Abstract
Inland aquaculture practice is becoming popular throughout the world to suffice the increasing protein demand of the growing population. Aquaculture ponds in general emit methane (CH4) towards the atmosphere. However, available data are scarce from India, where the number of aquaculture plots is growing at a fast pace. We measured the partial pressure of CH4 in surface water [pCH4(w)], the atmosphere-pond CH4 fluxes, and several relevant biogeochemical parameters in sewage–fed freshwater (FWP) and oligohaline (OHP) aquaculture ponds situated in an eastern Indian wetland. We hypothesized that pCH4(w) and the atmosphere-pond CH4 effluxes would significantly vary between FWP and OHP as salinity plays a crucial role in regulating the methanogens in any water column. Measurements were carried out in both FWP and OHP throughout an annual cycle. FWP and OHP emitted CH4 at the rate of 22.4 ± 16.2 mg m−2 h−1 and 13.4 ± 13.6 mg m−2 h−1, respectively. Apart from low salinity, turbidity was higher in FWP, which in turn led to reduced photosynthetic activities and lower dissolved oxygen levels compared to OHP. pH was also substantially lower in FWP compared to OHP. More anaerobic and low pH conditions in FWP compared to OHP favored methanogenic activities and methane oxidation was discouraged, which led to higher atmosphere-pond CH4 fluxes from FWP compared to OHP. However, both FWP and OHP exhibited annual mean CH4 effluxes much higher than the efflux rates observed in most of the Chinese aquaculture ponds.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Methane emission
- GHG
- Aquaculture
- Sewage–fed
- Freshwater
- Brackish water
- Wetland
- East Kolkata Wetland
- Sundarban Biosphere Reserve
1 Introduction
Aquaculture ponds have become an essential land-use class and encompass a substantial part of the surface water ecosystems of the Earth (Yang et al., 2018a). Since the 1970s, aquaculture ponds have come up as an alternative to capture fisheries and it has been serving well to meet the ever-increasing demand for aquatic foods like fish, shrimp, crabs, etc. (Hu et al., 2012). Distributed over a wide range of tropical to temperate regions, the freshwater and brackish water aquaculture ponds comprise an area of about 1,10,832 km2 throughout the world (Verdegem & Bosma, 2009). However, like many other inland lentic ecosystems (e.g. lakes, ponds, reservoirs), aquaculture ponds have been also found to emit a substantial quantity of carbon dioxide, methane, and nitrous oxides (Boyd et al., 2010; Chen et al., 2016; Yang et al., 2015a). Among the several greenhouse gases, methane (CH4) emission has perhaps received the highest attention (Bastviken et al., 2011; Hu et al., 2014, 2016). The aquaculture ponds receive a substantial amount of organic load which when remains unutilized acts as a substrate for the microbes to act upon and under anaerobic conditions, the methanogens produce a substantial amount of CH4 from it (Yang et al., 2019). According to the estimates made at the beginning of the present decade, inland aquatic bodies are capable of emitting 650 Tg C year−1 in the form of methane (Bastviken et al., 2011). However, it is believed that these magnitudes include considerable uncertainty as the CH4 emissions from shallow aquaculture ponds are mostly not considered while drawing these estimates (Long et al., 2016; Yang et al., 2018a). Moreover, field observations of CH4 emission are still very few and mostly concentrated in Chinese pisciculture plots (Chen et al., 2016; Long et al., 2016; Yang et al., 2015a, 2018b, 2019).
CH4 transport from the water column of any aquatic ecosystem towards the atmosphere mainly takes place through either diffusion (Chen et al., 2016) or ebullition (Dutta et al., 2013). A wide range of biotic and abiotic factors are known to regulate the production of CH4 by the methanogens and its consumption by methanotrophs which in turn govern the partial pressure of CH4 in surface water [pCH4(w)] and hence the atmosphere-pond CH4 flux (Yang et al., 2019). Earlier pieces of research have highlighted that water temperature plays a critical role in governing the CH4 biogeochemistry (Knox et al., 2016; Olsson et al., 2015; Palma‐Silva et al., 2013). Factors like pH (Hu et al., 2017), dissolved oxygen (DO) (Liu et al., 2015), primary productivity (Xiao et al., 2017), water table (Yang et al., 2013) and substrate availability (Venkiteswaran et al., 2013) also regulates the water column CH4 production. In addition to these factors, one of the most important and decisive factors that substantially alters the pCH4(w) is the salinity of the water (Hu et al., 2017; Vizza et al., 2017; Welti et al., 2017), based on which aquaculture ponds are differentiated into freshwater and brackish water categories. An increase in salinity is often found to reduce pCH4(w) and hence lead to lower atmosphere-pond CH4 fluxes (Yang et al., 2018b). Earlier studies exhibited that higher salinity leads to ion stress to methanogens (Chambers et al., 2013; Neubauer et al., 2013). It provides alternative electron acceptors like sulfate ion to the water medium, which in turn suppresses the methanogens from producing CH4 in the water column (Laanbroek, 2010; Sun et al., 2013). So far, very few attempts have been made to analyze the difference in pCH4(w) dynamics in aquaculture ponds of varying salinity (Yang et al., 2018a, 2019) and most of these studies were carried out in highly saline mariculture ponds. Moreover, almost all of the studies where pCH4(w) dynamics of aquaculture ponds are characterized are carried out in such aquaculture ponds where daily fish feeds are provided. Comparisons between sewage-fed freshwater aquaculture ponds and oligohaline aquaculture ponds are not at all available at the present date.
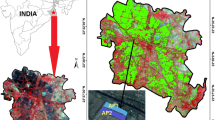
Keeping in view this background, the present research work was carried out to expand the knowledge by quantifying atmosphere-pond CH4 fluxes from sewage fed freshwater as well as an oligohaline aquaculture pond in the eastern part of India, located in East Kolkata Wetlands (EKW), and Minakhan block, within the Sundarban Biosphere Reserve (SBR), respectively (West Bengal, India). It is worth mentioning that aquaculture practice is steadily increasing in India and they encompass a substantial area (7,900 km2) of India’s total areal extent (Adhikari et al., 2012). However, endeavors of quantifying the atmosphere-pond CH4 fluxes from such water bodies are very few (Adhikari et al., 2012; Pathak et al., 2013). This is why; this data set generated from this study is expected to contribute to the global database of CH4 fluxes from aquaculture ponds. Atmosphere-pond CH4 fluxes have been found to exhibit potential variations in different seasons (Heyer & Berger, 2000). Thus we have carried out sampling all-round the year covering three seasons [monsoon season (June, July, August, and September), pre-monsoon season (February, March, April, and May), and post-monsoon season (October, November, December, and January)] for this piece of research. We hypothesized that pCH4(w) dynamics and hence atmosphere-pond CH4 flux would significantly vary between freshwater and an oligohaline aquaculture pond situated close and experience the same climate conditions due to different salinity. Following this hypothesis, the main aims of this research were to (i) characterize and compare the pCH4(w) and atmosphere-pond CH4 fluxes from freshwater as well as an oligohaline aquaculture pond throughout an annual cycle, (ii) examine seasonal variations of CH4 fluxes from the two aquaculture ponds and (iii) characterize the relationship between pCH4(w) and the associated biogeochemical factors, with special emphasis on salinity.
