Abstract
Organic pollutants are predominantly synthetic compounds made or extracted by humans, and it is therefore not surprising that they are common contaminants in urban soils. This chapter provides details of the immense range of potential organic pollutants in urban soils, including their types, physical properties, chemical structures, and sources. The behaviour of non-polar, polar, and ionic organic compounds in soils is explained in terms of their key chemical reactions, including abiotic and biological degradation, and transport phenomena are also addressed, before a brief discussion of toxicity. Sampling, extraction, and chemical analysis methods are covered briefly, together with background concentrations, regulatory contamination thresholds, and contamination indices. A comprehensive case study is focused on polycyclic aromatic hydrocarbons (PAH) in the highly urbanised Pearl River Delta region of China. This case study uses the PAH family of compounds to illustrate many of the concepts needed to understand the behaviour of persistent organic pollutants in urban soils, concluding with their fluxes and a conceptual PAH cycle.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords

What you could learn from this chapter:
-
The types of organic contaminants that are of concern are in urban soils and their sources.
-
The processes controlling immobilisation, mobility, and bioavailability of organic contaminants.
-
What the fluxes of organic contaminants are between air, water, sediment, and soil and the factors controlling these, including a case study of the Pearl River Delta, Guangdong, PR China.
-
Being aware of chemical analysis methods for organic contaminants in soils.
7.1 Sources of Organic Contaminants in Urban Soils
An enormous diversity of organic compounds has been added to soils in urban environments. Very few of these compounds are naturally occurring in soils, and therefore even low concentrations represent contamination . The much less common natural occurrence of organic pollutants is an important point of difference from inorganic pollutants such as metals. There is some natural occurrence of compounds in the PAH and dioxin categories from phenomena such as wildfires, but even for completely anthropogenic compounds, there are still ‘background’ concentrations in soils in minimally disturbed natural environments (Biasioli et al. 2012). Background concentrations of organic contaminants represent widespread dispersion of anthropogenic compounds by global atmospheric circulation by the late twentieth century (Wania and MacKay 1996). For example, Fuoco et al. (2009) describe the occurrence of persistent organic pollutants in Antarctica, derived from both remote sources and local use of fuels and other organic chemicals. Some important categories of organic pollutants and their main sources are listed on Table 7.1.
7.1.1 Types of Organic Compounds
McBride (1994) identifies some important properties of organic compounds which affect their behaviour, such as retention or mobility, in soils. These include the type and chemical reactivity of organic functional groups, the size and shape of organic molecules, and polarity/polarisability or charge of organic molecules and ions. The susceptibility of an organic compound to oxidation or reduction is also important. These molecular properties also affect physical properties such as volatility (e.g. vapour pressure or boiling point), density, or solubility in water or other solvents which, in turn, affect their behaviour in soils and similar environments. One of the more important aspects of the behaviour of organic compounds in soils is whether or not they are readily degraded by soil (micro)organisms, a distinction which contributes to separating the persistent organic pollutants (POPs) from other organic chemicals.
7.1.1.1 Categories of Organic Contaminants Based on General Chemical Properties
Polarity and charge (ionisation) of organic molecules are some of the more important properties affecting their behaviour (McBride 1994). We also distinguish organic molecules based on whether they consist of linear or branched chains of carbon atoms (aliphatic) or contain ring structures with carbon-carbon double bonding that involves delocalisation of electrons (aromatic); some examples of chemical structures are shown in Table 7.2 together with information on ionisability and polarity and potential sources. In both aliphatic and aromatic compounds, carbon atoms are bonded either to other carbon atoms or to hydrogen atoms. Much differentiation between organic compounds also depends on the presence in the molecules of specific arrangements of atoms, or functional groups . These functional groups often define a class of organic compounds and may include atoms other than carbon and hydrogen: oxygen, nitrogen, sulphur, phosphorus, or halogens such as fluorine, chlorine, or bromine.
The chemical structure of organic pollutants is an important determinant of their toxicity and persistence in environments. Of the 12 ‘persistent organic pollutant’ (POP) compounds originally listed by the United Nations’ Stockholm Convention, all contain both chlorine atoms and rings of carbon atoms. In some cases, the compounds were designed to be toxic (the original 12 POPs included 8 pesticides), but their effects on non-target species, including humans, were not initially considered. Other chemicals such as the PCBs, hexachlorobenzene, and the dioxins and dibenzofurans (see Tables 7.1 and 7.2) were either intended for other uses or, in the case of the PCDD/F compounds, were unintentional by-products of other anthropogenic processes. In humans and other mammals, exposure to the Stockholm Convention POPs can result in adverse effects to multiple physiological systems including endocrine, immune, digestive, and reproductive systems. Many of the compounds are also known or suspected to increase the incidence of cancers and have been shown to produce birth defects.
Much of the risk associated with persistent organic pollutants relates to their tendency to bioaccumulate and biomagnify – that is, they are not excreted efficiently from the body, so they accumulate and also tend to increase in concentration towards higher levels in food chains which increases the risk to predators and humans.
Persistent organic pollutants (POPs) are anthropogenic organic chemicals which persist for many years once released into the environment. In addition, POPs tend to bioaccumulate and are toxic to humans and other organisms (Rodríguez-Eugenio et al. 2018). The POPs (Table 7.3) are generally highly regulated, based on the revised Stockholm Convention of the United Nations Environment Programme (Secretariat of the Stockholm Convention 2018).
7.2 Controls on Organic Contaminant Behaviour in Urban Soils
7.2.1 Non-polar, Non-ionic Compounds
The organic compounds which are both non-ionic and non-polar include the unsubstituted hydrocarbons (i.e. those composed of only C and H) and some halogenated hydrocarbons having molecules with high symmetry such as carbon tetrachloride, tetrachloroethylene, or 1,3,5-trichlorobenzene. Some molecules with low polarity (e.g. 1,2-dichlorobenzene, DCB) behave similarly and are also classified as non-polar in some contexts (Kile et al. 1995). A good operational measure of whether an organic compound behaves as polar or not is the octanol-water partition coefficient, Kow, (Eq. 7.1):
where Co is the concentration of compound in n-octanol and Cw is the concentration of compound in n-octanol, both in contact and at equilibrium.
A very non-polar compound will be much more soluble in the low-polarity liquid n-octanol compared with water and have a large Kow (or, commonly, log10(Kow)) value compared with a polar compound.
Very non-polar organic compounds, if present in soils in large enough concentrations (e.g. from a leak or spill), can be present as a separate liquid phase, commonly called a non-aqueous-phase liquid or NAPL. The NAPL group is subdivided on the basis of density relative to water into the LNAPL (light non-aqueous-phase liquid) and DNAPL (dense non-aqueous-phase liquid) subcategories (Fig. 7.1). The separation of LNAPLs and DNAPLs on the basis of density relative to water is important, since their density is one of the primary factors affecting how separate NAPL phases will behave in a soil-groundwater system. Light NAPLs, such as petrol/gasoline, will ‘float’ on top of the groundwater; conversely, dense NAPLs will sink through the groundwater until reaching a confining layer such as a clay lens or impermeable bedrock. This tendency to float or sink also affects how contamination with LNAPLs or DNAPLs is treated, as we discuss in Chap. 11.
7.2.1.1 Adsorption of Non-polar Organics
The Kow value is also extremely useful in explaining the retention of organic chemicals by soils or sediments. Natural organic matter in soils and sediments behaves as an organic ‘solvent’ into which non-polar organic compounds preferentially partition into, by a related mechanism to their partitioning into n-octanol. The contents of soil organic matter and the associated black carbon are therefore the main factor affecting adsorption of non-polar organic compounds in soils. The partition coefficient for soil organic matter is related to Kow by a linear log-log relationship, and so retention can be predicted if the organic carbon content of soil or sediment is known. Retention of organic contaminants in soils is commonly described by KD, the adsorption (or partition) coefficient:
where Cs is the concentration of compound adsorbed to soil and Cw is the concentration of compound in water at equilibrium with soil.
The organic carbon normalised adsorption coefficient, KOC, is defined by (Eq. 7.3):
where foc is the mass fraction of organic carbon in soil.
The affinity of an organic compound for soil can then, in theory, be estimated from Kow and foc, without the need for direct measurement of adsorption on soil. In real soils and sediments, the relationship between KOC and Kow is not perfectly linear, conferring some statistical uncertainty to estimation of KD. Figure 7.2 shows an example of a measured relationship between log KOC and log Kow, which illustrates this uncertainty (Li and Ran 2012). In this example, prediction of log KOC from any log Kow value may fall within a range 1.2–1.33 units wide, corresponding to a 16- to 22-fold difference in possible log KOC values. For accurate prediction of adsorption-dependent behaviour such as leaching or degradation, therefore, laboratory measurement of KD on the actual soil or sediment involved is likely to be necessary.
Relationship between log10Kow and log10Koc for PAH adsorption on sediment from the Pearl River, Guangdong, China, showing the prediction and confidence intervals for the linear regression model. (Redrawn from Li and Ran (2012); used under the terms of a CC-BY-3.0 license)
The dependence of pesticide adsorption on soil on the soil organic carbon content is shown by the example in Fig. 7.3. The positive slopes of the relationship between (log10)-KD and soil organic carbon suggest that, for a 1% increase in soil organic carbon content, the strength of adsorption as measured by KD increases by about fivefold. More recently, it has been recognised that organic matter in the form of [micro]plastic contamination can also act as an adsorbent for organic contaminants (Rodríguez-Eugenio et al. 2018). If the organic matter itself is mobile (as is the case for some naturally occurring humic macromolecules), adsorption may actually increase transport of organic pollutants (Enfield et al. 1989).
Dependence of adsorption coefficient KD on soil organic carbon content for various organic pollutants (pesticides) on several soils. (Drawn by Andrew W. Rate from data in Hiller et al. (2008))
7.2.1.2 Volatilisation
Many organic compounds, particularly those with low polarity and low molecular weight, evaporate readily at ambient temperatures. Such compounds are often called volatile organic hydrocarbons, or VOCs, and include common pollutants such as light petroleum hydrocarbons, trichloroethylene, and the BTEX group of compounds. Volatilisation of these compounds is known to be an important mechanism for transfer of some pollutants from soils to the atmosphere (Petersen et al. 1996; Martí et al. 2014). As the case study later in this chapter (Sect. 7.5) will discuss, however, volatilisation from soil to air of organic compounds other than VOCs, such as PAHs , does occur and needs to be considered in order to fully understand their behaviour and fate in the environment.
