Abstract
Primary open-angle glaucoma (POAG) is a chronic neurodegenerative disease that may progress to the irreversible blindness. Unstable ocular blood flow is associated with a stronger patients’ reaction to psychological stress, which was described in patients with primary vascular dysregulation. Excessive activity of the sympathetic autonomic nervous system (ANS) may lead to both a blood supply violation of the nervous system and to a decrease in ocular perfusion pressure in the vessels of the optic nerve and choroid. It plays the crucial role in the pathogenesis of normal tension glaucoma. Systemic autonomic dysfunction may show a higher risk of glaucoma progression due to the stronger sensitivity of the optic nerve to fluctuations in intraocular pressure and intraocular perfusion pressure. Heart rate variability is a common tool for studying the autonomic modulation of the sympathovagal balance.
As optic nerve head and retinal microcirculation is of great importance in glaucoma development and progression, it makes it reasonable to search for new methods of the vascular bed’s visualization for the early diagnosis and monitoring of glaucoma. New biomarkers of glaucoma progression including vascular parameters may improve glaucoma treatment and outcome in accordance with a goal of the predictive preventive personalized medicine (PPPM/3PM).
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Predictive preventive personalized medicine (PPPM/3PM)
- Primary vascular dysregulation
- Flammer syndrome
- Ocular blood flow
- Autonomic nervous system (ANS)
- Heart rate variability (HRV)
- Cold provocation test (CPT)
- Early glaucoma detection
- Glaucoma progression
- Color Doppler imaging (CDI)
- OCT angiography (OCTA)
- Choroidal blood flow
- Normal tension glaucoma (NTG)
1 Introduction
Primary open-angle glaucoma (POAG) is a neurodegenerative disease characterized by a progressive course and an irreversible blindness worldwide.
Accurate prediction of optimal treatment beneficial and adverse effects could improve the results of therapy. The early detection of the specific features of the patient is a key point of the personalized approach in glaucoma treatment. Individualized patient profiling is an instrumental for implementing 3PM strategies in glaucoma management [1, 2].
It is believed that there are two groups of factors responsible for the development of glaucomatous optic neuropathy (GON): (1) vascular dysregulation associated with the decrease of ocular blood flow of the optic nerve disc [3] and (2) mechanical dysregulation associated with the scleral membrane damage and infringement of the optic nerve axons. According to well-known Flammer syndrome, patients with an instable ocular blood flow respond stronger to psychological stress as it has been described in patients with primary vascular dysregulation (PVD) [4, 5]. It has also been emphasized that any psychological stress leads to vascular dysfunction [6]. PVD is believed to be a main cause of local vasospasm and impaired autoregulation as well as a possible contributing factor in glaucoma pathogenesis [7, 8]. It has been shown that PVD patients have stronger reaction to psychological stress than non-PVD subjects. It is known that any psychological stress leads to vascular dysfunction and may become a risk factor of glaucoma development and progression.
The small branches of the central retinal artery provide the blood supply of the superficial ONH layer. The prelaminar region (a small area anterior to the lamina cribrosa), however, is mainly supplied by branches from the choroidal arteries and directly from the short posterior ciliary arteries [9]. As the choroidal microcirculation is regulated by the autonomic nervous system (ANS), the ANS dysfunction is involved in glaucoma pathogenesis.
Indeed, there is a growing body of evidence suggesting that glaucoma pathogenesis is related to vascular dysfunction [10,11,12,13,14,15,16]. The consensus on this issue, however, still has not been found due to the lack of adequate techniques for the study of ocular blood flow despite of different measurement tools [17,18,19,20]. Therefore, it highly recommended to search for new visualization methods of the vascular bed for early diagnosis and monitoring of glaucoma. Optical coherence tomography (OCT) is a common tool for diagnosis and treatment of glaucoma disease. Doppler OCT has been used to obtain precise measurements of total retinal blood flow [21]. Although Doppler OCT may be effectively used to detect blood flow in the large vessels around the optic disc, it is not sensitive enough for the accurate measurement of low velocities in the small vessels forming the disc microcirculation. The same refers to the most widely used method—color Doppler imaging (CDI). A new method—OCT angiography (OCT-A)—has been recently introduced. This method allows measuring vessel density in the retina and choroid in the peripapillary and macular areas using high-speed OCT to perform quantitative angiography.
This book chapter will primarily discuss a role of heart rate variability (HRV) assessment and OCTA in glaucoma diagnostics and monitoring.
2 Heart Rate Variability in Glaucoma Patients
According to the literature sources, patients with cardiovascular diseases have 2.33 times more rapid glaucoma progression despite significantly lower intraocular pressure (IOP) values [22]. Moreover, the concept that vascular changes in the eye may be an early indicator of heart diseases is also discussed in literature [23].
Recent studies have shown the role of vascular disturbances and vascular dysregulation in glaucoma [24,25,26,27,28,29]. This makes glaucoma common with such forms of pathology as arterial hypertension/hypotension, migraine, and vascular spasm [29].
According to the existing literature, POAG patients have significantly smaller diameter of the arterial and venous retinal vessels compared to the control subjects. Nevertheless, both venous and arterial dilatation was normal during the activation of neurons, despite their smaller diameter. The dilatation varied among patients and did not depend on the visual field deterioration. This fact was explained by chronic vasoconstriction leading to the limited energy flow to the retinal and brain neurons, followed by hypometabolism (so-called, silent neurons) and, finally, by the death of neurons [30].
Excessive activity of the sympathetic ANS is among the possible causes of ONH blood supply violation and decreased ocular perfusion pressure (OPP) in the optic nerve and choroidal vessels. Moreover, it has been demonstrated that excessive activity of the sympathetic link of the ANS is associated with glaucoma progression due to instability of the ocular blood flow [26].
Cold stimulation, or cold provocation test (CPT) is a well-established provocation test used for detecting abnormal vascular reactivity in patients with autonomic failures [31]. The testing procedure is rather simple: a patient’s hand is dipped into cold water (+4 ° C) with small pieces of ice for 30 seconds and the cardiovascular response is registered (Fig. 1). It has been revealed that the cold provocation test (CPT) may increase the ET-1 level in plasma in glaucoma patients reflected their vascular dysregulation [22]. This phenomenon may also indicate the imbalance of ANS that is manifested mostly during provocation tests, including CPT [33].
Cold provocation test (CPT). Photoplethysmography with infrared sensor records from a phalanx with an infrared sensor, located in the microprocessor module of data on HRV and peripheral blood flow [32]
Altered ocular blood flow or reduced visual field sensitivity during sympathetic provocation tests has been demonstrated in POAG patients [34,35,36].
2.1 HRV Assessment
HRV assessment is a standard method to evaluate ANS function. The lower the HRV, the more severe the ANS dysfunction with sympathetic predominance [37].
Compared with other methods of assessing autonomic dysfunction, which include cardiovascular reflex tests, sudomotor tests, Valsalva maneuver, the tilting test, HRV assessment is simpler and non-invasive. Numerous studies have validated HRV as a reliable measure of ANS function in cardiac and non-cardiac diseases. The studies showed that POAG patients had autonomic dysfunction characterized by a HRV drop [32, 38].
We have introduced a special hardware–software complex Rhythm-MET that is based on a comprehensive analysis of HRV, systemic hemodynamics, and vegetative regulation [32].
Photoplethysmograms showing the blood flow measurements in a phalanx were recorded using an infrared detector, located in the microprocessor module of data input and processing, and served as the source of HRV and supplemental blood inflow data. Cardiointervals obtained from photoplethysmograms is processed in accordance with the recommendations for the assessment of HRV parameters and their subsequent generalization, including hemodynamics parameters, and for assessment of the functional status (FS) and functional reserves (FR) of the cardiovascular system according to the results of the examination at rest and after CPT in order to form groups homogeneous in both FS and FR.
The ensuing characteristics should be considered in agreement with the transnational standard:
-
Standard divagation of NN-interval (SDNN) is the HRV parameter characterizing the total effect of autonomic blood circulation regulation. A reduction in SDNN reflects low HRV indicating a high tone of heart sympathetic exertion. The drop in SDNN reflects a drop in HRV, which indicates an increase in the heart sympathetic exertion tone.
-
The parameter of parasympathetic autonomic regulation exertion (RMSSD).
-
Total spectral power (TP) is the parameter of absolute exertion level of non-supervisory systems.
-
Power in the high-frequency range (HF) is the parameter of the spectral power of heart rate respiratory undulations reflecting the exertion position of respiratory center. The high-frequency band reflects rapid changes in beat-to-beat variability due to parasympathetic exertion.
-
Power in the low-frequency range (LF). The low-frequency band is considered to be a fair approximation of sympathetic exertion. The low-frequency band reflects substantially sympathetic stimulation.
-
The low/high-frequency rate is a rate of low-frequency to high- frequency power (LF/HF). An advanced rate indicates increased sympathetic exertion or reduced parasympathetic exertion.
-
The number of pairs of successive NN-intervals is the parameter of ascendance degree of parasympathetic regulation over sympathetic one (pNN50).
-
Autonomic regulation indicator (ARI) is the parameter for assessment and exertion of ANS. The increased ARI shows the activation of sympathetic regulation, but the decreased ARI shows the activation of parasympathetic regulation.
-
Variation range characterizing the degree of HRV (TINN).
SDNN is a representative parameter of HRV. The lower HRV is associated with enhanced SNS exertion, which may be characterized by ANS dysfunction [37].