2 Methodology
2.1 Study Sites
The EKW (Fig. 1) lies on the east of the Kolkata metropolis. Being tagged as a ‘wetland of international importance’, EKW found its place in the list of Ramsar Sites in 2002. EKW is known to be the ‘kidney of the city of Kolkata’ because of its unique natural purification system (Kundu et al., 2008). This wetland complex stands tall as the largest conglomeration of human-built pisciculture ponds in the world. EKW receives the wastewater load from the adjacent city of Kolkata and treats the water mass through activities like pisciculture, agriculture, and solid waste farms. In this way, the bulk sewage water load of Kolkata is naturally treated. The sewage canal flows into the Bidyadhari River that ends in the Bay of Bengal through the Sundarban mangrove ecosystem. According to the estimates of Aich et al. (2012), EKW has almost 250 functional aquaculture ponds that encompass 12 km2. This system altogether produces substantial fish and vegetables that engage close to 0.5 million people and acts as the primary food source to the residents of the metropolis (Chaudhuri et al., 2012). EKW’s performance has continued in this fashion since the late eighteenth century and it has been acting as an economic, ecosystem-resilient, and effective system of both aquatic and solid waste management (Kundu et al., 2008). These ponds are usually very shallow (1 to 1.5 m depth). The fishermen maintain a flat bottom in these ponds. These ponds vary in size from 10,000 m2 to 100,000 m2. The infrastructural characteristics of these ponds are portrayed in detail by Ghosh and Furedy (1984) and Ghosh (2005). The concept behind this natural engineering is discussed by Chaudhuri et al. (2007) and Chaudhuri et al. (2008). All the ponds within this system are mostly freshwater as a mixture of groundwater and sewage water is utilized for pisciculture in this setup.
The OHP which was sampled in this study is situated in the Minakhan community development Block situated almost 25 km to the east of EKW near the bank of Bidhyadhari River. This block is a part of the SBR, known to shelter the world’s largest continuous stretch of mangrove forest. Local people of this region mainly practiced agriculture since the 1970s (Naskar, 1985), however, after the construction of dikes and embankments by the Department of Irrigation, Govt. of West Bengal people started switching for aquaculture with the help of the oligohaline water flowing through the Bidyadhari River (Bunting et al., 2017). Coupled rice–shrimp farming is quite popular in Minakhan. In the present date, the number of pisciculture ponds has observed a drastic increase in the Minakahn Block (Mondal & Bandyopadhyay, 2015).
2.2 Sampling Strategy
Samples were collected once a month covering the entire annual cycle from March 2018 to February 2019. Sampling was conducted in two aquaculture ponds; one situated within the EKW (freshwater pond) and the other in Minakhan Block (oligohaline pond) [hereafter referred to as FWP (22.514744 N, 88.482124 E) (depth: 1.2 m; area: ~ 45,000 m2) and OHP (22.50693 N, 88.75452 E) (depth: 3 m; area: 54,000 m2) respectively]. Samples were collected at every 2 h intervals over a complete diel cycle. The ambient temperature, wind velocity, and atmospheric pressure were measured by deploying a portable weather station. The water surface physicochemical parameters were monitored in-situ using typical probes. For other parameters like CH4 concentration in water and air, chlorophyll–a (chl–a), and biochemical oxygen demand (BOD), samples were retrieved and relocated to the laboratory after taking necessary measures of preservation.
2.3 Pisciculture in FWP and OHP
Oreochromis nilotica (Tilapia) and Penaeus monodon (Tiger prawn) was cultured in FWP and OHP respectively. Unlike other aquaculture ponds, no external fish feed is used in these ponds. In the EKW ponds, the organic detritus of the sewage are utilized by the fish as their food throughout the year. However, during the monsoon season, the sewage load sometimes becomes overdiluted and can not provide sufficient food to the fish. Bunting et al. (2010) mentioned that under such occasional circumstances, external fish feed is deployed by the fisher community. Quantifying the total quantity of feed or the feed conversion ratio is an almost impossible endeavor in EKW ponds, as the quality and quantity of sewage vary significantly over a short-term temporal scale (Chanda et al., 2019). Ponds of the Minakhan area utilize the oligohaline water Bidhyadhari River channelized through lock gates to maintain the salinity levels. The fisher community of this region practices variable stocking density and periodically harvest fish at new moon and full moon phases of the lunar cycle (Alagarswamy, 1995; De Roy, 2012).
2.4 Biogeochemical Analysis
2.4.1 Ancillary Environmental Measurements
Atmospheric temperature and wind velocity were recorded by a field-operable weather station (WS–2350, La Crosse Technology, USA). Electrical conductivity (EC) (precision: 1 µS/cm) and water surface temperature (precision: 0.1 ºC) were recorded by a digital EC meter (Thermo Scientific, Eutech, Germany). Dissolved oxygen (DO) (accuracy: ± 1%; precision: 0.01 mg l−1) was recorded using a FiveGo portable F4 Dissolved Oxygen meter, Mettler Toledo. The DO readings were cross-checked by performing Winkler’s titration. pH was monitored by an Orion PerpHecT ROSS Combination pH Micro Electrode fitted to a micro–pH data reader (Thermo Scientific, USA) (analytical resolution – 0.001; precision – 0.009). NBS scale technical buffer solutions were used to calibrate the glass-calomel electrodes. Nephelometric turbidity was measured with Eutech TN–100 turbidity meter. Determination of underwater photosynthetically active radiation (UWPAR) was carried out using LI–192SA, LiCor, USA (precision 0.1 µ mol m−2 s−1) and a data reader (Li–250A, LiCor, USA). Quantification of chl–a was carried out using a spectrophotometer (precision 0.01 mg m−3). The community respiration (CR) and gross primary productivity (GPP) in both the ponds were monitored by the standard light-bottle-dark-bottle incubation method. A 24-h incubation was followed to monitor the alterations in DO concentrations. BOD was estimated by incubating water samples from each pond at 27 °C for 3 days. Chl–a, BOD, CR, and GPP were quantified according to the protocols of APHA (2005).
