Abstract
Invariant natural killer T (iNKT) cells are a subpopulation of T lymphocytes with unique specificities against (glyco)lipids presented in the context of CD1d belonging to major histocompatibility complex (MHC) class Ib. They recognise microbially encoded or synthetic glycolipids directly through their CD1d-restricted T-cell receptors (TCRs) other than microbial components or lipids including pathogen-associated molecular patterns (PAMPs) and bacterial superantigens. Once activated, iNKT cells participate as early effectors and/or regulators of immune responses. Immunoregulatory cytokines produced in copious amounts by these cells target a wide range of downstream effector cells and help shape the ensuing immune responses. Mammals comprise various numbers and isoforms of CD1 molecules, suggesting that (glyco)lipid presentation is a rapidly evolving component of the immune system, which adapts to environmental threats. Recent progress in our understanding of CD1d-restricted iNKT cells contributes to their true potentials in immunotherapeutic applications for various diseases. Recent findings about iNKT cell subtypes (iNKT1, 2, 17, 10) and their roles in pathological inflammation are also introduced and discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The immune system consists of separate but interacting components, the innate and acquired immune systems. Invariant natural killer T (iNKT) cells play unique intermediary roles, bridging between both immune systems.
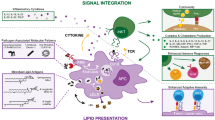
NKT cells were originally characterised as having cell surface markers such as NK1.1 in mice (MacDonald 1995) and CD161 in humans (Schmidt et al. 1986), as well as a T-cell receptor (TCR) (Makino et al. 1995; Porcelli et al. 1993). However, recent studies have indicated that a certain but not all of the NKT cells express NK1.1 in mice or CD161 in humans, but all NKT cells are defined by a particular invariant TCR α chain from Trav11_Traj18 (Vα14-Jα18) mostly paired with Trbv13-2 (Vβ8.2), Trbv13-3 (Vβ8.1), Trbv29 (Vβ7), or Trbv1 (Vβ2) in mice and TRAV10_TRAJ18/TRBV25-1_TRBD2_TRBJ2-7 (Vα24-Jα18/Vβ11-Dβ2-Jβ2.7) in humans, giving rise to the moniker ‘invariant’ for NKT cells. Different from the conventional αβT cells which recognise the peptide/MHC complex, these stereotypic TCRs endow iNKT cells with some types of glycolipid antigens presented by monomorphic MHC class I-like molecule CD1d (Godfrey et al. 2004; Salio et al. 2014). The prototypic antigen is glycosphingolipid α-galactosylceramide (α-GalCer), originally isolated from the marine sponge Agelas mauritianus and identified from structure-activity relationship studies around Agelasphin 9b by the pharmaceutical division of Kirin Brewery Co. Ltd. in a screen for naturally occurring molecules that prevented tumour metastases in mice in vivo (Natori et al. 1994). The synthetic compound, also known as KRN7000, retains the activity of Agelasphin 9b while being much easier to synthesise (Morita et al. 1995). α-GalCer has been used in many different studies and is a highly potent iNKT cell agonist both in humans and in mice (Hayakawa et al. 2004).
When activated by α-GalCer, iNKT cells immediately produce Th1 (IFN-γ), Th2 (IL-13, IL-4), Th17 (IL-17A), and inhibitory (IL-10) cytokines , resulting in bystander immune modulating functions, activating and inhibiting various immune effector cells, including NK cells , macrophages, granulocytes, dendritic cells (DCs) , basophils, and eosinophils in the innate system as well as CD4+ T and CD8+ T cells in the acquired system. Therefore, iNKT cells participate in the regulation of various disease states in a broad spectrum, including infection, autoimmunity , allergy, antitumour response, allograft rejection, and graft-versus-host disease.
The numerous studies investigating the role of iNKT cells were represented by mouse models of deficiency in iNKT cells . One makes use of mice deficient in CD1d (Cd1d1 -/-) (Mendiratta et al. 1997), which prevents the development of any CD1d -reactive T cells including iNKT cells . Another model directly targets Jα18 (Traj18 -/-) (Cui et al. 1997), which is required for iNKT-TCR formation. However, TCR repertoire diversity was impaired in Traj18 -/- mice, in which the PGK-Neo r cassette replaced from Traj18 has inadvertent substantial effects on transcription and TCR rearrangements (Bedel et al. 2013). Consequently, any changes in immunological activity associated with Traj18 -/- and Cd1d1 -/- mice and for which a role has been ascribed to iNKT cells needs to be reassessed.
