Abstract
The deep sea is Earth’s most typical environment and meiofauna its most common and arguably its most diverse metazoan inhabitants. They are therefore key in understanding temporal and spatial patterns in biodiversity and biogeography and are major contributors to ecological processes and functions. Meiofauna are integral to deep-sea benthic communities, with numerous links to other benthic organisms and the interstitial environment, the habitat from where they experience life around them. Although many meiofaunal patterns and relations have been identified, limited progress has been made in answering questions as to “why” and “how” these patterns and relations exist or are formed and maintained, and in many cases such knowledge does not exist. In this chapter, we review the knowledge we do have and present interpretations and explanations that bring a better understanding of how meiofauna patterns in the deep sea can be explained in terms of processes and ecological interactions. We applied this approach in four distinct fields of study: trophic interactions; biodiversity and ecosystem function; distribution and diversity patterns; and connectivity patterns. All four illustrate the extent to which meiofauna relate to other biological components and the abiotic environment. Moreover, technological advances and the increase in multidisciplinary approaches (inherent to offshore deep-sea research) show that meiofauna studies are becoming better integrated with other fields of deep-sea research. Meiofauna, therefore, offer an exciting scientific and diverse future of discovery with research operating at the frontiers of deep-sea science.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
8.1 Introduction
The deep sea (generally defined as deeper than 200 m water depth) covers vast expanses of the globe, providing an unimaginable amount of habitat space for microscopic organisms, to the point where it becomes difficult to grasp—at least for one not immersed in the study of meiobenthos. Those tiny animals, the size of a sand grain or smaller, can make a meaningful contribution to ecological processes on such a vast scale. Ubiquity and abundance, two important characteristics of meiofauna worldwide, make these organisms an essential part of deep-sea habitats and ecosystems. They should therefore be seen as central to the study of temporal and spatial patterns in biodiversity and biogeography, as key contributors to ecological processes and functions.
This is in opposition to misguided descriptions of meiofauna as an interesting but non-essential sideshow. Deep-sea meiofauna are, after all, the most common metazoan organisms living in the largest ecosystem on the planet. In other words, they are among the planet’s most typical organisms living in the planet’s most typical environment. Not only are they connected to deep-sea habitats by the simple fact that the abiotic environment, or environmental envelope, of these habitats provides them with the means to survive, but they are also an integral component of benthic communities. It is therefore not surprising to see links between the meiofauna and their biotic and abiotic surroundings everywhere in the deep sea. However, it behooves us to investigate questions beyond establishing a link and investigate the “why” and “how” of the observed relationships.
From the continental slope, to canyons, seamounts, guyots, abyssal plains, trenches, hydrothermal vents, and seeps, meiofauna are prevalent (Fig. 8.1). Foraminiferans, nematodes, and copepods constitute the most abundant taxa in deep-sea sediments (Zeppilli et al. 2018) and, at depths below 3000 m, meiofauna surpass larger size groups such as macrofauna and demersal fish in terms of total biomass (Rex et al. 2006; Wei et al. 2010). How can this be the case? Oligotrophy and intense natural selection for particle selection and resource exploitation ability have been posited as potential mechanisms for the prevalence of meiofauna in the deep sea and for the diminutive size of macrofaunal taxa in the deep sea (Jumars et al. 1990; Kaariainen and Bett 2006).
Considering the increasing relative abundance and biomass of meiofauna with water depth, we must also consider contributions of meiofauna to ecosystem functions (and ultimately services) in the deep sea. Their dominance over other deep-sea metazoan life forms needs to be explained by ecological processes and mechanisms providing them a competitive advantage. We should therefore also expect meiofauna to make an important contribution to ecosystem function, especially on continental margins, the transition zone from the shallow shelf to the oligotrophic abyssal plains, where much of the essential ecosystem services provided by the deep sea are concentrated [e.g., hydrocarbons, fisheries, and carbon burial (Levin and Dayton 2009; Sarmiento and Gruber 2002)]. However, despite their importance, many deep-sea ecological processes are still not understood. This lack of knowledge is particularly acute for meiofauna, and most microscopic organisms, including protozoans, bacteria, archaea, and viruses.
In this chapter, we will address advances in our understanding of four broad topics in meiofauna research that illustrate their relation to the deep ocean, the environment they inhabit, and the other biological components they relate to. This is by no means an attempt to review the entirety of available literature, but rather to present in-depth case studies of existing paradigms and ecological questions that merit further investigation and have relevance to the general field of marine ecology:
-
1.
Trophic interactions
-
2.
Biodiversity and ecosystem function
-
3.
Distribution and diversity patterns
-
4.
Connectivity
These four fields of study illustrate how meiofauna species and communities are connected to each other and the world around them. These disciplines have seen substantive increases in understanding in the last decade or so, supported by advances in technology and the tools with which to investigate important scientific questions. These make them ideal study topics to highlight what we currently understand to be frontiers in deep-sea meiofauna research.
8.2 Trophic Interactions of Deep-Sea Meiofauna: “You Are What You Eat” But What Do Deep-Sea Meiofauna Eat?
When staring down a microscope looking at an intact meiofauna sediment sample, one is immediately confronted with the hidden nature and intricacies of interstitial space. Jansson (1971), in his work “The “Umwelt” of the Interstitial Fauna” in the Proceedings of the First International Conference on Meiofauna, describes elegantly the functional environment of meiofauna from their point of view in an attempt to understand better the environmental parameters that drive the behavior and function of the microscopic organisms living between the sediment grains. It is easy to imagine the grain surfaces to be analogous to vast landscapes filled with green hills of micro-algae and meadows of bacteria on which meiofauna may feast (Meadows and Anderson 1966, 1968). It is evident that such an image evokes the necessity of a myriad of ecological connections between meiofauna and other organisms in their immediate surroundings, trophic, or otherwise. Ecological interactions between different meiofauna taxa and between meiofauna and other benthic components have been amply documented for shallow waters, but much less so for the deep sea (Schratzberger and Ingels 2018).
The deep ocean relies almost exclusively on a food supply originating from primary production in surface waters (except for chemosynthetic ecosystems where chemical energy is harnessed as the basis for the food web). Benthic–pelagic coupling, the exchange of energy, mass and nutrients between the water column and seafloor ecosystems, is crucial to deep-sea benthic life (Woolley et al. 2016). This organic matter, produced in surface waters as a result of photosynthetic primary production (phytoplankton), is converted into sinking particles by food web processes (zooplankton, other grazers, microbes), by aggregation and fecal pellet production (Cavan et al. 2015), as well as microbial colonization and growth during sinking (Azam and Malfatti 2007). Once on the seafloor, bacterial activity normally increases, and together with the deposited material, serves as a rich food for protozoans and small metazoans (Gooday 1993; Moodley et al. 2002; Witte et al. 2003a, b).
Meiofauna play a key role in linking detrital and prokaryotic food resources with higher-level consumers, making available energy that may remain unused in their absence, therefore fulfilling an essential ecological role (Schratzberger and Ingels 2018). High metabolic and reproductive rates and high standing stocks of meiofauna in coastal areas and shallow waters imply that meiofauna play an important role in benthic energy transfer. However, what about deep-sea settings? Owing to a lack of direct observations of deep-sea meiofauna life histories and metabolic characteristics, the same trophic linkages have not yet been explicitly documented in deep-sea settings. There, the limited evidence available suggests that meiofaunal energy demand is met through consuming labile and refractory detritus, bacterial resources, or dissolved organic matter (Gontikaki et al. 2011; Guilini et al. 2010; Ingels et al. 2010; Pape et al. 2013c; van Oevelen et al. 2006, 2011a, b;) and potentially other sources [e.g., fungi and protozoa such as foraminifera and ciliates (Bhadury et al. 2011; Majdi et al. 2020)]. However, they also respond to episodic food falls (Debenham et al. 2004; Soltwedel et al. 2018) and may consume other meiofauna organisms as evidenced by gut-content observations (Fonseca and Gallucci 2008). In addition, stable isotope and fatty acid analyses have shown the capacity of the nematode Deontostoma tridentum in deep-sea sediments off New Zealand, to consume a relatively wide range of food sources, spanning three trophic levels, while the species itself is highly nutritious to other predators as shown by elevated poly-unsaturated fatty acid content (Leduc et al. 2015). In some deep-sea habitats, such as canyons, increased numbers of predatory and scavenging nematodes also suggest that preying on small metazoans may be a successful strategy (Ingels et al. 2009; Soetaert and Heip 1995).
Meiofauna have been shown to exploit newly arriving phytodetrital matter in deep-sea settings, which can enhance both meiofauna densities and diversity (Lins et al. 2014; Pape et al. 2013b). Response times vary widely and range from days to weeks (Ingels et al. 2011a; Witte et al. 2003b). In contrast, other studies have shown metazoan meiofauna may in fact fail to exploit and use phytodetritus and associated microorganisms, unlike foraminifera. There is a distinct likelihood that foraminifera are more responsive to food arrival through efficient food-gathering organelles and their ability to raise metabolic activity very rapidly (Gooday 1993; Gooday et al. 1996). Metazoan meiofauna, on the other hand, may be hampered owing to potentially slower growth rates and higher reproductive expense, which puts them at a disadvantage in competitive interactions with foraminifera.
Veit-Köhler et al. (2011) noticed metazoan meiofauna becoming more concentrated at the sediment surface following a phytoplankton bloom deposition event, suggesting either active migration to where fresh phytodetritus had arrived to feed on bacteria and/or more degraded material, or migration to avoid increased respiration and declining oxygen concentration in deeper sediment layers. Further experimental evidence suggests (1) potential passive uptake of bacterial carbon in limited time periods (Guilini et al. 2010); (2) preferential bacterial carbon consumption by meiofauna (Ingels et al. 2010; Pape et al. 2013c); (3) or even dissolved organic matter uptake (Pape et al. 2013c).
