Abstract
The demand for more food production and the pollution of ecosystems by pesticides is calling for sustainable methods to improve crop yields, such as the management of rhizobacteria that grow in the root zone. For instance, rhizobacteria induce systemic resistance against a large number of pathogens in plants. Here we review induced systemic resistance in plants with focus on plant immunity, systemic versus local resistance, molecular mechanisms, signaling, the role of salicylic acid, hormones and genes, and the control of crop diseases.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Plants
- Resistance
- Induced systemic resistance
- Signalling
- Hormones
- Plant-immunity
- Systemic acquired resistance
- Commercial products
7.1 History of Resistance
Defining induced resistance in the plants is a daunting task due to the absence of defense-dedicated cells (Ruano and Scheuring 2020). Some pioneer scientists define induced resistance as an enhanced expression of the natural defensive attitude of plants in the presence of pathogenic invaders. This defensive behaviour is aggravated by alien factors of diverse type and subsequent infection lead to the expression of deterrence (Edreva 2004). The concept of systemic resistance in the plant is more than a century old. Beauverie published a paper titled, “Testing the immunization of plants against fungal diseases”. He discovered that begonia plants become more resistant to vigorously virulent strains of Botrytis cinerea fungus when a feebly virulent strain of this fungus (Botrytis cinerea) is inoculated to the begonia plants (Beauverie 1901). In the same year, in a different study, titled “Cryptogamic diseases of plants” Ray also indicated that exposure to various external or alien factors/pathogens; impart immunity to plants against different invaders. This provocation enhances the explicit expression of the natural defense mechanism of plants (Ray 1901). Thus, both of them, in independent studies proposed, plants previously exposed to a pathogen could better withstand second exposure.
Further, Chester in his famous study summarised different reports regarding the existence of various induced disease resistance phenomena in plants and gave the first-ever generic concept of plant defense mechanism (Chester 1933). J. Kuć is widely known as “the father of induced resistance research”. He gave biochemical evidence of induced resistance in the 1960s. He explained the phenomenon of induced systemic resistance with the help of phenylalanine (amino acid derivative). He studied the effects of phenylalanine regarding its resistance imparting properties against apple scab disease (Venturia inaequalis) (Williams and Kuc 1969). Kuc and co-workers became the torch-bearer in the area of induced resistance and its use as a method in plant defense mechanisms (Dalisay and Kuć 1955). After this initiation, induced systemic resistance phenomenon had been widely applauded and promoted by numerous authors from all around the globe (Benhamou and Picard 1999; Bokshi et al. 2003; Cohen 2001; Schönbeck et al. 1993; Gozzo 2003; Kessmann et al. 1994; Schneider et al. 1996; Soylu et al. 2003).
The terminology “induced systemic resistance” was envisaged specifically for soil-borne non-pathogenic bacteria; when these non-harmful rhizobacteria infect roots, the resistance was found to be induced in the leaves of the infected plants. This newly introduced type of defense system was unveiled in Arabidopsis thaliana plants. Roots of a few A. thaliana plants were inoculated with the strain of Pseudomonas fluorescens (non-pathogenic). Leaves of the plant in reference became somewhat resistant to Pseudomonas syringae (bacterial leaf pathogen) after a one-time exposure to infection (Pieterse 1998). Further, the induced systemic resistance involving non-pathogenic bacteria also demonstrates resistance against bacteria, viruses, and fungi in cucumber, tomato, tobacco, Arabidopsis, bean, and radish (van Loon et al. 1998). Later on, systemic resistance was characterized as plants’ ability to recall past experiences and as an example of “plant memory” (Conrath 2006). A variety of biotic and abiotic agents was found to be the reason behind the induction of such resistance. The resulting broad-spectrum and long-lasting resistance was called by different terms for example “plant immunity”, “resistance displacement”, “acquired physiological immunity” and “induced system resistance” (Conrath 2006).
7.2 Plant Systemic Immunity
Understanding of plant immunity mechanism is still in its infancy. Scientists are not certain of the underneath mechanisms involved in Induced systemic resistance for many plant species, plant diseases, or pests of these plants, despite the elucidation of the presence and identification of several pathways and chemical signals related to induced systemic resistance. For example, only a few studies are available regarding the resistance mechanisms involved in plant viruses (Satish et al. 2019). But it is well established that like animal immunity mechanisms, plant immunity also has layered characteristics. In the plant fraternity, the first line of defense i.e., pathogen-associated molecular patterns-triggered immunity is composed of pattern recognition (Boller and Felix 2009; Zipfel 2009). This shield prevents most potential enemies’ invasion. Further, the component of the second line of defense is a bit different than the components of the first line. For instance, Nucleotide-binding leucine-rich repeat receptor proteins [similar to Resistance (R) gene] is part of the second line of defense in plants. These resistance proteins identify specific effector molecules of an attacker, causing Effector-triggered immunity. This second line of defense is genetically more specific and generally followed by apoptosis in order to prevent further infection. The programmed killing of infected cells and extensive host cell reprogramming is part of the local immune response against pathogens. The first line and second line of defense, generally pave the way for enhanced defensive capacity in plant parts that are still not damaged by the invader. When such induced resistance is developed in a distant location from point of infection, resistance is known as systemic resistance.
7.3 Systemic Acquired Resistance Versus Local Acquired Resistance
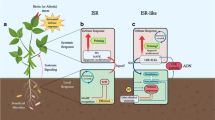
Depending upon the area and method of its expression, invader-derived immunity in plants might be classified as ‘local acquired resistance’ and ‘systemic acquired resistance’. Carefully conducted laboratory experiments with the Tobacco Mosaic Virus system helped Ross and co-workers in coining the terminology of local acquired resistance and systemic acquired resistance (Ross 1961a, b). In this historic experiment, leaves of the Nicotiana tabacum (cv. Xanthi-nc) were inoculated with the tobacco mosaic virus. After this tobacco mosaic virus infection, small necrotic abrasions protruded on the leaves. After a few days, the same leaves were again infected with Tobacco Mosaic Virus but this time smaller-sized and less numerous lesions appeared. Hence severity of infection was reduced to a great extent.
Local acquired resistance was defined by an experiment involving the secondary infection to the nearby leaves where the first inoculation was done. It was found that due to previous exposure to the inoculation, leaves in close vicinity also got immune. In the same system, Ross and colleagues infected the leaves, which were not exposed to the infection previously. They observed that these leaves also showed signs of resistance. They term this phenomenon as ‘local acquired resistance’ (Ross 1961a). Ross successfully induced resistance to tobacco mosaic virus in the tobacco leaves (which were distantly located on the same plant), by inoculation (primary) of an aerial part of the plant with the virus. They refer to this phenomenon as ‘systemic acquired resistance’ (Ross 1961b).
This systemic immunity response is known by different names depending upon the invader nature (Fig. 7.1) for example, if elicitor is a pathogen then immunity imparted will be known as ‘pathogen-induced systemic acquired resistance’ if the infection is caused by mycorrhiza ‘mycorrhiza-induced resistance’, while in case of herbivores, name of resistance will be ‘herbivore-induced resistance’, and if immunity is triggered by a non-pathogenic invader or beneficial soil-borne microbes then it will be acknowledged as ‘induced systemic resistance’.
