Abstract
Trochleoplasty is a safe and effective surgical technique for addressing trochlear dysplasia in the setting of patellar instability. The following chapter discusses the strict clinical and radiographic indications for deepening trochleoplasty including a discussion of when other associated procedures should be performed simultaneously. This chapter also includes an up-to-date review of the available literature regarding outcomes of contemporary trochleoplasty techniques.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Deepening trochleoplasty
- Dejour trochleoplasty
- Bereiter trochleoplasty
- Goutallier recession wedge
- Indications
- Outcomes
- Results
- Patellar instability
- MPFL
25.1 Indications
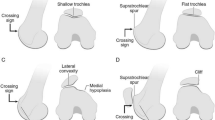
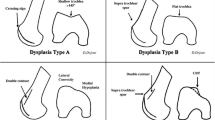
Correct patient selection is critical to the success of the deepening trochleoplasty procedure. Furthermore, the treating surgeon must have a complete understanding of the type and severity of the dysplasia as well as associated anatomic risk factors (ARFs) such as patella alta or a lateralized tibial tuberosity. It must be emphasized that the mere presence of type A dysplasia, a shallow groove, is not an indication for a deepening trochleoplasty (Fig. 25.1).
The most commonly utilized indications for trochleoplasty include persistent patellar instability refractory to conservative treatment in patients with a Dejour type B or D trochlea and a spur height >5–7 mm and a “J sign” on physical exam [1,2,3,4,5]. The key feature of Dejour type B and D dysplasia is the presence of the large supratrochlear spur (Figs. 25.2 and 25.3). This spur can act in a manner analogous to a “ski ramp.” As the knee moves into flexion, the patella will encounter the spur producing lateral subluxation or dislocation. Additionally, biomechanical testing has demonstrated that Dejour B, C, and D types have increased chondral contact pressures, decreased contact areas, and altered kinematics with resultant patellar instability [6]. High-grade dysplasia (greater than type B) is also associated with increasing patellofemoral arthrosis severity [7, 8].
The presence of a “J sign,” an indicator of pathological patellar tracking, is a critical finding on physical examination as it demonstrates the influence of the spur on the patella [9, 10]. The “J sign” occurs when the knee is actively brought from flexion to extension and the patella demonstrates a sudden lateral translation as it exits the trochlear groove when approaching full extension (Video 25.1). This physical examination finding reflects either trochlea dysplasia, patella alta, or a combination of both. In patients with a spur (Dejour B or D dysplasia), the J sign is seen when the patella “jumps” or “falls” off the supratrochlear spur. A large or tall supratrochlear spur often leads to a more dramatic J sign.
The importance of the supratrochlear spur as a requirement for deepening trochleoplasty cannot be overstated. A primary objective of trochleoplasty is removing the spur and bringing the height of the trochlea to a level flush with the anterior femoral cortex. In our experience, the presence of patella alta (patellotrochlear index (PTI) <0.20 or Caton–Deschamps (CD) index >1.4) lowers the surgical threshold to perform a deepening trochleoplasty (Fig. 25.4). The presence of patella alta magnifies the effect of the supratrochlear spur as the patella must navigate past this spur each time the knee flexes. These knees typically have the most pronounced J signs on exam.
Trochleoplasty should also be strongly considered in revision procedures in patients with Dejour B and D dysplasia who have previously undergone an isolated soft tissue procedure. Multiple studies have demonstrated an association between the severity of a patient’s trochlear dysplasia and their clinical outcome and risk of persistent instability following an isolated soft tissue procedure such as a medial patellofemoral ligament (MPFL) reconstruction [11,12,13,14]. Hopper et al. noted in their series of 68 patients treated with MPFL reconstruction that all 7 of the patients with Dejour C or D dysplasia had recurrent or persistent instability [15]. More recently, Balcarek et al. performed a systematic review of published studies of patients with high-grade trochlear dysplasia treated with (1) medial patellofemoral ligament reconstruction or (2) combined trochleoplasty and extensor apparatus balancing [16]. The MPFL group was comprised of four studies with a total of 210 patients (221 knees), and the trochleoplasty group was comprised of six studies with a total of 164 patients (186 knees). The rate of persistent instability was 7% in the isolated MPFL group compared to 2.1% in the trochleoplasty group.