2 iNKT Cells and Host Defence
There is evidence that iNKT cells participate in protection of mice from a variety of bacterial, viral, protozoan, and fungal parasites, although some of these results are controversial because susceptibility is different between Traj18 -/- and Cd1d1 -/- mice or between C57BL/6 and BALB/c genetic backgrounds.
In some examples where the mechanism of iNKT cell action in host defence has been partially defined, IFN-γ secretion is important. iNKT cells act not only as effectors by themselves in these cases, but also as conductors of IFN-γ secretion from NK cells and CD8+ T cells that have been stimulated as a result of iNKT cell activation (Hayakawa et al. 2001; Carnaud et al. 1999; Smyth et al. 2002a). However, iNKT cells can operate in either a stimulating or suppressive manner. For example, clearance of herpes simplex virus type 1 is reduced in Traj18 -/- and Cd1d1 -/- mice (Grubor-Bauk et al. 2003). By contrast, iNKT cells negatively regulate the response to lymphocytic choriomeningitis virus (Roberts et al. 2004).
A defect in iNKT cell development has been reported both in SAP (signalling lymphocytic activation molecule associated protein)-deficient mice and in humans with mutations in SH2D1A, the gene encoding SAP (Chung et al. 2005; Nichols et al. 2005; Pasquier et al. 2005). The Sh2d1a -/- mice are relatively normal in the development of T and B cells, whereas humans with SAP mutations are affected by a severe X-linked lymphoproliferative (XLP) syndrome and fail to control primary Epstein–Barr virus (EBV) infection, presenting with uncontrolled expansions of T and B cells. Although SAP-deficient XLP patients have a plethora of immune defects that could contribute to the lack of control of EBV infection, another group of XLP patients with defective iNKT cell numbers has been reported who bear mutations in the inhibitor of apoptosis XIAP gene (Rigaud et al. 2006). This raises questions of whether and how iNKT cells might be involved in the control of EBV and perhaps other viral infections.
Some microbes produce glycolipids different from viruses . iNKT cells are known to respond directly to some exogenous microbial glycolipids. Sphingomonas bacteria widely distributed in the environment are essentially universal antigens for iNKT cells and their glycosphingolipids containing either a galacturonic or a glucuronic acid have structures similar to α-GalCer and have an ability to activate iNKT cells directly (Kinjo et al. 2005; Mattner et al. 2005; Sriram et al. 2005).
Another example of the importance of iNKT cells is provided by a study of a Lyme disease model infected with Borrelia burgadorferi. After intradermal infection with Borrelia burgadorferi, Cd1d1 -/- mice developed a greater thickening of the knee and tibiotarsal joints, indicative of arthritis compared to BALB/c mice (Kumar et al. 2000).
It has been suggested that some Bacteroides sphingolipids might influence host immune homeostasis. Species of Bacteroides and their relatives are a prevalent commensal organism of the human intestine, and many of them also have the capacity to produce sphingolipids. Recently, a glycosphingolipid antigen from Bacteroides fragilis has been purified and characterised and it also activates mouse and human iNKT cells (Wieland Brown et al. 2013).
The role of iNKT cells in infection and host defence is clearly an exciting area of investigation with clinical potential, which makes them important targets for future vaccine design.
3 iNKT Cells and Antitumour Responses
iNKT cells have been shown to be potentially important to tumour rejection (Smyth et al. 2002b). iNKT cells were found to be necessary for IL-12–mediated tumour therapy in mice (Cui et al. 1997), and α-GalCer/KRN7000 has an ability to promote iNKT cell-dependent rejection of a broad range of experimental tumour cell lines including melanoma, carcinoma, thymoma, and sarcoma (Hayakawa et al. 2004; Smyth et al. 2002b; Kawano et al. 1997). Recent evidence has also shown that treatment with α-GalCer/KRN7000 can protect against spontaneous oncogene- or carcinogen-induced primary tumour formation in mice (Hayakawa et al. 2003). Based on these findings in mice, a phase I clinical study of KRN7000 was carried out in patients with solid tumours (Giaccone et al. 2002). However, a detectable level of IFN-γ and IL-12 in serum was never observed after 3 times intravenous (i.v.) injection of KRN7000 (Giaccone et al. 2002). This may be partially due to the small population of iNKT cells in cancer patients compared to the healthy, although it is not clear whether this is a cause or effect of the cancer. Another possible reason is repeated injection of KRN7000 elicits changes in immunity as regulatory properties by IL-10 production from iNKT cells and dendritic cells (DCs) as shown in mice (Kojo et al. 2005).