7.4 Induced Systemic Resistance
Induced plant resistance is a broad terminology for the defense mechanism of plants, evoked by infection or chemical or biological infections. Despite knowing systemically acquired resistance in plants, the concept of induced systemic resistance became apparent very late. During the early years of the 1990s, three research groups independently demonstrated that root colonization by certain non-pathogenic bacteria leads to enhancement in the defense capabilities of plants against pathogens/invaders (Alström 1991; van Peer et al. 1991; Wei et al. 1991). It has been demonstrated that after root infection by Pseudomonas fluorescens WCS417r, aerial plant parts attained an amplified immunity against Fusarium oxysporum infection and produced significantly more antimicrobial phytoalexins (van Peer et al. 1991). Thus, van Peerandco-workers gave testimony that P. fluorescens infection to the root system can provide systemic resistance in plants. Similarly, Wei and co-worker and Alström explicated enhanced plant immunity after infection of Pseudomonas and Serratia PGPR strains on cucumber and PGPR strain P. fluorescens S97 infection on common bean roots, respectively (Wei et al. 1991; Alström 1991). Among these pioneer research teams in the induced systemic resistance area, gave evidence about the spatial difference between the site of plant growth-promoting rhizobacterial infection and challenging pathogen (Wei et al. 1991; van Peer et al. 1991). Thus, after rigorous contemplation concept of induced systemic resistance came to the scientific sphere and the search for causing agents begun.
Different research groups suggested that induced resistance can be triggered by various elicitors for example; avirulent forms of pathogens or by virulent pathogens under certain environmental conditions, non-pathogens, chemicals, incompatible races of pathogens, etc. After the establishment of the correlation between bacteria and induced systemic resistance, rhizo-fungi (Trichoderma spp. or Piriformospora indica) were also shown to have a similar impact on plant immunity (Boller and Felix 2009; van Peer et al. 1991; Wei et al. 1991). Induced immunity provides enhanced resistance against a broad spectrum of invaders, for example, foliar, root and fruit pathogen, parasitic plants, and pests (even in some cases, invertebrates such as nematodes). Inoculation of A. thaliana roots by Pseudomonas fluorescens (WCS417r) bacteria, insulate the plants from various other invaders (bacteria, fungus, and oomycete). Various research groups demonstrated this induced resistance against bacteria (for example, bacterial leaf pathogens P. Syringae pv. tomato and Xanthomonas campestris pv. Armoraciae), fungi (for example; root-infecting fungi – Fusarium oxysporum f.sp. raphani, leaf infecting fungi – Alternaria brassicicola), and the oomycete leaf pathogen (Phytophthora parasitica) (Pieterse 1996; Ton et al. 2002; van Wees et al. 1997). A comprehensive list of induced systemic resistance-inducing beneficial microbes is given in Table 7.1.
7.5 Molecular Mechanism of Induced Systemic Resistance
Induced resistance in plants comprises of an intensified expression of resistance against several invaders simultaneously. This is a plants’ way to avoid infection from plant pathogens. Both induced systemic resistance and systemic acquired resistance lead to resistance against a wide range of invaders hence prima facie the mechanism of both was assumed to be similar. But many factors indicated otherwise; for example, salicylic acid is both necessary and sufficient for systemic acquired resistance whereas induced systemic resistance can work without accumulation of salicylic acid and is reliant on jasmonic acid and ethylene reaction pathways in plants (Yuan et al. 2019).
In the case of induced systemic resistance, no damage/localized necrosis is involved, thus evoking factors generated by induced systemic resistance-triggering bacteria are generally not similar to elicitor molecules produced by pathogenic invaders (Ebel and Mithöfer 1998). In an interesting study, it was revealed that induced systemic resistance-engineered P. Fluorescens treated Raphanus raphanistrum plant, did not amass pathogenesis related proteins, even though these plants exhibit amplified immunity against wilt disease caused by Fusarium. But if we take cognizance of the mechanism of systemic acquired resistance, pathogen related protein accumulation is indispensable (Hoffland et al. 1995). A brief description of the differences between systemic acquired resistance and induced systemic resistance is given in Table 7.2.
Despite having many differences induced systemic resistance and systemic acquired resistance pathway converge at the NPR1 protein (Cao 1994). NPR1 protein is downstream of the salicylic acid in the systemic acquired resistance pathway, whereas in induced systemic resistance it is situated downstream of ethylene response. This protein is found to be necessary for both systemic acquired resistance and induced systemic resistance as mutant npr1 plants are unable to express induced systemic resistance after exposure to WCS417. Hence, it has been proved that NPR1 protein might be involved in a critical reaction in achieving the evoked state in both the cases of systemic acquired resistance and induced systemic resistance. But perhaps in the mechanism after NPR1, the signaling reactions and pathways might become deviated as pathogen related proteins are not accumulated in the case of induced systemic resistance (Fig. 7.2). A deep insight into the induced systemic resistance mechanism can be achieved if we explore three main and distinctive steps of induced systemic resistance i.e., induction, signaling, and expression.
7.5.1 Induction
During initial research findings, salicylic acid produced by bacteria was considered to be the inducing factor in the case of bean (De Meyer and Höfte 1997), tobacco, and tomato (De Meyer et al. 1999), but later on, many other elicitors were shown to be effective in triggering induced systemic resistance. In the light of limited knowledge present in the scientific literature about bacterial determinants that trigger induced systemic resistance, few important conclusions can be made. For example, crude microbial cell wall preparations of the WCS358 (rhizobacterial strain) can invoke induced systemic resistance if absolute components of this strain are inoculated to roots of A. thaliana plants (Bakker et al. 2003; Meziane et al. 2005). A brief list of induced systemic resistance triggering agents has been enlisted in Table 7.3.
It has been identified that few ‘plant growth-promoting fungi’ also have cell components that explicitly behave as an elicitor of defense mechanisms (Conrath 2006). For example, Sm1 from Trichoderma virens is a protein that has a particular defense-eliciting function (Djonovic et al. 2007). Not only this, few cellulases and xylanases also act as defense-elicitor. Further, the concept of “differential induction of systemic resistance” emerged in various experimental reports related to different plant species or ecotypes (Ton et al. 1999; van Wees et al. 1997). According to the notion of differential induction pattern, few beneficial microbe strains have an impact on a variety of plant species i.e., they can elicit systemic resistance in various plant species, while other bacteria exhibit more specificity i.e., they can impact the defense mechanism of only a few plant species. This differential induction pattern indicates bacteria act according to a species-specific recognition pattern. It is proposed that bacteria recognize receptors, on the root surface before eliciting induced systemic resistance (van Loon et al. 1998). For example, WCS374 strain elicits induced systemic resistance in Raphanus raphanistrum but not in Arabidopsis thaliana, whereas WCS358 elicits induced systemic resistance in A. thaliana, Phaseolus vulgaris, and Solanum lycopersicum, but not in Daucus carota or Raphanus raphanistrum (Duijff et al. 1993; Leeman et al. 1995; van Wees et al. 1997; Meziane et al. 2005).
7.5.2 Priming of Infected Plants
Preparation of the whole plant to better resist the invader’s attack is called priming. In response to the primary attack of the induced systemic resistance-inducing invader, some of the induced systemic resistance-related compounds are produced in uninfected plant tissues. But the explicit induced systemic resistance-associated compounds are expressed only after secondary infection. This second exposure is characterized by comparatively faster and stronger defense responses. It is often not possible to assess if a plant is primed, in the absence of the invader. Only after the secondary exposure to the invaders’ attack, amplified/altered transcriptional changes in a plant can be observed.
