Abstract
In vivo imaging of β-amyloid (Αβ) has transformed the assessment of Αβ pathology and its changes over time, extending our insight into Aβ deposition in the brain by providing highly accurate, reliable, and reproducible quantitative statements of regional or global Aβ burden in the brain, proving essential in anti-Αβ therapeutic trials. Although cross-sectional evaluation of Αβ burden does not strongly correlate with cognitive impairment in AD, it does correlate with memory impairment and a higher risk for cognitive decline in the aging population and MCI subjects. This correlation with memory impairment, one of the earliest symptoms of AD, suggests that Αβ deposition is not part of normal aging. Longitudinal observations, coupled with different disease-specific biomarkers to assess potential downstream effects of Aβ, have confirmed that Αβ deposition in the brain starts decades before the onset of symptoms. Aβ imaging studies continue to refine our understanding of the role of Αβ deposition in Alzheimer’s disease.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Alzheimer’s disease
- Amyloid imaging
- Mild cognitive impairment
- Neuroimaging
- Positron emission tomography (PET)
- Neurodegeneration
1 Introduction
Alzheimer’s disease (AD) is a progressive and irreversible neurodegenerative disorder clinically characterized by memory loss and cognitive decline that severely affect the activities of daily living (Masters et al. 2006). AD is the leading cause of dementia in the elderly, leading invariably to death, usually within 7–10 years after diagnosis (Khachaturian 1985). The progressive nature of the neurodegeneration suggests an age-dependent process that ultimately leads to synaptic failure and neuronal damage in cortical areas of the brain essential for memory and other cognitive domains (Isacson et al. 2002). AD not only has devastating effects on the sufferers and their caregivers, but it also has a tremendous socioeconomic impact on families and the health system, a burden which will only increase in the upcoming years as the population of most countries ages (Johnson et al. 2000). In the absence of reliable biomarkers, direct pathologic examination of brain tissue derived from either biopsy or autopsy remains the only definitive method for establishing a diagnosis of AD (O'Brien et al. 2000). The typical macroscopic picture is gross cortical atrophy, while, microscopically, there are widespread cellular degeneration and diffuse synaptic and neuronal loss, accompanied by reactive gliosis and the presence of the pathological hallmarks of the disease: intracellular neurofibrillary tangles (NFT) and extracellular amyloid plaques (Jellinger 1990; Masters 2005; Masters and Beyreuther 2006). While NFT are intraneuronal bundles of paired helical filaments mainly composed of the aggregates of an abnormally phosphorylated form of tau protein (Jellinger and Bancher 1998; Michaelis et al. 2002), neuritic plaques consist of dense extracellular aggregates of β-amyloid (Αβ) (Masters et al. 1985), surrounded by reactive gliosis and dystrophic neurites. Αβ is a 4 kDa 39–43 amino acid metalloprotein derived from the proteolytic cleavage of the amyloid precursor protein (APP), by β- and γ-secretases (Cappai and White 1999). To date, all available genetic, pathological, biochemical, and cellular evidence strongly supports the notion that an imbalance between the production and removal of Αβ leading to its progressive accumulation is central to the pathogenesis of AD (Villemagne et al. 2006). The “Aβcentric theory” (Masters et al. 2006) postulates that Aβ plaque deposition is the primary event in a cascade of effects that lead to neurofibrillary degeneration and dementia (Hardy 1997).
Despite new diagnostic criteria (McKhann et al. 2011), clinical diagnosis of AD is still largely based on progressive impairment of memory and decline in at least one other cognitive domain and by excluding other diseases that might also present with dementia such as frontotemporal lobar degeneration (FTLD), dementia with Lewy bodies (DLB), stroke, brain tumor, normal pressure hydrocephalus, or depression (Larson et al. 1996; Cummings et al. 1998). A variable period of up to 5 years of prodromal decline in cognition characterized by a relatively isolated impairment in short-term memory that may also be accompanied by impairments of working memory, known as amnestic mild cognitive impairment (MCI), usually precedes the formal diagnosis of AD (Petersen et al. 1999; Petersen 2000). At this point, there is no cure for AD nor proven way to slow the rate of disease progression. Symptomatic treatment with acetylcholinesterase inhibitors or a glutamatergic moderator provides modest benefit in some patients usually by temporary stabilization rather than a noticeable improvement in memory function (Masters et al. 2006).
2 Aβ Imaging Radiotracers
Αβ plaques and NFTs are the pathological hallmark brain lesions of AD. These microscopic aggregates are still well beyond the resolution of conventional neuroimaging techniques used for the clinical evaluation of patients with AD. Positron emission tomography (PET) is a sensitive molecular imaging technique that allows in vivo quantification of radiotracer concentrations in the picomolar range, allowing the non-invasive assessment of molecular processes at their sites of action, detecting disease processes at asymptomatic stages when there is no evidence of anatomic changes on computed tomography (CT) and magnetic resonance imaging (MRI) (Phelps 2000). Since Αβ is at the center of AD pathogenesis, and given that several pharmacological agents aimed at reducing Αβ levels in the brain are being developed and tested, many efforts have been focused on generating radiotracers for imaging Αβ in vivo (Sair et al. 2004; Mathis et al. 2005; Villemagne et al. 2005). Several applications of Aβ imaging have been proposed (Table 10.1).
