Abstract
The detection of clandestine graves or concealed remains can pose a challenge to investigators. Research into the chemical signatures of decomposition, including volatile organic compounds (VOCs), can aid in the development of improved methods for the detection of remains and can further the understanding of decomposition processes. Over the last decade a number of studies have investigated decomposition VOCs from a variety of soil environments. However due to the variety of environments and methods used during these investigations a consistent odour signature remains elusive. This paper will discuss the complexity of decomposition odour and the current knowledge base of decomposition VOCs within soil environments including the impact of the entire death assemblage on the production of VOCs. The use of advanced instrumentation such as comprehensive two dimensional gas chromatography – time-of-flight mass spectrometry for the characterisation of decomposition odour is proposed. Incorporating advanced instrumentation and data handling tools into the analysis of decomposition odour will facilitate the comparison of odour profiles and generation of a consistent decomposition odour signature.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Forensic taphonomy is a multidisciplinary field within forensic science that investigates the chemical processes that occur after death and their impact on the surrounding environment. The changes to the visible appearance, chemical properties and biodiversity of the soil and surrounding environment are the result of soft tissue decomposition (France et al. 1997; Carter et al. 2007; Carter and Tibbett 2008). Investigators can utilize the changes that occur at a deposition site in order to aid in the detection and recovery of remains. During a forensic investigation or in the event of a mass disaster a variety of conditions may be encountered, therefore the method for detection of victims must be able to efficiently and accurately locate human remains (Statheropoulos et al. 2006, 2007, 2011). One potential method is the use of the chemical signature, particularly the volatile organic compounds (VOCs) produced by decomposition (Statheropoulos et al. 2011). Decomposition odour has only been studied over the last decade and the variety of studies conducted has identified a range of odour profiles from different environments. Therefore this work will explore the current state of knowledge in this area and highlight the application of advanced instrumentation to this growing field.
2 Decomposition VOCs
Over the last decade there has been an increase in research aimed at identifying the chemical components of decomposition odour. This work aims to understand the volatile compounds that elicit a response in cadaver dogs and attracts carrion insects to remains. Additionally this research can be applied to the development of portable instrumentation for the detection of remains as well as provide insight into the chemical processes of decomposition.
During decomposition, the putrefactive breakdown of macromolecules produces a variety of decomposition gases including methane, carbon dioxide and hydrogen sulfide as well as volatile organic compounds (VOCs) (Gill-King 1997; Dent et al. 2004; Statheropoulos et al. 2005; Boumba et al. 2008). VOCs can be generally defined as any compound with an appreciable vapour pressure and can have a variety of chemical properties (ASTM International 2004). The degradation of soft tissue produces hundreds of decomposition VOCs from numerous chemical classes including alcohols, aldehydes, alkanes/alkenes, aromatics, carboxylic acids, esters, ethers, halogens, ketones, nitrogen and sulfur compounds (Vass et al. 2004; Statheropoulos et al. 2005, 2007; Boumba et al. 2008; Vass et al. 2008; Dekeirsschieter et al. 2009; DeGreeff and Furton 2011; Paczkowski and Schutz 2011; Statheropoulos et al. 2011; Brasseur et al. 2012; Cablk et al. 2012; Dekeirsschieter et al. 2012; Stadler et al. 2013). Although similar chemical classes have been reported in a variety of studies the complexity of these samples has precluded the complete characterisation of decomposition odour (Statheropoulos et al. 2011; Dekeirsschieter et al. 2012).
Decomposition is a highly complex process and characterising decomposition odour is a challenge. Many studies have determined that decomposition odour is dynamic and changes its composition over time (Vass et al. 2004; Dekeirsschieter et al. 2009; Statheropoulos et al. 2011; Cablk et al. 2012; Dekeirsschieter et al. 2012; Stadler et al. 2013). All stages of decomposition produce VOCs, however outside the use of human analogues; few studies have monitored decomposition headspace throughout soft tissue decomposition. The rapid loss of soft tissue and perceived odour associated with remains during the active and advanced decay stages correlates with the number and variety of compounds identified during these stages (Anderson and VanLaerhoven 1996; Dekeirsschieter et al. 2009, 2012; Stadler et al. 2013). The overall process of decomposition in a soil environment can be characterised by a combination of chemical classes. The early stages are characterised by higher levels of polysulfides and alcohols, the transition to the later stages produces increased levels of aromatics and carboxylic acids which persist through the later stages of decay along with aldehydes and ketones (Dekeirsschieter et al. 2009, 2012; Stadler et al. 2013). Fresh and dry / skeletonized remains produce fewer VOCs, however cadaver dogs are known to locate remains shortly after death and after prolonged post-mortem intervals (Komar 1999; Rebmann et al. 2000; Lasseter et al. 2003; Oesterhelweg et al. 2008). In order to locate human remains, these canines rely on odorous compounds available for olfaction, therefore sufficient VOCs must be produced during these stages and additional research is required, including trace analysis, in order to elucidate the chemical composition of decomposition odour during these stages.
The chemical pathways and exact origins of these compounds are uncertain however it is clear that the decomposition environment and the bacteria present during decomposition will affect their production (Boumba et al. 2008; Dekeirsschieter et al. 2009; Paczkowski and Schutz 2011; Statheropoulos et al. 2011). Currently there is a large amount of variability in the compounds reported (DeGreeff and Furton 2011; Paczkowski and Schutz 2011). Studies have identified tens to hundreds of decomposition VOCs, however very few compounds have been reported in all studies, with the polysulfides being the most commonly cited (Stadler et al. 2013). The high degree of variability in the reported compounds is in part due to the range of decomposition environments studied. Decomposition VOCs have been analysed from buried remains and grave soil (Vass et al. 2004, 2008; Brasseur et al. 2012; Vass 2012), deposition on the soil surface (Dekeirsschieter et al. 2009, 2012; Stadler et al. 2013), case studies from aqueous environments (Statheropoulos et al. 2005, 2007;), enclosed spaces (Statheropoulos et al. 2011) and individual tissue samples (Hoffman et al. 2009; Cablk et al. 2012;). Each of these environments has a unique set of environmental conditions that will affect the production and availability of VOCs. The complete odour profile is complex and dynamic requiring the interaction of all aspects of the decomposition assemblage including the entomological fauna and micro-organisms.
