Abstract
Infrared thermography presents some important advantages in the determination of skin temperature, as it is a safe, non-invasive and non-contact technique with wide applications in the field of sports sciences. Like many others techniques, valid measurement in thermography requires following strict methodological steps from data acquisition to analyses and interpretation. In this chapter, we discuss the methodological aspects that must be taken into account when acquiring thermic images, along with some practical examples and recommendations based on the current literature.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
3.1 Thermography: Advantages and Limitations
In recent years, infrared thermography has become a popular technique to determine the temperature of human skin during exercise [1–4]. The reasons accounting for its popularization were the technological advances permitting assessment in a variety of sporting conditions and the more accessible price of the cameras. Previously used mostly with engineering applications, thermography became known by sports scientists who detected its potential for field assessment of physical activity and sport. Many recent studies are available presenting data from experiments that can illustrate the different applications of thermography in the assessment of exercise performance [5–7]. However, thermography in sports is still a recent topic and there are many fundamental discussions concerning different methodological aspects (e.g., determination of the regions of interest, strategy for analysis of the data, etc.), protocols of data acquisition, and the interpretation of results concerning its physiological or even mechanical meaning in the context of sports performance. Although the practice of infrared thermography has become more and more popular, these points of concern need further research. The good news is that the implication of the skin temperature information on athletic training is endorsed by basic research. For example, high skin temperatures are known to reduce the temperature gradient between the skin and the core, limiting heat transfer and producing a negative effect on aerobic performance [8, 9].
It is true that infrared thermography presents some methodological advantages , but also has some limitations . All these aspects must to be considered when designing, developing and analyzing data from experiments involving infrared thermography. De Andrade Fernandes et al. [3] described some of the advantages and limitations of using thermography in the assessments of skin temperature in humans. In Table 3.1 we summarize the description of advantages and limitations presented by De Andrade Fernandes et al. [3] and we also include some additional comments concerning the relevance of each advantage and limitation mentioned by the authors.
One important advantage of infrared thermography over other methods to determine skin temperature is that infrared thermography is considered to be a distance technique, which means that the measurement can be performed without interaction with the environment or subject. This advantage is essential in some applications, such as the assessment of electric or nuclear facilities in which the evaluator can analyze the components without being in danger. In the context of human assessment, this characteristic is also beneficial in order to determine skin temperature without interference in the processes of heat exchanges via convection, conduction, radiation and evaporation [3, 10, 11]. This is an inconvenient aspect of the sensors based on thermal contact , such as thermistors and thermocouples (see the circle in the Fig. 3.1). It is known that the interaction between these sensors, the skin and the environment can reduce the reliability of the measurement [12]. Additionally, the method used to attach these sensors to the skin (e.g., the clinical tape used) has been shown to affect the local heat transfer [13, 14], and hence the local thermal regulation and skin temperature might be altered [3, 10, 11, 15].
The non-invasive and non-harmful characteristics of infrared thermography make it a very attractive technique in medical areas, since it allows human physiological study without the risk invasive radiation presents in other imaging techniques (e.g., X-rays) [7, 16]. One attractive application of thermography is to detect injury. However, the association between thermographic data and injury remains debatable [16], although it has been used as an complementary tool in the physical examination [17, 18]. Some physiological disorders are associated with an alteration of skin temperature in the patient [7, 16]. In the context of sports injury, there are some studies discussing the potential associations between local temperature and specific injuries, but conclusions are still vague and further investigation is needed [7, 19].
One possible reason for the lack of relationship with some physiological manifestations of diseases or injury is that infrared thermography allows determination of surface temperatures . While very useful for discussing heat dissipation, inferences on central or tissues temperature are not possible. Some studies used infrared cameras to obtain direct data from deep tissues such as muscles, and this approach is limited due to all the processes of heat exchanges via convection, conduction, radiation and evaporation, that make the temperature from the internal tissues and the superficial skin temperature differ [20]. Furthermore, neuromuscular electrical activity, even measured by surface electromyography in the muscle, does not show a significant relationship with skin temperature in most of the lower limb muscles involved in the pedalling gesture [21]. However, there are many situations in which information regarding skin temperature is useful. The information about heat dissipation in the different body sites and the responses to exercise are important in the context of sportswear design [22] or in the assessment of the capacity of heat dissipation due to the physical fitness level [1, 4].
To draw consistent conclusions, accurate data are essential. To achieve this, a rigorous methodology must be followed [17, 23, 24]. Infrared thermography camera captures the infrared radiation, and the different levels of radiation define a thermal image. However, the proper determination of the temperature from this infrared radiation depends on technical and methodological factors that can influence the capture and the determination of temperatures (e.g., angle and distance of the camera, emissivity, etc.). Therefore, it is very important to strictly follow a protocol for the image acquisition. The main aspects that must be taken into account during image acquisition will be discussed in the next sections.
3.2 Methodology for Infrared Thermography Assessment in Humans
Different organizations proposed standard recommendations for the use of thermal imaging in studies with humans. Some examples are the technical guidelines published in 1986 by the American Academy of Thermology [25], the guidelines for neuromusculoskeletal thermography published by the same association in 2006 [26], and the Glamoral Protocol published in 2008 by the European Association of Thermology [23]. However, different methodological aspects remain unclear and, for this reason, researchers are still working to clarify different issues. To this end, recent scientific articles analyzed specific aspects of the methodology and thermography outcomes in human assessment [11, 27–30]. Taking together guidelines and publications, the main methodological aspects for assessment of humans with thermography should attempt to:
-
1.
Reduce the error of measurement of the infrared thermography camera (minimum error is commonly established by the manufacturers at ±2%).
-
2.
Reduce the variability in the measure of the skin temperature between participants.
-
3.
Increase the reproducibility of the measurements.
To achieve these three methodological concerns, both technical and methodological requirements must be considered and, more importantly, must be carefully administrated to improve the measurements and therefore lead to valid conclusions. The next sections will introduce each of these requirements.
3.2.1 Determination of the Region of Interest (ROI)
Determination of the ROI is one of the most important steps in the design of a thermography study. The determination of the ROI will depend on the purpose of the study. Therefore, the ROI must be determined in the early phase of study design because this will affect other aspects such as the body position during the thermal room adaptation or the position of the camera. The variables of interest, such as descriptive statistics, will be extracted from each ROI obtained from the thermal image. Determination of the ROI is important because it can also help in improving data acquisition quality, especially when camera resolution is limited. ISO normative determines the size of the ROI. To reduce error, a minimum of 25 pixels is recommended [31, 32]. The camera should be placed as close as possible to the ROI in order to increase the number of pixels of measurement [33]. Additionally, the size of the ROI will directly influence the temperature measurement, as a smaller ROI will present a higher temperature than a larger ROI due to the greater influence of hot spots in these smaller areas [11].
The Glamorgan Protocol was published in 2008 with the purpose of standardizing the ROIs determination in thermographic studies [23]. This document presents the definition of 90 different ROIs. Although the Glamorgan protocol provides important guidance in the determination of the ROI, there are a number of studies with very specific demands. Because of this, researchers have developed their own criteria to define ROIs, with different geometries and methodologies [24, 30]. Differences in skin temperature between ROIs are due to the tissue composition, muscular activity and capacity of sweating [21, 29, 30, 34], and these factors are important to take into account during the definition of the ROI.
The reproducibility in the determination of the ROIs for different images is another important factor in ensuring the accuracy of the results, especially when different subjects are compared. Different strategies were observed in the literature in order to increase the reproducibility of thermographical data. Some researchers use software with an automatic ROI selection feature [22, 35, 36]. However, these software are not accessible to all users, and for this reason one of the most used strategies is the determination of ROI according to anatomical proportions [21] or body segments [37–39].
