Abstract
Mesophotic coral ecosystems (MCEs) at 30–100 m depth are the dominant hard bottom habitat in the US Virgin Islands of the northeastern Caribbean Sea. Well-developed MCEs surpass the extent of shallow coral reefs by almost a factor of three. These habitats range from high coral cover structural reefs to low coral cover hard bottoms on banks, slopes, and walls. The most common communities in the United States Virgin Islands (USVI) are star coral (Orbicella franksi) banks that are concentrated in a relatively large area (187 km2) south and southwest of St. Thomas in depths of 30–43 m. Wall and slope coral communities in upper (30–60 m) and lower MCE (60–100 m) depths are dominated by lettuce corals (Agaricia spp.) Most MCEs are heavily covered in macroalgae, particularly Lobophora variegata. Corals are species rich in many upper MCE habitats (20–28 species) compared with shallow reefs (31), but not in upper and lower MCE slopes and walls (6–8). Sponges are species rich (24–53 species) in the MCEs of the USVI and may contribute much to nutrient cycling. Fishes are also species rich across habitats (45–151 species), but often their biomass is dominated by a few species, particularly on edges with high abundances of planktivores and piscivores. Despite their importance to the ecology and economy of USVI, MCEs are threatened by a variety of local and global stressors and disturbances that are causing declines in coral cover.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
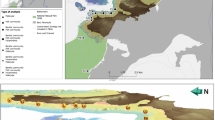
Caribbean mesophotic coral ecosystems (MCEs) are hard bottom habitats containing or dominated by scleractinian corals and spanning upper (30–60 m) and lower (60–100 m) depth ranges. The most extensively developed MCEs known in the Eastern Caribbean are found around the three major islands of the United States Virgin Islands (USVI): St. Croix, St. John, and St. Thomas (Fig. 8.1). USVI is one of the best-studied shallow coral reef systems in the world (Rogers et al. 2008); however, the insular shelves are actually dominated by understudied MCEs (Fig. 8.2). The extent of well-developed coral reefs (204 km2) at mesophotic depths surpasses that of shallow coral reefs (71 km2) by almost a factor of three. Because of its wealth of mesophotic coral resources and access to United States research resources, some of the longest and most detailed studies of MCEs globally were conducted in USVI (Turner et al. 2017).
(a) USVI located between the western Atlantic Ocean and northeastern Caribbean Sea. (b) Large insular shelves at mesophotic depths surround the northern USVI in (c) St. Thomas and St. John, whereas (d) St. Croix has a shallower insular shelf and a much narrower mesophotic margin. Red areas indicate marine protected areas with MCEs
Spatial extent of the insular shelf and predicted coral and hard bottom habitat in the northern USVI and St. Croix. In the northern USVI, habitat was measured from the British Virgin Islands Exclusive Economic Zone on the east to the 65.3°W, encompassing the entirety of the Virgin Passage MCE, which partially lies in Puerto Rican territorial waters. Classified habitat maps or habitat representation in subregional surveys taken from NOAA (2001), Costa et al. (2017), and Groves (2016) that have characterized about 45% of the insular shelf and which do not include the MCEs of the St. Thomas north mid-shelf, the western portion of the Virgin Passage, and mesophotic areas around St. Croix. Shallow-water (0–30 m) habitats included in the analysis do not include classified habitat surrounding Culebra and Vieques Islands, Puerto Rico. “Coral” habitats included were aggregate reef, linear reef, patch reef, and spur and grove reef. “Hard bottom” habitats included colonized bedrock/pavement and scattered coral and rock in unconsolidated sediment. “Insular shelf” indicates other unconsolidated sediment types or unclassified areas. Coral habitats from 60 to 90 m are poorly characterized, and it was estimated that 15% of the insular shelf area in that depth range is coral reef and 50% is colonized hard bottom
USVI has MCE communities representative of those found across the wider Caribbean. Communities within mesophotic habitats range from banks dominated by the scleractinian coral genus Orbicella, to steep slopes and walls fringed by the scleractinian coral genus Agaricia, to hard bottoms harboring mixed communities of macroalgae, sponges, and gorgonians. These communities show distinct spatial distributions across the insular shelves surrounding the islands, pointing to factors that potentially control their occurrence and distribution. A unique feature of USVI MCEs is the extensive Orbicella banks that make up the largest single community type.
1.1 Research History
1.1.1 1970s–1980s
USVI was one of the seminal areas for coral reef research in the 1970s and early 1980s. USVI was home to two underwater research laboratories focused on coral reefs, the Tektite II program and Virgin Islands Ecological Research Station on St. John and the National Oceanic and Atmospheric Administration’s Hydrolab operated by the Fairleigh Dickinson West Indies Laboratory on St. Croix. A wealth of shallow-water research studies were conducted prior to 1990, but relatively little was done at mesophotic depths. Some studies conducted at the Hydrolab in Salt River, St. Croix (located at 19 m depth), incorporated depth comparisons that had sites in upper mesophotic depths (30–37 m).
Suchanek et al. (1983) studied epibenthic sponge communities in Salt River wall reefs and showed that sponge diversity and sponge overgrowth of stony corals were highest at 36 m depth compared with 3–27 m, with the genera Verongia and Chondrilla being particularly common. Steneck (1983) found that grazing on algae was reduced at 36 m at the Salt River wall. Hubbard (1989), Hubbard et al. (2008), and Sadd (1984) described the geology, geomorphology, and sedimentary environment of northwestern St. Croix walls. Hubbard and Scaturo (1985) showed a decline in coral growth (linear extension) from shallow to mesophotic (30 m) depths on wall environments and attributed this to decreasing light availability. Aronson et al. (1994) described the impacts of Hurricane Hugo to the wall environments of Salt River as increasing with depth. This was mainly due to a loss of 11–26% absolute stony coral cover at 33 m, which was attributed to direct physical breakage of Agaricia lamarcki from oscillatory currents. Highly developed Orbicella banks were incidentally mentioned in papers describing fish spawning aggregations south of St. Thomas, but no comprehensive descriptions were made (Olsen and LaPlace 1978).
1.1.2 1990s–2010s
Starting in the 1990s, there was increasing awareness of MCEs south of St. Thomas and St. John. Beets and Friedlander (1999) conducted habitat observations of the Hind Bank Marine Conservation District (HBMCD; Fig. 8.1) and were the first to describe the mushroomlike growth form of mesophotic coral pillars composed primarily of Orbicella spp. High coral cover reefs (> 30%) dominated by Orbicella spp. were described in the HBMCD from initial larger-scale quantification using autonomous underwater vehicles and remotely operated vehicles (ROV; Armstrong et al. 2006). This study also gave some of the first indication that shallow-water species predominated at mesophotic depths and that orbicellid banks were an extension of shallow-water reef types. Menza et al. (2007) described a 30–40 m reef system (now known as Meri Shoal; Smith et al. 2016a) in the south of St. John in February 2005 by ROV with 38% coral cover and a structure similar to the HBMCD. They also noted an estimated 25% loss of coral cover, due to unknown causes within a year prior to observation.