Using experimental pulse chase and natural isotopic data in modeling has brought valuable perspectives into the contribution of meiofauna to total organic matter and carbon processing in various deep-sea settings (Gontikaki et al. 2011; van Oevelen et al. 2011a, 2011b). In subarctic conditions (−0.7 °C) in the Faroe–Shetland Channel at about 1000 m water depth, Gontikaki et al. (2011) reported that bacteria dominated carbon flow and respiration in the sediments, but nematodes were responsible for 96% of the refractory detritus uptake, almost entirely covering their energy requirements in this way. This implies an important role for nematodes in phytodetrital carbon turnover. However, only 2% of overall macrofaunal consumption was linked to predation on nematodes (but 35% of that of polychaetes), suggesting limited trophic transfer. In the upper sections of submarine canyons on the other hand, meiofauna can be responsible for over one fifth of carbon remineralization, suggesting high current speeds and sediment resuspension could favor meiofauna communities compared to macrofauna and megafauna (van Oevelen et al. 2011b).
Experiments and in situ observations have demonstrated how surface bloom fallouts and subsequent deep-sea arrival of food sources fuel the benthic food web. However, there is still large uncertainty about the mechanistics of meiofauna responses to this arrival and how selectively they can feed on various resources. Deducting from different buccal or mouth structures, as well as pharyngeal characteristics, some degree of selectivity or competitive advantage must exist if we accept the benefits of evolutionary adaptations to efficient feeding and niche segregation. However, Schuelke et al. (2018), investigating nematode-associated microbiomes in various ocean regions, could not find any relationship between these microbiomes (from the different parts of the nematode’s body) and nematode feeding morphology nor nematode taxonomy. This would suggest decoupling of food source identities and nematode taxonomic and functional diversity.
Experimental observations have not yet revealed how meiofauna meet their energetic requirements. How much energy is needed to sustain growth and reproduction? Metabolism rates of meiofauna (and hence also their energy requirements) may be currently overestimated depending on environmental conditions (Braeckman et al. 2013). The apparent lack of uptake of administered food in experiments could be explained (1) by non-selectively feeding on the bulk organic matter already present in the sediments, (2) by very high trophic specialization so that uptake, measured on assemblage or community level would strongly dilute the signal of administered food sources. In addition, the oligotrophic deep-sea floor provides an environment where small metazoans are most of the time exposed to limiting resource conditions, likely responsible for body-shape, behavioral, and metabolic adaptations that enable them to survive long periods without abundant food sources.
An overview of meiofauna diets in the deep sea would be incomplete without considering chemosynthetic food sources. Seeps and associated bacterial mats, pockmarks, etc., often exhibit increased meiofaunal abundance compared to adjacent, phytodetrital-dependent deep-sea environments (Cepeda et al. 2020; Vanreusel et al. 2010, 1997). Deep-sea hydrothermal vents on the other hand exhibit generally low meiofauna standing stock (Vanreusel et al. 2010). Three reasons are evoked why vents are characterized by low meiofauna abundances: (1) unsuitability of hard vent substrates, (2) possibility that quality and quantity of vent food sources can’t sustain high standing stocks, and (3) predation and competition with abundant macro-invertebrates (Vanreusel et al. 2010).
Chemosynthetic systems harness chemical energy through bacterial activity, rendering it directly or indirectly (through bacterial lysis) available to heterotrophic meiofaunal consumers or in close association with meiofauna in the form of symbioses such as for the nematode species Oncholaimus dyvae, Parabostrichus bathyalis, and Astomonema southwardorum (Bellec et al. 2018; Tchesunov et al. 2012). This is exemplified in isotopic work on deep-sea nematodes and copepods in or near chemosynthetic systems: thiotrophic and methanotrophic bacteria are likely contributors to their diets (Pape et al. 2011; Van Gaever et al. 2006, 2009).
Intricate, potentially symbiotic, relationships between bacteria and meiofauna (especially nematodes) have long been reported, (see Chap. 4), but in the deep sea, their relevance has only been considered relatively recently (Bellec et al. 2018; Tchesunov et al. 2012). Evidence is emerging that chemosynthetic and photosynthetically derived heterotrophic pathways are not mutually exclusive, suggesting that a black-and-white approach to resolving deep-sea benthic food webs may be inadequate to assess carbon flows (Ingels et al. 2011b; Tchesunov et al. 2012; Zeppilli et al. 2019).
While meiofauna can feed on a variety of food sources (of photosynthetic or chemosynthetic origin), evidence of higher consumers feeding on meiofauna has been much less documented. Selective predation on meiofauna taxa may occur in shallow waters (Coull 1990; Jochum et al. 2012), and in fresh water where meiofauna are prey to a large number of macroinvertebrates (Ptatscheck and Traunspurger 2020). However, to our knowledge, such data do not exist for the deep sea (e.g., Gontikaki et al. 2011). Experimental results from exclusion studies suggest that megafaunal grazing in the deep sea reduces meiofauna abundance and diversity (Gallucci et al. 2008a). This is likely caused by indirect effects through competition of resources, sediment disturbance, and burrowing of megafauna, rather than through direct grazing; an observation that was also supported in shallow-water experiments under a controlled environment with different megafauna species (Ingels et al. 2014).
So far, studies have attempted to unravel meiofauna diets and nutritional selectivity in various deep-sea habitats of the world’s oceans. Different approaches have been used, from field studies on natural lipid biomarkers and stable isotope signatures to enrichment experiments performed ex situ and in situ, supported or not by carbon flow modeling studies. However, out of necessity modeling studies still include assumptions and parameterizations for meiofauna metabolism and physiology that stem from shallow-water investigations and may exclude potentially important food sources such as dissolved organic matter and protozoa.
Moreover, in many deep-sea studies, meiofauna are typically considered as one functional group, thus ignoring the structural and functional diversity found across the many meiofauna taxa. Since meiofauna comprise minute organisms, traditional approaches such as stable isotope analysis require extraction of many individuals to obtain sufficient biomass. Moreover, traditional methods involve destructive manipulation so that individuals are not preserved for further taxonomic analysis. Although new technological developments such as NanoSIM allow for isotope identification on a (sub)cellular level, such techniques are currently still prohibitively expensive or not widely accessible. This normally hampers dietary assessments on the level of genera or species to enhance the resolution of intricate deep-sea food webs.
In the few habitats that are dominated by a few species, these methods are more easily applicable, but for most of the deep sea, where meiofauna are characterized by high diversity and low abundance (i.e., most species are rare, and abundance is relatively low) we must rely on more advanced technology to move forward in this field of study. However, technological advances, progress in microscopy, and novel molecular and isotopic procedures, should bring the elucidation of meiofauna diets and their trophic interactions with other benthic components within reach (Majdi et al. 2020).
8.3 Meiofauna Biodiversity and Ecosystem Function in the Deep Sea
There is still ample scientific debate about the relation between deep-sea meiofauna taxonomic diversity, their functional diversity, and ecosystem function. Functional diversity can be identified as a measure of how diverse functional traits (i.e., traits that bear importance to processes and functions in an ecosystem, such as body size and buccal cavity structure) are within a group of taxa. Ecosystem function, on the other hand, is a term encompassing many different processes operating in an ecosystem, such as nutrient fluxes or secondary productivity. Ecosystem function results from the interaction of organisms with their environment. Although linked, functional diversity and ecosystem function are largely different and influenced by environmental constraints depending on the habitat or ecosystem. This is a topic worthy of intense future study since the relationship “biodiversity–ecosystem function” is central in understanding the role of biodiversity: Which processes and functions within an ecosystem are maintained? How do these processes and functions generate and maintain the services the ecosystem provides, and how does biodiversity contribute to them (Loreau et al. 2001)? The deep sea and meiofauna, in particular, may offer unique insights into our understanding of species interactions, ecosystem functioning, and, thus, implications of biodiversity loss (Loreau 2008).
Several studies have shown that in the deep-sea, meiofaunal structural, or taxonomic diversity can be closely linked to meiofauna functional diversity (often expressed in terms of trophic diversity or life-history characteristics for nematodes) (Danovaro et al. 2008; dos Santos et al. 2020; Gambi et al. 2014; Leduc et al. 2013). Yet, meiofaunal diversity and ecosystem function/efficiency, the latter measured by means of biomass, production, respiration, bacterial processes, or enzyme concentrations, have been found to co-vary either positively or negatively in the deep sea (Danovaro et al. 2008; Leduc et al. 2013; Pape et al. 2013a) or not to vary at all (Pape et al. 2013a).
Leduc et al. (2013) highlighted the importance of environmental drivers in affecting the biodiversity–ecosystem function relation. They noted that the previously documented exponential relation between nematode diversity and ecosystem function [sensu Danovaro et al. (2008)] appears to largely break down beyond a certain diversity. This is potentially owing to increased competition or greater functional redundancy under a given set of environmental constraints, such as available resources or carrying capacity of the habitat or ecosystem. In other words, adding species to an already high-diversity assemblage will likely not additionally contribute to ecosystem function (Leduc et al. 2013). Loreau (2008) expresses how extraordinary the findings by Danovaro et al. (2008) are, considering that, in general, diversity has a positive but saturating influence on ecosystem function. Generally, a plateau is reached in the biodiversity–ecosystem function relation at high biodiversity levels, and meta-analyses have shown that this paradigm is remarkably consistent across different ecosystem types and trophic levels (Balvanera et al. 2006; Cardinale et al. 2006; Loreau 2008; Worm et al. 2006). Future research into this phenomenon should consider the full range of deep-sea meiofauna diversity in assessing its relationship with deep-sea ecosystem function, and, importantly, move further into investigating the causal mechanisms responsible for that relation (Fig. 8.2). It is possible that characteristics of deep-sea ecosystems such as low disturbance and low food availability somehow promotes facilitation among species, unlike what has been observed in other ecosystems. However, that does not mean that coexistence and facilitation among (nematode) species is a relation that does not eventually saturate.