7.2.2 Behaviour of Polar or Ionisable Organic Compounds
Compounds which have high polarity, or readily ionisable compounds, have much greater solubility in water (such as soil pore water) than do the non-polar compounds discussed above. High polarity compounds tend to be those having short (1–4 atom) carbon chains, with functional groups in asymmetric positions containing atoms such as oxygen, nitrogen, or sulphur with different electronegativity than carbon, such as simple alcohols or ketones. Readily ionisable compounds include carboxylic acids, amides, and amines, and the tendency to ionise is related to acid-base reactions with water as a solvent.
Volatilisation of polar organic solvents tends to be less than for non-polar organic solvents, since intermolecular attraction of polar molecules lowers their vapour pressure. Polar compounds do volatilise, however, such as low molecular weight alcohols (e.g. methanol, ethanol) and ketones (e.g. acetone).
Ionisation. Organic molecules containing carboxylic, sulfonic, or phosphoric acid functional groups are able to ionise in water, by acid dissociation reactions, to form carboxylate or sulfonate anions, leading to high water solubility, since water can easily solvate most ionic chemical species. Similarly, organic molecules containing amino functional groups are basic and can accept hydrogen ions from water to form cationic species. Ionisable pollutants include many pesticides (e.g. anionic 2,4-D or metsulfuron-methyl; cationic triazine herbicides) (McBride 1994; Kah and Brown 2006). In addition, some organic compounds (e.g. glyphosate, the active ingredient of the widely used herbicide Roundup®) can form ‘zwitterions’, having combinations of functional groups which can allow molecules to have both positive and negative charge.
7.2.3 Adsorption of Ionisable and Polar Organics
Organic compounds can accumulate as ions or molecules at the surface of finely particulate substances in soils, such as secondary minerals and organic matter, by similar mechanisms to those described for inorganic substances in Chaps. 4 and 6.
7.2.3.1 Cation Exchange
Organic compounds which are ionisable to form organic cations tend to be adsorbed onto the surfaces of negatively charged soil solids such as clays and organic matter by cation exchange (McBride 1994). Other mechanisms such as hydrophobic interactions (i.e. non-polar partitioning as described in Sect. 7.2.1), hydrogen bonding, and charge transfer can also result in adsorption of cationic pesticides (Kah and Brown 2006). Adsorption of organic cations such as the triazine herbicides on soils generally increases with increasing soil organic carbon content, since much of the CEC of many soils is conferred by negative charge on soil organic matter and since adsorption by non-polar partitioning also increases with soil organic carbon content (Fig. 7.3). The effect of soil pH on organic cation adsorption is complex; generally, organic cations have decreased adsorption as pH increases, which is counter-intuitive if we only consider increasing pH as increasing the pH-dependent negative charge on soil colloids. The decrease in adsorption of organic cations with increasing pH relates to the ionisation of the organic molecules themselves; at low pH the molecules are cationic since their weakly basic amino or pyridyl functional groups accept a hydrogen ion to become positively charged. As the pH increases beyond the pKa value (−log10 acid dissociation constant) of the organic cation, the molecule loses its charge and can no longer interact electrostatically with a negatively charged soil surface. Adsorption of organic cations on clay minerals such as smectites is probably by physical processes rather than chemical bonding. The ability of clays such as smectites with expanding interlayers to adsorb organic cations is restricted by the molecular size of the cations, such that larger cations (e.g. quaternary ammonium compounds) are excluded from interlayer spaces (McBride 1994).
7.2.3.2 Adsorption of Anionic Organics
Molecules with weakly acidic functional groups (carboxylic acids, –NHSO2– acids, sulfonic acids) become progressively more dissociated as pH increases, producing organic anions and hydrogen ions. The acid dissociation constants of the undissociated acids are usually large enough, however, that the molecules are almost completely dissociated within normal soil pH ranges. Adsorption occurs by ion exchange if positively charged soil colloids such as ferric oxides exist, by ligand exchange on clays or oxide minerals, or by other mechanisms such as cation bridging (Kah and Brown 2006). Organic anion adsorption generally decreases with increasing pH, due to the increasing negative charge on soil colloids as pH increases. Adsorption of anionic organic contaminants increases with increasing content of soil organic carbon, reactive ferric oxides, or cation exchange capacity (Fig. 7.4).
Association, based on component weightings from principal component analysis, of adsorption constants for anionic organic contaminants ( ) and soil properties (
) and soil properties ( ), with observation scores for individual soils 1–10 (◻). MSM metsulfuron-methyl, IMHP 2-isopropyl-4-methyl-6-hydroxypyrimidine (a metabolite of diazinon), TCP 3,5,6-trichloro-2-pyridinol (a metabolite of chlorpyrifos). The clustering of adsorption constants with organic carbon (OC), amorphous iron oxides (FeOx), and CEC in principal component space suggests that these soil properties exert the most control on adsorption. (Redrawn from Báez et al. (2015); used with permission from Springer)
), with observation scores for individual soils 1–10 (◻). MSM metsulfuron-methyl, IMHP 2-isopropyl-4-methyl-6-hydroxypyrimidine (a metabolite of diazinon), TCP 3,5,6-trichloro-2-pyridinol (a metabolite of chlorpyrifos). The clustering of adsorption constants with organic carbon (OC), amorphous iron oxides (FeOx), and CEC in principal component space suggests that these soil properties exert the most control on adsorption. (Redrawn from Báez et al. (2015); used with permission from Springer)
7.2.4 Chemical Degradation (Abiotic) of Organic Compounds
Organic contaminants degrade in soils, with the main cause of degradation being metabolism by soil microorganisms, a topic that will be addressed in sections of Chaps. 8 and 11. The biochemical degradation processes do not always convert organic contaminants directly to CO2, and intermediary degradation products, or metabolites, are produced which may have similar toxicity to the original compound. For example, the organochlorine insecticide DDT (now banned in many jurisdictions) is metabolised to several products including o,p′-DDE, p,p′-DDE, and o,p′-DDD. The persistence and toxicity of DDE, the predominant DDT metabolite, is greater than for DDT itself; DDD has also been used as a pesticide. Conversely, the metabolites of 2,3,7,8-TCDD, the most toxic of the dioxin (PCDD/PCDF) group of compounds, are considerably less toxic than the parent 2,3,7,8-TCDD.
Many organic compounds are oxidisable or reducible, and chemical oxidation or reduction is pathway for abiotic degradation of organic pollutants in soils (Mulligan and Yong 2004). The principal oxidising agent, or electron acceptor, in soils is oxygen; other substances, such as FeIII or MnIV oxides, can also react as oxidising agents, and some clays can catalyse oxidation by oxygen by forming reactive oxygen species (Yong and Mulligan 2003). In the presence of aluminosilicate clays, for example, reactive oxygen species can degrade phenols by oxidation to quinones. Barrett and McBride (2005) showed that glyphosate (N-(phosphonomethyl)glycine) could also be degraded by an oxidation reaction where a manganese oxide was the electron acceptor. Reductive processes are perhaps better known in soils, especially reductive dechlorination of chlorinated hydrocarbons such as PCE (perchloroethylene = tetrachloroethene), TCE (trichloroethene), and carbon tetrachloride. Reduction of chlorinated organic contaminants can occur if other substances (e.g. polycarboxylic acids such as oxalate or citrate, which can be released by plant roots, or natural organic matter) reduce Fe3+ to Fe2+ at a ferric oxide surface; the surface Fe2+ ions can then facilitate the reductive degradation (Li et al. 2008a). Zerovalent (metallic) iron (ZVI) is a powerful reducing agent which can also reductively dechlorinate organic compounds, and the use of ZVI in remediation is discussed in Chap. 11. Other naturally occurring substances such as Fe2+ or pyrite can also reduce organic compounds; their presence in soil would normally require anoxic conditions.
Many organic molecules, including persistent and other pollutants, can be degraded by ultraviolet light derived from solar radiation (e.g. Zhong et al. 1994; John and Shaike 2015). The efficiency of photodegradation is lower in soil than in air or water (Kromer et al. 2004), and the importance of photodegradation for organic pollutant removal in urban soils is essentially unknown, but not considered to be significant relative to other removal mechanisms.
7.2.5 Biological Degradation
Some soil microorganisms (including species of bacteria and fungi) can use contaminant compounds as a metabolic energy source and growth substrate, decomposing the contaminants in the process. We call this biological decomposition process biodegradation , and the end products are carbon dioxide (or intermediary carbon compounds, or metabolites). We discuss the biodegradation process in more detail in Chap. 8, including explanations of the factors affecting bioavailability of different organic compounds.
From a perspective of chemical kinetics, the biodegradation rates of organic contaminants in soils are commonly first-order processes, meaning that the degradation rate is dependent only on the concentration of the contaminant compound, and the half-life is constant. First-order behaviour was confirmed for a range of pesticides and soils by Jury et al. (1987), whose analysis showed that the reaction rates observed were dependent on the half-life for a specific compound (i.e. related to the pollutant) and the KOC value (Eq. 7.3) for adsorption (i.e. related to soil properties). Other soil properties are also known to affect the rates (and therefore amounts) or biodegradation that occurs. In many cases, the ability of microorganisms to degrade pollutants is limited by the external supply of water and/or nutrients such as nitrogen and phosphorus (Shahsavari et al. 2017). In many pollutant environments, organic contaminants coexist with other contaminants such as metals. The presence any one of several metals (e.g. Cd, Cr, Cu, Hg, Pb, or Zn) at sufficient concentrations can inhibit biodegradation of compounds such as pesticides, PAHs , or petroleum hydrocarbons (Sandrin and Maier 2003).
As with inorganic contaminants (Chap. 6), the bioavailability of organic pollutants is affected by their residence time in soil, and this is known as an ‘aging effect’. Biodegradation is slower and occurs to a lesser extent, for contaminants which have been in contact with soil for longer, compared with very recently added contaminants (Hatzinger and Alexander 1995). In processes such as composting , where microbial stabilisation of organic materials is the primary objective, the bioavailability of organic pollutants is also known to decrease (Chen et al. 2015b).
