Abstract
The supraorbital-pterional approach was first proposed in 1987 by Al-Mefty for large tumors extending in both intracranial and intraorbital compartments. It conjugates a pterional approach with the removal of the lateral orbital wall and roof, thus allowing large exposure of the orbital content. The skull base structures, including anterior clinoid, optic canal and strut, superior orbital fissure, may be exposed by extradural route and resected by microsurgical technique and by using a diamond high-speed drill. The orbital apex may be exposed by extending posteriorly the resection of the orbital roof. The damage of the optic and oculomotor nerves during the bone drilling, significant periorbital ecchymosis and cerebrospinal fluid (CSF) leak are the most significant complications. The advantages and limits of the supraorbital-pterional approach versus other microsurgical cranio-orbital and orbital ones and versus endoscopic transorbital and endonasal approaches are discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The approach to the cranio-orbital lesions is evolved over the years with the aims to obtain better control of the intraorbital structures and lesser brain retraction.
In the first years of the past century, some neurosurgeons [1, 2] proposed resecting the supraorbital arch in the frontal approach. In 1982, Jane et al. [3] reviewed this approach and considered it the best supraorbital route for orbital tumors. Al-Mefty and Fox [4] suggested a supero-lateral approach and reconstruction to improve the orbital exposure. The pterional approach, first described by Heuer [5], was refined by Yasargil [6], who suggested drilling of the sphenoid ridge and occasional removal of part of the orbital ridge and roof.
All the above cited approaches are not adequate for large tumors extending in both intracranial and intraorbital compartments. To this aim, Al-Mefty [7] proposed the supraorbital pterional approach, which conjugates the pterional approach with removal of the lateral orbital wall and roof.
2 Surgical Technique
2.1 Patient Positioning and Skin Flap
The patient is positioned in the operating table with the head fixed at the Mayfield head holder and rotated 30° toward the contralateral side. The neck is slightly extended, allowing spontaneous retraction of the frontal lobe.
The scalp incision (Fig. 6.1) begins at level of the zygomatic arch, 1 cm anterior to the tragus, then it proceeds up to the superior temporal line and curves anterior and medially, up to the midline, about 2 cm posterior to the hairline. The temporal muscle flap is detached from the temporal fossa and the lateral orbital wall is exposed; a small muscle margin is left attached to the bone to be used for the muscle closure. During the scalp dissection, the periosteum and galea must be preserved with the aim of being used over an eventual defect of the frontal sinus. The frontal and supraorbital nerves must be preserved to avoid paralysis of the frontal muscle and anesthesia in the forehead.
2.2 Craniotomy
The craniotomy (Figs. 6.1 and 6.2) is performed by using 2 or 3 burr holes. A craniotome is used to create a fronto-temporal bone flap centered on the sphenoid wing that exposes the anterior and middle cranial fossa floors and temporal tip. Through a burr hole over the frontal sinus an osteotomy is made posterior to the orbital rime to detach the orbital roof. In this way, when the bone flap is elevated, the orbital roof remains attached to the flap itself. During the flap elevation, careful dissection of the dura mater and periorbita is necessary to avoid interruption. The superior orbital rime remains intact. When tumor invasion of the orbital roof and rime is evidenced, these structures may be resected using a rongeur. If the frontal sinus has been opened, the mucosa must be exenterated and the sinus must be filled with antibiotics and occluded by cottonoids.
2.3 Management of the Skull Base Structures
The skull base structures, including orbital apex, optic canal, optic strut, anterior clinoid and superior orbital fissure, must be managed by microsurgical technique and the use of a high-speed diamond drill, and by extradural route. The orbital apex may be exposed by extending posteriorly the resection of the orbital roof. The optic canal must be opened by drilling its wall with a diamond drill during continuous washing, to avoid excessive heating. No attempt must be made to use rongeurs in the optic canal, because this maneuver may result in optic nerve damage.
