Abstract
Background: Non-invasive measurement of intracranial pressure (ICP) can be invaluable in the management of critically ill patients. Invasive measurement of ICP remains the “gold standard” and should be performed when clinical indications are met, but it is invasive and brings some risks. In this project, we aim to validate the non-invasive ICP (nICP) assessment models based on arterious and venous transcranial Doppler ultrasonography (TCD) and optic nerve sheath diameter (ONSD).
methods: We included brain injured patients requiring invasive ICP monitoring (intraparenchymal or intraventricular). We assessed the concordance between ICP measured non-invasively with arterious [flow velocity diastolic formula (ICPFVd) and pulsatility index (PI)], venous TCD (vPI) and ICP derived from ONSD (nICPONSD) compared to invasive ICP measurement.
Results: Linear regression showed a positive relationship between nICP and ICP for all the methods, except PIv. ICPONSD showed the strongest correlation with invasive ICP (r = 0.61) compared to the other methods (ICPFVd, r = 0.26, p value = 0.0015; PI, r = 0.19, p value = 0.02, vPI, r = 0.056, p value = 0.510). The ability to predict intracranial hypertension was highest for ICPONSD (AUC = 0.91; 95% CI, 0.85–0.97 at ICP > 20 mmHg), with a sensitivity and specificity of 85%, followed by ICPFVd (AUC = 0.67; 95% CI, 0.54–0.79).
Conclusions: Our results demonstrate that among the non-invasive methods studied, ONSD showed the best accuracy in the detection of ICP.
Access provided by CONRICYT-eBooks. Download conference paper PDF
Similar content being viewed by others
Keywords
Introduction
Elevated intracranial pressure (ICP) is an important cause of secondary brain injury and may be associated with a poor outcome [1]. Clinical signs of elevated ICP, such as headache, altered level of consciousness and vomiting, are considered to be non-specific and unreliable predictors of brain damage [2]. The “gold standard” for continuous ICP monitoring is an intraventricular catheter connected to an external pressure transducer [3]. However, the procedure is invasive, and it is at times complicated by infection, haemorrhage, malfunction, obstruction and malpositioning of the catheter [4, 5]. Therefore, non-invasive measurement of ICP (nICP) can be invaluable in the management of critically ill patients [6]. This project is focused on comparing and refining methods for nICP assessment based on arterial and venous transcranial Doppler ultrasonography (TCD) and optic nerve sheath diameter (ONSD) ultrasonography.
Methods
This single-centre non-invasive clinical trial was approved by the institutional ethics committee and written informed consent was obtained from all participants’ next of kin. Patients admitted to the Neurocritical Care Unit, Addenbrookes Hospital, Cambridge, UK, for intracranial diseases (including traumatic brain injury, subarachnoid haemorrhage and stroke) that necessitate monitoring of ICP with continuous intraparenchymal or intraventricular monitoring were enrolled in this study.
Inclusion criteria were age >18 and necessary ICP monitoring in situ (intraparenchimal or intraventricular). Exclusion criteria were a history of optic nerve lesion or previous optic nerve trauma, cerebral venous thrombosis, skull base fracture with cerebral spinal fluid leakage, inaccessible ultrasound window and the absence of informed consent.
ONSD ultrasound measurements were performed and recorded twice a day from day 1 to day 5 of admission, and in the case of high dynamics of ICP (like plateau waves) on event. Doppler velocity in the middle cerebral artery (MCA) and venous TCD on the straight sinus (SS) was simultaneously measured in all the patients.
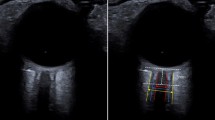
ONSD was measured 3 mm behind the retina [7]. A single investigator used a 7.5-MHz linear ultrasound probe oriented perpendicularly in the vertical plane and at around 30° in the horizontal plane on the closed eyelids of both eyes of supine subjects. Ultrasound gel was applied to the outside of each eyelid and recordings made in the axial and longitudinal planes of the widest diameter visible. Measurements were performed in the transverse and sagittal planes of both eyes, and the final ONSD value was calculated by averaging four measured values.
Arterious TCD was performed on the MCA through the temporal window. TCD measurements were conducted trans-temporally using a traditional 2-MHz transducer as previously described [7].
Venous TCD was performed on the SS through an occipital and transforaminal bone window at a depth of 50–80 mm for flow directed towards the probe, as described by Schoser et al. [8].