7.2.6 Transport of Organic Contaminants
Organic compounds can be transferred within soils or between soils and other environmental compartments (air, surface water, groundwater, biota) by a number of processes (Fig. 7.5). The existence of widespread diffuse pollution of Earth’s environments with organic contaminants is strong evidence that transport of organic contaminants is a significant issue (Fuoco et al. 2009; Rodríguez-Eugenio et al. 2018; Sect. 7.1). Some transport phenomena specifically related to polycyclic aromatic hydrocarbons (PAH) are described in Sect. 7.5 later in this chapter, with Sect. 7.5.7 explicitly covering fluxes of PAH compounds to and from soils.
Fluxes from direct human use or production of organic compounds to soils (also called ‘primary emissions’; Wang et al. 2016) include:
-
Accidental loss, leakage, or spillage (e.g. of fuel hydrocarbons or during manufacture or use of most classes of organic compound – see Table 7.1).
-
Deliberate application to soils (e.g. pre-emergent herbicides).
-
Incidental addition to soils from other deliberate activities (e.g. spraying of pesticides onto crops, use of fire retardants).
-
Unintentional generation during combustion (e.g. formation of PAHs or dioxins from coal or biomass burning).
-
Entry of organic pollutants into wastewater streams which may eventually be deliberately or accidentally applied to soil (e.g. detergents or antibiotics in sewage sludge).
Atmosphere-soil fluxes include additions to soils from wet and dry deposition and emissions from soils by volatilisation. The additions to soils are from the primary emission processes described above. Secondary emissions from soil to atmosphere occur for volatile organic pollutants, depending on several factors. Higher temperatures favour volatilisation (Rodríguez-Eugenio et al. 2018), and greater soil water contents suppress volatilisation by reducing air-filled pore space and by Henry’s Law partitioning of organic compounds into soil water (Grathwohl 2011). Emissions from soil by volatilisation are also limited by adsorption of organic compounds (see Sect. 7.5.7) or if biodegradation is rapid (Grathwohl 2011).
Soil-surface water fluxes are mainly from run-off, although wind erosion and subsequent deposition of atmospheric particulates containing organic pollutants are also possible. The partitioning of organic pollutants into organic matter may increase the transport of these pollutants in the dissolved form by association with dissolved organic matter (Enfield et al. 1989). Transport from soil to water can also occur when pollutants are adsorbed to particulate organic matter, by soil erosion or loss of soil during urbanisation (Wang et al. 2016).
Soil-groundwater fluxes occur via leaching, that is, vertical transport through soil by percolating water. As we would expect, leaching is greater for organic compounds with greater water solubility (Mulligan et al. 2001) and depends on sufficient precipitation or other water input to soil to allow transport of water through the soil matrix or preferential flow through macropores. Reactions between organic contaminants and soil sold phases suppress leaching to groundwater, as do biodegradation (Grathwohl 2011). The partitioning of hydrophobic organic compounds into dissolved organic matter macromolecules may increase the transport of pollutants in soil water, even if the pollutants have low water solubility (Enfield et al. 1989).
Transport mechanisms for pesticides within and between environmental compartments. (Adapted from Rodríguez-Eugenio et al. 2018; reproduced with permission from UNEP)
7.3 Effects of Organic Soil Contaminants on Ecosystem Services
The toxicity of organic pollutants to a range of organisms is well established (Rodríguez-Eugenio et al. 2018), and we address some of the effects and issues involved in Chap. 8, particularly for soil microorganisms. Organic pollutants such as petroleum hydrocarbons, PAHs , PCBs, organochlorine or organophosphate pesticides, and PCDDs/PCDFs have also been shown to be toxic to organisms other than microorganisms in soils or sediments, including plants, insects, earthworms, crustaceans, and burrowing vertebrates. In addition, many of the persistent organic pollutants (POPs) are known to undergo biomagnification as pollutants taken up by organisms lower in food webs are consumed by predators (Rodríguez-Eugenio et al. 2018). The toxicity of POPs varies widely with type and species of organism. While some plants show adverse effects such as reduced growth and photosynthetic activity (Ahammed et al. 2012), others are more resistant to toxic effects and may be able to take up organic pollutants to an extent that they can be used for phytoremediation (Wyrwicka et al. 2014). Similarly, while some microbial processes such as respiration or nitrification may be affected by organic pollutants such as PAHs or pesticides (Sverdrup et al. 2007; John and Shaike 2015), some microorganisms are able to degrade organic pollutants, a phenomenon which may also be used for remediation of contaminated soils (Chap. 11).
The toxicity of many organic pollutants to humans is well-known; for example, benzo[a]pyrene is a recognised human carcinogen. In contrast, however, benzo[a]pyrene in soil does not always show adverse effects on plants or invertebrate animals (Sverdrup et al. 2007). Vertebrates other than humans have negative responses to organic pollutants; for example, Gonzalez-Mille et al. (2013) showed DNA damage could occur to a toad (amphibian) species exposed to urban soil contaminated with POPs (mainly organochlorine pesticides such as hexachlorocyclohexane and DDT and also PCBs).
7.4 Measurements and Data
7.4.1 Total and Partial Analyses
7.4.1.1 Soil Sampling for Organic Compounds
The sampling methods for hydrocarbons differ depending on whether the target organic compound(s) are volatile or semi/non-volatile. For volatile compounds such as C5–C10 alkanes, BTEX, and 2–3-ring PAHs (see Sect. 7.5.2), sampling techniques must avoid the loss of target compounds by evaporation. Such methods include zero-headspace sampling in specialised containers, or solvent extraction (Sadler and Connell 2003). Less rigorous sampling procedures are required for semi- or non-volatile compounds.
7.4.1.2 Extraction of Organic Compounds from Soil
Materials sampled for volatile organic compounds by zero-headspace or solvent extraction methods need no further extraction prior to analysis. Non-volatile or semi-volatile compounds, however, may be strongly adsorbed to soil materials (Sect. 7.2.1) and require extraction with a solvent able to remove target compounds from adsorbed forms. These include the commonly used Soxhlet extraction using solvents such as acetone/hexane or methylene chloride/acetone (US EPA 1996a) or extraction in specialised high-pressure vessels with supercritical fluids such as liquid CO2 (e.g. US EPA 1996b). The supercritical fluid extraction methods may also be applied to volatile organic compounds. Adaptations of these methods are applicable to several other categories of organic contaminants, such as organochlorine pesticides, phenols, PCBs, and so on.
It is worthwhile noting that solvent extraction procedures do not extract all of the organic contaminants present in soil or sediment samples. The extractability, for example, of PAHs , is controlled by organic carbon and black carbon content (Ma et al. 2010). He et al. (2008) found that between 34% and 57% of PAHs in soils and sediments was non-extractable in dichloromethane in a Soxhlet procedure, with between 235 and 1425 mg/kg of total PAHs not extracted in some samples.
7.4.1.3 Analysis of Organic Compounds in Extracts
Since soils usually contain a range of potential organic contaminant compounds, analytical methods usually involve a chromatography technique which separates compounds according to their polarity or other chemical property. The most common of these is gas chromatography (US EPA 2018), which may be coupled with a mass-spectrometer detector to increase the method’s ability to identify individual compounds unambiguously. This textbook does not have space to cover all the details of chemical analytical techniques, so readers are referred to a general analytical chemistry text such as the excellent open-access book by Harvey (2016).
7.4.2 Background Concentrations
Many potentially polluting organic compounds have natural sources, and so background concentrations in urban soils must be considered. Natural phenomena such as volcanism, forest fires, crude oil seeps, decomposition of organic residues, or plant emissions can emit organic compounds such as hydrocarbons including those in the BTEX and PAH categories, phenols, dioxins, and others. In contrast, numerous organic compounds such as many chlorinated hydrocarbons including PCBs, PFAS, and most pesticides (i.e. most of the compounds identified as POPs in Table 7.3) have no known natural sources. Even for compounds which do not occur naturally, however, measurable background concentrations may occur in urban soils due to the widespread use and emissions of these substances.
Any assessment of organic contaminants should therefore account for unavoidable background concentrations of potential pollutants caused by diffuse source emissions from urban areas. This type of background concentration is called the ‘anthropic background’ by Biasioli et al. (2012), who used the 90th percentile (excluding outliers) of multiple observations to calculate background concentrations of benzo[a]pyrene (31.5 μg/kg), PCBs (8.9 μg/kg), and PCDDs+PCDFs (3.5 μg/kg). Most other studies which report background concentrations for organic pollutants in urban or peri-urban areas rely either on measurements at non-urban sites or concentrations cited in other scientific literature.
7.4.3 Regulatory Contamination Thresholds
Contamination thresholds for organic contaminants soil are based on an analysis of risk, including the desired level of protection, exposure pathways, and the types of organisms which may potentially be affected (e.g. National Environment Protection Council 2013a). More detail on risk assessment processes is presented in Chap. 9. Some examples of regulatory contamination threshold concentrations in soil, for organic contaminants, appear in Table 7.4, for compounds from the BTEX, PAH, and PCDD/PCDF categories.
7.4.4 Contamination Indices
The concept of contamination indices, as discussed in Chap. 6 for trace elements (metals and metalloids), is seldom used explicitly, probably because the practice of summing concentrations of pollutants in a particular category is so common. As the case study below (see Sect. 7.5.2) observes, it is normal for polycyclic aromatic hydrocarbon (PAH) concentrations to be presented as the sum of several (typically 9–28) critical compounds (even though there are hundreds of PAH compounds). The same practice is used for other classes of compounds; for example, Biasioli et al. (2012) summed the concentrations of polychlorinated biphenyls (PCBs) and also polychlorinated dibenzo-dioxins and furans (PCDDs/PCDFs) rather than presenting the concentrations of individual compounds. Similarly, summed concentrations of phthalic acid esters (Mo et al. 2009) and organochlorine pesticides (You 2015) have been reported instead of separate compounds. Effectively, a sum of concentrations of a class of contaminants (e.g. ∑PAH) is analogous to a simple additive contamination index, as presented in Chap. 6.