Contraindications to trochleoplasty include advanced patellofemoral arthrosis [1, 17]. Attempts to perform a deepening trochleoplasty in the presence of brittle, eburnated, and sclerotic trochlear bone can lead to fracture propagation when attempting to cut the osteochondral shell with a scalpel or osteotome. This could potentially result in unstable fragments that might require additional fixation or alterations to postoperative rehabilitation. Likewise, if using the “thin-flap” technique, the flap may be too brittle and stiff to reshape properly.
Open physes or skeletal immaturity is also frequently cited as contraindication to trochleoplasty due to the risk of growth arrest or angular abnormality [17]. Interestingly, Nelitz et al. performed thin-flap trochleoplasty in 18 adolescents with less than 2 years of growth remaining [18]. There were no cases of growth arrest or disturbance and no episodes of recurrent instability. Additional research, however, is necessary before trochleoplasty can be advocated in skeletally immature patients, and caution should be exercised.
25.1.1 Associated Procedures
Trochleoplasty is frequently combined with other procedures in order to fully address the multifactorial contributors to patellar instability. In patients with chronic patellar instability, the medial soft tissue restraints, including the MPFL, are disrupted or attenuated. For this reason, a MPFL reconstruction should be included with any trochleoplasty procedure [1, 17].
Patella alta is a critical ARF for patellar instability and frequently coexists with trochlear dysplasia [19, 20]. Patella alta promotes instability, as the patella does not engage the trochlear groove until deeper degrees of flexion. A distalizing tibial tubercle osteotomy may be considered in patients with a CD >1.3, a PTI <0.32, or a sagittal patellofemoral engagement (SPE) ratio <0.45 [21, 22]. Given the additional morbidity of a distalizing osteotomy, most surgeons (including the authors) generally accept a CD ratio >1.4 or a PTI <20% as an indication. It is important to note that distalizing the patella will help minimize the influence of the supratrochlear spur in producing lateral patellar dislocation as the patella is brought further into the groove, thereby avoiding the effects of the spur proximally. This is especially important in “borderline cases” with significant patella alta (CD ratio of >1.4) and a smaller spur (≤5 mm). Distalizing the tubercle in these cases will effectively diminish the effect of the spur and potentially obviate the need for trochleoplasty.
In patients who also have genu valgum or a laterally positioned tibial tuberosity, the effect of trochlear dysplasia is magnified. The combination of all three anatomic risk factors (trochlear dysplasia, patella alta, and elevated tibial tubercle-trochlear groove (TT-TG) distance) is especially problematic for surgical success unless all three are addressed. An elevated TT-TG >20 mm is often utilized as criteria for a bony procedure such as a tibial tubercle osteotomy (TTO) [23, 24]. Measurement of TT-TG is most accurate using CT, as MRI can underestimate this measurement by up to 4 mm [25]. A trochleoplasty may obviate the need for TTO in patients with type B or D dysplasia and borderline elevated TT-TG. In our experience, the new trochlear groove achieved during trochleoplasty can be created up to 3–5 mm laterally, thus effectively decreasing the TT-TG distance. Schöttle et al. noted an even more dramatic improvement in TT-TG (20 mm preoperatively to 9.9 mm postoperatively) in 19 patients treated with a thin-shell technique [26]. Thus, in the setting of a trochleoplasty, a more elevated TT-TG of >23 mm may be considered a threshold for TTO given that a minimally elevated TT-TG (18–22 mm) can be normalized by lateralization of the newly created trochlear sulcus (Video 25.2).
25.2 Results
Results following trochleoplasty are generally positive but demonstrate some variability which may be attributed to the heterogeneity of the patient population afflicted with patellar instability and the resulting complex surgical decision-making. Since trochleoplasty is relatively rarely performed, no current level I trials investigating trochleoplasty exist. Therefore, the available studies are smaller case series with short- and midterm follow-up which can be challenging to use as a basis for clinical decision-making due to the varying combinations of procedures utilized. A recent consensus statement was issued by a work group jointly funded by the American Orthopedic Society for Sports Medicine (AOSSM) and the Patellofemoral Foundation (PFF) that based its recommendations largely on expert opinion and basic anatomic principles in the absence of high-level evidence in the literature [27]. Nevertheless, a review of published outcomes reveals several trends to guide practice and helps lay a foundation for future investigations.
