Abstract
Dispersive liquid–liquid microextraction (DLLME) was initially described as an effective sample preparation technology in 2006. However, researchers are still interested in making it more efficient, and ecologically friendly. The dispersion of extraction solvent in aqueous samples is the critical stage in DLLME, which is commonly accomplished using dispersive solvents. Because hazardous dispersive solvents offer a significant environmental danger, attempts have been undertaken to produce greener dispersion procedures while maintaining high extraction efficiency. When it comes to ordinary DLLME, the number of fascinating approaches for changing disperser solvents has expanded. As a result, the goal of this chapter is to provide a overview of current developments in DLLME dispersion modes. Different strategies are covered, including the employment of environmentally-benign dispersers as well as other dispersion methodologies. The most noteworthy approaches that have been implemented to date are highlighted. The problems and prospects for the future of these techniques are discussed. The chapter offer new study avenues, reinforce existing hypotheses, and discover trends among existing DLLME research papers.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Dispersive liquid–liquid microextraction
- Sample preparation
- Green analytical chemistry
- Air-assisted
- Supramolecular
1 Introduction
Liquid–liquid extraction (LLE) is the most commonly used technique of sample preparation. In LLE, a few milliliters of a water immiscible organic solvent is mixed and shaken with the aqueous to allow for analyte partitioning. After that, the extract is left to evaporate under a stream of nitrogen to evade sample oxidation. Then, the residue is dissolved is the least possible amount of a suitable organic solvent to keep the sample highly concentrated. These procedure are not only time consuming, but also health hazardous due to the large volume of organic solvents that either evaporate or disposed after extraction [1]. In addition, The automation of LLE steps is a real challenge [2]. For these reasons, LLE is considered ecologically unfriendly and laborious.
Miniaturization of extraction technologies has grown significantly in recent decades [3] to solve the problems of classical LLE while benefiting from its cost-effectiveness and high efficiency. A tiny amount of a water-immiscible organic solvent is employed in liquid–liquid microextraction (LLME) to extract target analytes from aqueous samples. This smaller variant of LLE offers a more environmentally friendly approach to improving extraction efficiency with fewer chemicals and quicker analytical times. Moreover, LLME was readily automated, which aided in the analytical process and safeguarded workers [4].
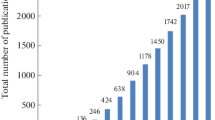
DLLME technique was developed in 2006 by Rezaee and colleagues as a modification of the LLME technique [5, 6]. The purpose was to improve the recovery rate of LLME. In DLLME, an immiscible organic solvent is combined with an organic disperser to create an emulsion. Manual shaking is then used to disperse the organic extractant into tiny droplets, resulting in a homogeneous solution. This dispersion process extends the contact surface area between the extractant and the sample, thereby increasing extraction kinetics. Following this, the sample is centrifuged to separate the extractant and break up the emulsion. In 2007, Zanjani et al. proposed a new variation of DLLME that uses low-density solvents of long-chain alcohol. These solvents solidify at low temperatures, allowing for easy phase separation. This method, called DLLME with solidified organic droplet (DLLME-SFOD), has been widely used in various applications [7]. A year later, ultrasound was utilized instead of manual or mechanical shaking to induce dispersion, eliminating the need for a dispersing solvent [8]. In 2010, Chen et al. introduced the solvent-terminated DLLME (ST-DLLME) technique, which avoids the centrifugation step by adding an auxiliary solvent to break the dispersion and induce phase separation [9]. This mode aided in the automation of the method. In 2011, Jafarvand and Shemirani used coacervates and reverse micelles to form supramolecular self-assemblies, resulting in higher extraction efficiency and selectivity for specific analytes [10, 11]. The following year, Farajzadeh and Mogaddam introduced air-assisted DLLME (AA-DLLME) using repeated aspiration/injection cycles to induce dispersion [12]. In 2014, magnetic ionic liquids were employed in DLLME to induce phase separation using a strong magnet, eliminating the need for centrifugation [13]. In 2020, water-immiscible natural deep eutectic solvents (NADES) were utilized in DLLME to extract various analytes, including nine phthalic acid esters [14]. Figure 1 illustrates the milestones of DLLME development over the last years. DLLME is one of the most successful miniaturized sample preparation techniques, due to the high EF, high sensitivity, acceptable precision, accuracy and selectivity according to the acceptance criteria and guidelines of the Food and Drug Administration (FDA). In addition, DLLME is a fast mode of sample preparation in comparison with conventional techniques. The speed of DLLME could be even accelerated by using semiautomated-DLLME or fully automated DLLME [15].
2 Fundamentals
The efficiency of the DLLME technique is governed by the same experimental conditions as LLE. Both extraction and microextraction processes are equilibrium-based and are controlled by the partition coefficient (K), which can be calculated using the following equation:
where, Corg,Eq represents the concentration of the analyte in the extracting solvent, and Caq,Eq represents the concentration of the analyte in the aqueous sample, both measured at equilibrium. The main difference between microextraction and extraction lies in the use of tiny amounts of the extractant (microliters) in DLLME, compared to milliliters in conventional LLE. As a result, Corg,Eq is substantially higher in DLLME when compared to LLE for two primary reasons. Firstly, the small volumes of organic solvents used in DLLME leads to the analyte being highly concentrated due to the inverse relationship between volumes and concentrations. Secondly, Caq,Eq at equilibrium is very high because only a small amount of the analyte migrates to the small layer of organic extractant. However, K must remain constant, which only occurs if Corg,Eq is also very high to maintain the (Corg,Eq/Caq,Eq) ratio.
In DLLME, the analyte partitioning takes place at the interface between the aqueous sample and the immiscible organic extracting solvent. Increasing this interface enhances the efficacy of partitioning and in turn, the efficiency of microextraction. In DLLME, the organic extract is dispersed in the aqueous sample with the aid of a disperser, mechanical force, or both. This dispersion step increases the contact surface area between the two layers, leading to better extraction and higher efficiency. The efficacy of the process can be assessed by calculating the enrichment factor (EF) using the following formula:
where, Caq,int denotes the starting concentration of the analyte in the aqueous sample. The EF can be enhanced by selecting appropriate organic solvents and optimizing the experimental settings that influence the DLLME process. These optimization techniques will be discussed in the following sections.
2.1 Requirements of Organic Solvents Used in DLLME
The DLLME technique relies on the utilization of water immiscible solvents as extractants, while a disperser is used to increase their miscibility in the aqueous sample. A diverse range of organic solvents can serve as organic extractants, and their properties are determined by the specific DLLME mode employed. However, there are certain fundamental characteristics that must be satisfied before utilizing organic solvents as extractants in DLLME. Firstly, the extractant must exhibit low miscibility with the aqueous medium to achieve proper phase separation; this is especially critical as the use of a disperser increases the extractant’s miscibility in the aqueous medium. Secondly, the extractant should possess the capacity to dissolve the target analyte, with high partition coefficients being desirable. Unfortunately, partition coefficient data for all analytes across various solvents is not widely reported, so the documented Kow value for the octanol/water system is often utilized to estimate the lipophilicity of the target analyte. Thirdly, after manual or instrumental shaking, the organic solvent should be dispersible either using an organic disperser or not. Fourthly, the extractant used must be compatible with the subsequent procedure, or else it must be evaporated first. This additional phase may negatively affect the accuracy of the sample preparation procedure, besides the effort and time involved. Finally, the cost of the extractant should also be taken into consideration, as it should be inexpensive to minimize the overall cost of the analytical procedure. Table 1 summarizes the properties of the most widely used solvents in DLLME.
2.2 Experimental Variables in DLLME
There are several experimental factors that can be optimized to increase extraction efficiency in DLLME including solvents types and volume used in extraction and dispersion, sample temperature and pH, salt addition, extraction duration and stirring rate. The most significant of these factors are proper choices of the kind and amount of disperser and extractant. In traditional DLLME, Halogenated hydrocarbons, including chloroform, are frequently employed as extractants, however, in cases where low density solvents are utilized in DLLME modes, 1-undecanol has emerged as the most prevalent extractant. Typically, maximum extraction efficiency is found at lower extractant quantities (20–100 µL). The type and volume of disperser come next in significance. Acetonitrile (ACN) [17] and methanol [18, 19] are the most often utilized dispersants. A few hundreds of microliters (200–800 µL) are frequently sufficient to spread the extracting solvent in the sample. Greater quantities of dispersants are mot recommended due to the unwanted co-solvency, which reduces the efficiency of DLLME [20]. While optimizing the extraction conditions, it is essential to consider the potential interactions between variables. The extraction efficiency of ionizable solutes can be affected by sample pH. The use of acids or bases can potentially alter the ionization process towards the unionized form of the analyte, which is theoretically easier to extract. Similarly, the salting out effect can boost extraction efficiency. Investigating the effects of sample temperature, salt concentration, stirring rate, and extraction duration may aid in achieving the best extraction conditions. Figure 2 shows the effect of extractant volume, pH, dispersion method and centrifugation time on the EF of four different antivirals. As the figure indicated, the most crucial factor was the extractant volume, with markedly higher EFs at lower volumes of the extractant [21]. These factors may be modified at the same time utilizing chemometrics, which can predict the optimal conditions for DLLME with the fewest experimentation, while also predicting variables’ interactions [22, 23].
Optimization of a extractant volume, b pH of diluent, c dispersion technique and d centrifugation time (n = 3). Reprinted from [21] with permission from Elsevier
2.3 Modes of DLLME
The conventional mode of DLLME (also known as normal DLLME or n-DLLME) employs high density organic solvents as extractants, and manual shaking to facilitate in dispersion [24]. The different modes of DLLME can be classified according to the extractant type or the dispersion technique. As for the extractant type, different solvents have been utilized such as low density solvent (LDS), ionic liquids (ILs), deep eutectic solvent (DES), and supramolecular (SUPRAS). On the other hand, DLLME can be categorized according to the dispersion technique into vortex-assisted (VA), ultrasound-assisted (USA), AA and magnetic stirrer assisted (MSA)-DLLME. Phase separation after dispersion is usually induced by centrifugation [25], although solvent-terminated DLLME has been frequently reported [26]. When a dispersion is subjected to centrifugal force, the tiny droplets within the dispersion experience a radial outward force. This force causes these droplets to move away from the axis of rotation, towards the outer edges of the sample centrifuge tube. The centrifugal force in this case accelerates the phase separation. Denser solvents will settle faster than lighter ones, causing them to migrate towards the bottom of the sample, while lighter solvents float on the top [27]. Figure 3 depicts the categorization of the various DLLME modes.
2.3.1 n-DLLME
The most widely employed method for biological analysis is conventional dispersive liquid–liquid microextraction (n-DLLME), which involves combining a suitable disperser with an extractant that is heavier than water [28]. Upon injection of the combined extractant/disperser solvents into the sample, shaking the solution leads to the formation of an unstable emulsion which can be rapidly disrupted by centrifugation. The bottom layer is then collected using a syringe and supplied to the assay equipment. In this context, n-DLLME has been successfully utilized to determine different classes of drugs including antipsychotics [29], antidepressants [30, 31], antimicrobials [32, 33], immunosuppressants [34], antiarrhythmics [35], and drugs of addiction [36,37,38]. Chloroform [39,40,41] is the most commonly used extractant, while methylene chloride [35] and carbon tetrachloride [37] are other often used halogenated hydrocarbons. It should be noted that the disperser needs to possess miscibility with both the sample and the extracting solvent in order to serve as a dispersant. Commonly employed dispersers in n-DLLME include ACN [30], methanol [29], acetone [33], ethanol [37], and tetrahydrofuran (THF) [40]. In some instances, the organic solvent is evaporated before utilizing the analytical tool, and the sample residue is reconstituted in a compatible solvent [41]. n-DLLME has been employed in a range of analytical techniques, including ultraviolet spectrophotometry (UV) [30], mass spectrometry (MS) [34], gas chromatography (GC) [36], high performance liquid chromatography (HPLC) [32], and capillary electrophoresis (CE) [40, 41]. The n-DLLME technique has certain limitations, such as low manual shaking efficiency, high toxicity of organic extractants, and difficulties in automation. Researchers have addressed these challenges by modifying the default processes and developing new modes of DLLME [28].