7.5 Case Study: Polycyclic Aromatic Hydrocarbons (PAHs ) in the Pearl River Delta
The focus on the Pearl River Delta region of China in this textbook is quite deliberate – we think that it is representative of many urban environments on Earth. As well as being one of the largest conurbations worldwide, it is also one of the fastest growing in terms of population, and like many large urban areas, it has a legacy of soil contamination with a range of substances including metals and persistent organic pollutants. Similarly, we have chosen to focus on polycyclic aromatic hydrocarbons (PAHs) as representative organic pollutants. This does not mean that other organic pollutants are less important or relevant, but PAHs have been studied now for a long time, and we think that many aspects of their behaviour in the Pearl River Delta are more widely applicable to other pollutants or other regions.
7.5.1 The Pearl River Delta
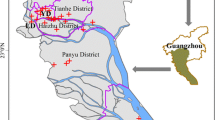
The Pearl River Delta (PRD) in Guangdong Province, People’s Republic of China, is a large (54,156 km2) and industrialised area which contains a series of interlinked megacities, including Guangzhou, Shenzhen, and Hong Kong. Table 7.5 shows the sizes of these urban areas in the PRD region in terms of their land areas and human populations. The area, extending from Yangjiang in the west to Shanwei in the east, to approximately Qingyuan and Heyuan in the north, and bounded in the south by the South China Sea (Nan Hai) (Fig. 7.6), is still rapidly urbanising, and an estimated 120 million people live in the greater Pearl River Delta area (UN-Habitat 2016). The PRD region is known to be heavily polluted with a wide range of contaminants and other anthropogenic substances, including nutrients, trace metals, a range of organic pollutants including PAHs and many other POPs, as well as emerging contaminants such as pharmaceuticals, perfluoroalkyl compounds, and microplastics (Wang and Rainbow 2020).
Location map of the Pearl River (珠江口, Zhujiang) Delta, showing the major waterways and urban centres. (Modified by Andrew W. Rate from Liang et al. (2019), used under the terms of a CC-BY-4.0 license)
7.5.2 Polycyclic Aromatic Hydrocarbons
The polycyclic aromatic hydrocarbons (PAHs) are a group of persistent organic pollutant compounds with chemical structures based on chemically bonded benzene (6-carbon aromatic) ring structures sharing at least two carbon atoms (see the example of phenanthrene in Table 7.2). They range in molecular size from the smallest, naphthalene, with two rings, up to molecules with six to nine rings such as benzo[ghi]perylene (see Table 7.6) and numerous other compounds which are generally less common as environmental pollutants (Achten and Andersson 2015a). In some of the compounds, 5-membered rings are also present (e.g. acenaphthene, fluorene, indeno[1,2,3-cd]pyrene, etc.).
The concentrations of PAHs in soils may be reported individually, and it is very common for a sum of PAH concentrations to be reported, such as the 16 PAHs prioritised by the US EPA (2020; see Table 7.6 and Fig. 7.7). The abbreviation Σ16PAHs is commonly used to represent the combined concentrations of the 16 US EPA priority PAHs (or Σ15PAHs since naphthalene is sometimes omitted), or a different number subscript may be used to show how many individual PAH compounds were analysed for (e.g. Σ28PAHs). Seven of the 16 US EPA priority PAHs are known to be carcinogenic: benzo[a]anthracene, chrysene, benzo[b]fluoranthene, benzo[k]fluoranthene, benzo[a]pyrene, indeno[1,2,3-cd]pyrene, and dibenzo[a,h]anthracene (abbreviated variously as Σ7PAHs, Σ7CarPAHs, ΣcarcPAHs, etc.).
The PAHs occur naturally in many soils in small concentrations due to their presence in geogenic hydrocarbon deposits such as oil shales or crude oil and emission by natural wildfires or volcanic activity (National Pollutant Inventory 2020). In environments affected by urbanisation, however, PAHs are released in greater amounts into various environmental compartments due to their presence in fossil fuels and solvents and their release during combustion of coal, other fossil fuels, and biomass for energy production (Chen et al. 2005). Another source of PAH pollution is waste disposal, especially disposal or burning of e-waste (Leung et al. 2006; Wang et al. 2017).
Lower molecular weight PAHs may volatilise significantly or be transported in the atmosphere by sorption to airborne particulates. Contamination of soils, surface water, and groundwater with PAHs is well known in many urban environments worldwide (Menzie et al. 1992; Wang and Rainbow 2020), and exchange between environmental compartments does occur, including uptake by plants from soils.
7.5.3 PAH Concentrations in Soils of the Pearl River Delta
The concentrations of PAHs in soils are generally reported in units of μg/kg (or equivalently ng/g); both units correspond to parts per billion to reflect the low concentrations of PAHs in uncontaminated environments. In a range of studies across the PRD, in urban and peri-urban soils used for vegetable growing, parklands, urban forest, and even e-waste recycling, ∑16PAH concentrations ranged from undetectable (Li et al. 2007) to >12,000 μg/kg (Wei et al. 2014 and Fig. 7.8). The mean concentrations in urban surface soils, if presented, were generally in the range 150–320 μg/kg. The focus on surface soils is reasonable since the risk of transfers by wind erosion, plant uptake, or direct soil contact is greatest in the soil surface. Some of the difference between findings of different studies may be caused by the differences in sampling depth for ‘surface’ soils; while most studies sampled 0–20 cm depth, a few sampled 0–10 cm (e.g. Leung et al. 2006; Ni et al. 2011), and one study sampled soil from 0 to 5 cm (Zhang et al. 2016).
Map of the distribution of ∑28PAHs in soils of the Pearl River Delta. (From Wei et al. (2014) showing PAH pollution centred on Foshan, Guangzhou, and Dongguan. Reproduced with permission from Elsevier)
The concentration ranges measured in PRD soils are often lower than other urbanised areas worldwide (Ma et al. 2009; Cao et al. 2010), perhaps reflecting the shorter history of intense urbanisation. Chen et al. (2005) found that background concentrations of ∑16PAHs in vegetable-growing soils in Guangzhou were between 1 and 10 μg/kg, lower than background concentrations between 20 and 50 μg/kg in the Netherlands. Menzie et al. (1992) reviewed 15 studies of PAHs in urban soils and found a median concentration of Σ7CarPAHs (carcinogens) of 1100 μg/kg with a range of 60–5800 μg/kg, suggesting that the Pearl River Delta soils are genuinely less contaminated with PAHs than elsewhere (especially since Σ7CarPAHs omits 9 of the US EPA priority PAHs and so underestimates Σ16PAHs). Nevertheless, Ke et al. (2017) calculated that the PAH concentrations in urban parks in Guangzhou represented a ‘potentially high’ cancer risk for people using the parks, based on established exposure pathways and likely dosage for adults and children.
PAHs in street dust . Street dusts can contribute PAHs to air, water, and soils in urban areas and have been suggested to do so in the Pearl River Delta region (Mai et al. 2003; Liang et al. 2008; Wu et al. 2014). These dusts are not PAH sources in themselves but are a transitional reservoir of PAHs and other contaminants derived from traffic (e.g. liquid fossil fuel combustion, direct petroleum emissions) and other sources. The concentrations of PAHs are commonly greater in street dusts than in soils; Menzie et al. (1992) gave a range of measured concentration of Σ7CarPAHs in street dusts between 8000 and 336,000 μg/kg and suggested that very high ΣPAHs measured in the literature for urban soils probably include a contribution from street dusts. Apart from emissions or incomplete combustion of vehicle fuels and oils, PAHS may also be derived from asphalt-based road materials (Abdel-Shafy and Mansour 2016).
7.5.4 Fractionation of Individual PAHs
Fractionation refers to changes in relative amounts of related substances, such as individual PAHs as a result of environmental processes. The concepts of fractionation are very well developed in inorganic geochemistry, with considerable focus on fractionation of isotopes of various elements, the rare-earth elements, and many others. The concentrations of different PAH compounds in urban soils will differ, since different PAHs will be released into receiving environments in various amounts depending on the nature of the source and physical and chemical properties of the PAHs themselves. For example, dominance of PAHs with two or three rings may suggest background additions (Li et al. 2007; Xiao et al. 2014) or more recent additions from a contaminant source (Cai et al. 2007). The tendency of lower molecular weight PAHs to be present at greater concentrations, when contamination is recent, probably reflects the greater volatilisation and water solubility (lower Kd values) of these compounds relative to the larger PAH molecules. Low molecular weight PAHs also have a greater tendency to be chemically reduced or oxidised (Abdel-Shafy and Mansour (2016), and see Sect. 7.5.7 for more information on factors affecting PAH fluxes). The smaller PAH molecules would therefore tend to persist for shorter times in soils, before being lost relative to heavier PAHs as the contamination ages. Biodegradation of PAHs is also more rapid for compounds having lower molecular weight; for example, Zhu et al. (2019) found the degradation rate of phenanthrene (3 rings, MW = 178 g/mole) to be much greater than for benzo[a]pyrene (5 rings, MW = 252 g/mole). The same factors lead to greater concentrations of 2–3-ring PAHs in water and in plant tissue following uptake from PAH-contaminated soil. You (2015) found that 2–3-ring PAHS were dominant in water and that in vegetable tissue 2–4-ring PAHs were most common. In relation to fractionation of PAHs by plants, Zhang et al. (2008) found that PAH uptake by vegetables from (peri)urban soils was greater for PAHs with lower KOC values, with 4-ring PAHs being most common in soils but 2–3-ring PAHs taken up by vegetables in greatest amounts. The relative amounts of PAH compounds also reflect the source of contamination . At an e-waste recycling/disposal facility in Guiyu, Leung et al. (2006) found mainly 2–3-ring PAHs associated with a printer roller dump, but where wastes had been burned, 3–5-ring PAHs were the most common. In general, combustion yields PAHs with greater molecular weights than other sources (Chen et al. 2005). Most commonly, however, additional information is necessary to identify the probable source of PAH pollution, and the next section discusses how we can use PAH data to obtain information about their sources.