2.2 The Results of HRV Assessment in High Tension Glaucoma (HTG) and Normal Tension (NTG) Glaucoma
The strict definition of POAG includes HTG and NTG. As far as HTG and NTG are concerned, they appear to be a continuum of glaucomatous process, in which the underlying mechanisms shifts from predominantly elevated IOP in HTG to hemodynamic change in NTG. In other words, both HTG and NTG are related to hemodynamics, but it was hypothesized that the evidence of vascular dysfunction would be more pronounced in NTG patients. One of the possible reasons for this is autonomic dysfunction that may contribute to unstable or fluctuating blood pressure and thereby may induce the dysfunction of autoregulation leading to glaucoma development and progression [3].
Some authors report on autonomic dysfunction in HTG with short term and daily analysis of heart rate variability [26, 34, 38,39,40]. However, the existing data on autonomic dysfunction in HTG and HTG are contradictory. According to Riccadonna M. et al., HRV and nocturnal diastolic BP variability were reduced in NTG compared to HTG [39]. Furthermore, these differences were more prominent in more severe clinical forms of NTG. The authors suggested a correlation between the extent of autonomic dis-order and severity of glaucoma.
Brown et al. assessed the baroreflective control of the blood and heart vessels using sinusoidal cervical aspiration and showed that the ANS response in healthy subjects was significantly higher than in glaucoma patients. However, they did not detect any difference between NTG and HTG. According to their data, the decreased sympathetic and parasympathetic modulation during baroreceptor stimulation in the patients with HTG and NTG suggested that autonomic dysfunction that may contribute to the pathogenesis of both diseases [40].
Mroczkowska et al. compared NTG and HTG patients with early glaucoma using 24-h outpatient blood pressure monitoring and measurement of peripheral pulse-wave analysis and thickness of the intima-media complex of the carotid arteries. The authors also evaluated reactivity of retinal vessels to flickering of light. Similar changes in systemic and ocular circulation were observed in glaucoma patients of both groups compared to healthy subjects, but no significant differences were revealed in nocturnal blood pressure, arterial or venous retinal atrial fibrillation, systemic arterial stiffness, and intima-media thickness between patients with NTG and HTG [41].
Bossuyt et al. reported on significantly reduced OPP in patients with HTG and NTG compared to the control healthy subjects. They suggested that perfusion-associated vascular changes play an important role in the pathogenesis of both conditions [42].
On the other hand, there are some important differences between HTG and NTG. The nature of VF progression in HTG differs from other types of glaucoma [43]. It is worth noting that NTG of the eye usually progressed in the central region of the VF, and this response was associated with unstable or strong fluctuations in the average 24-h ocular perfusion pressure and excessive nocturnal drops in systemic arterial blood pressure [26, 44]. Consequently, the division into NTG and HTG in clinical practice is still accepted [45].
A significant decrease in retrobulbar blood flow in HTG is described in the literature [46, 47]. Furthermore, Kaiser et al. revealed that ocular blood flow was decreased both in patients with NTG and HTG who progressed despite normal IOP values [46].
Vascular risk factors varied in HTG and NTG [7, 23, 25]. It was hypothesized in the literature that the vascular dysfunction would be more pronounced in NTG patients compared to HTG [7, 31, 48]. However, some authors reported on similar changes in systemic and ocular circulation in the early stages of the disease in patients with HTG and NTG [41].
The decrease in arterial ocular blood flow was more significant in HTG than in NTG, while lower venous blood flow was detected in patients with NTG [47]. It was emphasized in the literature that decreased blood flow rates in the central retinal artery and central retinal vein were significantly associated with the glaucoma progression both in patients with NTG and HTG with well-controlled IOP (21 mmHg or less) [46]. Circulatory disorders can occur in both NTG and HTG, regardless of the IOP level. One of the reasons is increased sympathetic nervous activity (SNA). This leads to increased vascular resistance and, especially in conditions of endothelial dysfunction, may have consequences for blood circulation related to the pathogenesis of glaucoma. The SNA activation causes an increase in stroke volume, heart rate, and vasoconstriction, as well as regulates circadian blood pressure fluctuations, and it is closely related to night dives.
Recently, we compared the shift in HRV indicators in patients with NTG and HTG after a cold provocation test (CPT). MOPP, 24-h blood pressure and HRV were studied in 30 NTG, 30 patients with HTG and 28 healthy individuals. The cardiovascular system condition was assessed before and after CPT. We applied a method of comparing regression lines to identify the differences between groups. Minimum daily diastolic blood pressure and MOPP were reduced in patients with HTG and NTG in comparison with healthy subjects. There were no differences in MOPP between HTG and NTG before CPT. However, all HRV parameters reflected the predominance of sympathetic innervation in glaucoma patients compared to healthy subjects (p < 0.05). Up to CPT standard deviation of NN intervals (SDNN) HRV was lower at HTG compared to NTG, 27.2 ± 4.1 ms and 35.33 ± 2.43 ms (P = 0.02), respectively. After CBT, SDNN decreased in NTG by 1.7 ms and increased in HTG and healthy individuals by 5.0 ms and 7.09 ms, respectively (P < 0.05). Analysis of the relative shift of other HRV parameters after CPT also revealed a significant difference between NTG and HTG in relation to the predominance of sympathetic innervation in NTG compared to HTG. The conclusion was made that NTG patients had a more pronounced violation of ANS than HTG patients, which was manifested by sympathetic nervous system activation in response to CPT. This discovery relates to the pathogenesis of NTG and suggests the use of HRV assessment in the diagnosis and monitoring of glaucoma [32].
Besides, we observed a significant dipping of diastolic BP both in NTG and HTG patients compared to healthy subjects. Probably, this was a consequence of the sympathetic innervation activation. There is evidence of the presence of a choroidal nerve plexus, represented by numerous internal autonomic ganglia forming an autonomous perivascular network around choroidal vessels [49]. It is considered that it plays a vasodilating function aimed at increasing ocular blood flow. Apparently, vascular mechanisms of optic nerve and retinal trophic disorders and their autonomic regulation play an important role in the ocular pathophysiology and physiology in general and in glaucoma. Vasoconstriction occurs against the background of the predominance of sympathoadrenal effects on arterioles and capillaries, as well as due to a decrease in the activity of parasympathetic effects on retinal vessels.
The abovementioned study contained the evidence of altered MOPP both in HTG and NTG patients compared to healthy subjects. However, there were no difference in the MOPP between HTG and NTG that is consistent with the results of previous studies [35, 39, 41]. The obtained results demonstrate that there may be a significant overlap in the development of NTG and HTG, especially at early glaucoma stage [41, 50]. From this point of view, it has been assumed that provocation tests may be used to reveal alterations in cardiovascular function in NTG patients [42]. Prior to CPT, there was a significant difference in all HRV parameters at rest both between glaucoma patient’s groups and between HTG patients and control subjects. CPT confirmed a significant difference between the control group and glaucoma patients. In the present study we described a significant increase in the activity of the sympathetic ANS in NTG patients in response to CPT. Changes in the main HRV parameters (SDNN, HF, LF, S, and ARI) after CPT emphasize a significant difference between HTG and NTG patients. It is a well-known fact that PVD people have an increase in sympathetic ANS as a response to provocation tests. The NTG development is associated with the presence of PVD [7, 23]. But currently, this fact is not absolutely reliable, and therefore NTG is considered to be a form of open-angle glaucoma. Although the role of PVD in the pathogenesis of GON has been discussed for many years, only recent studies due to the use of modern technologies could prove that patients with NTG, but not healthy individuals, suffer from the retinal blood flow autoregulation failure in the conditions of provocation tests [51]. From this point of view, the dysfunction of the autonomic blood flow regulation seems to be of high importance and its study attracts attention of the researches. Wierzbowska et al. revealed that the sympathovagal balance of ANS in NTG patients shifted towards sympathetic exertion with no change of 24-h pattern of BP variability as compared to the control healthy group [34]. Na et al. demonstrated significantly reduced SDNN values in NTG [38].
There are new highly relevant data indicating the influence of vascular factors on the NTG development. It can be concluded that the disorders of autonomic innervation underlying PVD lead to the development of NTG, but not its specific feature. The presence of ANS imbalance in POAG patients, including HTG, can also be considered as a risk factor for an unfavorable course of GON. In any case, the obtained results demonstrate the significant role of PVD in the pathogenesis of NTG. The results of our study demonstrating the ascendance of SNA in NTG can be useful for distinguishing HTG and NTG.
This conclusion has an important practical implication for detecting NTG (or if it is suspected), determining the prognosis and choosing more appropriate therapy, as well as making recommendations to patients concerning the proper lifestyle. Further studies are needed to verify our findings as well as studies on any therapies that favorably influence ANS activity in patients with glaucoma.
2.3 The Effect of Autonomic Nervous System Dysfunction on the Progression of Primary Open-Angle Glaucoma
Patients with systemic autonomic dysfunction might be at higher risk for glaucoma progression due to higher susceptibility of the optic nerve to fluctuations of IOP or MOPP.
Park et al. in their study described NTG patients with different types of HRV and reported on the fact that the VF progression in patients with sympathetic predominance occurs faster than in patients with higher HRV. The authors concluded that autonomic dysfunction, especially a decrease in SDNN, is a predictor of the progression of central VF in NTG [26]. This study concluded that IOP-independent risk factors, such as orthostatic hypotension, migraine, and autonomic dysfunction, are associated with the progression of central VF.
In another retrospective study of 40 cases of POAG patients who underwent regular reexamination with heart rate variability (HRV) assessment for more than 3 years Liu and co-authors revealed that patients with POAG in the lowest HRV group showed a faster thinning rate of RNFL than those in the highest HRV group. The progression was accompanied by greater fluctuation of intraocular pressure and a decrease of blood pressure and ocular perfusion pressure. Moreover, thinning rate of RNLF was negatively correlated with SDNN values: the more severe the ANS dysfunction is, characterized, the faster the glaucoma progression in POAG patients. The authors explained this by the enhanced activity of the sympathetic tone and concluded that the treatment of ANS may be useful in glaucoma [37].