Overcoming these problems has been demonstrated. Induced pluripotent stem cells (iPSCs) are known to hold tremendous potential for cell replacement therapy and functionally competent mouse iNKT cells from iPSCs were successfully generated that suppressed tumour growth in vivo (Watarai et al. 2010), which is an important advance in solving the problem of the limited number of iNKT cells in advanced cancer patients (Watarai et al. 2012a). Concerning the lesser effectiveness of KRN7000 in humans, i.v. injection of monocyte -derived DCs that were KRN7000-pulsed was evaluated and led to sustained expansion of iNKT cells in advanced cancer patients (Chang et al. 2005). iNKT-cell–mediated immunotherapy has clearly provided a proof of concept for future perspectives for cancer immunotherapy even though numerical tests and trials should be done and there is much to be learned before these cells can be safely and effectively manipulated in the clinic.
4 iNKT Cells and Autoimmunity
It has been suggested that iNKT cells naturally influence autoimmunity from some mouse models and the greatest number of studies was investigated in the type 1 diabetes model. iNKT cells are reduced in nonobese diabetic (NOD) mice (Ortaldo et al. 2004) and produce less IL-4 after stimulation (Hammond et al. 1998). Nonetheless, stimulation of iNKT cells with repeated administration of KRN7000 has a therapeutic effect and protection from diabetes by iNKT cells was associated with the induction of a Th2 response to islet autoantigens (Hong et al. 2001). Interestingly however, iNKT cells prevented differentiation of IFN-γ producing diabetogenic T cells in NOD mice in an iNKT-derived IL-4–independent manner but through activating NK cells and DCs (Beaudoin et al. 2002). Taken collectively, iNKT cells may act by inducing KRN7000-mediated anergy in islet-specific T cells and/or by altering the function of DCs with regulatory, in addition to Th2 polarisation .
Similar to the findings in NOD mice, KRN7000 was found to prevent experimental autoimmune encephalomyelitis (EAE; Jahng et al. 2001), an animal model of multiple sclerosis (MS) , by shifting the balance from a pathogenic Th1 toward a Th2 response. A decrease in iNKT cells has also been found in the peripheral blood of MS patients, suggesting a regulatory role for iNKT cells in this human disease (Araki et al. 2003).
5 The Growing iNKT Cell Subsets
A major paradox is the ability of iNKT cells to produce various types of cytokines and to both promote and suppress immune responses as described above. This is probably due to the existence of functionally distinct subtypes of iNKT cells producing different cytokines. It was previously established that iNKT cells include both CD4-positive and CD4-negative subtypes (Godfrey et al. 2004; Bendelac et al. 2007), each of which produces different cytokines. Although a functional distinction was originally less apparent for mouse CD4+ and CD4− iNKT cell subsets, the division of mouse iNKT cells on the basis of NK1.1 expression has revealed striking differences between NK1.1-positive and NK1.1-negative subsets. NK1.1+ iNKT cells produce a large amount of IFN-γ and little IL-4, whereas NK1.1− iNKT cells produce less IFN-γ and more IL-4 (Benlagha et al. 2002; Pellicci et al. 2002).
More recently, there is further heterogeneity in CD4+ iNKT cells, a subset of which bears IL-17 receptor B (also known as IL-25 receptor; Terashima et al. 2008). IL-25 is a key cytokine in Th2 immunity that eliminates helminth and promotes airway hyperreactivity (Fallon et al. 2006). IL-17RB+ CD4+ iNKT cells which can be designated iNKT2 cells produce large amounts of IL-13, IL-9, and IL-4 in response to IL-25 (Terashima et al. 2008; Angkasekwinai et al. 2010; Motomura et al. 2011) and are phenotypically NK1.1− and present mainly in thymus , spleen, lung, and lymph nodes (Watarai et al. 2012b). Another newly identified subset of iNKT cells is producing predominant IL-17A (Michel et al. 2007). IL-17A is produced from Th17 cells and/or γδ T cells, which have a key role in autoimmune pathology in disease models such as EAE and collagen-induced arthritis (CIA; Weaver et al. 2007). IL-17A–producing iNKT cells (iNKT17 cells) express the transcription factor RORγt, IL-23 receptor, and CCR6, the same as Th17 cells (Michel et al. 2008) and are enriched in lymph nodes and the skin, resulting in rapid production of IL-17A in response to inflammation (Watarai et al. 2012b; Doisne et al. 2009, 2011; Fig. 27.1). Concerning the IL-10 producing iNKT cells with regulatory property, it is still unclear whether this type of cell exists in steady state. In regard to the α-GalCer/KRN7000-induced protection from EAE, iNKT cells have been reported to regulate immune responses by inducing IL-10 production by other cells or to produce IL-10 themselves (Kojo et al. 2005; Singh et al. 2001). However, in these cases, a distinct population of iNKT cells with a unique phenotype was not described. Recently, IL-10–producing iNKT cells were determined from mice pretreated with α-GalCer retaining cytotoxic activity and maintaining the ability to respond to TCR-dependent as well as -independent cytokine-mediated stimulation, phenotypically and functionally similar to inducible regulatory T cells (Tregs; Sag et al. 2014). Furthermore, it has been reported that early after α-GalCer injection, iNKT cells acquire a T follicular helper (Tfh) phenotype, characterised by the expression of the transcription factor BCL6 (Chang et al. 2011; King et al. 2011). Taken collectively, iNKT cells consist of functionally distinct subtypes rather than a multifunctional uniform population, similar to helper T cells or innate lymphoid cells (ILCs) . It is important to elucidate the complexity, stability, plasticity, and the role of iNKT subsets in immunity and inflammation, including crosstalk with other immune cells and subtypes.