2.3.2 Ultrasound Assisted DLLME
The most critical step in DLLME is the dispersion. In USA-DLLME, ultrasonic waves are employed to induce better dispersion than manual shaking. This mode has also been termed USA emulsification microextraction (USAEME). The ultrasonic energy was preferentially employed to increase the turbidity of the solution and spread the extractant droplets into the aqueous phase. As a result, the analyte was trapped in these tiny droplets, which make it easily separated from the aqueous solution. Furthermore, the ultrasonic power hastens the transfer of analyte to the extractant phase. To monitor these effects, the ultrasonic settings (temperature, duration, and amplitude of sonication) could be optimized [42] to increase the frequency and rate of material molecular motion, enhance solvent penetration, and thus increase the dispersion degree of extraction solvents. This accelerates the speed of the analytes in the extraction phase, and promote extraction efficiency. Altunay et al. [42] developed USA-DLLME using NADES as extractants for extraction of trace metals from honey by using flame atomic absorption spectroscopy (FAAS). This application studied the effect of ultrasound time and temperature. The effect of sonication period on mass transfer and metal ion recovery was studied from 0 to 20 min at a maximum amplitude of 70%. The recovery values for metal ions were relatively low when ultrasound was not used. The recovery rate for all metal ions rose significantly as the ultrasound period increased up to 10 min, and there was no significant difference in recovery at longer ultrasound times. The influence of ultrasonic bath temperature on the production of NADES droplets with metal ion recovery was also investigated at temperatures ranging from 25 to 60 °C. The best phase separation was attained at 35 °C. No phase separation was accomplished, especially at temperatures over 45 °C. As a result, an ultrasound period of 10 min and a temperature of 35 °C were determined to be appropriate. Generally, ultrasonic bath [43,44,45,46,47] was widely used in USA-DLLME in addition to the ultrasound homogenizer probe, which could be more suitable for limited sample volumes or small extraction vessels [48].
Fernández et al. [49] examined n-DLLME and USA-DLLME for the detection of benzodiazepines in biological fluids; USA-DLLME had greater efficiencies due to the increased dispersion. Moreover, the ultrasonic waves in USA-DLLME obviated the need for a dispersant, reducing solvent usage [50]. Yet, most USA-DLLME applications employ both a disperser and an ultrasonic bath for enhancing the extraction. Fernández and coworkers [51] used USA-DLLME for determination of antidepressants in human plasma by adding 2.5 mL of ACN to samples to act as protein precipitant and dispersant. As an extractant, a volume of 200 µL of chloroform was used. The extracted drugs were tested using UPLC/UV, and the findings were compared to those obtained using traditional LLE connected to GC/MS, there were no substantial differences between the two techniques which indicated that DLLME could enable UV detection to give comparable results to the highly sensitive MS detection. USA-DLLME was also utilized in flow injection analysis (FIA) with inline derivatization through diazotized p-sulfanilic acid to determine tetracyclines in egg supplement samples [52].The reaction was carried out at 45 °C in a slightly alkaline media, and the absorbance at 435 nm was measured using a liquid waveguide capillary cell (LWCC). GC–MS was also utilized to detect seven recreational drugs in human plasma, including meperidine, ketamine, methadone, amphetamine, and amphetamine derviatives. The impact of ultrasound application duration ranged from 0 to 5 min. After 2 min, the optimal sonication was reached. Because of the potential demulsification impact, prolonged duration in the ultrasounds application through a bath was undesired. USA-DLLME was used to extract different analytes from different matrices including aqueous samples [43, 48], food [42] and biological samples [53].
2.3.3 VA-DLLME
Instead of shaking or using ultrasonic waves, the sample can be vortexed with an extractant and a dispersant to induce dispersion. A principal role of vortex is to break down any extraction solvent into tiny droplets, increase the contact area between the droplets and water, and speed up extraction equilibrium, which is dependent on vortex speed and duration [54]. As a result, the vortex promotes the equilibration and dispersion of the target analytes in the extractant and aqueous solution, reducing the equilibrium period [55]. So, vortex time should be enough to achieve equilibration between the aqueous sample and the extractant [56]. Usually, the vortex step is performed with the aid of a vortex agitator [56,57,58]. The multi-tube vortexers were also employed to increase sample throughput. This allows for more applications within the same timeframe and facilitates automation.
Compared with other modes of DLLME, the vortex outperformed the other mechanical agitators in terms of extraction efficiency [59]. The sample/extractant combination is vortexed with or without an organic disperser to generate an emulsion in VA-DLLME. Herrera-Herrera et al. [60] created the VA-DLLME technique for extracting various sulfonamides and quinolones prior to HPLC–UV. As an extractant, chloroform was employed, while acetonitrile was used as a dispersant. Before centrifugation, the ternary mixture was vortexed for 3 min. Interestingly, vortexing had little effect on extraction efficiency, but it dramatically enhanced accuracy. This describes how vortices help to accelerate the equilibrium process. VA-DLLME was widely used for extraction various analytes from diverse sample types including beverages [61], biologicals [62, 63], food [64], and sewage [65]. However, VA-DLLME biological applications are still lower than expected [66].
2.3.4 AA-DLLME
To eliminate the need of equipment in DLLME, an AA-DLLME technique was introduced, in which dispersion was produced simply by aspirating and injecting the extraction mixture with a syringe repeatedly. This approach requires no extra instruments, facilitating the automation process [67]. The principle of AA-DLLME was similar to DLLME in many ways, but there was no requirement for an organic solvent to disperse an extractant into the sample solution. A hydrophobic organic solvent at µL-concentration (extraction solvent) was dispersed into the sample solution using a syringe fitted with a needle and sucking/dispersing cycles numerous times. Despite the absence of a dispersant solvent, this approach considerably enhanced the contact area of the extraction solvent with the sample solution. The investigations revealed that the two primary factors in liquid phase micro extraction (LPME) procedures were extraction solvent viscosity and interfacial tension [68]. These parameters influence both the extractant droplet size and the analyte mass transfer rate. Aspirating-dispersing cycles transform the extraction solvent into extremely small droplets, increasing the contact area of the sample solution with the extraction solvent dramatically. During the aspirating/dispersing cycles in AA-DLLME, there is intense turbulence in the solution, and mass transfer of the analytes is mostly regulated by the convective process [67].
A syringe is used in AA-DLLME to repetitively withdarw and inject the extractant and sample until a hazy solution forms. Farajzadeh et al. [31] used AA-DLLME to preconcentrate NSAIDs in biological fluids. The hazy solution was back-extracted into 10 µL of ammonia buffer (0.1 M, pH 9) after four rounds of recurrent withdrawal and injection before being delivered to an HPLC equipment with UV detection. When the analytical figures of merit were compared to other methods of LPME, it was discovered that AA-DLLME had the highest EF, the maximum sensitivity, and a suitable extraction duration of 10 min. NSAIDs might potentially be chemically derivatized and extracted concurrently with AA-DLLME [69]. The derivatizing agent for GC-FID was butyl chloroformate, while the catalyst was picoline. A syringe was used to aspirate and disseminate the material, which was combined with the chloroform, in presence of the catalyst and the reagents. The number of extraction cycles was investigated; four rounds of repeated injection and withdrawal were found to be adequate to generate dispersion. With five or more extraction cycles, no further improvements in signal intensities were detected. The discovered method’s sensitivity was higher than previously reported GC–MS approaches, and it was less expensive and time-saving. The AA-DLLME techniques’ simplicity and ease of automation improve their potential in biological applications. Barfi et al. [70] compared the performance of ultrasound-enhanced AA-DLLME (USE-AA-DLLME) with previous DLLME techniques that extracted NSAIDs using an organic disperser. Higher EFs were reported with USE-AA-DLLME, which might be attributable to disperser-induced improved solubility in the aqueous sample. So, because of these advantages AA-DLLME including facilitating of the automation and absence of disperser, AA-DLLME was widely used for determination different analytes from different matrices including water [71,72,73], beverages [74], food [75,76,77], biological [78] and biodiesel samples [79].
Rahmani and coworkers [80] compared USA-DLLME, AA-DLLME and VA-DLLME to extract benzene, toluene, ethylbenzene and xylene isomers (BTEX) from water samples. There was no need for a dispersive solvent in any of these procedures, as the extractant is dispersed by air bubbles, vortex, and ultrasound for AA-DLLME, VA-DLLME, and USA-DLLME, respectively. The findings revealed that the three techniques used were highly effective, and the hazy solutions formed were capable of extracting analytes in a relatively short time and with good recoveries (BTEX was used as a simple analyte in these tests). These three procedures were faster, simpler, more sensitive, less costly, and more environmentally friendly than the previous DLLME methods due to the absence of the dispersive solvent. AA-DLLME required a larger volume of extractant and, as a result, required less time to centrifuge. Consequently, AA-DLLME had the shortest analysis time (3 min). The linear dynamic range of AA-DLLME was greater (50–2600 µg /L), although VA-DLLME utilizes less solvent (only 25 µL) and had the best RSD. USA-DLLME has the highest enrichment factor, up to 245-fold.
3 New Developments in DLLME
Since the introduction of DLLME in 2006, tremendous efforts have been exerted to enhance the performance and widen its scope of application. These advances can be categorized into three main trends. The first involves exploring new extractants such as LDS, IL, or DES. The second direction focuses on facilitating the phase separation step such as in DLLME-SFOD. The third direction is geared towards method automation. The following sections discuss these new trends in more details.
3.1 New Extractants in DLLME
3.1.1 Using Low Density Solvents in DLLME
The main obstacles in n-DLLME is the restricted number of extraction solvents and the high toxicity of the halogenated hydrocarbons. These barriers were overcome by utilizing nontoxic organic solvents, with densities lower than water, such as hexane, toluene, xylene, octanol and others in a mode known as low density solvent DLLME (LDS-DLLME). Following dispersion and termination, the floating layer could be delivered to the analytical equipment using a syringe. The LDS-DLLME theory has been studied [81]; nonetheless, applicability in biological fluids are still quite beyond expectations. Ghambari et al. [82] used LDS-DLLME followed by HPLC/UV to extract and evaluate warfarin in plasma. The extraction was carried out in a separate cell. The extraction cell contains deproteinized plasma (pH 2.3 adjusted), an LDS (octanol, 150 µL), a disperser (methanol, 150 µL), and a magnetic stir bar. After the extraction procedure, the extractant collection was facilitated by the long and narrow neck of a special extraction cell. Warfarin extraction recovery was 91%. Applying ultrasonic waves during the dispersion stage boosted LDS-DLLME efficiency. Meng et al. [83] employed SA-LDS-DLLME to determine illicit drugs in plasma. The authors compared their approach to hollow fiber liquid-phase microextraction (HFLPME); USA-LDS-DLLME achieved greater efficiency in shorter extraction times. The fundamental benefit of LDS-DLLME is that the LDS organic extractants are compatible with routinely used HPLC mobile phases. As a result, there is no need for solvent evaporation or reconstitution prior to sample injection. Unfortunately, LDS-DLLME has intrinsic limitations due to the incomplete phase separation following extraction and the challenging automation of the centrifugation step. Other DLLME modes, such as SFOD [84] and ST-DLLME [9, 85] could overcome these problems.