2.4.2 Measuring CH4 Concentrations in Water and Air
Surface water from FWP and OHP was directly filled in 40 ml glass ampoules equipped with a latex septum. No headspace was left during the sampling. Supersaturated HgCl2 solution (100 µl) was pushed through the septum to cease all microbial activities till further analysis. Before analysis, half of the sample was injected out of the vial and 99.99% pure nitrogen gas was used to purge the remaining half to equilibrate the sample. The samples were equilibrated for 2 h. A Hamilton syringe was used to collect the headspace gas (5 ml). The collected gas was flown through a gas chromatograph (GC) (Systronics GC–8205) to estimate CH4 concentrations. The uncertainty in estimation was ± 2.9%. The carrier gas was pure nitrogen and the retention time for CH4 was 37s. Moisture removal was done from the system by enhancing the injector temperature to 105 °C. The GC was regularly calibrated by reference standard CH4 gas of known concentrations. A battery-operated pump was attached to a glass sampling bulb to draw in air samples from above the pond water interface. The glass bulbs were carefully evacuated and washed with distilled water before sampling. While bringing the air samples to the laboratory, parafilm coverings were used to seal the knobs and outlets. These samples were analyzed in GC using the same method discussed above.
2.4.3 Air–water CH4 Flux Estimation
pCH4(w) and pCH4(a) were transformed to concentrations of CH4 in surface water (CH4wc) and air (CH4ac) as per the Eqs. (1) and (2) (Morel, 1983) and (3) (Lide, 2007).
where KH stands for the gas partition coefficient of CH4 in water at sampling temperature, expressed in mole l−1 atm−1, and TK refers to the temperature (Kelvin).
The CH4 flux is estimated as per Eq. (4) (MacIntyre et al., 1995).
where kx denotes the mass transfer coefficient (cm h−1) and it is computed according to Eq. (5) (Wanninkhof, 1992)
where Sc is the Schmidt number for CH4. It depends on water temperature as per Eq. (6). k600 is computed from the wind velocity (U10), as per Cole and Caraco (1998) (Eq. 7) and ‘x’ = 0.66 for wind speed ≤ 3 m s−1 and ‘x’ = 0.5 for wind speed > 3 m s−1.
2.4.4 Statistical Computations
We carried out a one-way analysis of variance (ANOVA) to test whether the seasonality exhibited by all the parameters in each of the ponds is statistically significant or not. Independent samples Student’s t-test was applied to examine the difference in the average of all the parameters between FWP and OHP. Pearson correlation coefficients were computed to study the relationship between pCH4(w) and the measured physicochemical parameters. We used the SPSS version 16.0 (SPSS, Inc., USA) to carry out these analyses. 95% confidence level (p < 0.05) was set as the threshold in this study to determine the statistical significance.
3 Results
3.1 Variability of Physicochemical Parameters
Seasonal mean pH values were higher in OHP compared to FWP in all the seasons [pre–monsoon: 8.189 ± 0.096 (FWP) and 8.291 ± 0.108 (OHP); monsoon: 8.030 ± 0.066 (FWP) and 8.071 ± 0.068 (OHP); post–monsoon: 8.187 ± 0.089 (FWP) and 8.273 ± 0.122 (OHP)] (Table 1). The difference in seasonal pH between OHP and FWP were significant in all the seasons (pre–monsoon: t = −4.9, p < 0.001; monsoon: t = −3.0, p = 0.003; post–monsoon: t = −3.9, p < 0.001). The seasonal variability in pH within FWP (F = 55.7, p < 0.001) and OHP (F = 69.3, p < 0.001) were also statistically significant (Fig. 2a).
The EC values were also significantly different in OHP and FWP in all the seasons (see Table 1) with almost 5 to 7 times higher values in OHP compared to FWP. EC varied in FWP from 434 µS cm−1 to 1403 µS cm−1, whereas, in OHP it varied from 3018 µS cm−1to 6267µS cm−1. The seasonal variability in EC was statistically significant in both FWP (F = 46.1, p < 0.001) and OHP (F = 30.8, p < 0.001) with considerably lower values during monsoon season compared to the other two seasons (Fig. 2b).
The seasonal mean water temperature was found highest during the monsoonal months, followed by the pre-monsoonal months and the lowest was observed during the post-monsoonal months. The seasonal variability in water temperature was statistically significant in both FWP (F = 86.8, p < 0.001) and OHP (F = 82.5, p < 0.001). However, water temperature did not exhibnit any statistical difference between FWP and OHP in any of the seasons (pre–monsoon: t = −0.02, p = 0.981; monsoon: t = −0.14, p = 0.883; post–monsoon: t = −0.02, p = 0.986) (Fig. 2c).
The seasonal mean DO concentration was significantly higher in OHP (pre–monsoon: 8.3 ± 1.4 mg l−1; monsoon: 8.0 ± 1.3 mg l−1; post–monsoon: 9.1 ± 1.5 mg l−1) compared to FWP (pre–monsoon: 9.4 ± 1.5 mg l−1; monsoon: 8.0 ± 1.3 mg l−1; post–monsoon: 9.8 ± 1.4 mg l−1) during pre-monsoonal months (t = −3.8, p < 0.001) and post-monsoonal months (t = −2.57, p = 0.011), however, no significant difference (t = −0.05, p = 0.957) was observed during the monsoonal months. The seasonal difference in DO was statistically significant for both FWP (F = 7.8, p = 0.001) and OHP (F = 22.6, p < 0.001) (Fig. 2d).
Like pH and EC, UWPAR exhibited higher magnitudes in OHP compared to FWP in all the three seasons, however, the difference was not statistically significant in any of the three seasons (pre–monsoon: t = −0.99, p = 0.326; monsoon: t = −2.0, p = 0.052; post–monsoon: t = −1.9, p = 0.062). In monsoon and post–monsoon seasons the p–value was marginally higher than 0.05. However, the seasonal difference in UWPAR within FWP (F = 18.2, p < 0.001) and OHP (F = 16.4, p < 0.001) was statistically significant (Fig. 2e).
BOD in FWP was significantly higher than OHP in all the three seasons (pre–monsoon: t = 13.8, p < 0.001; monsoon: t = 9.3, p < 0.001; post–monsoon: t = 14.8, p < 0.001). Over the annual cycle, BOD ranged between 6.5 mg l−1and 9.2 mg l−1 in OHP, whereas, it varied between 9.4 mg l−1and 14.7 mg l−1 in FWP. The seasonal difference in BOD was also statistically significant within FWP (F = 18.2, p < 0.001) and OHP (F = 7.3, p = 0.002) (Fig. 2f).
3.2 Variability of Primary Productivity-Related Parameters
The chl–a concentration was higher in FWP compared to OHP in all the seasons, however, the difference was significant during pre–monsoon season (t = 7.4, p < 0.001) and post–monsoon season (t = 4.5, p < 0.001). During monsoon season the p–value was marginally not significant (t = 1.9, p = 0.059). The inter–seasonal variation in chl–a was statistically significant in case of FWP (F = 5.1, p = 0.012), however, in OHP there was no significant variation over the annual cycle (F = 1.8, p = 0.172) (Fig. 3a).