3 Impact of the Environment on Decomposition VOCs
Abiotic factors such as temperature, humidity, availability of oxygen and pH affect the process of decomposition (Mann et al. 1990; Clark et al. 1997; Gill-King 1997; Dent et al. 2004) and therefore the production of VOCs. However, the interaction between VOCs and the environment also dictates which compounds are available within the headspace of decomposition. The majority of decomposition VOC studies have been conducted within soil environments (Vass et al. 2004, 2008; Dekeirsschieter et al. 2009; Brasseur et al. 2012; Dekeirsschieter et al. 2012; Stadler et al. 2013) however the impact of the soil on the overall profile has received little attention. In a burial situation, the compounds produced by the remains must travel through the soil column in order to be available at the surface for olfaction/detection (Vass et al. 2004; Brasseur et al. 2012). The migration of compounds is affected by the properties of the soil such as soil type, moisture content and pH but also by the properties of the compound itself (Vass et al. 2004; Statheropoulos et al. 2007; Brasseur et al. 2012; Vass 2012). The polarity of a compound indicates the likelihood the compound will migrate through the soil versus becoming bound to the soil particles, with less polar compounds such as hydrocarbons being detected near or at the surface (Brasseur et al. 2012). Decomposition soil is an integral part of the death assemblage and taphonomic studies have demonstrated the exchange of material between remains and the environment by identifying a variety of decomposition by-products within soil (Vass et al. 1992, 2002; Dent et al. 2004; Carter et al. 2007, 2008; Benninger et al. 2008; Van Belle et al. 2009; Swann et al. 2010a,b,c). Several of these compounds have also been identified within the headspace of decomposition (Dekeirsschieter et al. 2012; Stadler et al. 2013) and indicate that soil may act as a scent reservoir for decomposition odour (Brasseur et al. 2012). A challenge in the analysis of decomposition odour from soil environments is the odour signature from the soil itself. Soil VOCs have not been studied directly however, through the use of control samples and reference soils, it is clear that soil produces its own volatile signature consisting of equal complexity and diversity (Brasseur et al. 2012; Dekeirsschieter et al. 2012). This volatile signature will vary between environments and seasons and needs to be taken into consideration when interpreting decomposition VOC profiles.
In addition to the abiotic conditions of the decomposition environment, the entire ecosystem must be considered. The deposition of remains represents a large input of nutrients that will consequently alter the composition of the soil, flora and fauna of the area (Carter et al. 2007; Benninger et al. 2008; Janaway et al. 2009). Little is known about the microflora involved with decomposition, however it is known to be a complex combination of intrinsic bacteria from the respiratory and digestive systems and extrinsic bacteria from soil and the surrounding environment (Carter et al. 2007; Janaway et al. 2009). This diverse ecosystem encompasses both aerobic and anaerobic bacteria as well as fungi (Janaway et al. 2009), whose metabolic pathways degrade proteins, lipids and carbohydrates to produce numerous breakdown products, including VOCs (Boumba et al. 2008; Dekeirsschieter et al. 2009; Paczkowski and Schutz 2011). This metabolism may be the result of several bacteria or may be products of a sequential food chain with one organism working on the products of the next (Boumba et al. 2008; Paczkowski and Schutz 2011). Microbial modification of VOCs may also occur within soil as compounds migrate from buried remains (Vass et al. 2004). By altering the decomposition environment including the micro-organisms present, a different odour profile is created (Dekeirsschieter et al. 2009). The odour profile may also be affected by the entomological fauna that colonize the remains as it has been demonstrated that many decomposition VOCs are present within the headspace of isolated blowfly larvae and pupae (Frederickx et al. 2012). An investigation into the decomposition VOCs from three different environments was able to demonstrate that although the overall odour profile varies between environments there was a common core of VOCs (Dekeirsschieter et al. 2009).
The complexity of decomposition odour is further elucidated when the headspace of individual tissues are examined. Hoffman et al. (2009) examined a selection of tissue samples commonly utilized as canine training aids. None of the 14 samples analysed produced the same odour profile, and no single compound was found in common between all samples (Hoffman et al. 2009). Removal of the environmental influences and separation of tissues from the decomposition processes that occur within whole remains produces a sub-set of the profile of decomposition odour. This can be problematic when trying to conduct comparisons between individuals or to human analogues. Compounds that have been reported as being absent from the headspace of human analogue tissue such as pig carcasses and therefore unique to human remains have in fact been identified when the headspace from the entire death assemblage is analysed (Stadler et al. 2013). Development of a method for remains detection needs to consider the entire death assemblage (i.e. the remains, intrinsic and extrinsic micro flora, entomological fauna, plants and soil) in order to be an accurate representation of remains deposition encountered by investigators.
4 Signature of Decomposition Odour
Recently, decomposition odour was presented as evidence for the prosecution in the State of Florida v. Casey Marie Anthony (case no. 48-2008-CF-15606-O). Decomposition VOCs were utilized to identify a decomposition event of human origin, however the odour signature of human decomposition is not generally accepted in the scientific community and currently the analysis of decomposition VOCs has not been validated as a method for identifying the presence of human remains (Perry 2011).