The first permanent monitoring sites for USVI MCEs were established as part of the USVI Territorial Coral Reef Monitoring Program in 2003 at the HDMCD (41 m) and Grammanik Bank (38 m) (Nemeth et al. 2004). These sites have been monitored annually since 2003, and an additional seven MCE sites were added between 2009 and 2011 (Smith et al. 2016a). Two additional permanent monitoring sites have been added by the US National Park Service north of Buck Island, St. Croix, in 2017 (T.B. Smith, pers. obs.), making USVI one of the most comprehensively monitored MCE locations globally. Benthic cover, diseases, general coral health, and sediment trap flux rates were studied at permanent monitoring sites by Herzlieb et al. (2005) and Smith et al. (2008) and, in contrast to MCE walls in St. Croix, showed almost no sediment accumulation in traps at the HDMCD. These studies showed general coral health and coral cover increased in deeper offshore MCE. In addition, permanent monitoring sites were used to study the response of MCE to thermal stress in 2005, 2010, and 2012 (Smith et al. 2016b).
Studies of different MCE habitat characteristics and MCE fish spawning aggregations between 2003 and 2016 have provided an increasingly comprehensive description of the deeper environments of the USVI. Fish spawning aggregations are a common feature of USVI’s MCEs (Nemeth et al. 2007; Kadison et al. 2010; Nemeth and Kadison 2013; Biggs and Nemeth 2016). Large-bodied grouper and snapper spawning aggregations have been documented in at least seven MCE sites around the USVI and adjacent El Seco, Vieques, in Puerto Rico. Increasing use of side-scan and multibeam sonar to produce high resolution bathymetric maps of the seafloor in mesophotic depths revolutionized the understanding of the distribution of mesophotic bank systems in the USVI. Riviera et al. (2006) used side-scan sonar to make the first map of the HBMCD and Lang Bank, St. Croix. Subsequently, NOAA’s National Centers for Coastal Ocean Science’s Biogeography Program has conducted near annual field cruises to map MCEs using the NOAA Ship Nancy Foster. The result as of 2016 was partial mapping of MCEs of St. Croix and northern St. Thomas and a complete mapping and habitat characterization of the southern St. Thomas and St. John MCEs (Costa et al. 2017). The first wide-scale and spatially randomized description of an Orbicella bank system was conducted in the HBMCD in 2007 (Nemeth et al. 2008; Smith et al. 2010). Subsequent studies have documented MCE structure using spatially randomized or targeted surveys north of St. Thomas (Groves 2016), southern St. Thomas, and the St. John shelf edge (Smith et al. 2016c), portions of the Virgin Passage orbicellid bank complex outside the HBMCD (M. Brandt, unpubl. data), and the Lang Bank (Garcia et al. 2014). Furthermore, deep wall and slope MCE (30–100 m) were sampled, by divers and ROV by the Caribbean Coral Reef Institute at the University of Puerto Rico, Mayagüez, south of St. Thomas and St. John and at various points around St. Croix in 2011 and 2012 (CCRI 2011, 2012).
2 Environmental Setting
2.1 Geographic and Geological Setting
USVI sits at the northeastern edge of the Antillean island arc at the meeting of the Leeward Islands and Greater Antilles (18 °N, 64.7 °W; Fig. 8.1). The two northern islands of St. John (54 km2) and St. Thomas (91 km2) are part of the Puerto Rican shelf and connected to Puerto Rico to the west and the British Virgin Islands to the east. The Puerto Rican shelf forms a border between the western Atlantic Ocean and the Puerto Rican Trench (max. depth of 8400 m) to the north and the Caribbean Sea to the south. The more isolated island of St. Croix (216 km2) is located in the Caribbean Sea about 55 km to the south of St. Thomas and St. John and separated by the 4000 m deep Anegada-Jungfern Passage (Fratantoni et al. 1997). There are also 50 smaller islands on the insular shelves. All main islands have a mountainous topography, weakly defined wet and dry season, and very few areas with continuous freshwater flow into the marine environment. However, intense periods of heavy rain often cause sediment plumes of eroded soil to extend several km offshore.
Geologically, the islands are composed of island arc-related Cretaceous and Tertiary volcanic and volcanoclastic rock, carbonates, and near-surface intrusives (Alminas and Foord 1994). The islands are tectonically active (Hubbard et al. 2008) and prone to locally generated tsunamis (Watlington 2006); however, this section of the Antillean arc is slipping perpendicular to the subduction of oceanic crust, and, thus, there is no modern-day volcanism. The islands are strongly impacted by hurricanes and tropical storms (Hubbard et al. 2008; Chollett et al. 2012), with recent impacts from major hurricanes in 1989 (Hugo), 1995 (Marilyn), and 2017 (Irma and Maria).
2.2 Oceanographic Setting
The sea surface waters of USVI are comprised of Caribbean surface water typified by warm temperatures, high salinity, and increased water clarity (Chollett et al. 2012). Surface flow is primarily northward due to Ekman transport, 90° to strong easterly trade winds (Morrison and Smith 1990). The surface waters of the USVI are driven annually by changes in circulation, rainfall, and mesoscale eddies influenced by the extension of Amazon and Orinoco river plumes emanating from South America (Hu et al. 2004). These eddies can reach USVI and have large impacts on surface water characteristics and biological productivity (Johns et al. 2014). Riverine eddies have stable seasonal patterns of appearance, with the highest influence between June and December (Corredor and Morell 2001), 4 months after peak rains in northeast South America (Chérubin and Garavelli 2016). Largely because of the influence of the riverine eddies, peak values of near-surface chlorophyll a and colored dissolved organic matter occur in the warm season (June–November), whereas salinity peaks in the cool season (December–May). The El Niño Southern Oscillation and the North Atlantic Oscillation act as large-scale drivers by changing the strength and directions of the prevailing winds. Large-scale drivers impact biophysical properties of the Caribbean surface water, including how far the river plumes extend into the Caribbean and the production of mesoscale eddies in the plume fronts, which in turn influences nutrient availability (Corredor and Morell 2001; Hu et al. 2004; Chérubin and Richardson 2007; Chérubin and Garavelli 2016).
Whether or not surface conditions reflect conditions at mesophotic depths is dependent on the depth of the upper mixed layer (UML). The UML is the area from the sea surface to the first pycnocline (abrupt seawater density change) where wind mixing and other forms of turbulence homogenize the water’s physical characteristics. In the USVI oceanographic warm season (June–November), the UML is shallow because winds tend to be calmest, and steep vertical gradients of salinity are created primarily by riverine eddies and to a lesser degree by increased rainfall (Corredor and Morell 2001). Because salinity and then temperature have the largest proportional effect on water density, this leads to a relatively shallow pycnocline at 30–40 m depth and a decoupling of mesophotic depth waters from surface water characteristics in the warm season (Fig. 8.3a). In the oceanographic cool season (December–May), increased wind, turbulence, and salinity and decreased temperature deepen the UML to about 80–90 m, causing most MCEs to experience the water conditions of the UML. Thus, cool season temperatures are homogenous from shallow to mesophotic depths, whereas there is strong divergence of temperatures in the warm season (Fig. 8.3b). The chlorophyll maximum layer, an area of high pelagic productivity associated with a high concentration of phytoplankton, is associated with the first thermocline and can potentially influence mesophotic corals through heterotrophic feeding (Leichter and Genovese 2006). As should be expected, the depth of the chlorophyll maximum layer is seasonally dependent, located at the upper mesophotic zone in the warm season and at the base of the lower mesophotic in the cool season (Smith et al. 2016b; Fig. 8.3a), which has implications for coral energetics (Brandtneris et al. 2016).