Three different types of relationships between ecosystem function and diversity have been used to explain observed patterns in the deep sea. a Saturation: as diversity increases, ecosystem function increases, but only to a certain point where the relationship becomes saturated and increases in diversity do not increase ecosystem function, possibly because of competitive processes or functional redundancy. This relationship has been observed in most ecosystems (Loreau 2008). b Exponential (unsaturated) relationship: as suggested by Danovaro et al. (2008), an exponential relationship between diversity and ecosystem function suggests facilitation between biological components, where diversity increases only lead to increased ecosystem functioning. c Combined exponential–saturation relationship: The diversity–ecosystem function relationship starts exponentially but eventually saturates due to functional redundancy (Leduc et al. 2013)
More broadly, there are many examples of facilitation between different components of deep-sea benthic communities. A few cases in point are as follows:
-
(1)
facilitative meiofauna–bacteria relationships; whereby bacteria serve as nutrition for meiofauna (symbiotic or otherwise) and are stimulated for growth when grazed upon by meiofauna;
-
(2)
bioturbation activities by macrofauna (and large meiofauna) and sediment disturbance by megafauna species enhance biogeochemical cycling and nutrient exchange, or may redistribute food sources to consumers (Levin et al. 2001; Lohrer et al. 2004);
-
(3)
deep-sea species such as large xenophyophorans (megafauna-sized foraminifera) may cause enhanced microbial activity, while itself processing dissolved organic matter and acting as particle traps as do other structure-forming organisms, thereby enhancing sedimentary food-source retention and promoting meiofaunal abundance and diversity (Levin and Gooday 1992);
-
(4)
positive relationships between meiofauna and macrofauna taxonomic and functional diversity (including feeding morphology, predator richness, and bioturbation potential) (Baldrighi and Manini 2015).
These are just four examples of how the relation between biodiversity and ecosystem function in the deep sea may be characterized by enhanced facilitative interactions to maintain important ecosystem processes and functions involving meiofauna. Theoretically, these examples could also occur in shallow-water benthos, but it can be argued that such facilitative relationships have a better chance of developing and being sustained in the deep sea. Deep-sea sediments seem to generally have a more even distribution of individuals, faster accumulation of species with individuals, and often a higher proportion of singletons in a given sample compared to their shallow-water counterparts (Snelgrove and Smith 2002). This suggests species populations in deep-sea sediments are smaller than in shallow waters, but also distributed over larger areas, allowing increased habitat and resource space that may attenuate intense competition processes and promote facilitation. In more dynamic shallow-water environments, facilitative relationships may break down more quickly. Perhaps it is not surprising that increased niche space, as found in the deep sea, in addition to habitat patchiness (caused by biotic activity and abiotic regimes) plus intermediate levels of disturbance allows for the smaller meiofauna to become more dominant than larger organisms, and more diverse and successful, especially below 3000 m water depth, as previously noted.
8.4 Distribution and Diversity of Deep-Sea Meiofauna: Local to Global Scale Patterns
Standing stocks—Food availability [i.e., particulate organic carbon (POC) flux to the seafloor] is the main factor influencing the abundance and biomass of deep-sea meiofauna, explaining not only bathymetric patterns but also geographical variation among and within ocean basins. On a global scale, the highest biomass is found in areas with coastal upwelling (e.g., continental margins), and the lowest biomass is found in central abyssal plains of major ocean basins (Wei et al. 2010). The bathymetric decline of benthic standing stocks is not equal across different size classes; meiofaunal abundance and biomass decrease more slowly with depth than macro- and megafauna, resulting in the dominance of meiofaunal-sized organisms below 3000 m depth. Indeed, the reduction in average animal body size with depth is among the best established macro-ecological patterns in the deep sea (Rex et al. 2006; Thiel 1975). This pattern is thought to originate from the effect of food limitation, which disproportionately affects the ability of large species with a high energy demand to maintain populations above a critical threshold required for reproduction (Thiel 1975). Although the meiofauna-dominated seafloor below 3000 m depth is characterized by low benthic standing stocks relative to coastal, shelf and slope environments, it accounts for more than half of global benthic biomass due to its large surface area (Wei et al. 2010). The oceans’ deepest ecosystems therefore play a critical role in global biogeochemical cycles (see Chap. 2) despite the paucity of large fauna.
Species richness and diversity patterns—As Rex and Etter (2010, p. 25) noted, ‘[benthic] standing stock is the culmination of pelagic–benthic coupling, (…) and is the most directly relevant measure of ecological and evolutionary opportunity in the deep sea’. Given the importance of POC flux in determining deep-sea meiofaunal standing stocks, it is perhaps not surprising that food availability (i.e., chemical energy) has emerged as the main driver of deep-sea meiofauna species richness, in addition to standing stocks (Mokievsky et al. 2007; Soltwedel 2000). This contrasts with coastal marine environments where thermal energy has a dominant role in determining species richness (Yasuhara and Danovaro 2016).
The relationship between productivity and local species richness in the deep sea generally takes a unimodal shape, at least when the full range of productivities is considered. In studies confined to a relatively narrow range of productivities, the unimodal curve can be skewed to the right or left or can show positive or negative relationships with productivity (Rex and Etter 2010). The unimodal relationship was demonstrated for deep-sea nematodes by using biomass as a proxy of productivity (Leduc et al. 2012b) and the relationship also seems to hold in newly studied deep-sea systems (e.g., Dos Santos et al. 2020). Because this relationship persists at both regional and global scales, the mechanisms involved are likely to be of an ecological rather than an evolutionary nature. The ascending limb of the unimodal curve may be the result of higher proportions of rare (Preston 1962) and specialized species (Evans et al. 2006), whereas the descending limb may result from increased competitive exclusion (Grime 1973; Rosenzweig and Abramsky 1993), temporal variability in productivity (Chown and Gaston 1999), or environmental stress (e.g., hypoxia; Levin and Gage 1998). Gray (2002) suggested that the role of productivity lies primarily in setting the upper limit of species diversity, with other factors such as disturbance and habitat heterogeneity (e.g., grain size diversity; Leduc et al. 2012b) influencing the realized diversity at a given location.
The richness of infauna also decreases with distance to the nearest landmass (Woolley et al. 2016). Although it is difficult to disentangle the effects of water depth (i.e., POC flux) and distance from landmass, it is possible that the transport of propagules (Rex et al. 2005), land-derived organic matter (Leduc et al. 2020) and sediment particles of various sizes and mineralogy (Cerrano et al. 1999) have an impact on deep-sea meiofaunal diversity. The fact that meiofauna lack pelagic larval stages and still have abyssal populations of relatively high density means they are more likely to be self-sustaining. Passive dispersal of meiofaunal adults may be more widespread than once thought (see Sect. 8.5 on connectivity and dispersal), and source-sink processes could be influencing deep-sea meiofaunal diversity patterns in low productivity areas.
As seen for standing stocks, meiofaunal diversity is less sensitive to the declines in productivity than that of larger fauna, as might be expected from meiofauna’s smaller body size and energy requirements. Relative to shallow water environments, local species richness of meiofauna in bathyal and abyssal environments is often elevated perhaps reflecting not only lower rates of competitive exclusion due to lower overall productivity, but also lower levels of physical disturbance, and/or lower likelihood of species extinction due to wider species distribution ranges in the vast deep-sea environments (Rex and Etter 2010; Zeppilli et al. 2018).
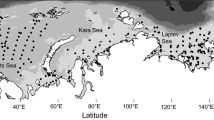
Data on nematode species richness obtained from across the full range of ocean depths (200–11,000 m), from across the Pacific Ocean and including data from Tonga, Kermadec, and Mariana trenches show a trend of decreasing species richness with depth (Fig. 8.3; P < 0.0001, R2 = 0.49). A closer look at the data, however, shows that there is no significant relationship between depth and nematode species richness within each of the 200–6000 and > 6000 m depth ranges (P > 0.1). The overall decrease across the full ocean depth range is therefore due to lower species richness values in hadal trenches (mean ES(51) = 16) relative to slope and abyssal environments (mean ES(51) = 33).
Nematode species richness (ES(51)) from 200 to 11,000 m depth in the Pacific Ocean. The dotted line indicates a significant negative correlation with depth (P < 0.0001, R2 = 0.49) across the full depth range; however, there is no relationship with depth within each of the 200–6000 (empty circles, mean species richness = 33) and >6000 m depth ranges (filled circles, mean species richness = 16). Note the paucity of data below 3000 m depth. (Data from Danovaro et al. 2008; Danovaro et al. 2002; Frank Wenzhöfer, unpublished; Guilini et al. 2012; Lambshead et al. 2002; Leduc et al. 2010b; Leduc and Rowden 2018; Leduc et al. 2012a; Leduc et al. 2016; Miljutin et al. 2010; Miljutin et al. 2011)
Although at a glance these data suggest that species richness does not respond to depth-related environmental gradients in slope and abyssal environments, it seems more likely that the lack of trend reflects wide variation in bathymetric environmental gradients across different locations in the Pacific Ocean. The apparently sudden twofold decrease in nematode species richness observed as we cross the abyssal–hadal trench boundary at ca. 5500–6000 m depth (Fig. 8.3) indicates that habitat-related factors are limiting species richness in the oceans’ deepest environment.
Relative to abyssal plains, the limited extent of hadal trench environments as well as their recent geological formation and isolation likely limit the size of the species pool available for colonization. The greater frequency of physical disturbance induced by turbidity flows in hadal trenches is also likely to limit local species richness. These findings are not irreconcilable with the unimodal model relationship between nematode species richness and productivity since habitat-related factors may affect the general decline of available energy with increasing water depth. Also, food input may be considered a disturbance at levels consistent with high organic loads. To expand our understanding of these patterns, more data on species richness from the full range of ocean depths (200–11,000 m) should be obtained. Our knowledge has so far been hampered by the limited amount of data available for abyssal plains (ca. 4000–6000 m) and hadal trenches (>6000 m) in particular.
Species richness from local to regional and global scales—Meiofauna species number estimates in the deep sea is still fodder for rich debate. Lambshead (1993) noted that local diversity of deep-sea nematodes is roughly the same as that of polychaetes (which typically dominate macrofaunal species richness and abundance), but that nematode abundance is one order of magnitude higher. He concluded that both local and global nematode diversity should therefore be about one order of magnitude greater than macrofaunal diversity. It has since been shown that when all meiofaunal individuals from a site are identified (as opposed to the common practice of estimating species richness based on relatively small subsamples) local nematode and harpacticoid species richness is indeed much greater than polychaete species richness. Perhaps the best illustration of this comes from a study conducted at an abyssal site in the Angola basin, where a total of 600 harpacticoid copepod species were identified based on the identification of 1804 adult specimens, with over half of species represented by a single specimen (George 2014). Extrapolating this order of magnitude difference between meiofaunal and macrofaunal species richness at the global scale, however, relies on the potentially flawed assumption that meiofaunal species turnover is also high.