7.5.5 Sources of PAH Pollution in the Pearl River Delta
The sources of PAH pollution in soil can be assessed to some extent by analysing the relative concentrations of PAHs of different ring number or molecular weight. More commonly, however, additional information from the molar ratios of PAH isomers is used, where isomers are one or more compounds which share the same chemical formula (and therefore the same molecular weight) but have a different chemical structure (see Table 7.6 for molecular weights and Fig. 7.7 for chemical structures of PAHs ). The sets of isomers commonly used are anthracene-phenanthrene (ANT-PHE, MW 178), benzo[a]anthracene-chrysene (BaA-CHR, MW 228), benzo[b]fluoranthene-benzo[k]fluoranthene-benzo[a]pyrene (BbF-BkF-BaP, MW 252), and benzo[ghi]perylene-indeno[1,2,3-cd]pyrene (BghiP-IcdP, MW 278). For example, Cao et al. (2010) interpreted the ratios listed in Table 7.7 to infer a combustion source for PAH contamination of soil in Shenzhen. We should note that isomeric ratios of PAHs should always be interpreted with caution, since even PAHs which are members of the same isomer set can have different mobilities, transformation rates, or degradation rates in various environmental compartments (Zhang et al. 2005).
In most studies, two or more PAH isomeric ratios are plotted in bivariate ‘cross plots’, so that more than one ratio informs the identification of likely PAH source(s). Figure 7.9 presents an example of the plots from Ma et al. (2008), who used the PAH isomeric ratios to deduce that the PAHs in the Dongguan, Guangzhou, and Foshan areas of the PRD had multiple sources. These sources were mainly combustion of solid fuels (coal, plant matter), with fewer samples suggesting that PAHs were derived from liquid fuel combustion or direct pollution by petroleum (Fig. 7.9). This interpretation was supported by relative concentrations of phenanthrene (3-ring) and other 4- or 5-ring PAHs accounting for, on average, 68% of PAHs by mass, also suggesting combustion sources.
Graphical analysis of PAH isomeric ratios using ‘cross plots’, implying a dominance of combustion sources for PAHs in urban soils in Dongguan and Foshan. (From Ma et al. 2008; used with permission from Springer)
In a variation on the analysis of PAH isomer ratios, source information can also be obtained from the concentrations of substituted PAHs , in which a hydrogen bonded to the PAH carbon framework is replaced by another atom or functional group (such as a methyl group, –CH3, or a hydroxyl group –OH). The ratio of methyl-phenanthrene/phenanthrene, for example, was used to infer a mainly petroleum-based origin for PAHs from vegetable-growing soils in urban Guangzhou (Chen et al. 2005). In a somewhat different context, Gao et al. (2019) concluded that the spatially different ratios between individual PAHs and their hydroxyl-substituted counterparts represented different degrees of microbial transformation depending on land use.
Multivariate statistical analyses (see Chap. 3) have also been used to identify the potential sources of PAHs in contaminated urban soils of the PRD. For example, Ke et al. (2017) used principal component analysis (PCA) in combination with multiple regression to assess the contributions of traffic emissions and coal combustion to PAH contamination in urban parklands in Guangzhou. Ma et al. (2009) also analysed data for PAHs in soils from Huizhou using principal component analysis, in combination with hierarchical cluster analysis (another multivariate statistical method). The cluster analysis broadly grouped PAHs by ring number, which Ma et al. (2009) attributed to the trends in parameters affecting environmental behaviour of PAHs having different molecular weights (e.g. see Table 7.6 and Fig. 7.13).
7.5.6 Controls on PAH Concentrations and Fractionation
Soil organic carbon and ‘black carbon ’. As we discussed in Sect. 7.2, soil organic carbon content strongly affects the adsorption of non-polar (or low-polarity) organic compounds such as PAHs . In many examples of PAH contamination of urban soils in the Pearl River Delta, a positive relationship between PAH concentration and soil organic carbon content is observed (Li et al. 2008b; Ma et al. 2008; Zhu et al. 2019). Similarly, there can also be a positive relationship between PAH concentration and black carbon content of soil, where black carbon is composed of partially combusted solid materials such as soot or finely divided charcoal (Ma et al. 2008). Figure 7.10 shows examples of relationships of ∑16PAHs concentration with both total soil organic carbon and black carbon, in soils of the Pearl River Delta, which suggest that PAHs in these soils are adsorbed to one or both forms of organic carbon. (It is worth noting here that not all methods of measuring soil organic carbon measure total carbon. For example, the widely used methods employing wet oxidation of soil organic matter with acid dichromate solution are known to exclude black carbon, so do not measure a true ‘total’ organic carbon content (TOC).)
Relationships between ∑16PAHs concentration and both total soil organic carbon and black carbon, in urban and peri-urban soils in Guangzhou, Dongguan, and Foshan (Pearl River Delta, Guangdong, China). (Redrawn from Ma et al. (2008); used with permission from Springer)
In some urban environments, PAH concentrations are not (or poorly) correlated with either soil organic carbon or black carbon. The lack of a strong correlation between ∑16PAHs concentration and soil organic/black carbon is usually attributed to recent additions of PAHs or proximity to ongoing PAH sources, so that PAHs are not in equilibrium with the soil environment (Cai et al. 2007; Ke et al. 2017).
The microbial community composition of urban soil, although not strictly an effect of soil organic matter (which is usually defined as the non-living organic material), may also affect the concentrations of fractionation of PAHs . For example, Zhu et al. (2019) found shifts in bacterial community structure when exposed to phenanthrene and benzo[a]pyrene, suggesting that some groups of soil bacteria may be able to degrade PAHs more effectively than others.
Urban spatial and land use effects. The concentrations of PAHs in urban soils should reflect spatial factors such as distance from point sources or from the urban centre, since PAH concentrations into soils should be greater where industrial or traffic emissions are greater. For similar reasons, land use should also have an effect on PAH concentrations in urban soils. Unless there have been significant changes in land use, we would expect lower PAH concentrations in less urbanised environments, such as residential suburbs or peri-urban agriculture, than in industrial areas or in the urban centre.
In the Pearl River Delta, PAH concentrations in urban and peri-urban soils seem to follow these expected trends. For example, Liu et al. (2011), Xiao et al. (2014), and You (2015) all found that PAH concentrations decreased with increasing distance from urban centres across several cities in the PRD (Fig. 7.11). The greatest ∑PAH concentrations observed in Shenzhen (Zhang et al. 2014) in traffic-affected soils are consistent with the greater expected concentrations of PAHs in street dusts (Menzie et al. 1992). Low-intensity land uses such as gardens, urban woodland, and urban green space tend to have the lowest PAH concentrations (Fig. 7.11a).
(a) Decreasing trends in ∑PAH concentrations with decreasing intensity of urban land use, redrawn and translated from Zhang et al. (2014); (b) trends in the proportions of ∑PAHs by ring number, on an urban-rural gradient. (Redrawn from Xiao et al. (2014) and used under the terms of a CC-BY-3.0 license (http://creativecommons.org/licenses/by/3.0/))
When individual PAHs , or smaller categories based on ring number, are considered, the trends do not necessarily match those of the ∑PAHs . As the data from Xiao et al. (2014) show (Fig. 7.11b), the proportions of 2-ring and 3-ring PAHs (e.g. [2-ring PAHs ] ∕ [∑16PAHs]) are both greater in soil in rural land than in urban land, tending to increase from urban, to suburban, to rural. In contrast, the proportions of 4-, 5-, and 6-ring PAHs follow the same trend as ∑16PAHs in soil and decrease from urban, to suburban, to rural land. The combination of low ∑16PAHs and greater proportions of 2- and 3-ring PAHs in rural soils may represent small ongoing background additions from other environmental compartments such as wet or dry deposition from air.
Uptake of PAHs from soil by plants. The concentrations of PAHs in plants are generally lower than in the soils they are grown in. Song et al. (2013) determined mean ∑16PAHs of 318 μg/kg in soil, which was approximately 1.4 times greater than the 234 μg/kg in vegetables. Similarly, Zhang et al. (2008) measured approximately 1.3-fold greater concentrations of ∑16PAHs in soils than in vegetables. In contrast, You (2015) found greater ∑16PAH concentrations in vegetable matter (Fig. 7.12), with mean ∑16PAH concentrations across 5–6 sites of 232 μg/kg in soils and 641 μg/kg in vegetables.
In addition, plant uptake is not the same for all PAH compounds, resulting in fractionation during the transfer between soil and plant. For example, Song et al. (2013) showed that vegetable tissues were depleted in 5- and 6-ring PAHs relative to soils (Fig. 7.12). The differences in uptake for different PAHs are consistent with their water solubilities. The PAH compounds with greater molecular weights (i.e. greater ring numbers) have lower water solubilities and larger KOW values, meaning that they are more strongly associated with soil solids (mainly soil organic carbon), a situation which restricts plant uptake.
Within plant biomass, PAHs partition differently between above-ground (shoots) and below-ground (roots) tissues. Transfer within plants is in water-based media (the phloem and xylem), so, being non-polar compounds, PAHs would be expected to have limited mobility in plants. Individual PAHs with greater water solubility (lower KOW) are more likely to move from soil to roots to shoots. For a range of plant species grown at e-waste recycling sites in Longtang, Guangdong, Wang et al. (2017) measured mean ∑16PAH concentrations in shoots of 196 μg/kg compared with 284 μg/kg in roots. In addition, there was some evidence of differential accumulation of individual PAHs in root and shoot tissues of plants.
7.5.7 Fluxes of PAHs to and from Soils in the Pearl River Delta
Emissions of PAHs from industry and other sources are mainly into the atmosphere from combustion processes, so that atmospheric deposition represents the dominant input flux into environmental compartments such as soils (Abdel-Shafy and Mansour 2016). The soil budget for PAHs in the Pearl River Delta region is not known completely, but the amounts are large; Ma et al. (2008) calculated values for Guangzhou of 920,000 kg, for Dongguan of 143,000 kg, and for Foshan of 229,000 kg. In Shenzhen, Ni et al. (2011) calculated the soil ∑15PAH inventory to be 152,000 kg. With such large reservoirs of PAHs in urban and peri-urban soils of the PRD, the transfers to and from the soil environment are therefore very significant.