Mean oceanographic conditions of USVI MCEs represented by (a) vertical water column profiles, (b) mean benthic temperatures, and (c) a week of benthic temperatures in November 2016. (a) Composite mean water column profiles of temperature, salinity, density, chlorophyll, and turbidity (fluorometric) from the peak of the sea surface warm (August 15–November 15; solid red line ± S.D.) and cool (January 01–April 01; solid blue line ± S.D.) seasons (See Smith et al. 2016b). Casts were taken in an area off the south shelf of St. Thomas near the Grammanik Bank (18.18°N, 64.96°W) with the N showing the number of casts contributing to the mean at each depth and season. Dashed lines indicate the depth of the upper mesophotic and lower mesophotic zones. (b) Composite mean diel benthic water temperature (black line ± SD) at coral reef monitoring sites indicated by a “T” in Fig. 8.1. Means are the average from each Julian day over multiple year of monitoring (n14m = 14 years, n38m = 12, n63m = 6), with the standard deviation the mean of the diel standard deviation for that day. (c) A 15-min frequency record of benthic temperatures in the first week of November 2016 at sites represented in (b)
Internal waves along the pycnocline result in higher diel temperature variability during the warm season (Fig. 8.3b). At the southern shelf edge of St. Thomas, internal waves are largely associated with internal tides and occur approximately twice per day (Fig. 8.3c). This diel variation creates an oscillating benthic current that may deliver onshelf and offshelf water (e.g., benthic currents tend to have a north-south shift with the tide; Smith et al. 2016a) to MCEs. The activity of internal waves differs between MCE habitats, with little evidence of internal wave induced upwelling in an Orbicella basin in the HBMCD (Smith et al. 2010) and somewhat weaker activity in upper mesophotic zone walls on northwest St. Croix (T.B. Smith, unpub. data). This heterogeneity may contribute to different nutrient and temperature regimes at a given depth with implications for coral energetics and exposure to thermal stress.
3 Habitat Description
The MCEs of USVI show remarkable heterogeneity in terms of geomorphology, stony coral abundance, and dominant species. USVI’s shelf environments can be divided by geomorphology into horizontal banks on the insular shelf to steep slopes and walls along the shelf edges. Across these features community composition can vary dramatically and range from low coral abundance hard bottoms (~4% cover) dominated by macroalgae, sponges, and octocorals to high scleractinian coral abundance (~20%) with high habitat complexity (Smith et al. 2016c; Fig. 8.4). Where scleractinian corals are abundant, the dominant species are strongly dependent on depth. In the upper mesophotic zone (30–43 m depth), coral cover is dominated by star coral (Orbicella) banks that are flat or nearly flat and located at least 2 km from shore. In the lower mesophotic zone (60–75 m), coral cover is typified by sheet and scroll coral (Agaricia) communities growing on steep slopes. In high coral cover systems, there appears to be a community break between 43 m depth, the lower limit of Orbicella reefs, and 60 m, the upper limit of dense agariciid (Agaricia undata and A. grahamae) reefs (Smith and Holstein 2016; Smith et al. 2016b). This intermediate zone at 45–60 m often consists of low stony coral cover hard bottom, colonized by macroalgae, sponges, octocorals, and antipatharians. This break supports a distinction of the upper mesophotic (30–60 m) and lower mesophotic (60–100 m) zones. However, the break in community composition is absent on the north facing walls of St. Croix, where coral abundance tends to be low across the mesophotic depth range and plating agariciids dominate (CCRI 2012).
Representative MCEs of USVI. (a) An Orbicella bank southeast of St. Thomas at 40 m depth, with inset showing close up of pillar-like coral reef structure (Hammerhead Shoal, St. Thomas). (b) The agariciid fringe at 65 m depth along permanent transects of the USVI Territorial Coral Reef Monitoring Program (Ginsburg’s Fringe, St. Thomas). (c) Close-up of structure of the agariciid fringe showing a colony of Agaricia lamarcki and pair of reef butterflyfish (Chaetodon sedentarius). (d) Upper mesophotic wall of Salt River at 35 m. (e) Lower mesophotic wall at Cane Bay, St. Croix at 60 m depth showing overlapping colonies of A. undata. (f) A shelf edge hard bottom MCE on the Grammanik Bank, St. Thomas at a depth of 35 m. (Photos: (a), (c) Viktor W. Brandtneris; (b), (f) Tyler B. Smith; (d) Rosmin Ennis; (e) Doug Kesling)
Extent of Habitats
Slightly more than half of the known and predicted coral reef and hard bottom habitats of the USVI are in mesophotic depths between 30–100 m (Fig. 8.2). However, this 382 km2 is unequally distributed among communities. By far the highest proportion of habitat is concentrated in upper mesophotic banks (307 km2), which are dominated by Orbicella (187 km2). Hard bottom habitats supporting corals form the remainder of the consolidated substrate in the upper mesophotic zone (120 km2). However, the upper mesophotic zone also supports large areas (1354 km2) of unconsolidated sediments, largely composed of rhodolith beds, which are very important habitats for local fisheries (Tobias 1997), and solitary coralliths and small stony coral patches (Authors, pers. obs.). These “soft” habitats are particularly concentrated north and south of St. Thomas and St. John and on the tip of the Lang Bank, St. Croix. USVI’s lower mesophotic zone covers a much smaller area (106 km2) that is exclusively composed of moderately steep slopes or very steep wall environments. These areas are poorly characterized in terms of community distribution because the slope angle makes them difficult to capture in multibeam sonar surveys and drop camera surveys, and their depth precludes large-scale diver-based observations. Based on the work of CCRI (2012), Smith et al. (2016a), and unpublished surveys, we estimate an area of 17 km2 of high abundance coral communities (15% of the habitat) and 58 km2 of consolidated hard bottom communities with corals (50%). However, these estimates will likely change as lower MCEs in the USVI are more thoroughly explored.
3.1 Upper Mesophotic Zone (30–60 m)
3.1.1 Orbicella Banks
Star corals form very dense and topographically complex mesophotic communities along the outer shelves of the USVI (Fig. 8.4). These communities are often a continuation of shallow-water reefs on a gently sloping seafloor, with maximum depths of dense communities of about 43 m (> 4% cover). The well-studied Orbicella banks of the Virgin Passage between the Spanish Virgin Islands (Culebra and Vieques, Puerto Rico) and western St. Thomas are exceptional examples (Fig. 8.5), with mean coral cover of >20% and up to 60% (Smith et al. 2016c; Fig. 8.6). The area of the larger Orbicella bank reef complex of the Virgin Passage (170 km2) is about 2.4 times larger than the combined area of well-developed shallow-water coral reef habitat in the northern USVI (53 km2) and St. Croix (19 km2) (Fig. 8.2).