The supraorbital pterional approach allows good exposure and opening of the superior and lateral walls of the optic canal and the optic strut; on the other hand, the inferomedial wall is less controlled [8, 9]. Thus, the circumferential decompression of the optic canal may be accomplished with an anterior clinoidectomy and removal of the optic strut.
The resection of the skull base structures depends on the direction of tumor growth and orbital location. If the tumor is in the apex or in the optic canal, the entire orbital roof must be removed, and the optic canal must adequately be opened. If the tumor enters to the orbit through its lateral wall without involvement of the optic canal, it may not be opened [8,9,10,11].
2.4 Incision of the Dura Mater and Periorbita
The incision of the dura mater and periorbita must follow rigorous criteria to obtain good exposure and to allow watertight closure. The dura mater is opened in a semicircular fashion, then the dural flap is bisected by a second dural incision that points toward the optic canal. The dural incision along the optic canal must be made by leaving a cuff of the dura around the optic nerve for an adequate closure [12]. The incision of the anulus of Zinn and the periorbita must be made medial to the levator and superior rectus muscles to avoid injury to the oculomotor nerve.
2.5 Reconstruction
The closure of the dura mater must be watertight also over the optic canal, when preserved. The frontal sinus must be obliterated by muscle tissue and covered by a galeal flap mobilized and sutured to the dura mater and reinforced by fibrin glue.
We do not reconstruct the orbital roof in our cases. This usually does not result in cosmetic problems. The bone flap is replaced and fixed by wires and the temporal muscle is sutured to the small muscle margin left attached to the bone. Then, the skin is closed in layers.
Postoperatively, moderate periorbital ecchymosis and slight-to-moderate swelling occur for several days.
3 Complications
Complications of the supraorbital pterional approach may be observed during both intraoperative and post-operative periods.
Intraoperative complications may occur in all phases of the approach. During the turning of the skin flap, the frontal and supraorbital nerves may be injured, resulting in frontal nerve palsy or decreased sensation of the forehead, respectively; it may be avoided by carefully dissecting the nerves from the subcutaneous tissues and supraorbital notch.
At the elevation of the bone flap, the frontal sinus may accidentally be opened; this often results in post-operative cerebrospinal fluid (CSF) rhinorrhea. In such event, the careful closure of the defect by periosteum and galea flaps must be achieved.
Injuries to the cranial nerves may occur during the bone resection at the skull base and/or at the entry into the orbit. The injury to the optic nerve may be due to excessive retraction, particularly in presence of adherent tumors, to the occlusion of the central retinal artery, and particularly to heat transmission during drilling of the optic nerve canal. This complication is serious and not an exception (5% in the series of Maroon et al. [13]). It may be avoided by careful microsurgical drilling using a high-speed drill and a diamond bur with continuous irrigation. The injury to the oculomotor nerves may occur at the superior orbital fissure, when opened, or within the orbit. This complication may be prevented by avoiding undue retraction of levator and superior rectus muscles and by approaching the intraorbital tumor component medially, between the superior rectus and levator and medial rectus muscles.
Intraoperative bleeding from the internal carotid and ophthalmic arteries, although very rare, may occur mainly at the reoperations for tumor recurrence.
Postoperative complications include pulsating proptosis, enophthalmos, hematoma, CSF rhinorrhea, and meningitis.
Pulsating proptosis is rare and may be due to cerebral edema and CSF; remission usually occurs within 1–2 months. Enophthalmos may occur after resection of large tumors with significant residual cavity, but it is not significant. In exceptional cases, where the cosmetic problem is severe, the reoperation for orbital repair may be evaluated.
Postoperative intraorbital hematoma is rare and results from insufficient hemostasis of the surgical field. As already discussed, moderate periorbital ecchymosis lacks pathological significance, while severe chemosis should suggest a hematoma. The evacuation of the blood collection is often required to prevent eye damage.