After extensive study of priming, it has been ascertained that priming enhances the fitness quotient of a plant, and the advantage of priming seems to be greater than its costs during invader combat. This indicates that priming serves as a survival mechanism of the plant against damage caused in an adverse environment. During conditioning/sensitization, augmentation of structural barriers and elevation in several miRNAs of various transcription factors genes is observed.
In a study, it has been shown that strain WCS417r (P. fluorescens) inoculated A. thaliana plant display crucial changes. On the entry of Hyaloperonospora arabidopsidis in A. thaliana, an enhanced frequency of callose accumulation was witnessed, which efficiently arrests the invasion of the pathogen (van der Ent et al. 2009a). Callose is a polysaccharide comprised of β-1,3-glucan with β-1,6-branches. Callose is generally present in the cell walls of higher plants. Further, the same study threw some light on the fact that this phenomenon of ingression hindrance is controlled by abscisic acid (plant hormone). In a different but related study of structural barriers by priming, stomata closure to a significant extent has been reported after a second exposure to infection. In this study, A. thaliana leaves were inoculated by P. syringae. This primary exposure of P. syringae results in the infection of the FB17 strain of Bacillus subtilis to the roots of A. thaliana FB17 triggered induced systemic resistance, which provides immunity to the unexposed plant parts from P. syringae infection (Rudrappa et al. 2008). This plant growth promoting rhizobacteria-induced priming indicated a potent structural barrier that can delay/inhibit the progression of disease in induced systemic resistance-primed plants.
Along with structural barriers, transcriptional factors play an important role during priming. This induced systemic resistance-involved transcription factors often remain inactive in plants which not exposed to the invaders, but upon the cognizance of pathogen/pest presence, these transcription factors provide the plant accelerated defense response. Several members of the APETALA2/ethylene-responsive factor family are predicted to have a crucial role in the regulation of jasmonic acid- and ethylene-dependent defences (Memelink 2009). Priming effects are an integral part of the induced systemic resistance mechanism and can be elicited by biological, chemical-induced systemic resistance inducers. After the primary infection/inoculation, responses such as cell wall lignification or phytoalexin synthesis occur more expeditiously. The in-depth molecular mechanisms behind priming and its significance in the overall plant immunity are still not explored to the fullest.
7.5.3 Signalling in Beneficial Microbe-Induced Systemic Resistance
Signaling in induced systemic resistance is a considerably complex phenomenon. For example, several induced systemic resistance-evoking rhizobacterial strains produce salicylic acid, whereas others do-not. Some rhizobacteria are capable of producing salicylic acid on minimal media in-vitro in the absence of iron (Fig. 7.3). If the soil confronts a similar environment in the rhizosphere, these bacteria are expected to show analogous expression. But in nature i.e., under in-vivo conditions, salicylic acid is not released into the rhizosphere and is destined to the salicylic acid-containing siderophore. Thus, induced systemic resistance induction can happen only when the determinant agent is siderophore and no resistance happens if siderophore is not able to act as elicitor (Aznar and Dellagi 2015).
7.5.4 Expression of Induced Systemic Resistance
Induced systemic resistance articulation is pretty much analogous to systemic acquired resistance, this is the reason why the discovery of induced systemic resistance took too long while the mechanism of systemic acquired resistance had been already explored to a great extent. Due to overlapping responses and few proteins like NPA1, finding induced systemic resistance-specific mechanisms has become a challenge to the scientific community. Both, the induced systemic resistance and systemic acquired resistance, reduce the disease severity and in some cases, the extent of invasion in plants is minimized along with depreciated growth of the pathogen itself. These signs are a testament to the enhanced immunity of plants.
Though the plant seems to be well equipped against the invader, expression of neither induced systemic resistance nor systemically acquired resistance saves plants thoroughly from all types of infections. But the reduction in disease development generally saves a crop to some extent from the natural attack of invaders. As stated earlier, the complete mechanism of induced systemic resistance has not been discovered yet thus the entire conclusions drawn are based on the limited information available. Systemic acquired resistance has characteristic pathogenesis-related genes but the search for characteristic protein for induced systemic resistance was not able to produce substantial results. Further, there is no major shift found in the defense-related gene expression during induced systemic resistance activation (Heil 2002).
Using, transcriptome analyses (cDNA microarrays technique) Verhagen and co-workers confirmed the hypothesis that beneficial microbes interfere in plant transcriptome to only a negligible extent (Verhagen et al. 2004). For example, WCS417-infected Arabidopsis plants were taken for the analysis of over 8000 genes. Out of these whooping 8000 genes, only 102 genes in the roots exhibit changes in expression and no gene showed the change in expression in leaves at all, despite full-blown expression of induced systemic resistance by leaves. As the studies to date are in primitive stage, we are not able to draw any concrete conclusion regarding players involved in induced systemic resistance. We can assume only that, plants must possess some undiscovered defense-related products, which impart resistance to plants against broad spectrum invaders. Transgenic plants with altered enzyme activities, exposed to induced systemic resistance-evoking invader can help in exploring the molecular mechanism behind induced systemic resistance. But so far, no such successful studies are known to the scientific world.
7.6 Is Salicylic Acid Necessary for Induced Systemic Resistance?
Many studies have been conducted to ascertain if induced systemic resistance-triggering strains cause activation of the salicylic acid-independent pathway. Association of pathogen-related proteins with induced systemic resistance and nahG gene mutant studies clear the air to a great extent. Induced activation study on tobacco plant demonstrated that induction of induced systemic resistance by 7NSK2 strain was salicylic acid independent and there was no sign of enhanced production of the pathogenesis related PR-1 protein along with the enhanced immunity (De Meyer et al. 1999). Interpretation of results revealed that salicylic acid produced in this case is insufficient for generating pathogen-related proteins but certainly enough for eliciting induced systemic resistance mechanism. Further, Press and co-workers (1997) demonstrated that mutants of Serratia marcescens, which could not produce salicylic acid, were able to induce defense mechanisms in tobacco against P. syringae and in cucumber against C. Orbiculare. In a different but related study, van Wees et al. (1997) provide experimental proof that WCS358 does not produce salicylic acid but it can elicit systemic resistance in A. thaliana.
On the other hand, they also proved that WCS374 when invade, A. thaliana can produce salicylic acid in-vitro but this incidence does not provoke any induced systemic resistance (van Wees et al. 1997) or elicit induced systemic resistance in a “Salicylic acid-independent” pathway (Press et al. 1997). In the light of the above-mentioned evidence and observation, it can be concluded that salicylic acid production by rhizobacteria is not imperatively required for the expression of systemic resistance.