25.2.1 Biomechanical Studies
Biomechanical and anatomic studies have examined the effect of trochlear dysplasia on patellofemoral biomechanics and demonstrated the effectiveness of trochleoplasty through normalizing patellofemoral kinematics and reducing instability in cadaveric specimens. Van Haver et al. designed a study with four cadaveric knees and implanted in them a series of 3D-printed trochleas with typical dysplasia patterns (based on Dejour’s classification scheme) to objectively demonstrate increased contact pressures, increased lateral translation, and decreased stability in trochlear dysplasia compared to normal [6]. Amis et al. used fresh frozen cadaver knees to simulate patellar instability by surgically raising the central anterior trochlear which significantly increased the degree of instability in their model [28]. The researchers then subsequently performed a trochleoplasty procedure and recorded a decrease in instability. An anatomic study was performed by Fucentese et al., who utilized CT scans before and after sulcus-deepening trochleoplasty in 14 patients (17 knees) to demonstrate the improvement in key anatomic features [29]. The trochlear groove was lateralized by a mean of 6.1 mm proximally and 2.5 mm distally, while the patella medialized 5 mm. The depth of the groove also improved, increasing by 5.9 mm proximally and 2.8 mm distally. Others have assessed the sulcus angle pre- and postoperative with a reported correction from 12° to 39° following deepening trochleoplasty [30,31,32].
25.2.2 Clinical Results
There has been a growing body of literature investigating the clinical outcomes of trochleoplasty with some early results being unfavorable. Surgical techniques have evolved over time, and some techniques have fallen out of favor with contemporary authors. Albee was the first to propose a trochleoplasty technique in 1915 that involved elevating the lateral aspect of the trochlea with a bone wedge graft in an attempt to restore normal patellar tracking [33]. This technique was later found to significantly increase patellofemoral contact pressures and subsequently observed to result in poor outcomes [34, 35]. Masse then introduced the concept of the deepening trochleoplasty in 1978 which involved removing subchondral bone and molding the articular cartilage with direct impaction using a mallet [36]. A study by Rouanet presented 15-year results for this technique in 34 patients which showed a 20% failure rate, including seven patients that subsequently underwent arthroplasty procedure during the study period due to advanced patellofemoral arthritis [37]. Despite this high failure rate, the authors reported no recurrent instability episodes in their 34 patients.
Techniques have been further refined to include the three most commonly performed techniques today: (1) the Lyon procedure, i.e., Dejour or “thick shell,” (2) the Bereiter procedure or “thin flap” and its arthroscopic modification, and (3) the Goutallier recession wedge [1, 38,39,40]. The outcomes of these modern techniques have been more promising, and the following reviewed articles focus on these contemporary techniques. The inclusion (or exclusion) of other patellofemoral-stabilizing procedures continues to be confounding as the indications for these procedures are not universally agreed upon, and consequently the outcomes must be interpreted in light of these differences.
25.2.3 Lyon Technique
The Lyon technique involves creating a thick, 3–5 mm osteochondral shell by undermining the trochlea with a high-speed burr to remove the subchondral bone. An osteotomy is then created using an osteotome or scalpel forming two “shingles” which are fixed in place using small anchors and absorbable sutures draped over each side to form a new trochlea [1]. A large level IV study utilizing the Lyon technique was published by McNamara et al., including 90 patients (107 knees) with severe dysplasia who underwent deepening trochleoplasty in addition to other indicated stabilizing procedures [41]. In this cohort, 83% of patients reported satisfaction with their outcome. At 6-year follow-up, there where were significant improvements in Kujala scores from a mean of 63 to 84 postoperatively (p < 0.05). Of note, only 14 (13.1%) of patients had MPFL reconstruction at the index procedure, and the authors reported 21 knees (19.6%) required a second operation for recurrent instability including 10 MPFL reconstructions, 2 of which were revisions. These results suggest that additional patellar stabilization procedures should be considered at the index procedure, which has been suggested by other authors [1, 17]. In their series, sports participation increased from 40% to 67% at final follow-up.
In another level IV study, Ntagiopoulous et al. presented midterm results on 31 knees treated with trochleoplasty for high-grade dysplasia and demonstrated substantial improvements from preoperative Kujala and IKDC scores [32]. Radiographically, their mean sulcus angles improved, and there were no reported postoperative patellar dislocations reported although a patellar apprehension sign was found on exam in six knees.
Other smaller series include Dejour et al. which examined 22 patients (24 knees). The authors showed no instability events and improvements in Kujala score from 44 to 81 over a mean follow-up period of 66 months. Pain decreased in 72% of patients, and the apprehension sign was negative in 75%. No patients exhibited evidence of osteoarthritis during the study period [30].