Characterising the signature of human decomposition odour has been confounded by the variability in the compounds reported in the literature as well as some ambiguity surrounding the precise goal of these investigations. It has been proposed that there are two complimentary but opposing goals in the analysis of decomposition VOCs (Cablk et al. 2012). First is the goal of developing portable instrumentation for the detection of human remains. This instrumentation would detect compounds unique to humans, these compounds need not be products of decomposition but could include anthropogenic compounds that as humans we are uniquely exposed to (Cablk et al. 2012). In contrast is the characterisation of decomposition odour in order to determine the compounds required for odour recognition by cadaver dogs (Cablk et al. 2012).
Cadaver dogs are currently the best detectors for human remains. They are able to differentiate human from animal remains and locate human remains in a variety of conditions including submerged, buried and skeletal (Komar 1999; Lasseter et al. 2003; Lorenzo et al. 2003; Oesterhelweg et al. 2008). Their ability to differentiate these complex odours may be due to their ability to chromatographically separate odorants (Lawson et al. 2012), however the mechanism of odour recognition and the key compounds identified remains unclear. Research on the odour signatures of static odorants such as explosives, have indicated that canines may be responding to the most abundant compounds in an odour profile (Johnston 1999; Lorenzo et al. 2003). Recognition of complicated and dynamic target odours such as decomposition may require a variety of compounds in addition to the dominant ones as many of the compounds identified within the headspace of decomposition are not unique to decomposition and can be found from a variety of sources (Vass et al. 2008). In order to gain a better understanding of decomposition odour and evaluate the uniqueness of the entire profile, an in depth analysis of these compounds from a variety of death assemblages is required.
5 Analysis of Decomposition VOCs
Over the last decade this area of study has gained more attention and numerous methods have been explored. A variety of collection methods are available including solid-phase microextraction (SPME) (Hoffman et al. 2009; Kalinova et al. 2009; DeGreeff and Furton 2011; Cablk et al. 2012), thermal desorption (TD) (Vass et al. 2004; Statheropoulos et al. 2005, 2007; Vass et al. 2008; Stadler et al. 2013) and solvent desorption (Brasseur et al. 2012; Dekeirsschieter et al. 2009, 2012). Although all of these sampling methods utilize a solid sorbent to trap the VOCs they each have particular advantages and disadvantages.
SPME is a passive sampling device and is comprised of a small fused silica fibre with a sorbent coating which acts as an integrated system for both sample extraction and sample introduction. It is a fast and simple sample collection method however competition between analytes for adsorptive sites within the fibre can limit the accuracy and precision of the results (Augusto et al. 2001). SPME is generally utilized to sample smaller volumes of headspace and requires that an equilibrium status be reached between the sample matrix, the headspace and the fiber (Agelopoulos and Pickett 1998; Augusto et al. 2001). This equilibrium condition is not always feasible in a field setting especially when sampling whole remains.
The field portability and variety of sorbents available for sorbent tubes makes them a popular alternative. The choice of sorbent is dictated by the compounds of interest, however the stainless steel or glass tubes can be packed with multiple sorbents in order to increase the volatility range of compounds that can be sampled (McClenny 1999; Statheropoulos et al. 2011). This trait is particularly desirable for the analysis of decomposition VOCs and multi-sorbent tubes have been utilized in a number of studies (Vass et al. 2004; Statheropoulos et al. 2005, 2007; Vass et al. 2008; Statheropoulos et al. 2011; Stadler et al. 2013). Desorption of these sample tubes can be done either thermally or through the use of a solvent. Solvent desorption allows retention of a liquid sample that can be stored and analysed at a later date. However, the large solvent peak that is generated may mask many of the early eluting compounds (Agelopoulos and Pickett 1998; Brasseur et al. 2012; Dekeirsschieter et al. 2012). Conversely thermal desorption (TD) is a solvent free system that offers many advantages including increased sensitivity and applicability to complex samples with a wide range of compound volatility and polarity (Agelopoulos and Pickett 1998; Ribes et al. 2007). TD analysis generally utilizes the entire sample however many TD systems can overcome the ‘one-shot’ nature of this analysis by running the instrument in split mode and collecting the split flow prior to sample injection. TD instrumentation can also provide improved peak shape and compound resolution via a focusing step in the desorption sequence (McClenny 1999; Sanchez and Sacks 2006). TD is preferable for sampling large volumes of headspace, e.g. collecting VOCs above decomposed remains; whereas SPME is preferred for sampling small volumes of headspace e.g. collecting VOCs from blood or decomposition fluid used as cadaver dog training aids.
Despite the range of sampling methods available the analysis of VOCs has been primarily carried out using conventional gas chromatography – mass spectrometry (GC-MS). Chromatographic efficiency is influenced by numerous parameters, including temperature programming, flow rate and primarily, column selection. Published studies on the analysis of decomposition VOCs have utilized general purpose capillary columns suitable for the analysis of a variety of compound classes and volatile organics. These include non-polar 100 % dimethyl polysiloxane (Vass et al. 2004, 2008), low polarity 5 % phenyl/95 % dimethyl polysiloxane (Hoffman et al. 2009; Cablk et al. 2012) and slightly polar cyanopropylphenyl/ dimethyl polysiloxane (Statheropoulos et al. 2005, 2007; Dekeirsschieter et al. 2009; Statheropoulos et al. 2011). These studies were able to identify tens to hundreds of compounds using retention times and mass spectral data. However, chromatograms published from these studies illustrate the difficulty in achieving compound resolution (Statheropoulos et al. 2005; Dekeirsschieter et al. 2009;). The complexity of these samples in terms of the large number of components, the combination of compound classes and the significant dynamic range across the decomposition process presents an analytical challenge.
