Abstract
Type II diabetes mellitus (T2DM) is a metabolic disorder characterized by chronically elevated glucose caused by insulin resistance. Although T2DM is manageable through insulin therapy, the disorder is a risk factor for much more dangerous diseases, including cardiovascular disease, kidney disease, retinopathy, Alzheimer’s disease, and more. T2DM affects 450 million people worldwide and is attributed to causing over 4 million deaths each year. Current methods for detecting diabetes typically involve randomly or after fasting testing a person’s glycated hemoglobin and blood sugar levels. However, these methods can be problematic due to an individual’s daily levels or being affected by diet or environment and the lack of sensitivity and reliability within the tests themselves. Vibrational spectroscopic methods have been pursued as a novel method for detecting diabetes accurately and early on in a non-invasive manner. This review summarizes recent research which has used infrared or Raman spectroscopy to develop a fast, simple, and accurate method for non-invasively diagnosing diabetes. It is proposed that vibrational spectroscopy can improve and revolutionize how diabetes is diagnosed, allowing for faster and more effective treatment.
Adopted from Ralbovsky, N.M. and Lednev, I.K., Applied Spectroscopy. Copyright © 2021 Sage Journals. https://doi.org/10.1177/00037028211019130
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Diabetes is commonly regarded as a group of metabolic diseases characterized by elevated glucose levels due to defects in insulin secretion and insulin action. Chronic levels of hyperglycemia in an individual can lead to severe complications, including the damage to, and even failure of, organs such as the kidneys and heart [1]. Due to many health complications associated with the disease, accurate and early detection is of incredible significance [2]. However, one-third of current type II diabetes cases are undiagnosed, and current diagnostic tests are heavily debated [3, 4]. Most commonly, various blood glucose tests, such as the oral glucose tolerance test and the fasting blood sugar test, are performed, which require the individual being tested to undergo some level of fasting and can be affected by short-term lifestyle influences and changes. Another test, called the glycated hemoglobin (HbA1c) test, provides an average blood sugar level for the past two to three months. Although the HbA1c test does not require fasting, it is expensive and does not accurately reflect glycemia. Further information regarding these tests is highlighted elsewhere [5, 6]. Because limits exist in the currently used methods, this review critically evaluates vibrational spectroscopy and its potential to contribute toward the identification of diabetes simply and accurately.
The two major methods associated with vibrational spectroscopy include Raman spectroscopy and infrared (IR) spectroscopy. Raman spectroscopy involves irradiating a sample with monochromatic (i.e., laser) light resulting in molecules scattering incident light. The majority of scattered light is at the same wavelength as the incident light and is called Rayleigh scattering. The small portion of the light scattered at a different wavelength is called Raman scattering. The difference in energy between the incident and Raman scattered light is a “Raman shift” and corresponds to a frequency for the vibration, measured in wavenumbers (cm−1). The resultant Raman spectrum is considered a vibrational “fingerprint,” specific to the analyzed sample. IR spectroscopy is complementary to Raman spectroscopy and uses infrared light to irradiate the sample, exciting molecular vibrations. The resultant spectrum describes the absorption of the light by the molecules in the sample as a function of its frequency, again measured in wavenumbers (cm−1). Vibrational spectroscopy is useful for identifying different functional groups present in a sample. Both methods provide complementary information regarding the molecular structure and composition of the sample. Importantly, due to their specificity, each can be used to identify differences between biological samples obtained from different types of donors, such as those with or without a disease. Furthermore, research has already shown that known differences in the biochemical composition of biological fluids exist because of diabetes. It has been observed that higher levels of certain enzymes, total cholesterol, triglycerides, and low-density lipoprotein and lower levels of high-density lipoprotein, hemoglobin, and red blood cell content were found in the blood of individuals with type 2 diabetes mellitus compared to non-diabetic subjects [7, 8]. Due to these differences, which have been previously observed and documented [9, 10], it is hypothesized that vibrational spectroscopy may be successful in detecting the alterations in composition, in addition to others that have been reported, within biological samples and capitalize on this detection for diagnostic success.
Due to the importance of detecting diabetes accurately and effectively, other reviews have been published on the topic. However, the scope of this alternative but informative review differs greatly from this review. Many reviews focus broadly on any methods useful for monitoring glucose [11,12,13,14,15], glycated hemoglobin [3, 16], or other biomarkers [17] levels to generate a diagnosis. Different reviews have focused on any method which could be useful for detecting diabetes [4, 18], and some work has been done to investigate non-enzymatic methods for glucose sensing [19,20,21,22]. While further reviews focus on the utility of either Raman spectroscopy [23, 24] or IR spectroscopy [23,24,25,, 26], or both [27,28,29,30,31] for general medical diagnostics, there is a gap in the literature that focuses specifically on vibrational spectroscopy for diagnosing diabetes. In this regard, the current review will analyze and discuss research published between 2015 and the present. In particular, articles that focus on applying either Raman spectroscopy or IR spectroscopy to diagnose diabetes are considered. Modifications of either method will be considered, such as incorporating fiber-optic techniques or hand-held devices. Studies conducted using any form of biological material (including cells, tissue, and body fluids) will be reviewed, in addition to those studies which may or may not utilize chemometric methods. Although it is preferred to incorporate the use of multivariate analysis for objective and accurate diagnostic results, the use of such methods is not always necessary for identifying diabetes, as will be discussed. This work will review the many applications of Raman spectroscopy and infrared spectroscopy for the inexpensive, rapid, simple, and accurate identification of diabetes.
2 Raman Spectroscopy
In more than half of the manuscripts reviewed herein, Raman spectroscopy has been used to successfully identify various spectroscopic biomarkers to identify diabetes. The spectral fingerprint produced using Raman spectroscopy can be vital for identifying differences between healthy donors and those donors with a disease. One of the first reports on the application of Raman spectroscopy for investigating the disease mechanism of diabetes was made by Professor Ozaki et al. in 1982. Raman spectroscopy was employed to investigate the biochemical differences between a diabetic cataractous lens and a normal lens [32]. Amongst the many important projects Professor Ozaki carried out, this work, in particular, was crucial to opening the door for further investigations into using vibrational spectroscopy as a tool to detect and monitor this disease.
The articles reviewed in this section include those using regular Raman spectroscopy to identify diabetes and different variations of the method. For example, several research projects have incorporated the use of surface-enhanced Raman spectroscopy (SERS). The advantage of SERS resides in its capacity to detect biomolecules at ultralow concentrations due to the adsorption of molecules onto rough metal surfaces, such as silver or gold nanoparticles. The SERS effect can enhance Raman scattering by factors up to 1010 or larger, allowing for a more sensitive analysis of the analyte in question [33, 34] Portable [35] and fiber-optic [36] Raman spectroscopic systems are other popular variations of Raman spectroscopy used in the following reviewed manuscripts for diagnostic applications. These methods represent the transition of the instrument toward use in clinical settings; the systems are typically much smaller, more portable, and easily able to be adapted into different settings as compared to a typical Raman spectrometer. The instruments can be less expensive than bench-top instruments, occupy significantly less space, and can often be used intraoperatively, making real-time analysis much more achievable.
In addition, many of the studies reviewed in this section incorporated chemometric methods for identifying and diagnosing diabetes. Generally, chemometrics refers to extracting chemically relevant information from complex datasets [37]. By applying chemometric methods to data that exists as a matrix (e.g., spectral data), machine learning algorithms can be built to separate, sort, and recognize patterns within chemical data. The built models can recognize differences and similarities between classes or groups of data and can use that information to generate predictions on new data presented. Incorporating multivariate analysis into a study can lead to more accurate and objective results than studies that do not rely on chemometrics. In this way, these algorithms can be used for many different types of medical screening and diagnostic applications [28, 38,39,40].
The following research studies focus on identifying diabetes through detection of glycated hemoglobin, blood glucose levels, other novel biomarkers, or strictly through chemometric models.
2.1 Monitoring Glycated Hemoglobin (HbA1c) Levels for Indicating Diabetes
Several research studies focused on detecting glycated hemoglobin (HbA1c) within the individuals they studied. Elevated levels of HbA1c have been indicated as a well-known biomarker for diabetes, and the HbA1c test provides an average blood sugar measurement of the past two to three months by measuring the percentage of blood sugar attached to hemoglobin [41, 42]. A recent review has focused on the future outlook of using Raman spectroscopy for sensing glycated hemoglobin [43]. One of the earlier papers to investigate Raman spectroscopy for detecting HbA1c was reported by Barman et al. in 2012 [44]. More recently, González-Viveros investigated various commercial lyophilized HbA1c in distilled water. Principal component analysis (PCA), an unsupervised chemometric method, showed good separation between the commercial HbA1c and two solutions with known concentrations (Fig. 1). A nonlinear regression model based on a feed-forward neural network (FFNN) was then built to predict the unknown concentration of HbA1c in different solutions, which resulted in a low root mean square error of 0.08% ± 0.04 after five-fold cross-validation (CV) [45]. While this work does not directly investigate diagnosing diabetes, the successful results indicate that future work could extend toward monitoring the levels of HbA1c in blood samples for both detecting and monitoring the progression of the disease.

Reproduced with permission from Elsevier B.V. [45]
HbA1c concentrations representation by three PC. Each point represents a sampled spectrum.
Using a Raman probe system (Fig. 2), Villa-Manríquez et al. collected Raman spectra from three different regions of the body, including the index fingertip of the right hand, ear lobe, and the forehead of 15 healthy volunteers and 71 patients with diabetes (of whom, 49 had high HbA1c levels and 22 had low HbA1c levels). Chemometrics analysis methods were used for discrimination, including PCA and support vector machine (SVM) analysis. Raman spectra of the forehead were the most successful for differentiating the low HbA1c level group and healthy volunteers, reaching 100% sensitivity and specificity each. The fingertip Raman spectra showed 100% sensitivity and 80% specificity for separating the healthy volunteers and the high HbA1c level group. A receiver operating characteristic (ROC) curve was used to confirm the results obtained after external validation conducted using an independent test dataset, indicating a successful in vivo method for identifying diabetic individuals [46].