A habitat map of MCEs of the southeastern Puerto Rican insular shelf south of St. Thomas and St. John, USVI. Methods for map creation are detailed in Costa et al. (2017). Edges are the limits of multibeam bathymetry data and do not represent habitat boundaries (Map created by B. Costa and L. Kracker, NOAA)
Composite mean benthic cover of different stony coral genera, algae, gorgonians, sponges, and sediment over different mesophotic and shallow habitats. Wall and slope environments are represented by the lower MCE “agariciid fringe” at the site of Ginsburg’s Fringe (year of sampling in italics) and upper MCE walls on the northeast coast of St. Croix (2016), the upper mesophotic banks categorized as Orbicella (cover >20%; 2007 & 2016), Caribbean shelf edge south of St. Thomas and St. John (2014), and Atlantic shelf edge, north of St. Thomas (2015). Shallow sites are part of the USVI Territorial Coral Reef Monitoring Program and are distributed throughout the USVI (2016). Samples sizes for benthic cover given in Table 8.1. Cover taken from benthic video transects or drop camera photos and data from Smith et al. (2010, 2016a, 2016c) and Groves (2016). Orbicellids listed in the upper panel are predominately Orbicella franksi
Of the Orbicella species, the dominant constructional species are predominantly Orbicella franksi, which account for about 90% of the coral cover and form a complex network of mushroomlike coral pillars over sediment-covered pavement (Beets and Friedlander 1999; Armstrong et al. 2006; Smith et al. 2010, 2016c; Figs. 8.4 and 8.6). A conservative estimate of the population of this coral genus in the Virgin Passage is 300 million individual colonies (based on scaling of estimate in Smith 2013). This area of the USVI includes the HBMCD and Grammanik Bank fisheries managed areas that were designated to protect spawning aggregations of red hind (Epinephelus guttatus) and yellowfin grouper (Mycteroperca venenosa) but have subsequently been found to support aggregations of at least six additional fish species, including the last known significant US Caribbean aggregation of the threatened Nassau grouper, Epinephelus striatus (Kadison et al. 2010). The Nassau grouper aggregation consists of several hundred spawning individuals (Kadison et al. 2010). The MCEs between these two managed areas also serve as important migration pathways for spawning groupers (Rowell et al. 2015). On the southern shelf of St. Thomas and St. John, dense Orbicella banks also occur on eastern mid-shelf areas, east of French Cap, and south of the Pillsbury Sound (e.g., Meri Shoal).
The island of St. Croix supports Orbicella banks of much narrower extent near the shelf edge but with the same characteristic dominance of orbicellid colonies growing atop mushroom-shaped pillars. On the eastern edge of Lang Bank, Orbicella communities of moderate to high cover (10–55%) are located around and within a mesophotic basin (30–55 m depth) (Nemeth et al. 2006; Garcia et al. 2014; Smith et al. 2016a). This feature is part of a relict barrier reef formation corresponding to the sea level of the early Holocene (8 ka) or earlier periods (Hubbard et al. 2013). The eastern end of Lang Bank has been seasonally closed to bottom tending fishing gear (December–February) since 1993 to protect a spawning aggregation of red hind (Nemeth et al. 2006; Garcia et al. 2014; Fig. 8.1). Mesophotic Orbicella banks have also recently been described on the southwestern shelf edge of Lang Bank (Lang Bank East End Marine Park, coral cover of 16%; Smith et al. 2016a) and north of Buck Island within the Virgin Islands National Monument (coral cover of 31%; M. Feeley, US National Park Service, unpub. data). The remainder of the St. Croix shelf tends to be steep slopes and walls that are unfavorable for the development of Orbicella-dominated reefs.
Orbicella banks can have different coral abundance and benthic features that may depend on antecedent topography, as well as modern-day disturbances. In the Virgin Passage of the northern USVI, Orbicella reefs form distinct banks oriented away from the shelf edge (Smith et al. 2010). Orbicella corals also form mesophotic patch reefs surrounded by rhodoliths that are often hotspots for large-bodied fishes and motile invertebrates (Nemeth et al. 2008; Garcia et al. 2014). Smith et al. (2010) described a novel basin and hillock Orbicella habitat type from the HBMCD. It is unclear how much antecedent topography controls the mesophotic bank reef macrostructure and how much is formed through modern Holocene coral growth, since the issue has not been studied directly. However, low coral cover pavements often gradually transition to high coral cover orbicellid pillar communities, where pavement is often visible between coral stacks. The evidence of a pavement bottom under modern coral communities suggests that modern Orbicella reefs are a structurally complex veneer atop antecedent carbonate frameworks. Bank formation could have occurred when these areas were near sea level throughout the late Tertiary and composed of fast-growing branching acroporids (Hubbard et al. 2008); however, the geology of these MCE banks needs further study. In addition, Orbicella banks are absent in areas that seem suitable for colonization and reef formation. This includes the drowned barrier reef along the southern shelf edge between Vieques and the British Virgin Islands (Smith et al. 2016c), the outer rim of Lang Bank (Garcia et al. 2014), and the northern bank edge of St. Thomas (Groves 2016). It was hypothesized that low-frequency storm and swell disturbances and low coral growth (Weinstein et al. 2016; Groves et al. 2018) and recovery rates (Smith et al. 2016b) are contributing to the lack of Orbicella reef formation (Groves 2016; Smith et al. 2016c). Thus, the occurrence of dense mesophotic orbicellid banks may be controlled by a combination of suitable antecedent topography, buffering from disturbance, and other biophysical characteristics, such as clear water and moderate flushing.
3.1.2 Hard Bottom Banks
One of the dominant habitats of USVI are upper mesophotic hard bottom banks constructed of carbonate pavements (17% of consolidated substrate habitats; Fig. 8.2). These habitats are concentrated on outer shelf edges north and south of St. Thomas and St. John (Groves 2016; Smith et al. 2016c) and on the outer edge of Lang Bank (Garcia et al. 2014; Fig. 8.1). Coral cover is low, typically less than 4%, and dominant benthic constituents are macroalgae, epilithic algal turfs, crustose coralline algae, and epibenthic sponges (Figs. 8.4 and 8.6). Gorgonians are only common on the Caribbean banks (Fig. 8.6). Groves (2016) studied the bank systems north of St. Thomas and found that mean coral cover was low and never exceeded 15% even though many of the banks are in the same depth ranges of the vibrant south shelf Orbicella banks. Growth rates were not an explanation for the lack of reef development, as orbicellids grew very slowly on both the north and south mesophotic bank systems (Weinstein et al. 2016; Groves et al. 2018).
3.1.3 Walls and Slopes
Upper mesophotic zone walls and steep slopes are ubiquitous features of the shelf edge and range from moderate slopes on the north shelf edge of St. Thomas to nearly vertical walls along the northwest coast of St. Croix. The latter is one of the steepest submarine slopes in the world (Hubbard et al. 2008). Upper mesophotic seaward slopes tend to largely consist of low stony coral cover habitats, with higher relative coverage of sponges, gorgonians, and macroalgae (Figs. 8.4 and 8.6; Smith et al. 2016c). Despite their lack of coral cover, these slopes can be critical areas for fish spawning aggregations (Smith et al. 2016a). In contrast, wall MCEs on the northwestern coastline of St. Croix are exceptionally well developed (Sadd 1984; Hubbard et al. 1986; Hubbard 1989; Smith et al. 2016a). These walls are composed of large buttresses surrounded by channels that transport sediments from the upper reef top into the abyss (Fig. 8.4; Sadd 1984; Hubbard 1989). Thus, the majority of coral growth tends to occur on top of the spurs. The majority of corals are agariciids (Fig. 8.6), particularly A. lamarcki and A. grahamae, which account for about 90% of living coral cover at Salt River (30–33 m depth) and Cane Bay (33–40 m) (Smith et al. 2016a). Coral cover appears more continuous with depth if there is suitable wall habitat (CCRI 2012) and there is no faunal break to hard bottom between 43 and 60 m depth, as seen on the southern slopes of the northern USVI.
3.1.4 Lower Mesophotic Zone (60–90 m): The Agaricia Fringe
Lower mesophotic communities of the USVI (> 60 m depth) are not well studied because of their depth. However, observations suggest that corals of the genus Agaricia are dominant and form what has been termed the “agariciid fringe” or “ring around the bathtub” in the Caribbean (Robert Ginsburg, pers. comm.). This community forms on steep slopes and walls and is the exclusive MCE habitat in these depth ranges in USVI. From available observations, it appears that almost all scleractinian corals disappear below about 90 m depth in USVI (Nelson and Appeldoorn 1985; CCRI 2012), which is what defines the lower limit of MCEs herein.