7.7 Role of Hormones in the Signaling of Induced Systemic Resistance
Jasmonic acid and ethylene are known to be the central players in induced systemic resistance signaling (Fig. 7.4). In the series of events where beneficial microbes (present in the soil) impart salicylic acid-independent systemic resistance in plants, jasmonic acid and ethylene play crucial role (Audenaert et al. 2002; Hossain et al. 2008; Pieterse 1998, Korolev et al. 2008; Ryu et al. 2004; Stein et al. 2008, Ahn et al. 2007; De Vleesschauwer et al. 2008; Weller et al. 2012, Hase et al. 2008; van der Ent et al. 2009b; Yan et al. 2002). The essentiality of jasmonic acid in the induced systemic resistance pathway is ascertained using jar1 mutant plants. jar1 gene is supposed to encode a jasmonic acid amino acid synthetase, required for jasmonic acid signaling activation. Arabidopsis plant mutants; jar1, jin1, and coi1 are found to be defective in induced systemic resistance signaling (Kloepper et al. 2004; Pieterse 1998) whereas ethylene signaling mutants etr2, ein1, ein3, and eir1, were found non-functional in WCS417r-induced systemic resistance strain of P. fluorescens (Pieterse 1998; Kloepper et al. 2004; Pozo et al. 2008). A jar1–1 plant (Jasmonic acid response mutant) and the etr1–1 plant (ethylene insensitive mutant) were checked for the capability to showcase induced systemic resistance in A. thaliana. Even after inoculation of roots by WCS417r strain of Pseudomonas fluorescens both mutants were not show-casing any sign of increased resistance against P. Syringae pv. Tomato (Pieterse 1998). Moreover, exogenous application of jasmonic acid and ethylene also often results in an elevated level of resistance against Pseudomonas syringae. This leads to the conclusion that both of the hormones are equally essential in induced systemic resistance mechanism, a defect in the synthesis mechanism of any one of them hamper the induced systemic resistance expression.
Further, Pieterse et al. (1998) postulated that jasmonic acid and ethylene components are successively engaged in the induced systemic resistance pathway, which causes deeper insight into the essentiality of jasmonic acid and ethylene simultaneously. Regarding jasmonic acid and ethylene, van Wees et al. (1999) reached an interesting conclusion during a study. They suggested jasmonic acid and ethylene involvement in induced systemic resistance have relation to enhanced sensitivity to these hormones and the production level of these concerned hormones is not enhanced during induced systemic resistance response. To prove this point, van Wees et al. (1999) examined the expression of a combination of jasmonic acid and ethylene-responsive genes (i.e., PDF1.2, VSP, LOX1, LOX2, PAL1, CHI-B, and HEL) in A. thaliana plants expressing Pseudomonas fluorescens WCS417r-ISR. But there were no signs of change in the regulation pattern of jasmonic acid and ethylene genes in inoculated plants, neither locally or systemically. Hence it was suggested that production levels of either jasmonic acid or ethylene have little/no role in resistance achieved. Further, Pieterse et al. (1998) using transgenic line S-12 confirmed that induced systemic resistance involved enhanced sensitivity of ethylene and jasmonic acid rather than elevated production.
7.8 Genes Involved in the Induced Systemic Resistance Mechanism
The repertoire of genes involved in the induced systemic resistance mechanism is neither complete nor well understood (Fig. 7.5). Till now the role of transcription co-regulator of pathogen-related genes (for example, NPR1) could not be ascertained in the case of induced systemic resistance, because in the induced systemic resistance mechanism there is no accumulation of pathogen-related genes. But it is quite clear that the induced systemic resistance mechanism cannot take place in the absence of NPR1 (Pieterse 1998). Apart from systemic acquired resistance and induced systemic resistance, plant growth-promoting rhizobacteria, and plant growth-promoting fungi also witness the crucial role of NPR1 (Iavicoli et al. 2003; Ahn et al. 2007; Hossain et al. 2008). Further, in contrast to nuclear function in the case of systemic acquired resistance, many pieces of evidence indicate the cytoplasmic function of NPR1 protein in the case of ethylene/jasmonic acid signaling (Pieterse et al. 2012; Ramírez et al. 2010; Spoel 2003). The sequence of induced systemic resistance signaling events established that the role of NPA1 is downstream that of ethylene and jasmonic acid.
Genes involved in the induced systemic resistance mechanism. Solid purple lines mark established interactions; dotted purple lines are indicative of hypothetical inter-connections. A visibly large arrow indicates long-distance translocation of molecular signals. MAMP microbe-associated molecular pattern, ABA abscisic acid, ET Ethylene, JA jasmonic acid, PRR pattern-recognition receptor, PTI pattern-triggered immunity, SA salicylic acid, and TF transcription factor
In the quest of searching role of various genes in the mechanism of induced systemic resistance, induced systemic resistance was induced chemically in wild-type Arabidopsis plants by external application of 1-aminocyclopropane-1-carboxylic acid. 1-aminocyclopropane-1-carboxylic acid was capable of inducing induced systemic resistance in the jar1 mutant plants. This hints towards the fact that ethylene is involved in the reaction after the jasmonic acid-involved signaling reaction. Contrarily, ‘methyl jasmonate’ is not capable of triggering induced systemic resistance in the plants with the mutated etr1 gene. Thus, receptivity to jasmonic acid and ethylene are structured in precedence, where jasmonic acid is required first. Neither ‘methyl jasmonate’ nor ‘1-aminocyclopropane-1-carboxylic acid’ could trigger induced systemic resistance in the plants with mutated npr1 genes. This indicates the requirements for both jasmonic acid and ethylene upstream of NPR1 in the signaling pathway. Hence it is very much clear that NPA1 has a crucial role not only in the accumulation of pathogen-related genes in the case of systemic acquired resistance but also in the case of ethylene/jasmonic acid signaling pathways.
As NPR1 is common and inseparable to both systemic acquired resistance and induced systemic resistance, it is a matter of scientific interest to know, how the selection of downstream reaction takes place. A major challenge in this regard is the identification of components necessary for signaling in the induced systemic resistance and systemic acquired resistance so that NPR1-dependent defense gene activation can be ascertained. Along with NPR1, another most important gene involved in induced systemic resistance is MYB72 (Segarra et al. 2009). This is the regulator of induced systemic resistance, which can be identified during early infection events in roots.
7.9 Controlling Crop Diseases Using Induced Resistance
Induced systemic resistance in plants is a long-lasting mechanism but the main hindrance in using this phenomenon as an alternative to available disease management programs is that it is generally not complete. Along with this, many of the induced systemic resistance-inducing agents minimize disease impact by 20–70% only. In the presence of highly specific, easily available, and immensely effective chemical reagents, the use of non-specific and less effective plant resistance inducers does not seem to be a lucrative and easily acceptable idea by the average farmer. Using non-toxic plant resistance inducers in agriculture to combat plant diseases is a very advantageous concept. It has the potential to reduce the use of conventional pesticides hence their indiscriminate addition to the environment.
Further, by easing the financial burden on consumers and small/marginal farmers, plant resistance inducers could become a potential product for use in modern agriculture. Plant resistance inducers might also be easily used in combination with organisms used as “biocontrol agents”, in comparison to conventional pesticides. In addition to this, induced systemic resistance induction might prolong the effective time of resistance (R) genes. From the economic perspective, some of the plant resistance inducer compounds are relatively cheaper than chemicals available in the market, for example, Probenazole (commercially available as Oryzemate) was the first plant resistance inducers. It was registered in Japan as a chemical resistance activator in 1975 (Iwata et al. 2004). Since then, many plant resistance inducers have been listed for commercial use. Some commercialized plant resistance inducers popular in the market are as follows (Table 7.4).