25.2.4 Bereiter Technique
The Bereiter technique involves making a thin osteochondral flap or “flake” 2–3 mm thick, which is elevated from the anterior femur using a burr or a combination of osteotomes. A bony sulcus is fashioned beneath the flap to create depth, and the flap is molded into the new sulcus and fixed with absorbable sutures and anchors draped over the top [38]. The largest published study on trochleoplasty to date comes from Metcalf et al., who reported on prospectively collected outcomes in 199 Bereiter trochleoplasties performed on 173 patients over a study period of 12 years [42]. Their mean follow-up time was 4.4 years (range 1–12). Eighty-eight percent of patients were satisfied with their outcomes, 90% of patients said their symptoms had resolved, and 73.6% had returned to sport. Kujala scores increased from 51.5 to 82.5 and IKDC scores increased from 44.3 to 71.3. Sixteen patients experienced a recurrent patellar dislocation, for an 8.3% recurrence rate. Twenty-seven patients (14%) underwent reoperations, nine of which were MPFL reconstructions. During the course of the investigation, it appears that the authors began to augment trochleoplasty with soft tissue balancing procedure which resulted in a changing incidence of subsequent MPFL reconstructions. Four of the first 40 patients underwent later MPFL compared to 5 of the next 174. In their cohort, patients were less likely to be satisfied if they had undergone a previous procedure. Of knees with X-rays more than 5 years after surgery, 7.7% demonstrated evidence of OA. In a subgroup analysis of 29 patients who had had been seen more than 8 years following surgery, the results appeared durable with Kujala scores of 83 and IKDC scores of 79.
Utting reported on prospectively collected outcomes for 59 knees undergoing deepening trochleoplasty with 2-year follow-up and showed improvement in all scores: Kujala, IKDC, Lysholm, Oxford, and WOMAC [43]. Nelitz et al. reported minimum 2-year follow-up on 23 patients (26 knees) who underwent combined trochleoplasty and MPFL reconstruction and showed no recurrent dislocations, Kujala score improvement from 79 to 96, IKDC improvement from 74 to 90, and VAS improvement from 3 to 1, with 95.7% of patients being satisfied with their outcome [44].
Isolated trochleoplasty was investigated by Camathias et al. in 44 adolescent patients (50 knees) with recurrent instability and trochlear dysplasia [45]. The authors reported improvements in both outcome measures and exam findings; Kujala scores increased from 71 to 92, and Lysholm scores increased from 71 to 95, while the J sign disappeared in 39 of 45, and apprehension disappeared in 33 of 41. A single redislocation was observed at 38 months. This patient group was reported to have no torsional or axial malalignment.
25.2.5 Goutallier Technique
The Goutallier technique involves an osteotomy to remove a section of bone from beneath the trochlea with a distal osteochondral hinge that is closed and secured with two or three 3.5 mm cancellous screws. The primary goal is to decrease the height of the supratrochlear spur rather than reposition or deepen the trochlear groove [40, 46]. Results from Thaunat et al. demonstrated that functional results trended toward, but did not reach, statistical significance in their study of 17 patients (19 knees) with a mean of 34 months of follow-up [40]. The supratrochlear spur height was decreased from a mean of 4.8 to −0.8 mm, and two patients experienced recurrent instability.
25.2.6 Systematic Reviews
Systematic reviews have also been conducted to compare available literature including a 2017 review of 15 articles regarding trochleoplasty for patellofemoral instability which showed improvements in Kujala scores (from 61.4 to 80.8), Lysholm scores (from 55.5 to 78.5), and a low redislocation rate of 2% [47]. Longo et al. published a systematic review that encompassed 392 knees in 371 patients across 14 articles [48]. Interestingly, this review also stratified which technique was performed including Bereiter, Dejour, or Goutallier recession trochleoplasty. All resulted in significant improvements in instability and outcome scores in the majority of patients. The Bereiter technique was associated with the lowest rate of recurrence and least range of motion deficiency. The Dejour technique was associated with the highest mean Kujala scores postoperatively. The authors concluded that there was no superiority for one technique over the others.
25.2.7 Our Results
At our institution, we have had high satisfaction rates following deepening trochleoplasty. A total of 64 patients (71 knees) with severe trochlear dysplasia were prospectively enrolled from 2011 to 2017. All patients underwent sulcus-deepening trochleoplasty using the Dejour, thick shell osteotomy method. All patients in our study underwent concomitant procedures during their trochleoplasty which included MPFL reconstruction (100%), lateral release or lengthening (50.7%), tibial tubercle osteotomy (32.8%), and some type of cartilage procedure (41.8%) to include shaving chondroplasty (38.8%), chondral allograft (10.4%), microfracture (7.5%), and/or removal of loose body (22.4%).