Based on a dataset from the equatorial central Pacific Ocean, Lambshead and Boucher (2003) later showed that his estimate of nematode species globally had to be revised down because “similar patches with similar species are duplicated over large areas resulting in a more modest regional diversity.”
In other words, while local diversity is high, turnover diversity and hence regional/global diversity are lower than previously assumed. This pattern of very high local species richness but limited spatial turnover was also demonstrated in a study where the identification of 1850 nematode specimens from a single bathyal site on Chatham Rise, New Zealand continental margin, revealed 247 morphospecies, which represents about a third of all 775 morphospecies species identified from the entire Chatham Rise (Leduc et al. 2010a, 2012a).
The assumption of high turnover diversity in deep-sea meiofauna communities followed from the premise that meiofauna have limited dispersal capabilities and therefore restricted geographical distributions. However, the dispersal potential of meiofauna may have been underestimated, at least in shallow-water environments where processes such as zoochory, rafting, drifting, and anthropogenic vectors can disperse meiofaunal organisms across vast distances (Ptatscheck and Traunspurger 2020). On the other hand, the presence of cryptic species complexes suggests that morphospecies distributions may hide true species distributions, resulting in high turnover diversity that can only be detected through molecular analysis. Although the nature and extent of transport mechanisms are less clear in the deep sea, some molecular data provide support for widely distributed deep-sea nematode species (Bik et al. 2010; see next Sect. 8.5 “Connectivity and dispersal”).
Of great interest in determining local to global diversity patterns of meiofauna is the notion that biogeographic distribution patterns of species are likely driven by the size of the organism in the context of the ubiquity model of global distribution. Azovsky et al. (2020) demonstrated how body size is the “master trait” that shapes global biogeographic patterns of marine micro- and meiofauna. The smallest of organisms, such as flagellates, tend to show low endemicity, high regional but low global diversity, and relatively little sign of dispersal limitation. Larger organisms within the size range investigated, such as harpacticoid copepods, on the other hand are posited to have higher endemicity and more restricted global distributions caused by dispersal limitation. These findings support the idea that body size is shaping global biogeographical patterns toward higher predictability and greater dispersal limitation for larger organisms (Azovsky et al. 2020).
Scalability of drivers and mechanisms of diversity distributions—While the size of the organisms seems important in shaping diversity patterns, the scales at which environmental variables operate and drive communities in the deep sea may vary substantially. A meta-analysis of deep-sea meiofaunal studies (Rosli et al. 2018) has shown that most of the variability in communities occurs at the largest (100–10,000 km) and smallest spatial scales (0.001–0.1 m), while the effect of intermediate scales (0.1 m–100 km) is usually less pronounced. At the small scale, gradients in meiofaunal communities are primarily linked with vertical gradients in food and oxygen availability into the sediments (see also Ingels and Vanreusel 2013). However, at the scale of ocean basins, evolutionary/geological history, dispersal barriers, and environmental variables such as surface productivity may all act to increase variability. Although studies have shown that some meiofaunal species can have distributions spanning several 1000s of km within an ocean basin (Ingels et al. 2006; Lambshead and Boucher 2003; Vermeeren et al. 2004), minimal overlap in species distributions across basins has also been observed (Fonseca et al. 2007; Ingels et al. 2006).
The causes behind the high local species richness of meiofauna in some parts of the deep sea are not yet fully understood. While we know that the relatively low levels of productivity in the deep sea should slow down rates of competitive exclusion, thus promoting species coexistence, we have very little information on the nature and extent of competitive interactions and niche segregation among meiofaunal species. The limited information that is available shows high levels of horizontal heterogeneity in meiofaunal communities at the centimeter scale, with the size of patches varying from less than 4–64 cm2 depending on the mobility of different taxa (e.g., nematodes vs copepods and small sedentary nematode species vs large and mobile nematode species) (Gallucci et al. 2008b). Therefore, if we assume that competitive exclusion takes place among meiofauna, we could expect that a greater number of species can coexist in a given area of sediments when species sizes decrease, which could partly explain the high species richness in the deep sea relative to shallow-water environments. Microhabitat heterogeneity resulting from steep vertical gradients in food availability, oxygen and sulfide concentrations as well as particle size diversity, and biogenic structures are also likely to play an important role.
It has been shown that closely related, cryptic meiofaunal species can differ markedly in their competitive abilities, response to environmental stressors, and trophic preferences despite having almost identical morphologies (De Meester et al. 2012, 2015). This indicates that a considerable degree of niche segregation may be occurring, which would facilitate species coexistence. While it will be difficult to investigate niche segregation among deep-sea species, predictions based on species size and mobility could be tested using existing data sets.
Major challenges, which have prevented further investigations of deep-sea species distributions, particularly at ocean basin and global scales, include the time-consuming nature of species identifications, the paucity of taxonomic expertise, the large number of species (many of which are undescribed), and the limited geographical extent of most deep-sea investigations. Although methodologies such as environmental DNA have been touted as potential solutions to this problem, sequence libraries remain far from complete and most sequence data obtained from deep-sea sediment samples cannot be assigned to species (Laroche et al. 2020). Taxonomists working on deep-sea meiofaunal taxa should therefore give sequence data in their species descriptions a high priority so to maximize the uptake of their research among the wider scientific community.
8.5 Connectivity, Dispersal, and Origins of Deep-Sea Meiofauna
The knowledge on population connectivity for deep-sea meiofauna taxa is limited in general. While meiofauna are relatively abundant in the deep sea, most meiofaunal species are rare, with a distinct lack of abundant, dominant species. This results in high species richness in deep-sea assemblages. The exceptions to this impediment may be presented by foraminifera, copepods, and nematodes, which can reach abundances sufficient for such studies. Furthermore, deep-sea meiofauna have a tendency for dwarfism, which is an additional hampering factor for widespread connectivity studies on bathyal, abyssal, and of course hadal meiofauna species.
Despite the fact that the deep seafloor comprises the largest contiguous biotope on Earth, deep-sea populations of benthic species are in general fragmented with evidence of cryptic speciation (Taylor and Roterman 2017). Benthic species from sediments show in general a more restricted dispersal compared to species from hard substrate, demersal, or pelagic habitats. Obviously species with pelagic and/or feeding (planktotrophic) larvae are more dispersive than other larval types (Baco et al. 2016). There is controversy as to the importance of geography versus bathymetry as a barrier for gene flow (Havermans et al. 2013; Zardus et al. 2006). Most studies seem to support the notion that vertical divergence between deep-sea populations is more pronounced than horizontal divergence over similar scales (reviewed in Taylor and Roterman 2017). The mechanisms responsible for this pattern are not clear at present but could relate to differences in currents and oceanographic patterns. This is not just of scientific relevance: Growing interest in deep-sea resources makes connectivity research critical for understanding the mechanisms that regulate population sizes and its function in the context of spatial management and mitigation of recovery from anthropogenic disturbance.
In contrast to the majority of macrobenthic species, connectivity in meiofauna taxa is not achieved through planktonic larval stages, and the dispersal processes are still a major gap in our understanding. Nematodes carry and/or lay eggs, while harpacticoid copepods have naupliar larvae but are still mainly restricted to a benthic lifestyle [despite documented emergence patterns of copepods, see Thistle et al. (2007)]. Also, other meiofauna taxa tend to lack life stages with pelagic dispersal capacities. While some shallow-water species exhibit a relatively wide geographic distribution, especially for opportunistic taxa (Bhadury et al. 2008; Sahraean et al. 2017), evidence is growing, based on an increasing number of molecular-based studies, that so-called cosmopolitan species are part of species complexes with a high degree of cryptic speciation (Bhadury et al. 2008; Cerca et al. 2018; Derycke et al. 2007, 2005; Sahraean et al. 2017). While these cases demonstrate a “hidden diversity” in shallow waters, for the deep sea, information is scant so far. Hauquier et al. (2019) demonstrated that gene flow and cryptic speciation can be very different among nematode species on the deep Antarctic shelf (300–500 m) depending on whether the species is a surface dwelling or deep burrowing species, with the latter showing lower gene flow. There are still many questions surrounding our understanding of meiofauna distributions and dispersal, especially in the deep sea. It seems that the meiofauna paradox is not so much a paradox, but rather a paradigm fed by a lack of understanding and underestimation of true species diversity and dispersal mechanisms (Cerca et al. 2018; Ptatscheck and Traunspurger 2020).
It is little understood to what extent the underestimation of species diversity and dispersal mechanisms holds for deep-sea meiofauna. Here, no major environmental drivers or obvious dispersal barriers are present, except for depth, certain topographies, water mass characteristics, and surface productivity. According to a study by Bik et al. (2010), Enoplids, an important nematode order, are not represented by endemic deep-sea lineages nor do their populations cluster according to bathymetry or geography. The study provides evidence for several cosmopolitan marine species at least at a trans-Atlantic scale. Moreover, Lecroq et al. (2009) and Pawlowski et al. (2007) found a wide bathymetric and geographic distribution of benthic foraminifera species in the deep sea. In contrast to shallow-water benthic foraminiferans, the common species Epistominella exigua showed a remarkably high genetic similarity between Arctic, Atlantic, Pacific, and Antarctic populations, implying a true cosmopolitan distribution (Lecroq et al. 2009). Pawlowski et al. (2007), on the other hand, discovered very limited genetic differentiation between Arctic and Antarctic specimens of two species (E. exigua, Cibicides wuellerstorfi), suggesting huge metapopulations for these species extending from pole to pole across vast bathymetric ranges (1351–4975 m, 573–4407 m, respectively). They also found genetically similar specimens of the same species in the North Atlantic Ocean, suggesting distributions of the species beyond the Arctic and Antarctic.