7.10 Conclusion
Systemically induced resistance expressed itself through elevated defense response upon the attack of the invader. The discovery of systemic acquired resistance dates back to the eighteenth century, while induced systemic resistance is newly discovered and still not completely explored by the scientific community. Systemic acquired resistance and induced systemic resistance, both exhibit resistances against invaders, but the major difference is that systemic acquired resistance negatively affects plant growth while the induced systemic resistance has plant growth-promoting properties. Though, the extent of correlation between plant growth and induced systemic resistance cannot be established with certainty. Both induced systemic resistance and systemic acquired resistance can act independently as well as can have an additive effect. This in-built plant immunity after induction can reduce the plant disease to some extent but it will take scientific time and effort to replace chemical methods of pest control with induced systemic resistance and systemic acquired resistance.
Induced disease resistance i.e., induced systemic resistance and systemic acquired resistance are good and attractive solution against potential environment degrading chemical agents. The molecular mechanism behind systemic acquired resistance is well discovered but molecular mechanism detail behind the induced systemic resistance is still the bottleneck. As systemic acquired resistance and induced systemic resistance pathways to act independently as well as additively, the experimental revelation of induced systemic resistance molecular biology can be proved instrumental in the development of an environment-friendly crop protection method. By exploiting the unique and natural plant potential to contest against pathogens, the induced systemic resistance might help in minimizing the use of toxic and eventually extremely harmful chemicals for plant ailment control.
Due to the least harmful nature, induced systemic resistance could be seen as a substitutive, non-genetically modified organism, non-traditional and eco-friendly approach for the protection of plants against diseases. Thus, induced systemic resistance can be envisaged as one of the foundation stones of the major pillar of sustainable agriculture. This unique and inherent plant power to combat pathogens can be exploited as an alternative, non-conventional, non-biocidal, and eco-friendly approach for plant protection, sustainable agriculture, and the welfare of humanity at large.
Abbreviations
- ISR:
-
Induced systemic resistance
- NPR1:
-
Natriuretic peptide receptor1
- PR:
-
Pathogenesis related
- SAR:
-
Systemic acquired resistance
References
Ahn IP, Lee SW, Suh SC (2007) Rhizobacteria-induced priming in Arabidopsis is dependent on ethylene, jasmonic acid, and NPR1. Mol Plant Microbe Int 20:759–768. https://doi.org/10.1094/MPMI-20-7-0759
Alabouvette C, Olivain C, Migheli Q, Steinberg C (2009) Microbiological control of soil-borne phytopathogenic fungi with special emphasis on wilt-inducing Fusarium oxysporum. New Phytol 184:529–544. https://doi.org/10.1111/j.1469-8137.2009.03014.x
Alkooranee JT, Aledan TR, Ali AK, Lu G, Zhang X, Wu J, Fu C, Li M (2017) Detecting the hormonal pathways in oilseed rape behind induced systemic resistance by Trichoderma harzianum TH12 to Sclerotinia sclerotiorum. PLoS One 12:e0168850. https://doi.org/10.1371/journal.pone.0168850
Alström S (1991) Induction of disease resistance in common bean susceptible to halo blight bacterial pathogen after seed bacterization with rhizosphere pseudomonads. J Gen Appl Microbiol 37:495–501. https://doi.org/10.2323/jgam.37.495
Audenaert K, Pattery T, Cornelis P, Höfte M (2002) Induction of systemic resistance to Botrytis cinerea in tomato by Pseudomonas aeruginosa 7NSK2: role of Salicylic Acid, Pyochelin, and Pyocyanin. Mol Plant Microbe Int 15:1147–1156. https://doi.org/10.1094/MPMI.2002.15.11.1147
Aznar A, Dellagi A (2015) New insights into the role of siderophores as triggers of plant immunity: what can we learn from animals? J Exp Bot 66:3001–3010. https://doi.org/10.1093/jxb/erv155
Bakker PAHM, Ran LX, Pieterse CMJ, van Loon LC (2003) Understanding the involvement of rhizobacteria-mediated induction of systemic resistance in biocontrol of plant diseases. Can J Plant Pathol 25:5–9. https://doi.org/10.1080/07060660309507043
Bakker PAHM, Pieterse CMJ, van Loon LC (2007) Induced systemic resistance by Fluorescent Pseudomonas spp. Phytopathology 97:239–243. https://doi.org/10.1094/PHYTO-97-2-0239
Beauverie J (1901) Essaisd’immunisation des végétauxcontre les maladies cryptogamiques. C R Acad Sci Ser III:107–110
Beneduzi A, Ambrosini A, Passaglia LMP (2012) Plant growth-promoting rhizobacteria (PGPR): their potential as antagonists and biocontrol agents. Genet Mol Biol 35:1044–1051. https://doi.org/10.1590/S1415-47572012000600020
Benhamou N, Picard K (1999) La résistanceinduite : une nouvelle stratégie de défense des plantescontre les agentspathogènes. Phytoprot 80:137. https://doi.org/10.7202/706189ar
Bokshi AI, Morris SC, Deverall BJ (2003) Effects of benzothiadiazole and acetylsalicylic acid on beta-1,3-glucanase activity and disease resistance in potato. Plant Pathol 52:22–27. https://doi.org/10.1046/j.1365-3059.2003.00792.x
Boller T, Felix G (2009) A renaissance of elicitors: perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Ann Rev Plant Biol 60:379–406. https://doi.org/10.1146/annurev.arplant.57.032905.105346
Cameron DD, Neal AL, van Wees SCM, Ton J (2013) Mycorrhiza-induced resistance: more than the sum of its parts? Trends Plant Sci 18:539–545. https://doi.org/10.1016/j.tplants.2013.06.004
Cao H (1994) Characterization of an Arabidopsis mutant that is nonresponsive to inducers of systemic acquired resistance. Plant Cell 6:1583–1592. https://doi.org/10.1105/tpc.6.11.1583
Chester KS (1933) The problem of acquired physiological immunity in plants. Q Rev Biol 8:275–324
Cohen Y (2001) The BABA story of induced resistance. Phytoparasitica 29:375–378. https://doi.org/10.1007/BF02981855
Conrath U (2006) Systemic acquired resistance. Plant Sign Behav 1:179–184. https://doi.org/10.4161/psb.1.4.3221
Dalisay RF, Kuć J (1955) Persistence of induced resistance and enhanced peroxidase and chitinase activities in cucumber plants. Physiol Mol Plant Pathol 47:315–332