In 2011, Statheropoulos et al. explored more advanced instrumentation for the analysis of decomposition VOCs and utilized thermal desorption – gas chromatography – time-of-flight mass spectrometry (TD-GC-TOFMS) (Statheropoulos et al. 2011). In contrast to other studies that utilized mass spectrometers such as quadrupoles or ion traps (Vass et al. 2004; Statheropoulos et al. 2005, 2007; Vass et al. 2008; Dekeirsschieter et al. 2009; Hoffman et al. 2009; Cablk et al. 2012), TOFMS is a non-scanning instrument and collects full mass spectra during each acquisition (Cochran 2002; Semard et al. 2009). These nonskewed spectra ensure that consistent ion ratios are maintained as a peak elutes into the detector (Erickson et al. 1990). The nonskewed spectra allow for software algorithms to perform peak finding and spectral deconvolution of overlapping peaks (Focant et al. 2004). The advantage of utilizing hyphenated techniques for the analysis of these complex samples was noted (Statheropoulos et al. 2011) and the benefit of exploring additional technologies became evident. The spectral deconvolution of the TD-GC-TOFMS system was able to identify numerous decomposition VOCs (Statheropoulos et al. 2011) however this only provides a starting point for the full characterisation and non-target analysis of these complex samples.
The goal of non-target analysis is to confidently identify all components within the sample. To achieve this goal improvements in the chromatographic resolution in addition to the spectral resolution are required. Conventional GC systems offer high peak capacities and generally provide sufficient resolution of compounds. However when analysing highly complex samples with a variety of components such as decomposition odour, the peak capacities offered by such systems are not able to sufficiently resolve all components (Dalluge et al. 2003; Dekeirsschieter et al. 2012). Comprehensive two dimensional gas chromatography (GC × GC) is an emerging technique that provides the additional peak capacity required for the analysis of complex samples, however its wide range of potential applications are not well represented in the literature (Dalluge et al. 2003). Recent studies into decomposition odour have shown the applicability of this analytical technique to this complex sample type (Kalinova et al. 2009; Brasseur et al. 2012; Dekeirsschieter et al. 2012; Stadler et al. 2013).
6 Comprehensive Two Dimensional Gas Chromatography – Time-of-Flight Mass Spectrometry
GC × GC has been developed for the trace analysis or in-depth investigations of complex samples and matrices. In these situations, the peak capacity of conventional GC systems might not be adequate to achieve efficient separation of sample components. GC × GC-TOFMS provides multi-dimensional information about the entire sample in approximately the same amount of time as conventional one dimensional GC systems and can be combined with a variety of sample types and injection systems (Semard et al. 2009).
Multidimensional GC techniques started as heart-cut GC (GC-GC) where individual fractions of the column effluent were subjected to a secondary separation. The additional peak capacity gained from this analysis can be represented as the sum of the two columns (first dimension (1D) peak capacity + second dimension (2D) peak capacity) (Dalluge et al. 2003). This is in contrast to comprehensive two dimensional GC (GC × GC) where all the column effluent from the 1D undergoes separation on a second GC column in the 2D (Dalluge et al. 2003; Schoenmakers et al. 2003). In this case, the overall peak capacity can be represented as the product of the two columns (1D peak capacity × 2D peak capacity) (Giddings 1987; Venkatramani et al. 1996; Dalluge et al. 2003). Comprehensive GC × GC provides a distinct advantage because the entire sample undergoes thorough analysis and therefore the maximum amount of information is gained.
The first and second dimension columns of GC × GC are connected in series (Fig. 19.1) and are selected to ensure the orthogonal separation of components i.e. the compounds are separated by two different properties (Semard et al. 2009). As two compounds are not likely to have the same boiling point and the same 2nd dimension column interaction, they can be efficiently resolved. The key feature of the GC × GC instrumentation is the modulator. The modulator is situated between the two columns and serves several functions; to continuously trap and re-focus fractions of effluent from the first dimension column, and to re-inject these fractions onto the second dimension column (Dalluge et al. 2003). As a peak elutes from the 1D column the modulator collects small fractions of it at a time, producing multiple slices across the peak (Fig. 19.1). The peaks resulting from the two dimensional separation and modulation are quite narrow (100–600 milliseconds) and require fast detectors in order to reconstruct the two dimensional chromatograms. Time of flight mass spectrometry (TOFMS) fulfils this requirement with its acquisition rates of >50 Hz (Dalluge et al. 2003). The combination of the chromatographic resolution provided by GC × GC along with the analytical or mass spectral resolution provides an extremely powerful tool for the analysis of complex samples as each sample component will be described by its first dimension retention time (1tR), second dimension retention time (2tR) and mass spectra.
Schematic of GC × GC system. Sample inlet can be liquid injector or thermal desorption apparatus, detector is typically a TOFMS. The black trace represents the coelution of the red and blue peak on a conventional GC system. 1tR: first dimension retention time, 2tR: second dimension retention time, pM: modulation period (Figure adapted from Semard et al. 2009)
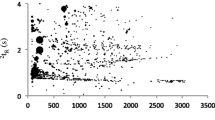
The initial data processing required to generate an output for GC × GC-TOFMS analysis requires extensive computing power and integrated software tools (Dalluge et al. 2002, 2003). The raw output of the TOFMS is a linear trace of the modulated peaks or slices. Each slice has four pieces of information associated with it; the 1tR, 2tR, a mass spectrum and signal intensity. A reconstructed peak matrix is then plotted by their first and second retention times. In a 3D surface plot the intensity of a peak is shown on the z-axis. A contour plot is a 2D plot of the data and is essentially a birds-eye view of the 3D surface plot (Fig. 19.2). In both, the colour scale indicates the intensity of a peak. Once processed, the data may also be presented as an apex plot. In this case there is no scaling to indicate the intensity of the peak and the two retention times of the peak apex are plotted as one point. The contour plot can be overlaid with the apex plot for further clarity (Dalluge et al. 2002).