Atmosphere to soil fluxes of PAHs . The inputs of PAHs across the entire Pearl River Delta region are substantial (Table 7.8); for example, Li et al. (2010) sampled sites across the PRD including urban and rural locations and calculated that particle-phase deposition of ∑15PAHs was 2950 kg/y (excluding Hong Kong), with ∑15PAHs deposition of 86 kg/y in Hong Kong (both fluxes are possibly underestimates, given that only particle-phase deposition was measured). Including Hong Kong in the regional estimate, this represents 0.054 kg/km2/y over the 54,156 km2 of the PRD, which includes both urban and non-urban land. In Shenzhen, Ni et al. (2011) calculated that total atmospheric fluxes (wet and dry deposition) accounted for inputs of 1156 kg/y over the 2050 km2 area of Shenzhen (i.e. 0.56 kg/km2/y). The differences are likely to be due to more comprehensive measurement in Shenzhen, which included ‘rain washing’, wet deposition, and dry deposition; the rain washing flux was dominant, being about 98% of total atmosphere to soil transfer (Ni et al. 2011). In addition, the proportion of non-urban land in Shenzhen may be greater than in the PRD region as a whole. PAH fluxes to soil via precipitation were also important in a peri-urban forest in Guangzhou (Chen et al. 2015a); importantly, interception of precipitation by tree canopies decreased PAH inputs to soil by nearly 25% relative to the original precipitation.
Soil to atmosphere fluxes of PAHs . Despite the large atmospheric deposition of PAHs to soil, Ni et al. (2011) found that diffusion of PAHs from soil into air was also an important pathway, accounting for over 10,000 kg/y of ∑15PAHs over the 2050 km2 area of Shenzhen (i.e. approximately 4.9 kg/km2/y). As Fig. 7.13 shows, the net flux may be positive or negative for individual PAHs (Wei et al. 2014). Lower molecular weight PAH molecules such as acenaphthene or fluorene are more volatile and have less affinity for soil organic matter so have positive soil to air fluxes. In contrast, high molecular weight PAHs such as benzo[a]pyrene or indeno[1,2,3-cd]pyrene have net air-soil fluxes (Fig. 7.13).
Relationship of soil-atmosphere flux for priority pollutant PAHs to their molecular mass (redrawn by Andrew W. Rate using data for central Pearl River Delta soils from Wei et al. 2014). Abbreviations for individual PAHs are from Table 7.6 and are followed by the number of rings in parentheses; the trend ellipse is just a visual guide. A similar plot is obtained for soil-air flux vs. log10KOW (octanol-water partition coefficient). Larger PAH molecules are less volatile and have greater affinity for soil organic matter so have small or negative soil to air fluxes
Other soil fluxes of PAHs : run-off, leaching, and erosion. In their Guangzhou peri-urban forest study, Chen et al. (2015a) calculated a loss of ∑9PAHs in run-off of 0.034 kg/km2/y. In the same forest environment, leaching of ∑9PAHs through soil transferred 0.065 kg/km2/y to a depth of 30 cm and 0.008 kg/km2/y to 60 cm. The decrease in PAHs during leaching through soil was attributed to a combination of processes including biodegradation and adsorption (and presumably also volatilisation from surface soil, although the authors did not consider this). A component of the run-off flux was soil erosion, which also removed PAHs from the soil environment. Run-off was separated into water and particulate (i.e. erosion) components by Ni et al. (2011), who estimated a water (dissolved) run-off flux of ∑15PAHs of 0.94 kg/km2/y and soil erosion (particulate run-off) flux of 0.32 kg/km2/y.
7.5.8 Other Issues for PAHs in the Pearl River Delta
The PAHs released by combustion, petroleum, and other sources into urban soils may react to give new compounds which are not measured in routine PAH analyses. For example, small but significant concentrations of several hydroxylated PAHs were measured in 15 cities across the Pearl River Delta region by Gao et al. (2019). Ni and Zeng (2012) found non-negligible concentrations of a range of chlorinated and brominated PAHs in urban soils in Shenzhen. For hydroxylated, chlorinated, and brominated PAHs , the absence of correlations between concentrations of substituted PAHs and their probable parent compounds inferred that they were formed by reactions in the soil environment. The toxicity profiles of these compounds are not currently well understood, but both studies recommended that the potential risks to human health be considered.
The occurrence and behaviour of polycyclic aromatic hydrocarbons in China’s Pearl River Delta is a globally relevant case study. Based on the material covered in this chapter, Fig. 7.14 presents a generalised conceptual model for cycling of organic pollutants in urban environments.
7.6 Summary
-
A wide range of organic compounds are potential contaminants in urban soils and sediments, with these compounds having widely varying structures and properties. Soils are one of the largest reservoirs of organic pollutants.
-
Organic compounds can be highly toxic to a wide range of organisms, including humans.
-
The behaviour of organic contaminants in soils is affected by the properties of the individual compounds, or categories of compounds, involved, including: polarity, volatility, density, ionisation, substitution with atoms other than C and H, and molecular weight.
-
The natural organic carbon content of soils (including black carbon) is very important for the retention of organic contaminants by adsorption and cation exchange.
-
Organic contaminants can degrade chemically in soils (as opposed to biologically mediated degradation), but the most effective and common form of degradation is mediated by soil microorganisms such as bacteria and fungi.
-
Using the behaviour of polycyclic aromatic hydrocarbons in soils of the large urban agglomeration of the Pearl River delta as a case study, it is apparent that:
-
Categories of closely related organic contaminants such as PAHs can have systematic differences in properties which lead to fractionation in soils. This fractionation can provide information about the sources of contamination .
-
Transfers of organic contaminants such as PAHs between urban soils and the atmosphere can be particularly important, as can transfers between soils and other environmental compartments.
-
7.7 Further Reading
-
Albanese S, Cicchella D (2012) Legacy problems in urban geochemistry. Elements 8:423–428. https://doi.org/10.2113/gselements.8.6.423
-
Kah M, Brown C (2006) Adsorption of ionisable pesticides in soils. Rev Environ Contam Toxicol 188:149–217. https://doi.org/10.1007/978-0-387-32,964-2_5
-
Berkowitz B, Dror I, Yaron B (2008). Contaminant geochemistry: interactions and transport in the subsurface environment. Springer, Berlin
-
Rodríguez-Eugenio N, McLaughlin M, Pennock D (2018) Soil pollution – a hidden reality. Food and Agriculture Organization of the United Nations, Rome
7.8 Questions
7.8.1 Checking Your Understanding
-
1.
Give as many examples as you can of non- or weakly polar organic pollutants.
-
2.
Which organic pollutants could be derived from use of solvents?
-
3.
Explain the acronyms LNAPL and DNAPL and how they relate to soil and groundwater contamination .
-
4.
What are the properties of persistent organic pollutants that distinguish them from other organic compounds?
-
5.
Why are KOW values of organic compounds useful – what do they represent, and what can they predict?
-
6.
What are the two main steps in analysis of organic compounds in soils, and what is involved in each step?
7.8.2 Thinking About the Topics More Deeply
-
7.
Explain how fractionation of polycyclic aromatic hydrocarbons occurs and what information that fractionation can provide.
-
8.
Why is biodegradation of organic pollutants usually more significant for attenuation than abiotic degradation?
-
9.
The intensity of soil pollution with organic contaminants is often related to the intensity of land use, especially along a rural-urban gradient, with land in industrial or urban-core zones being the most contaminated. Can you think of any scenarios where this may NOT be the case?
7.8.3 A Question ‘Out of Left Field’
-
10.
Different countries have different limits for acceptable concentrations of organic contaminants in soils (even for the same organic compound). Do you think that there is a scientific reason for this or do other factors contribute to the diversity in contaminant limits?