Reproduced with permission from Wiley‐VCH Verlag GmbH & Co. KGaA, Weinheim [46]
Experimental setup scheme. a Laser of 785 nm, b spectrometer, c computer, d Raman probe, and e sample.
Lin et al. utilized a near-IR laser tweezers Raman spectroscopy (LTRS) system, a method that allows for analysis of single biological particles or cells in suspension [47], to investigate variation in hemoglobin levels within red blood cells obtained from T2DM individuals (n = 45) and healthy volunteers (n = 45). Linear discriminant analysis (LDA) could accurately discriminate between the groups, reaching 100% sensitivity and 90% specificity after external validation. The major spectral differences were assigned to proteins and heme groups [48]. These two studies are clear examples of successful methods which employ Raman spectroscopy for detecting elevated or variated levels of HbA1c, with the potential to improve accurate detection of T2DM.
2.2 Monitoring Blood Glucose Levels for Indicating Diabetes
The classic gold standard for diagnosing diabetes is testing an individual’s blood sugar levels, such as through the oral glucose tolerance test (OGTT); however, this test, and others, require the patient to fast, are time-consuming, and may have poor reproducibility [49]. Through Raman spectroscopy, numerous researchers have pursued improved and alternative methods for identifying elevated blood glucose levels within individuals. In one work, a method for noninvasive monitoring of blood glucose levels was explored through Raman spectroscopic analysis of microvessels in the superficial layer of the human nail fold of 12 random volunteers. PCA in combination with a backpropagation artificial neural network (BP-ANN) was used to predict the blood glucose levels of the individuals. The levels were compared to those obtained using the OGTT, with the results showing a root mean square error of prediction of 0.45 mmol/L and R2 of 0.95; the predicted glucose concentrations were further evaluated using the Clarke error grid, which compares how similar blood glucose values are to sensor readings at isolated points in time [50]. Here, results indicated the predicted concentrations fell within Clarke error zones A and B, which means they were within 20% similarity to that provided by the OGTT or outside of 20% similarity but would not lead to inappropriate treatment, respectively. Additional validation of the chemometric model was not reported [51].
A fiber-optic Raman probe was also used to relate Raman spectra to blood glucose levels in similar work. Here, 20 individuals were given a standard glucose drink, typically used in OGTT. Raman spectra were recorded transcutaneously every 10 min for 160 min, while finger-prick measurements were drawn to record the blood glucose levels. Partial least squares regression (PLSR) modeling was used to predict glucose concentration in the blood using Raman spectral data. Results after external validation showed an accurate comparison could be made, and 97% of the predictions fell within zones A and B of the Clarke error grid [52]. Alternatively, Ju et al. used a SERS sensor to detect in situ glucose levels in a mouse model of Streptozotocin (STZ)-induced type I diabetes via a functional poly(methyl methacrylate) microneedle (F-PMMA MN) array (Fig. 3). It was shown that the microneedle array could directly measure glucose levels within interstitial fluid without causing lasting damage to the skin. A commercial glucometer was used to compare glucose level measurements; results show 93% of the glucose readings obtained using the F-PMMA MN array fell within zones A and B of the Clarke error grid, indicating a novel minimally invasive method determining blood glucose levels for diabetes detection [53].

Reproduced with permission from the American Chemical Society [53]
a Photograph of the F-PMMA MN array pressed onto the skin on the back of a mouse. b Mouse under anesthesia on the stage of the Raman microspectroscopy system for measurements. c Schematic illustration of the glucose measurement using the F-PMMA MN array for in vivo transdermal detection based on surface-enhanced Raman spectroscopy. d Glucose level measured using SERS biosensor for a range of dwelling time from 0 to 15 min. e Glucose levels were measured using our SERS glucose biosensor (red) compared to those obtained from a commercial glucometer (blue). We selected three mice with different blood glucose concentrations for testing in which the excitation power was 16.5 mW out of the needle tip at 785 nm and the exposure time was 10 000 ms. Each mouse was tested sequentially five times. f Clarke error grid analysis of the in vivo glucose measurements using our SERS glucose biosensor in a mouse model of STZ-induced type I diabetes.
Lastly, in different works, a wearable Raman probe system was employed. Ten human volunteers were administered 2 g/kg of sucrose. Both Raman spectra were obtained transcutaneously from the inside of the wrist, and glucose reference values, via a glucose meter were recorded every 20 min for two hours. A nonlinear PLS model was built to predict the glucose values based on the Raman spectra, with results indicating an average R2 value of 0.844 after CV. This work indicates a very intriguing potential to investigate blood glucose levels for monitoring disease progression in a noninvasive manner [54].
These four studies show that Raman spectroscopy has great potential to measure blood glucose levels accurately and is minimally invasive. While this research requires repetition in large-scale human studies, the success shown here indicates this method could be considered in future work.
2.3 Monitoring Novel Biomarkers for Indicating Diabetes
In addition to monitoring HbA1c and blood glucose levels, other biomarkers were also explored as a potential route toward identifying diabetes with greater levels of accuracy and repeatability. Although not all novel biomarkers have a well-known biochemical connection to diabetes, the exploratory nature of Raman spectroscopy allows for the detection of biomolecules that have not been considered in the past.
The first set of research studies in this section focused on analyzing serum for identifying novel biomarkers. Recently, leucine and isoleucine amino acids were investigated as biomarkers for early T2DM screening using SERS. Here, blood from 40 rats was deposited on substrates prepared from conductive silver paste smeared onto glass and analyzed; Specific Raman bands were found to correlate with the two amino acids, in addition to glucose, with the intensities corresponding to T2DM biomarkers. Further, when the rats were administered two different antidiabetic drugs (pioglitazone and herbal extract Momordica spinosa (Glig.) Chiov), the intensity of these Raman bands in newly collected spectra were shown to decrease, thus indicating these amino acids as potential spectroscopic markers for monitoring the progression of the disease as well as the efficacy of treatment [55]. Similarly, the same research group used SERS substrates to analyze blood collected from 50 rats. Here, they identified valine, leucine, isoleucine, creatine, glucose, and fructose spectral bands as early indicators for predicting the presence of diabetes. These spectral bands were also sensitive to antidiabetic drug treatment in the rats. Here, the herbal extract Rotheca myricoides Hochst and the antidiabetic drug pioglitazone resulted in the decrease in intensity of the spectroscopic bands associated with the aforementioned biomolecules; PCA also indicated spectral differences existed between the various groups [56]. Both papers indicate an interesting potential for the early identification and treatment monitoring of T2DM based on the novel and alternative Raman spectroscopic biomarkers. Early detection of the disease can help mitigate potential issues that arise due to it and provide the afflicted individual with more effective treatment opportunities [57,58,59].
In human studies, Silveira Jr. et al. leveraged Raman spectroscopy for investigating the levels of glucose and lipid fractions in 44 serum samples. The concentration of glucose, triglycerides, cholesterol, and high- and low-density lipoproteins were determined using a colorimetric method. A PLSR model with leave-one-out cross-validation (LOOCV) was then built to predict the known concentrations of the biochemical components based on the Raman spectra and indicated triglycerides and cholesterol concentrations could be estimated with r values of 0.98 and 0.96, respectively. The r values were slightly lower (0.75–0.86) for the other biochemicals [60]. González-Solís et al. analyzed serum samples from 15 individuals diagnosed with T2DM and from 20 healthy controls, with spectral differences due to glutathione, polysaccharides, phenylalanine, tryptophan, and proteins being observed. PCA with LDA was then employed to discriminate between the two groups, reaching 96% sensitivity and 99% specificity after CV [61]. In one last report using blood, albumin was purified using membrane electrophoresis from plasma samples of 40 T2DM patients and 50 healthy volunteers (where five donors from each class were set aside for an independent external validation group). SERS spectra were collected, and PCA with LDA was shown to successfully differentiate between diabetic and healthy spectra with 100% specificity and 80% sensitivity after external validation [62]. These studies indicate that alternative blood-based biomarkers may increase the sensitivity and specificity for identifying and diagnosing diabetes using Raman spectroscopy.
In non-blood-based studies, urinary extracellular vesicles were shown in one paper to be useful as a potential diabetes biomarker, with cluster analysis (CA) of the obtained Raman spectra showing significant differences between controls (n = 10) and T2DM (n = 45, 19 with good glycemic control and 26 with unsatisfactory glycemic control) individuals. On the other hand, endothelium-derived extracellular vesicles successfully separated cells cultured in differing glycemic conditions. PLSR analysis indicated spectral bands associated with saccharides, lipids, proteins and protein conformation, and nucleic acids could separate the three groups. Although no validation was reported, this research indicates a different and much less invasive method for detecting potential diabetes biomarkers [63]. Flores-Guerrero utilized a probe-based Raman spectrometer to investigate urinary albumin in individuals with T2DM. Diabetic kidney disease is a main complication of T2DM and is commonly identified through urinary albumin excretion. Urine samples from ten individuals with T2DM were analyzed, indicating several specific Raman peaks that could be assigned to albumin. Due to the ability of Raman spectroscopy to detect urinary albumin, the authors propose a promising method for detecting T2DM noninvasively in future work [64]. Each of these independent studies is important for their potential to be used for the early identification of diabetes via the minimally invasive monitoring of biomarkers that have previously not been focused.
In a different study, the nonenzymatic glycation of collagen scaffolds was analyzed in T2DM mice at various time points. While the Raman peak positions due to collagen did not change between the groups, the relative intensity of the peaks after normalization increased as diabetic time progressed. These bands were positively correlated to the expression of anti-advanced glycation end products obtained by immunofluorescence imaging of the scaffolds, suggesting Raman spectroscopy can be used to monitor how the structure of collagen scaffolds is affected by nonenzymatic glycation in T2DM mice [65].