Sample output of comprehensive two dimensional chromatography – time-of-flight mass spectrometry of decomposition headspace from human analogue in a soil environment. Linear trace at rear of image is a one dimensional projection of the chromatogram similar to what would be produced from the analysis with conventional GC-MS systems. The grey oval outlines the baseline of the GC × GC chromatogram. 1tR: first dimension retention time, 2tR: second dimension retention time
Few taphonomic studies have utilized this technique however the application of this advanced instrumentation to the analysis of decomposition odour has shown several benefits over conventional GC-MS. Utilization of this method for the analysis of decomposition headspace demonstrated that the increased peak capacity is capable of producing a more detailed profile by separating and identifying up to ten times the components (Dekeirsschieter et al. 2012). The enhanced peak capacity and separation of sample components is also enhanced by the utilization of the large chromatographic space available in GC × GC (Stadler et al. 2013). Figure 19.2 demonstrates the separation of all sample components including large overloading compounds from smaller peaks. Component peaks that align in the 2nd dimension of Fig. 19.2 would have co-eluted in conventional chromatography as shown by the linear trace along the rear of the figure and thus their identification may have been inhibited. Optimization of the two dimensional method to ensure that the majority of the chromatographic space is utilized allows the sample components to be separated from the instrumental background, shown by the oval outline in Fig. 19.2, thus ensuring better compound detection and identification (Dekeirsschieter et al. 2012; Stadler et al. 2013). This is particularly advantageous when characterising samples such as fresh or skeletonized remains, that have lower or trace amounts of compounds which would be otherwise masked within the baseline (Kalinova et al. 2009; Dekeirsschieter et al. 2012).
The non-target analysis of decomposition odour by GC × GC-TOFMS allows for the complete characterisation and comparison of these samples. This approach produces large datasets that can be difficult to manage, however there are several data handling tools that make reduction of the data set and cross-sample comparisons fast and efficient (Brasseur et al. 2012; Dekeirsschieter et al. 2012; Stadler et al. 2013). Scripting is a data handling tool that utilizes characteristic mass spectral features to extract all peaks from a particular chemical class (Brasseur et al. 2012). The unique aspects of the fragmentation pattern are written in a computer language and applied to the entire data set, peaks whose mass spectra match that of the script are highlighted allowing for a large number of compounds to be extracted simultaneously (Brasseur et al. 2012). Scripts for multiple chemical classes can be applied to a data set facilitating the extraction of the decomposition odour signature (Brasseur et al. 2012). In addition to scripting, statistical analysis tools are available to facilitate the comparison of samples. By comparing the peak tables generated from initial data processing the unknown chemical variations between sample groups i.e. experimental and control, can be determined. Specifically this tool allows for the alignment of multiple chromatograms and the statistical comparison of the peak tables from groups or classes of samples (Stadler et al. 2013). This approach to non-target analysis and the identification of all components within a sample facilitates the extraction of decomposition products from environmental VOCs producing more accurate and consistent profiles of decomposition odour (Brasseur et al. 2012; Dekeirsschieter et al. 2012; Stadler et al. 2013). This can be of additional benefit when working with difficult matrices, such as soil (Brasseur et al. 2012). Due to the complex nature of both the soil matrix and decomposition signatures, sophisticated data handling tools can be used to quickly screen raw data sets and extract particular compounds or classes of interest (Brasseur et al. 2012). With ongoing research these tools can be used to generate profiles from various environments and decomposition scenarios thereby facilitating the production of a more holistic profile and improving our understanding of decomposition assemblages and their volatile signatures.
7 Application of GCxGC-TOFMS to Human Analogue Decomposition in a Soil Environment
A field study was conducted at the Geoforensic Research Facility in Oshawa Ontario, Canada during the summer months (July–August 2011) to investigate the application of GCxGC-TOFMS for the chemical analysis of decomposition products. Pig carcasses (Sus scorfa domesticus) were utilized as human analogues and were placed on a soil surface on top of light grassy vegetation. Wire scavenging cages were placed over the carcasses between sampling periods in order to prevent avian and mammalian scavenging. Within the Geoforensic Research Facility, control sites of similar vegetation were delineated and the headspace was sampled following the same schedule and procedure as the experimental sites which contained the carcasses. At the time of sampling, a stainless steel hood (100 cm x 70 cm x 40 cm) was placed over the remains for a period of 30 min in order to develop a headspace. A multi-sorbent (Tenax GR & Carbopack B) thermal desorption tube was connected to a sampling port and 1 L of headspace was collected.
The decomposition headspace was analysed using TD-GCxGC-TOFMS. Thermal desorption was carried out by a Markes International Ltd. (Llantrisant U.K.) Unity 2 series thermal desorber, which combines sample desorption, focusing and injection. The two dimensional chromatography and spectroscopy was completed with an Agilent 7890 GC (Palo Alto, CA, USA) equipped with a secondary oven and modulator which was coupled with a Pegasus 4D GCxGC TOFMS from LECO Corporation (St. Joseph, MI, USA). An Rxi5Sil-MS column (30 m × 0.25 mm × 0.25 μm) was utilised in the first dimension and a BPX-50 column (1.2 m × 0.1 mm × 0.1 μm) was placed in the second dimension. A detailed description of the methods and analytical parameters utilised have been described previously (Stadler et al. 2013).