Mathematical methods can also be used in the determination of ROIs. One example of this is the Tmax method, which was proposed by Ludwig et al. [27]. In this method, the operator selects an ROI and the software will automatically select the five warmest pixels within the ROI, with a minimum distance of at least of five pixels between each. The software selects an area of five × five pixels around each of the five pixels previously selected, determining the mean temperature across the 125 pixels included in the selection [27]. These authors suggested that the main advantage of this method is the use of the same number of pixels for the calculation, removing the effect of the different anatomical sizes between participants [27].
It is also possible to determine the ROIs using markers in anatomical references or outer delimiting areas of the body surface (Fig. 3.2) [7, 29]. This strategy can be useful in ensuring the reproducibility in the delimitation of the ROI and it facilitates further data analysis. After this recapitulation of information, we can provide some overall recommendations for the determination of ROIs:
-
To record the maximum number of pixels of the ROI in the thermal image, put the camera as close as possible to the region (always considering that the image must be focused). A higher number of pixels will provide more robust descriptive statistical data relating to the ROI.
-
Whenever the objective of the study permits, use large ROIs instead of small ROIs. The large ROIs will reduce the possible effects of punctual points of the ROI such as hot spots.
-
To reduce the subjective aspect for the delimitation of the ROIs as far as possible, increase the reliability. Software with automatic ROI selection, the use of markers in the skin, or the delimitation using anatomical references are strategies that can help in this objective.
-
To determine the ROIs, take aspects like tissue composition, muscular activity and capacity of sweating into account. In this sense, we recommend avoiding measurements in the same ROI, regions in which the muscles are close to the skin (e.g., thigh or leg) and regions with a higher proportion of bone and connective tissue close to the skin (e.g., knee or Achilles).
3.2.2 Methodological Aspects of the Camera
The choice of camera is an important step in the use of thermography. Cameras for thermographical assessment can be cooled or uncooled cameras (Fig. 3.3). Cooled cameras normally operate at cryogenic temperatures [40]. The older cameras were cooled by the addition of liquid nitrogen, argon gas or sterling cooler, resulting in a limitation of the angle of operation of the camera [40–42]. In this case, a high camera angle could put the liquid nitrogen or other cooling system in contact with the electronics and cause damage. Other cooling systems (e.g., thermoelectric systems) were introduced to overcome the angle problem of the camera [40, 41]. The cooling system is usually associated with power and time taken to cool the camera, and it is bulky and expensive [33, 40]. However, the cooled detectors provide a superior image quality and a greater sensitivity to small differences in the scene temperatures than the uncooled detectors [33, 40]. The more recent generation of uncooled cameras can be used without cooling, but using a microbolometer [42]. These cameras will be stabilized at ambient temperatures using temperature control elements (based for example on changes on resistance and voltage) to reduce the image noise [40]. Although these cameras provided a lower quality of image and have lower sensitivity, they are less expensive in price and maintenance, smaller and easier to use [40, 41].
Also related to the choice of the camera, infrared resolution is an important technical aspect to be considered (Fig. 3.4). Although infrared resolution is not the most important factor in the accuracy of the calculation of the temperature [43], it is always preferable to have a camera with the best resolution that you can afford [24]. Each pixel in the thermal image represents one thermal data, and a higher number of pixels will result in a more robust measure when average temperature is calculated in the ROI. One example of this is that 25 is considered to be the minimum number of pixels to determine average temperature for a region of interest for the ISO normative [31, 32]. In this regard, an infrared resolution of 320 × 240 pixels is commonly considered to be the minimum resolution necessary for satisfactory data in the human assessments [7, 24].
For sports science applications in particular, the capacity to record thermographic video is an important feature in the thermographic camera . There are a few studies using this feature in sport assessment [2, 44, 45]. Despite the higher cost and specific configuration required for proper image acquisition during thermographical video records, the calculation of the temperature in an sports assessment could be disturbed by nearby electronic equipment (e.g., ergometer, computers, etc.) [41]. This is the reason why researchers usually try to analyze static images recorded before and after exercise [11, 46].
As observed in many other instruments, a proper calibration is also necessary for thermographical assessment. Infrared cameras are calibrated by the manufactures with a high number of black body sources at different temperatures when the signal of each pixel is determined [47]. A black body source is an instrument with a stable and accurate temperature surface, with a high emissivity, configured with a specific temperature emission (Fig. 3.5). These black body sources usually have an emissivity as high as 0.98 [47]. In addition to the manufacture calibration, in research studies, the camera usually is calibrated again before every experiment starts. To do this, the black body sources need time to stabilize temperature before the calibration. The calibration is more precise when the black body source is set at a higher temperature than the room temperature, and it is important to know the accuracy and the emissivity of the black body source in order to perform the best calibration possible. For example, black body source can be configured to some specific temperature emission (e.g., 50 °C). When the black body is stabilized, thermography camera can record a thermal image where is the black body. After that, we can analyze the difference between the temperature of the black body source in the thermography and the set temperature of the black body source. If the difference is inside of the accuracy of black body source, the camera is measuring correctly, but if we have a higher difference, we can compensate this difference in the data analysis. However, if the difference is very high it is recommendable to send the camera to the manufacturer for calibration. In addition, the black body (and also a black plate thermistor) can be placed in the space of measurement in order to have this reference in the image, which also permits any drift in the temperature sensitivity during the laboratory daily routine to be checked and increases reliability [17, 41] (Fig. 3.6).
Another important aspect to consider is that the camera should be turned on some time before the measurements start. Infrared thermography cameras have a condenser to stabilize the electronics and compensate its temperature variations. However, it needs time to adjust to the environmental conditions and to work properly. The time necessary for this stabilization may differ between camera models. For example, the minimum time determined for the uncooled camera used by the authors of this chapter was 5 min [30]. However, since their camera measurements became stable after 5 min of turning on the camera, they determined a 10 min stabilization period to ensure this process [30]. There are cameras that may need a minimum of 10 min to stabilize [47]. The most common procedure to ensure camera stabilization time is to set some system with a stable temperature (black body source or hot plate system), turn on the camera and record thermal images continuously. When there is no variation in the temperature data with the time, it means that the camera is stable.
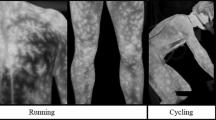
The position of the camera ( distance and angle in relation to the ROI) can affect the measurements and needs to be controlled [48–50]. The camera lens should be placed perpendicularly to the ROI. Angles higher than 60º between the camera and the ROI considerably modify the data [50]. It is important to take all the thermal images of one study at the same distance to minimize these errors. In this sense, Tkáčová et al. [50] observed differences of 0.2 °C between measurements performed between 0.2 and 2.5 m between the camera and the ROI. Although the distance is an important parameter to control, it also depends on the infrared resolution of the camera and the ROI (Fig. 3.7). Most of the studies with humans registered the measurements at distances between 1 and 3 m [1, 3, 11, 22, 30, 39, 46]. The camera should be placed as close as possible to the ROI and record the highest number of pixels in that region [33]. In order to have some reference values, a distance of 1–1.5 m it can be enough for a ROI that is a body segment (Fig. 3.7a), and a distance of 3–4 m it can be the necessary when the ROI is all the whole surface of the subject body (Fig. 3.7b). Similarly, when taking thermographic images, the focus of the camera must be adjusted. Errors in the focus adjustment consistently produce errors in the temperature calculation of the ROI (see details in the Fig. 3.8).