Pulsatility index (PI) was calculated according to Gosling’s method [9]: [PI = (FVs − FVd)/FVm]. FVd-based non-invasive ICP (ICPFVd) was derived from the work of Czosnyka et al. [10], in which the authors describe a method for non-invasive estimation of cerebral perfusion pressure (nCPP) in traumatic brain-injured patients:
Non-invasive ICP was estimated as the difference between inflow (ABPm) and non-invasive cerebral perfusion pressure:
PI-derived non-invasive ICP from the SS (vPI) was calculated as [8]:
The value of ONSD-derived ICP was calculated according to our preliminary unpublished results, from the regression plot in a cohort of 23 neurocritical care patients, where invasive ICP was compared with ONSD ultrasound measurements:
Results
A total of 22 patients were included in this study, with a total of 110 measurements. The characteristics of the patients are shown in Table 1. Median value of ICP was 10 (IQR = 17–5.0).
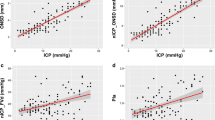
Results from linear regression showed a positive relationship between ICP and nICP calculated with ICPONSD, ICPFVd and PI, but not for vPI (Fig. 1). ICPONSD showed the strongest correlation with invasive ICP (r = 0.61) compared to the other methods (ICPFVd: r = 0.26, p value = 0.001; PI: r = 0.19, p value = 0.02; vPI: r = 0.056, p value = 0.51). Results from the receiver operator characteristic (ROC) curve analysis are shown in Fig. 2.
The ability to predict intracranial hypertension (threshold 20 mmHg) was highest for ICPONSD (AUC = 0.91; 95% CI, 0.85–0.97 at ICP > 20 mmHg), with a sensitivity and specificity of 85%, followed by ICPFVd (AUC = 0.67; 95% CI, 0.54–0.79).
Discussion
According to our results, ONSD showed the best accuracy to assess non-invasively ICP when compared with TCD-based methods. To our knowledge, this is the first study comparing ONSD ultrasonography with different (arterious or venous) TCD methods. Non-invasive ICP is a poorly developed technique, and several attempts have been made in order to find an accurate method to estimate ICP, but so far none of these can substitute the invasive gold standard [6].
The optic nerve sheath (ONS) is continuous with dura mater; as such, the space within the sheath is continuous with the cranial subarachnoid space. When ICP increases, the pressure in the ONS increases linearly, which distends the ONS. Several studies have directly correlated ONSD measurements on ultrasound with ICP measured invasively [6]. The cut-off value for normal ONSD, measured 3 mm posterior to the globe, ranges from 5.2 to 5.9 mm. The sensitivity is 74–95% and the specificity is 74–100% to identify ICP >20 mmHg [11, 12].
TCD is a non-invasive, safe and bedside tool, and it has been used for multiple applications, including detection of changes in cerebral blood flow, vasospasm and circulatory arrest [13]. The non-invasive evaluation of ICP using TCD has also been studied. TCD generates a velocity–time waveform of cerebral blood flow from which the peak systolic (PSV) and end-diastolic (EDV) flow rates can be measured.
The Gosling pulsatility index (PI) has been for many years the most commonly used formula, but many studies have demonstrated that PI value cannot determine the corresponding ICP with an acceptable clinical precision [13, 14]. Similarly, different formulas and mathematical approaches have been proposed for ICP and CPP estimation [10]. Schmidt et al. [15] proposed a new non-PI-related formula for estimation of CPP and therefore ICP based on FVd, which proved that the absolute difference between real CPP and nCPP so calculated was less than 10 mmHg in 89% of measurements and less than 13 mmHg in 92% of measurements in their study. The same group of authors, in another study [10], reinforced the results of the above-mentioned study (correlation between and CPP and measured CPP was r = 0.73; p < 0.0001). Venous TCD ultrasonography is an evolving technique [8]. Within a closed skull, cerebral compliance depends secondarily on the compressibility of the low-pressure venous or capacitance segment of the vascular bed. This venous capacitance segment encompasses 70% of the complete cerebral vascular volume. With progressive increases in ICP, venous blood flow is impaired (venous congestion) before secondary arterial blood flow came to stasis. These venous changes were observed at approximately 50 mmHg of ICP. The main study in this field has been conducted by Schoser et al. [8], who performed venous TCD on 30 control volunteers and 25 patients with raised ICP. Venous blood-flow velocities (BFVs) in the basal vein of Rosenthal showed, within a certain range, a linear relationship between mean ICP and maximal venous BFV (r = 0.645; p < 0.002). Moreover, a linear relationship was found for maximal venous BFVs in the SS and mean ICP (r = 0.928; p < 0.0003) [8].
Conclusions
Non-invasive estimation of ICP is a developing field. The ideal non-invasive ICP monitoring method should be safe, low cost, easily available, suitable for emergency settings and accurate. At present, none of these methods seem to be reliable and accurate enough to substitute the invasive ICP measurement. However, ONSD ultrasonography is a simple repeatable, bedside tool, widely used and, among the methods described here, it seems to have highest accuracy in the detection of ICP.