De Meyer G, Höfte M (1997) Salicylic acid produced by the rhizobacterium Pseudomonas aeruginosa 7NSK2 induces resistance to leaf infection by botrytis cinerea on bean. Phytopathology 87:588–593. https://doi.org/10.1094/PHYTO.1997.87.6.588
De Meyer G, Audenaert K, Höfte M (1999) Pseudomonas aeruginosa 7NSK2-induced systemic resistance in Tobacco depends on in planta salicylic acid accumulation but is not associated with PR1a expression. Eur J Plant Pathol 105:513–517. https://doi.org/10.1023/A:1008741015912
De Vleesschauwer D, Höfte M (2009) Rhizobacteria-induced systemic resistance. In: van Loon LC (ed) Advances in botanical research, vol 51. Elsevier, pp 223–281. https://doi.org/10.1016/S0065-2296(09)51006-3
De Vleesschauwer D, Djavaheri M, Bakker PAHM, Hofte M (2008) Pseudomonas fluorescens WCS374r-induced systemic resistance in Rice against Magnaporthe oryzae is based on Pseudobactin-mediated priming for a salicylic acid-repressible multifaceted defense response. Plant Physiol 148:1996–2012. https://doi.org/10.1104/pp.108.127878
Djonovic S, Vargas WA, Kolomiets MV, Horndeski M, Wiest A, Kenerley CM (2007) A proteinaceous elicitor Sm1 from the beneficial fungus Trichoderma virens is required for induced systemic resistance in Maize. Plant Physiol 145:875–889. https://doi.org/10.1104/pp.107.103689
Duijff BJ, Meijer JW, Bakker PAHM, Schippers B (1993) Siderophore-mediated competition for iron and induced resistance in the suppression of fusarium wilt of carnation by fluorescent Pseudomonas spp. Netherl J Plant Pathol 99:277–289. https://doi.org/10.1007/BF01974309
Ebel J, Mithöfer A (1998) Early events in the elicitation of plant defense. Planta 206:335–348. https://doi.org/10.1007/s004250050409
Edreva A (2004) A novel strategy for plant protection: induced resistance. J Cell Mol Biol 3:61–69
El Modafar C, Elgadda M, El Boutachfaiti R, Abouraicha E, Zehhar N, Petit E, El Alaoui-Talibi Z, Courtois B, Courtois J (2012) Induction of natural defense accompanied by salicylic acid-dependant systemic acquired resistance in tomato seedlings in response to bio-elicitors isolated from green algae. Scientia Horticul 138:55–63. https://doi.org/10.1016/j.scienta.2012.02.011
Elad Y, David DR, Harel YM, Borenshtein M, Kalifa HB, Silber A, Graber ER (2010) Induction of systemic resistance in plants by biochar, a soil-applied carbon sequestering agent. Phytopathology 100:913–921. https://doi.org/10.1094/PHYTO-100-9-0913
Fatima S, Anjum T (2017) Identification of a potential ISR determinant from Pseudomonas aeruginosa PM12 against fusarium wilt in tomato. Front Plant Sci 8:848. https://doi.org/10.3389/fpls.2017.00848
Franken P (2012) The plant strengthening root endophyte Piriformospora indica: potential application and the biology behind. Appl Microbiol Biotechnol 96:1455–1464. https://doi.org/10.1007/s00253-012-4506-1
Gozzo F (2003) Systemic acquired resistance in crop protection: from nature to a chemical approach. J Agricul Food Chem 51:4487–4503. https://doi.org/10.1021/jf030025s
Hase S, Takahashi S, Takenaka S, Nakaho K, Arie T, Seo S, Ohashi Y, Takahashi H (2008) Involvement of jasmonic acid signalling in bacterial wilt disease resistance induced by biocontrol agent Pythium oligandrum in tomato. Plant Pathol 57:870–876. https://doi.org/10.1111/j.1365-3059.2008.01858.x
Heil M (2002) Induced systemic resistance (isr) against pathogens in the context of induced plant defences. Ann Bot 89:503–512. https://doi.org/10.1093/aob/mcf076
Hoffland E, Pieterse CMJ, Bik L, van Pelt JA (1995) Induced systemic resistance in radish is not associated with accumulation of pathogenesis-related proteins. Physiol Mol Plant Pathol 46:309–320. https://doi.org/10.1006/pmpp.1995.1024
Hossain MM, Sultana F, Kubota M, Hyakumachi M (2008) Differential inducible defense mechanisms against bacterial speck pathogen in Arabidopsis thaliana by plant-growth-promoting-fungus Penicillium sp. GP16-2 and its cell-free filtrate. Plant Soil 304:227–239. https://doi.org/10.1007/s11104-008-9542-3
Iavicoli A, Boutet E, Buchala A, Métraux J-P (2003) Induced systemic resistance in Arabidopsis thaliana in response to root inoculation with Pseudomonas fluorescens CHA0. Mol Plant Microbe Int 16:851–858. https://doi.org/10.1094/MPMI.2003.16.10.851
Iwata M, Umemura K, Midoh N (2004) Probenazole (Oryzemate®) – A plant defense activator. In: Kawasaki S (ed) Rice blast: interaction with rice and control. Springer, Dordrecht, pp 163–171. https://doi.org/10.1007/978-0-306-48582-4_19
Jaulneau V, Lafitte C, Corio-Costet M-F, Stadnik MJ, Salamagne S, Briand X, Esquerré-Tugayé M-T, Dumas B (2011) An Ulva armoricana extract protects plants against three powdery mildew pathogens. Eur J Plant Pathol 131:393–401. https://doi.org/10.1007/s10658-011-9816-0
Jung SC, Martinez-Medina A, Lopez-Raez JA, Pozo MJ (2012) Mycorrhiza-induced resistance and priming of plant defenses. J Chem Ecol 38:651–664. https://doi.org/10.1007/s10886-012-0134-6
Kessmann H, Staub T, Hofmann C, Maetzke T, Herzog J, Ward E, Uknes S, Ryals J (1994) Induction of systemic acquired disease resistance in plants by chemicals. Annu Rev Phytopathol 32:439–459. https://doi.org/10.1146/annurev.py.32.090194.002255
Kloepper JW, Ryu C-M, Zhang S (2004) Induced systemic resistance and promotion of plant growth by Bacillus spp. Phytopathology 94:1259–1266. https://doi.org/10.1094/PHYTO.2004.94.11.1259
Korolev N, Rav David D, Elad Y (2008) The role of phytohormones in basal resistance and Trichoderma-induced systemic resistance to Botrytis cinerea in Arabidopsis thaliana. Biol Control 53:667–683. https://doi.org/10.1007/s10526-007-9103-3
Lee B, Farag MA, Park HB, Kloepper JW, Lee SH, Ryu C-M (2012) Induced resistance by a long-chain bacterial volatile: elicitation of plant systemic defense by a C13 volatile produced by Paenibacillus polymyxa. PLoS One 7:e48744. https://doi.org/10.1371/journal.pone.0048744
Leeman M, van Pelt JA, den Ouden FM, Heinsbroek M, Bakker PAHM, Schippers B (1995) Induction of systemic resistance by Pseudomonas fluorescens in radish cultivars differing in susceptibility to fusarium wilt, using a novel bioassay. Eur J Plant Pathol 101:655–664. https://doi.org/10.1007/BF01874869
Li H, Soares MA, Torres MS, Bergen M, White JF (2015) Endophytic bacterium, Bacillus amyloliquefaciens, enhance ornamental host-resistance to diseases and insect pests. J Plant Int 10:224–229. https://doi.org/10.1080/17429145.2015.1056261
Maurhofer M, Hase C, Meuwly P, Metraux JP (1994) Induction of systemic resistance of tobacco to tobacco necrosis virus by the root-colonizing Pseudomonas fluorescens strain CHA0: influence of the gacA gene and pyoverdine production. Phytopathology 84:139–146