3.2.3 Methodological Aspects Related to the Space Where the Measurement Will Be Conducted
Considering the space measurement, the main methodological aspects to take into account are the room space, the room temperature, the relative humidity, and all sources of infrared radiation that might affect the thermographic measures by standing around the subject. The parameters of the room temperature and its relative humidity must be informed in the camera setup. The minimum space dedicated for the measurement is 6–9 m2, but spaces of 12 m2 are preferable [41, 47]. The room temperature is one of the most important factors affecting skin temperature, because both are directly related [51–53]. Different authors have suggested that room temperature should be between 18 and 25 °C during thermography assessment with humans [42, 49]. This is because the room temperatures outside of this range could affect the thermoregulation mechanisms, resulting in sweating when the room temperature is higher than 25 °C, or shivering when the room temperature is lower than 18 °C [42]. Furthermore, temperature variation in the room should be no higher than 1–2 °C. When data from measurements made in the trunks of 64 male cyclists were analyzed, it was observed that an increase of 1 °C in the room temperature resulted in 0.35 °C higher skin temperature (r = 0.5 and p < 0.01). This influence was minimized when temperature variations (difference before and after) were determined (Fig. 3.9). Ring and Ammer suggested that an environmental temperature of 20° is better for visualizing inflammatory injuries [41]. Therefore, the ideal environmental temperature for thermographical measures may depend on the purpose of the experiment.
The relative humidity in the room where the measurements are taken affects the sweat evaporation rate during physical activity and sport performance [54]. The influence of relative humidity can be higher when the camera is positioned far from the ROI [47]. It is recommended to control relative humidity to values between 40 and 70% [1–4, 11, 22, 38, 46, 55, 56]. It is also important to avoid the presence of particles in the air, such as dust or vapor, because these can absorb the radiation emitted by the participant and therefore cause measurement errors because all the infrared radiation of the participant does not arrive at the camera.
Reflected temperature is defined as the temperature of other objects that are reflected by the target into the infrared thermography camera [57]. This temperature should be measured and informed during camera setup configuration. The software of the camera will disregard this parameter from the calculation of temperature of the ROI. It is recommended to measure the temperature reflected in each test. The reflected temperature can change when the environmental conditions change (e.g., variation in the room temperature), the conditions of the evaluator (e.g., changes in clothing) or any feature of the space changes. One of the most used methods to measure the reflected temperature is the “reflector method” described in the international normative ISO 18434-1:2008 [57]. This method consists of:
-
1.
Informing the following parameters to the camera: distance with a value of 0 and an emissivity with a value of 1.
-
2.
Positioning cardboard with aluminum foil at the same level as the participant (Fig. 3.10a).
-
3.
Measuring the average temperature of the aluminum foil (using a rectangle ROI) (Fig. 3.10b).
-
4.
Introducing the thermal average obtained, such as the reflected temperature.
The presence of infrared radiation sources close to the measurement space should be avoided. Infrared radiation sources include the incidence of sunlight in the space of measurement, the presence of electronic devices and the number of people in the measurement space (evaluator and participant). Other sources of radiation are heating ducts, water pipes, heating lights and airflow. Likewise, reflections of infrared radiation around the participant should be avoided, so we recommend placing anti-reflective material behind the plane of interest [7, 47].
3.2.4 Methodological Aspects Related to the Person
Considering the characteristics of the subject going to be evaluated, one of the factors to take into account is the emissivity of human skin. Different studies determined the skin emissivity and described values between 0.97 and 0.99 [58–60]. Thermographic studies with humans commonly consider an emissivity of 0.98 [1, 3, 7, 21, 22, 61]. These studies used this emissivity value for all the participants due to the lower variability observed—standard deviation of 0.01 [58, 60]. This standard deviation in the emissivity resulted in approximately ±0.05 °C or ±0.12% of change in the absolute temperature, and such error will be smaller than the thermal sensitivity of the camera or the variability of the skin temperature between participants and trials. These values of emissivity of the skin means that humans are a good emitter of infrared radiation, and therefore we can obtain accurate surface temperatures [59, 62]. The emissivity value must be introduced into the camera or thermographic software for the correct calculation of the temperature.
The thermal variability of the human skin temperature can be very high due to a great number of factors, such as the individual metabolism, blood flow, adipose tissue, etc. [24]. The present chapter has an applied orientation, and for this reason the factors will be classified considering the experimental process and when they will be taken into account: (1) in the design of the experiment; (2) in the criteria for selection of the participants; and (3) in order to elaborate the instruction that participants need to follow (Fig. 3.11). Some of the points related to the selection of the participants or instructions they will receive can be interpreted in a different classification depending of the researcher. For example, some researches will consider it important to classify the participant as a smoker or non-smoker in the inclusion criteria for the research, while for others such information will be pertinent when sending the instructions to the subject before the assessment. For this reason, it is important to understand that the authors of the present chapter do not think that their classification is the only one to be considered, but rather is the classification considered suitable for the purpose of this chapter. On the other hand, the most important aspects in which the thermography evaluator can act were addressed in this section. Other factors, such as genetics, were not addressed in this chapter.
3.2.4.1 Factors to Take into Account in the Design of the Assessment Protocol
Some factors are essential to take into account in order to obtain the maximum reproducibility in the accomplishment of thermographic measurements. The temperature of the skin usually presents high variability, which may limit the comparison of results between and within subjects. Therefore, some general recommendation can be followed to reduce these intervenient factors.
Room time adaptation
One of the most important methodological aspects is the time required to achieve a stable measurement of skin temperature in a controlled environment [28, 41]. When the individual arrives at the measurement space (typically he/she comes from a different environmental temperature) and prepares for the thermographic measurements (e.g., undressing), it will produce a change in thermal conditions, and hence a variation in the skin temperature. Therefore, an adaptation period is necessary; otherwise the repeatability of the measurements will be very low because the external conditions of each day and each subject are different. During this adaptation time, the participant must avoid all the movements and contact that can affect the skin temperature of the ROIs; for instance, actions such as folding or crossing the extremities, placing bare feet on a cold surface, etc. [41]. In this sense, the use of a carpet or rug in the ground is recommended. Studies commonly use room time adaptation ranging from 10 to 20 min [4, 11, 21, 38, 56, 63, 64]. Longer periods are not recommended because when the time adaptation exceeds 30 min, the temperature oscillation are higher and may result in thermal asymmetries [28, 41]. Marins and colleagues concluded that a minimum time of 10 min is suitable for temperature stabilization, but they suggested that longer times can be required when participants come from extreme environments (cold or hot) [28].
Body posture
Body posture during the image acquisition will be dependent on the ROI defined. Some examples and guidelines are present in the catalogue of ROIs described in the Glamorgan Protocol [23]. In this sense, the anatomical position is usually a very common position for thermal measurement using infrared thermography. However, there will be some cases not considered in that protocol and, for these cases, special attention should be given. The body posture needs to be the same during the whole room time adaptation [41]. In some exercises, it is possible to take the thermal images during the moments of force production or along the movement (e.g., in a static contraction or during a body segmental movement), [4, 64, 65]. We do not recommend comparison of thermal images taken in different body postures because the posture changes the body surface exposed to the environment and then the skin temperature may also be changed [17, 41, 66] (Fig. 3.12).
Effect of the body posture on the heat flow. Figure obtained from Houdas and Ring [66]
Circadian rhythm
The circadian rhythm affects the core temperature [67, 68], skin blood flow [67] and skin temperature [69, 70]. Skin temperatures are lower in the morning and increase during the day [69, 70]. The hands are the body region with the highest increase in skin temperature during the day [69, 70]. These effects of skin temperature could be related to the same trend observed by Smolander et al. about the relationship between skin blood flow and the circadian rhythm [67]. In order to minimize this effect, some studies have performed all measurements in the same period of time [1, 3, 4, 27, 38]. Another possible strategy to reduce the intra-subject effect of the circadian rhythm, when the study has a cross-over experimental design, is to ensure that measurements of each participant are always performed at the same time of the day [55, 56].
