Abstract
In this work, a simple and sensitive approach for VEGF detection using antibody-aptamer assay and gold screen-printed electrodes as transducers is presented. The assay was performed in a sandwich format. Anti-VEGF antibody was first chemically immobilized on the gold working electrode surface of screen-printed cell. After the incubation with the antigen, the sandwich assay was realized by incubation step with biotinylated anti-VEGF aptamer. The sensor was then incubated with streptavidin-alkaline phosphatase and with 1-naphthyl phosphate. Differential pulse voltammetry (DPV) measurements were performed to detect VEGF biomarker.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
- Vascular Endothelial Growth Factor
- Vascular Endothelial Growth Factor Level
- Differential Pulse Voltammetry
- Vascular Endothelial Growth Factor Protein
- Vascular Endothelial Growth Factor Concentration
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
The possibility of performing reliable cancer diagnosis even before any symptom of disease appears is crucial for increasing therapeutic treatment success and patient survival rates [1]. During the last decade, improved understanding of carcinogenesis and tumor progression has revealed a large number of potential tumor markers. Vascular endothelial growth factor (VEGF) is referred to a family of homodimeric glycoproteins which are involved in the development of the blood vascular system (vasculogenesis), of the lymphatic system (lymphangiogenesis), in the formation of new blood vessels from pre-existing one (angiogenesis), and in the vascularization of tumor. VEGF level has been extensively used as biomarker associated with diagnosis and prognosis of different cancer disease [2].
Different detection methods such as enzyme-linked immunosorbent assays (ELISA) and immunohistochemistry have been used for VEGF quantification. Nevertheless these methods do not satisfy the rapidity requirement and the necessity to use simple instrumentation for point of care diagnostics [3]. In this context, rapid non-immunochemical sensors based on electrochemical methods that can use aptamers are emerging. In this work, simple and sensitive approach for VEGF detection using antibody-aptamer assay and screen-printed cells as transducers is presented.
2 Materials and Methods
11-Mercaptoundecanoic acid (MUDA), 6-mercapto-1-hexanol (MCH), diethanolamine (DEA), 1-naphthyil phosphate, streptavidin-alkaline phosphatase, ethanolamine (EA), human vascular endothelial growth factor (VEGF), and anti-VEGF antibody were obtained from Sigma-Aldrich (Milan, Italy).
Aptamer used in this work [5] was purchased from MWG Biotech, Germany:
5′-GGGCCCGTCCGTATGGTGGGTGTGCTGGCCTTTTTTTTTTTTTTT(45) 3′-Biot.
All chemicals were used as received without any further purification. Milli-Q water was used throughout this work.
Electrochemical experiments were performed in a digital potentiostat/galvanostat Autolab PGSTAT 30(2)/FRA2 controlled with the General Purpose Electrochemical System (GPES) and Frequency Response Analyzer (FRA2) 4.9 software (Eco Chemie, Utrecht, The Netherlands). The immunosensor was assembled using screen-printed cells, comprising of gold working electrode (2.5 mm in diameter), counter graphite electrode, and a pseudo-silver [4].
Differential pulse voltammetry (DPV) was performed using the following parameters: potential range: −0.2 V to +0.5 V; pulse amplitude 0.070 V; and scan rate 0.033 V s−1. The measurements were carried out at room temperature.
3 Experimental Procedure
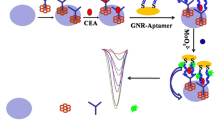
The scheme of antibody-aptamer assay developed for the detection of VEGF tumor marker was shown in Fig. 1.
Firstly, 11-mercaptoundecanoic acid (MUDA) was immobilized on the gold working electrode surface of screen-printed cell followed by a blocking step with 6-mercapto-1-exanhol. After the activation of –COOH groups by EDAC/NHS solution, incubation step with anti-VEGF antibody was performed. Then, the unreacted –COOH groups were blocked with ethanolamine (EA), and the sensor was incubated with VEGF protein buffered solution at different concentration. The sandwich assay was later completed by interaction with anti-VEGF biotinylated aptamer. Streptavidin-alkaline phosphatase was incubated at the end with the sensor.
Finally, 50 μL of 1-naphthyl phosphate was placed on the screen-printed cell for 6 min and then DPV measurements were carried out.
3.1 Results and Discussion
Calibration curve for the determination of VEGF protein in buffered solutions by antibody-aptamer assay was reported in Fig. 2.
A linear correlation between the current and the VEGF concentration in the range of 0–0.4 ng mL−1 was obtained. The assay was repeated in order to evaluate the reproducibility; at this purpose, three repetitions of each standard solution were carried out. The average coefficient of variation was 10 %, calculated as mean of all the concentrations considered. The limit of detection (LOD), calculated by the ratio between three times the deviation standard of blank and the slope of calibration curve (3Sblank/Sslope), was 0.1 ng mL−1.
4 Conclusion
In this work, simple and sensitive approach for VEGF detection using antibody-aptamer assay and gold screen-printed electrodes as transducers is presented. The assay was performed in a sandwich format. The proposed assay shows a linear calibration curve in the range of 0–0.4 ng mL−1 with a LOD of 0.1 ng mL−1.
References
J. J. Ott, A. Ullrich, A. B. Miller. The importance of early symptom recognition in the context of early detection and cancer survival. European Journal of Cancer 45, 2743-2748 (2009).
C. Sessa, A. Guibal, G. Del Conte, C. Ruegg. Biomarkers of angiogenesis for the development of antiangiogenic therapies in oncology: tools or decorations?. Nature clinical practice oncology 5, 378-91 (2008).
H. Chen, C. Jiang, C. Yu, S. Zhang, B. Liu, J. Kong. Protein chips and nanomaterials for application in tumor marker immunoassays. Biosensors and Bioelectronics 24 3399-3411 (2009).
F. Bettazzi, F. Lucarelli, I. Palchetti, F. Berti, G. Marrazza, M. Mascini. Disposable electrochemical DNA-array for PCR amplified detection of hazelnut allergens in foodstuffs. Anal. Chim. Acta 614, 93-102 (2008).
H. Hasegawa, K. Sode, K. Ikebukuro. Selection of DNA aptamers against VEGF(165) using a protein competitor and the aptamer blotting method. Biotechnol. Lett. 30, 829-834 (2008).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this paper
Cite this paper
Ravalli, A., Marrazza, G., Rivas, L., De La Escosura-Muniz, A., Merkoci, A. (2014). Electrochemical Antibody-Aptamer Assay for VEGF Cancer Biomarker Detection. In: Di Natale, C., Ferrari, V., Ponzoni, A., Sberveglieri, G., Ferrari, M. (eds) Sensors and Microsystems. Lecture Notes in Electrical Engineering, vol 268. Springer, Cham. https://doi.org/10.1007/978-3-319-00684-0_33
Download citation
DOI: https://doi.org/10.1007/978-3-319-00684-0_33
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-00683-3
Online ISBN: 978-3-319-00684-0
eBook Packages: EngineeringEngineering (R0)