References
Abdel-Shafy HI, Mansour MSM (2016) A review on polycyclic aromatic hydrocarbons: source, environmental impact, effect on human health and remediation. Egypt J Pet 25:107–123. https://doi.org/10.1016/j.ejpe.2015.03.011
Achten C, Andersson JT (2015a) Overview of polycyclic aromatic compounds (PAC). Polycycl Aromat Compd 35:177–186. https://doi.org/10.1080/10406638.2014.994071
Achten C, Andersson JT (2015b) Overview of polycyclic aromatic compounds (PAC) [supplementary material]. Polycycl Aromat Compd 35:177–186. https://doi.org/10.1080/10406638.2014.994071
Ahammed GJ, Wang MM, Zhou YH, Xia XJ, Mao WH, Shi K, Yu JQ (2012) The growth, photosynthesis and antioxidant defense responses of five vegetable crops to phenanthrene stress. Ecotoxicol Environ Saf 80:132–139. https://doi.org/10.1016/j.ecoenv.2012.02.015
Báez ME, Espinoza J, Silva R, Fuentes E (2015) Sorption-desorption behavior of pesticides and their degradation products in volcanic and nonvolcanic soils: interpretation of interactions through two-way principal component analysis. Environ Sci Pollut Res 22:8576–8585. https://doi.org/10.1007/s11356-014-4036-8
Barceló D (ed) (2012) Emerging organic contaminants and human health, The handbook of environmental chemistry, vol 20. Springer, Heidelberg, 466 pp
Barrett KA, McBride MB (2005) Oxidative degradation of glyphosate and aminomethylphosphonate by manganese oxide. Environ Sci Technol 39:9223–9228. https://doi.org/10.1021/es051342d
Biasioli M, Fabietti G, Barberis R, Ajmone-Marsan F (2012) An appraisal of soil diffuse contamination in an industrial district in northern Italy. Chemosphere 88:1241–1249. https://doi.org/10.1016/j.chemosphere.2012.03.081
Bromilow RH (2005) Pesticides. In: Hillel D (ed) Encyclopedia of Soils in the environment. Elsevier, Oxford, pp 188–195. https://doi.org/10.1016/B0-12-348530-4/00110-7
Cai QY, Mo CH, Li YH, Zeng QY, Katsoyiannis A, Wu QT, Férard JF (2007) Occurrence and assessment of polycyclic aromatic hydrocarbons in soils from vegetable fields of the Pearl River Delta, South China. Chemosphere 68:159–168. https://doi.org/10.1016/j.chemosphere.2006.12.015
Canadian Council of Ministers of the Environment (1991) Interim Canadian environmental quality criteria for contaminated sites. Canadian Council of Ministers of the Environment, Winnipeg
Canadian Council of Ministers of the Environment (2010) Canadian soil quality guidelines for the protection of environmental and human health – polycyclic aromatic hydrocarbons. Canadian Council of Ministers of the Environment, Winnipeg
Cao SP, Ni HG, Qin PH, Zeng H (2010) Occurrence and human non-dietary exposure of polycyclic aromatic hydrocarbons in soils from Shenzhen, China. J Environ Monit 12:1445–1450. https://doi.org/10.1039/c000549e
Chen L, Ran Y, Xing B, Mai B, He J, Wei X et al (2005) Contents and sources of polycyclic aromatic hydrocarbons and organochlorine pesticides in vegetable soils of Guangzhou, China. Chemosphere 60:879–890. https://doi.org/10.1016/j.chemosphere.2005.01.011
Chen B, Pei N, Huang J, Liu S, Zhang N, Xiao Y, Pan Y (2015a) Removal of polycyclic aromatic hydrocarbons from precipitation in an urban forest of Guangzhou, South China. Bull Environ Contam Toxicol 95:240–245. https://doi.org/10.1007/s00128-015-1567-8
Chen M, Xu P, Zeng G, Yang C, Huang D, Zhang J (2015b) Bioremediation of soils contaminated with polycyclic aromatic hydrocarbons, petroleum, pesticides, chlorophenols and heavy metals by composting: applications, microbes and future research needs. Biotechnol Adv 33:745–755. https://doi.org/10.1016/j.biotechadv.2015.05.003
CL:AIRE (2009) Soil guideline values – soil guideline value reports. Contaminated Land: Applications in Real Environments, Haddenham. https://www.claire.co.uk/information-centre/water-and-land-library-wall/44-risk-assessment/178-soil-guideline-values. Accessed 27 Aug 2020
Enfield CG, Bengtsson G, Lindqvist R (1989) Influence of macromolecules on chemical transport. Environ Sci Technol 23:1278–1286
Fuoco R, Capodaglio G, Muscatello B, Radaelli M (2009) Persistent organic pollutants (POPs) in the Antarctic environment: a review of findings. Scientific Committee on Antarctic Research (SCAR), Cambridge
Gao CJ, Xia LL, Wu CC, Shen HM, Guo Y (2019) Hydroxylated polycyclic aromatic hydrocarbons in surface soil in an emerging urban conurbation in South China. Sci Total Environ 692:1250–1256. https://doi.org/10.1016/j.scitotenv.2019.07.334
Gonzalez-Mille DJ, Espinosa-Reyes G, Rivero-Pérez NE, Trejo-Acevedo A, Nava-Montes AD, Ilizaliturri-Hernández CA (2013) Persistent organochlorine pollutants (POPs) and DNA damage in Giant Toads (Rhinella marina) from an industrial area at Coatzacoalcos, Mexico. Water Air Soil Pollut 224:1781. https://doi.org/10.1007/s11270-013-1781-0
Grathwohl P (2011) Fate and transport of organic compounds in (to) the subsurface environment. In: Xing B, Senesi N, Huang PM (eds) Biophysico-chemical processes of anthropogenic organic compounds in environmental systems, Wiley online books. Wiley, Hoboken, pp 215–231. https://doi.org/10.1002/9780470944479.ch8
Harvey DT (2016) Analytical chemistry 2.1 (Open access; CC-BY-VC-SA 4.0). http://dpuadweb.depauw.edu/harvey_web/eTextProject/version_2.1.html. Accessed 18 June 2021
Hatzinger PB, Alexander M (1995) Effect of aging of chemicals in soil on their biodegradability and extractability. Environ Sci Technol 29:537–545. https://doi.org/10.1021/es00002a033
He L, Song J, Peng P (2008) Characterization of extractable and non-extractable polycyclic aromatic hydrocarbons in soils and sediments from the Pearl River Delta, China. Environ Pollut 156:769–774. https://doi.org/10.1016/j.envpol.2008.06.003
Hiller E, Krascsenits Z, Čerňanský S (2008) Sorption of acetochlor, atrazine, 2,4-D, chlorotoluron, MCPA, and trifluralin in six soils From Slovakia. Bull Environ Contam Toxicol 80:412–416. https://doi.org/10.1007/s00128-008-9430-9
Joa K, Jefimova J, Irha N, Teinemaa E, Lintelmann J, Kirso U (2009) Determination of polycyclic aromatic hydrocarbons (PAHs) in oil shale processing wastes: current practice and new trends. Oil Shale 26. https://doi.org/10.3176/oil.2009.1.07
John EM, Shaike JM (2015) Chlorpyrifos: pollution and remediation. Environ Chem Lett 13:269–291. https://doi.org/10.1007/s10311-015-0513-7
Johnson D (2005) Pollutants | persistent organic (POPs). In: Hillel D (ed) Encyclopedia of soils in the environment. Elsevier, Oxford, pp 264–271. https://doi.org/10.1016/B0-12-348530-4/00560-9
Jury WA, Focht DD, Farmer WJ (1987) Evaluation of pesticides groundwater pollution potential from standard indices of soil-chemical adsorption and biodegradation. J Environ Qual 16:422–428
Kah M, Brown C (2006) Adsorption of ionisable pesticides in soils. Rev Environ Contam Toxicol 188:149–217. https://doi.org/10.1007/978-0-387-32964-2_5
Ke CL, Gu YG, Liu Q (2017) Polycyclic aromatic hydrocarbons (PAHs) in exposed-lawn soils from 28 urban parks in the megacity Guangzhou: occurrence, sources, and human health implications. Arch Environ Contam Toxicol 72:496–504. https://doi.org/10.1007/s00244-017-0397-6
Kile DE, Chiou CT, Zhou H, Li H, Xu O (1995) Partition of nonpolar organic pollutants from water to soil and sediment organic matters. Environ Sci Technol 29:1401–1406. https://doi.org/10.1021/es00005a037
Kostecki P, Morrison R, Dragun J (2005) Hydrocarbons. In: Hillel D (ed) Encyclopedia of soils in the environment. Elsevier, Oxford, pp 217–226. https://doi.org/10.1016/B0-12-348530-4/00098-9
Kromer T, Ophoff H, Stork A, Führ F (2004) Photodegradation and volatility of pesticides: chamber experiments. Environ Sci Pollut Res 11:107–120. https://doi.org/10.1007/BF02979710
Leung A, Cai ZW, Wong MH (2006) Environmental contamination from electronic waste recycling at Guiyu, southeast China. J Mater Cycles Waste Manage 8:21–33. https://doi.org/10.1007/s10163-005-0141-6
Li H, Ran Y (2012) Distribution and bioconcentration of polycyclic aromatic hydrocarbons in surface water and fishes. Sci World J 2012:Article 632910. https://doi.org/10.1100/2012/632910
Li Y, Li F, Zhang T, Yang G, Chen J, Wan H (2007) Pollution assessment, distribution and sources of PAHs in agricultural soils of Pearl River Delta – the biggest manufacturing Base in China. J Environ Sci Health A Toxic Hazard Subst Environ Eng 42:1979–1987. https://doi.org/10.1080/10934520701628890
Li FB, Wang XG, Li YT, Liu CS, Zeng F, Zhang LJ et al (2008a) Enhancement of the reductive transformation of pentachlorophenol by polycarboxylic acids at the iron oxide-water interface. J Colloid Interface Sci 321:332–341. https://doi.org/10.1016/j.jcis.2008.02.033
Li YT, Li FB, Chen JJ, Yang GY, Wan HF, Zhang TB et al (2008b) The concentrations, distribution and sources of PAHs in agricultural soils and vegetables from Shunde, Guangdong, China. Environ Monit Assess 139:61–76. https://doi.org/10.1007/s10661-007-9816-x
Li J, Liu X, Zhang G, Li XD (2010) Particle deposition fluxes of BDE-209, PAHs, DDTs and chlordane in the Pearl River Delta, South China. Sci Total Environ 408:3664–3670. https://doi.org/10.1016/j.scitotenv.2010.04.048
Liang Y, Fung PK, Tse MF, Hong HC, Wong MH (2008) Sources and seasonal variation of PAHs in the sediments of drinking water reservoirs in Hong Kong and the Dongjiang River (China). Environ Monit Assess 146:41–50. https://doi.org/10.1007/s10661-007-0058-8
Liang Y, Zhou Z, Li X (2019) Dynamic of regional planning and sustainable development in the Pearl River Delta, China. Sustainability 11:6074. https://doi.org/10.3390/su11216074
Liu G, Yu L, Li J, Liu X, Zhang G (2011) PAHs in soils and estimated air-soil exchange in the Pearl River Delta, South China. Environ Monit Assess 173:861–870. https://doi.org/10.1007/s10661-010-1429-0
Ma X, Ran Y, Gong J, Zou M (2008) Concentrations and inventories of polycyclic aromatic hydrocarbons and organochlorine pesticides in watershed soils in the Pearl River Delta, China. Environ Monit Assess 145:453–464. https://doi.org/10.1007/s10661-007-0054-z
Ma J, Zhou Y, Wan H (2009) Study on polycyclic aromatic hydrocarbons (PAHs) contents and sources in the surface soil of Huizhou City, South China, based on multivariate statistics analysis. Chin J Geochem 28:335–339. https://doi.org/10.1007/s11631-009-0335-z
Ma X, Ran Y, Gong J, Chen D (2010) Sequential accelerated solvent extraction of polycyclic aromatic hydrocarbons with different solvents: performance and implication. J Environ Qual 39:2072–2079. https://doi.org/10.2134/jeq2010.0163