The majority of patients were female (81.6%) with a mean age of 19.6 ± 6.8 years. Follow-up ranged from 12 months to 78.4 months (mean 27.7 ± 15.4). All knees were either Dejour B (81.3%) or D (18.8%) with a mean Caton–Deschamps index (CDI) of 1.20 (±0.2). Mean spur height preoperatively was 7.41 mm (±1.84 mm) with a mean trochlear depth of only −0.18 mm (±2.71). Mean patellotrochlear index (PTI) was 0.41 (±0.41).
Most importantly, there were zero episodes of recurrent instability. One patient had patellar apprehension and a recurrent J sign at terminal extension postoperatively requiring a distal femoral osteotomy for genu valgum. All patients reported clinically significant improvements compared with baseline preoperative outcome scores. The mean preoperative IKDC score improved from 49.99 to 79.86 (p < 0.001), and the mean preoperative Kujala score improved from 55.88 to 85.80 (p < 0.001). Patients reported high satisfaction rates (mean 9.5 ± 1.6 out of 10). All but one patient (96.9%) returned to work, while 88.2% of patients were able to return to sport. Ten knees (20.4%) developed arthrofibrosis and required manipulation under anesthesia, while eight of which underwent simultaneous arthroscopic lysis of adhesions. No patients had fixation failure or progression of arthritis noted on yearly X-rays.
25.2.8 Complications
Trochleoplasty is a technically challenging procedure that requires a three-dimensional understanding of the problem and steps to correction. Even with the most thoughtful approach, complications can and do occur. Injury to the articular cartilage, over- or under-correction, and recurrent instability have all been reported. An overall complication rate of 13.4% has been reported by Song et al. following trochleoplasty [3]. This includes a 6.8% of redislocation and 65.9% with increased patellofemoral pain. Nearly 8% went on to develop progression of patellofemoral joint arthritis at mean follow-up of 69.9 months [3].
The primary complication reported after trochleoplasty is stiffness, which has been reported as high as 46% of knees [3, 37, 41, 49]. More recent investigations have estimated a 0–20% incidence of stiffness which is more in line with our unpublished findings above [32, 50, 51]. In many cases, patients suffering from stiffness return to the operating room for manipulation under anesthesia (MUA) and/or arthroscopic lysis of adhesions (LOA). Several sources have identified satisfactory return to function after LOA/MUA [49, 50, 52]. During arthroscopic LOA, these knees have been described as having massive periarticular scar formation, and it has been proposed that early passive range of motion exercises may reduce the incidence of this complication [53]. Our experience echoes this with more stiffness noted early in our learning curve when our postoperative rehab protocol was more conservative. Emphasizing early motion has reduced the need for LOA/MUA for us dramatically.
A concerning potential complication of trochleoplasty is subchondral collapse. There are risk of direct damage to the articular cartilage while osteotomizing the osteochondral shell and also risk of indirect damage to the articular cartilage while burring beneath the surface due to thermal injury or over-resection of subchondral bone. Use of a measured offset guide with the burr or drill bit adds a measure of safety as well as reproducibility in our experience and seems to reduce this risk.
In many cases, patients undergoing trochleoplasty have already sustained multiple patella dislocations leading to a high burden of existing chondral damage and the possibility of postoperative patellofemoral pain despite surgical correction [31]. This complication has been observed in 10–14% of patients [48]. Despite radiographic improvement of the patellofemoral joint, it is possible that trochleoplasty may not be protective against progression of osteoarthritis. While osteoarthritis rates have been reported as low as 0–7%, the lack of long-term results of the contemporary techniques leaves this question unresolved [30, 42, 48].
25.3 Conclusion
Current literature and expert opinion favors trochleoplasty as an effective option to surgically treat patellofemoral instability in carefully selected patients. There is no consensus as to which technique should be employed; however, it is our opinion that whichever technique is chosen should be used in conjunction with MPFL reconstruction in all cases and paired with additional bony reconstructive procedures if indicated. With the lack of any large-scale randomized series, more efforts are needed to better understand this challenging clinical problem and the appropriate application of these surgical techniques.
References
Dejour D, Saggin P. The sulcus deepening trochleoplasty—the Lyon’s procedure. Int Orthop. 2010;34(2):311–6.