Memelink J (2009) Regulation of gene expression by jasmonate hormones. Phytochemistry 70:1560–1570. https://doi.org/10.1016/j.phytochem.2009.09.004
Meziane H, Van Der Sluis I, Van Loon LC, Höfte M, Bakker PAHM (2005) Determinants of Pseudomonas putida WCS358 involved in inducing systemic resistance in plants. Mol Plant Pathol 6:177–185. https://doi.org/10.1111/j.1364-3703.2005.00276.x
Pangesti N, Reichelt M, van de Mortel JE, Kapsomenou E, Gershenzon J, van Loon JJA, Dicke M, Pineda A (2016) Jasmonic acid and ethylene signaling pathways regulate glucosinolate levels in plants during rhizobacteria-induced systemic resistance against a leaf-chewing herbivore. J Chem Ecol 42:1212–1225. https://doi.org/10.1007/s10886-016-0787-7
Pieterse CMJ (1996) Systemic resistance in Arabidopsis induced by biocontrol bacteria is independent of salicylic acid accumulation and pathogenesis-related gene expression. Plant Cell 8:1225–1237. https://doi.org/10.1105/tpc.8.8.1225
Pieterse CMJ (1998) A novel signaling pathway controlling induced systemic resistance in Arabidopsis. Plant Cell 10:1571–1580. https://doi.org/10.1105/tpc.10.9.1571
Pieterse CMJ, van Wees SCM, van Pelt JA, Knoester M, Laan R, Gerrits H, Weisbeek PJ, van Loon LC (1998) A novel signaling pathway controlling induced systemic resistance in Arabidopsis. Plant Cell 10:1571–1158
Pieterse CMJ, Van der Does D, Zamioudis C, Leon-Reyes A, Van Wees SCM (2012) Hormonal modulation of plant immunity. Ann Rev Cell Develop Biol 28:489–521. https://doi.org/10.1146/annurev-cellbio-092910-154055
Pinto KMS, do Nascimento LC, de Souza Gomes EC, da Silva HF, dos Reis Miranda J (2012) Efficiency of resistance elicitors in the management of grapevine downy mildew Plasmopara viticola: epidemiological, biochemical and economic aspects. Eur J Plant Pathol 134:745–754. https://doi.org/10.1007/s10658-012-0050-1
Pozo MJ, Azcón-Aguilar C (2007) Unravelling mycorrhiza-induced resistance. Curr Opi Plant Biol 10:393–398. https://doi.org/10.1016/j.pbi.2007.05.004
Pozo MJ, Van Der Ent S, Van Loon LC, Pieterse CMJ (2008) Transcription factor MYC2 is involved in priming for enhanced defense during rhizobacteria-induced systemic resistance in Arabidopsis thaliana. New Phytol 180:511–523. https://doi.org/10.1111/j.1469-8137.2008.02578.x
Press CM, Wilson M, Tuzun S, Kloepper JW (1997) Salicylic acid produced by Serratia marcescens 90-166 is not the primary determinant of induced systemic resistance in Cucumber or Tobacco. Mol Plant Microbe Int 10:761–768. https://doi.org/10.1094/MPMI.1997.10.6.761
Pushpalatha HG, Sudisha J, Geetha NP, Amruthesh KN, Shekar Shetty H (2011) Thiamine seed treatment enhances LOX expression, promotes growth and induces downy mildew disease resistance in pearl millet. Biol Plant 55:522–527. https://doi.org/10.1007/s10535-011-0118-3
Quaglia M, Ederli L, Pasqualini S, Zazzerini A (2011) Biological control agents and chemical inducers of resistance for postharvest control of Penicillium expansum Link. on apple fruit. Posthar Biol Technol 59:307–315. https://doi.org/10.1016/j.postharvbio.2010.09.007
Ramírez V, Van der Ent S, García-Andrade J, Coego A, Pieterse CM, Vera P (2010) OCP3 is an important modulator of NPR1-mediated jasmonic acid-dependent induced defenses in Arabidopsis. BMC Plant Biol 10:199. https://doi.org/10.1186/1471-2229-10-199
Ray J (1901) Les maladies cryptogamiques des végétaux. Rev Gen Bot 13:145–151
Ren Y, Wang Y, Bi Y, Ge Y, Wang Y, Fan C, Deng H (2012) Postharvest BTH treatment induced disease resistance and enhanced reactive oxygen species metabolism in muskmelon (Cucumis melo L.) fruit. Eur Food Res Technol 234:963–971. https://doi.org/10.1007/s00217-012-1715-x
Ross AF (1961a) Localized acquired resistance to plant virus infection in hypersensitive hosts. Virology 14:329–339. https://doi.org/10.1016/0042-6822(61)90318-X
Ross AF (1961b) Systemic acquired resistance induced by localized virus infections in plants. Virology 14:340–358. https://doi.org/10.1016/0042-6822(61)90319-1
Ruano G, Scheuring D (2020) Plant cells under attack: unconventional endomembrane trafficking during plant defense. Plan Theory 9:389. https://doi.org/10.3390/plants9030389
Rudrappa T, Czymmek KJ, Pare PW, Bais HP (2008) Root-secreted malic acid recruits beneficial soil bacteria. Plant Physiol 148:1547–1556. https://doi.org/10.1104/pp.108.127613
Ryu C-M (2004) Bacterial volatiles induce systemic resistance in Arabidopsis. Plant Physiol 134:1017–1026. https://doi.org/10.1104/pp.103.026583
Ryu C-M, Murphy JF, Mysore KS, Kloepper JW (2004) Plant growth-promoting rhizobacteria systemically protect Arabidopsis thaliana against Cucumber mosaic virus by a salicylic acid and NPR1-independent and jasmonic acid-dependent signaling pathway. Plant J 39:381–392. https://doi.org/10.1111/j.1365-313X.2004.02142.x
Saldajeno MGB, Hyakumachi M (2011) The plant growth-promoting fungus fusarium equiseti and the arbuscular mycorrhizal fungus glomus mosseae stimulate plant growth and reduce severity of anthracnose and damping-off diseases in cucumber (Cucumis sativus) seedlings. Ann Appl Biol 159:28–40. https://doi.org/10.1111/j.1744-7348.2011.00471.x
Satish D, Mukherjee SK, Gupta D (2019) PAmiRDB: a web resource for plant miRNAs targeting viruses. Sci Rep 9:4627. https://doi.org/10.1038/s41598-019-41027-1
Schneider M, Schweizer P, Meuwly P, Métraux JP (1996) Systemic acquired resistance in plants. Int Rev Cytol 168:303–340
Schönbeck F, Steiner U, Kraska T (1993) Induced resistance: criteria, mechanisms, practical application and estimation. J Plant Dis Prot 100(5):541–557
Segarra G, Van der Ent S, Trillas I, Pieterse CMJ (2009) MYB72, a node of convergence in induced systemic resistance triggered by a fungal and a bacterial beneficial microbe. Plant Biol 11:90–96. https://doi.org/10.1111/j.1438-8677.2008.00162.x
Shetty R, Jensen B, Shetty NP, Hansen M, Hansen CW, Starkey KR, Jørgensen HJL (2012) Silicon induced resistance against powdery mildew of roses caused by Podosphaera pannosa: silicon induced resistance in roses. Plant Pathol 61:120–131. https://doi.org/10.1111/j.1365-3059.2011.02493.x
Shoresh M, Harman GE, Mastouri F (2010) Induced systemic resistance and plant responses to fungal biocontrol agents. Annu Rev Phytopathol 48:21–43. https://doi.org/10.1146/annurev-phyto-073009-114450