There was significant difference in turbidity between FWP and OHP in all the three seasons (pre–monsoon: t = 9.5, p < 0.001; monsoon: t = 5.1, p < 0.001; post–monsoon: t = 5.2, p < 0.001), with higher values in FWP compared to OHP. The seasonal mean difference in turbidity varied from ~ 6 NTU to ~ 11 NTU. The seasonal variability of turbidity was significant in FWP (F = 5.8, p = 0.007), however, like chl–a, it was not significant in OHP (F = 2.4, p = 0.110) (Fig. 3b).
The difference in GPP between FWP and OHP was statistically significant in pre–monsoon season (t = 3.6, p = 0.001) and monsoon season (t = 2.4, p = 0.024), however, during post–monsoon season the difference was not significant (t = −0.32, p = 0.747). Though the difference in GPP was significant in two seasons, in terms of magnitude, the mean difference ranged from 0.4 gO2 m−2 d−1 to 0.6 gO2 m−2 d−1. In contrast to chl–a, the inter–seasonal variation in GPP was not significant in FWP (F = 0.31, p = 0.733), however, it was statistically significant in case of OHP (F = 22.6, p < 0.001) (Fig. 3c).
Unlike GPP, CR was significantly higher in FWP compared to OHP in all the three seasons (pre–monsoon: t = 5.8, p < 0.001; monsoon: t = 23.6, p < 0.001; post–monsoon: t = 12.1, p < 0.001). The magnitude of difference varied from 6gO2 m−2 d−1 to 11gO2 m−2 d−1, which was much higher than the difference in GPP between FWP and OHP. At the same time, the seasonal variation within FWP (F = 20.1, p < 0.001) and OHP (F = 19.2, p < 0.001) was also statistically significant (Fig. 3d).
3.3 Variability in pCH4(a), pCH4(w) and Air–Water CH4 Flux
pCH4(a) varied over a very short range of 1.843 ppmv to 1.888 ppmv, however, it was marginally higher near FWP compared to OHP in all the three seasons (pre–monsoon: t = 3.3, p = 0.002; monsoon: t = 3.7, p < 0.001; post–monsoon: t = 3.1, p < 0.001). pCH4(a) also exhibited significant seasonal variation near FWP (F = 21.5, p < 0.001) as well as OHP (F = 15.2, p < 0.001) (Fig. 4a).
Seasonal mean pCH4(w) followed the trend monsoon > pre–monsoon > post–monsoon in both FWP and OHP, with significantly higher values in FWP compared to OHP (pre–monsoon: t = 7.5, p < 0.001; monsoon: t = 4.6, p < 0.001; post–monsoon: t = 5.6, p < 0.001). The seasonal difference in mean pCH4(w) was statistically significant in both FWP (F = 8.1, p < 0.001) and OHP (F = 4.9, p = 0.009) (Fig. 4a).
Both FWP (22.4 ± 16.2 mg m−2 h−1) and OHP (13.4 ± 13.6 mg m−2 h−1) acted as source of CH4 towards atmosphere throughout the year, with significantly higher values of CH4 efflux from FWP compared to OHP in all the seasons (pre–monsoon: t = 4.2, p < 0.001; monsoon: t = 3.1, p = 0.002; post–monsoon: t = 3.7, p < 0.001). Mirroring the trend of seasonal mean pCH4(w), air–water CH4 flux also followed the same trend monsoon > pre–monsoon > post–monsoon in both FWP and OHP. The inter–seasonal difference in air–water CH4 flux was statistically significant in both FWP (F = 27.3, p < 0.001) and OHP (F = 15.9, p < 0.001) (Fig. 4b).
3.4 Relationship Between pCH4(w) and Biogeochemical Variables
pH exhibited significant negative relationship with pCH4(w) in both FWP (r = −0.62, p = 0.031) and OHP (r = −0.68, p = 0.015) (Fig. 5a). EC showed significant positive relationship with pCH4(w) in FWP (r = 0.72, p = 0.008), however, in OHP the relationship was not significant (r = 0.38, p = 0.220) (Fig. 5b). Water temperature exhibited very strong positive relationship with pCH4(w) in both FWP (r = 0.91, p < 0.001) and OHP (r = 0.94, p < 0.001) (Fig. 5c). The relationship between DO and pCH4(w) was significantly negative in both FWP (r = −0.86, p < 0.001) and OHP (r = −0.83, p = 0.001) (Fig. 5d). Like water temperature, BOD also showed significant positive relationship with pCH4(w) in both FWP (r = 0.81, p = 0.001) and OHP (r = 0.65, p = 0.021) (Fig. 5e). Chl–a exhibited significant positive relationship with pCH4(w) in OHP (r = 0.86, p < 0.001), however, the relationship was not significant in case of FWP (r = 0.50, p = 0.098) (Fig. 5f). Turbidity and GPP did not exhibit any significant relationship with pCH4(w) (Fig. 5g,h). CR showed significant positive relationship with pCH4(w) in OHP (r = 0.83, p = 0.001), but in FWP it was marginally beyond the significance limit (r = 0.56, p = 0.057) (Fig. 5i).
The scatter plots displaying the relationship between monthly mean water CH4 concentration [pCH4(w)] and monthly mean a pH, b electrical conductivity, c water temperature, d DO, e BOD, f chl–a, g turbidity, h GPP, and i CR. Linear trend lines along with the goodness of fit (R2) are shown separately for FWP and OHP
3.5 Diurnal Variation in pCH4(w) and Atmosphere-Pond CH4 Flux
During all the seasons the pCH4(w) and hence the atmosphere-pond CH4 efflux exhibited a steady increase from dawn till noon and the peak was observed during 1400 h to 1600 h (Fig. 6). During night time both the pCH4(w) and the atmosphere-pond CH4 efflux was much lower compared to the day time.
4 Discussion
Analyzing the results it can be observed that OHP had significantly lower pCH4(w) and atmosphere-pond CH4 fluxes compared to FWP all-around the year. In terms of physicochemical variables, the difference in salinity was the major reason behind choosing these two ponds and comparing their dissolved CH4 dynamics. EC values in OHP were substantially higher than FWP throughout the year and this could be the most crucial factor which led to lower pCH4(w) in OHP due to reduced methanogenesis; as also observed in earlier studies like Poffenbarger et al. (2011) and Welti et al. (2017). Thus we could accept our hypothesis that a difference in salinity (or EC) leads to a difference in pCH4(w) and atmosphere-pond CH4 fluxes between OHP and FWP. However, unlike previous studies like Cotovicz et al. (2016) and Yang et al., (2018a, 2019), no significant negative relationship was observed between EC and pCH4(w) in neither FWP nor OHP. This shows that there are some other factors as well which are regulating the pCH4(w) in these two ponds apart from EC.