Fucentese SF, Zingg PO, Schmitt J, Pfirrmann CWA, Meyer DC, Koch PP. Classification of trochlear dysplasia as predictor of clinical outcome after trochleoplasty. Knee Surg Sports Traumatol Arthrosc. 2011;19(10):1655–61.
Song GY, Hong L, Zhang H, Zhang J, Li X, Li Y, et al. Trochleoplasty versus nontrochleoplasty procedures in treating patellar instability caused by severe trochlear dysplasia. Arthroscopy. 2014;30(4):523–32.
Weber AE, Nathani A, Dines JS, Allen AA, Shubin-Stein BE, Arendt EA, et al. An algorithmic approach to the management of recurrent lateral patellar dislocation. J Bone Joint Surg. 2016;98(5):417–27.
Carstensen SE, Menzer HM, Diduch DR. Patellar instability: when is trochleoplasty necessary? Sports Med Arthrosc. 2017;25(2):92–9.
Van Haver A, De Roo K, De Beule M, Labey L, De Baets P, Dejour D, et al. The effect of trochlear dysplasia on patellofemoral biomechanics: a cadaveric study with simulated trochlear deformities. Am J Sports Med. 2015;43(6):1354–61.
Grelsamer RP, Dejour D, Gould J. The pathophysiology of patellofemoral arthritis. Orthop Clin North Am. 2008;39(3):269–74.
Dejour D, Le Coultre B. Osteotomies in patello-eemoral instabilities. Sports Med Arthrosc Rev. 2018;26(1):8–15.
Post WR. Clinical evaluation of patients with patellofemoral disorders. Arthroscopy. 1999;15(8):841–51.
Johnson LL, Van Dyk GE, Green JR, Pittsley AW, Bays B, Gully SM, et al. Clinical assessment of asymptomatic knees: comparison of men and women. Arthroscopy. 1998;14(4):347–59.
Hiemstra LA, Kerslake S, Lafave MR. Quality-of-life outcomes of patients following patellofemoral stabilization surgery: the influence of trochlear dysplasia. J Knee Surg. 2017;30(9):887–93.
Hiemstra LA, Kerslake S, Heard M, Buchko G, Lafave M. Outcomes of surgical stabilization in patients with combined ACL deficiency and patellofemoral instability—a case series. Knee. 2016;23(6):1106–11.
Wagner D, Pfalzer F, Hingelbaum S, Huth J, Mauch F, Bauer G. The influence of risk factors on clinical outcomes following anatomical medial patellofemoral ligament (MPFL) reconstruction using the gracilis tendon. Knee Surg Sports Traumatol Arthrosc. 2013;21(2):318–24.
Kita K, Tanaka Y, Toritsuka Y, Amano H, Uchida R, Takao R, et al. Factors affecting the outcomes of double-bundle medial patellofemoral ligament reconstruction for recurrent patellar dislocations evaluated by multivariate analysis. Am J Sports Med. 2015;43(12):2988–96.
Hopper GP, Leach WJ, Rooney BP, Walker CR, Blyth MJ. Does degree of trochlear dysplasia and position of femoral tunnel influence outcome after medial patellofemoral ligament reconstruction? Am J Sports Med. 2014;42(3):716–22.
Balcarek P, Rehn S, Howells NR, Eldridge JD, Kita K, Dejour D, et al. Results of medial patellofemoral ligament reconstruction compared with trochleoplasty plus individual extensor apparatus balancing in patellar instability caused by severe trochlear dysplasia: a systematic review and meta-analysis. Knee Surg Sports Traumatol Arthrosc. 2017;25(12):3869–77.
LaPrade RF, Cram TR, James EW, Rasmussen MT. Trochlear dysplasia and the role of trochleoplasty. Clin Sports Med. 2014;33(3):531–45.
Nelitz M, Dreyhaupt J, Williams SRM. No growth disturbance after trochleoplasty for recurrent patellar dislocation in adolescents with open growth plates. Am J Sports Med. 2018;48(13):3209–16.
Dejour H, Walch G, Nove-Josserand L, Guier C. Factors of patellar instability: an anatomic radiographic study. Knee Surg Sports Traumatol Arthrosc. 1994;2(1):19–26.
Steensen RN, Bentley JC, Trinh TQ, Backes JR, Wiltfong RE. The prevalence and combined prevalences of anatomic factors associated with recurrent patellar dislocation: a magnetic resonance imaging study. Am J Sports Med. 2015;43(4):921–7.