It is also remarkable that non-chemosynthetic meiofauna taxa dominate deep-sea cold seeps or hydrothermal vents, in contrast with what has been observed for macrofauna and megafauna (Vanreusel et al. 2010; Zeppilli et al. 2018). Several studies have shown that meiofaunal species recorded from chemosynthetic environments are also distributed across proximate and distant habitats, suggesting that populations close to these chemosynthetic systems can be the result of a continuous colonization from adjacent areas (Gollner et al. 2015 and references therein; Zeppilli et al. 2018). A study by Gollner et al. (2016) based on the analysis of mitochondrial DNA of nine dirivultid copepod species occurring at hydrothermal vents in different ocean basins suggests wide dispersal capacity for all species. Despite different geological settings and wide geographic distances, the vents were occupied by these same species indicating high population expansion and high connectivity of these copepod populations with great dispersal capacity.
On the other hand, natural disturbance events (such as volcanic eruptions) at these ephemeral and harsh environments may prevent settlement and successful arrivals. They may even wipe out entire populations, thereby affecting realized rates of dispersal of these hydrothermal species. The authors put forward various reasons for the overall, large-scale genetic exchange between populations:
-
(1)
ocean current regimes may favor dispersal of these copepods since they have lecitotrophic nauplii and feeding copepodites that have been observed in the water column above vents;
-
(2)
intermediate habitats between vents may be used as stepping stones, since vent copepods have been observed up to 1 km away from active vents; and
-
(3)
the potential of biological traits such as lecitotrophy and directed migration during current entrainment may support successful dispersal (Gollner et al., 2016).
Very little is known about the evolutionary origins of deep-sea meiofaunal taxa. Most nematode genera are globally distributed, and most are found both in shallow and deep-sea settings, although some genera occur mostly (e.g., Acantholaimus) or exclusively (e.g., Bathyeurystomina, Manganonema, Thelonema, Cricohalalaimus) in the deep sea. Some recently discovered genera are so far only known from hadal trench environments (>6000 m, e.g., Lamyronema, Maragnopsia). Relative to macrofauna, little is known about the depth distribution of meiofaunal species. So far, forty-eight nematode species are known to have a depth range over 2000 m, and half a dozen have a depth range exceeding 4000 m (Miljutin et al. 2010), which suggests that species ranges for nematodes may be wider than for macrofaunal species.
On the other hand, phylogenetic analyses of deep-sea and shallow-water nematodes suggest repeated and recent interchanges between the deep-sea and intertidal zone and provide evidence for multiple, independent invasions of both deep-sea and shallow-water habitats (Bik et al. 2010). This supports the suggestion of Holterman et al. (2008) that habitat transitions are frequent and common in this taxon. Apart from generally smaller body size, there is no evidence for morphological adaptations specific to deep-sea meiofaunal taxa, although it seems likely that physiological and enzymatic adaptations have developed (Zeppilli et al. 2018).
Given the low abundances and high species richness of meiofauna taxa in the deep sea, our knowledge on species distribution and population connectivity is growing only slowly. To solve questions related to the dispersal, radiative adaptation, and genetic diversity of deep-sea meiofauna, high-throughput sequencing such as genome scanning approaches, metagenomics or metatranscriptomics is very important (Cerca et al. 2018). A metabarcoding approach applied at a wide geographic scale in the NE Pacific abyss provided some insights regarding the distribution of deep-sea nematodes and the tentative mechanisms defining their community structure (Macheriotou et al. 2020). From a phylogenetic perspective, nematode assemblages from the abyssal plains in the Pacific Clarion Clipperton Fracture Zone were characterized by high relatedness.
But factors controlling phylogenetic structuring, or the relative dominance thereof, remain mostly unknown and can differ between genera. Areas located at a few hundreds to thousands of km distance showed a high degree of rarity represented by a large proportion of unique single DNA sequences recovered from a high-throughput molecular analysis (~85% of the entire sequence assemblage). These unique Amplicon Sequence Variants (ASV) have a higher degree of re-usability, reproducibility, and comprehensiveness, and are increasingly replacing Operational Taxonomic Units (OTU) as the standard unit of marker-gene analysis: They can be based on single nucleotide changes instead of a cluster analysis using fixed dissimilarity thresholds (Callahan et al. 2017). However, interpretation of distributional isolation or connectivity between geographical areas, based on ASV’s units, remains, as yet, problematical.
8.6 Conclusions, Thoughts, and Future Perspectives
We attempted to address a common theme that is often discussed in deep-sea meiofauna research: How connected are meiofauna in the deep sea? We used four different topics to illustrate the advances in these research frontiers:
-
trophic interactions,
-
biodiversity and ecosystem function,
-
distribution and diversity patterns, and
-
connectivity.
In the field of deep-sea trophic ecology, it is obvious that meiofauna is inherently linked to the dominant food source—marine snow, and the remnants and microbes that travel to great depths to feed the benthos. Meiofauna respond to these fluxes of particulate rain coming down. However, it is also becoming clearer that in the deep sea, meiofauna are not exclusively reliant on phytodetrital food sources, but that they can acquire energy from microbial and chemosynthetic sources through consumption or symbiosis. Innovative modeling approaches together with stable isotope information (from natural data or experiments whereby food sources are labeled) have brought a better understanding of energetic pathways and carbon cycling and what role meiofauna plays in them. So far, habitat characteristics and their respective environmental conditions seem to affect to a great degree how food webs function and how much energy is transferred through the meiofaunal component to higher trophic levels. However, our understanding of dietary and metabolic mechanisms for meiofauna species is still limited, exacerbated by the complexity of obtaining in situ data. In addition, the observation that many meiofauna exhibit dietary plasticity and the fact that their energetic requirements are not met based on our current understanding of meiofauna metabolisms and food availability lays open a field of study that is certainly worth pursuing. Research into food selectivity and the energetic mechanisms used by meiofauna to cope with the general oligotrophy in the deep sea will give us a better idea of how meiofauna contribute to overall functioning of the deep seafloor and give much-needed insights into the adaptation of deep-sea organisms in general to the deep sea.
In recent years, insights into meiofauna biodiversity and ecosystem function patterns have drastically increased as more data from different deep-sea habitats that may cover larger swaths of seafloor become available. Also here, the connection between meiofauna, their environment, and other organisms is essential in shaping the observed patterns. This is particularly the case when assessing ecosystem function using microbial activity and biomass/production, or biogeochemical processes such as nutrient consumption and cycling. The paradigms associated with the idea that facilitative interactions may play a very important role, and according to some, cause an exponential relationship between meiofauna biodiversity and ecosystem function, must receive further attention in future studies. As more data from full water-depth spectra become available, a more complete picture of this relationship should become more apparent (e.g., Fig. 8.3).
Distribution and diversity patterns are perhaps the best-studied topics in deep-sea meiofauna research. Small-to large-scale studies, ranging from a very specific sampling area to sometimes global-scale assessments, have shown that meiofauna communities respond to environmental conditions in relatively predictable ways. However, new genetic information and insight into cryptic species have demonstrated that distribution patterns of deep-sea meiofauna species that are based on morphological identifications alone are smaller than originally thought. In other cases, globally distributed meiofauna species in the deep sea have been confirmed by molecular studies. Questions as to why distributions of different meiofauna species can differ so drastically have not been answered satisfactorily, but recent insights into body-size distributions suggest that being small may positively influence large-scale dispersal and distribution ranges. Further study into why meiofauna diversity is high in the deep sea is also needed. Ecological theory has been used to explain diversity patterns to a great extent, but our understanding of speciation and niche differentiation for meiofauna in the deep sea does not seem entirely consistent with resource ecology as used for larger organisms such as macrofauna and megafauna.
In the field of meiofauna connectivity research in the deep sea, great advances have been made in the past few years. Yet, the contrast between species that have wide distributions and high gene flow between distant locations, and species that have very limited distributions remains unexplained. Rarity seems to be the norm in the deep sea, with low abundance of many unique DNA sequences in any particular area, but some species do not answer to that paradigm. The evolution and origin of deep-sea meiofauna are relatively unstudied, but some studies suggest that repeated exchange between shallow waters and the deep sea lie at the cause for the large depth ranges observed for some meiofauna species. Morphological adaptations to living in the deep sea (apart from perhaps smaller body size as a consequence of oligotrophy) have not been posited, but it is likely that molecular or enzymatic adaptations exist that allow successful exploitation of the unique challenges the deep sea and its various habitats pose.
Concluding, it emerges that it is not particularly useful to make a distinction between marine benthic ecology and deep-sea benthic ecology. This distinction would contribute to the perception of the deep sea as a disconnected and atypical ecosystem not relevant to society (Jamieson et al. 2020). Deep-sea benthic ecology really is just benthic ecology since the deep sea comprises over 98% of the oceans’ depth range and most of the seabed surface.
Likewise, the study of deep-sea meiofauna is not some esoteric pursuit focusing on organisms that are tiny, strange, difficult to study, and ecologically irrelevant. On the contrary, meiofauna are a dominant component of benthic ecosystems and although there is still much to learn, our knowledge has increased dramatically owing to hundreds of deep-sea meiofauna studies covering thousands of sites sampled across the globe since the pioneering work of Wigley and McIntyre (1964).
Despite the progress, however, many questions about meiofauna in the deep sea remain. It is not surprising that also the diversity of topics being studied in this field seems to increase. Advances in technology and large integrative projects, often involving multiple nations, are improving access to the deep sea and therefore also access to meiofauna samples or ship time to conduct experiments. Advances in analytical procedures, notably in molecular science, with ever-decreasing sequencing costs and increasing resolution and reliability, help to generate vast amounts of new, previously inaccessible information. The multidisciplinary nature of the research is another notable advance. Many studies take advantage of diverse fields of study such as oceanography, modeling approaches, microbiology, geology, and biogeochemistry, to answer questions on the meiofauna patterns we observe: How do meiofauna organisms live and function in the largest, and therefore most characterizing environment on Earth—the deep sea?