Siwinska J, Siatkowska K, Olry A, Grosjean J, Hehn A, Bourgaud F, Meharg AA, Carey M, Lojkowska E, Ihnatowicz A (2018) Scopoletin 8-hydroxylase: a novel enzyme involved in coumarin biosynthesis and iron-deficiency responses in Arabidopsis. J Exp Bot 69:1735–1748. https://doi.org/10.1093/jxb/ery005
Sommer A, Wenig M, Knappe C, Kublik S, Foesel B, Schloter M, Vlot C (2021) Induced systemic resistance impacts the phyllosphere microbiome through plant-microbe-microbe interactions. Plant Biol. https://doi.org/10.1101/2021.01.13.426583
Soylu S, Baysal Ö, Soylu EM (2003) Induction of disease resistance by the plant activator, acibenzolar-S-methyl (ASM), against bacterial canker (Clavibacter michiganensis subsp. michiganensis) in tomato seedlings. Plant Sci 165:1069–1075. https://doi.org/10.1016/S0168-9452(03)00302-9
Spoel SH (2003) NPR1 modulates cross-talk between salicylate- and jasmonate-dependent defense pathways through a novel function in the cytosol. Plant Cell 15:760–770. https://doi.org/10.1105/tpc.009159
Srivastava P, George S, Marois JJ, Wright DL, Walker DR (2011) Saccharin-induced systemic acquired resistance against rust (Phakopsora pachyrhizi) infection in soybean: effects on growth and development. Crop Prot 30:726–732. https://doi.org/10.1016/j.cropro.2011.02.023
Stein E, Molitor A, Kogel K-H, Waller F (2008) Systemic resistance in Arabidopsis conferred by the mycorrhizal fungus Piriformospora indica requires jasmonic acid signaling and the cytoplasmic function of NPR1. Plant Cell Physiol 49:1747–1751. https://doi.org/10.1093/pcp/pcn147
Ton J, Pieterse CMJ, van Loon LC (1999) Identification of a locus in Arabidopsis controlling both the expression of rhizobacteria-mediated induced systemic resistance (ISR) and basal resistance against Pseudomonas syringae pv. Tomato. Mol Plant Microbe Int 12:911–918. https://doi.org/10.1094/MPMI.1999.12.10.911
Ton J, van Pelt JA, van Loon LC, Pieterse CMJ (2002) Differential effectiveness of salicylate-dependent and jasmonate/ethylene-dependent induced resistance in Arabidopsis. Mol Plant Microbe Int 15:27–34. https://doi.org/10.1094/MPMI.2002.15.1.27
van der Ent S, van Hulten M, Pozo MJ, Czechowski T, Udvardi MK, Pieterse CMJ, Ton J (2009a) Priming of plant innate immunity by rhizobacteria and β-aminobutyric acid: differences and similarities in regulation. New Phytol 183:419–431. https://doi.org/10.1111/j.1469-8137.2009.02851.x
van der Ent S, van Wees SCM, Pieterse CMJ (2009b) Jasmonate signaling in plant interactions with resistance-inducing beneficial microbes. Phytochemistry 70:1581–1588. https://doi.org/10.1016/j.phytochem.2009.06.009
van Loon LC (2007) Plant responses to plant growth-promoting rhizobacteria. Eur J Plant Pathol 119:243–254. https://doi.org/10.1007/s10658-007-9165-1
van Loon LC, Bakker PAHM, Pieterse CMJ (1998) Systemic resistance induced by rhizosphere bacteria. Annu Rev Phytopathol 36:453–483. https://doi.org/10.1146/annurev.phyto.36.1.453
van Peer R, Niemann GJ, Schippers B (1991) Induced resistance and phytoalexin accumulation in biological control of Fusarium wilt of carnation by Pseudomonas sp. strain WCS417r. Phytopathology 81:728–734
van Wees SCM, Pieterse CMJ, Trijssenaar A, vant’Westende YAM, Hartog F, van Loon LC (1997) Differential induction of systemic resistance in Arabidopsis by biocontrol bacteria. Mol Plant Microbe Int 10:716–724. https://doi.org/10.1094/MPMI.1997.10.6.716
van Wees SCM, Luijendijk M, Smoorenburg I, van Loon LC, Pieterse CMJ (1999) Rhizobacteria-mediated induced systemic resistance (ISR) in Arabidopsis is not associated with a direct effect on expression of known defense-related genes but stimulates the expression of the jasmonate-inducible gene Atvsp upon challenge. Plant Mol Biol 41:537–549. https://doi.org/10.1023/A:1006319216982
Verhagen BWM, Glazebrook J, Zhu T, Chang H-S, van Loon LC, Pieterse CMJ (2004) The transcriptome of rhizobacteria-induced systemic resistance in Arabidopsis. Mol Plant Microbe Int 17:895–908. https://doi.org/10.1094/MPMI.2004.17.8.895
Wang J, Zhou C, Xiao X, Xie Y, Zhu L, Ma Z (2017) Enhanced iron and selenium uptake in plants by volatile emissions of Bacillus amyloliquefaciens (BF06). Appl Sci 7:85. https://doi.org/10.3390/app7010085
Wei G, Kloepper JW, Tuzun S (1991) Induction of systemic resistance of cucumber to Colletotrichum orbiculare by select strains of plant-growth promoting rhizobacteria. Phytopathology 81:1508–1512
Weller DM, Mavrodi DV, van Pelt JA, Pieterse CMJ, van Loon LC, Bakker PAHM (2012) Induced systemic resistance in Arabidopsis thaliana against Pseudomonas syringae pv. Tomato by 2,4-diacetylphloroglucinol-producing Pseudomonas fluorescens. Phytopathology 102:403–412. https://doi.org/10.1094/PHYTO-08-11-0222
Williams EB, Kuc J (1969) Resistance in Malus to Venturia inaequalis. Annu Rev Phytopathol 7:223–246. https://doi.org/10.1146/annurev.py.07.090169.001255
Yan Z, Reddy MS, Ryu C-M, McInroy JA, Wilson M, Kloepper JW (2002) Induced systemic protection against tomato late blight elicited by plant growth-promoting rhizobacteria. Phytopathology 92:1329–1333. https://doi.org/10.1094/PHYTO.2002.92.12.1329
Yang K-H, Huang C-J, Liu Y-H, Chen C-Y (2011) Efficacy of probenazole for control of southern corn leaf blight. J Pest Sci 36:235–239. https://doi.org/10.1584/jpestics.G10-94
Yuan M, Huang Y, Ge W, Jia Z, Song S, Zhang L, Huang Y (2019) Involvement of jasmonic acid, ethylene and salicylic acid signaling pathways behind the systemic resistance induced by Trichoderma longibrachiatum H9 in cucumber. BMC Genomics 20:144. https://doi.org/10.1186/s12864-019-5513-8
Zipfel C (2009) Early molecular events in PAMP-triggered immunity. Curr Opin Plant Biol 12:414–420. https://doi.org/10.1016/j.pbi.2009.06.003
Acknowledgements
We would like to thank the funding agencies which provided financial support (JRF and SRF) to the authors. The duly acknowledged funding agencies are the Council of Scientific and Industrial Research (CSIR, India), and University Grants Commission (UGC, India).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Satish, D., Mehta, S. (2023). Induced Systematic Resistance and Plant Immunity. In: Singh, N., Chattopadhyay, A., Lichtfouse, E. (eds) Sustainable Agriculture Reviews 60. Sustainable Agriculture Reviews, vol 60. Springer, Cham. https://doi.org/10.1007/978-3-031-24181-9_7
Download citation
DOI: https://doi.org/10.1007/978-3-031-24181-9_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-24180-2
Online ISBN: 978-3-031-24181-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)