Bruce TJA, Chamberlain K, Field LM, Huttly AK, Martin JL, Parker R, Phillips AL, Pickett JA, Prosser IM, Shewry PR, Smart LE, Wadhams LJ, Woodcock CM, Zhang Y (2006) Aphid alarm pheromone produced by transgenic plants affects aphid and parasitoid behavior. P Natl Acad Sci USA 103:10509–10513
Bellostas N, Sorensen AD, Sorensen JC, Sorensen H, Sorensen MD, Gupta SK, Kader JC (2007) Genetic variation and metabolism of glucosinolates. Adv Bot Res 45:369–415
Berlinski K (1965) Studies on food intake and the effects of food plants on the beet aphid – Aphis fabae. Pol Pismo Entomol 34(1–2):163–168
Bhadoria NS, Jakhmola SS, Dhamdhere SV (1995) Relative susceptibility of mustard cultivars to Lipaphis erysimi in North West Madhya Pradesh (India). J Entomol Res 19:143–146
Bhatia V, Uniyal PL, Bhattacharya R (2011) Aphid resistance in Brassica crops: challenges, biotechnological progress and emerging possibilities. Biotech Adv 29:879–888
Birkett MA, Campbell CAM, Chamberlain K, Guerrieri E, Hick AJ, Martin JL, Matthes M, Napier JA, Pettersson J, Pickett JA, Poppy GM, Pow EM, Pye BJ, Smart LE, Wadhams GH, Wadhams LJ, Woodcock CM (2000) New roles for cis-jasmone as an insect semiochemical and in plant defense. P Natl Acad Sci USA 97:9329–9334
Bjorkman M, Klingen I, Birch ANE, Bones AM, Bruce TJA, Johansen TJ, Meadow R, Molmann J, Seljasen R, Smart LE, Stewart D (2011) Phytochemicals of Brassicaceae in plant protection and human health – influences of climate, environment and agronomic practice. Phytochemistry 72:538–556
Blackman RL, Eastop VF (1984) Aphids on the World’s crops. Wiley, Chichester
Blackman RL, Eastop VF (2000) Aphids on the World’s crops: an identification and information guide, 2nd edn. Wiley, Chichester
Blackman RL, Eastop VF (2007) Taxonomic issues. In: van Emden HF, Harrington R (eds) Aphids as crop pests. CABI, Wallingford, pp 1–29
Blande J, Pickett J, Poppy G (2007) A comparison of semiochemically mediated interactions involving specialist and generalist Brassica feeding aphids and the braconid parasitoid Diaeretiella rapae. J Chem Ecol 33:767–779
Bodnaryk RP (1992) Leaf epicuticular wax as an antixenotic factor in Brassicaceae that affects the rate and pattern of feeding of flea beetles Phyllotreta cruciferae (Goeze). Can J Plant Sci 72:1295–1303
Boman HG (1995) Peptide antibiotics and their role in innate immunity. Annu Rev Immunol 13:61–92
Bones AM, Rossiter JT (2006) The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry 67:1053–1067
Bones AM, Thangstad OP, Haugen O, Espevik T (1991) Fate of myrosin cells - characterization of monoclonal antibodies against myrosinase. J Exp Bot 42:1541–1549
Borregaard N, Elsbach P, Ganz T, Garred P, Svejgaard A (2000) Innate immunity: from plants to humans. Immunol Today 21:68–70
Bouchereau A, Clossais-Besnard N, Bensaoud A, Leport L, Renard M (1996) Water stress effects on rapeseed quality. Eur J Agron 5:19–30
Boulter D, Gatehouse AMR, Hilder V (1989) Use of cowpea trypsin inhibitor (CpTI) to protect plants against insect predation. Biotechnol Adv 7(4):489–497
Brar KS, Sandhu GS (1978) Comparative resistance of different Brassica species/varieties to the mustard aphid, Lipaphis erysimi (Kalt.) under natural and artificial conditions. Indian J Agric Res 12:198–200
Bridges M, Jones AME, Bones AM, Hodgson C, Cole R, Bartlet E, Wallsgrove R, Karapapa VK, Watts N, Rossiter JT (2002) Spatial organization of the glucosinolate-myrosinase system in Brassica specialist aphids is similar to that of the host plant. Proc Royal Soc London B 269:187–191
Broekgaarden C, Poelman EH, Steenhuis G, Voorrips RE, Dicke M, Vosman B (2008) Responses of Brassica oleracea cultivars to infestation by the aphid Brevicoryne brassicae: an ecological and molecular approach. Plant Cell Environ 31:1592–1605
Buxdorf K, Yaffe H, Barda O, Levy M (2013) The effects of glucosinolates and their breakdown products on necrotrophic fungi. PLoS One. doi:10.1371/journal.pone.0070771
Bwye AM, Proudlove W, Berlandier FA, Jones RAC (1997) Effects of applying insecticides to control aphid vectors and cucumber mosaic virus in narrow leafed lupins (Lupinus angustifolius). Aust J Exp Agric 37:93–102