References
Marmarou A, Anderson RL, Ward JD, Young HF, Marmarou A. Impact of ICP instability and hypotension on outcome in patients with severe head trauma. J Neurosurg. 1991;75:S59–66.
Tayal VS, Neulander M, Norton HJ, Foster T, Saunders T, Blaivas M. Emergency department sonographic measurement of optic nerve sheath diameter to detect findings of increased intracranial pressure in adult head injury patients. Ann Emerg Med. 2007;49:508–14.
Online TBI guidelines 2010. Available at www.braintrauma.org/coma/guidelines. Accessed May 14.
Holloway KL, Barnes T, Choi S, Bullock R, Marshall LF, Eisenberg HM, et al. Ventriculostomy infections: the effect of monitoring duration and catheter exchange in 584 patients. J Neurosurg. 1996;85:419–24.
Hoefnagel D, Dammers R, Ter Laak-Poort MP, Avezaat CJ. Risk factors for infections related to external ventricular drainage. Acta Neurochir. 2008;150:209–14.
Robba C, Bacigaluppi S, Cardim D, Donnelly J, Bertuccio A, Czosnyka M. Non-invasive assessment of intracranial pressure. Acta Neurol Scand. 2016;134(1):4–21. https://doi.org/10.1111/ane.12527. Epub 2015 Oct 30.
Robba C, Bragazzi NL, Bertuccio A, Cardim D, Donnelly J, Sekhon M, Lavinio A, Duane D, Burnstein R, Matta B, Bacigaluppi S, Lattuada M, Czosnyka M. Effects of prone position and positive end-expiratory pressure on noninvasive estimators of ICP: a pilot study. J Neurosurg. 1999;91(5):744–9.
Schoser BG, Riemenschneider N, Hansen HC. The impact of raised intracranial pressure on cerebral venous hemodynamics: a prospective venous transcranial Doppler ultrasonography study. J Neurosurg. 1999;91(5):744–9.
Gosling RG, King DH. Arterial assessment by Doppler-shift ultrasound. Proc R Soc Med. 1974;67:447–9.
Czosnyka M, Matta BF, Smielewski P, Kirkpatrick PJ, Pickard JD. Cerebral perfusion pressure in head-injured patients: a noninvasive assessment using transcranial Doppler ultrasonography. J Neurosurg. 1998;88:802–8.
Rajajee V, Vanaman M, Fletcher JJ, et al. Optic nerve ultrasound for the detection of raised intracranial pressure. Neurocrit Care. 2011;15:506–15.
Moretti R, Pizzi B, Cassini F, et al. Reliability of optic nerve ultrasound for the evaluation of patients with spontaneous intracranial hemorrhage. Neurocrit Care. 2009;11:406–10.
Zweifel C, Czosnyka M, Carrera E, et al. Reliability of the Blood Flow Velocity Pulsatility Index for assessment of intracranial and cerebral perfusion pressures in head injured patients. Neurosurgery. 2012;71:237–9.
Bellner J, Romner B, Reinstrup P, et al. Transcranial Doppler sonography pulsatility index (PI) reflects intracranial pressure (ICP). Surg Neurol. 2004;62:45–51.
Schmidt EA, Czosnyka M, Gooskens I, Piechnik SK, Matta BF, Whitfield PC, Pickard JD. Preliminary experience of the estimation of cerebral perfusion pressure using transcranial Doppler ultrasonography. J Neurol Neurosurg Psychiatry. 2001;70:198–204.
Conflicts of interest statement
DC is supported by a Cambridge Commonwealth European and International Trust Scholarship. JD is supported by a Woolf Fisher Trust Scholarship. XL is supported by a Gates Cambridge Trust Scholarship. BC is supported by a CNPQ Scholarship (Research Project 203792/2014-9). MC and DC are partially supported by NIHR Brain Injury Healthcare Technology Co-operative, Cambridge. For the remaining authors nothing was declared.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this paper
Cite this paper
Robba, C. et al. (2018). Non-invasive Intracranial Pressure Assessment in Brain Injured Patients Using Ultrasound-Based Methods. In: Heldt, T. (eds) Intracranial Pressure & Neuromonitoring XVI. Acta Neurochirurgica Supplement, vol 126. Springer, Cham. https://doi.org/10.1007/978-3-319-65798-1_15
Download citation
DOI: https://doi.org/10.1007/978-3-319-65798-1_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-65797-4
Online ISBN: 978-3-319-65798-1
eBook Packages: MedicineMedicine (R0)