Biedert RM, Albrecht S. The patellotrochlear index: a new index for assessing patellar height. Knee Surg Sports Traumatol Arthrosc. 2006;14(8):707–12.
Dejour D, Ferrua P, Ntagiopoulos PG, Radier C, Hulet C, Rémy F, et al. The introduction of a new MRI index to evaluate sagittal patellofemoral engagement. Orthop Traumatol Surg Res. 2013;99(8):S391–8.
Caton JH, Dejour D. Tibial tubercle osteotomy in patello-femoral instability and in patellar height abnormality. Int Orthop. 2010;34(2):305–9.
Anley CM, Morris GV, Saithna A, James SL, Snow M. Defining the role of the tibial tubercle-trochlear groove and tibial tubercle-posterior cruciate ligament distances in the work-up of patients with patellofemoral disorders. Am J Sports Med. 2015;43(6):1348–53.
Camp CL, Bond JR, Collins MS, Stuart MJ, Krych AJ, Levy BA, et al. CT and MRI measurements of tibial tubercle to trochlear groove distances (TT-TG) are not equivalent. Orthop J Sport Med. 2013;41(8):1835–40.
Schöttle PB, Fucentese SF, Pfirrmann C, Bereiter H, Romero J. Trochleaplasty for patellar instability due to trochlear dysplasia: a minimum 2-year clinical and radiological follow-up of 19 knees. Acta Orthop. 2005;76(5):693–8.
Post WR, Fithian DC. Patellofemoral instability: a consensus statement from the AOSSM/PFF patellofemoral instability workshop. Orthop J Sport Med. 2018;6(1):1–5.
Amis AA, Oguz C, Bull AMJ, Senavongse W, Dejour D. The effect of trochleoplasty on patellar stability and kinematics: a biomechanical study in vitro. J Bone Joint Surg. 2008;90(7):864–9.
Fucentese SF, Schöttle PB, Pfirrmann CWA, Romero J. CT changes after trochleoplasty for symptomatic trochlear dysplasia. Knee Surg Sports Traumatol Arthrosc. 2007;15(2):168–74.
Dejour D, Byn P, Ntagiopoulos PG. The Lyon’s sulcus-deepening trochleoplasty in previous unsuccessful patellofemoral surgery. Int Orthop. 2013;37(3):433–9.
Faruqui S, Bollier M, Wolf B, Amendola N. Outcomes after trochleoplasty. Iowa Orthop J. 2012;32:195–206.
Ntagiopoulos PG, Byn P, Dejour D. Midterm results of comprehensive surgical reconstruction including sulcus-deepening trochleoplasty in recurrent patellar dislocations with high-grade trochlear dysplasia. Am J Sports Med. 2013;41(5):998–1004.
Albee F. The bone graft wedge in the treatment of habitual dislocation of the patella. Med Rec. 1915;88:257–9.
Tigchelaar S, van Sambeeck J, Koeter S, van Kampen A. A stand-alone lateral condyle-elevating trochlear osteotomy leads to high residual instability but no excessive increase in patellofemoral osteoarthritis at 12-year follow-up. Knee Surg Sports Traumatol Arthrosc. 2018;26(4):1216–22.
Kuroda R, Kambic H, Valdevit A, Andrish J. Distribution of patellofemoral joint pressures after femoral trochlear osteotomy. Knee Surg Sports Traumatol Arthrosc. 2002;10(1):33,037.
Masse Y. Trochleoplasty. Restoration of the intercondylar groove in subluxations and dislocations of the patella. Rev Chir Orthop Reparatrice Appar Mot. 1978;64(1):3–17.
Rouanet T, Gougeon F, Fayard JM, Rémy F, Migaud H, Pasquier G. Sulcus deepening trochleoplasty for patellofemoral instability: a series of 34 cases after 15 years postoperative follow-up. Orthop Traumatol Surg Res. 2015;101(4):443–7.
Metcalfe AJ, Clark DA, Kemp M, Eldridge JD. The Bristol experience of the Bereiter trochleoplasty: indications and technique. Oper Tech Sports Med. 2015;23(2):123–8.
Blond L, Schöttle P. Arthroscopic deepening trochleoplasty for patella instability. In: Patellofemoral pain, instability, and arthritis: clinical presentation, imaging, and treatment. Berlin Heidelberg: Springer; 2010. p. 233–6.