The CSF rhinorrhea may occur because of accidental opening of the frontal and/or ethmoidal sinus or not correct repair of the sinus wall after resection of an invading tumor. Its incidence is about 3%. Most CSF fistulas undergo remission by lumbar puncture and antibiotic therapy, as done for other surgical approaches and sites. Cases where the CSF leak persists with the risk of meningitis must undergo reoperation with careful closure of the communication.
4 Supraorbital Pterional Versus Other Approaches: Advantages and Limits
The choice of the best surgical approach to the cranio-orbital mass lesions depends on several factors, including route of the orbital spread, intraorbital location, and mass size [14,15,16] (Table 6.1).
The supraorbital pterional approach provides good exposure of the lateral, superior, and partly medial orbital compartments, anterior and middle cranial fossa, orbital apex, optic canal, and superior orbital fissure [4, 8,9,10, 17, 18]. Its limits include the need for a large craniotomy, the bad exposure of the infratemporal fossa, and the infero-medial orbital region below the medial side of the optic foramen. Thus, mass lesions with significant infratemporal extension require a frontotemporal-orbitozygomatic approach; on the other hand, mass lesions of the medial intraorbital compartment with significant extension in the ethmoidal sinus may require a medial transorbital approach [15, 19].
Several other approaches may be used for cranio-orbital mass lesions. These include the mini-pterional craniotomy, the frontotemporal-orbitozygomatic approach, the lateral supraorbital, supraorbital key-hole and lateral orbital-cranial approaches, the endoscopic endonasal and transorbital approaches.
The mini-pterional craniotomy [20] is a less invasive modification of the standard pterional craniotomy [18, 21, 22]. However, the smaller craniotomy size may limit the space for frontal and temporal lobe mobilization in larger lesions. Moreover, the exposure at the medial orbital roof and inferolateral orbital wall is significantly more limited. Thus, the mini-pterional approach may be used only for small cranio-orbital lesions with no or limited extension in the medial orbital compartment.
The frontotemporal-orbitozygomatic approach is a frontotemporal approach with orbital roof opening associated to the resection of the zygomatic process [23, 24]. It allows good exposure of the orbital cavity, orbital apex, and middle fossa; the downward extension of the bone resection allows to expose the infratemporal fossa [25].
This approach enhances the surgical exposure and allows less brain retraction. However, it is more invasive and carries the risk of periorbital hematoma and enophthalmos. Thus, in the cranio-orbital surgery, this approach should be limited to more extensive tumors, mainly to those with significant extension into the infratemporal fossa [10].
The supraorbital approach is a variant of the pterional approach with no removal of the temporal bone [3]. It includes a standard pterional skin incision to provide supraorbital frontal bone exposure with minimal disruption of the temporalis muscle [15, 26]. Removal of the orbital roof and superior orbital rime allows to expose the orbital cavity.
The supraorbital approach provides good access to the anterior cranial fossa and superior orbital compartment; on the other hand, the access to the lateral intraorbital compartment is less easy. This approach may be used for cranio-orbital lesions extending in the superior compartment of the orbital region. It also carries the risk of injury to the frontal branch of the facial nerve.
The supraorbital keyhole approach, first introduced in 1998 by Perneczsky [27], is realized through an incision within the eyebrow and a supraorbital enlarged keyhole. The minimal soft-tissue dissection and the small craniotomy size reduce postoperative orbital and frontotemporal swelling [28]. The limited exposure and the risk of frontal sinus contamination are the main disadvantages.
This approach, more limited than the previously cited, may be used for treating orbital intraconal and extraconal lesions superior to the optic nerve.
The lateral orbito-cranial approach [29] includes a lateral orbitotomy and a small craniotomy at the level of the temporal fossa. It allows good exposure of the superolateral intraorbital compartments, the lateral wall of the optic canal, and the anterior temporal region [30, 31].
This approach is useful for cranio-orbital mass lesions located at the lateral compartment of the orbital cavity also when involving the wall of the optic canal [29]. On the other hand, the mass lesions extending medially to the axis of the optic nerve are not sufficiently controlled.