3 Vascular, Structural, and Functional Deterioration in Glaucoma
3.1 The Association of Vascular, Structural, and Functional Parameters in Glaucoma
For many years, there has been a debate concerning the issue which parameters—structural or functional—have the greatest diagnostic value in glaucoma [52]. Perimetry was considered a golden standard for the diagnosis of primary open-angle glaucoma (POAG) for a long time. The peripapillary retinal nerve fiber layer (RNFL) and the layer of macular ganglion cells are described in the existing literature as the most significant structural markers for the glaucoma diagnosis [53]. Some authors also reported that the molecular parameters have a high discriminating ability and high reproducibility for the early detection of glaucoma compared to the parameters of the peripapillary RNFL [54].
According to our recent studies, the circulatory parameters serve as diagnostic glaucoma markers [55]. Reduction of retinal hemoperfusion in glaucoma has been repeatedly mentioned in the literature [10, 11]. Nowadays, there is much data on hemodynamic disorders in retina, ONH, and retrobulbar circulation in glaucoma [49, 56,57,58]. Moreover, several authors have concluded that color Doppler imaging is associated with a prognostic value for damage to visual function in glaucoma patients [59, 60].
However, it is not clear yet if reduced blood flow is the cause or the consequence of glaucoma damage secondary to retinal ganglion cell (RGC) death. This issue can be solved only due to long-term observation of patients by using available methods of clinical examination of retinal vessels, optic nerve, and choriocapillaris. One of the candidates for this method is optical coherence tomography angiography.
3.2 Optical Coherence Tomography Angiography in Glaucoma Diagnostics and Monitoring
Optical coherence tomographic angiography (OCTA) is a new non-invasive diagnostic technique to study the microcirculation in optic nerve, retina, and choroid. OCTA opens up new prospects for examining the blood supply to main structures usually affected by glaucoma (peripapillary retina, optic disc, and internal macular layers) [61]. The studies have consistently demonstrated reduced ONH [61, 62], peripapillary [63], and macular [63,64,65,66,67] perfusion in glaucoma patients using OCTA.
The cross coefficients of variation (CV) range from 3.2% to 9.0% for the global OCT-A parameters of the macular and peripapillary regions [62] and from 5.0% to 6.9% for the peripapillary region [68]. According to the results of some studies, OCTA measurements of vascular density may complement the existing structural parameters for glaucoma detection and its progression by detecting changes in the microcirculatory bed supplying ganglion cells and axons before changes in structural thickness measurements [12, 65,66,67, 69,70,71]. OCTA has opened the prospects for novel imaging of retinal and ONH microcirculation [72]. OCTA is based on a new three-dimensional angiography algorithm called amplitude-decorrelation angiography with a split spectrum (SSADA), comparing successive B-scans at the same location to detect blood flow using motion contrast. The reproducibility of OCTA has been reported in several studies [61, 73, 74].
Reduced ONH and peripapillary perfusion parameters have been reported by different authors in subjects with glaucoma measured by OCTA [61, 64, 72,73,74,75]. The decreased vessel density (VD) was significantly associated with the severity of visual field damage independent of the structural loss [76, 77].
Different authors have found a significant decrease in IOP in patients with glaucoma compared with healthy people. Wang et al. reported reduced blood flow index in the entire optic disc and inferotemporal segment of the optic disc [73]. The study by Chichara et al. demonstrated the priority of detecting superficial peripapillary retinal VD to differentiate between glaucoma and ophthalmic hypertension and healthy eyes [78]. Liu et al. in their study revealed that there was a significant decrease in peripapillary VD in glaucoma patients compared to healthy subjects of the same age [74]. According to the authors, this indicator had a high diagnostic value for the early detection of glaucoma. Some other studies reported that quantitative OCT-A analysis made it possible to distinguish eyes with glaucoma from healthy eyes by evaluating the entire peripapillary vascular network, from the ILM to the Bruch membrane [76]. According to Yarmohammadi et al., the decreased VD was significantly associated with the severity of visual field damage independent of the structural loss, and whole image vessel density (wiVD) of the disc scan showed the best AUC in their study (AUC: 0.94) [77].
Previously, we have reported better diagnostic accuracy by using capillary density in the macular area over the peripapillary area and the optic disc in the early glaucoma detection [64]. These data are consistent with the literature data on early macular lesions with a high concentration of RGC in glaucoma and explain the localization of the vulnerable area of the retina affected at the very beginning of glaucoma [79, 80].
The functional activity of retinal ganglion cells can be measured using a pattern electroretinogram (PERG). The other objective method of checking visual function is the pattern of visual evoked potentials (PVEP). Glaucomatous changes in PVEP and PERG were reported before the appearance of anomalies in the peripapillary retina and ONH [80,81,82,83,84,85,86]. Moreover, PVEPs were used to assess reversible ganglion cell damage in the studies of neuroprotective agents for glaucoma treatment [87, 88].
Having compared the diagnostic ability of the vascular, structural, and functional parameters in differentiation between the normal eyes, early glaucoma, and moderate to severe glaucoma, we have revealed that the results of the electrophysiological testing along with the retinal microcirculation measured by OCTA demonstrated superiority over the structural variables in early glaucoma detection (Fig. 2) [89].
Clinical examples of the normal controls, early glaucoma, and severe glaucoma. GCC map and RNFL thickness map (a), SAP visual field results showing corresponding visual field defects (c), PVEP-protocols (d), PERG-protocols (e). Figure b show a stepwise decrease of vessel density both in the circumpapillary VD map and Fovea and circum parafovea VD map (wiVD Disc is reduced from 54.25% (normal eye) to 52.26% (early glaucoma) to 42.17% (severe glaucoma); wiVD Macula Superficial is reduced from 52.56% (normal eye) to 41.95% (early glaucoma) to 41.29% (severe glaucoma). Figure d show a stepwise decrease of the amplitude and prolonged latency of P100 component of PVEP and e show a decrease of the amplitude and prolonged latency of N95 component of PERG in glaucoma eyes compared to normal eye. (Modified from the National Journal of Glaucoma (RUS), 2019 with permission)
According to our study, a strong correlation between the amplitude of the P100 PVVP and the density of vessels in the ZEN and peripapillary retina, on the one hand, and a correlation between the density of vessels in the superficial macular plexus and the GCC thickness in inferior hemisphere, on the other hand, were revealed. It can be concluded that the damage of ganglion cells may be associated with decreased blood supply to the retina. It was shown that the macular capillary vessel area density strongly correlated with inferior hemimacula or structural damage [65]. Inferior hemimacular retinal structure is subject to a decrease in the area of the capillary vessels of the retina in eyes with glaucoma. Moreover, the blood flow parameters in ophthalmic artery, central retinal artery, and short posterior ciliary arteries in early glaucoma significantly correlate with the retinal thickness in the inferior hemisphere [90].
The existing results of studies demonstrate the importance of microcirculation parameters of the peripapillary retinal and macular region, PERGs and PVEPs, for early detection and monitoring of glaucoma.
3.3 The Detection of Glaucoma Progression Using OCTA
Successful monitoring of POAG depends on early detection of the disease progression. An individual treatment plan should be based on the risk factors and specific clinical markers that allow predicting the rate of disease progression and avoiding unreasonable prescriptions.
Increased intraocular pressure (IOP) [91,92,93,94,95] and its fluctuations [96] are commonly considered to be the main recognized factors for POAG progression. However, there is an increasing interest in the influence of other factors as it is known that the disease can progress at normal IOP [27, 97, 98]. These factors include a thin cornea [93, 99], low corneal hysteresis [100], optic disc hemorrhages [95, 101], peripapillary atrophy of the choroid [91, 100], age of patients [91, 95, 102], female sex [95, 103], presence of pseudoexfoliation [92], late detection of glaucoma [93], and arterial hypotension [104, 105] or/and hypertension [103, 106]. Nevertheless, researchers disagree on many issues regarding progression risk factors and recommend to take into account only highly reliable results concerning significant parameters [95, 107].
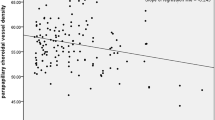
A number of studies demonstrate the importance of using OCT angiography for the detection of glaucoma progression. Moghimi S et al. showed that a higher rate of RNFL thinning was associated with an initially reduced density of macular and peripapillary vessels in glaucoma patients [70]. An increase in the area of depletion of vascular macular blood flow, according to the literature, significantly correlates with the presence of structural and functional markers of glaucoma progression, such as the appearance of visual field defects and thinning of the RNFL [69]. According to literature, there is a direct relationship between the vascular, structural, and functional changes in patients with advanced glaucoma [108]. Figure 3 demonstrates a clinical example of the structural and vascular loss that is accompanied by the functional deterioration.
Association of structural, functional progression with the decrease of microvascularity in the patient with early glaucoma. (a) Progressive RNFL and GCC loss; (b, c) A decrease of vessel density in the circumpapillary VD map; (d) A decrease of thickness of GCC and ONH corresponding to the loss of vessel density; (e) Fundus visualization of ONH; (f, g) SAP visual field results
Retinal microvascular loss may be detected more often than structural ones due to the presence of the so-called floor effect in the late stages of the disease, which certainly puts the use of OCT angiography in the forefront in assessing progression of glaucomatous optic neuropathy [109]. Thus, in advanced glaucoma, the measurement of parameters of the microvascular superficial parafoveal vessel density is more prognostic due to the lack of “floor effect” [110].
According to Kwon and co-authors, the visual field progression rate was significantly faster in eyes with parapapillary deep layer microvasculature dropout detected by OCT-A than in those without dropout, and the location of dropout and VF progression was spatially correlated. These findings implicate dropout as a structural parameter suggestive of past glaucomatous VF progression [111].