3.1.2 Using ILs in DLLME
ILs have inspired scientists in a variety of research and industrial fields over the last decade. This is demonstrated by the large number of articles in the area of analytical chemistry pertaining to ionic liquids. ILs can be effectively isolated and reused to greatly decrease application costs [86,87,88,89]. Another significant benefit of ionic liquids is the ability to select from a wide range of ions to create an IL with the desired physical and chemical characteristics such as melting point, viscosity, density, and miscibility with water and other solvents. As a result, ILs are frequently referred to as modelling solvents. Ionic liquids’ distinctive characteristics piqued the curiosity of scientists and engineers in the field of extraction and separation [90]. For these reasons, ILs and polymeric ionic liquids (PILs) are used in a variety of applications in DLLME [91,92,93].
The physical and chemical characteristics of ILs are primarily determined by the size, placement, and type of the organic cation and the organic/inorganic anion. The potential of constructing IL structures by selecting the cation and anion that give the necessary physicochemical qualities opens up the possibility of widespread usage of these substances in academic research and industrial applications [94, 95]. ILs are commonly regarded as “green solvents” for their excellent solvation characteristics, low vapour pressure, and low toxicity [92].
Zhou et al. [96] and Baghdadi et al. [97] were the first to use ionic solutions in the DLLME method and to coin the term IL-DLLME [60]. Liu et al. [98] used this IL-DLLME mode for preconcentration and isolation of heterocyclic pesticides in water before HPLC/DAD determination. The IL employed was [C6MIm] [PF6], and the dispersive liquid was methanol [99]. This approach has recently been modified by changing the sample temperature, using ultrasound, microwaves, or more radical modifications such as the in situ IL formation during ME. This in situ IL-DLLME mode was initially suggested by Bahdadi and Shemirani in 2009 and is often referred to as in situ solvent formation microextraction (ISFME) [100]. This in situ IL-DLLME technique works by dissolving hydrophilic IL in an aqueous solution containing the analytes of interest, then adding an ion-exchange reagent to create an insoluble IL. An ion-exchange reagent supports a metathesis reaction, which transforms the hydrophilic IL into a hydrophobic one that settles and preconcentrates the analytes. Yao and Anderson [101] used a similar method for the measurement of aromatic hydrocarbons.
Although ILs have been shown to be good extractants in DLLME, phase separation still needs centrifugation, which is time-consuming and difficult to automate. So, an innovative family of ionic liquids with magnetic properties, known as magnetic ionic liquids (MILs), has been synthesized, which frequently feature an imidazolium [102], choline [103], or phosphonium cation [104] and a paramagnetic metal (Fe, Co, Mn, or Gd) chloride anion. The higher the magnetic susceptibility, the easier the phase separation in the presence of an external magnet. For this reason, Abdelaziz et al. [105] used a hydrophobic gadolinium-based MILfor the first time as extraction solvent in DLLME. In this work, the produced Gd(III)-based MIL demonstrated hydrolysis resilience in aqueous samples as well as a minimal UV noise signal. Furthermore, the suggested MIL’s acceptable viscosity promotes analyte partitioning, speeds phase separation, and simplifies extract handling and transfer into the analytical instrument. Furthermore, the introduced Gd-mased MIL showed significantly high magnetic susceptibility, enabling for quicker extraction solvent recovery with a powerful magnet.
3.1.3 Using Deep Eutectic Solvent in DLLME
One of the objectives of implementing the DLLME method is to utilize environmentally friendly green solvents [106]. The critical step in this aspect is to prepare a solvent that is not only green but also offers efficient extraction [107]. Typically, the solvents used in DLLME methods are toxic, which has spurred the development of remarkable and ecologically favorable green solvents [108]. DESs are typically made up of hydrogen bond acceptors (HBAs) and hydrogen bond donors (HBDs). HBAs are frequently quaternary ammonium compounds, whereas HBDs are amines, carboxylic acids, alcohols, polyols, or carbohydrates [109, 110]. Because of the creation of intramolecular hydrogen bonds, DESs have a much lower melting point than their separate components. DESs have low volatility, low vapour pressure, a reasonably broad liquid range, and high heat durability [111]. Furthermore, DESs are readily produced without the need for purification stages, and they are made from low-cost compounds with low or minimal toxicity. DES are also biodegradable and easily reusable. These characteristics make DESs superior to traditional solvents used in extraction processes [112], especially DLLME extraction and isolation of bioactive substances [113, 114]. One important benefit of DESs, for example, is their ability to be tuned to accomplish specific functionality due to the numerous possibilities of beginning components. The selectivity of DESs for extraction and separation can be adjusted by altering the structure and molar ratio of their hydrogen-bonding components [115]. DESs have been categorized into four kinds, as shown in Fig. 4: Type I (metal halide and quaternary salt), Type II (quaternary salt and hydrated metal halide), Type III (quaternary salt and hydrogen bond donor), Type IV (metal halide and HBDs), and Type V (HBD and HBA). This class is particularly important in the microextraction and sample preparation of ionic and highly polar analytes [116,117,118].
3.1.4 Using SUPRAS in DLLME
SUPRASs are nano-structured liquids produced through self-assembly processes occurring at molecular and nanometer scales from amphiphiles [119]. These solvents have been used in extraction processes for many years under different names such as cloud point technique and coacervates [120, 121], and offer a set of appealing intrinsic properties, including the use of self-assembly based synthetic procedures, widespread availability of amphiphiles, tunability of solvent properties, and excellent solvation properties for various compounds [119]. SUPRASs are formed through consecutive self-assembly processes that occur at molecular and nanometer levels, where amphiphiles form three-dimensional aggregates that separate from the bulk solution as a new liquid phase via coacervation when the critical aggregation concentration is reached (Fig. 5) [122]. Two main types of SUPRAS, vesicle-based and reverse micelle-based, have been developed for analytical extractions, with driving forces for effective solubilization and high extraction efficiency being dispersion forces between hydrocarbon chains and analytes, cation interactions between aromatic rings of complexes and amphiphiles, and hydrogen bonding between nitrogen and oxygen atoms in complexes and carboxylic acids from [123, 124]. Reverse micelle-based SUPRAS using THF has shown greater potential for DLLME compared to vesicle-based SUPRAS [125, 126].
Self-assembly processes in supramolecular solvent formation. Reprinted from [127] with permission from Elsevier
The initial self-assembly process in supramolecular solvent production is the accumulation of amphiphilic molecules in a variety of nanostructures. As a critical aggregation concentration (CAC) is reached, amphiphiles spontaneously aggregate to minimise adverse solvophobic interactions [128, 129]. It becomes energetically advantageous for amphiphiles to interact with one another at the CAC. Colloidal self-assembled structures result from the intricate interplay of solute–solvent and solute–solute interactions.
Seidi et al. [123] developed DLLME-SFOD based on a vesicular SUPRAS of decanoic acid and quaternary ammonium compound for extraction of cadmium, the extraction mechanism of cadmium depended on the SUPRAS structure contains polar and apolar groups, their various interactions with analytes can boost extraction efficiency. The interactions between the vesicular SUPRAS and the Cd(II)-(2-pyridylazo)-2-naphthol (PAN), PAN complex are shown in Fig. 6. The main extraction driving forces appear to be three types of interactions: (1) dispersion forces between the hydrocarbon chains of the amphiphile and the analyte; (2) -cation interactions between the aromatic rings of the Cd(II)-PAN complex and Bu4N+; and (3) hydrogen bonding between the nitrogen and oxygen atoms in the Cd(II)-PAN complex and hydrogen of carboxylic acid. These interactions allow for effective solubilization of Cd (II)-PAN in the SUPRAS as well as high extraction efficiency.
a Chemical interaction can influence vesicle formation and its stability, b hydrogen bonding in vesicular formation, and c molecular mechanism of microextraction and different interactions between Cd (II)—PAN complex and the vesicle. Reprinted from [123] with permission from Springer Nature
Bendito et al. [124] proposed a novel type of SUPRAS-based extraction constituted of reverse micelles of decanoic acid (DeA) distributed in a water/THF combination in 2007. They demonstrated that polar and non-polar molecules were extracted into SUPRAS using hydrogen bonding and Van der Waals interactions that reverse micelles may generate. A series self-assembly model predicts that the dissolved DeA in THF producing reverse micelles has at least three critical micelle concentration (CMC) points (4.8 ± 0.2, 7.6 ± 0.4, and 51 ± 2 M).When water is added to this combination, the aggregates partially dissolve, facilitating contact and encouraging the formation of larger reverse micelles as an immiscible liquid phase separate from the THF/water bulk solution [125, 126]. It is worth mentioning that using THF based SUPRAS is much more than vesicular SUPRAS in DLLME.
3.2 Phase Separation by SFOD
DLLME-SFOD use low melting point solvents (10–25 °C), such as 1-undecanol and 1-dodecanol [84, 130]. The floating droplet is solidified using an icebox after dispersion and phase separation and then transferred using spatula or forceps. The key benefit of DLLME-SFOD is the ease with which the extracted phase may be separated. In pharmaceutical and biological analysis, DLLME-SFOD is the second most often utilized mode of DLLME. DLLME-SFOD has been used to determine several pharmaceuticals [131,132,133,134,135,136,137,138] and drugs of abuse [139,140,141,142] in dosage forms as well as biological fluids such as urine [136], plasma [138, 140], milk [143] and tissues. While being extensively recognised in biomedical analysis, DLLME-SFOD has several drawbacks; to extract the analyte of interest, two organic solvents (the extractant and the disperser) are required. This issue can be avoided by using mechanically-induced dispersion, as in USA-DLLME-SFOD and AA-DLLME-SFOD [144]. The other issue stemmed from the centrifugation stage, which slowed the extraction process and hampered automation. ST-DLLME [145] can be used to solve this problem by adding a demulsifying solvent. To break the emulsion and produce phase separation, a demulsifying solvent is added to the sample/extractant/disperser combination in ST-DLLME. In this situation, the centrifugation stage can be skipped, allowing for process automation and a reduction in overall analytical time. The main barrier in DLLME-SFOD is the limited number of solvents that can solidify at relatively low temperature without causing the whole sample to freeze. Exploring other solvents especially those from botanical origin with relatively low melting point is highly recommended.
3.3 Automation of DLLME
Automation is one of the DLLME technique’s ongoing problems. Several developments in DLLME have relied on flow analysis methods [146]. Initially, DLLME was automated using the sequential injection analysis (SIA) approach and used to determine metals by flame or electrothermal atomic absorption spectrometry [147,148,149,150]. A comparable method was developed utilizing the flow injection analysis methodology, which performed online DLLME using ionic liquids [151,152,153]. DLLME has also been automated utilizing a dual SIA system, which connects both syringe burettes by a conical tube that serves as the extraction container [154].
SIA rendered the automation of DLLME possible [155], through a multi-axis robotic arm with an integrated phase separator and temperature control. This setup allowed for the automatic solidification of the organic phase, followed by the collection of the organic extract for analyte measurement. The automated DLLME-SFOD of parabens was examined as a proof-of-concept, followed by analyte separation using liquid chromatography. Medina et al. [156] and coworkers developed an automated method in which everything was automated by combining a SIA technology with a custom-made robotic phase separator. Then, phase separation was performed in a 3D printed device incorporating a Peltier cell set and placed on a multi-axis robotic arm. A single software package controls the combined action of the flow system and the robotic arm, allowing for the solidification/melting and collection of the organic phase for subsequent analyte measurement as indicated in Fig. 7.