Water temperature has been regarded as one of the crucial factors which govern both methanogenesis (Inglett et al., 2012; Yang et al., 2015b) and methane oxidation (Lofton et al., 2014; Osudar et al., 2015). In the present study, a strong positive association between water temperature and pCH4(w) was portrayed in both the ponds, and no significant difference was observed in water temperature as well between the two ponds. Hence it can be affirmed in both the ponds increase in water temperature aided methanogenesis by enhancing organic matter degradation and hence providing suitable labile substrates from which CH4 is produced by microbial activity. Similar observations were made by Xiang et al. (2015) in coastal marshes and Yang et al. (2019) in aquaculture ponds. The difference in pCH4(w) magnitudes between FWP and OHP despite having the same water temperature further shows that methane oxidation is not prompted by the effect of temperature fluctuation, rather the intrinsic difference in water column substrates led to the difference in pCH4(w) between FWP and OHP as also observed by Roland et al. (2017).
Earlier studies emphasized that the growth and thriving of the methanogens are quite dependent on the pH of the aquatic column, and they prefer to grow at lower pH close to ~ 7.7 (Chang & Yang, 2003). The pH all over the annual cycle was significantly lower in FWP compared to OHP, which could have facilitated better growth of methanogens in FWP compared to OHP. This reason could be further ascertained as both the ponds exhibited significant negative relation between pH and pCH4(w), which showed that with decreasing pH, methanogens flourished and led to enhancement of pCH4(w). Datta et al. (2009) also recorded a similar negative association between pH and pCH4(w) while working in a rain-fed fish farming pond situated in Eastern India. Yang et al. (2018b) also observed a similar relationship in aquaculture ponds of China and attributed the reduced activity of methanogens behind such observations.
The present study also showed that OHP had significantly higher DO levels compared to FWP. This could be attributed to the lower turbidity and higher UWPAR in OHP which in turn facilitated a higher degree of autotrophic activities and hence higher production of DO compared to that in FWP. Several studies have observed that Oreochromis nilotica which is cultured in FWP is a fast-moving bottom feeder (Adeyemi et al., 2009; Jihulya, 2014; Njiru et al., 2004) and the strong fish movement in the sediment–water interface often causes enhanced turbidity (Chapman & Fernando, 1994; Frei & Becker, 2005). Penaeus monodon, on the other hand, being cultured in OHP is also known to be a bottom feeder but shrimps and prawns are usually more sluggish than fish species, hence the bottom churning due to their movement is expected to be quite less. This differential photosynthesis-induced difference in DO content between the two ponds could play a crucial role in having different pCH4(w) magnitudes between FWP and OHP. Kettunen et al. (1999) and Yang et al. (2013) reported that reduced DO levels promote methane production by enhancing anaerobic decomposition rates and at the same time lower DO levels reduce methane oxidation.
BOD serves as a proxy of biodegradable organic matter in any aquatic column and the present study observed significantly higher BOD in FWP compared to OHP. FWP utilizes sewage water to carry out fishing practice and since BOD concentration in city sewage remains very high, FWP reflected the higher levels of BOD (Sarkar et al., 2017). On the contrary, OHP utilizes the brackish water of the adjacent Bidhyadhari River, which though receives the sewage water of the Kolkata metropolis but the BOD levels are quite reduced when the sewage effluent reaches Bidhyadhari River after passing through EKW (Ghosh, 2018). Higher BOD in any aquatic ecosystem indicates the presence of the labile biodegradable substance and under reduced DO and lower pH levels and leads to a higher rate of methane emission [as also observed by Yang (1998) while working in the rivers and lakes of Taiwan].
The effect of lower DO and higher BOD was also reflected on the CR of the two selected ponds. Though both FWP and OHP showed CR magnitudes greater than the magnitudes of GPP, CR in FWP was significantly higher than OHP, which further indicated that the degree of net heterotrophy was substantially higher in FWP. Previous studies have clearly shown that there is a strong relationship between net heterotrophic conditions and supersaturation of CH4, especially in small ponds like that of the chosen aquaculture ponds (Holgerson, 2015). It should be also mentioned in this regard, that chl–a concentrations were also substantially higher in FWP compared to OHP. Chl–a magnitude in any lentic ecosystem acts as a proxy of trophic status and provides an idea about the primary productivity in shallow lentic ecosystems (Liu et al., 2017; Yang et al., 2015a). Higher chl–a magnitudes indicate higher algal production rates which in turn consequences the creation of autochthonous organic substrates (Palma-Silva et al., 2013). Chl–a and GPP portrayed a positive (significant) relation with the pCH4(w) clearly emphasizing that the autochthonous production of organic substrates facilitated methanogenesis (Flury et al., 2010; Furlanetto et al., 2012) and hence facilitated higher methane emission.
In terms of the annual mean magnitude of atmosphere-pond CH4 efflux, the methane emission observed in this piece of research (FWP: 22.4 ± 16.2 mg m−2 h−1 and OHP: 13.4 ± 13.6 mg m−2 h−1) was found higher than many of the recent measurements. Datta et al. (2009) observed a mean CH4 emission of 2.5 mg m−2 h−1 from the refuge rain-fed ponds of Cuttack, India. In Chinese aquaculture ponds, most of the recent estimates exhibited lower magnitudes than those observed in the present study. Wu et al. (2018) observed a mean CH4 emission of only 0.5 mg m−2 h−1in the experimental farm of Nanjing Agricultural University, China. Yang et al (2018b) observed a mean CH4 emission of 1.1 ± 0.9 mg m−2 h−1 and 10.5 ± 4.9 mg m−2 h−1from the undrained and drained ponds of Shanyutan Wetlands, China. Yang et al. (2015a) recorded mean CH4 efflux of 1.6 ± 0.5 mg m−2 h−1 from mixed polyculture ponds of China, whereas, from shrimp they observed a mean CH4 emission rate of 19.9 ± 4.3 mg m−2 h−1 (which was higher than that observed in the OHP of the present study). However, very few studies like Yang et al. (2017) observed substantially higher effluxes (123 ± 48 mg m−2 h−1) than the present estimates while working in the shrimp ponds near Min River Estuary, China. Thus in totality, it can be inferred that atmosphere-pond CH4 fluxes from the aquaculture ponds of EKW and Minakhan Block were found higher than the recent observations being made throughout the world, especially in China.
4.1 Uncertainties and Scope for Future Studies
The present study implemented the bulk formula method and thus the estimation of atmosphere-pond CH4 flux was carried out from the difference of CH4 concentration between pond and atmosphere. This is why we could measure only the diffusive flux in this study. In the future, the chamber method should be deployed to characterize the CH4 ebullition rates from this region. More aquaculture ponds of this region should be sampled based on different species being cultured, different depths, and so forth to draw a holistic CH4 budget of this crucial region. In the present study, sampling was conducted in all the months of a calendar year. However, different stages of aquaculture should be distinctly studied as they usually exhibit different signatures of fluxes in several annual studies (Liu et al., 2016; Wu et al., 2018). In addition to these, the dissolved organic carbon should be measured in the future and the methane dynamics in the sediment–water interface should be also studied.