Carrillo L, Martinez M, Álvarez-Alfageme F, Castanera P, Smagghe G, Diaz I, Ortego F (2011) A barley cysteine-proteinase inhibitor reduces the performance of two aphid species in artificial diets and transgenic Arabidopsis plants. Transgenic Res 20:305–319
Chen DQ, Purcell AH (1997) Occurrence and transmission of facultative endosymbionts in aphids. Curr Microbiol 34:220–225
Chen DQ, Campbell BC, Purcell AH (1996) A new Rickettsia from a herbivorous insect, the pea aphid Acyrthosiphon pisum (Harris). Curr Microbiol 33:123–128
Cherqui A, Tjallingii WF (2000) Salivary proteins of aphids, a pilot study on identification, separation and immunolocalisation. J Insect Physiol 46:1177–1186
Clossais-Besnard N, Larher F (1991) Physiological role of glucosinolates in Brassica napus. Concentration and distribution pattern of glucosinolates among plant organs during a complete life cycle. J Sci Food Agr 56:25–38
Cole RA (1994a) Locating a resistance mechanism to the cabbage aphid in two wild Brassicas. Entomol Exp Appl 71:23–31
Cole RA (1994b) Isolation of a chitin binding lectin, with insecticidal activity in chemically defined synthetic diets, from two wild brassica species with resistance to cabbage aphid, Brevicoryne brassicae. Entomol Exp Appl 72:181–187
Cole RA (1997) Comparison of feeding behaviour of two Brassica pests Brevicoryne brassicae and Myzus persicae on wild and cultivated Brassica species. Entomol Exp Appl 85:135–143
Dannenhoffer JM, Suhr RC, Thompson GA (2001) Phloem-specific expression of the pumpkin fruit trypsin inhibitor. Planta 212:155–162
Darby AC, Birkle LM, Turner SL, Douglas AE (2001) An aphid-borne bacterium allied to the secondary symbionts of whitefly. FEMS Microbiol Ecol 36:43–50
Dedryver CA, Le Ralec A, Fabre F (2010) The conflicting relationships between aphids and men: a review of aphid damages and of their control strategies. C R Biol 333:539–553
Dicke M (1999) Are herbivore-induced plant volatiles reliable indicators of herbivore identity to foraging carnivorous arthropods. Entomol Exp Appl 91:131–142
Dixon AFG (2005) Insect herbivore-host dynamics: tree dwelling aphids. Cambridge University Press, Cambridge
Duffey SS (1986) Plant glandular trichomes: their partial role in defence against insects. In: Juniper B, Southwood SR (eds) Insects and the plant surface. Arnold, London, pp 151–172
Duffey SS, Stout MJ (1996) Antinutritive and toxic components of plant defense against insects. Arch Insect Biochem Physiol 32:3–37
Dutta S (2007) Development and characterization of aphid tolerant Brassica juncea chromosome addition lines from Roripobrassica somatic hybrid (Roripa indica + Brassica juncea) through plant breeding approach. Ph.D. Dissertation, Jadhavpur University, Kolkata
Eigenbrode SD, Espelie KE, Shelton AM (1991) Behaviour of neonate diamondback moth larvae [Plutella xylostella (L.)] on leaves and on extracted leaf waxes of resistant and susceptible cabbages. J Chem Ecol 7:169l–l704
Ellis PR, Farrell JA (1995) Resistance to cabbage aphid (Brevicoryne brassicae) in six Brassica accessions in New Zealand. NZ J Crop Hort Sci 23:25–29
Ellis PR, Cole RA, Crisp P, Hardman JA (1980) The relationship between cabbage root fly egg laying and volatile hydrolysis products of radish. Ann Appl Biol 95:283–289
Ellis PR, Kiff NB, Pink DAC, Jukes PL, Lynn J, Tatchell GM (2000) Variation in resistance to the cabbage aphid (Brevicoryne brassicae) between and within wild and cultivated Brassica species. Genet Resour Crop Evol 47:395–401
Eskanderi F, Sylvester ES, Richardson J (1979) Evidence for lack of propagation of potato leaf roll virus in Myzus persicae. Phytopathology 68:45–47
Fahey JW, Zalcmann AT, Talalay P (2001) The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 56:5–51
Falk KL, Gershenson J (2007) The desert locust, Schistocerca gregaria, detoxifies the glucosinolates of Schowia purpurea by desulfation. J Chem Ecol 33:1542–1555
Felton GW, Eichenseer H (1999) Herbivore saliva and its effects on plant defense against herbivores and pathogens. In: Agrawal AA, Tuzun S, Bent E (eds) Induced plant defenses against pathogens. APS Press, St. Paul, pp 19–36
Fenwick R, Heaney RK, Mullin WJ (1983) Glucosinolates and their breakdown products in food and food plants. CRC Crit Rev Food Sci Nutr 18:123–201