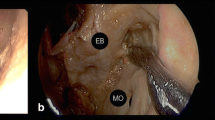
The endoscopic approaches to the orbit include the transorbital, the transsphenoidal endonasal, and the combined endonasal-transorbital approaches. The transorbital approaches [32,33,34] are realized by palpebral incision through medial and/or lateral surgical corridors; they allow to expose the sphenoid wing, the roof of the optic canal, and the superior orbital fissure. The endoscopic endonasal approach provides exposure to the inferomedial orbital region and the inferomedial wall of the optic canal [35,36,37].
Although these approaches are less invasive than the cranio-orbital microsurgical ones, they are often too limited for large cranio-orbital mass lesions.
5 Conclusion
The supraorbital pterional approach is, in our experience, the most useful approach to the cranio-orbital mass lesions of middle and large size. Those located in the lateral orbital compartment may be approached through a less invasive lateral orbito-cranial approach. On the other hand, the indication to the fronto-temporal-orbito-zygomatic approach may be limited to more extensive mass lesions with significant infratemporal extension. The significant tumor extension in the ethmoidal sinus and below the medial wall of the optic canal, although often exposed by the supraorbital pterional approach, may require a medial transorbital approach. The less invasive endoscopic endonasal and transorbital approaches may be used for treating not large mass lesions.
References
McArthur L. An aseptic surgical access to the pituitary body and its neighborhood. JAMA. 1912;58:2009–11.
Frazier CHI. An approach to the hypophysis through the anterior cranial fossa. Ann Surg. 1913;57(2):145–50. https://doi.org/10.1097/00000658-191302000-00001.
Jane JA, Park TS, Pobereskin LH, Winn HR, Butler AB. The supraorbital approach: technical note. Neurosurgery. 1982;11(4):537–42.
Al-Mefty O, Fox JL. Superolateral orbital exposure and reconstruction. Surg Neurol. 1985;23(6):609–13. https://doi.org/10.1016/0090-3019(85)90012-6.
Heuer G. Surgical experience with an intracranial approach to chasmal lesions. Arch Surg. 1920;1:368–81.
Yasargil M, Fox J, Ray M. The operative approach to aneurysms of the anterior communicating artery. In: Advances and technical standards in neurosurgery. Berlin: Springer-Verlag; 1975.
Al-Mefty O. Supraorbital-pterional approach to skull base lesions. Neurosurgery. 1987;21(4):474–7. https://doi.org/10.1227/00006123-198710000-00006.
Mariniello G, de Divitiis O, Bonavolontà G, Maiuri F. Surgical unroofing of the optic canal and visual outcome in basal meningiomas. Acta Neurochir. 2013;155(1):77–84. https://doi.org/10.1007/s00701-012-1485-z.
Mariniello G, Bonavolontà G, Tranfa F, Maiuri F. Management of the optic canal invasion and visual outcome in spheno-orbital meningiomas. Clin Neurol Neurosurg. 2013;115(9):1615–20. https://doi.org/10.1016/j.clineuro.2013.02.012.
Mariniello G, Maiuri F, Strianese D, Donzelli R, Iuliano A, Tranfa F, et al. Spheno-orbital meningiomas: surgical approaches and outcome according to the intraorbital tumor extent. Zentralbl Neurochir. 2008;69(4):175–81. https://doi.org/10.1055/s-2008-1077077.
Mariniello G, de Divitiis O, Corvino S, Strianese D, Iuliano A, Bonavolontà G, et al. Recurrences of spheno-orbital meningiomas: risk factors and management. World Neurosurg. 2022;161:e514. https://doi.org/10.1016/j.wneu.2022.02.048.
Mariniello G, Maiuri F. Letter: mini-pterional craniotomy and extradural clinoidectomy for clinoid meningioma: optimization of exposure using augmented reality template: 2-dimensional operative video. Oper Neurosurg (Hagerstown). 2020;20(1):E75. https://doi.org/10.1093/ons/opaa353.