The non-target analysis of the decomposition headspace revealed thousands of peaks, including instrumental background, environmental VOCs and compounds of interest. The comparison of the control and experimental samples using sophisticated software and statistical techniques identified 300 VOCs within the headspace of decomposition throughout the decay process. The compounds were from a variety of chemical classes and the odour profile formed was dynamic across decomposition. The dominant compounds of the various chemical classes are shown in Table 19.1. The key chemical families, alcohols, sulfides, aromatics, and carboxylic acids characterized the continuous process of decomposition and the dominant compounds (e.g., 1-butanol, 2-and 3-methyl butanoic acid, DMDS, DMTS, phenol, and indole) were identified as potential target odorants of decomposition. In addition to these major chemical classes the aldehydes, ketones and nitrogen compounds further characterized the complete profile.
A major challenge in the analysis of complex samples such as decomposition headspace is the combination of trace compounds with large overloading peaks. Using conventional GC-MS systems, this large dynamic range can cause compounds to be lost due to co-elution or misidentification of compounds due to concentration effects within the mass spectrometer. Decomposition odour has the highest complexity during the active decay (Dekeirsschieter et al. 2012; Stadler et al. 2013), including the presence of large overloading compounds which increase the potential for compound co-elution (Fig. 19.2). The linear trace along the rear of Fig. 19.2 represents a chromatogram obtained using a one dimensional GC. It is evident that the larger peaks would mask many of the smaller components. However the additional peak capacity provided by the two dimensional chromatography allows for the separation of sample components across the chromatographic plane thereby decreasing the amount of co-elution within the sample. The improved compound resolution along with the TOFMS utilized in this work compensated for the sample complexity and dynamic range. Compound identifications were made by a mass spectral library search. The TOFMS generated higher quality non-skewed spectra which facilitated the identification of decomposition VOCs by correcting for concentration effects as overloading peaks elute into the detector.
This profile generated from the non-target analysis of decomposition headspace was able to characterize the process of soft tissue decomposition within a surface soil environment (Stadler et al. 2013). The ability to identify numerous compounds of interest in order to characterize these complicated samples illustrates the main benefit of multidimensional chromatography. The production of large data sets facilitates the use of a number of data mining strategies to generate meaningful results (Brasseur et al. 2012). Currently there exists a large amount of variation in the VOC profiles reported within the literature; however the application of GCxGC-TOFMS to the comprehensive analysis of decomposition odour can generate more consistent profiles of decomposition odour. The odour profile identified in this work was consistent with another study on carrion decomposition within a surface soil environment conducted in a separate geographical location (Stadler et al. 2013). The use of GCxGC-TOFMS for the non-target analysis of decomposition VOCs allows for the comparison of VOC profiles from different experimental treatments and is able to generate a comprehensive and accurate decomposition odour profile.
8 Summary
VOCs are an integral part of decomposition; they are responsible for the attraction of forensically relevant insects and are utilized by cadaver dogs to locate human remains. The profile generated by the current literature indicates the major chemical families; polysulfides, alcohols, aromatics, carboxylic acids and aldehydes. The polysulfides are dominant compounds within decomposition odour however, the consistent identification of other profile components has been elusive. If the identification of compounds can be improved a decomposition odour fingerprint can be developed. This odour profile of decomposition can be further utilized to develop improved methods for remains detection as well as further our understanding of the chemistry of decomposition. In order to determine the components of human decomposition odour, extensive research into the chemical composition of decomposition headspace needs to be conducted utilizing instrumentation capable of the non-target analysis of complex samples such as thermal desorption coupled to comprehensive two dimensional gas chromatography – time-of-flight mass spectrometry. Non-target analysis is capable of identifying all chemical components within samples and can be used for the comparison of sample profiles. This level of analysis may also aid in the production of more consistent volatile profiles.
Using this instrumentation, investigations into the decomposition odour from a variety of geographical locations and decomposition environments will facilitate the development of a comprehensive odour signature. Studies isolating the variables that affect the production of decomposition VOCs including micro-organisms, and identifying the resultant changes in the VOCs produced would provide an expanded odour profile that would provide valuable information about this complex target odour and advance our knowledge of decomposition chemistry. A major limitation to this area of research is the difficulties associated with accessing human remains however this can be alleviated through the use of human analogues, such as pig carcasses. In addition to the analysis of soft tissue decomposition, research into skeletonised remains as well as potential scent reservoirs such as decomposition soil are required.