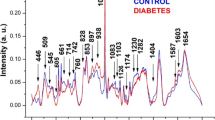
In one of the largest studies carried out using Raman spectroscopy, skin glycated proteins were investigated using a portable Raman spectroscopy system and fluorescence spectroscopy (Fig. 4). Ninety-four individuals who were either nondiabetic had insulin resistance or were diabetic were evaluated. Increased skin autofluorescence was noted for those individuals with insulin resistance and those who had diabetes compared to healthy individuals. Raman spectral bands related to changes in skin hydration, type I collagen, and protein glycation were noted for diabetic patients. A positive but weak correlation was also noted between the level of skin autofluorescence and the ratio of Raman bands indicative of glycated proteins. Although further work is needed, this information could be used in the future for noninvasive screening of diabetes and help to contribute to understanding the biochemical progression of the disease [66].

Reproduced with permission from WILEY‐VCH Verlag GmbH & Co. KGaA, Weinheim [66]
Clinical procedure with fluorescence and Raman techniques.
These studies indicate several opportunities to explore new potential biomarkers for diagnosing diabetes using Raman spectroscopy. Although the biochemical basis for many of these biomolecules is not well established, there exists an exciting opportunity to increase the sensitivity and specificity of diagnosis with additionally verified biomarkers.
2.4 General Application of Chemometric Methods for Indicating Diabetes Within Various Biological Samples
Although detecting new and known biomarkers using Raman spectroscopy for diagnosing diabetes has been fruitful, several research groups have leveraged the power of chemometric methods to simply investigate diagnosing the disease without requiring or depending on the presence of specific biomarkers. One of the greatest advantages of Raman spectroscopy is its ability to probe a sample’s entire biochemical signature. By leveraging the contribution of all biochemical components, instead of narrowing the focus to one or two specific biomarkers, chemometrics can capitalize on the multitude of information obtained in a spectrum to objectively achieve highly sensitive and specific levels of diagnostic accuracy.
Several studies focused on analyzing blood samples using Raman spectroscopy and chemometrics. In another study by González-Solís et al., superparamagnetic clustering, a type of clustering-based chemometric method, was investigated to analyze Raman spectra of serum samples from 15 individuals diagnosed with T2DM and from 20 healthy controls. Results showed 97.5% sensitivity and 91.2% specificity for correctly diagnosing the class of serum; however, no validation of the model was reported [67]. Blood plasma samples obtained from healthy (n = 8) and type I diabetic individuals (n = 12) were investigated using Raman spectroscopy and Raman optical activity, electronic circular dichroism, and IR spectroscopy. LDA was used to evaluate each method individually, as well as combined. Interestingly, the best results were obtained when the combined data reached 92% sensitivity and 100% specificity after LOOCV. Raman spectroscopy was used in combination with PCA to successfully differentiate serum from individuals with T2DM, diabetic retinopathy, or those who are healthy. Mahalanobis distance, which measures the similarity between two sets of data for discrimination, was also shown to separate the Raman spectral data successfully; no validation of either method was included in the manuscript. However, these results were found to be more successful than those obtained by relying on the comparison of prominent Raman peak positions and intensities [68].
Two different studies focused on analyzing blood from animal donors to diagnose the disease. Red blood cells from healthy humans, healthy rats, T2DM humans, and STZ-induced and Alloxan-induced diabetic rats were investigated using Raman spectroscopy. In a unique experimental decision, PCA combined with an SVM classifier could successfully separate all five red blood cells, although a validation mechanism was not reported. Additional spectral differences were noted between the classes. It was determined that the STZ-induced diabetic rats were more similar to the human T2DM group than the Alloxan-induced diabetic rats [69]. Most recently, a study was shown to successfully separate the blood serum of rats given a high-fat diet treatment and considered pre-diabetic from those fed a normal diet, using partial least squares discriminant analysis (PLS-DA) combined with a ROC curve. The external validation results showed the algorithm was 100% successful at making donor-level predictions [70]. While these studies’ potential is exciting, it must be noted that the work should be repeated in human trials.
Other attempts to use Raman spectroscopy combined with chemometrics for diagnosing diabetes were made using other, non-blood-based, biological samples. A portable SERS system was used in one study to analyze urine samples collected from 20 diabetic patients and 21 healthy volunteers. PCA and LDA were used for analyzing the SERS data and indicated 85% sensitivity and 90.5% specificity of the method for discriminating between the two groups. The model was deemed accurate, yielding an area under the ROC curve of 0.836, although no further validation was reported [71]. Alternatively, a portable Raman spectrometer was used to collect in vivo Raman spectra from four different skin sites: left earlobe left inner arm, left thumbnail, and left median cubital vein, each from 11 individuals with T2DM and 9 healthy controls. ANNs separated the two groups with 88.9–90.9% accuracy for the varying sample sites. A second model built using PCA and SVM resulted in lower levels of diagnostic accuracy. Both methods were validated using a tenfold CV. The results of the ANN model were comparable to those obtained using the invasive capillary blood glucose test, showcasing the technique’s success for generating objective and noninvasive diagnoses [72].
Vieira et al. used Raman spectroscopy to investigate spectroscopic changes in the dorsal root ganglia (DRG) due to diabetic neuropathy. STZ-induced diabetic neuropathic (hyperalgesia) rats were analyzed before and after photobiomodulation therapy (PBMT). PBMT is shown to treat neuropathy by relieving pain. Raman spectra showed characteristic DRG bands had increased intensities in the hyperalgesia rats, which were then reduced in the spectra collected after PBMT therapy. An LDA model was built to differentiate between the different groups with 86% success, although no validation was reported. Further research here may provide a new avenue for monitoring the treatment of diabetes and identifying potential routes for detecting the onset early on [73]. In one study by Pacia et al., a confocal Raman imaging system was used to analyze mice models’ endothelium representing diabetes, hypertension, or cancer metastasis from controls. Hierarchical cluster analysis (HCA) of the Raman spectra indicated sensitivity and specificity levels between 88 and 96% for successfully distinguishing between groups. However, no model validation was reported (Fig. 5) [74]. Interestingly, these works reveal that various chemometric methods can be used to analyze Raman spectral data to achieve the same task. While some methods work better than others, and some studies do not report using a validation mechanism, all are shown to achieve similar levels of success. Further, utilizing Raman spectroscopy with chemometric methods to distinguish between healthy and diabetic biological samples shows as much success as those studies that focus on utilizing specific new or known biomarkers for diagnoses.

Reproduced with permission from John Wiley & Sons, Ltd. [74]
The analysis of single and average spectra of the endothelium in the murine model of diabetes. The result of HCA analysis for the single endothelium spectra was obtained for db + (blue) and db/db (red) mice. a The average spectra of the endothelium of control (blue) and diabetes (red) mice with the standard error on each data point, b in the 1550–1200 cm−1 range, the Raman intensity is about threefold magnified relatively to the high wavenumber region.
Raman spectroscopy has been heavily explored in recent years to identify diabetes biomarkers and directly diagnose the disease itself. Many different avenues have been explored, but the results of all recent studies described herein signify the great potential of the method. The obvious next step would be to pursue large-scale studies that can confirm the method's statistical significance and indicate its potential application for clinical use. This is further discussed in the Critical Evaluation section.
3 Infrared Spectroscopy
Similar to the research completed using Raman spectroscopy, infrared spectroscopy was also explored for detecting diabetes in various recent research. Several groups capitalized on the advantages of IR spectroscopy to identify novel biomarkers or biochemical components useful for identifying the disease. The articles reviewed in this section include those which have used either far-, mid-, or near-IR radiation. Near-IR radiation (~14,000–4000 cm−1) is highest in energy and typically excites combination modes or overtones of molecular vibrations, mid-IR (~4000–400 cm−1) typically excites fundamental vibrations, and far-IR (~400–10 cm−1), which is the lowest in energy, is used for rotational spectroscopy and low-frequency vibrations. [75] The mid-IR region is most typically used in the research reviewed herein due to the absorption radiation of most organic compounds and inorganic ions being within that region. Notably, near-IR spectroscopy is not considered a form of vibrational spectroscopy, but due to its complementarity, the few studies which used it are still included in this review.
The most common infrared spectrometer used in the reviewed work herein is the Fourier transform infrared (FTIR) spectrometer due to its simultaneous collection of spectral data across a wide spectral range and transforming that data into a spectrum. Further, the attenuated total reflectance (ATR) accessory is also often used with FTIR spectroscopy. It enables the user to directly measure samples in the solid or liquid state without further sample preparation, typically required for transmittance FTIR [76]. In addition, many of the studies reviewed in this section incorporated chemometric methods for diagnosing diabetes due to the aforementioned benefits.
The following research studies focus on identifying diabetes through detection of glucose levels within bodily fluids, detection of novel biomarkers, or through the strict use of chemometric models.
3.1 Monitoring Blood and Saliva Glucose Levels for Indicating Diabetes
Similar to those studies conducted using Raman spectroscopy, numerous researchers have pursued using IR spectroscopy to improve the identification of elevated glucose levels to diagnose diabetes. Liu et al. evaluated the precision of four different non-invasive glucose sensing methods based on near-IR (NIR) spectroscopy, including pulse-based differential NIR spectroscopy, occlusion-based differential NIR spectroscopy, traditional NIR diffuse reflectance spectroscopy, and position diffuse NIR reflectance spectroscopy. By evaluating the measurement precision, it was determined that traditional NIR diffuse reflectance spectroscopy and position diffuse NIR spectroscopy have the greatest potential to be used in the future as glucose sensing methods [77]. Then, Jintao et al. employed a NIR fiber optic probe system to analyze plasma obtained from diabetic and normal rats to develop an in vivo blood glucose assay (Fig. 6). Spectral data were collected at 0, 15, 30, 45, 60, 90, 120, 180, and 360 min after glucose injection, with blood glucose levels, were recorded simultaneously. Two chemometric methods were employed for analyzing the data, including a PLSR model and an ANN non-regression model, each evaluated using external validation. After validation, the most optimal PLSR model reached a correlation coefficient of 96.22%. The ANN model was less successful, with the most optimum model achieving a correlation coefficient of 92.79% after validation. Unsurprisingly, the regression model showed great success for this type of study, suggesting the influence of selecting a chemometric method on accomplishing the desired diagnostic goals. [78]

Reproduced with permission from Elsevier B.V. [78]
The process of collecting NIR spectra a rat's hind leg shaved; b the NIR fiber-optical probe; c collection of the NIR spectra; d NIR spectra.