An analysis of FAZ in glaucoma monitoring revealed that, as it manifested, there was a significant thinning of the RNFL and GC-IPL, while no changes in perimetric data were observed [112].
3.4 Role of the Peripapillary Choriocapillaris Loss in Glaucoma Development and Progression
Choroid is another important structure for OCTA assessment. It has the highest blood flow compared to any other tissue in the body [113]. The choriocapillary layer is formed from small arteries and veins, which then break up into many capillaries, passing several red blood cells in one row, which allows more oxygen to enter the retina. The choriocapillary layer of the choroid plays a crucial role in supplying oxygen and nutrition to the outer cells of the retina, especially the retinal pigment epithelium [114]. It should be emphasized that a lesion of blood flow in the choriocapillary layer in the area of the peripapillary retina leads to damage to the lamina cribrosa of sclerae, resulting in a weakening of the structures of the latter.
Optical coherence tomography and angiography mode have opened up new prospects in the study of choriocapillaris blood flow, or rather, the loss of choriocapillaris of the peripapillary retina in glaucoma (Fig. 4).
(a, b, c). Determination of the area and localization of choriocapillary dropout within the beta zone on the density map using RTVue XR Avanti («Optovue», USA). On scans of 4.5 × 4.5 mm of ONH (a) at the level of the choroid inside the beta zone, the total dropout of choriocapillaris is determined (b: red arrows), with the help of ImageJ program, the area of choriocapillaris dropout in mm2 inside the beta zone is calculated by pixels (c: red areas)
According to literature, more than the half of patients with primary open-angle glaucoma have a choriocapillary dropout in the beta zone of the ONH [115].
Also, during the examination of 118 patients, scientists revealed that with primary open-angle glaucoma and the presence of defects in the lamina cribrosa sclerae (59 patients), the frequency of detection of choriocapillary dropout was more frequent than in patients with glaucoma without defects in the lamina cribrosa sclerae (59 patients) [116].
The study by Eun Ji Lee et al. found out that larger lamina cribrosa curvature index (LCCI), disc hemorrhage, and the presence of cMvD were associated with faster global RNFL thinning in multivariate regression analysis. The regression tree analysis revealed three stratified groups based on the RNFL thinning rate divided into LCCI and the presence of cMvD. Eyes with LCCI ≥11.87 had the fastest RNFL thinning (−2.4 ± 0.8 microns/year). Among the eyes with LCCI <11.87, the presence of cMvD was the strongest factor influencing the faster thinning of RNFL (−1.5 ± 0.8 microns/year). Eyes with LCCI <11.87 and without cMvD showed the lowest RNFL thinning (−0.8 ± 0.9 μm/year) [117].
In a recent study, we also showed that the higher the stage of glaucoma, the larger the area of loss of choriocapillaris measured in mm2 (Fig. 5).
Youn Hye Jo in their study showed that the initial parameters of choroidal microvasculature dropout (CMvD) can be predictors of the rapid development of visual field defects [118].
We have been obtaining glaucoma patients with the fast thinning of choroid that was associated with the rapid visual field deterioration despite the normal IOP (Fig. 6).
Clinical example of the choroidal thinning in correspondence with structural, functional and vascular deterioration. (a) A decrease of thickness of RNFL and GCC; (b) A decrease of amplitude of PVEP; (c) A decrease of end-diastolic velocity in the short posterior artery; (d) A deep layer microvasculature dropout; (e) Decrease of the peripapillary choroidal thickness
A thin peripapillary choroid is also considered a predictor of glaucoma progression [119]. The study by Kim et al. reported that the eyes with the loss of capillaries in the lower temporal quadrant of the peripapillary vasculature have more pronounced visual field damage compared to the eyes with preserved peripapillary choroidal microcirculation. Kim et al. noted an inverse relation between the VD in the peripapillary vasculature and the SNVS thickness [120]. The data of the present study on the prognostic role of the thickness of the peripapillary vasculature are consistent with these results.
Two-year observational study by Park H. et al. revealed the prolapse of peripapillary choroidal microvessels (MvD) in glaucomatous eyes with or without disc hemorrhage (DH). The authors demonstrated that MvD was significantly higher in patients with progressive glaucoma than in stable patients in both the DH and no-DH groups. Park H. et al. concluded that MvD is associated with progressive RNFL thinning. They suggested that OCTA was a new biological marker for glaucoma progression, and this biomarker is a peripapillary choroidal microvascular system [69]. The authors explained this phenomenon is caused by choroidal vascular insufficiency, which may play a significant role in the lack of prelaminar nutrition of the optic nerve during the progression of glaucoma.
3.5 The Prognosis of Glaucoma Progression on the Basis of Functional, Structural, and Circulatory Data
It is rather complicated to define glaucoma progression, since many various factors influence the course of glaucoma [92, 103]. The study by De Moraes reported on the fact that combining data can be useful when discussing risks and treatment options with individual patients, as well as when standardizing the quantitative assessment of the risk of progression in treated patients with glaucoma [100]. From this point of view, the application of new biomarkers as the vessel density, measured by OCTA, may improve the prediction of glaucoma progression.
We conducted a comparative study of microcirculation parameters, predictors of glaucoma progression, and other clinical data [121]. The following results were obtained: decreased blood circulation, including regional microcirculation and retrobulbar blood flow, is associated with the progression of glaucoma. According to the multilevel analysis of models of mixed effects, four predictors were revealed: parafoveal superficial plexus vessel density, the end-diastolic velocity of the central retinal artery, the volume of focal loss of the ganglion cell complex and the peak follow-up IOP. The rate of disease progression expressed as a change of perimetric index MD of visual field and RNFL thinning correlated with the peak follow-up IOP and the end-diastolic velocity in posterior ciliary arteries. A positive correlation was also observed between the thickness of the retinal inner layers in parafovea and the parafovea vessel density in superficial layer. Mean ocular perfusion pressure (MOPP) correlated with the average ganglion cells complex thickness and its focal loss volume [121].
The present study concluded that only a decrease in parafoveal VD in the superficial layer was associated with the glaucoma progression, which was confirmed by both functional and structural disorders in all statistical models. Several existing studies have shown that OCTA makes it possible to detect the disease at the preperimetric stage, and OCTA parameters correlate better with functional parameters, including electrophysiological parameters, than with structural ones [61, 77, 89].
Some authors have reviled that the disease progression is mainly associated with low end-diastolic velocity in short posterior ciliary arteries [59] and the high resistive index in central retinal artery [122].
A decrease in OCTA VD may actually precede both structural and functional losses and, therefore, may be useful in the diagnosis and monitoring of glaucoma at the earliest stages [123, 124]. This decrease detected at early glaucoma stages may be used as a glaucoma progression predictor.
Generally speaking, the role of OCTA in identifying glaucoma progression predictors and the dynamic range of vascular density is insignificant. Moreover, the study conducted by Moghimi and co-authors also has underlined that the correlation between the rate of RNFL loss and VD measurements was not strong. However, Moghimi et al. concluded that the OCTA parameters can predict RNFL loss during the long-term follow-up. The authors reported that OCTA may provide the data concerning early RGCs dysfunction with lower metabolic needs.
According to our study, a decrease in parafoveal vessel density can serve as a predictor of death of cells and subsequent tissue thinning with functional loss. The inferior sector of the macula as a vulnerable region for glaucoma damage makes sense, since most of the nerve fibers of the lower macula are projected into the lower quadrant of the optic disc, an area that is particularly susceptible to glaucomatous damage. Hood et al. described that thinning of RGC and the nerve fiber layer is already present in preperimetric glaucoma patients and progresses with increasing loss of mean deviation (MD) [79]. Lommatzsch et al. confirmed first that the VD of the inferior perimacular sector is lower than in all other sectors, and that this value decreases in early forms of glaucoma with progressive losses with worsening of progressive losses with deteriorating MD [125]. The recent study showed that a decrease in VF MD for every 1 dB was associated with a decrease in macular wiVD by 0.43% and pfVD by 0.46%. According to their study, the correlation between macular vessel density and MD of the visual field in the whole image was stronger than ONH whole image capillary density and GCC and RNFL thicknesses [108]. However, the authors did not sufficiently study the reasons for early macula damage in glaucoma. This fact is probably due to retinal ischemia with increased metabolic needs of the area with the highest concentration of RGC. According to the results of our study, the thickness of the RGC and its characteristics (GLV and FLV) correlated with the average ocular perfusion pressure.
The assessment of macular VD in glaucoma monitoring also makes it possible to determine the disease progression in such cases when structural parameters are unacceptable due to the presence of floor effect. According to Moghimi, even a pronounced loss of visual function (MD reached −19 dB) did not result in the “floor effect” of VD [126]. Similar results were obtained by Rao [127] who demonstrated that the floor effect for the specified parameter did not occur at MD -15 dB. Other authors reported that the floor effect in the peripapillary retinal VD occurs somewhat earlier, which is observed at MP < −14.0 dB, but at the same time later than for such morphometric parameters as RGC thickness and RNFL [79, 128]. According to Hood et al., this effect for RNFL is already visible at MD of −10 dB [79]. Other authors have also noted the advantages of studying GCC thickness compared to RNFL [129]. Furthermore, the importance of assessing peripapillary VD in the late glaucoma stages has been described in the recent studies [130]. It has been revealed that the detection of glaucoma progression at early stages is more reliable with the use of SD-OCT, while functional deterioration is more visible in the middle and late stages. In our previous studies we reported that the structural parameters (in particular, retinal GCС) have priority over functional ones in early glaucoma compared to advanced stage of the disease [89]. However, peripapillary VD had the highest diagnostic accuracy to distinguish between early, middle and late stages, while parafoveal VD in the surface layer had the highest diagnostic accuracy to distinguish between early glaucoma and healthy eyes. In general, the diagnostic ability of the OCTA parameters in early glaucoma was higher compared to GCC and RNFL thickness.