References
Azam F, Malfatti F (2007) Microbial structuring of marine ecosystems. Nat Rev Microbiol 5:782–791
Azovsky AI, Chertoprud ES, Garlitska LA, Mazei YA, Tikhonenkov DV (2020) Does size really matter in biogeography? Patterns and drivers of global distribution of marine micro- and meiofauna. J Biogeogr 47:1180–1192
Baco AR, Etter RJ, Ribeiro PA, Heyden S, Beerli P, Kinlan BP (2016) A synthesis of genetic connectivity in deep-sea fauna and implications for marine reserve design. Mol Ecol 25:3276–3298
Baldrighi E, Manini E (2015) Deep-sea meiofauna and macrofauna diversity and functional diversity: are they related? Mar Biodivers 45:469–488
Balvanera P, Pfisterer AB, Buchmann N, He J-S, Nakashizuka T, Raffaelli D, Schmid B (2006) Quantifying the evidence for biodiversity effects on ecosystem functioning and services. Ecol Lett 9:1146–1156
Bellec L, Cambon-Bonavita M-A, Cueff-Gauchard V, Durand L, Gayet N, Zeppilli D (2018) A nematode of the Mid-Atlantic Ridge hydrothermal vents harbors a possible symbiotic relationship. Frontiers in Microbiology 9
Bhadury P, Austen M, Bilton D, Lambshead P, Rogers A, Smerdon G (2008) Evaluation of combined morphological and molecular techniques for marine nematode (Terschellingia spp.) identification. Mar Biol 154:509–518
Bhadury P, Bik H, Lambshead JD, Austen MC, Smerdon GR, Rogers AD (2011) Molecular diversity of fungal phylotypes co-amplified alongside nematodes from coastal and deep-sea marine environments. PLoS ONE 6:e26445
Bik H, Thomas WK, Lunt D, Lambshead PJ (2010) Low endemism, continued deep-shallow interchanges, and evidence for cosmopolitan distributions in free-living marine nematodes (order Enoplida). BMC Evol Biol 10:389
Braeckman U, Vanaverbeke J, Vincx M, van Oevelen D, Soetaert K (2013) Meiofauna metabolism in suboxic sediments: currently overestimated. PLoS ONE 8:e59289
Callahan BJ, McMurdie PJ, Holmes SP (2017) Exact sequence variants should replace operational taxonomic units in marker-gene data analysis. ISME J 11:2639–2643
Cardinale BJ, Srivastava DS, Duffy JE, Wright JP, Downing JP, Sankaran M, Jouseau C (2006) Effects of biodiversity on the functioning of trophic groups and ecosystems. Nature 443:989–992
Cavan E, Le Moigne FA, Poulton AJ, Tarling GA, Ward P, Daniels CJ, Fragoso GM, Sanders RJ (2015) Attenuation of particulate organic carbon flux in the Scotia Sea, Southern Ocean, is controlled by zooplankton fecal pellets. Geophys Res Lett 42:821–830
Cepeda D, Pardos F, Zeppilli D, Sanchez N (2020) Dragons of the deep sea Kinorhyncha communities in a pockmark field at Mozambique Channel, with the description of three new species. Frontiers in Marine Science 7
Cerca J, Purschke G, Struck TH (2018) Marine connectivity dynamics: clarifying cosmopolitan distributions of marine interstitial invertebrates and the meiofauna paradox. Mar Biol 165:123
Cerrano C, Arillo A, Bavestrello G, Benatti U, Calcinai B, Cattaneo-Vietti R, Cortesogno L, Gaggero L, Giovine M, Puce S, Sarà M (1999) Organism–quartz interactions in structuring benthic communities: towards a marine bio-mineralogy? Ecol Lett 2:1–3
Chown SL, Gaston KJ (1999) Patterns in procellariiform diversity as a test of species-energy theory in marine systems. Evol Ecol Res 1:365–373
Coull BC (1990) Are members of the meiofauna food for higher trophic levels? Trans Am Microsc Soc 109
Danovaro R, Gambi C, Dell’Anno A, Corinaidesi C, Fraschetti S, Vanreusel A, Vincx M, Gooday AJ (2008) Exponential decline of deep-sea ecosystem functioning linked to benthic biodiversity loss. Curr Biol 18:1–8
Danovaro R, Gambi C, Della Croce N (2002) Meiofauna hotspot in the Atacama Trench, eastern South Pacific Ocean. Deep Sea Res Part 1 Oceanogr Res Pap 49:843–857
De Meester N, Derycke S, Moens T (2012) Differences in time until dispersal between cryptic species of a marine nematode species complex. PLoS ONE 7:e42674
De Meester N, Derycke S, Rigaux A, Moens T (2015) Temperature and salinity induce differential responses in life histories of cryptic nematode species. J Exp Mar Biol Ecol 472:54–62
Debenham NJ, Lambshead PJD, Ferrero TJ, Smith CR (2004) The impact of whale falls on nematode abundance in the deep sea. Deep Sea Res Part 1 Oceanogr Res Pap 51:701–706
Derycke S, Backeljau T, Vlaeminck C, Vierstraete A, Vanfleteren J, Vincx M, Moens T (2007) Spatiotemporal analysis of population genetic structure in Geomonhystera disjuncta (Nematoda, Monhysteridae) reveals high levels of molecular diversity. Mar Biol 151:1799–1812
Derycke S, Remerie T, Vierstraete A, Backeljau T, Vanfleteren J, Vincx M, Moens T (2005) Mitochondrial DNA variation and cryptic speciation within the free-living marine nematode Pellioditis marina. Mar Ecol-Prog Ser 300:91–103
dos Santos GAP, Silva AC, Esteves AM, Ribeiro-Ferreira VP, Neres PF, Valdes Y, Ingels J (2020) Testing bathymetric and regional patterns in the southwest Atlantic deep sea using infaunal diversity, structure, and function. Diversity 12:485
Evans KL, Jackson SF, Greenwood JJD, Gaston KJ (2006) Species traits and the form of individual species-energy relationships. Proceedings of the Royal Society b: Biological Sciences 273:1779–1787
Fonseca G, Gallucci F (2008) Direct evidence of predation in deep-sea nematodes: the case of Pontonema sp. Cah Biol Mar 49:295–297
Fonseca G, Muthumbi AW, Vanreusel A (2007) Species richness of the genus Molgolaimus (Nematoda) from local to ocean scale along continental slopes. Marine Ecology-an Evolutionary Perspective 28:446–459
Gallucci F, Fonseca G, Soltwedel T (2008a) Effects of megafauna exclusion on nematode assemblages at a deep-sea site. Deep Sea Res Part 1 Oceanogr Res Pap 55:332–349
Gallucci F, Moens T, Vanreusel A, Fonseca G (2008b) Active colonisation of disturbed sediments by deep-sea nematodes: evidence for the patch mosaic model. Mar Ecol-Prog Ser 367:173–183
Gambi C, Pusceddu A, Benedetti-Cecchi L, Danovaro R (2014) Species richness, species turnover and functional diversity in nematodes of the deep Mediterranean Sea: searching for drivers at different spatial scales. Glob Ecol Biogeogr 23:24–39
George KH (2014) Southern ocean harpacticoida. In: De Broyer C, Koubbi P, Griffiths HJ, Raymond B, d’Udekem d’Acoz C, Van de Putte AP, Danis B, David B, Grant S, Gutt J, Held C, Hosie G, Huettmann F, Post A, Ropert-Coudert Y (eds) Biogeographic Atlas of the southern ocean. Scientific committee on Antarctic research, Cambridge UK, pp 88–93
Gollner S, Govenar B, Arbizu PM, Mills S, Le Bris N, Weinbauer M, Shank TM, Bright M (2015) Differences in recovery between deep-sea hydrothermal vent and vent-proximate communities after a volcanic eruption. Deep Sea Res Part 1 Oceanogr Res Pap 106:167–182
Gollner S, Stuckas H, Kihara TC, Laurent S, Kodami S, Martinez Arbizu P (2016) Mitochondrial DNA analyses indicate high diversity, expansive population growth and high genetic connectivity of vent copepods (Dirivultidae) across different oceans. PLoS ONE 11:e0163776
Gontikaki E, van Oevelen D, Soetaert K, Witte U (2011) Food web flows through a sub-arctic deep-sea benthic community. Prog Oceanogr 91:245–259
Gooday AJ (1993) Deep-sea benthic foraminiferal species which exploit phytodetritus: characteristic features and controls on distribution. Mar Micropaleontol 22:187–205
Gooday AJ, Pfannkuche O, Lambshead PJD (1996) An apparent lack of response by metazoan meiofauna to phytodetritus deposition in the bathyal north-eastern Atlantic. J Mar Biol Assoc UK 76:297–310
Gray JS (2002) Species richness of marine soft sediments. Mar Ecol Prog Ser 244:285–297
Grime JP (1973) Competitive exclusion in herbaceous vegetation. Nature 242:344–347
Guilini K, Levin LA, Vanreusel A (2012) Cold seep and oxygen minimum zone associated sources of margin heterogeneity affect benthic assemblages, diversity and nutrition at the Cascadian margin (NE Pacific Ocean). Prog Oceanogr 96:77–92
Guilini K, Van Oevelen D, Soetaert K, Middelburg JJ, Vanreusel A (2010) Nutritional importance of benthic bacteria for deep-sea nematodes from the Arctic ice margin: results of an isotope tracer experiment. Limnol Oceanogr 55:1977–1989
Hauquier F, Macheriotou L, Bezerra TN, Egho G, Martínez Arbizu P, Vanreusel A (2019) Distribution of free-living marine nematodes in the Clarion-Clipperton Zone: implications for future deep-sea mining scenarios. Biogeosciences 16:3475–3489
Havermans C, Sonet G, d’Acoz CdU, Nagy ZT, Martin P, Brix S, Riehl T, Agrawal S, Held C (2013) Genetic and morphological divergences in the cosmopolitan deep-sea amphipod Eurythenes gryllus reveal a diverse abyss and a bipolar species. PLoS ONE 8:e74218
Holterman M, Holovachov O, van den Elsen S, van Megen H, Bongers T, Bakker J, Helder J (2008) Small subunit ribosomal DNA-based phylogeny of basal Chromadoria (Nematoda) suggests that transitions from marine to terrestrial habitats (and vice versa) require relatively simple adaptations. Mol Phylogenet Evol 48:758–763