In different work, attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy was used to evaluate the saliva of non-diabetic, diabetic, and insulin-treated diabetic rats to identify biomarkers related to glucose. Two different chemometric methods, including PCA-LDA and HCA, both with LOOCV, were used to differentiate between the three groups. Interestingly, both methods reached 95.2% accuracy. Further, two spectral bands correlate with glycemia strongly and were shown to classify diabetic rats with greater than 93% sensitivity and specificity. The potential for non-invasive diabetic detection is clearly illustrated through the analysis of saliva herein [79]. While more research is necessary to pursue IR spectroscopic detection of glucose levels for diagnosing diabetes, these studies present strong support for the potential of the method to achieve this goal.
3.2 Monitoring Novel Biomarkers for Indicating Diabetes
New and alternative biomarkers have also been explored using IR spectroscopy to identify diabetes easier and more accurate. Several research studies focused on the analysis of fingernails for meeting this need. In one in-depth study, the characterization of microstructures (including both surface morphology and roughness as well as density and calcium content), materials (modulus and hardness), and macromolecules (disulfide bond content, protein content and related secondary structure) of fingernail plates were investigated. Specifically, human fingernail plates of T2DM controlled (n = 20), T2DM uncontrolled (n = 25), and healthy people (n = 30) were analyzed using FTIR spectroscopy. The results indicated that the general quality of the nail plate degrades within the T2DM controlled group but degrades even further in the T2DM uncontrolled group. Specifically, the T2DM uncontrolled group has the most porous, the least amount of dense materials (minerals) present, highly altered surface morphology, increased surface roughness, decreased amount of modulus and hardness of the nail, and decreased calcium and protein content. These values were optimal in healthy individuals and fell in the middle for the controlled T2DM group. This research poses an interesting avenue for exploring secondary complications due to T2DM, with the potential to translate the changes observed within fingernail plates into an early and noninvasive diagnostic mechanism in the future [80]. Coopman et al. also investigated fingernails, this time using ATR-FTIR spectroscopy as a tool for assessing glycation in diabetics. Fingernail clippings were obtained and analyzed from 105 healthy individuals and 127 individuals with T2DM. Using fructosamine 3-kinase, glycation and deglycation experiments of the clippings were performed. Glycation was indicated by spectral features, including increased absorption at 1047 cm−1; after deglycation, there was a general decrease in the area under the curve between 970 and 1140 cm−1. It was found that the glycated nail protein concentrations of diabetics were significantly higher than those of healthy controls, with ROC analysis yielding 82% specificity and 90% sensitivity with a cut-off value of 1.28 μmol/g nail, illustrating an alternative method for the non-invasive and effective detection of diabetes [81]. Lastly, an investigation of fingernails was executed by Monteyne et al. Here, 52 individuals with T2DM and 107 healthy controls were included in the study. Of the 107 healthy control fingernail samples, 21 were glycated in vitro at different concentrations with a glucose solution, and all individuals’ fingernails were analyzed using NIR spectroscopy. The effect of glycation had a noticeable impact on the spectral signatures, indicating a potential avenue for monitoring the onset and progression of diabetes. PLS-DA was performed to differentiate between the T2DM group and the healthy individuals, where 100% diagnostic accuracy was achieved when tested using an independent validation set. Interestingly, the advantage of incorporating chemometrics for diagnosing diabetes completely non-invasive and objective is again supported herein [82]. This triplet of large studies increasingly indicates IR spectroscopy as a potential method for a completely noninvasive method for detecting diabetes, suggesting an emphasis may be placed on analyzing fingernails in future work.
Tissue samples were analyzed in two different studies for diabetes detection. In one study, Varma et al. analyzed tissue from histologically normal kidneys (n = 4), histologically normal kidneys obtained from diabetic subjects (n = 4), and kidneys with evidence of diabetic nephropathy (n = 5). Spectral data were obtained from the glomerular basement membrane, tubular basement membrane, and mesangium of the tissue samples. PCA with LDA was shown to distinguish between the two control groups and the diabetic group and between all three groups with a very high level of separation for each tissue section analyzed (Fig. 7). The authors also identified differences in intensities of two different spectral frequencies, which could be used for an alternative separation of the groups; notably, the results of chemometrics are more definitive. However, a validation mechanism was not reported [83]. Kidney tissue sections were then studied by a different group using probe-based NIR spectroscopy. The sections were obtained from 27 individuals with normal histological findings, 26 individuals with diabetic neuropathy, and 11 with T2DM. The spectral signatures indicated differences in carbamoylation and glycation between the groups; these differences were restored after treatment with the deglycating enzyme fructosamine 3-kinase. PCA and soft independent modeling of class analogy (SIMCA) with CV showed that the groups could easily be separated [84].

Reproduced with permission from the International Society of Nephrology; published by Elsevier Inc. [83]
Linear discriminant analysis (LDA) of spectral data extracted from the glomerular basement membrane (GBM), tubular basement membrane (TBM) and the mesangium (M) of patients categorized as normal diabetic (NLD), normal nondiabetic (NL), and diabetic nephropathy (DN). LDA was performed using the complete spectral data set for each of the features studied: a GBM, b TBM, and c M.
Other researchers studied bodily fluids, including blood and saliva, were studied by other researchers for developing a diagnostic test for diabetes using IR spectroscopic detection of alternative biomarkers. Mazmuder et al. used FTIR spectroscopy to study serum samples from 85 humans, including individuals with T2DM who did or did not have retinopathy (n = 30, each) and healthy controls (n = 25). SVM models could discriminate between all three groups with an overall accuracy of 90.5% after ten-fold CV. The differences between spectral signatures indicated a variety of biochemical components as potential spectroscopic biomarkers, including carbohydrate and polysaccharide content, total lipid content, protein phosphorylation, and the Amide II group [85]. Recently, a method was devised to detect methylglyoxal (MGO), a disease-causing factor of diabetic cardiovascular complications. Here, the reaction between MGO and o-phenylenediamine produced a product with strong absorption in the far-IR range. Spectral analysis indicated that MGO could be detected at concentrations between 5 and 2500 nmol/mL, and the concentration of MGO within test blood samples was determined with 95% accuracy. The results indicated the method could be used in future clinical applications to determine the concentration of MGO and relate its presence to diabetes. [86] ATR-FTIR spectroscopy was then used in one study to differentiate between the saliva of individuals with diabetes (n = 20), individuals with different kinds of psoriasis (n = 35), and healthy controls (n = 20). The collected spectral data showed differences in the Amide I and Amide II bands, suggesting the secondary structure of proteins is altered between the groups. It was further found that the protein secondary structure between individuals with plaque psoriasis is similar to that found within patients with diabetes. Based on this information, the authors were able to conclude ATR-FTIR spectroscopy could be used as a tool to explore any potential link between psoriasis and diabetes, and further aid in developing effective treatment plans [87].
Lastly, femurs of type I diabetic (n = 6) and control (n = 5) rats were investigated using FTIR spectroscopy. The results of the analysis indicated several important characteristic differences between the two groups; these include decreased levels of mineral content, microhardness, and collagen maturity in the diabetic femurs, as well as an increase in carbonate content and size and maturation of hydroxyapatite crystals. These factors suggest that diabetes harms bones, providing information for relating the structure and function of diabetes on bone health as well as for potential diagnostic applications [88].
Given the plethora of different biomarkers which were identified in the research reviewed herein, it is obvious that there is great potential in alternative methods to identify diabetes which may reach even greater levels of accuracy than current tests can achieve. Of course, large-scale studies are required to pursue and investigate these hypotheses further to understand which the most promising, and further analysis are is required to understand the biochemical basis for the novel IR spectroscopy-determined biomarkers.
3.3 General Application of Chemometric Methods for Indicating Diabetes Within Various Biological Samples
The ability to pinpoint new and known biomarkers for diagnosing diabetes using IR spectroscopy has shown varying levels of success. A great advantage of chemometric methods resides in the ability to overlook specific biomarkers while monitoring minute changes in overall spectral data. Further, the advantages of incorporating chemometrics into diagnostic studies include the ability to make accurate and quantitative decisions without the need for subjective interpretation. The building and use of chemometric models allow for the method to be used in a variety of settings using the same standard, increasing the efficiency and efficacy of early and accurate diagnoses. In this section, research that has applied chemometric methods to identify diabetes using IR spectroscopy and without the use of biomarkers are reviewed.
Analysis of blood was used in most of the research covered in this section. ATR-FT mid-IR spectroscopy was used to analyze serum samples from 65 patients with T2DM and 55 healthy volunteers. A SVM model optimized using a genetic algorithm (GA) reached 100% sensitivity, 95.45% specificity, and 97.87% accuracy for discriminating between the two groups during external validation, indicating one of the most successful reports yet for detecting T2DM [89] Yang et al. employed ATR-FTIR spectroscopy for the detection of prediabetes via analysis of peripheral blood. Here, fasting blood glucose levels and glucose levels at hour 2 during the OGTT were measured from 112 individuals to determine the control group and the prediabetic group. ATR-FTIR spectra were recorded from those blood samples simultaneously; classification and regression trees (CART) and extreme gradient Boosting (XGBoost) ensemble algorithms were both used to develop the prediabetes diagnostic tests. The CART model achieved 80% specificity and 95% sensitivity, while the XGBoost model reached 100% specificity and 85% sensitivity. The accuracy for the CART and XGBoost models were 86.67% and 93.33%, respectively. All results were reported after external validation. The superior XGBoost method indicates a real potential for the accurate detection of prediabetes within individuals [90]. Guang et al. also utilized ATR-FTIR spectroscopy in combination with XGBoost to analyze whole blood samples, here to diagnose T2DM. Whole blood was collected from 51 T2DM individuals and 55 healthy individuals. The most optimum XGBoost model achieved a sensitivity of 95.23%, specificity of 96%, and accuracy of 95.65% after external validation, further illustrating the success of IR spectroscopy with chemometrics for identifying diabetes [91]. Interestingly, this chemometric method of XGBoost was not explored in any other recent studies, despite its success in these two works.