Foissac X, Nguyen TL, Christou P, Gatehouse AMR, Gatehouse JA (2000) Resistance to green leaf hopper (Nephotettix virescens) and brown plant hopper (Nilaparvata lugens) in transgenic rice expressing snowdrop lectin (Galanthus nivalis agglutinin; GNA). J Insect Physiol 46:573–583
Font R, Del Rio-Celestion M, Rosa E, Aires A, De Hardo-Bailon A (2005) Glucosinolate assessment in Brassica oleracea leaves by near-infrared spectroscopy. J Agric Sci 143:65–73
Fukatsu T, Nikoh N, Kawai R, Koga R (2000) The secondary endosymbiotic bacterium of the pea aphid Acyrthosiphon pisum (Insecta: Homoptera). Appl Environ Microbiol 66:2748–2758
Fukatsu T, Tsuchida T, Nikoh N, Koga R (2001) Spiroplasma symbiont of the pea aphid Acyrthosiphon pisum (Insecta: Homoptera). Appl Environ Microbiol 67:1284–1291
Gabrys B, Tjallingii WF (2002) The role of sinigrin in host plant recognition by aphids during initial plant penetration. Entomol Exp Appl 104:89–93
Gao LL, Anderson JP, Klingler JP, Nair RM, Edwards OR, Singh KB (2007) Involvement of the octadecanoid pathway in bluegreen aphid resistance in Medicago truncatula. Mol Plant-Microbe Interact 20:82–93
Garcia-Brugger A, Lamotte O, Vandelle E, Bourque S, Lecourieux D, PoinssotB WD, Pugin A (2006) Early signaling events induced by elicitors of plant defenses. Mol Plant-Microbe Interact 19:711–724
Giamoustaris A, Mithen R (1995) The effect of modifying the glucosinolate content of leaves of oilseed rape (Brassica napus ssp. oleifera) on its interaction with specialist and generalist pests. Ann Appl Biol 126:347–363
Gibson RW (1972) The distribution of aphids on potato leaves in relation to vein size. Entomol Exp Appl 15:213–223
Gill RS, Bakhetia DRC (1985) Resistance of some Brassica napus and B. campestris strains to Lipaphis erysimi (Kalt.). J Oilseeds Res 2:227–239
Giordanengo P, Brunissen L, Rusterucci C, Vincent C, van Bel A, Dinant S, Girousse C, Faucher M, Bonnemain JL (2010) Compatible plant-aphid interactions: how aphids manipulate plant responses. C R Biol 333:516–523
Goggin FL (2007) Plant-aphid interactions: molecular and ecological perspectives. Curr Opinion Plant Biol 10:399–408
Gruber MY, Wang S, Ethier S, Holowachuk J, Bonham-Smith PC, Soroka JJ, Lloyd A (2006) “HAIRY CANOLA”—Arabidopsis GL3 induces a dense covering of trichomes on Brassica napus seedlings. Plant Mol Biol 60:679–698
Gupta S, Sangha MK, Kaur G, Banga S, Gupta M, Kumar H, Banga SS (2015) QTL analysis for phytonutrient compounds and the oxidant molecule in mustard (Brassica juncea L.). Euphytica 201:345–356. doi:10.1007/s10681-014-1204-3
Halkier BA, Gershenzon J (2006) Biology and biochemistry of glucosinolates. Annu Rev Plant Biol 57:303–333
Hart CA, Batt RM, Saunders JR, Getty B (1988) Lectin-induced damage to the enterocyte brush border: an electron-microscopic study in rabbits. Scand J Gastroenterol 23:1153–1159
Haukioja E (1999) Ecology: bite the mother, fight the daughter. Nature 40:22–23
Haynes S, Darby AC, Daniell TJ, Webster G, van Veen FJF, Godfray HCJ, Prosser JI, Douglas AE (2003) Diversity of bacteria associated with natural aphid populations. Appl Environ Microbiol 69:7216–7223
Hegedus DD, Erlandson M (2012) Genetics and genomics of insect resistance in brassicaceae crops. In: Edwards D, Batley J, Parkin I, Kole C (eds) Genetics, genomics and breeding of oilseed brassicas. CRC Press, Taylor and Francis, New York, pp 319–372
Henning E (1966) Zür histologie und funktion von einstichen der schwarzen bohnenlaus (Aphis fabae Scop.) in Vicia faba pflanzen. J Insect Physiol 12:67–76
Hines RL, Hutchison WD (2013) Cabbage aphids. VegEdge, vegetable IPM resource for the midwest. University of Minnesota, Minneapolis, MN. http://www.vegedge.umn.edu/vegpest/colecrop/aphid.htm
Höglund AS, Lenman M, Rask L (1992) Myrosinase is localized to the interior of myrosin grains and is not associated to the surrounding tonoplast membrane. Plant Sci 85:165–170
Hopkins RJ, van Dam NM, van Loon JJA (2009) Role of glucosinolates in insect plant relationships and multitrophic interactions. Annu Rev Entomol 54:57–83
Hossain MA, Maiti MK, Basu A, Sen S, Ghosh AK, Sen SK (2006) Transgenic expression of onion leaf lectin in Indian mustard offers protection against aphid colonization. Crop Sci 46:2022–2032
Imlau A, Truernit E, Sauer N (1999) Cell-to-cell and long-distance trafficking of the green fluorescent protein in the phloem and symplastic unloading of the protein in sink tissues. Plant Cell 11:309–322
