Summary
Introduction: Neuromonitoring analysis for intracerebral hemorrhage (ICH) is still rare, especially regarding vascular reactivity patterns. Our goal was to analyze neuromonitoring data and 28-day mortality for ICH patients.
Methods: Neuromonitoring records were retrospectively reviewed from a cohort of ICH patients admitted to a neurocritical care unit between 2013 and 2016. Variables considered were intracranial pressure (ICP), cerebral perfusion pressure (CPP), optimal CPP, and pressure reactivity index (PRx), as well as ICP dose, PRx dose, and time percentage above critical value (T%abv). Information regarding demographics, surgical drainage, external ventricular drain placement, and 28-day mortality was recorded. Statistical analysis was performed using the t-test and Kaplan-Meier curves.
Results: Forty-six patients were analyzed, with a mean of 263 ± 173 h of signal records and a median length of stay in the intensive care unit of 22 (interquartile range of 13) days. The mean age was 62.6 ± 11.8 years old, and 24 (52%) of the patients were male. Patients who died within 28 day (37.0%) had significantly higher mean ICP, PRx, ICP dose, PRx dose, and T%abv. Although their mean ICP was under 20 mmHg, they presented PRx > 0.25, indicating impaired cerebrovascular reactivity (0.30 ± 0.26). Also, patients with PRx > 0.25 had a lower survival rate, with a proportion of 14% at 28 days, as opposed to 85% of those with PRx < 0.25 (p < 0.001).
Conclusion: The data suggest that autoregulation indexes are associated with 28-day mortality for ICH patients.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Spontaneous intracerebral hemorrhage (ICH), also known as hemorrhagic stroke, is the second most common cerebrovascular event and carries exceptionally high morbidity and mortality [1]. Clinical presentation may range from a few symptoms to critical states with the need for intensive care, surgery, and extensive rehabilitation. This high variability is one of the main concerns when investigating ICH [2]. Mortality may affect as much as 60% of patients per year, and only 20% will have functional independence at 6 months after the event [3].
Neuromonitoring analysis for spontaneous intracerebral hemorrhage (ICH) is still scarce, especially regarding vascular reactivity patterns. Intracerebral pressure (ICP) is most frequently monitored by an intraparenchymal pressure sensor used in brain lesion patients that require mechanical ventilation for neurological assessment while sedated. Augmented ICP and impaired cerebrovascular autoregulation have been recognized as important mechanisms to contribute to secondary brain injury after brain trauma, subarachnoid, and ischemic stroke [4, 5].
There is some evidence that ICP variability and frequency above 20 mmHg is associated with mortality and poor outcome in ICH patients [6]. However, neuromonitoring data regarding ICH to guide neurocritical care are still scarce and are mainly based on transferred information from traumatic brain injury (TBI) patients [7].
PRx is an index, recorded by ICM+ software, obtained from the correlation between arterial blood pressure (ABP) and ICP. A negative correlation (up to a maximum value of 0.25) is considered normal, since, in physiological conditions, it is expected that with an increase in ABP, cerebral vasculature may be able to increase resistance to maintain a stable ICP. This is considered a preserved cerebrovascular autoregulation.
Cerebral perfusion pressure (CPP) is the driving force that leads blood into cranial vessels and is calculated by the difference between ABP and ICP (ABP-ICP). Its value may be calculated in real time at bedside and is frequently described in the literature as a value that should be maintained between 60 and 80 mmHg. However, in the last few years, tailoring CPP based on cerebrovascular autoregulation status driven by PRx has been associated, in retrospective studies, with better outcomes for TBI patients. This concept is referred to as optimal CPP (CPPopt) [8, 9].
The need for neuromonitoring research and defining its utility in neurocritical care for ICH patients has been expressed often. Considering ICH management guideline assessment of ICP and CPP is important for secondary brain injury prevention; however, information regarding cerebrovascular reactivity is still scarce and its importance for patient management and association with outcome is yet to be established [10].