References
Agelopoulos NG, Pickett JA (1998) Headspace analysis in chemical ecology: effects of different sampling methods on ratios of volatile compounds present in headspace samples. J Chem Ecol 24:1161–1172
Anderson GS, VanLaerhoven SL (1996) Initial studies on insect succession on carrion in southwestern British Colombia. J Forensic Sci 41:617–625
ASTM International (2004) D 6345 standard guide for selection of methods for active, integrative sampling of volatile organic compounds in air. ASTM Standards, West Conshohocken
Augusto F, Koziel J, Pawliszyn J (2001) Design and validation of portable SPME devices for rapid field air sampling and diffusion-based calibration. Anal Chem 73:481–486
Benninger LA, Carter DO, Forbes SL (2008) The biochemical alteration of soil beneath a decomposing carcass. Forensic Sci Int 180:70–75
Boumba VA, Ziavrou KS, Vougiouklakis T (2008) Biochemical pathways generating post-mortem volatile compounds co-detected during forensic ethanol analyses. Forensic Sci Int 174:133–151
Brasseur C, Dekeirsschieter J, Schotsmans EMJ, de Koning S, Wilson AS, Haubruge E, Focant JF (2012) Comprehensive two dimensional gas chromatography-time-of-flight mass spectometry for the forensic study of cadaveric volatile organic compounds released in soil by buried decaying pig carcasses. J Chromatogr A 1255:163–170
Cablk ME, Szelagowski EE, Sagebiel JC (2012) Characterization of the volatile organic compounds present in the headspace of decomposing animal remains, and compared with human remains. Forensic Sci Int 220:118–125
Carter DO, Tibbett M (2008) Cadaver decomposition and soil: processes: chemical and biological effects of buried human remains. In: Tibbett M, Carter DO (eds) Soil analysis in forensic Taphonomy. CRC Press, New York, pp 29–51
Carter DO, Yellowlees D, Tibbett M (2007) Cadaver decomposition in terrestrial ecosystems. Naturwissenschaften 94:12–24
Carter DO, Yellowlees D, Tibbett M (2008) Using ninhydrin to detect gravesoil. J Forensic Sci 53:397–400
Clark MA, Worrell MB, Plessm JE (1997) Postmortem changes in soft tissues. In: Haglund WD, Sorg MH (eds) Forensic taphonomy: the postmortem fate of human remains. CRC Press, New York, pp 151–164
Cochran JW (2002) Fast gas chromatography-time-of-flight mass spectrometry of polychlorinated biphenyls and other environmental contaminants. J Chromatogr Sci 40:254–268
Dalluge J, Vreuls RJ, Beens J, Brinkman UA (2002) Optimization and characterization of comprehensive two-dimentional gas chromatography with time-of-flight mass spectometric detection (GCxGC-TOF MS). J Sep Sci 25:201–214
Dalluge J, Beens J, Brinkman UA (2003) Comprehensive two-dimensional gas chromatography: a powerful and versatile analytical tool. J Chromatogr A 1000:69–108
DeGreeff LE, Furton KG (2011) Collection and identification of human remains volatiles by non-contact, dynamic airflow sampling and SPME-GC/MS using various sorbent materials. Anal Bioanal Chem 401:1295–1307
Dekeirsschieter J, Verheggen FJ, Gohy M, Hubrecht F, Bourguignon L, Lognay G, Haubruge E (2009) Cadaveric volatile organic compounds released by decaying pig carcasses (Sus domesticus L.) in different biotopes. Forensic Sci Int 189:46–53
Dekeirsschieter J, Stefanuto PH, Brasseur C, Haubruge E, Focant JF (2012) Enhanced characterization of the smell of death by comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry (GCxGC-TOFMS). PLoS ONE 7(6), e39005
Dent BB, Forbes SL, Stuart BH (2004) Review of human decomposition processes in soil. Eviron Geo 45:576–585
Erickson ED, Enke CG, Holland JF, Watson JT (1990) Application of time array detection to capillary column gas chromatography/conventional time-of-flight mass spectrometry. Anal Chem 62:1079–1084
Focant JF, Sjodin A, Turner WE, Patterson DG Jr (2004) Measurement of selected polybrominated diphenyl ethers, polybrominated and polychlorinated biphenyls, and organochlorine pesticides in human serum and milk using comprehensive two-dimensional gas chromatography isotope dilution time-of-flight mass spectrometry. Anal Chem 76:6313–6320
France DL, Griffin TJ, Swanburg JG, Lindemann JW, Davenport GC, Trammell V, Travis CT, Kondratieff B, Nelson A, Castellano K, Hopkins D, Adair T (1997) NecroSearch revisited: further multidisciplinary approaches to the detection of clandestine graves. In: Haglund WD, Sorg MH (eds) Forensic taphonomy: the postmortem fate of human remains. CRC Press, New York, pp 497–509
Frederickx C, Dekeirsschieter J, Brostaux Y, Wathelet JP, Verheggen FJ, Haubruge E (2012) Volatile organic compounds released by blowfly larvae and pupae: new perspectives in forensic entomology. Forensic Sci Int 219:215–220
Giddings JC (1987) Concepts and comparisons in multidimensional separation. J High Resolut Chromatogr 10:319–323
Gill-King H (1997) Chemical and ultrastructural aspects of decomposition. In: Haglund WD, Sorg MH (eds) Forensic taphonomy: the postmortem fate of human remains. CRC Press, New York, pp 93–108
Hoffman EM, Curran AM, Dulgerian N, Stockham RA, Eckenrode BA (2009) Characterization of the volatile organic compounds present in the headspace of decomposing human remains. Forensic Sci Int 186:6–13
Janaway RC, Percival SL, Wilson AS (2009) Decomposition of human remains. In: Percival SL (ed) Microbiology and aging. Springer Science + Business Media, New York, pp 313–334
Johnston JM (1999) Canine detection capabilities: operational implications of recent R & D Findings. Institute for Biological Detection Systems, Auburn
Kalinova B, Podskalska H, Ruzicka J, Hoskovec M (2009) Irresistible bouquet of death—how are burying beetles (Coleoptera: Silphidae: Nicrophorus) attracted by carcasses. Naturwissenschaften 96:889–899
Komar D (1999) The use of cadaver dogs in locating scattered, scavenged human remains: preliminary field test results. J Forensic Sci 44:405–408