5 Conclusion
Analyzing all the results and outcomes of the present study, it can be concluded that partial pressure of CH4 [pCH4(w)] and hence atmosphere-pond CH4 fluxes were much higher in the freshwater aquaculture ponds (FWP) of East Kolkata Wetlands compared to the oligohaline aquaculture ponds (OHP) of Minakhan Block, both being situated under the same climatic regime in eastern India. Higher salinity was found to inhibit methanogens which resulted in lower pCH4(w) in the OHP compared to FWP. The difference in fishing practice in the two ponds was also found to regulate the turbidity and hence the presence of photosynthetically active radiation in the two ponds. The higher turbidity in FWP led to lower dissolved oxygen levels and enhanced community respiration which in turn facilitated anaerobic conditions and thus the methanogen activity was much more in FWP compared to OHP. Aquaculture practice is becoming popular day by day in India, however, efforts of characterizing the methane fluxes from these ecosystems stand very few in the present date. The annual study revealed that the mean CH4 emission observed in these sites of India was much higher than most of the recent estimates being carried out in the Chinese aquaculture ponds. Thus this study is expected to provide an impetus to carry out similar measurements in other aquaculture ponds of India to meet the present need of the hour and fill the data gap.
References
Adeyemi, S. O., Bankole, N. O., Adikwu, A. I., & Akumbo, P. M. (2009). Age growth mortality of some commercially important fish species in Gbedikere. Rivers, 2, 45–51.
Adhikari, S., Lal, R., & Sahu, B. C. (2012). Carbon sequestration in the bottom sediments of aquaculture ponds of Orissa India. Ecological Engineering, 47, 198–202.
Aich, A., Chakraborty, A., Sudarshan, M., Chattopadhyay, B., & Mukhopadhyay, S. K. (2012). Study of trace metals in Indian major carp species from wastewater–fed fishponds of East Calcutta Wetlands. Aquaculture Research, 43, 53–65.
Alagarswamy, K. (1995). Regional study and workshop on the environmental assessment and management of shrimp farming. Organized by Food and Agriculture Organisation and Network of Aquaculture Centres in Asia–Pacific (NACA), 21–26.
APHA. (2005). Standard method for the examination of water and waste water. American Public Health Association, 20th ed., p. 541.
Bastviken, D., Tranvik, L. J., Downing, J. A., Crill, P. M., & Enrich-Prast, A. (2011). Freshwater methane emissions offset the continental carbon sink. Sci, 331, 50.
Boyd, C. E., Wesley Wood, C., Chaney, P. L., & Queiroz, J. F. (2010). Role of aquaculture pond sediments in sequestration of annual global carbon emissions. Environmental Pollution, 158, 2537–2540.
Bunting, S. W., Kundu, N., & Ahmed, N. (2017). Evaluating the contribution of diversified shrimp–rice agroecosystems in Bangladesh and West Bengal India to social–ecological resilience. Ocean and Coastal Management, 148, 63–74.
Bunting, S. W., Pretty, J., & Edwards, P. (2010). Wastewater–fed aquaculture in the East Kolkata Wetlands India: Anachronism or archetype for resilient ecocultures. Reviews in Aquaculture, 2, 138–153.
Chambers, L. G., Osborne, T. Z., & Reddy, K. R. (2013). Effect of salinity–altering pulsing events on soil organic carbon loss along an intertidal wetland gradient: A laboratory experiment. Biogeochem, 115, 363–383.
Chanda, A., Das, S., Bhattacharyya, S., Das, I., Giri, S., Mukhopadhyay, A., Samanta, S., Dutta, D., Akhand, A., Choudhury, S. B., & Hazra, S. (2019). CO2 fluxes from aquaculture ponds of a tropical wetland: Potential of multiple lime treatment in reduction of CO2 emission. Science of the Total Environment, 655, 1321–1333.
Chang, T. C., & Yang, S. S. (2003). Methane emissions from wetlands in Taiwan. Atmospheric Environment, 37, 4551–4558.
Chapman, G., & Fernando, C. H. (1994). The diets and related aspects of feeding Nile tilapia (Oreochromis niloticus L.) and common carp (Cyprinus carpio L.) in low land rice fields in northeast Thailand. Aquaculture, 123, 281–307.
Chaudhuri, S. R., Mishra, M., Salodkar, S., Sudarshan, M., & Thakur, A. R. (2008). Traditional aquaculture practice at East Calcutta Wetland: The safety assessment. American Journal of Environmental Sciences, 4, 140–144.
Chaudhuri, S. R., Mukherjee, I., Ghosh, D., & Thakur, A. R. (2012). East Kolkata Wetland: A multifunctional niche of international importance. Online Journal of Biological Sciences, 12, 80–88.
Chaudhuri, S. R., Salodkar, S., Sudarshan, M., & Thakur, A. R. (2007). Integrated resource recovery at East Calcutta wetland: How safe is these? American Journal of Agricultural Biological Sciences, 2, 75–80.
Chen, Y., Dong, S. L., Wang, F., Gao, Q. F., & Tian, X. L. (2016). Carbon dioxide and methane fluxes from feeding and no–feeding mariculture ponds. Environmental Pollution, 212, 489–497.
Cole, J. J., & Caraco, N. F. (1998). Atmospheric exchange of carbon dioxide in a low-wind oligotrophic lake measured by the addition of SF6. Limnology and Oceanography, 43, 647–656.
Cotovicz, L. C., Knoppers, B. A., Brandini, N., Poirier, D., Costa Santos, S. J., & Abril, G. (2016). Spatio-temporal variability of methane (CH4) concentrations and diffusive fluxes from a tropical coastal embayment surrounded by a large urban area (Guanabara Bay, Rio de Janeiro, Brazil). Limnology and Oceanography, 61, S238–S252.
Datta, A., Nayak, D. R., Sinhababu, D. P., & Adhya, T. K. (2009). Methane and nitrous oxide emissions from an integrated rainfed rice–fish farming system of Eastern India. Agriculture, Ecosystems & Environment, 129(1–3), 228–237.
De Roy, S. (2012). Impact of fish farming on employment and household income. Economic and Political Weekly, 47, 69.
Dutta, M. K., Chowdhury, C., Jana, T. K., & Mukhopadhyay, S. K. (2013). Dynamics and exchange fluxes of methane in the estuarine mangrove environment of Sundarbans, NE coast of India. Atmospheric Environment, 77, 631–639.
Flury, S., McGinnis, D. F., & Gessner, M. O. (2010). Methane emissions from a freshwater marsh in response to experimentally simulated global warming and nitrogen enrichment. Journal of Geophysical Research: Biogeoscience, https://doi.org/10.1029/2009J G001079.
Frei, M., & Becker, K. (2005). A greenhouse experiment on growth and yield effects in integrated rice–fish culture. Aquaculture, 244, 119–128.
Furlanetto, L. M., Marinho, C. C., Palma-Silva, C., Albertoni, E. F., Figueiredo-Barros, M. P., & Esteves, F. A. (2012). Methane levels in shallow subtropical lake sediments: Dependence on the trophic status of the lake and allochthonous input. Limnology, 42, 151–155.