Our goal was to determine the association, if any, between the presence/absence of cerebrovascular autoregulation and patient 28-day survival.
Materials and Methods
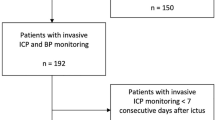
At our Neuro Critical Care Unit (NCCU), patients with intracerebral lesions that need to be sedated have placement of an ICP Codman® probe for intracranial neuromonitoring. The neuromonitoring integration data system used is ICM+ ® (Cambridge Enterprise UK). This allows us to calculate in real time and at bedside not only ICP, but also indices associated with ICP and ABP such as CPP, PRx, and optimal PRx. With the availability of these data from our unit, neuromonitoring records between 2013 and 2016 of adult patients, with spontaneous supra-tentorial ICH, were retrospectively reviewed. To be considered eligible, records had to have a minimum of 24 h of good-quality signals for each patient.
Variables considered for analysis were ICP, CPP, CPPopt, and PRx, as well as ICP dose, PRx dose, and time percentage above critical value (T%abv). ICP dose and PRx dose were calculated as secondary analysis using ICM® software and correspond to the area beneath the histogram curve relating time of exposure and ICP and PRx values. Information regarding demographics, surgical drainage, external ventricular drain (EVD) placement, and 28-day mortality was recorded. A t-test and Kaplan-Meier curves were used for statistical analysis. Significance value was considered at 0.05. Approval for data collection was obtained from our local ethics committee.
Results
We analyzed data from 46 patients, representing a mean duration of 263 ± 173 h of signal records and a median length of stay in the intensive care unit (ICU) of 22 (interquartile range (IQR) of 13) days. The mean age was 62.6 ± 11.8 years, and 24 (52%) of the patients were male. The EVD for cerebrospinal fluid (CSF) drainage was applied in addition to an ICP probe in 50% of patients, and 32.6% were sent to surgery for ICH evacuation (Table 1). Patients who died within 28 days (37.0%) had significantly higher mean ICP, PRx, ICP dose, PRx dose, and T%abv compared to those who survived (Table 2 and Fig. 1). Although patients who died had relatively low values of mean ICP (13.73 ± 7.75 mmHg), under the critical value of 20 mmHg, they presented a mean PRx of over 0.25, indicating impaired cerebrovascular reactivity (0.30 ± 0.26). Patients with PRx over 0.25 had significantly lower survival, with a proportion of 14% survival at 28 days, as opposed to 85% of those with PRx under 0.25 (p < 0.001) (survival curves in Fig. 2).
Discussion
Although mean ICP in ICH patients was under the critical value of 20 mmHg, the ICP dose and percentage of time above critical value was higher in patients with worse outcomes. The variability of intracranial pressure is high and the exposure of brain to higher pressure may not be well represented by mean ICP but better by ICP dose and T%abv. It is hypothesized that these measures of exposure representativity may have prognostic value in ICH patients.
Regarding cerebrovascular pressure reactivity, the mean values were higher than 0.25 in patients with worse outcomes (mortality at 28 days), as were PRx dose and T%abv.
In our cohort, patients with ICH were more exposed to autoregulation impairment than to increased ICP. When taking into account the patient’s outcome, ICH patients who died in the first 28 days had worse cerebrovascular reactivity indices, namely PRx, PRx dose, and time above the critical value of 0.25. Optimal CPP was particularly challenging to identify and achieve in these patients. However, our analysis had limitations in that our cohort was retrospectively analyzed. Based on our data, we may hypothesize that treatments aimed at keeping PRx as low as possible may be associated with better outcomes for ICH patients. Besides being an individualized value, most ICH patients have been chronically exposed to elevated blood pressure (one of the main risk factors in ICH), this is why it is possible that CPP intervals considered to TBI may not be adequate for all ICH patients. Individual adjustment, through continuous evaluation of PRx, may positively influence outcomes [9].