Slope communities show great heterogeneity in development, from spectacularly developed patches of A. undata to scattered corals on hard bottom. One highly developed agariciid coral reef is present below the Grammanik Bank seasonal fishery closure, St. Thomas, and has been under annual monitoring since 2011 (Smith et al. 2016a; Figs. 8.3b–c, 8.4, 8.6, and 8.7). This reef is dominated by the stony coral A. undata (75% of living coral cover), with lesser amounts of A. grahamae (20%) and A. lamarcki (5%) and isolated colonies of Montastrea cavernosa, Porites astreoides, and Meandrina meandrites (Table 8.1). Large whip corals and antipatharians are also present. Some of the A. undata colonies in these environments form whorls greater than 7 m in diameter that provide large amounts of structure for small depth-generalist and mesophotic fish species (CCRI 2012; Smith et al. 2016a). This site is named “Ginsburg’s Fringe” in honor of MCE pioneer Dr. Robert Ginsburg (1925–2017). From areas that have sustained damage to the overlying corals, it appears there is a true framework over the basement limestone that can be at least 1 m thick (see photo in Smith et al. 2019). Somewhat similar communities have been described from St. John and St. Croix (CCRI 2012). Although patches of high coral cover have been documented, these slopes have not been systematically characterized in USVI, and available observations suggest that low density and patchy coral colonies growing on pavement may be the most common habitat type.
Biomass of fishes divided by trophic group over different mesophotic and shallow habitats. Data sources as defined in Fig. 8.6. Estimates are from diver-based reef visual censuses along transects (4 × 25 m) with sample sizes of agariciid fringe slope = 16, upper MCE wall = 176, Orbicella bank = 659, Caribbean shelf edge = 44
Deeper wall environments of St. Croix were studied in 2011 and 2012 (CCRI 2011, 2012). They found great heterogeneity in coral development, with the most extensive development on the northwestern wall area of St. Croix (Fig. 8.4). In many sites, sedimentation appeared to limit reef development, even on steep walls.
4 Biodiversity
4.1 Macroalgae
Macroalgae are prominent components of USVI mesophotic habitats. Rather than diminishing with reduced light at depth, macroalgae can maintain similar relative abundances on shallow and mesophotic reefs (Smith et al. 2008, 2016a; Fig. 8.6). While the genus Dictyota is dominant in shallow water, it is present in much lower abundance in MCEs down to least 70 m. Instead, Lobophora variegata becomes the dominant macroalga in most habitats (up to 25% mean cover). However, the Atlantic shelf edge north of St. Thomas has a more mixed macroalgal community (species include Amphiroa spp., Dictyota spp., Halimeda spp., Lobophora spp., and Sargassum spp.). Epilithic algae of small to tall turfs and filamentous algae are the dominant benthic substrate on the Caribbean shelf edge south of St. Thomas and St. John (Smith et al. 2016c) and are also abundant on the outer Lang Bank (Garcia et al. 2014). Filamentous cyanobacterial mats are particularly abundant on Orbicella banks, where they cover almost 8% of the substrate at various times with an unknown seasonality.
4.2 Anthozoans
Caribbean scleractinian coral species are well represented in USVI’s MCE, with strong overlap of shallow-water species in the upper mesophotic zone and decreasing species richness and increasing representation of deepwater specialist species in the lower mesophotic zone. Variations in coral cover are foremost driven by the abundance of orbicellids on upper mesophotic banks and agariciids on mesophotic walls and slopes (Fig. 8.6). Depending on depth, specific coral species account for most coral cover and reef formation, with orbicellids, primarily O. franksi, at upper mesophotic depths, and A. undata or A. lamarcki at lower mesophotic depths. Although there is a high tendency toward dominance of one species in well-developed coral reef habitats, upper MCEs still have good representation of coral species relative to shallow water. From a compilation of studies, at least 31 species of scleractinian corals and hydrocorals were recorded in shallow reefs, 32 in Orbicella banks, 32 in Caribbean shelf edge banks, 22 in Atlantic shelf edge banks, 13 in upper mesophotic walls, and 7 in a lower mesophotic slope (Table 8.1). Only A. lamarcki was present across all habitats. In addition, 38 and 22 scleractinian species were documented in El Seco, southwest Virgin Passage (Garcia et al. 2011), and the Lang Bank, St. Croix (Garcia et al. 2014), respectively.
4.3 Sponges
As in shallow Caribbean reefs, epibenthic sponges are common and represented by many species in USVI’s MCEs. Cover ranged from 1 to 10% of the substrate in different MCEs (Fig. 8.6). Species level information has been collected for ~51species from El Seco (Garcia et al. 2011) and 53 species from Lang Bank (Garcia et al. 2014). Diversity was highest in colonized hard bottom habitats (40 and 42 species for El Seco and Lang Bank, respectively) and somewhat reduced in coral reef habitat (24 and 33 species). The giant barrel sponge Xestospongia muta dominated cover in hard bottom habitats and contributed greatly to habitat complexity. Given their abundance, it is likely that sponges in USVI’s MCEs are contributing significantly to nutrient cycling and reef energetics (de Goeij et al. 2013).
4.4 Fishes
USVI’s MCEs are critical as refuge areas for commercially important species and as sites of vulnerable fish spawning aggregations (Kadison et al. 2017). Fish populations show distinct assemblage structures both among habitat types and within different coral reef types (Fig. 8.7). Reef visual census data was compiled from the long-term USVI Territorial Coral Reef Monitoring Program (Smith et al. 2016a) and published and unpublished data from ad hoc research (Nemeth et al. 2008; Smith et al. 2010; Groves 2016; Smith et al. 2016c; T.B. Smith, unpubl. data). The highest fish biomass was found on Orbicella banks and Caribbean shelf edge habitats, which had approximately two to five times the mean biomass of shallow reefs. High biomass in these habitats was driven by a few species of planktivores, piscivores, and invertivores concentrated on bank edges. In particular, in Orbicella banks (151 species), over half of the biomass was from five species: large piscivorous cubera snapper (Lutjanus cyanoptera) and schoolmaster snapper (Lutjanus apodus) contributed 30% to overall biomass, and planktivorous creole wrasse (Clepticus parrae), black durgeon (Melichthys niger), and ocean triggerfish (Canthidermis sufflamen) contributed to 25%. On the shelf edge, hard bottom habitats bordering the Caribbean Sea (94 species), nearly half of the biomass was from five species: large schools of horse-eye jack (Caranx latus), the ocean triggerfish, and black durgeon contributed 35% to biomass, and the demersal species schoolmaster snapper (Lutjanus apodus) and the queen triggerfish (Balistes vetula) contributed a combined 13%. Wall and lower mesophotic slope habitats appear to support lower fish biomass than MCE banks and shallow-water coral reefs. In upper mesophotic wall habitats (124 species), fish biomass was more evenly distributed among species and trophic groups, likely reflecting a diverse range of food availability from pelagic, hard bottom/coral, and soft sediment sources. Half of the fish biomass was contributed by eight species (in descending order): yellow goatfish (Mulloidichthys martinicus), creole wrasse, redband parrotfish (Sparisoma aurofrenatum), yellowtail snapper (Ocyurus chrysurus), blue chromis (Chromis cyanea), black durgeon, princess parrotfish (Scarus taeniopterus), and mahogany snapper (Lutjanus mahogoni). In one reef of the lower MCE agariciid fringe (45 species), half of the biomass was from just four species: creole wrasse, the invasive Indo-Pacific lionfish (Pterois volitans), dog snapper (Lutjanus jocu), and the nurse shark (Ginglymostoma cirratum). The lower mesophotic zone also has a higher abundance of a deepwater fauna, including the blackfin snapper (Lutjanus buccanella), sunshinefish (Chromis insolata), cherubfish (Centropyge argi), longsnout butterflyfish (Prognathodes aculeatus), and peppermint bass (Liopropoma rubre).