A different study used IR spectroscopy to investigate non-alcoholic steatohepatitis (NASH), which is associated with the occurrence of T2DM as well as cardiovascular complications. In the largest study reviewed, 395 severely obese individuals who underwent a bariatric procedure were considered in the study; 66 of those individuals had NASH. Spectra of serum from the individuals were analyzed using a logistic regression model, with the performance evaluated using the area under the ROC curve (AUROC). After external validation, the AUROC was 0.77, with an associated sensitivity of 69% and specificity of 76%. When a composite model was built, incorporating aspartate aminotransferase levels, triglyceride levels, and waist circumference in addition to the IR spectral data, the AUROC increased to 0.84 after external validation. While intriguing, this study could potentially benefit from a different and less complicated method of analysis to reach higher classification results; however, based on the sample size, this study provides one of the most realistic evaluations of IR spectroscopy for identifying diabetes [92]. In a related manner, Bernardes-Oliveira utilized ATR-FTIR to investigate blood plasma obtained from 50 healthy pregnant women and 50 pregnant women with gestational diabetes mellitus. Several different chemometric methods were employed for separating the two groups, including LDA, quadratic discriminant analysis (QDA), and SVM. The best results were obtained with a GA-LDA model, which reached accuracy, sensitivity, and specificity levels all of 100% after validation using an independent test set. Lipids and proteins were found to be the most useful spectral features for separation. These results indicate a very successful route for low-cost and minimally invasive detection of gestational diabetes mellitus [93].
In a final study, pancreatic tissues were examined from healthy and the non-obese diabetic model for type 1 diabetes mice as well as from humans and analyzed using both Raman spectroscopy and FTIR spectroscopy. Analysis of the data collected through orthogonal PLS-DA with external validation resulted in the successful understanding of the biochemical profiles of the different pancreatic tissues. This work provides a stepping stone for generating in vivo diagnostic assessments through the analysis of pancreatic biochemistry, but results of the chemometric modeling were not reported [94].
Interestingly, the methods which were used to analyze IR spectroscopic data to detect diabetes were just as successful as those summarized using Raman spectroscopy. IR spectroscopy can identify potential novel biomarkers for monitoring the progression of the disease, and the spectra can be further analyzed via chemometric methods for objective and accurate detection of diabetes. Clearly, due to the high number of publications and research generated in the last five years alone, the utility of the method is quite promising.
4 Critical Evaluation
There has been an evident success in advancing vibrational spectroscopy for detecting diabetes in the past five years. Both infrared and Raman spectroscopy have shown the obvious potential to not only monitor spectroscopic biomarkers throughout both the onset and various forms of treatment of the disease but to also be able to objectively detect the disease within biological samples with high levels of diagnostic accuracy. It would be difficult to argue against the capacity of vibrational spectroscopy for future use in clinical settings for detecting and diagnosing diabetes. While other analytical methods for diagnosing diabetes have also been shown to be useful, including chromatography-based tests, enzymatic-based assays, and antibody-based immunoassays, these are beyond the scope of this review and the reader is referred to other work for more information [95,96,97,98].
A summary of all studies in this review which were conducted using Raman spectroscopy and using IR spectroscopy is seen in Tables 1 and 2, respectively. These tables synthesize the category of study, which was accomplished, the type of sample which was used (with animal models noted as appropriate), the number of samples analyzed in the study, the type of chemometric methods used, if any, and the type of validation that was employed if any. It is important to summarize these factors, as they can have a noticeable impact on the results that are reached, especially including the sample size used and the method of validation employed.
Based on this summary, there are some interesting conclusions that can be drawn. Although more research was accomplished using Raman spectroscopy, those studies which used IR spectroscopy more frequently analyzed a greater number of samples. This is an important distinction to make, as large-scale clinical trials are necessary to validate findings that are made in smaller work [100]. While small-scale studies can show success, the true nature of that success will not be realized until a large study is conducted. Additionally, 12 of the 43 studies reviewed utilize animal models instead of human samples. While animal models are well established for studying diabetes [101, 102], repeating the work using human donors, which typically have more complex regulatory measures. A positive study conducted within animals does not guarantee its success within humans [103, 104]. Therefore, a statistically significant number of human donors is of utmost importance to ensure that a research plan can be applied to the general population and not a small subset of individuals, as is commonly targeted in work reviewed herein. Unfortunately, no recent research has accomplished this goal yet.
The category of study employed is notably diverse between the research reviewed herein. Many (in fact, 19 of the 25 studies using Raman spectroscopy and 12 of the 18 studies using IR spectroscopy) focus on identifying new or known biomarkers for diabetes; interestingly, 22 of these also utilize chemometrics in addition to monitoring spectral biomarkers. Many of the noted novel biomarkers highlighted in the previous papers are common biochemical components that are not necessarily specific to diabetes or have not been previously linked to the development and progression of diabetes. While some biomarkers, such as glycated hemoglobin and blood glucose levels, are well-established biomarkers for the disease, the papers which applied chemometrics methods without searching for the presence of biomarkers are shown to be just as successful as those which focus solely on detecting them. While understanding the biochemical differences between healthy and diseased individuals is important, a significant advantage of vibrational spectroscopy is the ability to pinpoint spectroscopic differences without fully understanding the biochemical cause, saving both time and effort for making a diagnosis. Additionally, multiple biochemical components contribute to the vibrational spectrum produced, including contributions from biochemical components that the scientific community may not yet consider for the disease in question. Through probing the entire biochemical composition of the sample and not limiting the focus to a specific biomarker or two, the specificity and accuracy for identifying a disease can potentially be increased [105]. For this reason, those papers that incorporate chemometric methods should be focused on in the future. The advantages of chemometrics allow it to be used as an objective and accurate approach for identifying diabetes without limiting the focus to one or two notable biomolecules.
It should be noted that the studies reviewed herein which incorporated the use of chemometrics faced their own set of limitations. While almost every study mentioned reached high levels of diagnostic accuracy, comparable with currently employed diabetes diagnostic methods, proper validation of the applied chemometric method is not always observed (Tables 1 and 2) [106]. There are two main types of validation typically used in diagnostic studies. The first is considered cross-validation and refers to the general method of testing the model’s performance with the same set of data used to build it. Cross-validation may often overstate the success of a model due to overfitting and provide overly optimistic results; this is especially true when studies are conducted with small sample sizes [100, 107, 108]. Methods of cross-validation include leave-one-out (spectrum or sample) and n-fold cross-validation. Leave-one-out cross-validation involves the automatic process of leaving either a single spectrum or all spectra from one sample out of the model building process; the data left out is then used to test the model's performance. This process is repeated until all data has been left out. Similarly, n-fold cross-validation randomly divides the total dataset into n groups and builds the model with n-1 groups; the group which is left out is again repeatedly used for evaluating the model’s performance. On the other hand, external validation is a much more reliable and trustworthy method for interpreting the capabilities of a chemometric model. External validation refers to testing a model with independent data (i.e. data that was not involved in the model building process). Successful external validation is a key indication that a model is not biased to the data used to build it. It is likely to be successful when expanded to a real-world application, such as use within clinical settings [100, 107]. While crucial, external validation requires a larger number of samples to be incorporated into a study, which is not always feasible due to various issues such as the availability of volunteers or funding.
Interestingly, among those studies which incorporated chemometrics, all reported results achieved sensitivity, specificity, and accuracy levels greater than 80% and up to 100%. While the definition of a successful varies for several reasons, all studies that used chemometrics can be considered as erring on the side of success based on these parameters alone. Interestingly, the types of samples used, the number of samples studied, and the chemometric method employed vary among these studies widely. Within this small subset of research, a pattern for a useful combination of vibrational spectroscopy cannot be established. While various biological samples were used among these studies, including urine, saliva, fingernails, and others, blood seemed to be the most frequently employed, suggesting this biological specimen as the one to focus on in the future. Unsurprisingly, among those studies that focused on identifying biomarkers and using chemometrics, glucose and glycated proteins were the most commonly targeted biomolecules. A variety of chemometric techniques are used in relatively similar frequency levels, suggesting that the method chosen may not significantly impact the success of the research. Again, it should be noted that all studies reviewed here are considered small compared to large-scale clinical trials. While some results are reported after external validation, all results should be taken with a degree of caution.
Assuming the “perfect” (ideally a large-scale trial using human samples and with proper external validation of the chemometric method) trial can be implemented to verify the real success of vibrational spectroscopy for detecting diabetes, there remain further hurdles to overcome to introduce the method to clinical settings. While this topic is beyond the scope of this review, several prominent research groups have addressed this issue [21, 22, 109, 110]. Importantly, vibrational spectroscopists must prove to those in the medical community that spectroscopy can be used as a valid means for diagnosing T2DM for the method to enter clinical settings smoothly. A unified approach to analyzing samples and the chemometric model employed would also make implementation much more straightforward to understand.
Despite the many issues which still need to be addressed, it is clear that vibrational spectroscopy holds unique advantages for diagnosing diabetes. Both IR and Raman spectroscopy is easy-to-use, fast, and simple methods that provide objective and accurate diagnostic predictions. The specificity of the methods provides crucial details that can differentiate between stages of the disease and monitor disease progression and the effects of treatment. Vibrational spectroscopy is shown herein to have an incredible potential to revolutionize and simplify the way diabetes is diagnosed, creating great opportunities for early intervention and treatment, with the potential to prevent the onset of diabetes-related complications and even save lives.