Major steps of the automated DLLME-SFOD. Reprinted from [156] with permission from Elsevier
Another approach for DLLME automation is the completion of the extraction “in-syringe” [15]. In case, syringes are employed as DLLME containers, and the separated extractant droplets can be collected at the top of the syringe, ready to be automatically injected into the detection system, which is interfaced by an injection valve, utilizing solvents lighter than water [81]. Shishov et al. [157] developed an automated in syringe DLLME for chromium detection in beverages. As indicated in Fig. 8, in the first phase, 0.6 mL of extraction mixture (port a, valve) was aspirated into the syringe via channel 1 by back movement of the syringe pump plunger at a speed of 1.5 mL/ min. The valve was then switched to port b, and 4 mL of sample was aspirated into the syringe at a rate of 10 mL /min. Furthermore, 0.4 mL of air (port c, valve) was sucked to eliminate any leftover sample in channel 1. For 60 s, the sample and extraction liquid were mixed together. The syringe pump and stirrer were turned off for 30 s to allow for extraction and phase separation. Finally, the upper phase was transported into the flow cell of the UV–Vis detector (channel 2), and absorbance was measured under stopped-flow conditions for 5 s at 540 nm before the solution was supplied to trash. After each measurement, the syringe and flow cell were rinsed with 1 mL of isopropyl alcohol (port d). The automation did not compromise the analytical figures of merits, including linearity, selectivity, sensitivity, accuracy and precision. Maya et al. developed a fully automated DLLME for the determination of rhodamine B with integrated spectrophotometric detection [15]. The results indicated that rhodamine B was measured in a working range of 0.023–2 mg/L with a limit of detection of 0.007 mg/L. The method also showed good repeatability for 10 successive extractions, with % RSD values of up to 3.2%. The EF for a 1 mg/L rhodamine B standard was found to be 23, and the method was capable of performing 51 extractions in 1 h.
The manifold of automated procedure for the determination of chromium (VI) in beverages. Reprinted from [157] with permission from Elsevier
4 Application of DLLME
The different modes of DLLME have been extensively used to pre-concentrate analytes of different nature from a variety of samples. Plasma, urine, hair, milk, fruits, vegetables, seafood and water samples were treated by DLLME before analysis. Drugs, toxins, pesticides, preservatives and heavy metals were all enriched with the aid of different modes of DLLME. According to the dispersion technique, n-DLLME and USA-DLLME are the most commonly used modes, followed by VA-DLLME and AA-DLLME. The average sample size is 5–10 mL, but amounts as small as 0.05 mL were also reported. In this case, a dilution step is required before sample preparation to facilitate dispersion. Large sample volumes were also prepared using DLLME, to allow for ultrasensitive determination of heavy metals. The type of extracting solvent depends on the selected DLLME mode, where chloroform is the widely used solvent in n-DLLME, while decanol is very common in DLLME-SFOD. Methanol, ACN and THF are the most popular dispersers due to availability, and high miscibility with both organic solvents and aqueous samples. The volume of disperser is usually less than 1000 μL, and it is highly dependent on the sample size and the extractant volume and type. DLLME has been extensively coupled to HPLC with different detectors including UV, FLD and MS. Application of DLLME before CE was also reported. Both HPLC and CE require minimal sample volumes to be injected into the instrument, which may explain the wide spread of DLLME with these particular analytical techniques. DLLME could also be used before UV/Vis spectrophotometric and spectrofluorometric determinations, if a microcuvette was available. An alternative approach in UV/Vis spectrophotometry was to measure the extracted small sample via a Nanodrop spectrophotometer. Table 2 shows some selected applications of the different modes of DLLME.
5 Conclusions and Future Trends
DLLME has attracted the interest of the analytical community since its introduction in 2006, owing to its simplicity and strong analytical capabilities. However, traditional DLLME has had one shortcoming from its emergence: the use of high-density halogenated solvents. Despite the high efficiency, these halogenated solvents are very hazardous, and a process utilising this type of solvent cannot be termed green even if the amount required was in microliter units. As a result, scientists have been seeking for solvents that are not only safe for the environment and operators, but also capable of improving the extraction efficiency of DLLME-based procedures. Many approaches have been presented in this regard. The development of the various types of solvents utilized in DLLME over the last 5 years has been examined in this chapter. SUPRAs and DESs offer exceptional qualities for microextraction and some advantages from being termed green solvents. Nonetheless, the field of chemistry is conservative in certain ways, and many DLLME experiments continue to employ traditional halogenated solvents in accordance with the guideline. The benefits of these traditional solvents, such as their ease of use and high density are obvious but we must not overlook the significant impact that these solvents have on health and the environment. As a result, the adoption of newer and greener solvents must be the goal of DLLME in the next years, with an emphasis on tailorable green solvents with high extraction capabilities and simple and safe synthesis.
The authors have declared no conflict of interest.
Abbreviations
- ACN:
-
Acetonitrile
- DES:
-
Deep eutectic solvent
- DLLME:
-
Dispersive liquid–liquid microextraction
- HBA:
-
Hydrogen bond acceptor
- HBD:
-
Hydrogen bond donor
- HLLME:
-
Homogeneous liquid–liquid microextraction
- LLE:
-
Liquid–liquid extraction
- LPME:
-
Liquid phase microextraction
- LLME:
-
Liquid–liquid microextraction
- THF:
-
Tetrahydrofuran
- EF:
-
enrichment factor
- DLLME-SFOD:
-
solidified organic droplet
- ST-DLLME:
-
solvent terminated-DLLME
- AA-DLLME:
-
Air assisted-DLLME
- VA-DLLME:
-
vortex assisted-DLLME
- USA-DLLME:
-
Ultrasound assisted-DLLME
- MSA-assisted DLLME:
-
magnetic stirrer assisted DLLME
- n-DLLME:
-
normal DLLME
- DES:
-
deep eutectic solvent
- NADES:
-
Natural deep eutectic solvent
- IL:
-
ionic liquid
- PIL:
-
polymeric ionic liquid
- CAC:
-
critical aggregation concentration
- CMC:
-
critical micelle concentration
- K:
-
Partition coefficient
- LDS:
-
low density solvent
- UV:
-
ultraviolet spectrophotometry
- MS:
-
mass spectrometry
- GC:
-
gas chromatography
- HPLC:
-
high performance liquid chromatography
- CE:
-
capillary electrophoresis
- FAAS:
-
flame atomic absorption spectroscopy
- FIA:
-
flow injection analysis
- SIA:
-
sequential injection analysis
- USAEME:
-
USA emulsification microextraction
- LWCC:
-
liquid waveguide capillary cell
- ISFME:
-
in situ solvent formation microextraction
- DeA:
-
decanoic acid
- PAN:
-
2-pyridylazo-2-naphthol
References
Hammad SF, Abdallah IA, Bedair A, Mansour FR (2022) Homogeneous liquid–liquid extraction as an alternative sample preparation technique for biomedical analysis. J Sep Sci 45:185–209. https://doi.org/10.1002/jssc.202100452
Lee J, Lee HK, Rasmussen KE, Pedersen-Bjergaard S (2008) Environmental and bioanalytical applications of hollow fiber membrane liquid-phase microextraction: a review. Anal Chim Acta 624:253–268. https://doi.org/10.1016/j.aca.2008.06.050
Kannouma RE, Hammad MA, Kamal AH, Mansour FR (2022) Miniaturization of liquid–liquid extraction; the barriers and the enablers. Microchem J 182:107863. https://doi.org/10.1016/j.microc.2022.107863
Yamini Y, Rezazadeh M, Seidi S (2019) Liquid-phase microextraction—the different principles and configurations. TrAC—Trends Anal Chem 112:264–272. https://doi.org/10.1016/j.trac.2018.06.010
Rezaee M, Assadi Y, Milani Hosseini M-R, Aghaee E, Ahmadi F, Berijani S (2006) Determination of organic compounds in water using dispersive liquid–liquid microextraction. J Chromatogr A 1116:1–9. https://doi.org/10.1016/j.chroma.2006.03.007
Mansour FR, Khairy MA (2017) Pharmaceutical and biomedical applications of dispersive liquid–liquid microextraction. J Chromatogr B 1061–1062:382–391. https://doi.org/10.1016/J.JCHROMB.2017.07.055
Khalili Zanjani MR, Yamini Y, Shariati S, Jönsson JÅ (2007) A new liquid-phase microextraction method based on solidification of floating organic drop. Anal Chim Acta 585:286–293. https://doi.org/10.1016/j.aca.2006.12.049
Regueiro J, Llompart M, Garcia-Jares C, Garcia-Monteagudo JC, Cela R (2008) Ultrasound-assisted emulsification—microextraction of emergent contaminants and pesticides in environmental waters. J Chromatogr A 1190:27–38. https://doi.org/10.1016/j.chroma.2008.02.091
Chen H, Chen R, Li S (2010) Low-density extraction solvent-based solvent terminated dispersive liquid–liquid microextraction combined with gas chromatography-tandem mass spectrometry for the determination of carbamate pesticides in water samples. J Chromatogr A 1217:1244–1248. https://doi.org/10.1016/j.chroma.2009.12.062
Jafarvand S, Shemirani F (2011) Supramolecular-based dispersive liquid–liquid microextraction: a novel sample preparation technique utilizes coacervates and reverse micelles. J Sep Sci 34:455–461. https://doi.org/10.1002/jssc.201000630
Jafarvand S, Shemirani F (2011) Supramolecular—based dispersive liquid–liquid microextraction: a novel sample preparation technique for determination of inorganic species. Microchim Acta 173:353–359. https://doi.org/10.1007/s00604-011-0564-9
Farajzadeh MA, Mogaddam MRA (2012) Air-assisted liquid–liquid microextraction method as a novel microextraction technique; Application in extraction and preconcentration of phthalate esters in aqueous sample followed by gas chromatography–flame ionization detection. Anal Chim Acta 728:31–38. https://doi.org/10.1016/j.aca.2012.03.031
Wang Y, Sun Y, Xu B, Li X, Jin R, Zhang H, Song D (2014) Magnetic ionic liquid-based dispersive liquid–liquid microextraction for the determination of triazine herbicides in vegetable oils by liquid chromatography. J Chromatogr A 1373:9–16. https://doi.org/10.1016/j.chroma.2014.11.009
Ortega-Zamora C, González-Sálamo J, González-Sálamo J, Hernández-Sánchez C, Hernández-Sánchez C, Hernández-Borges J, Hernández-Borges J (2020) Menthol-based deep eutectic solvent dispersive liquid–liquid microextraction: a simple and quick approach for the analysis of phthalic acid esters from water and beverage samples. ACS Sustain Chem Eng 8:8783–8794. https://doi.org/10.1021/acssuschemeng.0c02603