6 iNKT-Cell–Mediated Type 2 Inflammation
iNKT cells are also involved in the development of asthma , because Traj18 -/- mice fail to develop or reduce ovalbumin (OVA) antigen-induced airway hyperreactivity (AHR) (Akbari et al. 2003). Th2 cells and ILC2 are not always essential for iNKT cell-mediated AHR development, because activation of iNKT cells induces AHR in the absence of CD4+ T cells in MHC class-II–deficient mice (Meyer et al. 2006). These findings suggest that iNKT cells are directly involved in the development of AHR, independent of Th2 response or ILC2 activation in some cases. IL-17RB expressing iNKT2 cells and IL-25, a ligand for IL-17RB, might be responsible for iNKT-cell–mediated OVA/IL-25–induced AHR (Terashima et al. 2008). IL-17RB is preferentially expressed in iNKT2 cells among cells in steady state. iNKT2 cells in the lung produce predominantly IL-13 upon stimulation with IL-25 in vitro. Depletion of iNKT2 cells but not of other subtypes by IL-17RB–specific monoclonal antibodies or Traj18 -/- mice failed to develop OVA/IL-25–dependent AHR. Cell transfer of iNKT2 cells into Traj18 -/- mice also successfully reconstituted AHR induction. These results strongly suggest that iNKT2 cells play a crucial role in the pathogenesis of allergen-induced asthma. Recently, IL-25–mediated mucosal inflammation appeared to be negatively regulated by IL-17B which antagonises shared receptor IL-17RB (Reynolds et al. 2015). It should be investigated whether iNKT2-mediated IL-25–dependent inflammation is also regulated by IL-17B (Fig. 27.2).
Not only allergens but also certain viruses, such as respiratory syncytial virus (RSV), Sendai virus (SeV), metapneumovirus, and parainfluenza virus, cause childhood asthma and chronic obstructive pulmonary disease (COPD)-like symptoms, which include AHR, airway inflammation, and mucus hypersecretion (Gern & Busse 2000; Sigurs et al. 2005; Hamelin et al. 2006). It has been reported that, in a mouse model of infection with a SeV, virus-induced chronic inflammation leads to asthma that resembles human asthma and COPD even long after the apparent clearance of viruses (Kim et al. 2008). The chronic pulmonary symptoms evolved independently of CD4+ T cells but required iNKT cells, leading to accumulation of M2 macrophage resulted in IL-13–dependent lung disease (Kim et al. 2008). The link between these events needs to be better defined as to whether iNKT2 cells are involved; it was found that TREM (triggering receptor expressed on myeloid cells)-2 promotes macrophage survival and lung disease after viral infection (Wu et al. 2015).
iNKT2 cells potentially trigger respiratory syncytial virus (RSV)-induced AHR (Watarai et al. 2012b). RSV is an unusual virus in that it can cause repeated reinfections throughout life and a major viral pathogen causing extensive outbreaks of respiratory tract infections both in the very young and in vulnerable adults. iNKT2 cells produce Th2 cytokine after RSV infection, although triggering molecular mechanisms are still unclear.
7 Conclusion
iNKT cells have been characterised as a T-cell subtype with diverse and miscellaneous functions that recognise (glyco)lipids. However, recent studies revealed that iNKT cells consist of functionally and phenotypically distinct subtypes, each of which mediates different types of immunity and inflammation. iNKT cells can regulate the function of innate and adaptive immune cells in antimicrobial immunity, tumour rejection, and inflammatory diseases. Further studies are warranted to dissect the molecular mechanisms by which iNKT subtypes can modulate both pro- and anti-inflammatory responses. The challenge remains to translate the iNKT cell biology to the clinic.