Sweat
Moistening of the skin surface due to perspiration may influence thermographic data [11]. Ammer [71] suggested that a film of water on the skin may act as a filter for infrared radiation and that could lead to an error in the estimation of the skin temperature. Removing the sweat or water from the skin has been tried as a solution [72, 73]. These studies [72, 73] tried to remove the sweat without friction of the skin. However, in other studies, temperature could increase as a consequence of rubbing the skin surface and also as a result of the reduction of the natural process of sweat evaporation [11]. On the other hand, the results of one study indicated that the accumulation of sweat during cycling is not enough to form a film of water, and that sweat produced under these conditions does not affect the thermographic data [11]. However, the influence of the sweat on the skin emissivity in infrared measurements as well as the possible strategies to deal with the effect of sweating on skin emissivity during thermographic examinations are unclear to date. Hence, future studies are necessary to explore this methodological aspect in more detail.
3.2.4.2 Factors to Take into Account in the Criteria Selection of the Participants
In order to reduce the variability of the skin temperature results, many factors should be considered. It starts with the definition of the criteria for the selection of the participants. Some of the most relevant factors are commented.
Sex
Women present higher core temperature [68, 74], lower whole-body sweat rate [75] and higher percentage of body fat than men, which influences heat loss and results in lower skin temperature in most of the body regions [37, 76]. The lower heat production during exercise presented in women is another explanation for these lower skin temperatures [77]. These gender effects are more evident in the morning than in the evening, possibly because the blood flow is lower and then the thermal insulation effect of the body fat is higher [70]. It is also important to consider the influence of the menstrual cycle. Different studies have shown that core temperature [78, 79] and skin blood flow [80] were higher during the luteal phase than in the follicular phase resulting in a higher skin temperature in the women.
Age
Older people present lower basal values of skin temperature [51, 64, 81] and after an intervention aimed at increase body temperature (e.g., exercise), a lower response of skin temperature was found among the elderly [51, 64]. The reasons for the skin temperature decreases with age may be related to a lower metabolic rate [82], lower core temperature [68], and the lower capacity of heat dissipation via vasodilation/vasoconstriction [51, 83] and sweat rate [82, 84].
Body surface and body composition
Body surface has an important role on whole-body heat exchange [85, 86]. Individuals with a greater body surface have a greater capacity for heat dissipation [85]. This happens because they have a greater skin surface and then greater absolute rates of convection, radiation and sweat evaporation [86]. For this reason, in order to compare different groups or participants, it is recommended that some body surface variables be normalized (e.g., sweat rate; mg cm−2 min−1). Body surface is usually measured using predictive equations considering the height and body mass, and one of the most used equations is the DuBois and DuBois’ equation [87]. In relation to body composition, the level of body fat influences the capacity for heat loss resulting in lower skin temperatures [88]. Body fat tissue had an insulation capacity resulting in impairment in heat dissipation between the core and the skin [46, 88, 89].
Injuries
An injury is often related to variations in the regional blood flow. Changes in blood flow affect skin temperature, which can increase in the case of an inflammation, or decrease in the case of tissues with poor perfusion, degeneration or reduced muscular activity [7, 42]. Individuals with overuse and traumatic injuries could present with an alteration of skin temperature and thermal symmetry [7, 42] (Fig. 3.13a). In addition, other disorders such as diabetic neuropathy, fever and vascular disease, among others, can influence skin temperature [16] (Fig. 3.13b). For this reason, studies selected participants without history of injuries in the previous months [3, 56, 90]. This is certainly a fertile topic for further investigation, since the medical application of the thermography, as mentioned before, has many advantages over other imaging methods.
Physical fitness level
Physical fitness influences skin temperature [1, 2, 4, 21, 46]. Larger heat loss is observed among participants with better physical fitness, which could be explained mainly through the higher capacity of evaporation of sweat during exercise [46, 91]. Moreover, trained people usually have greater capacity for heat transference between the core and the skin due to higher blood flow and lower body fat [21, 46, 89, 92]. The variability of fitness level will result in a high variability of the skin temperature if considering a heterogeneous group.
Medicaments
Medicaments could alter skin blood flow and then skin temperature. It is known that different kinds of medicaments such as analgesics, anti-inflammatories, vasoactives, hormonal medications, prophylactics and anesthetics can affect the normal values of skin temperature [24]. Furthermore, infrared thermography was used in different studies as a method to determine the response of a medicament [93–96]. In human assessment, when the objective of a study is not to assess a medicament, it is common to recruit participants without a medicament prescription or to avoid the intake of medicaments before a study in order to remove this factor [1, 4, 38, 90].
-
Factors to Take into Account in Instructing Participants
It is suggested that participants be instructed by means of a previous session or by means of a written letter or email containing the information and procedures necessary to avoid possible measurement errors. When possible, a familiarization session can be very valuable in order to fully address all the factors that can influence measurement. Some of the most important factors are described below.
Nutrition
It is commonly established that after food intake, blood supplies the stomach to the detriment of the skin, resulting in a lower skin temperature in the extremities [97, 98]. Heavy meals approximately 4 h before the measurement should be avoid for the human thermography assessments [1, 21, 30].
Hydration and drinks
Prior to the thermographical assessment, it is important to ensure that the subject is adequately hydrated. Hipohydration can affect the sweat rate response and then the skin temperature, as detected by infrared thermography [99]. On the other hand, intake of different liquids has been associated with alteration of skin temperature. For example, alcohol intake increases skin temperature due to the increase of the skin blood flow [100–103]. Liquids with caffeine, and others stimulants such as teas, stimulate the physiological function, resulting in an increase in skin temperature [104–106]. For these reasons, participants of the studies are usually requested to avoid ingestion of alcoholic or caffeine at least 4 h [1, 27, 38] or, preferentially, 12 h before the measurements [21, 30, 56].
Tobacco
It is well established that nicotine has a vasoconstrictive effect and therefore it may reduce skin temperature [107–109] (Fig. 3.14). In addition, it is known that smokers have a reduced circulatory system compared with non-smokers (e.g., they have impaired capillary recruitment and impaired vasodilation) [109]. Thus, it is usually recommended that thermographic studies do not include smokers [1, 38], or that smokers are instructed to avoid smoking for 12 h before the measurement [21, 30, 56].
Hair
The presence of the hair on skin surface can alter the estimation of the skin temperature using infrared thermography [24]. Hair is an avascular tissue [110] with a low heat capacity and then with a lower temperature [111] (very similar to room temperature). For this reason, if there is a high hair density in the skin, this hair could be considered to be an artifact resulting in an underestimation of the skin temperature (Fig. 3.15). For this reason, some studies asked for participants to remove their body hair before the study [1, 4, 27, 38]. However, this could be inconvenient during participant recruitment.
Application of cosmetics on the skin
This group of variables is especially important since they influence the radiation emitted by the skin and are difficult to detect. The use of cosmetics, creams, gels or any other type of skin treatment are relevant to the analysis of thermographic images, since they will act as a filter on the skin, distorting its radiation, influencing the emissivity and resulting in an underestimation of the skin temperature [62]. The different studies usually avoid the use of skin lotions or other cosmetic products in the day of the measurement [1, 4, 21, 27].
Physical activity
Exercise recovery is a process in which the body works to restore the different physiological processes that occurred in the body during exercise, such as muscle damage, DNA damage, oxidative stress, among others [112, 113]. In relation to the skin temperature, Fernández-Cuevas et al. observed that, after a training session, there is a progressive increase in the skin temperature up to 6 h after exercise [35]. This idea that exercise affects skin temperature a posteriori is an important factor to take into account, and for this reason the studies should request that participants avoid physical exercise on the day of the study [90] and also refrain from high intensity exercise the day before the study [1, 21, 55].
Sunbathing and UV rays
The exposure of the skin to sunbathing and UV radiation results in a higher absorption of radiation by the skin, and then in an increase of skin temperature [114]. A good recommendation for the participants in a human thermography assessment is to refrain from sunbathing or from being exposed to UV rays 12 H before the test [21, 30, 56].