Maya F, Estela JM, Cerdà V (2012) Completely automated in-syringe dispersive liquid–liquid microextraction using solvents lighter than water. Anal Bioanal Chem 402:1383–1388. https://doi.org/10.1007/s00216-011-5572-4
Dean JA (1999) Lange’s handbook of chemistry, fifteenth. McGraw Hill Inc., New York, USA
Mahmoudpour M, Mohtadinia J, Mousavi M, Ansarin M, Nemati M (2017) Application of the microwave-assisted extraction and dispersive liquid–liquid microextraction for the analysis of PAHs in smoked rice. Food Anal Methods 10:277–286. https://doi.org/10.1007/s12161-016-0579-2
Kalhor H, Hashemipour S, Yaftian MR, Shahdousti P (2016) Determination of carbamazepine in formulation samples using dispersive liquid–liquid microextraction method followed by ion mobility spectrometry. Int J Ion Mobil Spectrom 19:51–56. https://doi.org/10.1007/s12127-015-0184-x
Mookantsa SOS, Dube S, Nindi MM (2016) Development and application of a dispersive liquid–liquid microextraction method for the determination of tetracyclines in beef by liquid chromatography mass spectrometry. Talanta 148:321–328. https://doi.org/10.1016/j.talanta.2015.11.006
Ahmadi-Jouibari T, Fattahi N, Shamsipur M (2014) Rapid extraction and determination of amphetamines in human urine samples using dispersive liquid–liquid microextraction and solidification of floating organic drop followed by high performance liquid chromatography. J Pharm Biomed Anal 94:145–151. https://doi.org/10.1016/j.jpba.2014.01.044
Kannouma RE, Hammad MA, Kamal AH, Mansour FR (2021) A dispersive liquid–liquid microextraction method based on solidification of floating organic droplet for determination of antiviral agents in environmental water using HPLC/UV. Microchem J 171:106790. https://doi.org/10.1016/j.microc.2021.106790
Wang R, Qi X, Zhao L, Liu S, Gao S, Ma X, Deng Y (2016) Ionic-liquid-based dispersive liquid–liquid microextraction coupled with high-performance liquid chromatography for the forensic determination of methamphetamine in human urine. J Sep Sci 39:2444–2450. https://doi.org/10.1002/jssc.201600170
Monzón CM, Teglia CM, Delfino MR, Goicoechea HC (2016) Chemometric optimization and validation of a novel dispersive liquid–liquid microextraction–HPLC method for gliclazide, glibenclamide and glimepiride quantitation in serum samples. Microchem J 127:113–119. https://doi.org/10.1016/j.microc.2016.02.011
Šandrejová J, Campillo N, Viñas P, Andruch V (2016) Classification and terminology in dispersive liquid–liquid microextraction. Microchem J 127:184–186. https://doi.org/10.1016/j.microc.2016.03.007
Kamal AH, Hammad MA, Kannouma RE, Mansour FR (2022) Response surface optimization of a vortex-assisted dispersive liquid–liquid microextraction method for highly sensitive determination of repaglinide in environmental water by HPLC/UV. BMC Chem 16:33. https://doi.org/10.1186/s13065-022-00826-w
Mansour FR, Danielson ND (2018) Solvent-terminated dispersive liquid–liquid microextraction: a tutorial. Anal Chim Acta 1016:1–11. https://doi.org/10.1016/j.aca.2018.02.005
Cölfen H, Rosenberg R, Haffke D, Stemplinger S, Zemb T, Horinek D (2023) Two types of liquid phase separation induced by soft centrifugation in aqueous ethyl acetate using ethanol as cosolvent. Research 6:A67. https://doi.org/10.34133/research.0026
Viñas P, Campillo N, López-García I, Hernández-Córdoba M (2014) Dispersive liquid–liquid microextraction in food analysis. A critical review Microextraction Techniques. Anal Bioanal Chem 406:2067–2099. https://doi.org/10.1007/s00216-013-7344-9
Fisichella M, Odoardi S, Strano-Rossi S (2015) High-throughput dispersive liquid/liquid microextraction (DLLME) method for the rapid determination of drugs of abuse, benzodiazepines and other psychotropic medications in blood samples by liquid chromatography–tandem mass spectrometry (LC-MS/MS) and app. Microchem J 123:33–41
Bulatov A, Medinskaia K, Aseeva D, Garmonov S, Moskvin L (2014) Talanta Determination of antipyrine in saliva using the dispersive liquid–liquid microextraction based on a stepwise injection system. Talanta 133:66–70. https://doi.org/10.1016/j.talanta.2014.05.064
Farajzadeh MA, Reza M, Mogaddam A, Bamorowat M (2015) Determination of unconjugated non-steroidal anti-inflammatory drugs in biological fluids using air-assisted liquid–liquid microextraction combined with back extraction followed by high performance liquid chromatography. Anal Methods 7:1372–1379. https://doi.org/10.1039/c4ay01996b
Deng K-J, Lan X-H, Sun G, Ji LY, Zheng X-L (2016) Determination of sulfonamide residues in chicken liver using high-performance liquid chromatography. Food Anal Methods 9:3337–3344. https://doi.org/10.1007/s12161-016-0514-6
Lin SY, Chen PS, Chang SY (2015) Detection of posaconazole by surface-assisted laser desorption/ionization mass spectrometry with dispersive liquid–liquid microextraction. J Am Soc Mass Spectrom 26:530–533. https://doi.org/10.1007/s13361-014-1061-2
Chen P-S, Cheng Y-H, Lin S-Y, Chang SY (2016) Determination of immunosuppressive drugs in human urine and serum by surface-assisted laser desorption/ionization mass spectrometry with dispersive liquid–liquid microextraction. Anal Bioanal Chem 408:629–637
Jouyban A, Sorouraddin MH, Farajzadeh MA, Somi MH, Fazeli-Bakhtiyari R (2015) Determination of five antiarrhythmic drugs in human plasma by dispersive liquid–liquid microextraction and high-performance liquid chromatography. Talanta 134:681–689. https://doi.org/10.1016/j.talanta.2014.12.008
Cunha RL, Lopes WA, Pereira PAP (2016) Determination of free (unconjugated) amphetamine-type stimulants in urine samples by dispersive liquid–liquid microextraction and gas chromatography coupled to mass spectrometry (DLLME-GC-MS). Microchem J 125:230–235. https://doi.org/10.1016/j.microc.2015.11.017
Habibollahi S, Tavakkoli N, Nasirian V, Khani H (2015) Determination of tramadol by dispersive liquid–liquid microextraction combined with GC-MS. J Chromatogr Sci 53:655–661. https://doi.org/10.1093/chromsci/bmu118
Gardner MA, Sampsel S, Jenkins WW, Owens JE (2015) Analysis of fentanyl in urine by DLLME-GC-MS. J Anal Toxicol 39:118–125. https://doi.org/10.1093/jat/bku136
Shamsipur M, Mirmohammadi M (2014) High performance liquid chromatographic determination of ultra traces of two tricyclic antidepressant drugs imipramine and trimipramine in urine samples after their dispersive liquid–liquid microextraction coupled with response surface optimization. J Pharm Biomed Anal 100:271–278. https://doi.org/10.1016/j.jpba.2014.08.008
Hsieh M, Chang SY (2015) Sensitive detection of piperazinyl phenothiazine drugs by field-amplified sample stacking in capillary electrophoresis with dispersive liquid–liquid microextraction. Electrophoresis. https://doi.org/10.1002/elps.201500260
de Albuquerque NCP, de Gaitani CM, de Oliveira ARM (2015) A new and fast DLLME-CE method for the enantioselective analysis of zopiclone and its active metabolite after fungal biotransformation. J Pharm Biomed Anal 109:192–201. https://doi.org/10.1016/j.jpba.2015.02.039
Altunay N, Elik A, Gürkan R (2019) Monitoring of some trace metals in honeys by flame atomic absorption spectrometry after ultrasound assisted-dispersive liquid liquid microextraction using natural deep eutectic solvent. Microchem J 147:49–59. https://doi.org/10.1016/j.microc.2019.03.003
Ali J, Tuzen M, Citak D, Uluozlu OD, Mendil D, Kazi TG, Afridi HI (2019) Separation and preconcentration of trivalent chromium in environmental waters by using deep eutectic solvent with ultrasound-assisted based dispersive liquid–liquid microextraction method. J Mol Liq 291:111299. https://doi.org/10.1016/j.molliq.2019.111299
Campone L, Celano R, Piccinelli AL, Pagano I, Cicero N, Di SR, Carabetta S, Russo M, Rastrelli L (2019) Ultrasound assisted dispersive liquid–liquid microextraction for fast and accurate analysis of chloramphenicol in honey. Food Res Int 115:572–579. https://doi.org/10.1016/j.foodres.2018.09.006
Altunay N (2021) Optimization of ultrasound-assisted dispersive liquid–liquid microextraction of niacinamide in pharmaceutical and cosmetic samples using experimental design. Microchem J 170:106659. https://doi.org/10.1016/j.microc.2021.106659
Custodio-Mendoza JA, Caamaño-Fernandez C, Lage MA, Almeida PJ, Lorenzo RA, Carro AM (2022) GC–MS determination of malondialdehyde, acrolein, and 4-hydroxy-2-nonenal by ultrasound-assisted dispersive liquid–liquid microextraction in beverages. Food Chem 384:132530. https://doi.org/10.1016/j.foodchem.2022.132530
Hsieh CZ, Chung WH, Ding WH (2021) Experimental design approaches to optimize ultrasound-assisted simultaneous-silylation dispersive liquid–liquid microextraction for the rapid determination of parabens in water samples. RSC Adv 11:23607–23615. https://doi.org/10.1039/d1ra04195a
Werner J (2020) Ligandless, deep eutectic solvent-based ultrasound-assisted dispersive liquid–liquid microextraction with solidification of the aqueous phase for preconcentration of lead, cadmium, cobalt and nickel in water samples. J Sep Sci 43:1297–1305. https://doi.org/10.1002/jssc.201901184
Fernández P, González C, Pena MT, Carro AM, Lorenzo RA (2013) A rapid ultrasound-assisted dispersive liquid–liquid microextraction followed by ultra-performance liquid chromatography for the simultaneous determination of seven benzodiazepines in human plasma samples. Anal Chim Acta 767:88–96. https://doi.org/10.1016/j.aca.2013.01.016
Sajedi-Amin S, Assadpour-Zeynali K, Panahi-Azar V, Kebriaeezadeh A, Khoubnasabjafari M, Ansarin K, Jouyban-Gharamaleki V, Jouyban A (2015) Spectroscopic analysis of bosentan in biological samples after a liquid–liquid microextraction. Bioimpacts 5:191–197. https://doi.org/10.15171/bi.2015.28
Fernández P, Taboada V, Regenjo M, Morales L, Alvarez I, Carro AM, Lorenzo RA (2016) Optimization of ultrasound assisted dispersive liquid–liquid microextraction of six antidepressants in human plasma using experimental design. J Pharm Biomed Anal 124:189–197. https://doi.org/10.1016/j.jpba.2016.02.041
Rodríguez MP, Pezza HR, Pezza L (2016) Ultrasound-assisted dispersive liquid–liquid microextraction of tetracycline drugs from egg supplements before flow injection analysis coupled to a liquid waveguide capillary cell. Anal Bioanal Chem 408:6201–6211. https://doi.org/10.1007/s00216-016-9732-4
Zhou GS, Yuan YC, Yin Y, Tang YP, Xu RJ, Liu Y, Chen PD, Yin L, Duan JA (2020) Hydrophilic interaction chromatography combined with ultrasound-assisted ionic liquid dispersive liquid–liquid microextraction for determination of underivatized neurotransmitters in dementia patients’ urine samples. Anal Chim Acta 1107:74–84. https://doi.org/10.1016/j.aca.2020.02.027