References
Akbari O, Stock P, Meyer E, Kronenberg M, Sidobre S, Nakayama T, Taniguchi M, Grusby MJ, DeKruyff RH, Umetsu DT (2003) Essential role of NKT cells producing IL-4 and IL-13 in the development of allergen-induced airway hyperreactivity. Nat Med 9:582–588
Angkasekwinai P, Chang SH, Thapa M, Watarai H, Dong C (2010) Regulation of IL-9 expression by IL-25 signaling. Nat Immunol 11:250–256
Araki M, Kondo T, Gumperz JE, Brenner MB, Miyake S, Yamamura T (2003) Th2 bias of CD4+ NKT cells derived from multiple sclerosis in remission. Int Immunol 15:279–288
Beaudoin L, Laloux V, Novak J, Lucas B, Lehuen A (2002) NKT cells inhibit the onset of diabetes by impairing the development of pathogenic T cells specific for pancreatic beta cells. Immunity 17:725–736
Bedel R, Matsuda JL, Brigl M, White J, Kappler J, Marrack P, Gapin L (2013) Lower TCR repertoire diversity in Traj18-deficient mice. Nat Immunol 13:705–706
Bendelac A, Savage PB, Teyton L (2007) The biology of NKT cells. Annu Rev Immunol 25:297–336
Benlagha K, Kyin T, Beavis A, Teyton L, Bendelac A (2002) A thymic precursor to the NK T cell lineage. Science 296:553–555
Carnaud C, Lee D, Donnars O, Park SH, Beavis A, Koezuka Y, Bendelac A (1999) Cutting edge: Cross-talk between cells of the innate immune system: NKT cells rapidly activate NK cells. J Immunol 163:4647–4650
Chang DH, Osman K, Connolly J, Kukreja A, Krasovsky J, Pack M, Hutchinson A, Geller M, Liu N, Annable R, Shay J, Kirchhoff K, Nishi N, Ando Y, Hayashi K, Hassoun H, Steinman RM, Dhodapkar MV (2005) Sustained expansion of NKT cells and antigen-specific T cells after injection of alpha-galactosyl-ceramide loaded mature dendritic cells in cancer patients. J Exp Med 201:1503–1517
Chang P-P, Barral P, Fitch J, Pratama A, Ma CS, Kallies A, Hogan JJ, Cerundolo V, Tangye SG, Bittman R, Nutt SL, Brink R, Godfrey DI, Batista FD, Vinuesa CG (2011) Identification of Bcl-6-dependent follicular helper NKT cells that provide cognate help for B cell responses. Nat Immunol 13:35–43
Chung B, Aoukaty A, Dutz J, Terhorst C, Tan R (2005) Signaling lymphocytic activation moleculeassociated protein controls NKT cell functions. J Immunol 174:3153–3157
Cui J, Shin T, Kawano T, Sato H, Kondo E, Toura I, Kaneko Y, Koseki H, Kanno M, Taniguchi M (1997) Requirement for Valpha14 NKT cells in IL-12-mediated rejection of tumors. Science 278:1623–1626
Doisne JM, Becourt C, Amniai L, Duarte N, Le Luduec JB, Eberl G, Benlagha K (2009) Skin and peripheral lymph node invariant NKT cells are mainly retinoic acid receptor-related orphan receptor γt + and respond preferentially under inflammatory conditions. J Immunol 183:2142–2149
Doisne JM, Soulard V, Becourt C, Amniai L, Henrot P, Havenar-Daughton C, Blanchet C, Zitvogel L, Ryffel B, Cavaillon JM, Marie JC, Couillin I, Benlagha K (2011) Cutting edge: crucial role of IL-1 and IL-23 in the innate IL-17 response of peripheral lymph node NK1.1- invariant NKT cells to bacteria. J Immunol 186:662–666
Fallon PG, Ballantyne SJ, Mangan NE, Barlow JL, Dasvarma A, Hewett DR, Mcllgorm A, Jolin HE, McKenzie AN (2006) Identification of an interleukin (IL)-25-dependent cell population that provides IL-4, IL-5, and IL-13 at the onset of helminth expulsion. J Exp Med 203:1105–1116
Gern JE, Busse WW (2000) The role of viral infections in the natural history of asthma. J Allergy Clin Immunol 106:201–212
Giaccone G, Punt CJ, Ando Y, Ruijter R, Nishi N, Peters M, von Blomberg BM, Scheper RJ, van der Vliet HJ, van den Eertwegh AJ, Roelvink M, Beijnen J, Zwierzina H, Pinedo HM (2002) A phase I study of the natural killer T-cell ligand alpha-galactosylceramide (KRN7000) in patients with solid tumors. Clin Cancer Res 8:3702–3709