3.3 Analysis of the Thermal Data
The analysis process in thermography data still very conservative, as its use in the field of exercise science has only recently begun to grow. Before presenting conclusions from thermal data analyses, there are some important and mandatory steps for proper data analysis.
3.3.1 Qualitative Representation
The thermographic analysis can involve qualitative and quantitative approaches . Qualitative analysis consists of the representation and description of the images obtained. Although a quantitative analysis of the data is fundamental to drawing consistent conclusions, the qualitative analysis is very important for checking whether there is any factor that should be considered in the quantitative analysis. For example, in the qualitative analysis we can see that the assessed person has a varicose vein in the legs and therefore it is very possible that there is a thermal asymmetry. Because of this, it is necessary to always perform a qualitative analysis to ensure the correct interpretation of the numerical data. In addition, this analysis can provide a much faster feedback to the concerned subjects: the athlete, the coach or the medical staff.
Another utility of qualitative analysis is to obtain a graphical representation of what is being evaluated. In this sense, it is important to know the different types of representation currently offered by the thermographic software. Infrared cameras usually have an incorporated digital visual lens that provides interesting visualization tools. Besides the infrared image (Fig. 3.16a), infrared images superimposed on the visual can also be obtained (Fig. 3.16b) as well as an image that integrates infrared and the visual (Fig. 3.16c). According to the camera, the software, and how both correct the differences between lens in position and other characteristics, the overlap is more or less accurate.
Some of the recommendations usually performed for the qualitative representation of images with humans are listed below [42, 47]:
-
Use of a rainbow palette, with white-red as hot and blue-black as cold.
-
Use of an image range defined with a minimum of 23–24 °C and a maximum of 36–38 °C. In addition, use of the same image field for all the comparable images b comparable.
3.3.2 Quantitative Analysis
Different variables can be obtained from analysis of the ROI in the thermographic analysis, such as the average temperature, the maximum temperature, the minimum temperature and the standard deviation. In addition, it is possible to calculate other variables such as the variation (Δ) temperature or variation of the skin temperature (difference between before and after an intervention), the thermal symmetry (difference between both body hemispheres) and the Δ temperature of the ROI (difference between the maximum and minimum values). Different studies used different variables for the analysis of thermal images, and there are many still many open questions in this regard. The more common approaches to quantify thermal data are discussed below.
Average temperature is the most used skin temperature variable in the literature [1, 2, 22, 46, 64]. The advantage of using the average of skin temperature in large ROIs is that it can be a representative value and removes the effects of punctual higher values that can be a source of errors. However, when variations are important, the average temperature may not provide the experiment with significant information [30, 55, 56].
Maximum temperature is another thermal variable used in thermography studies [19, 115, 116]. Some previous studies with animals suggest associations between maximal temperature and injury [115, 116], but this needs to be further researched in humans. Related to the maximum temperature is the Tmax method suggested by Ludwig et al. [27], which is described in the Sect. 3.2.1 of this chapter. A positive feature of this method is that, using it, all the ROIs will have the same number of pixels for the calculation, which minimizes the problem of the differences in the anatomical size between participants.
Temperature variation , as produced by exercise, has been suggested as a valid measure for determining the effects of different interventions in studies using repeated measures design [30, 55, 56]. This idea is supported by some studies that observed differences in skin temperature variation after interventions without differences when looking at the absolute magnitudes of temperature [4, 56].
Thermal symmetries were explored in different studies assessing the normal thermal behavior of the participants or the risk of injuries [65, 117, 118]. Vardasca et al. [117] defined thermal symmetry as “the degree of similarity between two areas of interest, mirrored across the human body’s longitudinal main axes which are identical in shape, identical in size and as near identical in position as possible”. Thermal symmetry assessment is considered a valuable method to assess the physiological normality/abnormality in sport medicine [7, 117], because asymmetries higher than 0.5–0.7 °C are usually associated with a dysfunction in the musculoskeletal system [117, 119, 120].
Δ temperature of the ROI can be an useful measurement for exploring thermal gradients [121, 122]. For example, forearm-finger skin temperature gradient is usually considered to be an index of peripheral circulation and vasomotor tone [121].
Finally, the mean skin temperature is the most common variable used in the thermal studies with thermal contact sensors [123]. This variable is determined using different equations and including temperatures from different body regions [123, 124]. There are equations that make the calculations with temperature data from only three regions, whereas other equations that are considered more accurate will need temperature data from up to 15 regions [123, 124]. There is no clear conclusion about how many measurements represents the ideal condition. There are studies showing good results with different numbers of measurements with data from, for example, seven [124] or four [29] measurements. Although this variable is not commonly used in thermography studies, it is possible to measure infrared thermography in the same way, and it can be useful in studies when the main purpose is to obtain an overall value of skin temperature for the whole body.
Other authors suggested that skin temperature should be normalized using the difference between each body regions with one specific body region, rather than considering only absolute magnitudes of temperature [125, 126]. The aim of this normalization is to reduce the variability of the absolute temperatures [125]. Forehead [125] and chin [126] were some of the body regions used as references, probably due to the consideration of these regions as more temperature stable regions.
All of these methods described, in addition to another methods, such as the spectral analysis of the image, need to be investigated in order to elucidate which method is the most suitable for each situation.
3.4 Conclusions
In this chapter, we tried to briefly present the major concerns and implications of selecting a methodology for determining human skin temperature from measurements using infrared thermography. It is possible to note that thermography has many applications in the context of human movement, but more research is needed to determine the adequate procedures for data acquisition and analysis. Furthermore, unlike other techniques for the assessment of human movement, there are significant efforts prior to data acquisition to ensure that the participant will arrive in a condition that minimizes the influence of intervenient factors in the assessment.