The first Indo-Pacific lionfish was reported in USVI waters in 2009 on the west end of St. Croix in shallow water under the cruise ship pier. The following year, they were seen off St. Thomas and St. John and by early 2011 had become common throughout the territory (Smith et al. 2019). On MCEs, lionfish populations have thrived. Abundance climbed between years 2011 to 2014 based on fixed site surveys made by the USVI Territorial Coral Reef Monitoring Program, with peak values of 1–4 individuals 100 m−2, but have stabilized or decreased since 2014 (Smith et al. 2016a). Densities remain higher on MCEs than shallow reefs in the monitoring program. This may represent a preference for deeper habitats but more likely is a result of reduced fishing pressure for commercial, recreational, and management purposes (Andradi-Brown et al. 2017).
5 Ecology
The ecology of MCEs in USVI is only now beginning to be investigated. Basic information on the life, death, and growth of mesophotic organisms is lacking. Growth has been shown to decline in depth-generalist species with increasing depth in USVI (Hubbard and Scaturo 1985; Weinstein et al. 2016; Groves et al. 2018). However, internal and external bioerosion also tends to decrease with depth on Orbicella banks (Weinstein et al. 2016), and this may allow for reef construction even with very low rates of carbonate accretion (Weinstein et al. 2014). Grazing also likely declines with depth, given the general decline in grazing fish biomass (Fig. 8.7) and an almost complete absence of urchins in MCEs (Smith et al. 2016a). Opposing trends of growth and bioerosion may allow orbicellid-dominated MCEs to maintain positive rates of accretion (Weinstein et al. 2014). In fact, growth of mesophotic reefs with slow-growing foundational species may depend critically on attenuation of destructive processes that kill corals and erode reef frameworks (e.g., bioerosion, disease, bleaching, and mechanical disturbance) and may explain a positive relationship between coral cover and depth on modern USVI reefs. Slow growth in many stony coral species may be related to low-light availability and high reproductive output (Holstein et al. 2015, 2016), which may cause periods of low energy reserves as shown by tissue caloric content (Brandtneris et al. 2016). Growth rates in lower MCE environments have not been established for USVI, but have been shown to be relatively high for Caribbean agariciids in Curaçao (Bongaerts et al. 2015). In all MCEs of USVI, there is high heterogeneity in coral abundance and reef formation in the same depths and even where coral growth rates are similar (Hubbard and Scaturo 1985; Groves et al. 2018), indicating species interactions or extrinsic disturbances are major limiters of coral abundance and coral reef development.
6 Threats and Conservation Issues
The MCEs of USVI are not removed from all threats facing shallow reefs, and in some cases their anonymity makes them more vulnerable. All long-term MCE research sites in the USVI that have been monitored for over a decade have shown losses of coral cover (Smith et al. 2016a). The greatest threats have been thermal stress, disease, and mechanical disturbance. Alternatively, USVI’s MCEs may be partial refuges from fishing (Kadison et al. 2017) and sedimentation (depending on proximity to shore; Smith et al. 2008).
Thermal stress and disease. All long-term MCE sites have experienced moderate to severe bleaching linked to seawater exceeding local maximum mean temperatures (Smith et al. 2016b). Orbicella bank MCEs showed bleaching and loss of coral cover (~25%) associated with the 2005 thermal stress event (Fig. 8.8). Upper mesophotic wall environments were also heavily bleached in 2005 (> 80% of colonies; Smith et al. 2016b) and suffered an unknown amount of coral cover loss. More recently, in 2012, a lower MCE site bleached as a result of high temperatures, although the amount of coral cover loss, if any, is unknown (Smith et al. 2016b). While the presence of diseases may be low in lower MCEs, white plague has been recurrent in upper MCE Orbicella banks, often exceeding 5% prevalence in outbreaks, surpassing levels seen in shallow reefs (Smith et al. 2016a, 2016b, 2019).
Long-term trends in stony coral cover on MCEs of USVI and shallow reefs taken from the USVI Territorial Coral Reef Monitoring Program (Smith et al. 2016a). The agariciid fringe is represented by one site at 60–65 m depth. Orbicella banks are represented by four sites south of St. Thomas and St. John from 30 to 41 m depth. Agariciid wall sites are represented by two sites on northwestern St. Croix at 30–40 m depth. Shallow sites are represented by 22 locations from around USVI in 7–25 m depths
Fishing and fishing gear. Hard bottom banks in USVI face threats from fishing pressure. The hard bottom banks north of St. Thomas are some of the most intensively fished areas of USVI (Tobias 1997). The primary gear is West Indian fish traps connected in strings of 5–10 traps by subsurface polypropylene lines. These lines can drag across the bottom severing barrel sponges (Smith et al. 2019) and steel traps directly contact the benthos. Abandoned or lost traps can also continue fishing for some time, but mandatory escape panels limit their impacts (Renchen et al. 2014). Upper mesophotic wall environments in USVI also show impacts from fishing gear, particularly fouled lines and traps (Smith et al. 2016a).
Mechanical disturbance. The likelihood of damage to MCEs is increased by their relative invisibility to most people. The main Ginsburg’s Fringe site started at over 45% coral cover when first monitored in 2011, but by 2016 this had dropped to about 23% (Fig. 8.8; Smith et al. 2019). The suspected cause was physical damage from anchoring along the deep shelf edge, likely to facilitate fishing just outside or within the Grammanik Bank seasonal fisheries closure. Steel reef claw anchors and attached polypropylene line have been found abandoned at the site. It is likely that the responsible parties do not know what bottom habitat their anchors are contacting. In other cases damage may occur incidentally. The upper MCE wall sites on St. Croix are very popular as recreational diving sites and damage likely occurs from lack of caution by individuals.
Recovery. Conservation and management of MCEs in the USVI may also need to take into account the rate of recovery after disturbance. While this is an area in need of further study, it is worth noting that recovery has been lacking or weak in long-term sites. In Orbicella banks, recovery may be inhibited by recurrent white plague disease outbreaks (Smith et al. 2019). Upper mesophotic Cane Bay and Salt River walls have shown a very slight trend of increase in coral cover since the establishment of the monitoring sites in 2009 (Fig. 8.8). Thus, it may be that these MCEs in USVI will need special protection to avoid damage. Conservation of USVI MCEs will be enhanced greatly by better characterization and communication to the public and government agencies of their existence.