Jarvis JL (1970) Relative injury to some cruciferous oilseeds by the turnip aphid. J Econ Entomol 63:1498–1502
Jensen CR, Mogensen VO, Mortensen G, Fieldsend JK, Milford GFJ, Andersen MN, Thage JH (1996) Seed glucosinolate, oil and protein contents of field-grown rape (Brassica napus L.) affected by soil drying and evaporative demand. Field Crop Res 47:93–105
Jenson EB, Felkl G, Kristiansen K, Andersen SB (2002) Resistance to the cabbage root fly, Delia radicum within rassica fruticulosa. Euphytica 124:379–386
Jones P, Vogt T (2001) Glycosyltransferases in secondary plant metabolism: tranquilizers and stimulant controllers. Planta 213:164–174
Jones TH, Cole RA, Finch S (1988) A cabbage root fly oviposition deterrent in the frass of garden pebble moth caterpillars. Entomol Exp Appl 49:277–282
Kalra VK, Singh H, Rohilla HR (1987) Influence of various genotypes of Brassica juncea on biology of mustard aphid, Lipaphis erysimi (Kalt.). Indian J Agric Sci 57:277–279
Kanrar S, Venkateswari J, Kirti PB, Chopra VL (2002) Transgenic India mustard (Brassica juncea) with resistance to the mustard aphid (Lipaphis erysimi Kaltenbach). Plant Cell Rep 20:976–981
Kawada K, Murai T (1979) Apterous males and holocyclic reproduction of Lipaphis erysimi in Japan. Entomol Exp Appl 26:343–345
Kehr J (2006) Phloem sap proteins: their identities and potential roles in the interaction between plants and phloem-feeding insects. J Exp Biol 57:767–774
Kelly PJ, Bones A, Rossiter JT (1998) Sub-cellular immunolocalisation of the glucosinolate sinigrin in seedlings of Brassica juncea. Planta 206:370–377
Kift NB, Ellis PR, Tatchell GM, Pink DAC (2000) The influence of genetic background on resistance to the cabbage aphid (Brevicoryne brassicae) in kale (Brassica oleracea var. acephala). Ann Appl Biol 136:189–195
Kissen R, Rossiter JT, Bones AM (2009) The “mustard oil bomb”: not so easy to assemble?! Localization, expression and distribution of the components of the myrosinase enzyme system. Phytochem Rev 8:69–86
Kloth KJ, ten Broeke CJM, Thoen MPM, van den Brink MH, Wiegers GL, Krips OE, Noldus LPJJ, Dicke M, Jongsma MA (2015) High-throughput phenotyping of plant resistance to aphids by automated video tracking. Plant Methods 11:4. doi:10.1186/s13007-015-0044-z
Komath SS, Kavitha M, Swamy MJ (2006) Beyond carbohydrate binding: new directions in plant lectin research. Org Biomol Chem 4:973–988
Kumar S, Atri C, Sangha MK, Banga SS (2011) Screening of wild crucifers for resistance to mustard aphid, Lipaphis erysimi (Kaltenbach) and attempt at introgression of resistance gene(s) from Brassica fruticulosa to Brassica juncea. Euphytica 179:461–470. doi:10.1007/s10681-011-0351-z
Kusnierczyk A, Winge P, Jørstad T, Troczyńska J, Rossiter JT, Bones AM (2008) Towards global understanding of plant defence against aphids—timing and dynamics of early Arabidopsis defence responses to cabbage aphid (Brevicoryne brassicae) attack. Plant Cell Environ 31:1097–1115
Labana KS (1976) Release of mutant variety of raya (Brassica juncea). Mutat Breed Newsl 7:11
Lamb RJ (1980) Hairs protect pods of mustard (Brassica hirta ‘Gisilba’) from flea beetle feeding damage. J Plant Sci 60:1439–1440
Lamb RJ, Smith MAH, Bodnaryk RP (1993) Leaf waxiness and the performance of Lipaphis erysimi (Kaltenbach) (Homoptera: Aphididae) on three Brassica crops. Can Entomol 125:1023–1031
Lammerink J (1968) Rangi: new rape that resists aphids. N Z J Agric 117:61
LeCoz C, Ducombs G (2006) Plants and plant products. In: Frosch PJ, Menne T, Lepottevin JP (eds) Contact dermatitis, 4th edn. Springer, Berlin/Heidelberg, pp 751–800
Loe G, Torang P, Gaudeul M, Agren J (2007) Trichome production and spatiotemporal variation in herbivory in the perennial herb Arabidopsis lyrata. Oikos 116:134–142
Louda S, Mole S (1991) Glucosinolates, chemistry and ecology. In: Rosenthal GA, Berenbaum MR (eds) Herbivores. Their interactions with secondary plant metabolites, vol 1, 2nd edn. Academic Press, San Diego, pp 123–164
Lüthy B, Matile P (1984) The mustard oil bomb: rectified analysis of the subcellular organization of the myrosinase system. Biochem Physiol Pflanzen 179:5–12