References
Abate M, Di Carlo L, Di Donato L et al (2013) Comparison of cutaneous termic response to a standardised warm up in trained and untrained individuals. J Sports Med Phys Fitness 53:209–215
Akimov EB, Son’kin VD (2011) Skin temperature and lactate threshold during muscle work in athletes. Hum Physiol 37:621–628
de Andrade Fernandes A, dos Santos Amorim PR, Brito CJ et al (2014) Measuring skin temperature before, during and after exercise: a comparison of thermocouples and infrared thermography. Physiol Meas 35:189
Formenti D, Ludwig N, Gargano M et al (2013) Thermal imaging of exercise-associated skin temperature changes in trained and untrained female subjects. Ann Biomed Eng 41:863–871. doi:10.1007/s10439-012-0718-x
Arfaoui A, Polidori G, Taiar R, Popa C (2012) Infrared Thermography in Sports Activity. Infrared Thermogr 141–168
Bertucci W, Arfaoui A, Janson L, Polidori G (2013) Relationship between the gross efficiency and muscular skin temperature of lower limb in cycling: a preliminary study. Comput Methods Biomech Biomed Engin 16(Suppl 1):114–115. doi:10.1080/10255842.2013.815902
Hildebrandt C, Raschner C, Ammer K (2010) An overview of recent application of medical infrared thermography in sports medicine in Austria. Sensors 10:4700–4715
Sawka MN, Cheuvront SN, Kenefick RW (2012) High skin temperature and hypohydration impair aerobic performance. Exp Physiol 97:327–332. doi:10.1113/expphysiol.2011.061002
Cuddy JS, Hailes WS, Ruby BC (2014) A reduced core to skin temperature gradient, not a critical core temperature, affects aerobic capacity in the heat. J Therm Biol 43:7–12. doi:10.1016/j.jtherbio.2014.04.002
van den Heuvel CJ, Ferguson SA, Dawson D, Gilbert SS (2003) Comparison of digital infrared thermal imaging (DITI) with contact thermometry: pilot data from a sleep research laboratory. Physiol Meas 24:717. doi:10.1088/0967-3334/24/3/308
Priego Quesada JI, Martínez Guillamón N, Ortiz Cibrián, de Anda RM et al (2015) Effect of perspiration on skin temperature measurements by infrared thermography and contact thermometry during aerobic cycling. Infrared Phys Technol 72:68–76. doi:10.1016/j.infrared.2015.07.008
Smith ADH, Crabtree DR, Bilzon JLJ, Walsh NP (2010) The validity of wireless iButtons and thermistors for human skin temperature measurement. Physiol Meas 31:95–114. doi:10.1088/0967-3334/31/1/007
Buono MJ, Ulrich RL (1998) Comparison of mean skin temperature using “covered” versus “uncovered” contact thermistors. Physiol Meas 19:297–300
Psikuta A, Niedermann R, Rossi RM (2013) Effect of ambient temperature and attachment method on surface temperature measurements. Int J Biometeorol 1–9
Tyler CJ (2011) The effect of skin thermistor fixation method on weighted mean skin temperature. Physiol Meas 32:1541–1547. doi:10.1088/0967-3334/32/10/003
Lahiri BB, Bagavathiappan S, Jayakumar T, Philip J (2012) Medical applications of infrared thermography: a review. Infrared Phys Technol 55:221–235
Ng E-K (2009) A review of thermography as promising non-invasive detection modality for breast tumor. Int J Therm Sci 48:849–859
Fitzgerald A, Berentson-Shaw J (2012) Thermography as a screening and diagnostic tool: a systematic review. N Z Med J 125:80
Bouzas Marins JC, de Andrade Fernandes A, Gomes Moreira D et al (2014) Thermographic profile of soccer players’ lower limbs. Rev Andal Med Deporte 7:1–6. doi:10.1016/S1888-7546(14)70053-X
Priego Quesada JI, Martínez N, Salvador Palmer R et al (2016) Effects of the cycling workload on core and local skin temperatures. Exp Therm Fluid Sci 77:91–99. doi:10.1016/j.expthermflusci.2016.04.008
Priego Quesada JI, Carpes FP, Bini RR et al (2015) Relationship between skin temperature and muscle activation during incremental cycle exercise. J Therm Biol 48:28–35. doi:10.1016/j.jtherbio.2014.12.005
Fournet D, Ross L, Voelcker T et al (2013) Body mapping of thermoregulatory and perceptual responses of males and females running in the cold. J Therm Biol 38:339–344. doi:10.1016/j.jtherbio.2013.04.005
Ammer K (2008) The Glamorgan Protocol for recording and evaluation of thermal images of the human body. Thermol Int 18:125–144
Fernández-Cuevas I, Bouzas Marins JC, Arnáiz Lastras J et al (2015) Classification of factors influencing the use of infrared thermography in humans: A review. Infrared Phys Technol 71:28–55. doi:10.1016/j.infrared.2015.02.007
A Committee on Quality Control and Qualifications of the American Academy of Thermology (1986) Technical guidelines, Edition 2. Thermology 2:108–112
Schwartz RG (2006) Guidelines for neuromusculoskeletal thermography. Thermol Int 16:5–9
Ludwig N, Formenti D, Gargano M, Alberti G (2014) Skin temperature evaluation by infrared thermography: comparison of image analysis methods. Infrared Phys Technol 62:1–6
Marins JCB, Moreira DG, Cano SP et al (2014) Time required to stabilize thermographic images at rest. Infrared Phys Technol 65:30–35. doi:10.1016/j.infrared.2014.02.008
Maniar N, Bach AJE, Stewart IB, Costello JT (2015) The effect of using different regions of interest on local and mean skin temperature. J Therm Biol 49–50:33–38. doi:10.1016/j.jtherbio.2015.01.008
Priego Quesada JI, Lucas-Cuevas AG, Salvador Palmer R et al (2016) Definition of the thermographic regions of interest in cycling by using a factor analysis. Infrared Phys Technol 75:180–186. doi:10.1016/j.infrared.2016.01.014
ISO (2008) Particular requirements for the basic safety and essential performance of screening thermographs for human febrile temperature screening. TC121/SC3-IEC SC62D
ISO (2009) Medical electrical equipment-deployment, implementation and operational guidelines for identifying febrile humans using a screening thermograph. TR 13154:2009 ISO/TR 8-600
Bach AJ, Stewart IB, Minett GM, Costello JT (2015) Does the technique employed for skin temperature assessment alter outcomes? A systematic review. Physiol Meas 36:R27
Smith CJ, Havenith G (2011) Body mapping of sweating patterns in male athletes in mild exercise-induced hyperthermia. Eur J Appl Physiol 111:1391–1404. doi:10.1007/s00421-010-1744-8
Fernández-Cuevas I, Sillero-Quintana M, Garcia-Concepcion MA et al (2014) Monitoring skin thermal response to training with infrared thermography. New Stud Athl 29:57–71
Barcelos EZ, Caminhas WM, Ribeiro E et al (2014) A combined method for segmentation and registration for an advanced and progressive evaluation of thermal images. Sensors 14:21950–21967. doi:10.3390/s141121950
Chudecka M, Lubkowska A (2015) Thermal maps of young women and men. Infrared Phys Technol 69:81–87. doi:10.1016/j.infrared.2015.01.012
Merla A, Mattei PA, Di Donato L, Romani GL (2010) Thermal imaging of cutaneous temperature modifications in runners during graded exercise. Ann Biomed Eng 38:158–163. doi:10.1007/s10439-009-9809-8
Paolillo FR, Lins EC, Corazza AV et al (2013) Thermography applied during exercises with or without infrared light-emitting diode irradiation: individual and comparative analysis. Photomed Laser Surg 31:349–355. doi:10.1089/pho.2013.3505
Vardasca R, Simoes R (2013) Current issues in medical thermography. In: Topics in medical image processing and computational vision. Springer, Berlin, pp 223–237
Ring EFJ, Ammer K (2000) The technique of infrared imaging in medicine. Thermol Int 10:7–14
Ring EFJ, Ammer K (2012) Infrared thermal imaging in medicine. Physiol Meas 33:R33–46. doi:10.1088/0967-3334/33/3/R33
Ammer K (2005) Temperature readings from thermal images are less dependent on the number of pixels of the measurement area than on variation of room temperature. Thermol Int 15:131–133
Duc S, Arfaoui A, Polidori G, Bertucci W (2015) Efficiency and thermography in cycling during a graded exercise test. J Exerc Sports Orthop 2:1–8