References
Alminas HV, Foord EE (1994) Geochemistry, mineralogy, and geochronology of the U.S. Virgin Islands. U.S. Geological Survey Bulletin 2057. U.S. Geological Survey, Washington, DC, 35 pp
Andradi-Brown DA, Vermeij MJA, Slattery M, Lesser M, Bejarano I, Appeldoorn R, Goodbody-Gringley G, Chequer AD, Pitt JM, Eddy C, Smith SR, Brokovich E, Pinheiro HT, Jessup ME, Shepherd B, Rocha LA, Curtis-Quick J, Eyal G, Noyes TJ, Rogers AD, Exton DA (2017) Large-scale invasion of western Atlantic mesophotic reefs by lionfish potentially undermines culling-based management. Biol Invasions 19:939–954
Armstrong RA, Singh H, Torres J, Nemeth RS, Can A, Roman C, Eustice R, Riggs L, Garcia-Moliner G (2006) Characterizing the deep insular shelf coral reef habitat of the Hind Bank marine conservation district (US Virgin Islands) using the Seabed autonomous underwater vehicle. Cont Shelf Res 26:194–205
Aronson RB, Sebens KP, Ebersole JP (1994) Hurricane Hugo’s impact on Salt River submarine canyon, St. Croix, U.S. Virgin Islands. In: Proceedings of the colloquim on global aspects of coral reefs: health, hazards, and history. University of Miami, Miami, pp 189–195
Beets J, Friedlander A (1999) Evaluation of a conservation strategy: a spawning aggregation closure for red hind, Epinephelus guttatus, in the U.S. Virgin Islands. Environ Biol Fish 55:91–98
Biggs CR, Nemeth RS (2016) Spatial and temporal movement patterns of two snapper species at a multi-species spawning aggregation. Mar Ecol Prog Ser 558:129–142
Bongaerts P, Frade PR, Hay KB, Englebert N, Latijnhouwers KRW, Bak RPM, Vermeij MJA, Hoegh-Guldberg O (2015) Deep down on a Caribbean reef: lower mesophotic depths harbor a specialized coral-endosymbiont community. Sci Rep 5:7652
Brandtneris VW, Brandt ME, Glynn PW, Gyory J, Smith TB (2016) Seasonal variability in calorimetric energy content of two Caribbean mesophotic corals. PLoS ONE 11:e0151953
Caribbean Coral Reef Institute [CCRI] (2011) 2011 Mesophotic coral ecosystems cruise. University of Puerto Rico, Mayagüez. http://ccri.uprm.edu/media/Mesophotic_Web_page/MCE_Cruise_2011.html. Accessed 09 Jan 2018
CCRI (2012) 2012 Mesophotic coral ecosystems cruise. University of Puerto Rico, Mayagüez. http://ccri.uprm.edu/media/Mesophotic_Web_page/MCE_Cruise_2012.html. Accessed 09 Jan 2018
Chérubin LM, Garavelli L (2016) Eastern Caribbean circulation and island mass effect on St. Croix, US Virgin Islands: a mechanism for relatively consistent recruitment patterns. PLoS ONE 11:e0150409
Chérubin LM, Richardson PL (2007) Caribbean current variability and the influence of the Amazon and Orinoco freshwater plumes. Deep-Sea Res I Oceanogr Res Pap 54:1451–1473
Chollett I, Mumby PJ, Muller-Karger FE, Hu C (2012) Physical environments of the Caribbean Sea. Limnol Oceanogr 57:1233–1244
Corredor JE, Morell JM (2001) Seasonal variation of physical and biogeochemical features in eastern Caribbean Surface Water. J Geophys Res 106:4517–4525
Costa BM, Kracker LM, Battista T, Sautter W, Mabrouk A, Edwards K, Taylor C, Ebert E (2017) Benthic habitat maps for the Southeastern Puerto Rican insular shelf NOAA technical memorandum NOS NCCOS 241. National Oceanic and Atmospheric Administration, Silver Spring, 59 pp
de Goeij JM, van Oevelen D, Vermeij MJA, Osinga R, Middelburg JJ, de Goeij AFPM, Admiraal W (2013) Surviving in a marine desert: the sponge loop retains resources within coral reefs. Science 342:108–110
Fratantoni DM, Zantopp RJ, Johns WE, Miller JL (1997) Updated bathymetry of the Anegada-Jungfern Passage complex and implications for Atlantic inflow to the abyssal Caribbean Sea. J Mar Res 55:847–860
Garcia JR, Sabater-Clavell J, Esteves R, Capella J, Carlo M (2011) Characterization of benthic habitats and associated coral reef communities at El Seco, Southeast Vieques. Caribbean Fishery Management Council, San Juan, Puerto Rico, 96 pp
Garcia JR, Williams SM, Sabater-Clavell J, Esteves R, Carlo M (2014) Mesophotic benthic habitats and associated reef communities at Lang Bank, St. Croix, USVI. Caribbean Fishery Management Council, San Juan, Puerto Rico, 69 p + appendices
Groves S (2016) Physical drivers of community structure and growth among mesophotic coral ecosystems surrounding St. Thomas, U. S. Virgin Islands. M.Sc. University of the Virgin Islands
Groves SH, Holstein DM, Enochs IC, Kolodzeij G, Manzello DP, Brandt ME, Smith TB (2018) Growth rates of Porites astreoides and Orbicella franksi in mesophotic habitats surrounding St. Thomas, US Virgin Islands. Coral Reefs 37:345–354
Herzlieb S, Kadison E, Blondeau J, Nemeth RS (2005) Comparative assessment of coral reef systems located along the insular platform south of St. Thomas, US Virgin Islands and the relative effects of natural and human impacts. Proc 10th Int Coral Reef Symp 4(2):1144–1151
Holstein DM, Smith TB, Gyory J, Paris CB (2015) Fertile fathoms: Deep reproductive refugia for threatened shallow corals. Sci Rep 5:12407
Holstein DM, Smith TB, Paris CB (2016) Depth-independent reproduction in the reef coral Porites astreoides from shallow to mesophotic zones. PLoS ONE 11:e0146068
Hu C, Montgomery ET, Schmitt RW, Muller-Karger FE (2004) The dispersal of the Amazon and Orinoco River water in the tropical Atlantic and Caribbean Sea: observation from space and S-PALACE floats. Deep-Sea Res II Top Stud Oceanogr 51:1151–1171
Hubbard DK (1989) The shelf-edge reefs of Davis and Cane Bays, northwestern St. Croix, U.S.V.I. 12th Caribb Geol Conf Spec Publ 8:167–180
Hubbard DK, Scaturo D (1985) Growth rates of seven species of scleractinian corals from Cane Bay and Salt River, St. Croix, USVI. Bull Mar Sci 36:325–338
Hubbard DK, Burke RB, Gill IP (1986) Styles of reef accretion along a steep, shelf-edge reef, St. Croix, U.S. Virgin Islands. J Sediment Petrol 56:848–861
Hubbard DK, Burke RB, Gill IP, Ramirez WR, Sherman C (2008) Coral-reef geology: Puerto Rico and the US Virgin Islands. In: Riegl BM, Dodge RE (eds) Coral Reefs of the USA. Springer, New York, pp 263–302
Hubbard DK, Gill IP, Burke RB (2013) Holocene reef building on eastern St. Croix, US Virgin Islands: Lang Bank revisited. Coral Reefs 32:653–669
Johns EM, Muhling BA, Perez RC, Müller-Karger FE, Melo N, Smith RH, Lamkin JT, Gerard TL, Malca E (2014) Amazon River water in the northeastern Caribbean Sea and its effect on larval reef fish assemblages during April 2009. Fish Oceanogr 23:472–494
Kadison E, Nemeth RS, Blondeau J, Smith TB, Calnan JM (2010) Nassau grouper (Epinephelus striatus) in St. Thomas, US Virgin Islands, with evidence for a spawning aggregation site recovery. Proc Gulf Carib Fish Inst 62:273–279
Kadison E, Brandt M, Nemeth R, Martens J, Blondeau J, Smith T (2017) Abundance of commercially important reef fish indicates different levels of over-exploitation across shelves of the U.S. Virgin Islands. PLoS ONE 12:e0180063