Maroon JC, Kazim M, Kennerdell JS. Orbital meningiomas and other tumors. In: Brain surgery: complications avoidance and management, vol. 1. London: Churchill Livingstone; 1992.
Paluzzi A, Gardner PA, Fernandez-Miranda JC, Tormenti MJ, Stefko ST, Snyderman CH, et al. “Round-the-clock” surgical access to the orbit. J Neurol Surg B Skull Base. 2015;76(1):12–24. https://doi.org/10.1055/s-0033-1360580.
Abou-Al-Shaar H, Krisht KM, Cohen MA, Abunimer AM, Neil JA, Karsy M, et al. Cranio-orbital and orbitocranial approaches to orbital and intracranial disease: eye-opening approaches for neurosurgeons. Front Surg. 2020;7:1. https://doi.org/10.3389/fsurg.2020.00001.
Abussuud Z, Ahmed S, Paluzzi A. Surgical approaches to the orbit: a neurosurgical perspective. J Neurol Surg B Skull Base. 2020;81(4):385–408. https://doi.org/10.1055/s-0040-1713941.
Andrade-Barazarte H, Jägersberg M, Belkhair S, Tymianski R, Turel MK, Schaller K, et al. The extended lateral supraorbital approach and extradural anterior clinoidectomy through a frontopterio-orbital window: technical note and pilot surgical series. World Neurosurg. 2017;100:159–66. https://doi.org/10.1016/j.wneu.2016.12.087.
Martínez-Pérez R, Albonette-Felicio T, Hardesty DA, Prevedello DM. Comparative anatomical analysis between the minipterional and supraorbital approaches. J Neurosurg. 2020;134(3):1276–84. https://doi.org/10.3171/2019.12.JNS193196.
Galbraith JE, Sullivan JH. Decompression of the perioptic meninges for relief of papilledema. Am J Ophthalmol. 1973;76(5):687–92. https://doi.org/10.1016/0002-9394(73)90564-3.
Figueiredo EG, Deshmukh P, Nakaji P, Crusius MU, Crawford N, Spetzler RF, et al. The minipterional craniotomy: technical description and anatomic assessment. Neurosurgery. 2007;61(5 Suppl 2):256–64; discussion 64–5. https://doi.org/10.1227/01.neu.0000303978.11752.45.
Jägersberg M, Brodard J, Qiu J, Mansouri A, Doglietto F, Gentili F, et al. Quantification of working volumes, exposure, and target-specific maneuverability of the pterional craniotomy and its minimally invasive variants. World Neurosurg. 2017;101:710–717.e2. https://doi.org/10.1016/j.wneu.2017.02.011.
Rychen J, Croci D, Roethlisberger M, Nossek E, Potts M, Radovanovic I, et al. Minimally invasive alternative approaches to pterional craniotomy: a systematic review of the literature. World Neurosurg. 2018;113:163–79. https://doi.org/10.1016/j.wneu.2018.02.016.
Pellerin P, Lesoin F, Dhellemmes P, Donazzan M, Jomin M. Usefulness of the orbitofrontomalar approach associated with bone reconstruction for frontotemporosphenoid meningiomas. Neurosurgery. 1984;15(5):715–8. https://doi.org/10.1227/00006123-198411000-00016.
Hakuba A, Liu S, Nishimura S. The orbitozygomatic infratemporal approach: a new surgical technique. Surg Neurol. 1986;26(3):271–6. https://doi.org/10.1016/0090-3019(86)90161-8.
Dunklebarger M. Multimodal approaches in the management of malignancies involving the orbit. Oper Tech Otolaryngol-Head Neck Surg. 2018;29:232–42.