Merla A, Iodice P, Tangherlini A, et al (2005) Monitoring skin temperature in trained and untrained subjects throughout thermal video. In: Conference proceedings: annual international conference of the IEEE, Engineering in Medicine and Biology Society vol 2, pp 1684–1686. doi:10.1109/IEMBS.2005.1616767
Chudecka M, Lubkowska A (2010) Temperature changes of selected body’s surfaces of handball players in the course of training estimated by thermovision, and the study of the impact of physiological and morphological factors on the skin temperature. J Therm Biol 35:379–385
Vollmer M, Möllmann K-P (2011) Infrared thermal imaging: fundamentals, research and applications. Wiley, Weinheim
Ammer K (2006) Influence of imaging and object conditions on temperature readings from medical infrared images. Pol J Environ Stud (Submitted)
Ivanitsky GR, Khizhnyak EP, Deev AA, Khizhnyak LN (2006) Thermal imaging in medicine: A comparative study of infrared systems operating in wavelength ranges of 3-5 and 8-12 microm as applied to diagnosis. Dokl Biochem Biophys 407:59–63
Tkáčová M, Hudák R, Foffová P, Živčák J (2010) An importance of camera subject distance and angle in musculoskeletal applications of medical thermography. Acta Electrotech Inform 10:57–60
Petrofsky JS, Lohman E, Suh HJ, et al (2006) The effect of aging on conductive heat exchange in the skin at two environmental temperatures. Med Sci Monit Int Med J Exp Clin Res 12:CR400–408
Guéritée J, Tipton MJ (2015) The relationship between radiant heat, air temperature and thermal comfort at rest and exercise. Physiol Behav 139:378–385. doi:10.1016/j.physbeh.2014.11.064
Dumke CL, Slivka DR, Cuddy JS et al (2015) The effect of environmental temperature on glucose and insulin after an oral glucose tolerance test in healthy young men. Wilderness Environ Med 26:335–342. doi:10.1016/j.wem.2015.03.002
Maughan RJ, Otani H, Watson P (2011) Influence of relative humidity on prolonged exercise capacity in a warm environment. Eur J Appl Physiol 112:2313–2321. doi:10.1007/s00421-011-2206-7
Priego Quesada JI, Carpes FP, Salvador Palmer R et al (2016) Effect of saddle height on skin temperature measured in different days of cycling. SpringerPlus 5:205–214. doi:10.1186/s40064-016-1843-z
Priego Quesada JI, Lucas-Cuevas AG, Gil-Calvo M et al (2015) Effects of graduated compression stockings on skin temperature after running. J Therm Biol 52:130–136. doi:10.1016/j.jtherbio.2015.06.005
ISO (2008) 18434-1:2008: Condition monitoring and diagnostics of machines—thermography—part 1: general procedures
Steketee J (1973) Spectral emissivity of skin and pericardium. Phys Med Biol 18:686
Togawa T (1989) Non-contact skin emissivity: measurement from reflectance using step change in ambient radiation temperature. Clin Phys Physiol Meas 10:39
Sanchez-Marin FJ, Calixto-Carrera S, Villaseñor-Mora C (2009) Novel approach to assess the emissivity of the human skin. J Biomed Opt 14:24006–24006–6. doi:10.1117/1.3086612
James CA, Richardson AJ, Watt PW, Maxwell NS (2014) Reliability and validity of skin temperature measurement by telemetry thermistors and a thermal camera during exercise in the heat. J Therm Biol 45:141–149. doi:10.1016/j.jtherbio.2014.08.010
Bernard V, Staffa E, Mornstein V, Bourek A (2013) Infrared camera assessment of skin surface temperature—effect of emissivity. Phys Med 29:583–591. doi:10.1016/j.ejmp.2012.09.003
Zontak A, Sideman S, Verbitsky O, Beyar R (1998) Dynamic thermography: analysis of hand temperature during exercise. Ann Biomed Eng 26:988–993
Ferreira JJA, Mendonça LCS, Nunes LAO et al (2008) Exercise-associated thermographic changes in young and elderly subjects. Ann Biomed Eng 36:1420–1427. doi:10.1007/s10439-008-9512-1
Bertmaring I, Babski-Reeves K, Nussbaum MA (2008) Infrared imaging of the anterior deltoid during overhead static exertions. Ergonomics 51:1606–1619. doi:10.1080/00140130802216933
Houdas Y, Ring EFJ (1982) Temperature distribution. In: Human body temperature. Springer, berlin, pp 81–103
Smolander J, Härmä M, Lindgvist A et al (1993) Circadian variation in peripheral blood flow in relation to core temperature at rest. Eur J Appl Physiol 67:192–196. doi:10.1007/BF00376666
Chamberlain JM, Terndrup TE, Alexander DT et al (1995) Determination of normal ear temperature with an infrared emission detection thermometer. Ann Emerg Med 25:15–20. doi:10.1016/S0196-0644(95)70349-7
Costa CMA, Sillero-Quintana M, Piñonosa Cano S et al (2015) Daily oscillations of skin temperature in military personnel using thermography. J R Army Med Corps. doi:10.1136/jramc-2015-000462
Marins JCB, Formenti D, Costa CMA et al (2015) Circadian and gender differences in skin temperature in militaries by thermography. Infrared Phys Technol 71:322–328. doi:10.1016/j.infrared.2015.05.008
Ammer K (2009) Does neuromuscular thermography record nothing else but an infrared sympathetic skin response? Thermol Int 19:107–108
Zaidi H, Fohanno S, Polidori G, Taiar R (2007) The influence of swimming type on the skin-temperature maps of a competitive swimmer from infrared thermography. Acta Bioeng Biomech 9:47
Novotny J, Rybarova S, Zacha D et al (2015) The influence of breaststroke swimming on the muscle activity of young men in thermographic imaging. Acta Bioeng Biomech 17:121
van Ooijen AMJ, van Marken Lichtenbelt WD, Westerterp KR (2001) Individual differences in body temperature and the relation to energy expenditure: the influence of mild cold. J Therm Biol 26:455–459. doi:10.1016/S0306-4565(01)00060-2
Gagnon D, Kenny GP (2011) Sex modulates whole-body sudomotor thermosensitivity during exercise. J Physiol 589:6205–6217. doi:10.1113/jphysiol.2011.219220
Karki A, Karppi P, Ekberg J, Selfe J (2004) A thermographic investigation of skin temperature changes in response to a thermal washout of the knee in healthy young adults. Thermol Int 14:137–141
Gagnon D, Jay O, Lemire B, Kenny GP (2008) Sex-related differences in evaporative heat loss: the importance of metabolic heat production. Eur J Appl Physiol 104:821–829. doi:10.1007/s00421-008-0837-0
Gonzalez RR (1985) Blanchard LA (1998) Thermoregulatory responses to cold transients: effects of menstrual cycle in resting women. J Appl Physiol Bethesda Md 85:543–553
Stachenfeld NS, Silva C, Keefe DL (2000) Estrogen modifies the temperature effects of progesterone. J Appl Physiol Bethesda Md 1985 88:1643–1649
Bartelink ML, Wollersheim H, Theeuwes A et al (1990) Changes in skin blood flow during the menstrual cycle: the influence of the menstrual cycle on the peripheral circulation in healthy female volunteers. Clin Sci 78:527–532
Niu HH, Lui PW, Hu JS et al (2001) Thermal symmetry of skin temperature: normative data of normal subjects in Taiwan. Zhonghua Yi Xue Za Zhi Chin Med J Free China Ed 64:459–468
Inbar O, Morris N, Epstein Y, Gass G (2004) Comparison of thermoregulatory responses to exercise in dry heat among prepubertal boys, young adults and older males. Exp Physiol 89:691–700. doi:10.1113/expphysiol.2004.027979
Ho CW, Beard JL, Farrell PA et al (1997) Age, fitness, and regional blood flow during exercise in the heat. J Appl Physiol Bethesda Md 82:1126–1135
Stapleton JM, Poirier MP, Flouris AD et al (2015) Aging impairs heat loss, but when does it matter? J Appl Physiol Bethesda Md 1985 118:299–309. doi:10.1152/japplphysiol.00722.2014
Havenith G (2001) Human surface to mass ratio and body core temperature in exercise heat stress—a concept revisited. J Therm Biol 26:387–393. doi:10.1016/S0306-4565(01)00049-3
Cramer MN, Jay O (2016) Biophysical aspects of human thermoregulation during heat stress. Auton Neurosci Basic Clin 196:3–13. doi:10.1016/j.autneu.2016.03.001