Leichter JJ, Genovese SJ (2006) Intermittent upwelling and subsidized growth of the scleractinian coral Madracis mirabilis on the deep fore-reef of Discovery Bay, Jamaica. Mar Ecol Prog Ser 316:95–103
Menza C, Kendall M, Rogers C, Miller J (2007) A deep reef in deep trouble. Cont Shelf Res 27:2224–2230
Morrison JM, Smith OP (1990) Geostrophic transport variability along the Aves Ridge in the Eastern Caribbean Sea during 1985–1986. J Geophys Res Oceans 95:699–710
National Oceanic and Atmospheric Administration [NOAA] (2001) Benthic Habitats of Puerto Rico and the U.S. Virgin Islands. National Ocean Service
Nelson WR, Appeldoorn R (1985) Cruise report R/V Seward Johnson. A Submersible survey of the continental slope of Puerto Rico and the U.S. Virgin Islands. University of Puerto Rico, 76 pp
Nemeth R, Kadison E (2013) Temporal patterns and behavioral characteristics of aggregation formation and spawning in the Bermuda chub (Kyphosus sectatrix). Coral Reefs 32:1067–1076
Nemeth RS, Herzlieb S, Kadison ES, Taylor M, Rothenberger JP, Harold S (2004) Coral reef monitoring in St. Croix and St. Thomas, United States Virgin Islands year three final report submitted to Department of Planning and Natural Resources. Department of Planning and Natural Resources, United States Virgin Islands, 79 pp
Nemeth RS, Herzlieb S, Blondeau J (2006) Comparison of two seasonal closures for protecting red hind spawning aggregations in the US Virgin Islands. Proc 10th Int Coral Reef Symp Okinawa 4:1306–1313
Nemeth RS, Smith TB, Blondeau J, Kadison E, Calnan JM, Gass J (2008) Characterization of deep water reef communities within the marine conservation district, St. Thomas, U.S. Virgin Islands. Submitted to the Caribbean Fisheries Management Council. University of the Virgin Islands, St. Thomas, 90 p + appendices
Olsen D, LaPlace J (1978) A study of Virgin Islands grouper fishery based on a breeding aggregation. Proc 31st Gulf Caribb Fish Inst 31:130–144
Renchen GF, Pittman SJ, Clark R, Caldow C, Gall S, Olsen D, Hill RL (2014) Impact of derelict fish traps in Caribbean waters: an experimental approach. Bull Mar Sci 90:551–563
Rivera J, Prada M, Arsenault J-L, Moody G, Benoit N (2006) Detecting fish aggregations from reef habitats mapped with high resolution side scan sonar imagery. NOAA Prof Pap NMFS 5:88–104
Rogers C, Miller J, Muller E, Edmunds P, Nemeth R, Beets J, Friedlander A, Smith T, Boulon R, Jeffrey C, Menza C, Caldow C, Idrisi I, Kojis B, Monaco M, Spitzack T, Gladfelter E, Ogden J, Hillis-Starr Z, Lundgren I, Bane Schill W, Kuffner I, Richardson L, Devine B, Voss J (2008) Coral Reefs of the US Virgin Islands. In: Dodge R, Reigl B (eds) Coral reefs of the world vol 1 coral reefs of the United States, vol 1. Springer, New York, pp 303–373
Rowell TJ, Nemeth RS, Schärer MT, Appeldoorn RS (2015) Fish sound production and acoustic telemetry reveal behaviors and spatial patterns associated with spawning aggregations of two Caribbean groupers. Mar Ecol Prog Ser 518:239–254
Sadd JL (1984) Sediment transport and CaCO3 budget on a fringing reef, Cane Bay, St. Croix, U.S. Virgin Islands. Bull Mar Sci 35:221–238
Smith TB (2013) United States Virgin Islands’ response to the proposed listing or change in status of seven Caribbean coral species under the U.S. Endangered Species Act. University of the Virgin Islands 43
Smith TB, Holstein DM (2016) Chapter 3.4. The United States Virgin Islands, USA. In: Baker EK, Puglise KA, Harris PT (eds) Mesophotic coral ecosystems—a lifeboat for coral reefs? The United Nations Environment Programme and GRID-Arendal, Nairobi/Arendal, pp 26–27
Smith TB, Nemeth RS, Blondeau J, Calnan JM, Kadison E, Herzlieb S (2008) Assessing coral reef health across onshore to offshore stress gradients in the US Virgin Islands. Mar Pollut Bull 56:1983–1991
Smith TB, Blondeau J, Nemeth RS, Pittman SJ, Calnan JM, Kadison E, Gass J (2010) Benthic structure and cryptic mortality in a Caribbean mesophotic coral reef bank system, the Hind Bank Marine Conservation District, U.S. Virgin Islands. Coral Reefs 29:289–308
Smith TB, Ennis R, Kadison E, Nemeth RS, Henderson LM (2016a) The United States Virgin Islands Territorial Coral Reef Monitoring Program. 2016 annual report. University of the Virgin Islands, United States Virgin Islands, 286 pp
Smith TB, Gyory J, Brandt ME, Miller WJ, Jossart J, Nemeth RS (2016b) Caribbean mesophotic coral ecosystems are unlikely climate change refugia. Glob Chang Biol 22:2756–2765
Smith TB, Brandtneris VW, Canals M, Brandt ME, Martens J, Brewer RS, Kadison E, Kammann M, Keller J, Holstein DM (2016c) Potential structuring forces on a shelf edge upper mesophotic coral ecosystem in the US Virgin Islands. Front Mar Sci 3:115
Smith TB, Holstein DM, Ennis RS (2019) Disturbance in mesophotic coral ecosystems and linkages to conservation and management. In: Loya Y, Puglise KA, Bridge TCL (eds) Mesophotic coral ecosystems. Springer, New York, pp 911–929
Steneck R (1983) Quantifying herbivory on coral reefs: just scratching the surface or biting off more than we can chew. In: Reaka ML (ed) The ecology of deep and shallow reefs. NOAA Undersea Research Center, Rockville, pp 103–111
Suchanek TH, Carpenter RC, Witman JD, Harvell CD (1983) Sponges as important space competitors in deep Caribbean coral reef communities. In: Reaka ML (ed) The Ecology of deep and shallow coral reefs. NOAA/NURP, Rockville, pp 55–60
Tobias W (1997) Three year summary report: cooperative fisheries statistics program #SF-42 (NA27FT0301–01). Department of Fish and Wildlife, United States Virgin Islands, 41 pp
Turner JA, Babcock RC, Hovey R, Kendrick GA (2017) Deep thinking: a systematic review of mesophotic coral ecosystems. ICES J Mar Sci 74(9):2309–2320
Watlington R (2006) An 1867-class tsunami: potential devastation in the US Virgin Islands. In: Mercado-Irizarry A, Liu P (eds) Caribbean tsunami Hazard, vol 1. World Scientific, New Jersey, pp 255–266
Weinstein DK, Smith TB, Klaus JS (2014) Mesophotic bioerosion: variability and structural impact on U.S. Virgin Island deep reefs. Geomorphology 222:14–24
Weinstein DK, Sharifi A, Klaus JS, Smith TB, Giri SJ, Helmle KP (2016) Coral growth, bioerosion, and secondary accretion of living orbicellid corals from mesophotic reefs in the US Virgin Islands. Mar Ecol Prog Ser 559:45–63
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Smith, T.B. et al. (2019). The United States Virgin Islands. In: Loya, Y., Puglise, K., Bridge, T. (eds) Mesophotic Coral Ecosystems. Coral Reefs of the World, vol 12. Springer, Cham. https://doi.org/10.1007/978-3-319-92735-0_8
Download citation
DOI: https://doi.org/10.1007/978-3-319-92735-0_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-92734-3
Online ISBN: 978-3-319-92735-0
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)