References
Diagnosis and classification of diabetes mellitus. Diabetes Care 30, S42-S47 (2006)
Harris, M.I., Eastman, R.C.: Early detection of undiagnosed diabetes mellitus: a US perspective. Diabetes Metab. Res. Rev. 16(4), 230–236 (2000)
Malkani, S., Mordes, J.P.: Implications of using hemoglobin A1C for diagnosing diabetes mellitus. Am. J. Med. 124(5), 395–401 (2011)
Saudek, C.D., Herman, W.H., Sacks, D.B., Bergenstal, R.M., Edelman, D., Davidson, M.B.: A new look at screening and diagnosing diabetes mellitus. J. Clin. Endocrinol. Metab. 93(7), 2447–2453 (2008)
Makris, K., Spanou, L.: Is there a relationship between mean blood glucose and glycated hemoglobin? J. Diabetes Sci. Technol. 5(6), 1572–1583 (2011)
Goldstein, D.E.: How much do you know about glycated hemoglobin testing? Clinical Diabetes 13(4), 60–65 (1995)
Al Shehri, Z. S.: The relationship between some biochemical and hematological changes in type 2 diabetes mellitus. Biomed. Res. Ther. 4(11), 1760–1774 (2017)
Mediani, A., Abas, F., Maulidiani, M., Sajak, A.A.B., Khatib, A., Tan, C.P., Ismail, I.S., Shaari, K., Ismail, A., Lajis, N.H.: Metabolomic analysis and biochemical changes in the urine and serum of streptozotocin-induced normal-and obese-diabetic rats. J. Physiol. Biochem. 74(3), 403–416 (2018)
Abou-Seif, M.A., Youssef, A.-A.: Evaluation of some biochemical changes in diabetic patients. Clin. Chim. Acta 346(2), 161–170 (2004)
Salek, R.M., Maguire, M.L., Bentley, E., Rubtsov, D.V., Hough, T., Cheeseman, M., Nunez, D., Sweatman, B.C., Haselden, J.N., Cox, R.D.: A metabolomic comparison of urinary changes in type 2 diabetes in mouse, rat, and human. Physiol. Genomics 29(2), 99–108 (2007)
Vashist, S.K.: Non-invasive glucose monitoring technology in diabetes management: a review. Anal. Chim. Acta 750, 16–27 (2012)
Oliver, N.S., Toumazou, C., Cass, A.E.G., Johnston, D.G.: Glucose sensors: a review of current and emerging technology. Diabetic Med. 26(3), 197–210 (2009)
Yadav, J., Rani, A., Singh, V., Murari, B.M.: Prospects and limitations of non-invasive blood glucose monitoring using near-infrared spectroscopy. Biomed. Signal Process. Control 18, 214–227 (2015)
Pandey, R., Paidi, S.K., Valdez, T.A., Zhang, C., Spegazzini, N., Dasari, R.R., Barman, I.: Noninvasive monitoring of blood glucose with raman spectroscopy. Acc. Chem. Res. 50(2), 264–272 (2017)
Vashist, S.K., Zheng, D., Al-Rubeaan, K., Luong, J.H.T., Sheu, F.-S.: Technology behind commercial devices for blood glucose monitoring in diabetes management: a review. Anal. Chim. Acta 703(2), 124–136 (2011)
Jia, W.: Standardising HbA1c-based diabetes diagnosis: opportunities and challenges. Expert Rev. Mol. Diagn. 16(3), 343–355 (2016)
Pandey, R., Dingari, N.C., Spegazzini, N., Dasari, R.R., Horowitz, G.L., Barman, I.: Emerging trends in optical sensing of glycemic markers for diabetes monitoring. TrACx Trends Anal. Chem. 64, 100–108 (2015)
Zarkogianni, K., Litsa, E., Mitsis, K., Wu, P.-Y., Kaddi, C.D., Cheng, C.-W., Wang, M.D., Nikita, K.S.: A review of emerging technologies for the management of diabetes mellitus. IEEE Trans. Biomed. Eng. 62(12), 2735–2749 (2015)
Sakudo, A.: Near-infrared spectroscopy for medical applications: current status and future perspectives. Clin. Chim. Acta 455, 181–188 (2016)
Finlayson, D., Rinaldi, C., Baker, M.J.: Is infrared spectroscopy ready for the clinic? Anal. Chem. 91(19), 12117–12128 (2019)
Pahlow, S., Weber, K., Popp, J., Bayden, R.W., Kochan, K., Rüther, A., Perez-Guaita, D., Heraud, P., Stone, N., Dudgeon, A.: Application of vibrational spectroscopy and imaging to point-of-care medicine: a review. Appl. Spectrosc. 72(101), 52–84 (2018)
Baker, M.J., Byrne, H.J., Chalmers, J., Gardner, P., Goodacre, R., Henderson, A., Kazarian, S.G., Martin, F.L., Moger, J., Stone, N.: Clinical applications of infrared and Raman spectroscopy: state of play and future challenges. Analyst 143(8), 1735–1757 (2018)
Dayakar, T., Rao, K.V., Park, J., Sadasivuni, K.K., Rao, K.R.: Non-enzymatic biosensing of glucose based on silver nanoparticles synthesized from Ocimum tenuiflorum leaf extract and silver nitrate. Mater. Chem. Phys. 216, 502–507 (2018)
Dayakar, T., Rao, K.V., Bikshalu, K., Malapati, V., Sadasivuni, K.K.: Non-enzymatic sensing of glucose using screen-printed electrode modified with novel synthesized CeO2@CuO core shell nanostructure. Biosens. Bioelectron. 111, 166–173 (2018)
Dayakar, T., Ponnamma, D., Sadasivuni, K.K., Cabibihan, J.-J., Al-Ali, A.K., Malik, R.A., Min, B.: Progress of advanced nanomaterials in the non-enzymatic electrochemical sensing of glucose and H2O2. Biosensors 10(11), 151 (2020)
Yempally, S., Hegazy, S.M. Aly, A., Kannan, K., Sadasivuni, K.K.: Non-invasive diabetic sensor based on cellulose acetate/graphene nanocomposite. In: Macromolecular Symposia, pp. 2000024. Wiley Online Library (2020)
Ember, K.J.I., Hoeve, M.A., McAughtrie, S.L., Bergholt, M.S., Dwyer, B.J., Stevens, M.M., Faulds, K., Forbes, S.J., Campbell, C.J.: Raman spectroscopy and regenerative medicine: a review. NPJ Regener. Med. 2(1), 12 (2017)
Ralbovsky, N.M., Lednev, I.K.: Towards development of a novel universal medical diagnostic method: Raman spectroscopy and machine learning. Chem. Soc. Rev. 49, 7428–7453 (2020)
Tu, Q., Chang, C.: Diagnostic applications of Raman spectroscopy. Nanomed. Nanotechnol. Biology Med. 8(5), 545–558 (2012)
Loyola-Leyva, A., Hernández-Vidales, K., Loyola-Rodríguez, J.P., González, F.J.: Raman spectroscopy applications for the diagnosis and follow-up of type 2 diabetes mellitus. A brief review. Biomed. Spectrosc. Imaging 9(3–4), 119–140 (2020)
Jahn, I.J., Radu, A.I., Weber, K., Cialla-May, D., Popp, J.: Surface enhanced raman spectroscopy for medical diagnostics. In: Nanotechnology Characterization Tools for Biosensing and Medical Diagnosis, pp. 1–66. Springer (2018)
Ozaki, Y., Mizuno, A., Kamada, Y., Itoh, K., Iriyama, K.: Laser Raman spectroscopic study of a diabetic cataractous lens. Chem. Lett. 11(6), 887–890 (1982)
Haynes, C.L., McFarland, A.D., Van Duyne, R.P.: Surface-enhanced Raman spectroscopy. Anal. Chem. 338A-346A (2005)
Pilot, R., Signorini, R., Durante, C., Orian, L., Bhamidipati, M., Fabris, L.: A review on surface-enhanced Raman scattering. Biosensors 9(2), 57 (2019)
Gnyba, M., Smulko, J., Kwiatkowski, A., Wierzba, P.: "Portable Raman spectrometer-design rules and applications. Bull. Pol. Acad. Sci. Tech. Sci. 59(3), 325–329 (2011)
McCreery, R.L., Fleischmann, M., Hendra, P.: Fiber optic probe for remote Raman spectrometry. Anal. Chem. 55(1), 146–148 (1983)
Wold, S.: Chemometrics; what do we mean with it, and what do we want from it? Chemom. Intell. Lab. Syst. 30(1), 109–115 (1995)
Ralbovsky, N.M., Lednev, I.K.: Raman spectroscopy and chemometrics: a potential universal method for diagnosing cancer. Spectrochim. Acta, Part A 219, 463–487 (2019)
Kong, K., Kendall, C., Stone, N., Notingher, I.: Raman spectroscopy for medical diagnostics—from in-vitro biofluid assays to in-vivo cancer detection. Adv. Drug Delivery Rev. 89, 121–134 (2015)
Reddy, R.K., Bhargava, R.: Chemometric methods for biomedical Raman spectroscopy and imaging. In Emerging Raman Applications and Techniques in Biomedical and Pharmaceutical Fields, pp. 179–213. Springer (2010)
Little, R.R.: Glycated hemoglobin standardization–National glycohemoglobin standardization program (NGSP) perspective. Clin. Chem. Lab. Med. 41(9), 1191–1198 (2003)
Selvin, E., Steffes, M.W., Zhu, H., Matsushita, K., Wagenknecht, L., Pankow, J., Coresh, J., Brancati, F.L.: Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults. N. Engl. J. Med. 362(9), 800–811 (2010)
Pandey, R.: Raman spectroscopy-based sensing of glycated hemoglobin: critical analysis and future outlook. J. Postdoctoral Res. 3, 8–16 (2015)