Ingels J, Vanreusel A (2013) The importance of different spatial scales in determining structural and functional characteristics of deep-sea infauna communities. Biogeosciences 10:4547–4563
Ingels J, Vanhove S, De Mesel I, Vanreusel A (2006) The biodiversity and biogeography of the free-living nematode genera Desmodora and Desmodorella (family Desmodoridae) at both sides of the Scotia Arc. Polar Biol 29:936–949
Ingels J, Kiriakoulakis K, Wolff GA, Vanreusel A (2009) Nematode diversity and its relation to the quantity and quality of sedimentary organic matter in the deep Nazare Canyon, Western Iberian Margin. Deep-Sea Research Part I-Oceanographic Research Papers 56:1521–1539
Ingels J, Billett DSM, Van Gaever S, Vanreusel A (2011a) An insight into the feeding ecology of deep-sea canyon nematodes—results from field observations and the first in-situ C-13 feeding experiment in the Nazare Canyon. J Exp Mar Biol Ecol 396:185–193
Ingels J, Tchesunov AV, Vanreusel A (2011b) Meiofauna in the Gollum Channels and the Whittard Canyon, Celtic Margin–how local environmental conditions shape nematode structure and function. PLoS ONE 6:e20094
Ingels J, Dashfield SL, Somerfield PJ, Widdicombe S, Austen MC (2014) Interactions between multiple large macrofauna species and nematode communities—mechanisms for indirect impacts of trawling disturbance. J Exp Mar Biol Ecol 456:41–49
Ingels J, Van den Driessche P, De Mesel I, Vanhove S, Moens T, Vanreusel A (2010) Preferred use of bacteria over phytoplankton by deep-sea nematodes in polar regions. Mar Ecol Prog Ser 406:121–133
Jamieson AJ, Singleman G, Linley TD, Casey S (2020) Fear and loathing of the deep ocean: why don’t people care about the deep sea? ICES J Mar Sci. https://doi.org/10.1093/icesjms/fsaa234
Jansson B-O (1971) The “umwelt” of the interstitial fauna. In: Hulings NC (ed) Proceedings of the first international conference on meiofauna. Smithsonian Institution Press, Washington, pp 129–140
Jochum M, Schneider FD, Crowe TP, Brose U, O’Gorman EJ (2012) Climate-induced changes in bottom-up and top-down processes independently alter a marine ecosystem. Philosophical Transactions of the Royal Society b: Biological Sciences 367:2962–2970
Jumars PA, Mayer LM, Deming JW, Baross JA, Wheatcroft RA, Charnock H, Edmond John M, McCave IN, Rice AL, Wilson TRS (1990) Deep-sea deposit-feeding strategies suggested by environmental and feeding constraints. Philosophical Transactions of the Royal Society of London. Series a, Mathematical and Physical Sciences 331:85–101
Kaariainen JI, Bett BJ (2006) Evidence for benthic body size miniaturization in the deep sea. J Mar Biol Assoc UK 86:1339–1345
Lambshead PJD (1993) Recent developments in marine benthic biodiversity research. Oceanis 19:5–24
Lambshead PJD, Boucher G (2003) Marine nematode deep-sea biodiversity—hyperdiverse or hype? J Biogeogr 30:475–485
Lambshead PJD, Brown CJ, Ferrero TJ, Mitchell NJ, Smith CR, Hawkins LE, Tietjen J (2002) Latitudinal diversity patterns of deep-sea marine nematodes and organic fluxes: a test from the central equatorial Pacific. Mar Ecol-Prog Ser 236:129–135
Laroche O, Kersten O, Smith CR, Goetze E (2020) Environmental DNA surveys detect distinct metazoan communities across abyssal plains and seamounts in the western Clarion Clipperton Zone. Mol Ecol 29:4588–4604
Lecroq B, Gooday AJ, Pawlowski J (2009) Global genetic homogeneity in the deep-sea foraminiferan Epistominella exigua (Rotaliida: Pseudoparrellidae). Zootaxa 2096:23–32
Leduc D, Rowden AA (2018) Nematode communities in sediments of the Kermadec Trench, Southwest Pacific Ocean. Deep Sea Res Part I 134:23–31
Leduc D, Probert PK, Berkenbusch K, Nodder SD, Pilditch CA (2010a) Abundance of small individuals influences the effectiveness of processing techniques for deep-sea nematodes. Deep Sea Res Part 1 Oceanogr Res Pap 57:1363–1371
Leduc D, Probert PK, Nodder SD (2010b) Influence of mesh size and core penetration on estimates of deep-sea nematode abundance, biomass, and diversity. Deep Sea Res Part 1 Oceanogr Res Pap 57:1354–1362
Leduc D, Brown JCS, Bury SJ, Lörz A-N (2015) High intraspecific variability in the diet of a deep-sea nematode: stable isotope and fatty acid analyses of Deontostoma tridentum on Chatham Rise, Southwest Pacific. Deep Sea Res Part I 97:10–18
Leduc D, Nodder SD, Rowden AA, Gibbs M, Berkenbusch K, Wood A, De Leo F, Smith C, Brown J, Bury SJ, Pallentin A (2020) Structure of infaunal communities in New Zealand submarine canyons is linked to origins of sediment organic matter. Limnol Oceanogr 65:2303–2327
Leduc D, Rowden AA, Bowden DA, Nodder SD, Probert PK, Pilditch CA, Duineveld GCA, Witbaard R (2012a) Nematode beta diversity on the continental slope of New Zealand: spatial patterns and environmental drivers. Mar Ecol Prog Ser 454:37–52
Leduc D, Rowden AA, Bowden DA, Probert PK, Pilditch CA, Nodder SD (2012b) Unimodal relationship between biomass and species richness of deep-sea nematodes: implications for the link between productivity and diversity. Mar Ecol Prog Ser 454:53–64
Leduc D, Rowden AA, Pilditch CA, Maas EW, Probert PK (2013) Is there a link between deep-sea biodiversity and ecosystem function? Mar Ecol 34:334–344
Leduc D, Rowden AA, Glud RN, Wenzhöfer F, Kitazato H, Clark MR (2016) Comparison between infaunal communities of the deep floor and edge of the Tonga Trench: possible effects of differences in organic matter supply. Deep Sea Res Part I 116:264–275
Levin LA, Dayton PK (2009) Ecological theory and continental margins: where shallow meets deep. Trends Ecol Evol 24:606–617
Levin LA, Gage JD (1998) Relationships between oxygen, organic matter and the diversity of bathyal macrofauna. Deep Sea Res Part II 45:129–163
Levin LA, Etter RJ, Rex MA, Gooday AJ, Smith CR, Pineda J, Stuart CT, Hessler RR, Pawson D (2001) Environmental influences on regional deep-sea species diversity. Annu Rev Ecol Syst 32:51–93
Levin LA, Gooday AJ (1992) Possible roles for xenophyophores in deep-sea carbon cycling. In: Rowe GT, Pariente V (eds) Deep-sea food chains and the global carbon cycle, pp 93–104
Lins L, Guilini K, Veit-Köhler G, Hauquier F, Alves RMS, Esteves AM, Vanreusel A (2014) The link between meiofauna and surface productivity in the Southern Ocean. Deep Sea Res Part II 108:60–68
Lohrer AM, Thrush SF, Gibbs MM (2004) Bioturbators enhance ecosystem function through complex biogeochemical interactions. Nature 431:1092–1095
Loreau M (2008) Biodiversity and ecosystem functioning: the mystery of the deep sea. Curr Biol 18:R126–R128
Loreau M, Naeem S, Inchausti P, Bengtsson J, Grime JP, Hector A, Hooper DU, Huston MA, Raffaelli D, Schmid B, Tilman D, Wardle DA (2001) Biodiversity and ecosystem functioning: current knowledge and future challenges. Science 294:804–808
Macheriotou L, Rigaux A, Derycke S, Vanreusel A (2020) Phylogenetic clustering and rarity imply risk of local species extinction in prospective deep-sea mining areas of the Clarion-Clipperton Fracture Zone. Proceedings of the Royal Society b: Biological Sciences 287:20192666
Majdi N, Schmid-Araya JM, Traunspurger W (2020) Examining the diet of meiofauna: a critical review of methodologies. Hydrobiologia 847:2737–2754
Meadows PS, Anderson JG (1966) Micro-organisms attached to marine and freshwater sand grains. Nature 212:1059–1060
Meadows PS, Anderson JG (1968) Micro-organisms attached to marine sand grains. J Mar Biol Assoc UK 48:161–175
Miljutin D, Gad G, Miljutina M, Mokievsky V, Fonseca-Genevois V, Esteves A (2010) The state of knowledge on deep-sea nematode taxonomy: how many valid species are known down there? Mar Biodivers 40:143–159
Miljutin DM, Miljutina MA, Arbizu PM, Galéron J (2011) Deep-sea nematode assemblage has not recovered 26 years after experimental mining of polymetallic nodules (Clarion-Clipperton Fracture Zone, Tropical Eastern Pacific). Deep Sea Res Part I 58:885–897
Mokievsky VO, Udalov AA, Azovskii AI (2007) Quantitative distribution of meiobenthos in deep-water zones of the World Ocean. Oceanology 47:797–813
Moodley L, Middelburg JJ, Boschker HTS, Duineveld GCA, Pel R, Herman PMJ, Heip CHR (2002) Bacteria and Foraminifera: key players in a short-term deep-sea benthic response to phytodetritus. Mar Ecol-Prog Ser 236:23–29
Pape E, Bezerra TN, Vanneste H, Heeschen K, Moodley L, Leroux F, van Breugel P, Vanreusel A (2011) Community structure and feeding preference of nematodes associated with methane seepage at the Darwin mud volcano (Gulf of Cadiz). Mar Ecol-Prog Ser 438:71–83
Pape E, Bezerra T, Jones DO, Vanreusel A (2013a) Unravelling the environmental drivers of deep-sea nematode biodiversity and its relation with carbon mineralisation along a longitudinal primary productivity gradient. Biogeosciences 10:3127–3143