Macedo MLR, de Castro MM, Freire MDGM (2004) Mechanisms of the insecticidal action of TEL (Talisia esculenta Lectin) against Callosobruchus maculatus (Coleoptera: Bruchidae). Arch Insect Biochem Physiol 56:84–96
Maffei ME, Mithofer A, Boland W (2007) Before gene expression: early events in plant-insect interaction. Trends Plant Sci 12:310–316
Malik RS (1981) Morphological, anatomical and biochemical basis of aphid, Lipaphis erysimi (Kalt.) resistance in cruciferous species. Sver Utsaedesfoeren Tidskr 91:25–35
Mandal P (2003) Development and characterization of somatic hybrids between Rorippa indica and Brassica juncea. Dissertation, Jadhavpur University, Kolkata
Meisner J, Mitchell BK (1984) Phagodeterrency induced by some secondary plant substances in adults of the flea beetle Phyllotreta striolata. J Plant Dis Prot 91:301–304
Mewis IZ, Ulrich C, Schnitzler WH (2002) The role of glucosinolates and their hydrolysis products in oviposition and host plant finding by cabbage webworm, Hellula undalis. Entomol Exp Appl 105:129–139
Michiels K, van Damme EJM, Smagghe G (2010) Plant-insect interactions: what can we learn from plant lectins? Arch Insect Biochem Physiol 73:193–212
Miles PW (1999) Aphid saliva. Biol Rev 74:41–85
Milford GFJ, Fieldsend JK, Porter AJR, Rawlinson CJ, Evans EJ, Bilsborrow PE (1989) Changes in glucosinolate concentrations during the vegetative growth of single- and double-low cultivars of winter oilseed rape. Asp Appl Biol 23:83–90
Mitchell-Olds T (2001) Arabidopsis thaliana and its wild relatives: a model system for ecology and evolution. Trends Ecol Evol 16:693–700
Moran NA (1992) The evolution of aphid life cycles. Annu Rev Entomol 37:321–348
Moran NA, Russell JA, Koga R, Fukatsu T (2005) Evolutionary relationships of three new species of Enterobacteriaceae living as symbionts of aphids and other insects. Appl Environ Microbiol 71:3302–3310
Muir AD, Gruber MY, Hinks CF, Lees GL, Onyilagha J, Soroka J, Erlandson M (1999) Effect of condensed tannins in the diets of major crop insects. In: Gross G, Hemingway RW, Yoshida T (eds) Plant Polyphenols 2: Chemstry, biology, pharmacology Ecology. Kluwer Academic/Plenum Publ, NY, pp 867–881
Munson MA, Baumann P, Kinsey MG (1991) Buchnera gen. nov. and Buchnera aphidicola sp. nov., a taxon consisting of the mycetocyteassociated, primary endosymbionts of aphids. Int J Syst Bacteriol 41:566–568
Muratori F, Le Ralec A, Lognay G, Hance T (2006) Epicuticular factors involved in host recognition for the aphid parasitoid Aphidius rhopalosiphi. J Chem Ecol 32:579–593
Murdock LL, Huesing JE, Nielsen SS, Pratt RC, Shade RE (1990) Biological effects of plant lectins on the cowpea weevil. Phytochemistry 29:85–89
Mutti NS, Park Y, Reese JC, Reek GR (2006) RNAi knockdown of a salivary transcript leading to lethality in the pea aphid Acyrthosiphon pisum. J Insect Sci 6:38
Mutti NS, Louis J, Pappan LK, Pappan K, Begum K, Chen MS, Park Y, Dittmer N, Marshall J, Reese JC, Reeck GR (2008) A protein from the salivary glands of the pea aphid, Acyrthosiphon pisum, is essential in feeding on a host plant. Proc Natl Acad Sci U S A 105:9965–9969
Nachbar MS, Oppenheim JD (1980) Lectins in the United States diet: a survey of lectins in commonly consumed foods and a review of the literature. American J Clinical Nutr 33:2338–2345
Nguz K, van Gaver D, Huyghebaert A (1998) In vitro inhibition of digestive enzymes by sorghum condensed tannins [Sorghum bicolour L. (Moench)]. Sci Aliment 18:507–514
Nielsen JK (1978) Host plant discrimination within Cruciferae: feeding responses of four leaf beetles (Coleoptera: Chrysomelidae) to glucosinolates, cucurbitacins and cardenolides. Entomol Exp Appl 24:41–54
Nielsen JK, Larsen LM, Sørensen H (1979) Host plant selection of the horseradish flea beetle Phyllotreta armoraciae (Coleoptera: Chrysomelidae): identification of two flavonol glycosides stimulating feeding in combination with glucosinolates. Entomol Exp Appl 26:40–48
Nottingham SF, Hardie J, Dawson GW, Hick AJ, Pickett JA, Wadhams LJ, Woodcock CM (1991) Behavioral and electrophysiological responses of aphids to host and nonhost plant volatiles. J Chem Ecol 17:1231–1242
Oliver KM, Degnan PH, Burke GR, Moran NA (2010) Facultative symbionts in aphids and the horizontal transfer of ecologically important traits. Annu Rev Entomol 55:247–266
