Abstract
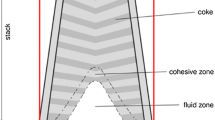
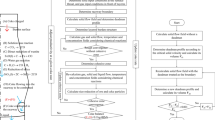
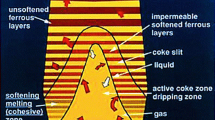
Blast furnaces are counter-current chemical reactors used to reduce iron ore into liquid iron. Hot reduction gases are blasted through a burden consisting of iron ore pellets, slag, flux, and coke. The chemical reactions that occur through the furnace reduce the iron ore pellets into liquid iron as they descend through the furnace. Experimental studies and live operation measurements can be extremely difficult to perform on a blast furnace due to the extremely harsh environment generated by the operational process. Computational Fluid Dynamics (CFD) modeling has been developed and applied to simulate the complex multiphase reacting flow inside a blast furnace shaft. The model is able to predict the burden distribution pattern, Cohesive Zone (CZ) shape, gas reduction utilization, coke rate, and other operational conditions. This paper details the application of this model to investigate the effects of coke size and porosity, iron ore pellet size, and burden descent speed on blast furnace efficiency.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Y. H. Dang, S. M. Zhang, “Discussion on Low Fuel Rate and High PCI Rate of Blast Furnace Operation,” Iron and Steel, 40 (2) (2005).
J. A. De Castro, H. Nogami, J. Yagi, “Numerical investigation of simultaneous injection of pulverized coal and natural gas with oxygen enrichment to the blast furnace,” ISIJ International, 42 (11) (2005), 1203–1211.
D. Fu, Y. Chen, C. Q. Zhou, “Application of the Blast Furnace Shaft CFD Simulator,” Baosteel Academic Steel Conference 2013, Shanghai, China, 10.
D. Fu, Y. Chen, C. Q. Zhou, “CFD Investigation of the Effects of Iron Ore Reducibility and Coke Reactivity on Blast Furnace Operation,” Proceedings of AISTech 2014, Indianapolis, U.S.A., 9.
T. Murayama, Y. Ono, Y. Kawai, “Step-Wise Reduction of Hematite Pellets with CO-CO2 Gas Mixtures,” Tetsu-to-Hagané, 63 (7) (1977), 1099–1107.
Q. T. Tsay, W. H. Ray, J. Szekely, “The Modeling of Hematite Reduction with Hydrogen Plus Carbon Monoxide Mixtures: Part I. The Behavior of Single Pellets,” AIChE Journal, 22 (6) (1976), 1064–1072.
The Iron and Steel Institute of Japan, Blast Furnace Phenomena and Modelling (New York, NY: Elsevier Applied Science Publishers LTD, 1987), 108–116.
J. B. Rawlings, J. G. Ekerdt, Chemical Beactor Analysis and Design Fundamentals (Madison, WI: Nob Hill Publishing, 2002).
S. Kobayashi, Y Omori, “The Chemical Reaction Rate of the Solution Loss of Coke,” Tetsu-to-Hagané, 63 (1977), 1081–1089
N. Miyasaka, N., S. Kondo, “The Rate of Cokes Gasification by Gas Consisting of CO2, H2O, CO, H2, and N2,” Tetsu-to-Hagané, 54 (1968), 1427–1431.
H. Kokubu et al., “Effect of Humidified Blast on Blast Furnace Operation from the Viewpoint of the Softening and Melting Process of Ore Burdens,” Tetsu-to-Hagané, 68 (15) (1982), 2338–2345.
W-K. Lu, “Chemical Reactions in the Blast Furnace as it Happens,” Proceedings of AISTECH2004, Volume I & II, 395–407.
G. Danloy, C. Stolz, “Shape and Position of the Cohesive Zone in a Blast Furnace,” Ironmaking Conference Proceedings, 50 (1991), 395–407.
D. J. Harris, I. W. Smith, “Intrinsic Reactivity of Coke and Char to Carbon Dioxide, Steam, and Oxygen,” Twenty-Third Symposium (International) on Combustion, 23 (1) (1991), 1185–1190.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 The Minerals, Metals & Materials Society
About this chapter
Cite this chapter
Okosun, T., Silaen, A.K., Tang, G., Wu, B., Zhou, C.Q. (2016). CFD Analysis of Blast Furnace Operating Condition Impacts on Operational Efficiency. In: Nastac, L., et al. CFD Modeling and Simulation in Materials Processing 2016. The Minerals, Metals & Materials Series. Springer, Cham. https://doi.org/10.1007/978-3-319-65133-0_10
Download citation
DOI: https://doi.org/10.1007/978-3-319-65133-0_10
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-65132-3
Online ISBN: 978-3-319-65133-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)