Wang Q, Fan J (2019) A hydrophobic deep eutectic solvent-based vortex-assisted dispersive liquid–liquid microextraction combined with HPLC for the determination of nitrite in water and biological samples. J Sep Sci 42(2):574–581. https://doi.org/10.1002/jssc.201800921
Li T, Song Y, Li J, Zhang M, Shi Y, Fan J (2019) New low viscous hydrophobic deep eutectic solvents in vortex-assisted liquid–liquid microextraction for the determination of phthalate esters from food-contacted plastics. Food Chem 309:125752. https://doi.org/10.1016/j.foodchem.2019.125752
Gholami Z, Hossein M, Yousefinejad S (2021) Vortex-assisted dispersive liquid–liquid microextraction based on hydrophobic deep eutectic solvent for the simultaneous identification of eight synthetic dyes in jellies and drinks using HPLC-PDA. Microchem J 170:106671. https://doi.org/10.1016/j.microc.2021.106671
Abdallah IA, Hammad SF, Bedair A, Mansour FR (2022) Menthol-assisted homogenous liquid–liquid microextraction for HPLC/UV determination of favipiravir as an antiviral for COVID-19 in human plasma. J Chromatogr B 1189:123087. https://doi.org/10.1016/j.jchromb.2021.123087
Abdallah IA, Hammad SF, Bedair A, Elshafeey AH, Mansour FR (2022) Determination of favipiravir in human plasma using homogeneous liquid–liquid microextraction followed by HPLC/UV. Bioanalysis 14:205–216. https://doi.org/10.4155/bio-2021-0219
Guiñez M, Martinez LD, Fernandez L, Cerutti S (2017) Dispersive liquid–liquid microextraction based on solidification of floating organic drop and fluorescence detection for the determination of nitrated polycyclic aromatic hydrocarbons in aqueous samples. Microchem J 131:1–8. https://doi.org/10.1016/j.microc.2016.10.020
Herrera-herrera AV, Hernández-borges J, Borges-miquel TM, Rodríguez-delgado MÁ (2013) Dispersive liquid–liquid microextraction combined with ultra-high performance liquid chromatography for the simultaneous determination of 25 sulfonamide and quinolone antibiotics in water samples. J Pharm Biomed Anal 75:130–137. https://doi.org/10.1016/j.jpba.2012.11.026
Santana-mayor A, Herrera-herrera AV, Rodr R, Miguel A (2021) Development of a green alternative vortex-assisted dispersive liquid−liquid microextraction based on natural hydrophobic deep eutectic solvents for the analysis of phthalate esters in soft drinks. ACS Sustain Chem Eng 9(5):2161–2170. https://doi.org/10.1021/acssuschemeng.0c07686
Golpayegani MR, Akramipour R, Gheini S, Amini MV, Fattahi F, Mohebbi A, Fattahi N (2022) Sensitive determination of vincristine in plasma of children with leukaemia using vortex-assisted dispersive liquid–liquid microextraction based on hydrophobic deep eutectic solvent. RSC Adv 12:3611–3617. https://doi.org/10.1039/d1ra07981f
Bocato MZ, Cesila CA, Lataro BF, de Oliveira ARM, Campíglia AD, Barbosa F (2020) A fast-multiclass method for the determination of 21 endocrine disruptors in human urine by using vortex-assisted dispersive liquid–liquid microextraction (VADLLME) and LC-MS/MS. Environ Res 189:109883. https://doi.org/10.1016/j.envres.2020.109883
Elencovan V, Joseph J, Yahaya N, Abdul N, Raoov M, Lim V, Nadhirah N, Zain M (2022) Exploring a novel deep eutectic solvents combined with vortex assisted dispersive liquid–liquid microextraction and its toxicity for organophosphorus pesticides analysis from honey and fruit samples. Food Chem 368:130835. https://doi.org/10.1016/j.foodchem.2021.130835
Wu Y, Chen M, Wang X, Zhou Y, Xu M, Zhang Z (2021) Development and validation of vortex-assisted dispersive liquid–liquid microextraction method based on solidification of floating hydrophobic deep eutectic solvent for the determination of endocrine disrupting chemicals in sewage. Microchem J 163:105915. https://doi.org/10.1016/j.microc.2020.105915
Bosch Ojeda C, Sánchez Rojas F (2014) Vortex-assisted liquid–liquid microextraction (VALLME): applications. Chromatographia 77:745–754. https://doi.org/10.1007/s10337-014-2669-x
Mogaddam A (2019) Air–assisted liquid–liquid microextraction; principles and applications with analytical instruments. Trends Anal Chem 112:115734. https://doi.org/10.1016/j.trac.2019.115734
Pena-pereira F, Lavilla I, Bendicho C (2010) Liquid-phase microextraction techniques within the framework of green chemistry. Trends Anal Chem 29:617–628. https://doi.org/10.1016/j.trac.2010.02.016
Farajzadeh MA, Nasrollahpour H, Mogaddam MRA, Khoshmaram L (2016) Determination of widely used non-steroidal anti-inflammatory drugs in biological fluids using simultaneous derivatization and air-assisted liquid–liquid microextraction followed by gas chromatography–flame ionization detection. J Iran Chem Soc 13:289–298. https://doi.org/10.1007/s13738-015-0736-3
Barfi B, Asghari A, Rajabi M, Goochani Moghadam A, Mirkhani N, Ahmadi F (2015) Comparison of ultrasound-enhanced air-assisted liquid–liquid microextraction and low-density solvent-based dispersive liquid–liquid microextraction methods for determination of nonsteroidal anti-inflammatory drugs in human urine samples. J Pharm Biomed Anal 111:297–305. https://doi.org/10.1016/j.jpba.2015.03.034
El-Deen AK, Shimizu K (2020) A green air assisted-dispersive liquid–liquid microextraction based on solidification of a novel low viscous ternary deep eutectic solvent for the enrichment of endocrine disrupting compounds from water. J Chromatogr A 1629:461498. https://doi.org/10.1016/j.chroma.2020.461498
Ge D, Zhang Y, Dai Y, Yang S (2018) Air-assisted dispersive liquid–liquid microextraction based on a new hydrophobic deep eutectic solvent for the preconcentration of benzophenone-type UV filters from aqueous samples. J Sep Sci 41:1635–1643. https://doi.org/10.1002/jssc.201701282
Jia L, Yang J, Zhao W, Jing X (2019) Microextraction based on solidification of the aqueous phase for the determination of triazole fungicides in water samples by high-performance. RSC Adv 9(63):36664–36669. https://doi.org/10.1039/c9ra07348e
Chaikhan P, Udnan Y, Ampiah-bonney RJ, Chuachuad W (2021) Air-assisted solvent terminated dispersive liquid–liquid microextraction (AA-ST-DLLME ) for the determination of lead in water and beverage samples by graphite furnace atomic absorption spectrometry. Microchem J 162:105828. https://doi.org/10.1016/j.microc.2020.105828
Rahmani M, Ghasemi E, Sasani M (2016) Application of response surface methodology for air assisted-dispersive liquid–liquid microextraction of deoxynivalenol in rice samples prior to HPLC—DAD analysis and comparison with solid phase extraction cleanup. Talanta 165:27–32. https://doi.org/10.1016/j.talanta.2016.12.031
Teglia CM, Gonzalo L, Culzoni MJ, Goicoechea HC (2017) Determination of six veterinary pharmaceuticals in egg by liquid chromatography: chemometric optimization of a novel air assisted-dispersive liquid–liquid microextraction by solid floating organic drop Abbreviated running title: Determination of six vet. Food Chem 273:194–202. https://doi.org/10.1016/j.foodchem.2017.08.034
Karami-osboo R, Maham M (2018) Pre-concentration and extraction of aflatoxins from rice using air-assisted dispersive liquid–liquid microextraction. Food Anal Methods 11(10):2816–2821
Ferrone AV, Cotellese R, Di ML, Carlucci G (2017) Air assisted dispersive liquid–liquid microextraction with solidification of the floating organic droplets (AA-DLLME-SFO) and UHPLC-PDA method: Application to antibiotics analysis in human plasma of hospital acquired pneumonia patients. J Pharm Biomed Anal 151:266–273. https://doi.org/10.1016/j.jpba.2017.12.039
Shishov A, Trufanov I, Nechaeva D, Bulatov A (2019) A reversed-phase air-assisted dispersive liquid–liquid microextraction coupled with colorimetric paper-based analytical device for the determination of glycerol, calcium and magnesium in biodiesel samples. Microchem J 150:104134. https://doi.org/10.1016/j.microc.2019.104134
Rahmani M, Kaykhaii M, Safari Z, Ansari O (2017) Comparison of air—assisted, vortex—assisted and ultrasound—assisted dispersive liquid–liquid microextraction for the determination of BTEX compounds in water samples prior to GC–FID analysis. Chromatographia 80:109–117. https://doi.org/10.1007/s10337-016-3216-8
Kocúrová L, Balogh IS, Šandrejová J, Andruch V (2012) Recent advances in dispersive liquid–liquid microextraction using organic solvents lighter than water: a review. Microchem J 102:11–17. https://doi.org/10.1016/j.microc.2011.12.002
Ghambari H, Hadjmohammadi M (2012) Low-density solvent-based dispersive liquid–liquid microextraction followed by high performance liquid chromatography for determination of warfarin in human plasma. J Chromatogr B 899:66–71. https://doi.org/10.1016/j.jchromb.2012.04.035
Meng L, Zhang W, Meng P, Zhu B, Zheng K (2015) Comparison of hollow fiber liquid-phase microextraction and ultrasound-assisted low-density solvent dispersive liquid–liquid microextraction for the determination of drugs of abuse in biological samples by gas chromatography–mass spectrometry. J Chromatogr B 989:46–53. https://doi.org/10.1016/j.jchromb.2015.02.039
Mansour FR, Danielson ND (2017) Solidification of floating organic droplet in dispersive liquid–liquid microextraction as a green analytical tool. Talanta 170:22–35. https://doi.org/10.1016/j.talanta.2017.03.084
Tolcha T, Merdassa Y, Megersa N (2013) Low-density extraction solvent based solvent-terminated dispersive liquid–liquid microextraction for quantitative determination of ionizable pesticides in environmental waters. J Sep Sci 36:1119–1127. https://doi.org/10.1002/jssc.201200849
Carda-broch S, Berthod A, Angel MJR (2008) Ionic liquids in separation techniques. J Chromatogr A 1184:6–18. https://doi.org/10.1016/j.chroma.2007.11.109
Nerín C, Salafranca J, Aznar M, Batlle R (2009) Critical review on recent developments in solventless techniques for extraction of analytes. Anal Bioanal Chem 393:809–833. https://doi.org/10.1007/s00216-008-2437-6
Sun P, Armstrong DW (2010) Analytica Chimica Acta Ionic liquids in analytical chemistry. Analytica Chimica Acta 661:1–16. https://doi.org/10.1016/j.aca.2009.12.007
Liu R, Liu J, Yin Y, Hu X (2009) Ionic liquids in sample preparation. Anal Bioanal Chem 393:871–883. https://doi.org/10.1007/s00216-008-2445-6
Danielson ND, Mansour FR, Zhou L, Connell CV, Dotlich EM, Gibler JN, Norman BE, Grossman S, Wei W, Zhang Y (2018) Liquid chromatography with alkylammonium formate ionic liquid mobile phases and fluorescence detection. J Chromatogr A 1559:128–135. https://doi.org/10.1016/j.chroma.2018.03.020
Borrull F, Marce RM (2012) Ionic liquids in solid-phase extraction. Trends Anal Chem 41:15–26. https://doi.org/10.1016/j.trac.2012.08.010
Rykowska I, Ziemblińska J, Nowak I (2018) Modern approaches in dispersive liquid–liquid microextraction (DLLME) based on ionic liquids: a review. J Mol Liq 259:319–339. https://doi.org/10.1016/j.molliq.2018.03.043