Lasseter AE, Jacobi KP, Farley R, Hensel L (2003) Cadaver dog and handler team capabilities in the recovery of buried human remains in the southeastern United States. J Forensic Sci 48:617–621
Lawson MJ, Craven BA, Paterson EG, Settles GS (2012) A computational study of odorant transport and deposition in the canine nasal cavity: implications for olfaction. Chem Senses 37:553–566
Lorenzo N, Wan T, Harper RJ, Hsu YL, Chow M, Rose S, Furton KG (2003) Laboratory and field experiments used to identify Canis lupus var. familiaris active odor signature chemicals from drugs, explosives, and humans. Anal Bioanal Chem 376:1212–1224
Mann RW, Bass WM, Meadows L (1990) Time since death and decomposition of the human body: variables and observations in case and experimental field studies. J Forensic Sci 35:103–111
McClenny EAWWA (1999) Compendium method TO-17: determination of volatile organic compounds in ambient air using active sampling onto sorbent tubes. U.S. Environmental Protection Agency, Cincinnati
Oesterhelweg L, Krober S, Rottmann K, Willhoft J, Braun C, Thies N, Puschel K, Silkenath J, Gehl A (2008) Cadaver dogs—a study on detection of contaminated carpet squares. Forensic Sci Int 174:35–39
Paczkowski S, Schutz S (2011) Post-mortem volatiles of vertebrate tissue. Appl Microbiol Biotechnol 91:917–935
Perry B (2011) State of Florida v. Casey Marie Anthony – Order denying motion to exclude unreliable evidence. 9th Judicial Circuit
Rebmann A, David E, Sorg MH (2000) Cadaver dog handbook: forensic training and tactics for the recovery of human remains. CRC Press, New York
Ribes A, Carrera G, Gallego E, Roca X, Berenguer MA, Guardino X (2007) Development and validation of a method for air-quality and nuisance odors monitoring of volatile organic compounds using multi-sorbent adsorption and gas chromatography/mass spectrometry thermal desorption system. J Chromatogr A 1140:44–55
Sanchez JM, Sacks RD (2006) Development of a multibed sorption trap, comprehensive two-dimensional gas chromatography, and time-of-flight mass spectrometry system for the analysis of volatile organic compounds in human breath. Anal Chem 78:3046–3054
Semard G, Mohamed A, Focant J-F (2009) Basic Instrumentation for GCxGC. In: Comprehensive Analytical Chemistry, vol 55. Elsevier, pp 15–48
Schoenmakers P, Marriott P, Beens J (2003) Nomenclature and conventions in comprehensive multidimensional chromatography. Coupling Matters, Ellesmere Port
Stadler S, Stefanuto PH, Brokl M, Forbes SL, Focant JF (2013) Characterization of volatile organic compounds from human analogue decompsoition using thermal desorption coupled to comprehensive two-dimensional gas chromatography - time-of-flight mass spectrometry. Anal Chem 85:998–1005
Statheropoulos M, Spiliopoulou C, Agapiou A (2005) A study of volatile organic compounds evolved from the decaying human body. Forensic Sci Int 153:147–155
Statheropoulos M, Mikedi K, Agapiou A, Georgiadou A, Karma S (2006) Discriminant analysis of volatile organic compounds data related to a new location method of entrapped people in collapsed buildings of an earthquake. Anal Chim Acta 566:207–216
Statheropoulos M, Agapiou A, Spiliopoulou C, Pallis GC, Sianos E (2007) Environmental aspects of VOCs evolved in the early stages of human decomposition. Sci Total Environ 385:221–227
Statheropoulos M, Agapiou A, Zorba E, Mikedi K, Karma S, Pallis GC, Eliopoulos C, Spiliopoulou C (2011) Combined chemical and optical methods for monitoring the early decay stages of surrogate human models. Forensic Sci Int 210:154–163
Swann L, Chidlow GE, Forbes S, Lewis SW (2010a) Preliminary studies into the characterization of chemical markers of decomposition for geoforensics. J Forensic Sci 55:308–314
Swann L, Forbes S, Lewis SW (2010b) Observations of the temporal variation in chemical content of decomposition fluid: a preliminary study using pigs as a model system. Aust J Forensic Sci 42:199–210
Swann LM, Forbes SL, Lewis SW (2010c) Analytical separations of mammalian decomposition products for forensic science: a review. Anal Chim Acta 682:9–22
Van Belle LE, Carter DO, Forbes SL (2009) Measurement of ninhydrin reactive nitrogen influx into gravesoil during aboveground and belowground carcass (Sus domesticus) decomposition. Forensic Sci Int 193:37–41
Vass AA (2012) Odor mortis. Forensic Sci Int 222:234–241
Vass AA, Bass WM, Wolt JD, Foss JE, Ammons JT (1992) Time since death determinations of human cadavers using soil solution. J Forensic Sci 37:1236–1253
Vass AA, Barshick SA, Sega G, Caton J, Skeen JT, Love JC, Synstelien JA (2002) Decomposition chemistry of human remains: a new methodology for determining the postmortem interval. J Forensic Sci 47:542–553
Vass AA, Smith RR, Thompson CV, Burnett MN, Wolf DA, Synstelien JA, Dulgerian N, Eckenrode BA (2004) Decompositional odor analysis database. J Forensic Sci 49:760–769
Vass AA, Smith RR, Thompson CV, Burnett MN, Dulgerian N, Eckenrode BA (2008) Odor analysis of decomposing buried human remains. J Forensic Sci 53:384–391
Venkatramani CJ, Xu J, Phillips JB (1996) Separation orthogonality in temperature-programmed comprehensive two-dimensional gas chromatography. Anal Chem 68:1486–1492
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this paper
Cite this paper
Stadler, S., Focant, JF., Forbes, S.L. (2016). Forensic Analysis of Volatile Organic Compounds from Decomposed Remains in a Soil Environment. In: Kars, H., van den Eijkel, L. (eds) Soil in Criminal and Environmental Forensics. Soil Forensics. Springer, Cham. https://doi.org/10.1007/978-3-319-33115-7_19
Download citation
DOI: https://doi.org/10.1007/978-3-319-33115-7_19
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-33113-3
Online ISBN: 978-3-319-33115-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)