Godfrey DI, MacDonald HR, Kronenberg M, Smyth MJ, Van Kaer L (2004) NKT cells: what’s in a name? Nat Rev Immunol 4:231–237
Grubor-Bauk B, Simmons A, Mayrhofer G, Speck PG (2003) Impaired clearance of herpes simplex virus type 1 from mice lacking CD1d or NKT cells expressing the semivariant V alpha 14-J alpha 281 TCR. J Immunol 170:1430–1434
Hamelin ME, Prince GA, Gomez AM, Kinkead R, Boivin G (2006) Human metapneumovirus infection induces long-term pulmonary inflammation associated with airway obstruction and hyperresponsiveness in mice. J Infect Dis 193:1634–1642
Hammond KJ, Poulton LD, Palmisano LJ, Silveira PA, Godfrey DI, Baxter AG (1998) Alpha/beta-T cell receptor (TCR) + CD4-CD8- (NKT) thymocytes prevent insulin-dependent diabetes mellitus in nonobese diabetic (NOD/Lt) mice by the influence of interleukin (IL)-4 and/or IL-10. J Exp Med 187:1047–1056
Hayakawa Y, Takeda K, Yagita H, Kakuta S, Iwakura Y, Van Kaer L, Saiki I, Okumura K (2001) Critical contribution of IFN-gamma and NK cells, but not perforin-mediated cytotoxicity, to anti-metastatic effect of alpha-galactosylceramide. Eur J Immunol 31:1720–1727
Hayakawa Y, Rovero S, Forni G, Smyth MJ (2003) Alpha-galactosylceramide (KRN7000) suppression of chemical- and oncogene-dependent carcinogenesis. Proc Natl Acad Sci U S A 100:9464–9469
Hayakawa Y, Godfrey DI, Smyth MJ (2004) Alpha-galactosylceramide: potential immunomodulatory activity and future application. Curr Med Chem 11:241–252
Hong S, Wilson MT, Serizawa I, Wu L, Singh N, Naidenko OV, Miura T, Haba T, Scherer DC, Wei J, Kronenberg M, Koezuka Y, Van Kaer L (2001) The natural killer T-cell ligand alpha-galactosylceramide prevents autoimmune diabetes in non-obese diabetic mice. Nat Med 7:1052–1056
Jahng AW, Maricic I, Pedersen B, Burdin N, Naidenko O, Kronenberg M, Koezuka Y, Kumar V (2001) Activation of natural killer T cells potentiates or prevents experimental autoimmune encephalomyelitis. J Exp Med 194:1789–1799
Kawano T, Cui J, Koezuka Y, Toura I, Kaneko Y, Motoki K, Ueno H, Nakagawa R, Sato H, Kondo E, Koseki H, Taniguchi M (1997) CD1d-restricted and TCR-mediated activation of valpha14 NKT cells by glycosylceramides. Science 278:1626–1629
Kim EY, Battaile JT, Patel AC, You Y, Agapov E, Grayson MH, Benoit LA, Byers DE, Alevy Y, Tucker J, Swanson S, Tidwell R, Tyner JW, Morton JD, Castro M, Polineni D, Patterson GA, Schwendener RA, Allard JD, Peltz G, Holtzman MJ (2008) Persistent activation of an innate immune response translates respiratory viral infection into chronic lung disease. Nat Med 14:633–640
King IL, Amiel E, Tighe M, Mohrs K, Veerapen N, Besra G, Mohrs M, Leadbetter EA (2011) Invariant natural killer T cells direct B cell responses to cognate lipid antigen in an IL-21-dependent manner. Nat Immunol 13:44–50
Kinjo Y, Wu D, Kim G, Xing GW, Poles MA, Ho DD, Tsuji M, Kawahara K, Wong CH, Kronenberg M (2005) Recognition of bacterial glycosphingolipids by natural killer T cells. Nature 434:520–525
Kojo S, Seino K, Harada M, Watarai H, Wakao H, Uchida T, Nakayama T, Taniguchi M (2005) Induction of regulatory properties in dendritic cells by Valpha14 NKT cells. J Immunol 175:3648–3655
Kumar H, Belperron A, Barthold SW, Bockenstedt LK (2000) Cutting edge: CD1d deficiency impairs murine host defense against the spirochete, Borrelia burgdorferi. J Immunol 165:4797–4801
MacDonald HR (1995) NK1.1+ T cell receptor-alpha/beta+ cells: new clues to their origin, specificity, and function. J Exp Med 182:633–638
Makino Y, Kanno R, Ito T, Higashino K, Taniguchi M (1995) Predominant expression of invariant Vα14+ TCR α chain in NK1.1+ T cell populations. Int Immunol 7:1157–1161