Du Bois D, Du Bois EF (1916) Clinical calorimetry: a formula to estimate the appropriate surface area if height and weight be known. Arch Intern Med 17:863–871
Chudecka M, Lubkowska A, Kempińska-Podhorodecka A (2014) Body surface temperature distribution in relation to body composition in obese women. J Therm Biol 43:1–6. doi:10.1016/j.jtherbio.2014.03.001
Savastano DM, Gorbach AM, Eden HS et al (2009) Adiposity and human regional body temperature. Am J Clin Nutr 90:1124–1131. doi:10.3945/ajcn.2009.27567
Zaproudina N, Varmavuo V, Airaksinen O, Närhi M (2008) Reproducibility of infrared thermography measurements in healthy individuals. Physiol Meas 29:515. doi:10.1088/0967-3334/29/4/007
Ichinose-Kuwahara T, Inoue Y, Iseki Y et al (2010) Sex differences in the effects of physical training on sweat gland responses during a graded exercise. Exp Physiol 95:1026–1032. doi:10.1113/expphysiol.2010.053710
Simmons GH, Wong BJ, Holowatz LA, Kenney WL (2011) Changes in the control of skin blood flow with exercise training: where do cutaneous vascular adaptations fit in? Exp Physiol 96:822–828. doi:10.1113/expphysiol.2010.056176
Ring EF, Collins AJ, Bacon PA, Cosh JA (1974) Quantitation of thermography in arthritis using multi-isothermal analysis. II. Effect of nonsteroidal anti-inflammatory therapy on the thermographic index. Ann Rheum Dis 33:353
Ring EF, Engel J, Page-Thomas DP (1984) Thermologic methods in clinical pharmacology-skin temperature measurement in drug trials. Int J Clin Pharmacol 22:20–24
Toshihiko U, Yoshiharu T, Atsuhiro M et al (1987) Application of thermography to the evaluation of the histamine skin test in man. J Pharmacol Methods 18:103–110
Wilczyński S (2015) The use of dynamic thermal analysis to distinguish between genuine and counterfeit drugs. Int J Pharm 490:16–21. doi:10.1016/j.ijpharm.2015.04.077
Westerterp-Plantenga MS, Wouters L, ten Hoor F (1990) Deceleration in cumulative food intake curves, changes in body temperature and diet-induced thermogenesis. Physiol Behav 48:831–836
Hirai A, Tanabe M, Shido O (1991) Enhancement of finger blood flow response of postprandial human subjects to the increase in body temperature during exercise. Eur J Appl Physiol 62:221–227
Kieser E, Dellimore K, Scheffer C, et al (2015) Development of diagnostic sensors for infant dehydration assessment using optical methods. In: Conference proceedings: annual international conference of the IEEE, Engineering in Medicine and Biology Society, vol 2015, pp 5537–5540. doi:10.1109/EMBC.2015.7319646
Hughes JH, Henry RE, Daly MJ (1984) Influence of ethanol and ambient temperature on skin blood flow. Ann Emerg Med 13:597–600. doi:10.1016/S0196-0644(84)80282-6
Mannara G, Salvatori GC, Pizzuti GP (1993) Ethyl alcohol induced skin temperature changes evaluated by thermography. Preliminary results. Boll Della Soc Ital Biol Sper 69:587
Wolf R, Tüzün B, Tüzün Y (1999) Alcohol ingestion and the cutaneous vasculature1. Clin Dermatol 17:395–403. doi:10.1016/S0738-081X(99)00023-1
Ammer K, Melnizky P, Rathkolb O (2003) Skin temperature after intake of sparkling wine, still wine or sparkling water. Thermol Int 13:99–102
Koot P, Deurenberg P (1995) Comparison of changes in energy expenditure and body temperatures after caffeine consumption. Ann Nutr Metab 39:135–142. doi:10.1159/000177854
Quinlan P, Lane J, Aspinall L (1997) Effects of hot tea, coffee and water ingestion on physiological responses and mood: the role of caffeine, water and beverage type. Psychopharmacology 134:164–173. doi:10.1007/s002130050438
Quinlan PT, Lane J, Moore KL et al (2000) The acute physiological and mood effects of tea and coffee: the role of caffeine level. Pharmacol Biochem Behav 66:19–28
Gershon-Cohen J, Borden AGB, Hermel MB (1969) Thermography of extremities after smoking. Br J Radiol 42:189–191
Bornmyr S, Svensson H (1991) Thermography and laser-Doppler flowmetry for monitoring changes in finger skin blood flow upon cigarette smoking. Clin Physiol 11:135–141
Ijzerman RG, Serne EH, van Weissenbruch MM et al (2003) Cigarette smoking is associated with an acute impairment of microvascular function in humans. Clin Sci 104:247–252
Barnes RB (1963) Thermography of the human body. Science 140:870–877
Togawa T, Saito H (1994) Non-contact imaging of thermal properties of the skin. Physiol Meas 15:291. doi:10.1088/0967-3334/15/3/007
Connolly DaJ, Sayers SP, McHugh MP (2003) Treatment and prevention of delayed onset muscle soreness. J Strength Cond Res 17:197–208
Ryu JH, Paik IY, Woo JH et al (2016) Impact of different running distances on muscle and lymphocyte DNA damage in amateur marathon runners. J Phys Ther Sci 28:450–455. doi:10.1589/jpts.28.450
Petersen B, Philipsen PA, Wulf HC (2014) Skin temperature during sunbathing–relevance for skin cancer. Photochem Photobiol Sci Off J Eur Photochem Assoc Eur Soc Photobiol 13:1123–1125. doi:10.1039/c4pp00066h
Whay HR, Bell MJ, Main DCJ (2004) Validation of lame limb identification through thermal imaging. In: Proceedings of 13th international symposium and 5th conference of lameness rumen. Maribor Slovenija, pp 11–15
Stokes JE, Leach KA, Main DCJ, Whay HR (2012) An investigation into the use of infrared thermography (IRT) as a rapid diagnostic tool for foot lesions in dairy cattle. Vet J 193:674–678
Vardasca R, Ring F, Plassmann P, Jones C (2012) Thermal symmetry of the upper and lower extremities in healthy subjects. Thermol Int 22:53–60
Sillero-Quintana M, Fernández-Jaén T, Fernández-Cuevas I et al (2015) Infrared thermography as a support tool for screening and early diagnosis in emergencies. J Med Imaging Health Inform 5:1223–1228
Nowakowski AZ (2006) Advances of quantitative IR-thermal imaging in medical diagnostics. Brain 10:3
Vainionpaa M, Tienhaara E-P, Raekallio M et al (2012) Thermographic imaging of the superficial temperature in racing greyhounds before and after the race. Sci World J. doi:10.1100/2012/182749
Keramidas ME, Geladas ND, Mekjavic IB, Kounalakis SN (2013) Forearm-finger skin temperature gradient as an index of cutaneous perfusion during steady-state exercise. Clin Physiol Funct Imaging 33:400–404. doi:10.1111/cpf.12043
Haddad DS, Brioschi ML, Baladi MG, Arita ES (2016) A new evaluation of heat distribution on facial skin surface by infrared thermography. Dentomaxillofacial Radiol 20150264. doi:10.1259/dmfr.20150264
Taylor NAS, Tipton MJ, Kenny GP (2014) Considerations for the measurement of core, skin and mean body temperatures. J Therm Biol 46:72–101. doi:10.1016/j.jtherbio.2014.10.006
Choi JK, Miki K, Sagawa S, Shiraki K (1997) Evaluation of mean skin temperature formulas by infrared thermography. Int J Biometeorol 41:68–75. doi:10.1007/s004840050056
Sun P-C, Jao S-HE, Cheng C-K (2005) Assessing foot temperature using infrared thermography. Foot Ankle Int 26:847–853. doi:10.1177/107110070502601010
Haddad DS, Brioschi ML, Vardasca R et al (2014) Thermographic characterization of masticatory muscle regions in volunteers with and without myogenous temporomandibular disorder: preliminary results. Dentomaxillofacial Radiol 43:20130440. doi:10.1259/dmfr.20130440
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Priego Quesada, J.I., Kunzler, M.R., Carpes, F.P. (2017). Methodological Aspects of Infrared Thermography in Human Assessment. In: Priego Quesada, J. (eds) Application of Infrared Thermography in Sports Science. Biological and Medical Physics, Biomedical Engineering. Springer, Cham. https://doi.org/10.1007/978-3-319-47410-6_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-47410-6_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-47409-0
Online ISBN: 978-3-319-47410-6
eBook Packages: Physics and AstronomyPhysics and Astronomy (R0)