According to some studies, a decrease in macula VD, OHN, and the peripapillary retina is associated with a higher rate of progression of RNFL loss in mild and moderate glaucoma, suggesting that a VD decrease may be a predictor of progression risk [126]. Their results have shown that vessel density measures tend to be more strongly associated with severity of visual field damage than thickness measures and may be an additional tool to monitor progression in advanced disease. These data are consistent with our results, according to which OCTA parameters serve as predictors of glaucoma progression [121].
There is a lack of information on the influence of lowering IOP on retinal microcirculation. The experimental studies have reported that microcirculation in the retina, choriocapillaris, sclera, and lattice plate remains unchanged even with significant IOP fluctuations [91]. On the other hand, according to some clinical studies, OCTA vessel density strongly correlates with IOP [131,132,133].
In conclusion, OCTA may significantly improve the early detection of glaucoma progression, as formerly OCT has provided more precise diagnostics in regard to this detection.
Abbreviations
- ANS:
-
Autonomic nervous system
- ARI:
-
Autonomic regulation index
- BP:
-
Blood pressure
- CDI:
-
Color Doppler imaging
- CPT:
-
Cold provocation test
- CRA:
-
Central retinal artery
- CV:
-
Coefficients of variation
- EPS:
-
Enhanced polarization-sensitive
- FLV:
-
Focal loss volume
- FR:
-
Functional reserves
- FS:
-
Functional status
- GCC:
-
Ganglion cell complex
- GLV:
-
Global loss volume
- GON:
-
Glaucomatous optic neuropathy
- HF:
-
High-frequency range
- HRV:
-
Heart rate variability
- HTG:
-
High tension glaucoma
- ILM:
-
Internal limiting membrane
- IOP:
-
Intraocular pressure
- IPL:
-
Inner plexiform layer
- LF:
-
Low-frequency range
- MD:
-
Mean deviation
- MOPP:
-
Mean ocular perfusion pressure
- MvD:
-
Microvasculature dropout
- NFI:
-
Nerve fiber indicator
- NTG:
-
Normal tension glaucoma
- OCTA:
-
Optic coherence tomography angiography
- ONH:
-
Optic nerve head
- OPP:
-
Ocular perfusion pressure
- PACG:
-
Primary angle closure glaucoma
- PERG:
-
Pattern electroretinogram
- pfVD:
-
Perifoveal vessel density
- POAG:
-
Primary open-angle glaucoma
- PPPM:
-
Predictive preventive personalized medicine
- PVD:
-
Primary vascular dysregulation
- PVEP:
-
Pattern visual evoked potential
- RGC:
-
Retinal ganglion cells
- RI:
-
Resistivity index
- RMSSD:
-
Parameter of parasympathetic autonomic regulation activity
- RNFL:
-
Retinal nerve fiber layer
- ROP:
-
Rate of progression
- SAP:
-
Standard automated perimetry
- SDNN:
-
Standard deviation of NN-interval
- SD-OCT:
-
Spectral-domain optic coherence tomography
- SNA:
-
Sympathetic neural activity
- SPCA:
-
Short posterior ciliary artery
- SSADA:
-
Split-spectrum amplitude-decorrelation angiography
- TP:
-
Total spectral power
- VD:
-
Vessel density
- VF:
-
Visual fields
- VFI:
-
Visual field index
- wiVD:
-
Whole image vessel density
References
Balendra SI, Zollet P, Casasca GCADGE, Cordeiro MF (2020) Personalized approaches for the management of glaucoma. Exp Rev Precis Med Drug Dev 5(3):145–164. https://doi.org/10.1080/23808993.2020.1756770
Nathan M. Radcliffe; Jason Bacharach, Murray Fingeret, Lorraine M Provencher. Glaucoma treatments in 2021: personalizing patient needs 2021
Flammer J, Konieczka K, Bruno RM, Virdis A, Flammer AJ, Taddei S (2013) The eye and the heart. Eur Heart J 34(17):1270–1278. https://doi.org/10.1093/eurheartj/eht023
Nicolela MT, Ferrier SN, Morrison CA et al (2003) Effects of cold-induced vasospasm in glaucoma: the role of endothelin-1. Invest Ophthalmol Vis Sci 44(6):2565–2572. https://doi.org/10.1167/iovs.02-0913
Golubnitschaja O (2019) Flammer syndrome: from phenotype to associated pathologies, prediction, prevention and personalisation. Flammer Syndrome
Neves VJ, Moura MJ, Almeida BS et al (2012) Chronic stress, but not hypercaloric diet, impairs vascular function in rats. Stress 15(2):138–148. https://doi.org/10.3109/10253890.2011.601369
Konieczka K, Ritch R, Traverso CE et al (2014) Flammer syndrome. EPMA J 5(1):11. Published 2014 Jul 8. https://doi.org/10.1186/1878-5085-5-11
Konieczka K, Flammer J, Sternbuch J, Binggeli T, Fraenkl S (2017) Leber’s hereditary optic neuropathy, Normal tension glaucoma, and Flammer syndrome: long term follow-up of a patient. Lebersche hereditäre Optikusneuropathie, Normaldruckglaukom und Flammer-Syndrom—eine langzeitige Beobachtung eines Patienten. Klin Monbl Augenheilkd 234(4):584–587. https://doi.org/10.1055/s-0042-119564
Schmidl D, Boltz A, Kaya S et al (2012) Comparison of choroidal and optic nerve head blood flow regulation during changes in ocular perfusion pressure. Invest Ophthalmol Vis Sci 53(8):4337–4346. Published 2012 Jul 1. https://doi.org/10.1167/iovs.11-9055
Grieshaber MC, Flammer J (2005) Blood flow in glaucoma. Curr Opin Ophthalmol 16(2):79–83. https://doi.org/10.1097/01.icu.0000156134.38495.0b
Hayreh SS (2001) Blood flow in the optic nerve head and factors that may influence it. Prog Retin Eye Res 20(5):595–624. https://doi.org/10.1016/s1350-9462(01)00005-2
Hwang JC, Konduru R, Zhang X et al (2012) Relationship among visual field, blood flow, and neural structure measurements in glaucoma. Invest Ophthalmol Vis Sci 53(6):3020–3026. Published 2012 May 17. https://doi.org/10.1167/iovs.11-8552
Jia Y, Tan O, Tokayer J et al (2012) Split-spectrum amplitude-decorrelation angiography with optical coherence tomography. Opt Express 20(4):4710–4725. https://doi.org/10.1364/OE.20.004710
Rusia D, Harris A, Pernic A et al (2011) Feasibility of creating a normative database of colour Doppler imaging parameters in glaucomatous eyes and controls. Br J Ophthalmol 95(9):1193–1198. https://doi.org/10.1136/bjo.2010.188219
Weinreb RN, Harris A (2007) Ocular blood flow in glaucoma: consensus series 6. Kugler Publications, Dordrecht
Yaoeda K, Shirakashi M, Fukushima A et al (2003) Relationship between optic nerve head microcirculation and visual field loss in glaucoma. Acta Ophthalmol Scand 81(3):253–259. https://doi.org/10.1034/j.1600-0420.2003.00073.x
Ben-Zion I, Harris A, Weizman Y, Ehrlich R, Rechtman E (2008) An updated review of methods for human retinal oximetry measurements and current applications. Harefuah 147(10):812–817. 836. Hebrew
Jonas JB, Harazny J, Budde WM, Mardin CY, Papastathopoulos KI, Michelson G (2003) Optic disc morphometry correlated with confocal laser scanning Doppler flowmetry measurements in normal-pressure glaucoma. J Glaucoma 12(3):260–265. https://doi.org/10.1097/00061198-200306000-00014
Kawasaki R, Wang JJ, Rochtchina E, Lee AJ, Wong TY, Mitchell P (2013) Retinal vessel caliber is associated with the 10-year incidence of glaucoma: the Blue Mountains Eye Study. Ophthalmology 120(1):84–90. https://doi.org/10.1016/j.ophtha.2012.07.007
Stalmans I, Vandewalle E, Anderson DR et al (2011) Use of colour Doppler imaging in ocular blood flow research. Acta Ophthalmol 89(8):e609–e630. https://doi.org/10.1111/j.1755-3768.2011.02178.x
Wang Y, Bower BA, Izatt JA, Tan O, Huang D (2008) Retinal blood flow measurement by circumpapillary Fourier domain Doppler optical coherence tomography. J Biomed Opt 13(6):064003. https://doi.org/10.1117/1.2998480
Chan TCW, Bala C, Siu A, Wan F, White A (2017) Risk factors for rapid glaucoma disease progression. Am J Ophthalmol 180:151–157. https://doi.org/10.1016/j.ajo.2017.06.003
Flammer J, Konieczka K, Flammer AJ (2013) The primary vascular dysregulation syndrome: implications for eye diseases. EPMA J 4(1):14. Published 2013 Jun 7. https://doi.org/10.1186/1878-5085-4-14
Galassi F, Sodi A, Ucci F, Renieri G, Pieri B, Baccini M (2003) Ocular hemodynamics and glaucoma prognosis: a color Doppler imaging study. Arch Ophthalmol 121(12):1711–1715. https://doi.org/10.1001/archopht.121.12.1711
Flammer J, Orgül S, Costa VP et al (2002) The impact of ocular blood flow in glaucoma. Prog Retin Eye Res 21(4):359–393. https://doi.org/10.1016/s1350-9462(02)00008-3
Park HY, Park SH, Park CK (2014) Central visual field progression in normal-tension glaucoma patients with autonomic dysfunction. Invest Ophthalmol Vis Sci 55(4):2557–2563. Published 2014 Apr 21. https://doi.org/10.1167/iovs.13-13742