References
Roh D, Sun CH, Schmidt JM, Gurol E, Murthy S, Park S, Agarwal S, Connolly ES, Claassen J (2018) Primary intracerebral hemorrhage: a closer look at hypertension and cerebral amyloid angiopathy. Neurocrit Care 29:77–83. https://doi.org/10.1007/s12028-018-0514-z
Guo R, Blacker DJ, Wang X, Arima H, Lavados PM, Lindley RI, Chalmers J, Anderson CS, Robinson T (2017) Practice patterns for neurosurgical utilization and outcome in acute intracerebral hemorrhage: intensive blood pressure reduction in acute cerebral hemorrhage trials 1 and 2 studies. Neurosurgery 81:980–985. https://doi.org/10.1093/neuros/nyx129
Cordonnier C, Demchuk A, Ziai W, Anderson CS (2018) Intracerebral haemorrhage: current approaches to acute management. Lancet 392:1257–1268. https://doi.org/10.1016/s0140-6736(18)31878-6
Carney N, Totten AM, O’Reilly C, Ullman JS, Hawryluk GW, Bell MJ, Bratton SL, Chesnut R, Harris OA, Kissoon N, Rubiano AM, Shutter L, Tasker RC, Vavilala MS, Wilberger J, Wright DW, Ghajar J (2017) Guidelines for the management of severe traumatic brain injury, fourth edition. Neurosurgery 80:6–15. https://doi.org/10.1227/NEU.0000000000001432
Ko SB (2013) Multimodality monitoring in the neurointensive care unit: a special perspective for patients with stroke. J Stroke 15:99–108. https://doi.org/10.5853/jos.2013.15.2.99
Sykora M, Steinmacher S, Steiner T, Poli S, Diedler J (2014) Association of intracranial pressure with outcome in comatose patients with intracerebral hemorrhage. J Neurol Sci 342:141–145. https://doi.org/10.1016/j.jns.2014.05.012
Diedler J, Sykora M, Rupp A, Poli S, Karpel-Massler G, Sakowitz O, Steiner T (2009) Impaired cerebral vasomotor activity in spontaneous intracerebral hemorrhage. Stroke 40:815–819. https://doi.org/10.1161/STROKEAHA.108.531020
Aries MJ, Czosnyka M, Budohoski KP, Steiner LA, Lavinio A, Kolias AG, Hutchinson PJ, Brady KM, Menon DK, Pickard JD, Smielewski P (2012) Continuous determination of optimal cerebral perfusion pressure in traumatic brain injury. Crit Care Med 40:2456–2463. https://doi.org/10.1097/CCM.0b013e3182514eb6
Beqiri E, Smielewski P, Robba C, Czosnyka M, Cabeleira MT, Tas J, Donnelly J, Outtrim JG, Hutchinson P, Menon D, Meyfroidt G, Depreitere B, Aries MJ, Ercole A (2019) Feasibility of individualised severe traumatic brain injury management using an automated assessment of optimal cerebral perfusion pressure: the COGiTATE phase II study protocol. BMJ Open 9:e030727. https://doi.org/10.1136/bmjopen-2019-030727
Hemphill JC 3rd, Greenberg SM, Anderson CS, Becker K, Bendok BR, Cushman M, Fung GL, Goldstein JN, Macdonald RL, Mitchell PH, Scott PA, Selim MH, Woo D (2015) Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American stroke association. Stroke 46:2032–2060. https://doi.org/10.1161/str.0000000000000069
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Ethics declarations
The authors declare that they have no conflict of interest.
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Ferreira, A.V., Maia, I., Dias, C. (2021). Monitoring of Cerebrovascular Reactivity in Intracerebral Hemorrhage and Its Relation with Survival. In: Depreitere, B., Meyfroidt, G., Güiza, F. (eds) Intracranial Pressure and Neuromonitoring XVII. Acta Neurochirurgica Supplement, vol 131. Springer, Cham. https://doi.org/10.1007/978-3-030-59436-7_37
Download citation
DOI: https://doi.org/10.1007/978-3-030-59436-7_37
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-59435-0
Online ISBN: 978-3-030-59436-7
eBook Packages: MedicineMedicine (R0)