Mattner J, Debord KL, Ismail N, Goff RD, Cantu C 3rd, Zhou D, Saint-Mezard P, Wang V, Gao Y, Yin N, Hoebe K, Schneewind O, Walker D, Beutler B, Teyton L, Savage PB, Bendelac A (2005) Exogenous and endogenous glycolipid antigens activate NKT cells during microbial infections. Nature 434:525–529
Mendiratta SK, Martin WD, Hong S, Boesteanu A, Joyce S, Van Kaer L (1997) CD1d1 mutant mice are deficient in natural T cells that promptly produce IL-4. Immunity 6:469–477
Meyer EH, Goya S, Akbari O, Berry GJ, Savage PB, Kronenberg M, Nakayama T, DeKruyff RH, Umetsu DT (2006) Glycolipid activation of invariant T cell receptor + NKT cells is sufficient to induce airway hyperreactivity independent of conventional CD4 + T cells. Proc Natl Acad Sci U S A 103:2782–2787
Michel ML, Keller AC, Paget C, Fujio M, Trottein F, Savage PB, Wong CH, Schneider E, Dy M, Leite-de-Moraes MC (2007) Identification of an IL-17-producing NK1.1− iNKT cell population involved in airway neutrophilia. J Exp Med 204:995–1001
Michel ML, Mendes-da-Cruz D, Keller AC, Lochner M, Schneider E, Dy M, Eberl G, Leite-de-Moraes MC (2008) Critical role of ROR-γt in a new thymic pathway leading to IL-17-producing invariant NKT cell differentiation. Proc Natl Acad Sci U S A 105:19845–19850
Morita M, Motoki K, Akimoto K, Natori T, Sakai T, Sawa E, Yamaji K, Koezuka Y, Kobayashi E, Fukushima H (1995) Structure-activity relationship of alpha-galactosylceramides against B16-bearing mice. J Med Chem 38:2176–2187
Motomura Y, Kitamura H, Hijikata A, Matsunaga Y, Matsumoto K, Inoue H, Atarashi K, Hori S, Watarai H, Zhu J, Taniguchi M, Kubo M (2011) The transcription factor E4BP4 regulates the production of IL-10 and IL-13 in CD4+ T cells. Nat Immunol 12:450–459
Natori T, Morita M, Akimoto K, Koezuka Y (1994) Agelasphins, novel antitumor and immunostimulatory cerebrosides from the marine sponge Agelas mauritianus. Tetrahedron 50:2771–2784
Nichols KE, Hom J, Gong SY, Ganguly A, Ma CS, Gong SY, Ganguly A, Ma CS, Cannons JL, Tangye SG, Schwartzberg PL, Koretzky GA, Stein PL (2005) Regulation of NKT cell development by SAP, the protein defective in XLP. Nat Med 11:340–345
Ortaldo JR, Young HA, Winkler-Pickett RT, Bere EW Jr, Murphy WJ, Wiltrout RH (2004) Dissociation of NKT stimulation, cytokine induction, and NK activation in vivo by the use of distinct TCR-binding ceramides. J Immunol 172:943–953
Pasquier B, Yin L, Fondaneche MC, Relouzat F, Bloch-Queyrat C, Lambert N, Fischer A, de Saint-Basile G, Latour S (2005) Defective NKT cell development in mice and humans lacking the adapter SAP, the X-linked lymphoproliferative syndrome gene product. J Exp Med 201:695–701
Pellicci DG, Uldrich AP, Kyparissoudis K, Crowe NY, Brooks AG, Hammond KJ, Sidobre S, Kronenberg M, Smyth MJ, Godfrey DI (2002) A natural killer T (NKT) cell developmental pathway involving a thymus-dependent NK1.1− CD4+ CD1d-dependent precursor stage. J Exp Med 195:835–844
Porcelli S, Yockey CE, Brenner MB, Balk SP (1993) Analysis of T cell antigen receptor (TCR) expression by human peripheral blood CD4-8-alpha/beta T cells demonstrates preferential use of several V beta genes and an invariant TCR alpha chain. J Exp Med 178:1–16
Reynolds JM, Lee YH, Shi Y, Wang X, Angkasekwinai P, Nallaparaju KC, Flaherty S, Chang SH, Watarai H, Dong C (2015) Interleukin-17B antagonizes interleukin-25-mediated mucosal inflammation. Immunity 42:692–703
Rigaud S, Fondaneche MC, Lambert N, Pasquier B, Mateo V, Soulas P, Galicier L, Le Deist F, Rieux-Laucat F, Revy P, Fischer A, de Saint BG, Latour S (2006) XIAP deficiency in humans causes an X-linked lymphoproliferative syndrome. Nature 444:110–114
Roberts TJ, Lin Y, Spence PM, Van Kaer L, Brutkiewicz RR (2004) CD1d1-dependent control of the magnitude of an acute antiviral immune response. J Immunol 172:3454–3461