Ghosh, D. (2005). Ecology and traditional wetland practice: Lessons from wastewater utilization in the East Calcutta Wetlands. Worldview, 1st ed., p. 120.
Ghosh, D., & Furedy, C. (1984). Resource conserving traditions and waste disposal: The garbage farms and sewage–fed fisheries of Calcutta. Conservation & Recycling, 7, 159–165.
Ghosh, S. (2018). Wastewater–fed aquaculture in East Kolkata Wetlands: State of the art and measures to protect biodiversity. In B. Jana, R. Mandal, & P. Jayasankar (Eds.), Wastewater management through aquaculture. Springer.
Heyer, J., & Berger, U. (2000). Methane emission from the coastal area in the southern Baltic Sea. Estuarine, Coastal and Shelf Science, 51(1), 13–30.
Holgerson, M. A. (2015). Drivers of carbon dioxide and methane supersaturation in small, temporary ponds. Biogeochemistry, 124, 305–318.
Hu, M. J., Ren, H. C., Ren, P., Li, J. B., Wilson, B. J., & Tong, C. (2017). Response of gaseous carbon emissions to low-level salinity increase in tidal marsh ecosystem of the Min River estuary, southeastern China. Journal of Environmental Sciences, 52, 210–222.
Hu, Z. Q., Wu, S., Ji, C., Zou, J. W., Zhou, Q. S., & Liu, S. W. (2016). A comparison of methane emissions following rice paddies conversion to crab-fish farming wetlands in southeast China. Environmental Science and Pollution Research, 23, 1505–1515.
Hu, Z., Lee, J. W., Chandran, K., Kim, S., & Khanal, S. K. (2012). Nitrous oxide (N2O) emission from aquaculture: A review. Environmental Science and Technology, 46, 6470–6480.
Hu, Z., Lee, J. W., Chandran, K., Kim, S., Sharma, K., & Khanal, S. K. (2014). Influence of carbohydrate addition on nitrogen transformations and greenhouse gas emissions of intensive aquaculture system. Science of the Total Environment, 470, 193–200.
Inglett, K. S., Inglett, P. W., Reddy, K. R., & Osborne, T. Z. (2012). Temperature sensitivity of greenhouse gas production in wetland soils of different vegetation. Biogeochemistry, 108, 77–90.
Jihulya, N. J. (2014). Diet and feeding ecology of Nile Tilapia, Oreochromis Niloticus and Nile Perch, Lates niloticus in protected and unprotected areas of Lake Victoria, Tanzania. International Journal of Scientific Technology Research, 3, 280–286.
Kettunen, A., Kaitala, V., Lehtinen, A., Lohila, A., Alm, J., Silvola, J., & Martikainen, P. J. (1999). Methane production and oxidation potentials in relation to water table fluctuations in two boreal mires. Soil Biology & Biochemistry, 31, 1741–1749.
Knox, S. H., Matthes, J. H., Sturtevant, C., Oikawa, P. Y., Verfaillie, J., & Baldocchi, D. (2016). Biophysical controls on inter–annual variability in ecosystem–scale CO2 and CH4 exchange in a California rice paddy. Journal of Geophysical Research: Biogeosciences, 121, 978–1001.
Kundu, N., Pal, M., & Saha, S. (2008). East Kolkata Wetlands: A resource recovery system through productive activities. Proceedings of Taal 2007: The 12th World Lake Conference, pp. 868–881.
Laanbroek, H. J. (2010). Methane emission from natural wetlands: Interplay between emergent macrophytes and soil microbial processes: A mini–review. Annals of Botany, 105, 141–153.
Lide, D. R. (2007). CRC handbook of chemistry and physics, 88th ed. CRC, New York, p. 2660.
Liu, S. W., Hu, Z. Q., Wu, S., Li, S. Q., Li, Z. F., & Zou, J. W. (2015). Methane and nitrous oxide emissions reduced following conversion of rice paddies to inland crab-fish aquaculture in southeast China. Environmental Science and Technology, 50, 633–642.
Liu, S. W., Hu, Z. Q., Wu, S., Li, S. Q., Li, Z. F., & Zou, J. W. (2016). Methane and nitrous oxide emissions reduced following conversion of rice paddies to inland crab−fish aquaculture in southeast China. Environmental Science and Technology, 50, 633–642.
Liu, X., Gao, Y., Zhang, Z., Luo, J., & Yan, S. (2017). Sediment–water methane flux in a eutrophic pond and primary influential factors at different time scales. Water, 9, 601. https://doi.org/10.3390/w9080601
Lofton, D. D., Whalen, S. C., & Hershey, A. E. (2014). Effect of temperature on methane dynamics and evaluation of methane oxidation kinetics in shallow Arctic Alaskan lakes. Hydrobiologia, 721, 209–222.
Long, L., Xiao, S. B., Zhang, C., Zhang, W. L., Xie, H., Li, Y. C., Lei, D., Mu, X. H., & Zhang, J. W. (2016). Characteristics of methane flux across the water–air interface in subtropical shallow ponds. Huan Jing Ke Xue Huanjing Kexue, 37, 4552–4559. (in Chinese).
MacIntyre, S., Wanninkhof, R., & Chanton, J. P. (1995). Trace gas exchange across the air–water interface in freshwater and costal marine environments. In P. A. Matson and R. C. Harriss (Eds.), Biogenic trace gases: Measuring emissions from soil and water, pp. 52–97. Blackwell Science Oxford.
Mondal, I., and Bandyopadhyay, J. (2015). Recent trend of aquaculture land of Bidyadhari River catchment area using geospatial techniques: A case study of Haroa and Minakhan Block, North–24 Parganas. Am Res Thoughts https://doi.org/10.6084/m9.figshare.1492986
Morel FM, M. (1983). Energetics and kinetics: Principles of aquatic chemistry, p. 446. Wiley.
Naskar, K. R. (1985). A short history and the present trends of brackish water fish culture in paddy fields at the Kulti-Minakhan areas of Sundarbans in West Bengal. Journal of the Indian Society Coastal Agricultural Research, 3, 115–124.
Neubauer, S. C., Franklin, R. B., & Berrier, D. J. (2013). Saltwater intrusion into tidal freshwater marshes alters the biogeochemical processing of organic carbon. Biogeosciences, 10, 8171–8183.
Njiru, M., Okeyo-Owuor, J. B., Muchiri, M., & Cowx, I. G. (2004). Shifts in food of Nile tilapia, Oreochromis niloticus in Lake Victoria. African Journal of Ecology, 44, 163–170.
Olsson, L., Ye, S., Yu, X., Wei, M., Krauss, K. W., & Brix, H. (2015). Factors in fluencing CO2 and CH4 emissions from coastal wetlands in the Liaohe Delta, northeast China. Biogeosciences, 12, 4965–4977.