Mai B, Qi S, Zeng EY, Yang Q, Zhang G, Fu J et al (2003) Distribution of polycyclic aromatic hydrocarbons in the coastal region off Macao, China: assessment of input sources and transport pathways using compositional analysis. Environ Sci Technol 37:4855–4863. https://doi.org/10.1021/es034514k
Martí V, Jubany I, Pérez C, Rubio X, De Pablo J, Giménez J (2014) Human health risk assessment of a landfill based on volatile organic compounds emission, immission and soil gas concentration measurements. Appl Geochem 49:218–224. https://doi.org/10.1016/j.apgeochem.2014.06.018
McBride MB (1994) Environmental chemistry of soils. Oxford University Press, New York
Menzie CA, Potocki BB, Santodonato J (1992) Exposure to carcinogenic PAHs in the environment. Environ Sci Technol 26:1278–1284. https://doi.org/10.1021/es00031a002
Ministry for the Environment (Manatū Mō Te Taiao) (2012) Users’ guide: national environmental standard for assessing and managing contaminants in soil to protect human health. New Zealand Government, Wellington
Mo CH, Cai QY, Tang SR, Zeng QY, Wu QT (2009) Polycyclic aromatic hydrocarbons and phthalic acid esters in vegetables from nine farms of the pearl river delta, South China. Arch Environ Contam Toxicol 56:181–189. https://doi.org/10.1007/s00244-008-9177-7
Mulligan CN, Yong RN (2004) Natural attenuation of contaminated soils. Environ Int 30:587–601. https://doi.org/10.1016/j.envint.2003.11.001
Mulligan CN, Yong RN, Gibbs BF (2001) Surfactant-enhanced remediation of contaminated soil: a review. Eng Geol 60:371–380. https://doi.org/10.1016/S0013-7952(00)00117-4
National Environment Protection Council (2013a) Schedule B5b: guideline on methodology to derive ecological investigation levels in contaminated soils, national environment protection (assessment of site contamination) measure (amended). Commonwealth of Australia, Canberra
National Environment Protection Council (2013b) Schedule B (1): guideline on the investigation levels for soil and groundwater, national environment protection (assessment of site contamination) measure (amended). Commonwealth of Australia, Canberra
National Pollutant Inventory (2020) Substances. Department of Agriculture, Water and the Environment, Canberra. http://www.npi.gov.au/substances. Accessed 17 June 2020
Ni HG, Zeng EY (2012) Environmental and human exposure to soil chlorinated and brominated polycyclic aromatic hydrocarbons in an urbanized region. Environ Toxicol Chem 31:1494–1500. https://doi.org/10.1002/etc.1854
Ni HG, Qin PH, Cao SP, Zeng H (2011) Fate estimation of polycyclic aromatic hydrocarbons in soils in a rapid urbanization region, Shenzhen of China. J Environ Monit 13:313–318. https://doi.org/10.1039/c0em00470g
Petersen LW, El-Farhan YH, Moldrup P, Rolston DE, Yamaguchi T (1996) Transient diffusion, adsorption, and emission of volatile organic vapors in soils with fluctuating low water contents. J Environ Qual 25:1054–1063. https://doi.org/10.2134/jeq1996.00472425002500050018x
Rodríguez-Eugenio N, McLaughlin M, Pennock D (2018) Soil pollution – a hidden reality. Food and Agriculture Organization of the United Nations, Rome. http://www.fao.org/3/I9183EN/i9183en.pdf
Sadler R, Connell D (2003) Analytical methods for the determination of total petroleum hydrocarbons in soil. In: Langley A, Gilbey M, Kennedy B (eds) Proceedings of the fifth national workshop on the assessment of site contamination. National Environment Protection Council Service Corporation, Adelaide. http://www.nepc.gov.au/system/files/resources/27b740ab-880f-d7f4-d10e-bab0fa964e48/files/asc-wkshoppaper-09-tphs-sadler-petrol-hydro-soil-200301.pdf
Sandrin TR, Maier RM (2003) Impact of metals on the biodegradation of organic pollutants. Environ Health Perspect 111:1093–1101. https://doi.org/10.1289/ehp.5840
Secretariat of the Stockholm Convention (2018) Stockholm convention on persistent organic pollutants: texts and annexes; revised in 2017. United Nations Environment Programme, Châtelaine
Shahsavari E, Poi G, Aburto-Medina A, Haleyur N, Ball AS (2017) Bioremediation approaches for petroleum hydrocarbon-contaminated environments. In: Anjum NA, Gill SS, Tuteja N (eds) Enhancing cleanup of environmental pollutants: volume 1: biological approaches. Springer, Cham
Song Y, Hao R, Peng S, Wan H (2013) Polycyclic aromatic hydrocarbons in different soils and vegetables from the Pearl River Delta, South China. Environ Eng Manag J 12:1891–1895
Sverdrup LE, Hagen SB, Krogh PH, van Gestel CAM (2007) Benzo(a)pyrene shows low toxicity to three species of terrestrial plants, two soil invertebrates, and soil-nitrifying bacteria. Ecotoxicol Environ Saf 66:362–368. https://doi.org/10.1016/j.ecoenv.2006.01.007
U.S. EPA (1996a) Method 3540C: soxhlet extraction, Test methods for evaluating solid waste, physical/chemical methods, EPA publication SW-846. United States Environmental Protection Agency, Washington, DC, pp 1–8. https://www.epa.gov/sites/production/files/2015-12/documents/3540c.pdf
U.S. EPA (1996b) Method 3560: supercritical fluid extraction of total recoverable petroleum hydrocarbons, Test methods for evaluating solid waste, physical/chemical methods, EPA publication SW-846. United States Environmental Protection Agency, Washington, DC, pp 1–8. https://www.epa.gov/sites/production/files/2015-12/documents/3540c.pdf
U.S. EPA (2018) Method 8000D: determinative chromatographic analysis, Test methods for evaluating solid waste, physical/chemical methods, EPA publication SW-846 Update VI. United States Environmental Protection Agency, Washington, DC. https://www.epa.gov/sites/production/files/2015-12/documents/8000d.pdf
U.S. EPA (2020) Integrated risk information system (IRIS). United States Environmental Protection Agency, Washington. https://www.epa.gov/iris. Accessed 22 July 2020
UN-Habitat (2016) World cities report 2016: urbanization and development – emerging futures. United Nations Human Settlements Programme, Nairobi. https://unhabitat.org/sites/default/files/download-manager-files/WCR-2016-WEB.pdf
Wang W-X, Rainbow PS (2020) Environmental pollution of the Pearl River Estuary, China, status and impact of contaminants in a rapidly developing region. Springer, Berlin/Heidelberg, 125 pp. https://doi.org/10.1007/978-3-662-61834-9
Wang X, Sun D, Yao T (2016) Climate change and global cycling of persistent organic pollutants: a critical review. Sci China Earth Sci 59:1899–1911. https://doi.org/10.1007/s11430-016-5073-0
Wang Y, He J, Wang S, Luo C, Yin H, Zhang G (2017) Characterisation and risk assessment of polycyclic aromatic hydrocarbons (PAHs) in soils and plants around e-waste dismantling sites in southern China. Environ Sci Pollut Res 24:22173–22182. https://doi.org/10.1007/s11356-017-9830-7
Wania F, MacKay D (1996) Tracking the distribution of persistent organic pollutants. Environ Sci Technol 30:390A–396A. https://doi.org/10.1021/es962399q
Weber R, Gaus C, Tysklind M, Johnston P, Forter M, Hollert H et al (2008) Dioxin- and POP-contaminated sites – contemporary and future relevance and challenges: overview on background, aims and scope of the series. Environ Sci Pollut Res 15:363–393. https://doi.org/10.1007/s11356-008-0024-1
Wei YL, Bao LJ, Wu CC, He ZC, Zeng EY (2014) Association of soil polycyclic aromatic hydrocarbon levels and anthropogenic impacts in a rapidly urbanizing region: spatial distribution, soil-air exchange and ecological risk. Science of the Total Environment, 473-474:676–684. https://doi.org/10.1016/j.scitotenv.2013.12.106
Wu SP, Yang BY, Wang XH, Yuan CS, Hong HS (2014) Polycyclic aromatic hydrocarbons in the atmosphere of two subtropical cities in Southeast China: seasonal variation and gas/particle partitioning. Aerosol Air Qual Res 14:1232–1246. https://doi.org/10.4209/aaqr.2013.01.0015
Wyrwicka A, Steffani S, Urbaniak M (2014) The effect of PCB-contaminated sewage sludge and sediment on metabolism of cucumber plants (Cucumis sativus L.). Ecohydrol Hydrobiol 14:75–82. https://doi.org/10.1016/j.ecohyd.2014.01.003
Xiao Y, Tong F, Kuang Y, Chen B (2014) Distribution and source apportionment of polycyclic aromatic hydrocarbons (PAHs) in forest soils from urban to rural areas in the Pearl River Delta of southern China. Int J Environ Res Public Health 11:2642–2656. https://doi.org/10.3390/ijerph110302642
Yong RN, Mulligan CN (2003) Natural attenuation of contaminants in soils. CRC Press, Baton Rouge
You Y (2015) Concentrations and distributions of organic pollutants in typical vegetable fields in Guangzhou, China. Jilin Daxue Xuebao (Diqiu Kexue Ban)/Journal of Jilin University (Earth Science Edition), 45:1205–1216. https://doi.org/10.13278/j.cnki.jjuese.201504207
Zhang XL, Tao S, Liu WX, Yang Y, Zuo Q, Liu SZ (2005) Source diagnostics of polycyclic aromatic hydrocarbons based on species ratios: a multimedia approach. Environ Sci Technol 39:9109–9114. https://doi.org/10.1021/es0513741
Zhang T, Wan H, Yang G, Gao Y, Luo W (2008) Distribution of polycyclic aromatic hydrocarbons in agricultural soil and vegetables of Foshan city in the Pearl River Delta. Huanjing Kexue Xuebao/Acta Sci Circumst 28:2375–2384
Zhang D, Cao SP, Sun JL, Zeng H (2014) Occurrence and spatial differentiation of polycyclic aromatic hydrocarbons in surface soils from Shenzhen, China. Huanjing Kexue/Enviro Sci 35:711–718
Zhang D, Wang J, Zeng H (2016) Soil polycyclic aromatic hydrocarbons across urban density zones in Shenzhen, China: occurrences, source apportionments, and spatial risk assessment. Pedosphere 26:676–686. https://doi.org/10.1016/S1002-0160(15)60076-5
Zhong Y, Overcash MR, McPeters AL (1994) Photodegradation of TCDD in soil containing organic solvents. Environ Geochem Health 16:235–240. https://doi.org/10.1007/BF01747921
Zhu Q, Wu Y, Zeng J, Wang X, Zhang T, Lin X (2019) Influence of bacterial community composition and soil factors on the fate of phenanthrene and benzo[a]pyrene in three contrasting farmland soils. Environ Pollut 247:229–237. https://doi.org/10.1016/j.envpol.2018.12.079
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Rate, A.W. (2022). Organic Contaminants in Urban Soils. In: Rate, A.W. (eds) Urban Soils. Progress in Soil Science. Springer, Cham. https://doi.org/10.1007/978-3-030-87316-5_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-87316-5_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-87315-8
Online ISBN: 978-3-030-87316-5
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)