Barman, I., Dingari, N.C., Kang, J.W., Horowitz, G.L., Dasari, R.R., Feld, M.S.: Raman spectroscopy-based sensitive and specific detection of glycated hemoglobin. Anal. Chem. 84(5), 2474–2482 (2012)
González-Viveros, N., Castro-Ramos, J., Gómez-Gil, P., Cerecedo-Núñez, H.H.: Characterization of glycated hemoglobin based on Raman spectroscopy and artificial neural networks. Spectrochim. Acta Part A. 247, 119077 (2020)
Villa-Manríquez, J.F., Castro-Ramos, J., Gutiérrez-Delgado, F., Lopéz-Pacheco, M.A., Villanueva-Luna, A.E.: Raman spectroscopy and PCA-SVM as a non-invasive diagnostic tool to identify and classify qualitatively glycated hemoglobin levels in vivo. J. Biophotonics 10(8), 1074–1079 (2017)
Chan, J.W.: Recent advances in laser tweezers Raman spectroscopy (LTRS) for label-free analysis of single cells. J. Biophotonics 6(1), 36–48 (2013)
Lin, J., Shao, L., Qiu, S., Huang, X., Liu, M., Zheng, Z., Lin, D., Xu, Y., Li, Z., Lin, Y.: Application of a near-infrared laser tweezers Raman spectroscopy system for label-free analysis and differentiation of diabetic red blood cells. Biomed. Opt. Express 9(3), 984–993 (2018)
Jesudason, D.R., Dunstan, K., Leong, D., Wittert, G.A.: Macrovascular risk and diagnostic criteria for type 2 diabetes: implications for the use of FPG and HbA1c for cost-effective screening. Diab. Care 26(2), 485–490 (2003)
Clarke, W.L., Cox, D., Gonder-Frederick, L.A., Carter, W., Pohl, S.L.: Evaluating clinical accuracy of systems for self-monitoring of blood glucose. Diab. Care 10(5), 622–628 (1987)
Li, N., Zang, H., Sun, H., Jiao, X., Wang, K., Liu, T.C.-Y., Meng, Y.: A noninvasive accurate measurement of blood glucose levels with raman spectroscopy of blood in microvessels. Molecules 24(8), 1500 (2019)
Singh, S.P., Mukherjee, S., Galindo, L.H., So, P.T., Dasari, R.R., Khan, U.Z., Kannan, R., Upendran, A., Kang, J.W.: Evaluation of accuracy dependence of Raman spectroscopic models on the ratio of calibration and validation points for non-invasive glucose sensing. Anal. Bioanal. Chem. 410(25), 6469–6475 (2018)
Ju, J., Hsieh, C.-M., Tian, Y., Kang, J., Chia, R., Chang, H., Bai, Y., Xu, C., Wang, X., Liu, Q.: Surface enhanced Raman spectroscopy based biosensor with a microneedle array for minimally invasive in vivo glucose measurements. ACS Sensors 5, 1777–1785 (2020)
Zheng, Y., Zhu, X., Wang, Z., Hou, Z., Gao, F., Nie, R., Cui, X., She, J., Peng, B.: Noninvasive blood glucose detection using a miniature wearable Raman spectroscopy system. Chin. Opt. Lett. 15(8), 083001 (2017)
Birech, Z., Mwangi, P.W., Bukachi, F., Mandela, K.M.: Application of Raman spectroscopy in type 2 diabetes screening in blood using leucine and isoleucine amino-acids as biomarkers and in comparative anti-diabetic drugs efficacy studies. PLoS One 12(9), (2017). e0185130
Chege, B.M., Birech, Z., Mwangi, P.W., Bukachi, F.O.: Utility of Raman spectroscopy in diabetes detection based on biomarker Raman bands and in antidiabetic efficacy studies of herbal extract Rotheca myricoides Hochst. J. Raman Spectrosc. 50(10), 1358–1366 (2019)
Clark, C.M., Fradkin, J.E., Hiss, R.G., Lorenz, R.A., Vinicor, F., Warren-Boulton, E.: Promoting early diagnosis and treatment of type 2 diabetesthe national diabetes education program. JAMA 284(3), 363–365 (2000)
Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352(9131), 837–853 (1998)
The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N. Engl. J. Med. 329(14), 977–986 (1993)
Silveira, L., Borges, R.d.C.F., Navarro, R.S., Giana, H.E., Zângaro, R.A., Pacheco, M.T.T., Fernandes, A.B.: Quantifying glucose and lipid components in human serum by Raman spectroscopy and multivariate statistics. Lasers Med. Sci. 32(4), 787–795 (2017)
González-Solís, J.L., Villafan-Bernal, J.R., Martinez-Zerega, B., Sánchez-Enríquez, S.: Type 2 diabetes detection based on serum sample Raman spectroscopy. Lasers Med. Sci. 33(8), 1791–1797 (2018)
Lin, J., Lin, X., Hu, C., Bai, P., Yang, H., Dai, Y., Qiu, H., Lin, M., Feng, S., Pan, J.: Silver nanoparticle based surface-enhanced Raman spectroscopy for label-free discrimination of diabetic albumin under near-infrared laser excitation. Laser Phys. Lett. 15(9), 095703 (2018)
Roman, M., Kamińska, A., Drożdż, A., Platt, M., Kuźniewski, M., Małecki, M.T., Kwiatek, W.M., Paluszkiewicz, C., Stępień, E.Ł: "Raman spectral signatures of urinary extracellular vesicles from diabetic patients and hyperglycemic endothelial cells as potential biomarkers in diabetes. Nanomed. Nanotechnol. Biology Med. 17, 137–149 (2019)
Flores-Guerrero, J.L., Muñoz-Morales, A., Narea-Jimenez, F., Perez-Fuentes, R., Torres-Rasgado, E., Ruiz-Vivanco, G., Gonzalez-Viveros, N., Castro-Ramos, J.: Novel assessment of urinary albumin excretion in type 2 diabetes patients by raman spectroscopy. Diagnostics 10(3), 141 (2020)
Shi, P., Liu, H., Deng, X., Jin, Y., Wang, Q., Liu, H., Chen, M., Han, X.: Label-free nonenzymatic glycation monitoring of collagen scaffolds in type 2 diabetic mice by confocal Raman microspectroscopy. J. Biomed. Opt. 20(2), 027002 (2015)
Paolillo, F.R., Mattos, V.S., de Oliveira, A.O., Guimarães, F.E.G., Bagnato, V.S., de Castro Neto, J.C.: Noninvasive assessments of skin glycated proteins by fluorescence and Raman techniques in diabetics and nondiabetics. J. Biophotonics 12(1), e201800162 (2019)
González-Solís, J.L., Torres-González, L.A., Villafán-Bernal, J.R.: Superparamagnetic clustering of diabetes patients Raman spectra. J. Spectrosc. 2019, 4296153 (2019)
Singh, A.K., Mazumder, A.G., Halder, P., Ghosh, S., Chatterjee, J., Roy, A.: Raman spectral probe and unique fractal signatures for human serum with diabetes and early stage diabetic retinopathy. Biomed. Phys. Eng. Expr. 5(1), 015021 (2018)
Wang, L., Liu, G.D., Mu, X., Xiao, H.B., Qi, C., Zhang, S.Q., Niu, W.-Y., Jiang, G.K., Feng, Y.N., Bian, J.Q.: Red blood cells Raman spectroscopy comparison of type two diabetes patients and rats. Guang Pu Xue Yu Guang Pu Fen Xi 35(10), 2776–2780 (2015)
Ralbovsky, N.M., Fitzgerald, G.S., McNay, E.C., Lednev, I.K.: Towards development of a novel screening method for identifying Alzheimer’s disease risk: Raman spectroscopy of blood serum and machine learning. Spectrochim. Acta Part A 119603 (2021)
Zou, Y., Huang, M., Wang, K., Song, B., Wang, Y., Chen, J., Liu, X., Li, X., Lin, L., Huang, G.: Urine surface-enhanced Raman spectroscopy for non-invasive diabetic detection based on a portable Raman spectrometer. Laser Phys. Lett. 13(6), 065604 (2016)
Guevara, E., Torres-Galván, J.C., Ramírez-Elías, M.G., Luevano-Contreras, C., González, F.J.: Use of Raman spectroscopy to screen diabetes mellitus with machine learning tools. Biomed. Opt. Express 9(10), 4998–5010 (2018)
Vieira, W.F., de Magalhães, S.F., Farias, F.H., de Thomaz, A.A., Parada, C.A.: Raman spectroscopy of dorsal root ganglia from streptozotocin‐induced diabetic neuropathic rats submitted to photobiomodulation therapy. J. Biophotonics 12(11), e201900135 (2019)
Pacia, M.Z., Mateuszuk, L., Buczek, E., Chlopicki, S., Blazejczyk, A., Wietrzyk, J., Baranska, M., Kaczor, A.: Rapid biochemical profiling of endothelial dysfunction in diabetes, hypertension and cancer metastasis by hierarchical cluster analysis of Raman spectra. J. Raman Spectrosc. 47(11), 1310–1317 (2016)
Yu, P., Kirkpatrick, R.J., Poe, B., McMillan, P.F., Cong, X.: Structure of calcium silicate hydrate (C-S-H): Near-, Mid-, and Far-infrared spectroscopy. J. Am. Ceram. Soc. 82(3), 742–748 (1999)
Glassford, S.E., Byrne, B., Kazarian, S.G.: Recent applications of ATR FTIR spectroscopy and imaging to proteins. Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics, vol. 1834, no. 12, pp. 2849–2858, 2013.