Sag D, Krause P, Hedrick CC, Kronenberg M, Wingender G (2014) IL-10-producing NKT10 cells are a distinct regulatory invariant NKT cell subset. J Clin Invest 124:3725–3740
Salio M, Silk JD, Jones EY, Cerundolo V (2014) Biology of CD1- and MR1-restricted T cells. Annu Rev Immunol 32:323–366
Schmidt RE, Murray C, Daley JF, Schlossman SF, Ritz J (1986) A subset of natural killer cells in peripheral blood displays a mature T cell phenotype. J Exp Med 164:351–356
Sigurs N, Gustafsson PM, Bjarnason R, Lundberg F, Schmidt S, Sigurbergsson F, Kjellman B (2005) Severe respiratory syncytial virus bronchiolitis in infancy and asthma and allergy at age 13. Am J Respir Crit Care Med 171:137–141
Singh AK, Wilson MT, Hong S, Olivares-Villagómez D, Du C, Stanic AK, Joyce S, Sriram S, Koezuka Y, Van Kaer L (2001) Natural killer T cell activation protects mice against experimental autoimmune encephalomyelitis. J Exp Med 194:1801–1811
Smyth MJ, Crowe NY, Pellicci DG, Kyparissoudis K, Kelly JM, Takeda K, Yagita H, Godfrey DI (2002a) Sequential production of interferon-gamma by NK1.1(+) T cells and natural killer cells is essential for the antimetastatic effect of alpha-galactosylceramide. Blood 99:1259–1266
Smyth MJ, Crowe NY, Takeda K, Yagita H, Godfrey DI (2002b) NKT cells – conductors of tumor immunity? Curr Opin Immunol 14:165–171
Sriram V, Du W, Gervay-Hague J, Brutkiewicz RR (2005) Cell wall glycosphingolipids of Sphingomonas paucimobilis are CD1d-specific ligands for NKT cells. Eur J Immunol 35:1692–1701
Terashima A, Watarai H, Inoue S, Sekine E, Nakagawa R, Hase K, Iwamura C, Nakajima H, Nakayama T, Taniguchi M (2008) A novel subset of mouse NKT cells bearing the IL-17 receptor B responds to IL-25 and contributes to airway hyperreactivity. J Exp Med 205:2727–2733
Watarai H, Fujii S, Yamada D, Rybouchkin A, Sakata S, Nagata Y, Iida-Kobayashi M, Sekine-Kondo E, Shimizu K, Shozaki Y, Sharif J, Matsuda M, Mochiduki S, Hasegawa T, Kitahara G, Endo TA, Toyoda T, Ohara O, Harigaya K, Koseki H, Taniguchi M (2010) Murine induced pluripotent stem cells can be derived from and differentiate into natural killer T cells. J Clin Invest 120:2610–2618
Watarai H, Yamada D, Fujii S, Taniguchi M, Koseki H (2012a) Induced pluripotency as a potential path towards iNKT cell-mediated cancer immunotherapy. Int J Hematol 95:624–631
Watarai H, Sekine-Kondo E, Shigeura T, Motomura Y, Yasuda T, Satoh R, Yoshida H, Kubo M, Kawamoto H, Koseki H, Taniguchi M (2012b) Development and function of invariant natural killer T cells producing Th2- and Th17-cytokines. PLoS Biol 10, e1001255
Weaver CT, Hatton RD, Mangan PR, Harrington LE (2007) IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol 25:821–852
Wieland Brown LC, Penaranda C, Kashyap PC, Williams BB, Clardy J, Kronenberg M, Sonnenburg JL, Comstock LE, Bluestone JA, Fischbach MA (2013) Production of α-galactosylceramide by a prominent member of the human gut microbiota. PLoS Biol 11, e1001610
Wu K, Byers DE, Jin X, Agapov E, Alexander-Brett J, Patel AC, Cella M, Gilfilan S, Colonna M, Kober DL, Brett TJ, Holtzman MJ (2015) TREM-2 promotes macrophage survival and lung disease after respiratory viral infection. J Exp Med 212:681–697
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Japan
About this chapter
Cite this chapter
Watarai, H. (2016). Elucidation and Control of the Mechanisms Underlying Chronic Inflammation Mediated by Invariant Natural Killer T Cells. In: Miyasaka, M., Takatsu, K. (eds) Chronic Inflammation. Springer, Tokyo. https://doi.org/10.1007/978-4-431-56068-5_27
Download citation
DOI: https://doi.org/10.1007/978-4-431-56068-5_27
Published:
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-56066-1
Online ISBN: 978-4-431-56068-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)