Thaunat M, Bessiere C, Pujol N, Boisrenoult P, Beaufils P. Recession wedge trochleoplasty as an additional procedure in the surgical treatment of patellar instability with major trochlear dysplasia: early results. Orthop Traumatol Surg Res. 2011;97(8):833–45.
McNamara I, Bua N, Smith TO, Ali K, Donell ST. Deepening trochleoplasty with a thick osteochondral flap for patellar instability. Am J Sports Med. 2015;43(11):2706–13.
Metcalfe AJ, Clark DA, Kemp MA, Eldridge JD. Trochleoplasty with a flexible osteochondral flap. Bone Joint J. 2017;99B(3):344–50.
Utting MR, Mulford JS, Eldridge JDJ. A prospective evaluation of trochleoplasty for the treatment of patellofemoral dislocation and instability. J Bone Joint Surg Br. 2008;90(2):180–5.
Nelitz M, Dreyhaupt J, Lippacher S. Combined trochleoplasty and medial patellofemoral ligament reconstruction for recurrent patellar dislocations in severe trochlear dysplasia: a minimum 2-year follow-up study. Am J Sports Med. 2013;41:1005.
Camathias C, Studer K, Kiapour A, Rutz E, Vavken P. Trochleoplasty as a solitary treatment for recurrent patellar dislocation results in good clinical outcome in adolescents. Am J Sports Med. 2016;44(11):2855–63.
Goutallier D, Raou D, Van Driessche S. Retro-trochlear wedge reduction trochleoplasty for the treatment of painful patella syndrome with protruding trochleae. Technical note and early results. Rev Chir Orthop Reparatrice Appar Mot. 2002;88(7):678–85.
Testa EA, Camathias C, Amsler F, Henle P, Friederich NF, Hirschmann MT. Surgical treatment of patellofemoral instability using trochleoplasty or MPFL reconstruction: a systematic review. Knee Surg Sports Traumatol Arthrosc. 2017;25(8):2309–20.
Longo UG, Vincenzo C, Mannering N, Ciuffreda M, Salvatore G, Berton A, et al. Trochleoplasty techniques provide good clinical results in patients with trochlear dysplasia. Knee Surg Sports Traumatol Arthrosc. 2018;26:2640–58.
Verdonk R, Jansegers E, Stuyts B. Trochleoplasty in dysplastic knee trochlea. Knee Surg Sports Traumatol Arthrosc. 2005;13(7):529–33.
Donell ST, Joseph G, Hing CB, Marshall TJ. Modified Dejour trochleoplasty for severe dysplasia: operative technique and early clinical results. Knee. 2006;13(4):266–73.
von Knoch F. Trochleaplasty for recurrent patellar dislocation in association with trochlear dysplasia: a 4 to 14-year follow-up study. J Bone Joint Surg Br. 2006;88(10):1331–5.
Beaufils P, Thaunat M, Pujol N, Scheffler S, Rossi R, Carmont M. Trochleoplasty in major trochlear dysplasia: current concepts. Sport Med Arthrosc Rehabil Ther Technol. 2012;4:7. https://doi.org/10.1186/1758-2555-4-7.
Diduch DR, Kandil A, Burrus MT. Lateral patellar instability in the skeletally mature patient: evaluation and surgical management. J Am Acad Orthop Surg. 2018;26(12):429–39.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
25.1 Electronic Supplementary Materials
Intra-articular view from a superolateral arthroscopic portal. As the knee moves from extension to flexion, the patella begins in a dislocated position and then abruptly reduces into the trochlea, generating the “J sign” described on physical examination. Also shown is the characteristic wear pattern of the lateral trochlea and patella due to chronic mal-tracking (MP4 14411 kb)
This video demonstrates the senior author’s preferred technique for deepening trochleoplasty. This technique mirrors the methods described by Dejour et al. and demonstrates a guide which is adjustable and in this case set to maintain a shell thickness of 5 mm using the burr [1] (MP4 475040 kb)
Rights and permissions
Copyright information
© 2020 ESSKA
About this chapter
Cite this chapter
Smith, J.M., Rush, J.K., Carstensen, S.E., Diduch, D.R. (2020). Trochleoplasty: Indications and Results. In: Dejour, D., Zaffagnini, S., Arendt, E., Sillanpää, P., Dirisamer, F. (eds) Patellofemoral Pain, Instability, and Arthritis. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-61097-8_25
Download citation
DOI: https://doi.org/10.1007/978-3-662-61097-8_25
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-61096-1
Online ISBN: 978-3-662-61097-8
eBook Packages: MedicineMedicine (R0)