Abdelaziz MA, Saleh AM, Mansour FR, Danielson ND (2022) A gadolinium-based magnetic ionic liquid for dispersive liquid–liquid microextraction of ivermectin from environmental water. J Chromatogr Sci. https://doi.org/10.1093/chromsci/bmac101
Kokosa JM (2013) Advances in solvent—microextraction techniques. Trends Anal Chem 43:2–13. https://doi.org/10.1016/j.trac.2012.09.020
Plechkova NV, Seddon KR (2008) Applications of ionic liquids in the chemical industry. Chem Soc Rev 37(1):123–150. https://doi.org/10.1039/b006677j
Zhou Q, Bai H, Xie G, Xiao J (2008) Trace determination of organophosphorus pesticides in environmental samples by temperature-controlled ionic liquid dispersive liquid-phase microextraction. J Chromatogr A 1188:148–153. https://doi.org/10.1016/j.chroma.2008.02.094
Baghdadi M, Shemirani F (2008) Cold-induced aggregation microextraction: a novel sample preparation technique based on ionic liquids. Anal Chim Acta 613:56–63. https://doi.org/10.1016/j.aca.2008.02.057
Liu Y, Zhao E, Zhu W, Gao H, Zhou Z (2009) Determination of four heterocyclic insecticides by ionic liquid dispersive liquid–liquid microextraction in water samples. J Chromatogr A 1216:885–891. https://doi.org/10.1016/j.chroma.2008.11.076
Soylak M (2016) Latest trends, green aspects, and innovations in liquid-phase—Based microextraction techniques: a review. Turkish J Chem 40. https://doi.org/10.3906/kim-1605-26
Baghdadi M, Shemirani F (2009) In situ solvent formation microextraction based on ionic liquids: a novel sample preparation technique for determination of inorganic species in saline solutions. Anal Chim Acta 634:186–191. https://doi.org/10.1016/j.aca.2008.12.017
Yao C, Anderson JL (2009) Dispersive liquid–liquid microextraction using an in situ metathesis reaction to form an ionic liquid extraction phase for the preconcentration of aromatic compounds from water. Anal Bioanal Chem 395:1491–1502. https://doi.org/10.1007/s00216-009-3078-0
Del Sesto RE, McCleskey TM, Burrell AK, Baker GA, Thompson JD, Scott BL, Wilkes JS, Williams P (2002) Structure and magnetic behavior of transition metal based ionic liquids. Chem Commun 8:447–449. https://doi.org/10.1039/b711189d
Rosatella AA, Siopa F, Frade RFM, Afonso CAM (2016) New low viscous cholinium-based magnetic ionic liquids. New J Chem 40:3124–3129. https://doi.org/10.1039/C5NJ03165F
Santos E, Albo J, Rosatella A, Afonso CAM, Irabien Á (2014) Synthesis and characterization of Magnetic Ionic Liquids (MILs) for CO2 separation. J Chem Technol Biotechnol 89:866–871. https://doi.org/10.1002/jctb.4323
Abdelaziz MA, Mansour FR, Danielson ND (2021) A gadolinium-based magnetic ionic liquid for dispersive liquid–liquid microextraction. Anal Bioanal Chem 413:205–214. https://doi.org/10.1007/s00216-020-02992-z
Mousavi L, Tamiji Z, Khoshayand MR (2018) Applications and opportunities of experimental design for the dispersive liquid–liquid microextraction method—a review. Talanta 190:335–356. https://doi.org/10.1016/j.talanta.2018.08.002
Sajid M, Alhooshani K (2018) Dispersive liquid–liquid microextraction based binary extraction techniques prior to chromatographic analysis: a review. TrAC—Trends Anal Chem 108:167–182. https://doi.org/10.1016/j.trac.2018.08.016
Paquin F, Rivnay J, Salleo A, Stingelin N, Silva C (2015) Multi-phase semicrystalline microstructures drive exciton dissociation in neat plastic semiconductors. J Mater Chem C 3:10715–10722. https://doi.org/10.1039/b000000x
Cvjetko Bubalo M, Ćurko N, Tomašević M, Kovačević Ganić K, Radojcic Redovnikovic I (2016) Green extraction of grape skin phenolics by using deep eutectic solvents. Food Chem 200:159–166. https://doi.org/10.1016/j.foodchem.2016.01.040
Khandelwal S, Tailor YK, Kumar M (2016) Deep eutectic solvents (DESs) as eco-friendly and sustainable solvent/catalyst systems in organic transformations. J Mol Liq 215:345–386. https://doi.org/10.1016/j.molliq.2015.12.015
Florindo C, Branco LC, Marrucho IM (2017) Development of hydrophobic deep eutectic solvents for extraction of pesticides from aqueous environments. Fluid Phase Equilib 448:135–142. https://doi.org/10.1016/j.fluid.2017.04.002
Duan L, Dou LL, Guo L, Li P, Liu EH (2016) Comprehensive evaluation of deep eutectic solvents in extraction of bioactive natural products. ACS Sustain Chem Eng 4:2405–2411. https://doi.org/10.1021/acssuschemeng.6b00091
Zeng Q, Wang Y, Huang Y, Ding X, Chen J, Xu K (2014) Deep eutectic solvents as novel extraction media for protein partitioning. Analyst 139:2565–2573. https://doi.org/10.1039/c3an02235h
Nam MW, Zhao J, Lee MS, Jeong JH, Lee J (2015) Enhanced extraction of bioactive natural products using tailor-made deep eutectic solvents: application to flavonoid extraction from Flos sophorae. Green Chem 17:1718–1727. https://doi.org/10.1039/c4gc01556h
Lasarte-Aragonés G, Lucena R, Cárdenas S, Valcárcel M (2015) Use of switchable hydrophilicity solvents for the homogeneous liquid–liquid microextraction of triazine herbicides from environmental water samples. J Sep Sci 38:990–995. https://doi.org/10.1002/jssc.201401224
Hamad A, Elshahawy M, Negm A, Mansour FR (2020) Analytical methods for determination of glutathione and glutathione disulfide in pharmaceuticals and biological fluids. Rev Anal Chem 38:20190019. https://doi.org/10.1515/revac-2019-0019
Mansour FR, Danielson ND (2012) Separation methods for captopril in pharmaceuticals and biological fluids. J Sep Sci 35:1213–1226. https://doi.org/10.1002/jssc.201200057
Mansour FR, Wei W, Danielson ND (2013) Separation of carnitine and acylcarnitines in biological samples: a review. Biomed Chromatogr 27:1339–1353. https://doi.org/10.1002/bmc.2995
Ballesteros-Gómez A, Rubio S, Pérez-Bendito D (2009) Potential of supramolecular solvents for the extraction of contaminants in liquid foods. J Chromatogr A 1216:530–539. https://doi.org/10.1016/j.chroma.2008.06.029
Carabias-Martínez R, Rodríguez-Gonzalo E, Moreno-Cordero B, Pérez-Pavón JL, García-Pinto C, Fernández Laespada E (2000) Surfactant cloud point extraction and preconcentration of organic compounds prior to chromatography and capillary electrophoresis. J Chromatogr A 902:251–265. https://doi.org/10.1016/S0021-9673(00)00837-2
Cantero M, Rubio S, Pérez-Bendito D (2004) Determination of non-ionic polyethoxylated surfactants in sewage sludge by coacervative extraction and ion trap liquid chromatography-mass spectrometry. J Chromatogr A 1046:147–153. https://doi.org/10.1016/j.chroma.2004.06.073
Menger FM, Sykes BM (1998) Anatomy of a coacervate. Langmuir 14:4131–4137. https://doi.org/10.1021/la980208m
Seidi S, Alavi L, Jabbari A (2018) Trace determination of cadmium in rice samples using solidified floating organic drop microextraction based on vesicular supramolecular solvent followed by flow-injection analysis–flame atomic absorption spectrometry. J Iran Chem Soc 15:2083–2092. https://doi.org/10.1007/s13738-018-1401-4
Ruiz FJ, Rubio S, Pérez-Bendito D (2007) Water-induced coacervation of alkyl carboxylic acid reverse micelles: Phenomenon description and potential for the extraction of organic compounds. Anal Chem 79:7473–7484. https://doi.org/10.1021/ac0708644
García-Prieto A, Lunar L, Rubio S, Pérez-Bendito D (2008) Decanoic acid reverse micelle-based coacervates for the microextraction of bisphenol A from canned vegetables and fruits. Anal Chim Acta 617:51–58. https://doi.org/10.1016/j.aca.2008.01.061
Tayyebi M, Yamini Y, Moradi M (2012) Reverse micelle-mediated dispersive liquid–liquid microextraction of 2,4-dichlorophenoxyacetic acid and 4-chloro-2-methylphenoxyacetic acid. J Sep Sci 35:2491–2498. https://doi.org/10.1002/jssc.201200150
Ballesteros-Gómez A, Sicilia MD, Rubio S (2010) Supramolecular solvents in the extraction of organic compounds: a review. Anal Chim Acta 677:108–130. https://doi.org/10.1016/j.aca.2010.07.027
Mabrouk MM, Hamed NA, Mansour FR (2022) Physicochemical and electrochemical methods for determination of critical micelle concentrations of surfactants: a comprehensive review. Monatshefte für Chemie—Chem Mon 153:125–138. https://doi.org/10.1007/s00706-022-02891-2
Mabrouk MM, Hamed NA, Mansour FR (2023) Spectroscopic methods for determination of critical micelle concentrations of surfactants; a comprehensive review. Appl Spectrosc Rev 58:206–234. https://doi.org/10.1080/05704928.2021.1955702
Viñas P, Campillo N, Andruch V (2015) Recent achievements in solidified floating organic drop microextraction. Trends Anal Chem 68:48–77. https://doi.org/10.1016/j.trac.2015.02.005
Farhadi K, Hatami M, Forough M, Molaei R (2013) Dispersive liquid–liquid microextraction of propranolol enantiomers from human plasma based on the solidification of a floating organic droplet. Bioanalysis 5:701–710. https://doi.org/10.4155/bio.13.23
Suh JH, Lee YY, Lee HJ, Kang M, Hur Y, Lee SN, Yang D-H, Han SB (2013) Dispersive liquid–liquid microextraction based on solidification of floating organic droplets followed by high performance liquid chromatography for the determination of duloxetine in human plasma. J Pharm Biomed Anal 75:214–219. https://doi.org/10.1016/j.jpba.2012.11.041
Jia S, Li J, Park S-R, Ryu Y, Park IH, Park JH, Hong S-S, Kwon SW, Lee J (2013) Combined application of dispersive liquid–liquid microextraction based on the solidification of floating organic droplets and charged aerosol detection for the simple and sensitive quantification of macrolide antibiotics in human urine. J Pharm Biomed Anal 86:204–213. https://doi.org/10.1016/j.jpba.2013.07.024
Taheri S, Jalali F, Fattahi N, Bahrami G (2015) Sensitive determination of atorvastatin in human plasma by dispersive liquid–liquid microextraction and solidification of floating organic drop followed by high-performance liquid chromatography. J Sep Sci 38:309–315. https://doi.org/10.1002/jssc.201401115
Zhao L, Zhao P, Wang L, Ma X, Hou X, Li F (2014) A dispersive liquid–liquid microextraction method based on the solidification of a floating organic drop combined with HPLC for the determination of lovastatin and simvastatin in rat urine. Biomed Chromatogr 28:895–900. https://doi.org/10.1002/bmc.3205
Shukri DSM, Sanagi MM, Ibrahim WAW, Abidin NNZ, Aboul-Enein HY (2015) Liquid chromatographic determination of NSAIDs in urine after dispersive liquid–liquid microextraction based on solidification of floating organic droplets. Chromatographia 78:987–994. https://doi.org/10.1007/s10337-015-2920-0
Pebdani AA, Shabani AMH, Dadfarnia S, Talebianpoor MS, Khodadoust S (2016) Preconcentration of valsartan by dispersive liquid–liquid microextraction based on solidification of floating organic drop and its determination in urine sample: central composite design. J Sep Sci 39:1935–1944. https://doi.org/10.1002/jssc.201600010