Osudar, R., Matoušů, A., Alawi, M., Wagner, D., & Bussmann, I. (2015). Environmental factors affecting methane distribution and bacterial methane oxidation in the German Bight (North Sea). Estuarine, Coastal Shelf Sciences, 160, 10–21.
Palma-Silva, C., Marinho, C. C., Albertoni, E. F., Giacomini, I. B., Figueiredo Barros, M. P., & Furlanetto, L. M. (2013). Methane emissions in two small shallow neotropical lakes: The role of temperature and trophic level. Atmospheric Environment, 81, 373–379.
Pathak, H., Upadhyay, R. C., Muralidhar, M., Bhattacharyya, P., & Venkateswarlu, B. (2013). Measurement of greenhouse gas emission from crop, livestock and aquaculture, p. 101. Indian Agricultural Research Institute.
Poffenbarger, H. J., Needelman, B. A., & Megonigal, J. P. (2011). Salinity influence on methane emissions from tidal marshes. Wetlands, 31, 831–842.
Roland, F. A., Darchambeau, E., Morana, F., Bouillon, C. S., & Borges, A. V. (2017). Emission and oxidation of methane in a meromictic, eutrophic and temperate lake (Dendre, Belgium). Chemosphere, 168, 756–764.
Sarkar, S., Tribedi, P., Gupta, A. D., Saha, T., & Sil, A. K. (2017). Microbial functional diversity decreases with sewage purification in stabilization ponds. Waste Biomass Valorization, 8(2), 417–423.
Sun, Z. G., Wang, L. L., Tian, H. Q., Jiang, H. H., Mou, X. J., & Sun, W. L. (2013). Fluxes of nitrous oxide and methane in different coastal Suaeda salsa marshes of the Yellow River estuary, China. Chemosphere, 90(2), 856–865.
Venkiteswaran, J. J., Schiff, S. L., St, V. L., Louis, C. J. D., Matthews, N. M., & Boudreau, E. M. (2013). Processes affecting greenhouse gas production in experimental boreal reservoirs. Global Biogeochemical Cycles, 27, 567–577.
Verdegem, M. C. J., & Bosma, R. H. (2009). Water withdrawal for brackish and inland aquaculture, and options to produce more fish in ponds with present water use. Water Policy, 11, 52–68.
Vizza, C., West, W. E., Jones, S. E., Hart, J. A., & Lamberti, G. A. (2017). Regulators of coastal wetland methane production and responses to simulated global change. Biogeosciences, 14, 431–446.
Wanninkhof, R. (1992). Relationship between gas exchange and wind speed over the ocean. Journal of Geophysical Research, 97, 7373–7381.
Welti, N., Hayes, M., & Lockington, D. (2017). Seasonal nitrous oxide and methane emissions across a subtropical estuarine salinity gradient. Biogeochemistry, 132(1–2), 55–69.
Wu, S., Hu, Z., Hu, T., Chen, J., Yu, K., Zou, J., & Liu, S. (2018). Annual methane and nitrous oxide emissions from rice paddies and inland fish aquaculture wetlands in southeast China. Atmospheric Environment, 175, 135–144.
Xiang, J., Liu, D. Y., Ding, W. X., Yuan, J. J., & Lin, Y. X. (2015). Invasion chronosequence of Spartina alterniflora on methane emission and organic carbon sequestration in a coastal salt marsh. Atmospheric Environment, 112, 72–80.
Xiao, Q. T., Zhang, M., Hu, Z. H., Gao, Y. Q., Hu, C., & Liu, C. (2017). Spatial variations of methane emission in a large shallow eutrophic lake in subtropical climate. Journal of Geophysical Research: Biogeosciences, 122, 1597–1614.
Yang, H., Andersen, T., Dörsch, P., Tominaga, K., Thrane, J. E., & Hessen, D. O. (2015a). Greenhouse gas metabolism in Nordic boreal lakes. Biogeochemistry, 126, 211–225.
Yang, J. S., Liu, J. S., Hu, X. J., Li, X. X., Wang, Y., & Li, H. Y. (2013). Effect of water table level on CO2, CH4 and N2O emissions in a freshwater marsh of Northeast China. Soil Biology and Biochemistry, 61, 52–60.
Yang, P., He, Q. H., Huang, J. F., & Tong, C. (2015b). Fluxes of greenhouse gases at two different aquaculture ponds in the coastal zone of southeastern China. Atmospheric Environment, 115, 269–277.
Yang, P., Lai, D. Y., Jin, B., Bastviken, D., Tan, L., & Tong, C. (2017). Dynamics of dissolved nutrients in the aquaculture shrimp ponds of the Min River estuary, China: Concentrations, fluxes and environmental loads. Science of the Total Environment, 603, 256–267.
Yang, P., Lai, D. Y., Huang, J. F., & Tong, C. (2018a). Effect of drainage on CO2, CH4, and N2O fluxes from aquaculture ponds during winter in a subtropical estuary of China. Journal of Environmental Sciences, 65, 72–82.
Yang, P., Lai, D. Y., Yang, H., Tong, C., Lebel, L., Huang, J., & Xu, J. (2019). Methane dynamics of aquaculture shrimp ponds in two subtropical estuaries, Southeast China: Dissolved concentration, net sediment release, and water oxidation. Journal of Geophysical Research: Biogeosciences, 124, 1430–1445.
Yang, P., Zhang, Y., Lai, D. Y., Tan, L., Jin, B., & Tong, C. (2018b). Fluxes of carbon dioxide and methane across the water–atmosphere interface of aquaculture shrimp ponds in two subtropical estuaries: The effect of temperature, substrate, salinity, and nitrate. Science of the Total Environment, 635, 1025–1035.
Yang, S. S. (1998). Methane production in river and lake sediments in Taiwan. Environmental Geochemistry Health, 20, 245–249.
Acknowledgements
All the authors are indebted to the National Remote Sensing Centre, Govt. of India for providing the research grant. Sania Shaher is indebted to the University Grants Commission (UGC), India for providing the UGC National Fellowship. The authors are also thankful to the East Kolkata Wetlands Management Authority, Govt. of West Bengal, and the local fisher community for extending their help and sharing their views and traditional knowledge. The authors are deeply grateful to Late Prof. Ananda Dev Mukherjee for his steady support and encouragement throughout this piece of research.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Ethics declarations
Conflict of Interest:
All the authors state that they do not have any competing conflict of financial or any other form of interest.
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Shaher, S. et al. (2022). Contrasting Diffusive Methane Emission from Two Closely Situated Aquaculture Ponds of Varying Salinity Situated in a Wetland of Eastern India. In: Islam, A., Das, P., Ghosh, S., Mukhopadhyay, A., Das Gupta, A., Kumar Singh, A. (eds) Fluvial Systems in the Anthropocene. Springer, Cham. https://doi.org/10.1007/978-3-031-11181-5_20
Download citation
DOI: https://doi.org/10.1007/978-3-031-11181-5_20
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-11180-8
Online ISBN: 978-3-031-11181-5
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)