Onyilagha JC, Lazorko J, Gruber MY, Soroka JJ, Erlandson MA (2004) Effect of flavonoids on feeding preference and development of the crucifer pest Mamestra configurata Walker. J Chem Ecol 30:109–124
Pedras MSC, Yaya EE (2010) Phytoalexins from Brassicaceae: news from the front. Phytochemistry 71:1191–1197
Pitino M, Coleman AD, Maffei ME, Ridout CJ, Hogenhout SA (2011) Silencing of aphid genes by dsRNA feeding from plants. PLoS One 6(10):e25709. doi:10.1371/journal.pone.0025709
Ponsen MB (1972) The site of potato leafroll virus multiplication in its vector, Myzus persicae-An anatomical study. Mededlingen Landbouwhogeschool Wageningen 72(16):1–147
Pope TW, Kissen R, Grant M, Pickett JA, Rossiter JT, Powell G (2008) Comparative innate responses of the aphid parasitoid Diaeretiella rapae to alkenyl glucosinolate derived isothiocyanates, nitriles and epithionitriles. J Chem Ecol 34:1302–1310
Porter AJR, Morton AM, Kiddle G, Doughty KJ, Wallsgrove RM (1991) Variation in the glucosinolate content of oilseed rape (Brassica napus L.). I. Effects of leaf age and position. Ann Appl Biol 118:461–467
Powell KS (2001) Antimetabolic effects of plant lectins towards nymphal stages of the plant hoppers Tarophagous proserpina and Nilaparvata lugens. Entomol Exp Appl 99:71–77
Powell KS, Gatehouse AMR, Hilder VA, Gatehouse JA (1993) Antimetabolic effects of plant lectins and plant and fungal enzymes on the nymphal stages of two important rice pests, Nilaparvata lugens and Nephotettix cinciteps. Entomol Exp Appl 66:119–126
Prakash S, Raut RN (1983) Artificial synthesis of Brassica napus and its prospects as an oilseeds crop in India. J Genet 43:282–290
Quaglia F, Rossi E, Petacchi R, Taylor CE (1993) Observations on an infestation by green peach aphids (Homoptera: Aphididae) on greenhouse tomatoes in Italy. J Econ Entomol 86:1019–1025
Rahbé Y, Deraison C, Bonadé-Bottino M, Girard C, Nardon C, Jouanin L (2003) Effects of the cysteine protease inhibitor oryzacystatin (OC-I) of different aphids and reduced performance of Myzus persicae on OC-I expressing transgenic oilseed rape. Plant Sci 164:441–450
Rajan SS (1961) Aphid resistance of autotetraploid toria. Indian Oilseeds J 8:251–255
Rana J (2005) Performance of Lipaphis erysimi (Homoptera: Aphididae) on different Brassica species in a tropical environment. J Pest Sci 78:155–160
Rask L, Andreasson E, Ekbom B, Eriksson S, Pontoppidan B, Meijer J (2000) Myrosinase: gene family evolution and herbivore defence in Brassicaceae. Plant Mol Biol 42:93–113
Ratzka A, Vogel H, Kliebenstein DJ, Mitchell-Olds T, Kroymann J (2002) Disarming the mustard oil bomb. P Natl Acad Sci USA 99:11223–11228
Raymer PL (2002) Canola: an emerging oilseed crop. In: Janwick J, Whipkey A (eds) Trends in new crops and new uses. ASHS Press, Alexandria, VA, pp 122–126
Reifenrath K, Riederer M, Müller C (2005) Leaf surface wax layers of Brassicaceae lack feeding stimulants for Phaedon cochleariae. Entomol Exp Appl 115:41–50
Remaudière G, Remaudière M (1997) Catalogue des Aphididae du Monde. INRA, Paris
Renwick JAA (2002) The chemical world of crucivores: lures, treats and traps. Entomol Exp Appl 104:35–42
Renwick JAA, Radke CD, Sachdev-Gupta K, Städler E (1992) Leaf surface chemicals stimulating oviposition by Pieris rapae (Lepidoptera: Pieridae) on cabbage. Chemoecology 3:33–38
Renwick JAA, Haribal M, Gouinguenѐ S, Städler E (2006) Isothiocyanates stimulating oviposition by the diamondback moth, Plutella xylostella. J Chem Ecol 32:755–766
Rossiter JT, Jones AM, Bones AM (2003) A novel myrosinase-glucosinolate defense system in cruciferous specialist aphids. Recent Adv Phytochem 37:127–142
Russell JA, Latorre A, Sabater-Munoz B, Moya A, Moran NA (2003) Independent origins and horizontal transfer of bacterial symbionts of aphids. Mol Ecol 12:1061–1075
Sadasivam S, Thayumanavan B (2003) Molecular host plant resistance to pests. Marcel Dekker Inc., New York
Sadeghi H (2002) The relationship between oviposition preference and larval performance in an aphidophagous hover fly, Syrphus ribesii L. (Diptera: Syrphidae). J Agric Sci Technol 4:1–10
Sandstrom JP, Russell JA, White JP, Moran NA (2001) Independent origins and horizontal transfer of bacterial symbionts of aphids. Mol Ecol 10:217–228