Drance S, Anderson DR, Schulzer M, Collaborative Normal-Tension Glaucoma Study Group (2001) Risk factors for progression of visual field abnormalities in normal-tension glaucoma. Am J Ophthalmol 131(6):699–708. https://doi.org/10.1016/s0002-9394(01)00964-3
Satilmis M, Orgül S, Doubler B, Flammer J (2003) Rate of progression of glaucoma correlates with retrobulbar circulation and intraocular pressure. Am J Ophthalmol 135(5):664–669. https://doi.org/10.1016/s0002-9394(02)02156-6
Phelps CD, Corbett JJ (1985) Migraine and low-tension glaucoma. A case-control study. Invest Ophthalmol Vis Sci 26(8):1105–1108
Zhou W, Sabel B (2023) Vascular dysregulation in glaucoma: retinal vasoconstriction and normal neurovascular coupling in altitudinal visual field defects. EPMA J 14(1):87–99. https://doi.org/10.1007/s13167-023-00316-6
Gherghel D, Hosking SL, Cunliffe IA (2004) Abnormal systemic and ocular vascular response to temperature provocation in primary open-angle glaucoma patients: a case for autonomic failure? Invest Ophthalmol Vis Sci 45(10):3546–3554. https://doi.org/10.1167/iovs.04-0290
Kurysheva NI, Ryabova TY, Shlapak VN (2018) Heart rate variability: the comparison between high tension and normal tension glaucoma. EPMA J 9(1):35–45. https://doi.org/10.1007/s13167-017-0124-4
Feke GT, Pasquale LR (2008) Retinal blood flow response to posture change in glaucoma patients compared with healthy subjects. Ophthalmology 115(2):246–252. https://doi.org/10.1016/j.ophtha.2007.04.055
Wierzbowska J, Wierzbowski R, Stankiewicz A, Siesky B, Harris A (2012) Cardiac autonomic dysfunction in patients with normal tension glaucoma: 24-h heart rate and blood pressure variability analysis. Br J Ophthalmol 96(5):624–628. https://doi.org/10.1136/bjophthalmol-2011-300945
Gherghel D, Hosking SL, Cunliffe IA, Heitmar R (2010) Transient cardiac ischaemia and abnormal variations in systemic blood pressure in unselected primary open angle glaucoma patients. Ophthalmic Physiol Opt 30(2):175–181. https://doi.org/10.1111/j.1475-1313.2009.00704.x
Chai E, Goldberg I, Chia A, Chen J (1999) Visual field responses to a hand vibration stimulus. Surv Ophthalmol 43(Suppl 1):S79–S86. https://doi.org/10.1016/s0039-6257(99)00051-x
Liu B, Zhao Y, Zhang H (2022) The effect of autonomic nervous system dysfunction on the progression of primary open-angle glaucoma. Int J Gen Med 15:4565–4573. Published 2022 May 2. https://doi.org/10.2147/IJGM.S362275
Na KS, Lee NY, Park SH, Park CK (2010) Autonomic dysfunction in normal tension glaucoma: the short-term heart rate variability analysis. J Glaucoma 19(6):377–381. https://doi.org/10.1097/IJG.0b013e3181c4ae58
Riccadonna M, Covi G, Pancera P et al (2003) Autonomic system activity and 24-hour blood pressure variations in subjects with normal- and high-tension glaucoma. J Glaucoma 12(2):156–163. https://doi.org/10.1097/00061198-200304000-00011
Brown CM, Dütsch M, Michelson G, Neundörfer B, Hilz MJ (2002) Impaired cardiovascular responses to baroreflex stimulation in open-angle and normal-pressure glaucoma. Clin Sci (Lond) 102(6):623–630. https://doi.org/10.1042/cs1020623
Mroczkowska S, Benavente-Perez A, Negi A, Sung V, Patel SR, Gherghel D (2013) Primary open-angle glaucoma vs normal-tension glaucoma: the vascular perspective. JAMA Ophthalmol 131(1):36–43. https://doi.org/10.1001/2013.jamaophthalmol.1
Bossuyt J, Vandekerckhove G, De Backer TLM et al (2015) Vascular dysregulation in normal-tension glaucoma is not affected by structure and function of the microcirculation or macrocirculation at rest: a case-control study. Medicine (Baltimore) 94(2):e425. https://doi.org/10.1097/MD.0000000000000425
Ahrlich KG, De Moraes CG, Teng CC et al (2010) Visual field progression differences between normal-tension and exfoliative high-tension glaucoma. Invest Ophthalmol Vis Sci 51(3):1458–1463. https://doi.org/10.1167/iovs.09-3806
Choi J, Jeong J, Cho HS, Kook MS (2006) Effect of nocturnal blood pressure reduction on circadian fluctuation of mean ocular perfusion pressure: a risk factor for normal tension glaucoma. Invest Ophthalmol Vis Sci 47(3):831–836. https://doi.org/10.1167/iovs.05-1053
Thonginnetra O, Greenstein VC, Chu D, Liebmann JM, Ritch R, Hood DC (2010) Normal versus high tension glaucoma: a comparison of functional and structural defects. J Glaucoma 19(3):151–157. https://doi.org/10.1097/IJG.0b013e318193c45c
Kaiser HJ, Schoetzau A, Stümpfig D, Flammer J (1997) Blood-flow velocities of the extraocular vessels in patients with high-tension and normal-tension primary open-angle glaucoma. Am J Ophthalmol 123(3):320–327. https://doi.org/10.1016/s0002-9394(14)70127-8
Kurysheva N, Kiseleva T, Irtegova E, Parshunina O (2015) Venous ocular blood flow in primary open angle glaucoma. Int J Ophthalmol Eye Res 3:1–7
Gasser P, Flammer J (1991) Blood-cell velocity in the nailfold capillaries of patients with normal-tension and high-tension glaucoma. Am J Ophthalmol 111(5):585–588. https://doi.org/10.1016/s0002-9394(14)73703-1
Grunwald JE, Piltz J, Hariprasad SM, DuPont J (1998) Optic nerve and choroidal circulation in glaucoma. Invest Ophthalmol Vis Sci 39(12):2329–2336
Koch EC, Staab J, Fuest M, Witt K, Voss A, Plange N (2015) Blood pressure and heart rate variability to detect vascular dysregulation in glaucoma. J Ophthalmol 2015:798958. https://doi.org/10.1155/2015/798958
Drance SM, Douglas GR, Wijsman K, Schulzer M, Britton RJ (1988) Response of blood flow to warm and cold in normal and low-tension glaucoma patients. Am J Ophthalmol 105(1):35–39. https://doi.org/10.1016/0002-9394(88)90118-3
Hood DC, Anderson SC, Wall M, Raza AS, Kardon RH (2009) A test of a linear model of glaucomatous structure-function loss reveals sources of variability in retinal nerve fiber and visual field measurements. Invest Ophthalmol Vis Sci 50(9):4254–4266. https://doi.org/10.1167/iovs.08-2697
Ojima T, Tanabe T, Hangai M, Yu S, Morishita S, Yoshimura N (2007) Measurement of retinal nerve fiber layer thickness and macular volume for glaucoma detection using optical coherence tomography. Jpn J Ophthalmol 51(3):197–203. https://doi.org/10.1007/s10384-006-0433-y
Nakatani Y, Higashide T, Ohkubo S, Takeda H, Sugiyama K (2011) Evaluation of macular thickness and peripapillary retinal nerve fiber layer thickness for detection of early glaucoma using spectral domain optical coherence tomography. J Glaucoma 20(4):252–259. https://doi.org/10.1097/IJG.0b013e3181e079ed
Kurysheva NI, Parshunina OA, Shatalova EO, Kiseleva TN, Lagutin MB, Fomin AV (2017) Value of structural and hemodynamic parameters for the early detection of primary open-angle glaucoma. Curr Eye Res 42(3):411–417. https://doi.org/10.1080/02713683.2016.1184281
Kurysheva NI, Kiseleva TN, Hodak NA (2012) The study of bioelectricactivity and regional hemodynamics in glaucoma. Klinicheskaya oftalmologiya 3:91–94
Flammer J, Orgül S (1998) Optic nerve blood-flow abnormalities in glaucoma. Prog Retin Eye Res 17(2):267–289. https://doi.org/10.1016/s1350-9462(97)00006-2
Tobe LA, Harris A, Hussain RM et al (2015) The role of retrobulbar and retinal circulation on optic nerve head and retinal nerve fibre layer structure in patients with open-angle glaucoma over an 18-month period. Br J Ophthalmol 99(5):609–612. https://doi.org/10.1136/bjophthalmol-2014-305780
Martínez A, Sánchez M (2005) Predictive value of colour Doppler imaging in a prospective study of visual field progression in primary open-angle glaucoma. Acta Ophthalmol Scand 83(6):716–722. https://doi.org/10.1111/j.1600-0420.2005.00567.x
Mokbel TH, Ghanem AA (2011) Diagnostic value of color Doppler imaging and pattern visual evoked potential in primary open-angle glaucoma. J Clin Exp Ophthalmol 2(1). https://doi.org/10.4172/2155-9570.1000127
Jia Y, Wei E, Wang X et al (2014) Optical coherence tomography angiography of optic disc perfusion in glaucoma. Ophthalmology 121(7):1322–1332. https://doi.org/10.1016/j.ophtha.2014.01.021
Manalastas PIC, Zangwill LM, Saunders LJ et al (2017) Reproducibility of optical coherence tomography angiography macular and optic nerve head vascular density in glaucoma and healthy eyes. J Glaucoma 26(10):851–859. https://doi.org/10.1097/IJG.0000000000000768