Zamani-Kalajahi M, Fazeli-Bakhtiyari R, Amiri M, Golmohammadi A, Afrasiabi A, Khoubnasabjafari M, Jouyban A (2013) Dispersive liquid–liquid microextraction based on solidification of floating organic droplet followed by spectrofluorimetry for determination of carvedilol in human plasma. Bioanalysis 5:437–448. https://doi.org/10.4155/bio.12.326
Yamini Y, Rezaee M, Khanchi A, Faraji M, Saleh A (2010) Dispersive liquid–liquid microextraction based on the solidification of floating organic drop followed by inductively coupled plasma-optical emission spectrometry as a fast technique for the simultaneous determination of heavy metals. J Chromatogr A 1217:2358–2364. https://doi.org/10.1016/j.chroma.2009.11.046
Ahmadi-Jouibari T, Fattahi N, Shamsipur M, Pirsaheb M (2013) Dispersive liquid–liquid microextraction followed by high-performance liquid chromatography-ultraviolet detection to determination of opium alkaloids in human plasma. J Pharm Biomed Anal 85:14–20. https://doi.org/10.1016/j.jpba.2013.06.030
Taheri S, Jalali F, Fattahi N, Jalili R, Bahrami G (2015) Sensitive determination of methadone in human serum and urine by dispersive liquid–liquid microextraction based on the solidification of a floating organic droplet followed by HPLC-UV. J Sep Sci 38:3545–3551. https://doi.org/10.1002/jssc.201500636
Akramipour R, Fattahi N, Pirsaheb M, Gheini S (2016) Combination of counter current salting-out homogenous liquid–liquid extraction and dispersive liquid–liquid microextraction as a novel microextraction of drugs in urine samples. J Chromatogr B 1012–1013:162–168. https://doi.org/10.1016/j.jchromb.2016.01.031
Asadi M, Dadfarnia S, Haji Shabani AM (2016) Simultaneous extraction and determination of albendazole and triclabendazole by a novel syringe to syringe dispersive liquid phase microextraction-solidified floating organic drop combined with high performance liquid chromatography. Anal Chim Acta 932:22–28. https://doi.org/10.1016/j.aca.2016.05.014
Hemmati M, Rajabi M, Asghari A (2017) Efficient and clean pre-concentration of ultra-trace calcium channel blockers from biological matrices via a hyphenated procedure of two sequential dispersive solid/liquid phase microextractions. Anal Chim Acta 960:138–150. https://doi.org/10.1016/j.aca.2017.01.001
Guo L, Lee HK (2014) Automated dispersive liquid−liquid microextraction−gas chromatography−mass spectrometry. Anal Chem
Silvestre CIC, Santos JLM, Lima JLFC, Zagatto EAG (2009) Liquid–liquid extraction in flow analysis: a critical review. Anal Chim Acta 652:54–65. https://doi.org/10.1016/j.aca.2009.05.042
Lenehan CE, Barnett NW, Lewis SW (2002) Sequential injection analysis. Analyst 127:997–1020. https://doi.org/10.1039/b106791p
Anthemidis AN, Ioannou K-IG (2009) On-line sequential injection dispersive liquid–liquid microextraction system for flame atomic absorption spectrometric determination of copper and lead in water samples. Talanta 79:86–91. https://doi.org/10.1016/j.talanta.2009.03.005
Anthemidis AN, Ioannou KIG (2010) Development of a sequential injection dispersive liquid–liquid microextraction system for electrothermal atomic absorption spectrometry by using a hydrophobic sorbent material: Determination of lead and cadmium in natural waters. Anal Chim Acta 668:35–40. https://doi.org/10.1016/j.aca.2009.10.063
Anthemidis AN, Ioannou KIG (2011) Sequential injection dispersive liquid–liquid microextraction based on fatty alcohols and poly(etheretherketone)-turnings for metal determination by flame atomic absorption spectrometry. Talanta 84:1215–1220. https://doi.org/10.1016/j.talanta.2010.12.017
Berton P, Martinis EM, Wuilloud RG (2010) Development of an on-line temperature-assisted ionic liquid dispersive microextraction system for sensitive determination of vanadium in environmental and biological samples. J Hazard Mater 176:721–728. https://doi.org/10.1016/J.JHAZMAT.2009.11.094
Berton P, Wuilloud RG (2011) An online ionic liquid-based microextraction system coupled to electrothermal atomic absorption spectrometry for cobalt determination in environmental samples and pharmaceutical formulations. Anal Methods 3:664–672. https://doi.org/10.1039/c0ay00616e
Martinis EM, Escudero LB, Berton P, Monasterio RP, Filippini MF, Wuilloud RG (2011) Determination of inorganic selenium species in water and garlic samples with on-line ionic liquid dispersive microextraction and electrothermal atomic absorption spectrometry. Talanta 85:2182–2188. https://doi.org/10.1016/J.TALANTA.2011.07.065
Andruch V, Acebal CC, Škrlíková J, Sklenářová H, Solich P, Balogh IS, Billes F, Kocúrová L (2012) Automated on-line dispersive liquid–liquid microextraction based on a sequential injection system. Microchem J 100:77–82. https://doi.org/10.1016/j.microc.2011.09.006
Ruzicka J, Marshall GD (1990) Sequential injection: a new concept for chemical sensors, process analysis and laboratory assays. Anal Chim Acta 237:329–343. https://doi.org/10.1016/S0003-2670(00)83937-9
Medina DAV, Santos-Neto ÁJ, Cerdà V, Maya F (2018) Automated dispersive liquid–liquid microextraction based on the solidification of the organic phase. Talanta 189:241–248. https://doi.org/10.1016/J.TALANTA.2018.06.081
Shishov A, Terno P, Moskvin L, Bulatov A (2020) In-syringe dispersive liquid–liquid microextraction using deep eutectic solvent as disperser: Determination of chromium (VI) in beverages. Talanta 206:120209. https://doi.org/10.1016/j.talanta.2019.120209
Mabrouk MM, Soliman SM, El-Agizy HM, Mansour FR (2020) Ultrasound-assisted dispersive liquid–liquid microextraction for determination of three gliflozins in human plasma by HPLC/DAD. J Chromatogr B 1136:121932. https://doi.org/10.1016/j.jchromb.2019.121932
Akramipour R, Fattahi N, Golpayegani MR (2021) Sensitive determination of methotrexate in plasma of children with acute leukemia using double-solvent supramolecular systemas a novel extractant for dispersive liquid–liquid microextraction. J Chromatogr B Anal Technol Biomed Life Sci 1171:122628. https://doi.org/10.1016/j.jchromb.2021.122628
Abdallah IA, Hammad SF, Bedair A, Abdelaziz MA, Danielson ND, Elshafeey AH, Mansour FR (2022) A gadolinium-based magnetic ionic liquid for supramolecular dispersive liquid–liquid microextraction followed by HPLC/UV for the determination of favipiravir in human plasma. Biomed Chromatogr 36:1–10. https://doi.org/10.1002/bmc.5365
Hammad MA, Kamal AH, Kannouma RE, Mansour FR (2021) Vortex-assisted dispersive liquid–liquid microextraction coupled with deproteinization for determination of Nateglinide in human plasma using HPLC/UV. J Chromatogr Sci 59:297–304. https://doi.org/10.1093/chromsci/bmaa096
Qiao L, Sun R, Yu C, Tao Y, Yan Y (2021) Novel hydrophobic deep eutectic solvents for ultrasound-assisted dispersive liquid–liquid microextraction of trace non-steroidal anti-inflammatory drugs in water and milk samples. Microchem J 170:106686. https://doi.org/10.1016/j.microc.2021.106686
Faraji M, Noormohammadi F, Adeli M (2020) Preparation of a ternary deep eutectic solvent as extraction solvent for dispersive liquid–liquid microextraction of nitrophenols in water samples. J Environ Chem Eng 8:103948. https://doi.org/10.1016/j.jece.2020.103948
Moradi M, Zarabi S, Heydari R (2021) Spectrophotometric determination of trace amounts of Sb(III) and Sb(V) in water and biological samples by in-tube dispersive liquid–liquid microextraction and air-assisted liquid–liquid microextraction. Chem Pap 75:6499–6508. https://doi.org/10.1007/s11696-021-01818-1
Kachangoon R, Vichapong J, Santaladchaiyakit Y, Burakham R, Srijaranai S (2020) An eco-friendly hydrophobic deep eutectic solvent-based dispersive liquid–liquid microextraction for the determination of neonicotinoid insecticide residues in water, soil and egg yolk samples. Molecules 25(12):2785. https://doi.org/10.3390/molecules25122785
Dias RAS, Sousa ER, Silva GS, Silva LK, Freitas AS, Lima DLD, Sousa ÉML (2021) Ultrasound-assisted dispersive liquid–liquid microextraction for determination of enrofloxacin in surface waters. Microchem J 160:105633. https://doi.org/10.1016/j.microc.2020.105633
Wang Y, Li J, Ji L, Chen L (2022) Simultaneous determination of sulfonamides antibiotics in environmental water and seafood samples using ultrasonic-assisted dispersive liquid–liquid microextraction coupled with high performance liquid chromatography. Molecules 27(7):2160. https://doi.org/10.3390/molecules27072160
Tuzen M, Elik A, Altunay N (2021) Ultrasound-assisted supramolecular solvent dispersive liquid–liquid microextraction for preconcentration and determination of Cr(VI) in waters and total chromium in beverages and vegetables. J Mol Liq 329:115556. https://doi.org/10.1016/j.molliq.2021.115556
Lian X, Wang N, Ma L, Jiang H, Bai D, Xue H, Ma Q (2020) Determination of aucubin by supramolecular solvent-based dispersive liquid–liquid microextraction and UPLC-MS/MS: application to a pharmacokinetic study in rats with type 1 diabetes. J Pharm Biomed Anal 186:113301. https://doi.org/10.1016/j.jpba.2020.113301
ALOthman ZA, Yilmaz E, Habila MA, Alhenaki B, Soylak M, Ahmed AYBH, Alabdullkarem EA (2022) Development of combined-supramolecular microextraction with ultra-performance liquid chromatography-tandem mass spectrometry procedures for ultra-trace analysis of carbaryl in water, fruits and vegetables. Int J Environ Anal Chem 102:1491–1501. https://doi.org/10.1080/03067319.2020.1738419
de Oliveira FM, Scheel GL, Augusti R, Tarley CRT, Nascentes CC (2020) Supramolecular microextraction combined with paper spray ionization mass spectrometry for sensitive determination of tricyclic antidepressants in urine. Anal Chim Acta 1106:52–60. https://doi.org/10.1016/j.aca.2020.01.061
Soylak M, Agirbas M, Yilmaz E (2021) A new strategy for the combination of supramolecular liquid phase microextraction and UV–Vis spectrophotometric determination for traces of maneb in food and water samples. Food Chem 338:128068. https://doi.org/10.1016/j.foodchem.2020.128068
Aydin F, Soylak M (2015) RSC Advances liquid microextraction of copper from water and hair samples. RSC Adv 5:40422–40428. https://doi.org/10.1039/C4RA17116K
Tao Y, Jia L, Qin H, Niu R, Qiao L (2022) A new magnetic ionic liquid based salting-out assisted dispersive liquid–liquid microextraction for the determination of parabens in environmental water samples. Anal Methods 14:4775–4783. https://doi.org/10.1039/d2ay01403c
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Bedair, A., Mansour, F.R. (2024). Dispersive Liquid–Liquid Microextraction. In: Rodríguez-Delgado, M.Á., Socas-Rodríguez, B., Herrera-Herrera, A.V. (eds) Microextraction Techniques. Integrated Analytical Systems. Springer, Cham. https://doi.org/10.1007/978-3-031-50527-0_9
Download citation
DOI: https://doi.org/10.1007/978-3-031-50527-0_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-50526-3
Online ISBN: 978-3-031-50527-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)