Sarkar P, Jana J, Chatterjee S, Sikdar SR (2016) Functional characterization of Rorippa indica defensin and its efficacy against Lipaphis erysimi. Springer Plus 5:511. doi:10.1186/s40064-016-2144-2
Sauvion N, Charles H, Febvay G, Rahbé Y (2004a) Effects of jackbean lectin (ConA) on the feeding behaviour and kinetics of intoxication of the pea aphid, Acyrthosiphon pisum. Entomol Exp Appl 110:31–44
Sauvion N, Nardon C, Febvay G, Gatehouse AMR, Rahbé Y (2004b) Binding of the insecticidal lectin Concanavalin A in pea aphid, Acyrthosiphon pisum (Harris) and induced effects on the structure of midgut epithelial cells. J Insect Physiol 50:1137–1150
Saxena AK, Bhadoria SS, Gadewadikar PN, Barteria AM, Tomar SS, Dixit SC (1995) Yield losses in some improved varieties of mustard by aphid, Lipaphis erysimi (Kalt.). Agric Sci Dig 15:235–237
Schnee C, Kollner TG, Held M, Turlings TCJ, Gershenzon J, Degenhardt J (2006) The products of a single maize sesquiterpene synthase form a volatile defense signal that attracts natural enemies of maize herbivores. Proc Natl Acad Sci 103(4):1129–1134
Schoonhoven LM, Dicke M, van Loon JJA (2007) Insect–plant biology. Oxford University Press, Oxford
Sekhon BS, Åhman I (1992) Insect resistance with special reference to mustard aphid. In: Lubana KS, Banga SS, Banga SK (eds) Monographs on theoretical and applied genetics: breeding oilseed brassicas. Springer, New York, pp 206–221
Shakesby AJ, Wallace LS, Isaacs HV, Pritchard J, Roberts DM, Douglas AE (2009) A water-specific aquaporin involved in aphid osmoregulation. Insect Biochem Mol Biol 39:1–10
Sharma A, Khan AN, Subrahmanyam S, Raman A, Taylor GS, Fletcher MJ (2014) Salivary proteins of plant feeding hemipteroids – implications in phytophagy. Bull Entomol Res 104:117–136. doi:10.1017/S0007485313000618
Singh CP, Sachan GC (1994) Assessment of yield losses in yellow sarson due to mustard aphid, Lipaphis erysimi (Kalt.). J Oilseeds Res 11:179–184
Singh SR, Narain A, Srivastava KP, Siddiqui RA (1965) Fecundity of mustard aphid on different rape and mustard species. Indian Oilseeds J 9:215–219
Smallegange R, van Loon J, Blatt S, Harvey J, Agerbirk N, Dicke M (2007) Flower vs. leaf feeding by Pieris brassicae: glucosinolate rich flower tissues are preferred and sustain higher growth rate. J Chem Ecol 33:1831–1844
Smith CM (1989) Plant resistance to insects- a fundamental approach. Wiley, New York, p 286
Smith CM, Chuang WP (2014) Plant resistance to aphid feeding: behavioral, physiological, genetic and molecular cues regulate aphid host selection and feeding. Pest Manag Sci 70:528–540
Sotelo T, Soengas P, Velasco P, Rodriguez VM, Cartea ME (2014) Identification of metabolic QTLs and candidate genes for glucosinolate synthesis in Brassica oleracea leaves, seeds and flower buds. PLoS One. doi:10.1371/journal.pone.0091428
Southwood SR (1986) Plant surfaces and insects-an overview. In: Juniper B, Southwood SR (eds) Insects and the plant surface. Arnold, London, pp 1–22
Srinivasachar D, Malik RS (1972) An induced aphid resistant, non-waxy mutant in turnip, Brassica rapa. Curr Sci 41:820–821
Srinivasachar D, Verma PK (1971) Induced aphid resistance in Brassica juncea (L.) Coss. Curr Sci 49:311–313
Städler E, Reifenrath K (2009) Glucosinolates on the leaf surface perceived by insect herbivores: review of ambiguous results and new investigations. Phytochem Rev 8:207–225
Stern DL (1995) Aphidomorpha. Aphids, green flies, plant lice, adelgids, phylloxerids. In: the tree of life web project, http://tolweb.org/Aphidomorpha/10985/1995.01.01
Stork NE (1980) Role of waxblooms in preventing attachment to brassicas by the mustard beetle, Phaedon cochleariae. Entomol Exp Appl 28:100–107
Thangstad OP, Evjen K, Bones A (1991) Immunogold-EM localization of myrosinase in Brassicaceae. Protoplasma 161:85–93
Thomma BPHJ, Penninckx IAMA, Cammue BPA, Broekaert WF (2001) The complexity of disease signaling in Arabidopsis. Curr Opin Immunol 13:63–68
Thomma B, Cammue B, Thevissen K (2002) Plant defensins. Planta 216:193–202
Thompson GA, Goggin FL (2006) Transcriptomics and functional genomics of plant defence induction by phloem-feeding insects. J Exp Bot 57:755–766
Tjallingii WF (1988) Electrical recording of stylet penetration activities. In: Minks AK, Harrewijn P (eds) Aphids, their biology, natural enemies and control, vol 2B. Elsevier, Amsterdam, pp 95–108