Park HH, Sung KS, Moon JH, Kim EH, Kim SH, Lee KS, et al. Lateral supraorbital versus pterional approach for parachiasmal meningiomas: surgical indications and esthetic benefits. Neurosurg Rev. 2020;43(1):313–22. https://doi.org/10.1007/s10143-019-01147-8.
Perneczky A. The keyhole concept in neurosurgery: with endoscope-assisted microsurgery and case studies. New York: Thieme; 1999.
Eroglu U, Shah K, Bozkurt M, Kahilogullari G, Yakar F, Dogan İ, et al. Supraorbital keyhole approach: lessons learned from 106 operative cases. World Neurosurg. 2019;124:e667. https://doi.org/10.1016/j.wneu.2018.12.188.
Mariniello G, Maiuri F, de Divitiis E, Bonavolontà G, Tranfa F, Iuliano A, et al. Lateral orbitotomy for removal of sphenoid wing meningiomas invading the orbit. Neurosurgery. 2010;66(6 Suppl Operative):287–92; discussion 92. https://doi.org/10.1227/01.NEU.0000369924.87437.0B.
Chabot JD, Gardner PA, Stefko ST, Zwagerman NT, Fernandez-Miranda JC. Lateral orbitotomy approach for lesions involving the middle fossa: a retrospective review of thirteen patients. Neurosurgery. 2017;80(2):309–22. https://doi.org/10.1093/neuros/nyw045.
Zhou G, Ju X, Yu B, Tu Y, Shi J, Wu E, et al. Navigation-guided endoscopy combined with deep lateral orbitotomy for removal of small tumors at the lateral orbital apex. J Ophthalmol. 2018;2018:2827491. https://doi.org/10.1155/2018/2827491.
Dallan I, Sellari-Franceschini S, Turri-Zanoni M, de Notaris M, Fiacchini G, Fiorini FR, et al. Endoscopic transorbital superior eyelid approach for the management of selected spheno-orbital meningiomas: preliminary experience. Oper Neurosurg (Hagerstown). 2018;14(3):243–51. https://doi.org/10.1093/ons/opx100.
Zoia C, Bongetta D, Gaetani P. Endoscopic transorbital surgery for spheno-orbital lesions: how I do it. Acta Neurochir. 2018;160(6):1231–3. https://doi.org/10.1007/s00701-018-3529-5.
De Rosa A, Pineda J, Cavallo LM, Di Somma A, Romano A, Topczewski TE, et al. Endoscopic endo- and extra-orbital corridors for spheno-orbital region: anatomic study with illustrative case. Acta Neurochir. 2019;161(8):1633–46. https://doi.org/10.1007/s00701-019-03939-9.
Abhinav K, Acosta Y, Wang WH, Bonilla LR, Koutourousiou M, Wang E, et al. Endoscopic endonasal approach to the optic canal: anatomic considerations and surgical relevance. Neurosurgery. 2015;11(Suppl 3):431–45; discussion 45–6. https://doi.org/10.1227/NEU.0000000000000900.
Cabrilo I, Dorward NL. Endoscopic endonasal intracanalicular optic nerve decompression: how I do it. Acta Neurochir. 2020;162(9):2129–34. https://doi.org/10.1007/s00701-020-04476-6.
Jeon C, Hong SD, Woo KI, Seol HJ, Nam DH, Lee JI, et al. Use of endoscopic transorbital and endonasal approaches for 360° circumferential access to orbital tumors. J Neurosurg. 2020;135:103. https://doi.org/10.3171/2020.6.JNS20890.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Maiuri, F., Mariniello, G., Corvino, S. (2023). The Supraorbital Pterional Approach. In: Bonavolontà, G., Maiuri, F., Mariniello, G. (eds) Cranio-Orbital Mass Lesions. Springer, Cham. https://doi.org/10.1007/978-3-031-35771-8_6
Download citation
DOI: https://doi.org/10.1007/978-3-031-35771-8_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-35770-1
Online ISBN: 978-3-031-35771-8
eBook Packages: MedicineMedicine (R0)