Chen HS, Liu CH, Wu WC, Tseng HJ, Lee YS (2017) Optical coherence tomography angiography of the superficial microvasculature in the macular and Peripapillary areas in glaucomatous and healthy eyes. Invest Ophthalmol Vis Sci 58(9):3637–3645. https://doi.org/10.1167/iovs.17-21846
Kurysheva NI (2016) Macula in glaucoma: vascularity evaluated by OCT angiography. Res J Pharm, Biol Chem Sci 7(5):651–662
Xu H, Yu J, Kong X, Sun X, Jiang C (2016) Macular microvasculature alterations in patients with primary open-angle glaucoma: a cross-sectional study. Medicine (Baltimore) 95(33):e4341. https://doi.org/10.1097/MD.0000000000004341
Akil H, Chopra V, Al-Sheikh M et al (2017. bjophthalmol-2016-309816) Swept-source OCT angiography imaging of the macular capillary network in glaucoma [published online ahead of print, 2017 Aug 9]. Br J Ophthalmol. https://doi.org/10.1136/bjophthalmol-2016-309816
Shoji T, Zangwill LM, Akagi T et al (2017) Progressive macula vessel density loss in primary open-angle glaucoma: a longitudinal study. Am J Ophthalmol 182:107–117. https://doi.org/10.1016/j.ajo.2017.07.011
Holló G (2016) Intrasession and Between-Visit Variability of Sector Peripapillary Angioflow Vessel Density Values Measured with the Angiovue Optical Coherence Tomograph in Different Retinal Layers in Ocular Hypertension and Glaucoma [published correction appears in PLoS One. 2016 Dec 9;11(12 ):e0168231]. PloS One 11(8):e0161631. Published 2016 Aug 18. https://doi.org/10.1371/journal.pone.0161631
Park HL, Kim JW, Park CK (2018) Choroidal microvasculature dropout is associated with progressive retinal nerve fiber layer thinning in glaucoma with disc hemorrhage. Ophthalmology 125(7):1003–1013. https://doi.org/10.1016/j.ophtha.2018.01.016
Moghimi S, Zangwill LM, Penteado RC et al (2018) Macular and optic nerve head vessel density and progressive retinal nerve fiber layer loss in glaucoma. Ophthalmology 125(11):1720–1728. https://doi.org/10.1016/j.ophtha.2018.05.006
Yarmohammadi A, Zangwill LM, Diniz-Filho A et al (2016) Relationship between optical coherence tomography angiography vessel density and severity of visual field loss in glaucoma. Ophthalmology 123(12):2498–2508. https://doi.org/10.1016/j.ophtha.2016.08.041
Jia Y, Morrison JC, Tokayer J et al (2012) Quantitative OCT angiography of optic nerve head blood flow. Biomed Opt Express 3(12):3127–3137. https://doi.org/10.1364/BOE.3.003127
Wang X, Jiang C, Ko T et al (2015) Correlation between optic disc perfusion and glaucomatous severity in patients with open-angle glaucoma: an optical coherence tomography angiography study. Graefes Arch Clin Exp Ophthalmol 253(9):1557–1564. https://doi.org/10.1007/s00417-015-3095-y
Liu L, Jia Y, Takusagawa HL et al (2015) Optical coherence tomography angiography of the peripapillary retina in glaucoma. JAMA Ophthalmol 133(9):1045–1052. https://doi.org/10.1001/jamaophthalmol.2015.2225
Lee EJ, Lee KM, Lee SH, Kim TW (2016) OCT angiography of the peripapillary retina in primary open-angle glaucoma. Invest Ophthalmol Vis Sci 57(14):6265–6270. https://doi.org/10.1167/iovs.16-20287
Lévêque PM, Zéboulon P, Brasnu E, Baudouin C, Labbé A (2016) Optic disc vascularization in glaucoma: value of spectral-domain optical coherence tomography angiography. J Ophthalmol 2016:6956717. https://doi.org/10.1155/2016/6956717
Yarmohammadi A, Zangwill LM, Diniz-Filho A et al (2016) Optical coherence tomography angiography vessel density in healthy, glaucoma suspect, and glaucoma eyes. Invest Ophthalmol Vis Sci 57(9):OCT451–OCT459. https://doi.org/10.1167/iovs.15-18944
Chihara E, Dimitrova G, Amano H, Chihara T (2017) Discriminatory power of superficial vessel density and prelaminar vascular flow index in eyes with glaucoma and ocular hypertension and normal eyes. Invest Ophthalmol Vis Sci 58(1):690–697. https://doi.org/10.1167/iovs.16-20709
Hood DC (2017) Improving our understanding, and detection, of glaucomatous damage: an approach based upon optical coherence tomography (OCT). Prog Retin Eye Res 57:46–75. https://doi.org/10.1016/j.preteyeres.2016.12.002
Hood DC, Raza AS, de Moraes CG, Johnson CA, Liebmann JM, Ritch R (2012) The nature of macular damage in glaucoma as revealed by averaging optical coherence tomography data. Transl Vis Sci Technol 1(1):3. https://doi.org/10.1167/tvst.1.1.3
Bowd C, Tafreshi A, Zangwill LM, Medeiros FA, Sample PA, Weinreb RN (2011) Pattern electroretinogram association with spectral domain-OCT structural measurements in glaucoma. Eye (Lond) 25(2):224–232. https://doi.org/10.1038/eye.2010.203
May JG, Ralston JV, Reed JL, Van Dyk HJ (1982) Loss in pattern-elicited electroretinograms in optic nerve dysfunction. Am J Ophthalmol 93(4):418–422. https://doi.org/10.1016/0002-9394(82)90131-3
Bobak P, Bodis-Wollner I, Harnois C et al (1983) Pattern electroretinograms and visual-evoked potentials in glaucoma and multiple sclerosis. Am J Ophthalmol 96(1):72–83. https://doi.org/10.1016/0002-9394(83)90457-9
Wanger P, Persson HE (1983) Pattern-reversal electroretinograms in unilateral glaucoma. Invest Ophthalmol Vis Sci 24(6):749–753
Bach M, Hiss P, Röver J (1988) Check-size specific changes of pattern electroretinogram in patients with early open-angle glaucoma. Doc Ophthalmol 69(3):315–322. https://doi.org/10.1007/BF00154412
Parisi V, Miglior S, Manni G, Centofanti M, Bucci MG (2006) Clinical ability of pattern electroretinograms and visual evoked potentials in detecting visual dysfunction in ocular hypertension and glaucoma. Ophthalmology 113(2):216–228. https://doi.org/10.1016/j.ophtha.2005.10.044
Rejdak R, Toczołowski J, Kurkowski J et al (2003) Oral citicoline treatment improves visual pathway function in glaucoma. Med Sci Monit 9(3):PI24–PI28
Tong Y, Wang P, Xia Z, Xia X, Xu X (2009) Color pattern reversal visual evoked potentials in primary open angle and angle closure glaucoma. Zhong Nan Da Xue Xue Bao Yi Xue Ban 34(8):771–775. Chinese
Kurysheva NI, Maslova EV, Zolnikova IV, Fomin AV, Lagutin MB (2018) A comparative study of structural, functional and circulatory parameters in glaucoma diagnostics. PloS One 13(8):e0201599. Published 2018 Aug 23. https://doi.org/10.1371/journal.pone.0201599
Kurysheva NI, Maslova EV, Trubilina AV, Fomin AV, Likhvantseva VG, Lagutin MB (2017) OCT angiography and color doppler imaging in glaucoma diagnostics. J Pharm Sci Res 9(5):527–536
Investigators AGIS (2002) The advanced glaucoma intervention study (AGIS): 12. Baseline risk factors for sustained loss of visual field and visual acuity in patients with advanced glaucoma. Am J Ophthalmol 134(4):499–512. https://doi.org/10.1016/s0002-9394(02)01659-8
Leske MC, Heijl A, Hussein M et al (2003) Factors for glaucoma progression and the effect of treatment: the early manifest glaucoma trial. Arch Ophthalmol 121(1):48–56. https://doi.org/10.1001/archopht.121.1.48
Leske MC, Heijl A, Hyman L et al (2007) Predictors of long-term progression in the early manifest glaucoma trial. Ophthalmology 114(11):1965–1972. https://doi.org/10.1016/j.ophtha.2007.03.016
Miglior S, Zeyen T, Pfeiffer N et al (2005) Results of the European glaucoma prevention study. Ophthalmology 112(3):366–375. https://doi.org/10.1016/j.ophtha.2004.11.030
Chauhan BC, Mikelberg FS, Balaszi AG et al (2008) Canadian glaucoma study: 2. Risk factors for the progression of open-angle glaucoma [published correction appears in arch Ophthalmol. 2008 Oct;126(10):1364]. Arch Ophthalmol 126(8):1030–1036. https://doi.org/10.1001/archopht.126.8.1030
Kim SH, Lee EJ, Han JC, Sohn SW, Rhee T, Kee C (2016) the effect of diurnal fluctuation in intraocular pressure on the evaluation of risk factors of progression in normal tension glaucoma. PloS One 11(10):e0164876. Published 2016 Oct 24. https://doi.org/10.1371/journal.pone.0164876
De Moraes CG, Juthani VJ, Liebmann JM et al (2011) Risk factors for visual field progression in treated glaucoma [published correction appears in arch Ophthalmol. 2011 Jul;129(7):878]. Arch Ophthalmol 129(5):562–568. https://doi.org/10.1001/archophthalmol.2011.72
Musch DC, Gillespie BW, Niziol LM, Lichter PR, Varma R, CIGTS Study Group (2011) Intraocular pressure control and long-term visual field loss in the collaborative initial glaucoma treatment study. Ophthalmology 118(9):1766–1773. https://doi.org/10.1016/j.ophtha.2011.01.047