Pape E, Jones DO, Manini E, Bezerra TN, Vanreusel A (2013b) Benthic-pelagic coupling: effects on nematode communities along Southern European continental margins. PLoS ONE 8:e59954
Pape E, van Oevelen D, Moodley L, Soetaert K, Vanreusel A, (2013c) Nematode feeding strategies and the fate of dissolved organic matter carbon in different deep-sea sedimentary environments. Deep sea research part I: oceanographic research papers
Pawlowski J, Fahrni J, Lecroq B, Longet D, Cornelius N, Excoffier L, Cedhagen T, Gooday AJ (2007) Bipolar gene flow in deep-sea benthic foraminifera. Mol Ecol 16:4089–4096
Preston FW (1962) The canonical distribution of commonness and rarity: part I. Ecology 43:185–215
Ptatscheck C, Traunspurger W (2020) The ability to get everywhere: dispersal modes of free-living, aquatic nematodes. Hydrobiologia 847(17):3519–3547
Rex MA, Etter RJ (2010) Deep-sea biodiversity: pattern and scale. Harvard Univesity Press
Rex MA, McClain CR, Johnson NA, Etter RJ, Allen JA, Bouchet P, Waren A (2005) A source-sink hypothesis for abyssal biodiversity. Am Nat 165:163–178
Rex MA, Etter RJ, Morris JS, Crouse J, McClain CR, Johnson NA, Stuart CT, Deming JW, Thies R, Avery R (2006) Global bathymetric patterns of standing stock and body size in the deep-sea benthos. Mar Ecol-Prog Ser 317:1–8
Rosenzweig ML, Abramsky Z (1993) How are diversity and productivity related? In: Rickleffs RE, Schluter D (eds) Species diversity in ecological communities: historical and geographical perspectives. University of Chicago Press, Chicago, IL, pp 52–65
Rosli N, Leduc D, Rowden AA, Probert PK (2018) Review of recent trends in ecological studies of deep-sea meiofauna, with focus on patterns and processes at small to regional spatial scales. Mar Biodivers 48:13–34
Sahraean N, Van Campenhout J, Rigaux A, Mosallanejad H, Leliaert F, Moens T (2017) Lack of population genetic structure in the marine nematodes Ptycholaimellus pandispiculatus and Terschellingia longicaudata in beaches of the Persian Gulf Iran. Marine Ecology 38:e12426
Sarmiento JL, Gruber N (2002) Sinks for anthropogenic carbon. Phys Today 55:30–36
Schratzberger M, Ingels J (2018) Meiofauna matters: the roles of meiofauna in benthic ecosystems. J Exp Mar Biol Ecol 502:12–25
Schuelke T, Pereira TJ, Hardy SM, Bik HM (2018) Nematode-associated microbial taxa do not correlate with host phylogeny, geographic region or feeding morphology in marine sediment habitats. Mol Ecol 27:1930–1951
Snelgrove PVR, Smith CR (2002) A riot of species in an environmental calm: the paradox of the species-rich deep-sea floor. In: Gibson RN, Barnes M, Atkinson RJA (eds) Oceanography and marine biology, vol 40. Taylor & Francis Ltd., London, pp 311–342
Soetaert K, Heip C (1995) Nematode assemblages of deep-sea and shelf break sites in the North-Atlantic and Mediterranean-Sea. Mar Ecol-Prog Ser 125:171–183
Soltwedel T (2000) Metazoan meiobenthos along continental margins: a review. Prog Oceanogr 46:59–84
Soltwedel T, Guilini K, Sauter E, Schewe I, Hasemann C (2018) Local effects of large food-falls on nematode diversity at an arctic deep-sea site: results from an in situ experiment at the deep-sea observatory HAUSGARTEN. J Exp Mar Biol Ecol 502:129–141
Taylor M, Roterman C (2017) Invertebrate population genetics across Earth’s largest habitat: the deep-sea floor. Mol Ecol 26:4872–4896
Tchesunov AV, Ingels J, Popova EV (2012) Marine free-living nematodes associated with symbiotic bacteria in deep-sea canyons of north-east Atlantic Ocean. J Mar Biol Assoc UK 92:1257–1271
Thiel H (1975) The size-structure of the deep-sea benthos. Internationale Revue Der Gesamten Hydrobiologie 60
Thistle D, Sedlacek L, Carman KR, Fleeger JW, Barry JP (2007) Emergence in the deep sea: evidence from harpacticoid copepods. Deep Sea Res Part 1 Oceanogr Res Pap 54:1008–1014
Van Gaever S, Moodley L, de Beer D, Vanreusel A (2006) Meiobenthos at the Arctic Hakon Mosby Mud Volcano, with a parental-caring nematode thriving in sulphide-rich sediments. Mar Ecol-Prog Ser 321:143–155
Van Gaever S, Moodley L, Pasotti F, Houtekamer M, Middelburg J, Danovaro R, Vanreusel A (2009) Trophic specialisation of metazoan meiofauna at the Håkon Mosby Mud Volcano: fatty acid biomarker isotope evidence. Mar Biol 156:1289–1296
van Oevelen D, Soetaert K, Middelburg JJ, Herman PMJ, Moodley L, Hamels I, Moens T, Heip CHR (2006) Carbon flows through a benthic food web: Integrating biomass, isotope and tracer data. J Mar Res 64:453–482
van Oevelen D, Bergmann M, Soetaert K, Bauerfeind E, Hasemann C, Klages M, Schewe I, Soltwedel T, Budaeva NE (2011a) Carbon flows in the benthic food web at the deep-sea observatory HAUSGARTEN (Fram Strait). Deep Sea Res Part I 58:1069–1083
van Oevelen D, Soetaert K, Garcia R, de Stigter HC, Cunha MR, Pusceddu A, Danovaro R (2011b) Canyon conditions impact carbon flows in food webs of three sections of the Nazaré canyon. Deep Sea Res Part II 58:2461–2476
Vanreusel A, VandenBossche I, Thiermann F (1997) Free-living marine nematodes from hydrothermal sediments: similarities with communities from diverse reduced habitats. Mar Ecol-Prog Ser 157:207–219
Vanreusel A, De Groote A, Gollner S, Bright M (2010) Ecology and biogeography of free-living nematodes associated with chemosynthetic environments in the deep sea: a review. PLoS ONE 5:e12449
Veit-Köhler G, Guilini K, Peeken I, Sachs O, Sauter EJ, Würzberg L (2011) Antarctic deep-sea meiofauna and bacteria react to the deposition of particulate organic matter after a phytoplankton bloom. Deep Sea Res Part II 58:1983–1995
Vermeeren H, Vanreusel A, Vanhove S (2004) Species distribution within the free-living marine nematode genus Dichromadora in the Weddell Sea and adjacent areas. Deep-Sea Research Part II: Topical Studies in Oceanography 51:1643–1664
Wei CL, Rowe GT, Escobar-Briones E, Boetius A, Soltwedel T, Caley MJ, Soliman Y, Huettmann F, Qu FY, Yu ZS, Pitcher CR, Haedrich RL, Wicksten MK, Rex MA, Baguley JG, Sharma J, Danovaro R, MacDonald IR, Nunnally CC, Deming JW, Montagna P, Levesque M, Weslawski JM, Wlodarska-Kowalczuk M, Ingole BS, Bett BJ, Billett DSM, Yool A, Bluhm BA, Iken K, Narayanaswamy BE (2010) Global patterns and predictions of seafloor biomass using random forests. Plos One 5
Wigley RL, McIntyre AD (1964) Some quantitative comparisons of offshore meiobenthos and macrobenthos south of Martha’s Vineyard. Limnol Oceanogr 9:485–493
Witte U, Aberle N, Sand M, Wenzhofer F (2003a) Rapid response of a deep-sea benthic community to POM enrichment: an in situ experimental study. Mar Ecol-Prog Ser 251:27–36
Witte U, Wenzhofer F, Sommer S, Boetius A, Heinz P, Aberle N, Sand M, Cremer A, Abraham WR, Jorgensen BB, Pfannkuche O (2003b) In situ experimental evidence of the fate of a phytodetritus pulse at the abyssal sea floor. Nature 424:763–766
Woolley SNC, Tittensor DP, Dunstan PK, Guillera-Arroita G, Lahoz-Monfort JJ, Wintle BA, Worm B, O’Hara TD (2016) Deep-sea diversity patterns are shaped by energy availability. Nature 533:393–396
Worm B, Barbier EB, Beaumont N, Duffy JE, Folke C, Halpern BS, Jackson JBC, Lotze HK, Micheli F, Palumbi SR, Sala E, Selkoe KA, Stachowicz JJ, Watson R (2006) Impacts of biodiversity loss on ocean ecosystem services. Science 314:787–790
Yasuhara M, Danovaro R (2016) Temperature impacts on deep-sea biodiversity. Biol Rev 91:275–287
Zardus JD, Etter RJ, Chase MR, Rex MA, Boyle EE (2006) Bathymetric and geographic population structure in the pan-Atlantic deep-sea bivalve Deminucula atacellana (Schenck, 1939). Mol Ecol 15:639–651
Zeppilli D, Leduc D, Fontanier C, Fontaneto D, Fuchs S, Gooday AJ, Goineau A, Ingels J, Ivanenko VN, Kristensen RM, Neves RC, Sanchez N, Sandulli R, Sarrazin J, Sørensen MV, Tasiemski A, Vanreusel A, Autret M, Bourdonnay L, Claireaux M, Coquillé V, De Wever L, Rachel D, Marchant J, Toomey L, Fernandes D (2018) Characteristics of meiofauna in extreme marine ecosystems: a review. Mar Biodivers 48:35–71
Zeppilli D, Bellec L, Cambon-Bonavita M-A, Decraemer W, Fontaneto D, Fuchs S, Gayet N, Mandon P, Michel LN, Portail M, Smol N, Sørensen MV, Vanreusel A, Sarrazin J (2019) Ecology and trophic role of Oncholaimus dyvae sp. nov. (Nematoda: Oncholaimidae) from the lucky strike hydrothermal vent field (Mid-Atlantic Ridge). BMC Zoology 4:6
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Ingels, J., Leduc, D., Zeppilli, D., Vanreusel, A. (2023). Deep-Sea Meiofauna—A World on Its Own or Deeply Connected?. In: Giere, O., Schratzberger, M. (eds) New Horizons in Meiobenthos Research. Springer, Cham. https://doi.org/10.1007/978-3-031-21622-0_8
Download citation
DOI: https://doi.org/10.1007/978-3-031-21622-0_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-21621-3
Online ISBN: 978-3-031-21622-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)