Traw BM (2002) Is induction response negatively correlated with constitutive resistance in black mustard? Evolution 56:2116–2205
Traw MB, Dawson TE (2002) Reduced performance of two specialist herbivores (Lepidoptera: Pieridae, Coleoptera: Chrysomelidae) on new leaves of damaged black mustard plants. Environ Entomol 31:714–722
Trebicki P, Tjallingii WF, Harding RM, Rodoni BC, Powell KS (2012) EPG monitoring of the probing behaviour of the common brown leafhopper Orosius orientalis on artificial diet and selected host plants. Arthropod Plant Interact 6:405–415
Tripathi MK, Mishra AS (2007) Glucosinolates in animal nutrition: a review. Animal Feed Sci Technol 132:1–27
Tumlinson JH, Pare P, Lewis WJ (1999) Plant production of volatile semiochemicals in response to insect-derived elicitors. In: Insect-plant interactions and induced plant defense. Wiley, Chichester, pp 95–109
Ulmer BJ, Gillott C, Woods D, Erlandson M (2002) Diamondback moth, Plutella xylostella (L.), feeding and oviposition preferences on glossy and waxy Brassica rapa (L.) lines. Crop Prot 21:327–331
Urbanska A, Tjallingii WF, Dixon AFG, Leszczynski B (1998) Phenol oxidizing enzymes in the grain aphid’s saliva. Entomol Exp Appl 86:197–203
van Loon JJA, Chen ZW, Nielsen JK, Gols R, Yu TQ (2002) Flavonoids from cabbage are feeding stimulants for diamondback moth larvae additional to glucosinolates: chemoreception and behaviour. Entomol Exp Appl 104:27–34
Vandenborre G, Smagghe G, van Damme EJM (2011) Plant lectins as defense proteins against phytophagous insects. Phytochemistry 72:1538–1550. doi:10.1016/j.phytochem.2011.02.024
Vasconcelos IM, Oliveira JT (2004) Antinutritional properties of plant lectins. Toxicon 44:385–403
Walling LL (2008) Avoiding effective defenses: strategies employed by phloem-feeding insects. Plant Physiol 146:859–866
Walton TJ (1990) Waxes, cutin and suberin. In: Dey PM, Harborne JB (eds) Methods in plant biochemistry. Academic Press, San Diego, pp 105–158
Werker E (2000) Trichome diversity and development. Adv Bot Res 31:1–35
Wietsma R (2010) The effect of differences in aliphatic glucosinolate concentrations in Arabidopsis thaliana on herbivores of different feeding guilds and different levels of specialization. Dissertation, Wageningen University and Research Centre, Wageningen
Will T, Vilcinskas A (2013) Aphid proof plants: biotechnology-based approaches for aphid control. Adv Biochem Eng Biotechnol. doi:10.1007/10_2013_211
Will T, Tjallingii WF, Thönnessen A, van Bel AJE (2007) Molecular sabotage of plant defense by aphid saliva. P Natl Acad Sci USA 104:10536–10541
Will T, Kornemann SR, Furch ACU, Tjallingii WF, van Bel AJE (2009) Aphid watery saliva counteracts sieve-tube occlusion: a universal phenomenon? J Exp Biol 212:3305–3312
Will T, Steckbauer K, Hardt M, van Bel AJE (2012) Aphid gel saliva: sheath structure, protein composition and Secretory dependence on Stylet-tip milieu. PLoS One 7(10):e46903. doi:10.1371/journal.pone.0046903
Will T, Furch ACU, Zimmermann MR (2013) How phloem feeding insects face the challenge of phloem located defenses. Front Plant Sci 4:1–12. doi:10.3389/fpls.2013.00336
Williams IS, Dixon AFG (2007) Life cycles and polymorphism. In: van Emden HF, Harrington R (eds) Aphids as crop pests. CABI, Wallingford, pp 69–85
Wittstock U, Agerbirk N, Stauber EJ, Olsen CE, Hippler M, Mitchell-Olds T, Gershenzon J, Vogel H (2004) Successful herbivore attack due to metabolic diversion of a plant chemical defense. P Natl Acad Sci USA 101:4859–4864
Zhu-Salzman K, Salzman RA, Ahn JE, Koiwa H (2004) Transcriptional regulation of sorghum defense determinants against a phloem-feeding aphid. Plant Physiol 134:420–431
Zust T, Agrawal AA (2016) Mechanisms and evolution of plant resistance to aphids. Nature Plants. doi:10.1038/NPLANTS.2015.206
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Kumar, S., Banga, S.S. (2017). Breeding for Aphid Resistance in Rapeseed Mustard. In: Arora, R., Sandhu, S. (eds) Breeding Insect Resistant Crops for Sustainable Agriculture. Springer, Singapore. https://doi.org/10.1007/978-981-10-6056-4_6
Download citation
DOI: https://doi.org/10.1007/978-981-10-6056-4_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-6055-7
Online ISBN: 978-981-10-6056-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)