Liu, J., Liu, R., Xu, K.: Accuracy of noninvasive glucose sensing based on near-infrared spectroscopy. Appl. Spectrosc. 69(11), 1313–1318 (2015)
Jintao, X., Liming, Y., Yufei, L., Chunyan, L., Han, C.: Noninvasive and fast measurement of blood glucose in vivo by near infrared (NIR) spectroscopy. Spectrochim. Acta, Part A 179, 250–254 (2017)
Caixeta, D.C., Aguiar, E.M.G., Cardoso-Sousa, L., Coelho, L.M., Oliveira, S.W., Espindola, F.S., Raniero, L., Crosara, K.T.B., Baker, M.J. Siqueira, W.L.: Salivary molecular spectroscopy: a sustainable, rapid and non-invasive monitoring tool for diabetes mellitus during insulin treatment. PloS One 15(3), e0223461 (2020)
Sihota, P., Yadav, R.N., Dhiman, V., Bhadada, S.K., Mehandia, V., Kumar, N.: Investigation of diabetic patient’s fingernail quality to monitor type 2 diabetes induced tissue damage. Sci. Rep. 9, 3193 (2019)
Coopman, R., Van de Vyver, T., Kishabongo, A.S., Katchunga, P., Van Aken, E.H., Cikomola, J., Monteyne, T., Speeckaert, M.M., Delanghe, J.R.: Glycation in human fingernail clippings using ATR-FTIR spectrometry, a new marker for the diagnosis and monitoring of diabetes mellitus. Clin. Biochem. 50(1–2), 62–67 (2017)
Monteyne, T., Coopman, R., Kishabongo, A.S., Himpe, J., Lapauw, B., Shadid, S., Van Aken, E.H., Berenson, D., Speeckaert, M.M., De Beer, T.: Analysis of protein glycation in human fingernail clippings with near-infrared (NIR) spectroscopy as an alternative technique for the diagnosis of diabetes mellitus. Clin. Chem. Lab. Med. 56(9), 1551–1558 (2018)
Varma, V.K., Kajdacsy-Balla, A., Akkina, S.K., Setty, S., Walsh, M.J.: A label-free approach by infrared spectroscopic imaging for interrogating the biochemistry of diabetic nephropathy progression. Kidney Int. 89(5), 1153–1159 (2016)
De Bruyne, S., Van Dorpe, J., Himpe, J., Van Biesen, W., Delanghe, S., Speeckaert, M.M., Delanghe, J.R.: Detection and characterization of a biochemical signature associated with diabetic nephropathy using near-infrared spectroscopy on tissue sections. J. Clin. Med. 8(7), 1022 (2019)
Mazumder, A.G., Banerjee, S., Zevictovich, F., Ghosh, S., Mukherjee, A., Chatterjee, J.: Fourier-transform-infrared-spectroscopy based metabolomic spectral biomarker selection towards optimal diagnostic differentiation of diabetes with and without retinopathy. Spectrosc. Lett. 51(7), 340–349 (2018)
Wu, X., Dai, Y., Wang, L., Peng, Y., Lu, L., Zhu, Y., Shi, Y., Zhuang, S.: Diagnosis of methylglyoxal in blood by using far-infrared spectroscopy and o-phenylenediamine derivation. Biomed. Opt. Express 11(2), 963–970 (2020)
Bottoni, U., Tiriolo, R., Pullano, S.A., Dastoli, S., Amoruso, G.F., Nisticò, S.P., Fiorillo, A.S.: Infrared saliva analysis of psoriatic and diabetic patients: similarities in protein components. IEEE Trans. Biomed. Eng. 63(2), 379–384 (2015)
Bozkurt, O., Bilgin, M.D., Evis, Z., Pleshko, N., Severcan, F.: Early alterations in bone characteristics of type I diabetic rat femur: a fourier transform infrared (FT-IR) imaging study. Appl. Spectrosc. 70(12), 2005–2015 (2016)
Fang, T., Li, Y., Li, F., Huang, F.: Rapid diagnosis of type II diabetes using fourier transform mid-infrared attenuated total reflection spectroscopy combined with support vector machine. Anal. Lett. 51(9), 1400–1416 (2018)
Yang, X., Fang, T., Li, Y., Guo, L., Li, F., Huang, F., Li, L.: Pre-diabetes diagnosis based on ATR-FTIR spectroscopy combined with CART and XGBoots. Optik 180, 189–198 (2019)
Guang, P., Huang, W., Guo, L., Yang, X., Huang, F., Yang, M., Wen, W., Li, L.: Blood-based FTIR-ATR spectroscopy coupled with extreme gradient boosting for the diagnosis of type 2 diabetes: A STARD compliant diagnosis research. Medicine 99(15), e19657 (2020)
Anty, R., Morvan, M., Le Corvec, M., Canivet, C.M., Patouraux, S., Gugenheim, J., Bonnafous, S., Bailly-Maitre, B., Sire, O., Tariel, H., Bernard, J., Piche, T., Loréal, O., Aron-Wisnewsky, J., Clément, K., Tran, A., Iannelli, A., Gual, P.: The mid-infrared spectroscopy: a novel non-invasive diagnostic tool for NASH diagnosis in severe obesity. JHEP Reports 1(5), 361–368 (2019)
Bernardes-Oliveira, E., de Freitas, D.L.D., de Morais, C.d.L.M., de Mesquita Cornetta, M.d.C., Camargo, J.D.d.A.S., de Lima, K.M.G., de Oliveira Crispim, J.C.: Spectrochemical differentiation in gestational diabetes mellitus based on attenuated total reflection Fourier-transform infrared (ATR-FTIR) spectroscopy and multivariate analysis, Sci. Rep. 10, 19259 (2020)
Nord, C., Eriksson, M., Dicker, A., Eriksson, A., Grong, E., Ilegems, E., Mårvik, R., Kulseng, B., Berggren, P.-O., Gorzsás, A.: Biochemical profiling of diabetes disease progression by multivariate vibrational microspectroscopy of the pancreas. Sci. Rep. 7, 6646 (2017)
Gupta, S., Jain, U., Chauhan, N.: Laboratory diagnosis of HbA1c: a review. J. Nanomed Res. 5(4), 00120 (2017)
Grant, D.A., Dunseath, G.J., Churm, R., Luzio, S.D.: Comparison of a point-of-care analyser for the determination of HbA1c with HPLC method. Pract. Lab. Med. 8, 26–29 (2017)
International expert committee report on the role of the A1C assay in the diagnosis of diabetes. Diab. Care 32(7), 1327–1334 (2009)
Sultanpur, C.M., Deepa, K., Kumar, S.V.: Comprehensive review on HbA1c in diagnosis of diabetes mellitus. Int. J. Pharm. Sci. Rev. Res. 3(2), 119–122 (2010)
Šťovíčková, L., Tatarkovič, M., Logerová, H., Vavřinec, J., Setnička, V.: Identification of spectral biomarkers for type 1 diabetes mellitus using the combination of chiroptical and vibrational spectroscopy. Analyst 140(7), 2266–2272 (2015)
Jain, A.K., Chandrasekaran, B.: 39 Dimensionality and sample size considerations in pattern recognition practice. Handbook Statist. 2, 835–855 (1982)
Rees, D.A., Alcolado, J.C.: Animal models of diabetes mellitus. Diabet. Med. 22(4), 359–370 (2005)
King, A.J.F.: The use of animal models in diabetes research. Br. J. Pharmacol. 166(3), 877–894 (2012)
Roberts, I., Kwan, I., Evans, P., Haig, S.: Does animal experimentation inform human healthcare? Observations from a systematic review of international animal experiments on fluid resuscitation. Br. Med. J. 324(7335), 474–476 (2002)
Hackam, D.G., Redelmeier, D.A.: Translation of research evidence from animals to humans. JAMA 296(14), 1727–1732 (2006)
Ralbovsky, N., Lednev, I.K.: Raman hyperspectroscopy shows promise for diagnosis of Alzheimer’s. Biophotonics 4(25), 33–37 (2018)
Steyerberg, E.W., Harrell, F.E., Jr.: Prediction models need appropriate internal, internal-external, and external validation. J. Clin. Epidem. 69, 245–247 (2016)
Kanal, L., Chandrasekaran, B.: On dimensionality and sample size in statistical pattern classification. Pattern Recogn. 3(3), 225–234 (1971)
Vabalas, A., Gowen, E., Poliakoff, E., Casson, A.J.: Machine learning algorithm validation with a limited sample size. PloS One 14(11), e0224365 (2019)
Santos, I.P., Barroso, E.M., Bakker Schut, T.C., Caspers, P.J., van Lanschot, C.G.F., Choi, D.-H., Van Der Kamp, M.F., Smits, R.W.H., Van Doorn, R., Verdijk, R.M., Noordhoek Hegt, V., von der Thüsen, J.H., van Deurzen, C.H.M., Koppert, L.B., van Leenders, G.J.L.H., Ewing-Graham, P.C., van Doorn, H.C., Dirven, C.M.F., Busstra, M.B., Hardillo, J., Sewnaik, A., ten Hove, I., Mast, H., Monserez, D.A., Meeuwis, C., Nijsten, T., Wolvius, E.B., Baatenburg de Jong, R.J., Puppels, G.J., Koljenović, S.: Raman spectroscopy for cancer detection and cancer surgery guidance: translation to the clinics. Analyst 142(17), 3025–3047 (2017)
Wilson, B.C., Jermyn, M., Leblond, F.: Challenges and opportunities in clinical translation of biomedical optical spectroscopy and imaging. J. Biomed. Opt. 23(3), 030901 (2018)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Ralbovsky, N.M., Lednev, I.K. (2022). Infrared and Raman Spectroscopy Assisted Diagnosis of Diabetics. In: Sadasivuni, K.K., Cabibihan, JJ., A M Al-Ali, A.K., Malik, R.A. (eds) Advanced Bioscience and Biosystems for Detection and Management of Diabetes. Springer Series on Bio- and Neurosystems, vol 13. Springer, Cham. https://doi.org/10.1007/978-3-030-99728-1_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-99728-1_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-99727-4
Online ISBN: 978-3-030-99728-1
eBook Packages: EngineeringEngineering (R0)