Medeiros FA, Sample PA, Zangwill LM, Bowd C, Aihara M, Weinreb RN (2003) Corneal thickness as a risk factor for visual field loss in patients with preperimetric glaucomatous optic neuropathy. Am J Ophthalmol 136(5):805–813. https://doi.org/10.1016/s0002-9394(03)00484-7
De Moraes CV, Hill V, Tello C, Liebmann JM, Ritch R (2012) Lower corneal hysteresis is associated with more rapid glaucomatous visual field progression. J Glaucoma 21(4):209–213. https://doi.org/10.1097/IJG.0b013e3182071b92
Bengtsson B, Leske MC, Yang Z, Heijl A, EMGT Group (2008) Disc hemorrhages and treatment in the early manifest glaucoma trial. Ophthalmology 115(11):2044–2048. https://doi.org/10.1016/j.ophtha.2008.05.031
Martínez A, Sanchez-Salorio M (2010) Predictors for visual field progression and the effects of treatment with dorzolamide 2% or brinzolamide 1% each added to timolol 0.5% in primary open-angle glaucoma. Acta Ophthalmol 88(5):541–552. https://doi.org/10.1111/j.1755-3768.2009.01595.x
Actis AG, Versino E, Brogliatti B, Rolle T (2016) Risk factors for primary open angle glaucoma (POAG) progression: a study ruled in Torino. Open Ophthalmol J 10:129–139. Published 2016 Apr 29. https://doi.org/10.2174/1874364101610010129
Leske MC, Wu SY, Nemesure B, Hennis A (2002) Incident open-angle glaucoma and blood pressure. Arch Ophthalmol 120(7):954–959. https://doi.org/10.1001/archopht.120.7.954
Gugleta K (2009) Vaskuläre Risikofaktoren beim Glaukom - Diagnostik [Vascular risk factors in glaucoma - diagnostics]. Praxis 98(4):201–207. https://doi.org/10.1024/1661-8157.98.4.201
Memarzadeh F, Ying-Lai M, Chung J, Azen SP, Varma R, Los Angeles Latino Eye Study Group (2010) Blood pressure, perfusion pressure, and open-angle glaucoma: the Los Angeles Latino eye study. Invest Ophthalmol Vis Sci 51(6):2872–2877. https://doi.org/10.1167/iovs.08-2956
Weinreb RN, Aung T, Medeiros FA (2014) The pathophysiology and treatment of glaucoma: a review. JAMA 311(18):1901–1911. https://doi.org/10.1001/jama.2014.3192
Ghahari E, Bowd C, Zangwill LM et al (2019) Association of macular and circumpapillary microvasculature with visual field sensitivity in advanced glaucoma. Am J Ophthalmol 204:51–61. https://doi.org/10.1016/j.ajo.2019.03.004
Hou H, Moghimi S, Zangwill LM et al (2020) Association between rates of retinal nerve fiber layer thinning after intraocular pressure-lowering procedures and disc hemorrhage. Ophthalmol Glaucoma 3(1):7–13. https://doi.org/10.1016/j.ogla.2019.11.002
Kamalipour A, Moghimi S, Hou H et al (2022) Multilayer macula vessel density and visual field progression in glaucoma. Am J Ophthalmol 237:193–203. https://doi.org/10.1016/j.ajo.2021.11.018
Kwon JM, Weinreb RN, Zangwill LM, Suh MH (2019) Parapapillary deep-layer microvasculature dropout and visual field progression in glaucoma. Am J Ophthalmol 200:65–75. https://doi.org/10.1016/j.ajo.2018.12.007
Li F, Lin F, Gao K et al (2022) Association of foveal avascular zone area with structural and functional progression in glaucoma patients. Br J Ophthalmol 106(9):1245–1251. https://doi.org/10.1136/bjophthalmol-2020-318065
Alm A, Bill A (1973) Ocular and optic nerve blood flow at normal and increased intraocular pressures in monkeys (Macaca irus): a study with radioactively labelled microspheres including flow determinations in brain and some other tissues. Exp Eye Res 15(1):15–29. https://doi.org/10.1016/0014-4835(73)90185-1
Lejoyeux, R., Benillouche, J., Ong, J., , et al. Choriocapillaris: fundamentals and advancements. Prog Retin Eye Res 2022;87:100997. https://doi.org/10.1016/j.preteyeres.2021.100997
Suh MH, Zangwill LM, Manalastas PI et al (2016) Deep retinal layer microvasculature dropout detected by the optical coherence tomography angiography in glaucoma. Ophthalmology 123(12):2509–2518. https://doi.org/10.1016/j.ophtha.2016.09.002
Lee SH, Kim TW, Lee EJ, Girard MJA, Mari JM (2020) Focal lamina cribrosa defects are not associated with steep lamina cribrosa curvature but with choroidal microvascular dropout. Sci Rep 10:6761. Published 2020 Apr 21. https://doi.org/10.1038/s41598-020-63681-6
Lee EJ, Kim TW, Kim JA et al (2019) Elucidation of the strongest factors influencing rapid retinal nerve fiber layer thinning in glaucoma. Invest Ophthalmol Vis Sci 60(10):3343–3351. https://doi.org/10.1167/iovs.18-26519
Jo YH, Shin JW, Song MK, Won HJ, Kook MS (2021) Baseline choroidal microvasculature dropout as a predictor of subsequent visual field progression in open-angle glaucoma. J Glaucoma 30(8):672–681. https://doi.org/10.1097/IJG.0000000000001853
Lee EJ, Kim TW, Kim JA, Lee SH, Kim H (2022) Predictive modeling of long-term glaucoma progression based on initial ophthalmic data and optic nerve head characteristics. Transl Vis Sci Technol 11(10):24. https://doi.org/10.1167/tvst.11.10.24
Kim SH, Park KH (2006) The relationship between recurrent optic disc hemorrhage and glaucoma progression. Ophthalmology 113(4):598–602. https://doi.org/10.1016/j.ophtha.2005.12.018
Kurysheva NI, Shatalova EO (2022) Parafoveal vessel density dropout may predict glaucoma progression in the long-term follow up. J Ophthalmol Res 5:150–166
Janulevičiene I, Ehrlich R, Siesky B, Nedzelskienė I, Harris A (2011) Evaluation of hemodynamic parameters as predictors of glaucoma progression. J Ophthalmol 2011:164320. https://doi.org/10.1155/2011/164320
Chen CL, Zhang A, Bojikian KD et al (2016) Peripapillary retinal nerve fiber layer vascular microcirculation in glaucoma using optical coherence tomography-based microangiography. Invest Ophthalmol Vis Sci 57(9):OCT475–OCT485. https://doi.org/10.1167/iovs.15-18909
Yarmohammadi A, Zangwill LM, Diniz-Filho A et al (2017) Peripapillary and macular vessel density in patients with glaucoma and single-Hemifield visual field defect. Ophthalmology 124(5):709–719. https://doi.org/10.1016/j.ophtha.2017.01.004
Lommatzsch C, Rothaus K, Koch JM, Heinz C, Grisanti S (2018) OCTA vessel density changes in the macular zone in glaucomatous eyes. Graefes Arch Clin Exp Ophthalmol 256(8):1499–1508. https://doi.org/10.1007/s00417-018-3965-1
Moghimi S, Bowd C, Zangwill LM et al (2019) Measurement floors and dynamic ranges of OCT and OCT angiography in glaucoma. Ophthalmology 126(7):980–988. https://doi.org/10.1016/j.ophtha.2019.03.003
Rao HL, Pradhan ZS, Weinreb RN et al (2017) Relationship of optic nerve structure and function to peripapillary vessel density measurements of optical coherence tomography angiography in glaucoma. J Glaucoma 26(6):548–554. https://doi.org/10.1097/IJG.0000000000000670
Mwanza JC, Budenz DL, Warren JL et al (2015) Retinal nerve fibre layer thickness floor and corresponding functional loss in glaucoma. Br J Ophthalmol 99(6):732–737. https://doi.org/10.1136/bjophthalmol-2014-305745
Belghith A, Medeiros FA, Bowd C et al (2016) Structural change can be detected in advanced-glaucoma eyes. Invest Ophthalmol Vis Sci 57(9):OCT511–OCT518. https://doi.org/10.1167/iovs.15-18929
Holló G (2019) Peripapillary capillary vessel density progression in advanced glaucoma: a case report. BMC Ophthalmol 19(1):2. Published 2019 Jan 5. https://doi.org/10.1186/s12886-018-1021-x
In JH, Lee SY, Cho SH, Hong YJ (2018) Peripapillary vessel density reversal after trabeculectomy in glaucoma. J Ophthalmol 2018:8909714. Published 2018 Jun 26. https://doi.org/10.1155/2018/8909714
Shin JW, Sung KR, Uhm KB et al (2017) Peripapillary microvascular improvement and lamina Cribrosa depth reduction after trabeculectomy in primary open-angle glaucoma. Invest Ophthalmol Vis Sci 58(13):5993–5999. https://doi.org/10.1167/iovs.17-22787
Holló G (2017) Influence of large intraocular pressure reduction on peripapillary OCT vessel density in ocular hypertensive and glaucoma eyes. J Glaucoma 26:e7
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kurysheva, N.I., Kim, V.Y., Kim, V.E. (2023). Does the Evaluation of Ocular Blood Supply Play a Role in Glaucoma Diagnostics and Prognosis of Progression?. In: Podbielska, H., Kapalla, M. (eds) Predictive, Preventive, and Personalised Medicine: From Bench to Bedside. Advances in Predictive, Preventive and Personalised Medicine, vol 17. Springer, Cham. https://doi.org/10.1007/978-3-031-34884-6_18
Download citation
DOI: https://doi.org/10.1007/978-3-031-34884-6_18
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-34883-9
Online ISBN: 978-3-031-34884-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)