As a quantitative neuroimaging probe, the Aβ radiotracer must possess a number of key general properties: it should be a lipophilic, non-toxic small molecule with a high specificity and selectivity for Αβ, amenable for high specific activity labeling with 18F or other long-lived radioisotope, with no radiolabeled metabolites that enter the brain, while reversibly binding to Αβ in a specific and selective fashion (Laruelle et al. 2003; Nordberg 2004; Pike 2009). Overall, binding affinity and lipophilicity are the most crucial properties for in vivo neuroimaging tracers. While high affinity for Αβ is desirable to provide an adequate signal-to-noise ratio, this high affinity might also delay reaching binding equilibrium requiring an extended scanning time. And while lipophilicity is necessary for the tracer to cross the blood-brain barrier (BBB), if the radiotracer is too lipophilic, its non-specific binding might be too high (Laruelle et al. 2003; Pike 2009).
Through the years, several compounds have been evaluated with different fortune as potential Aβ probes: monoclonal and anti-Aβ antibodies fragments, serum amyloid P, basic fibroblast growth factor, Aβ fragments, and derivatives of histopathological dyes such as Congo red, Chrysamine-G, and Thioflavin S and T (for review, see (Villemagne & Rowe 2010)). While selective tau imaging for in vivo NFT quantification is still in the early stages of development (Okamura et al. 2005; Maruyama et al. 2009; Ojida et al. 2009; Fodero-Tavoletti et al. 2011), several Αβ radiotracers found their way into human clinical trials.
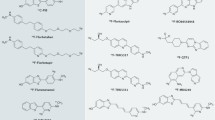
Almost a decade after unsuccessful trials with anti-Αβ antibodies (Majocha et al. 1992), Αβ imaging came to fruition with the first report of successful imaging in an AD patient with 18F-FDDNP, a tracer claimed to bind both plaques and NFT (Shoghi-Jadid et al. 2002). Since then, human Αβ imaging studies have been conducted in AD patients, normal controls, and patients with other dementias using 11C-PiB (Klunk et al. 2004), 11C-SB13 (Verhoeff et al. 2004), 11C-ST1859 (Bauer et al. 2006), 11C-BF227 (Kudo et al. 2007), 11C-AZD2138 (Nyberg et al. 2009), 18F-florbetaben (Rowe et al. 2008), 18F-flutemetamol (Serdons et al. 2009a; b), 18F-florbetapir (Wong et al. 2010), and 18F-NAV4694—a.k.a. AZD4694 (Cselenyi et al. 2012; Rowe et al. 2013a; b)—with PET.
In order to test compounds that could have more widespread application, preliminary studies with SPECT Aβ radiotracers 123I-CQ (Opazo et al. 2006) and 123I-IMPY (Newberg et al. 2006) showed limited utility for the evaluation of Aβ burden in AD, although 123I-IMPY might be useful in the evaluation of transmissible spongiform encephalopathies (Song et al. 2008). New SPECT radiotracers labeled with 123I or those that could be potentially labeled with 99mTc are being evaluated (Qu et al. 2007a; b; Serdons et al. 2007; Lin et al. 2009).
While all of the aforementioned tracers bind with varying degrees of success to Αβ fibrils and brain homogenates of AD patients, Congo Red and Thioflavin T—and some of their derivatives—have recently been shown to also bind to soluble oligomeric forms of Αβ (Maezawa et al. 2008). On the other hand, Αβ-soluble species represent less than 1% of the total brain Αβ (McLean et al. 1999; Roberts et al. 2017), and the reported affinity of PiB for these soluble oligomers seems to be significantly lower than for Αβ fibrils (Maezawa et al. 2008). Until highly selective radiotracers are developed to bind the Αβ-soluble species, the contribution of these oligomers to the PET signal in sporadic AD from tracers such as 11C-PiB is considered to be negligible (Mathis et al. 2007).
2.1 11C-Labeled Radiotracers
2.1.1 11C-PiB
11C-PiB (PiB) was the most successful and, early on, the most widely used of available Αβ tracers. PiB has been shown to possess high affinity and high selectivity for fibrillar Αβ in plaques and in other Αβ-containing lesions (Klunk et al. 2001; Mathis et al. 2002; Mathis et al. 2005; Price et al. 2005; Ye et al. 2005; Cohen et al. 2012). In vitro studies with high specific activity 3H-PiB demonstrated two binding sites for PiB (Klunk et al. 2005), and in in vitro studies, it has been used to establish the different binding sites in frontal cortex and hippocampus (Ni et al. 2013). Recent studies have shown that 11C-PiB binds with high affinity to the N-terminally truncated and modified Αβ and AβN3-pyroglutamate species in senile plaques (Maeda et al. 2007). In vitro assessment of 3H-PiB binding to white matter homogenates failed to show any specific binding (Fodero-Tavoletti et al. 2009a; b). PiB is a derivative of Thioflavin T, a fluorescent dye commonly used to assess fibrillization into β-sheet conformation (LeVine 3rd 1999), and as such PiB has been shown to bind to a range of additional Αβ-containing lesions, including diffuse plaques and cerebral amyloid angiopathy (CAA) (Lockhart et al. 2007), as well as to Αβ oligomers—albeit with lower affinity (Maezawa et al. 2008). PiB also displayed lower affinity toward other misfolded proteins with a similar β-sheet secondary structure such as α-synuclein (Fodero-Tavoletti et al. 2007; Ye et al. 2008) and tau (Lockhart et al. 2007; Ikonomovic et al. 2008). This is relevant particularly since AD has been described as a “triple brain amyloidosis” (Trojanowski 2002). Most importantly, these studies have shown that, at the concentrations achieved during a PET scan, 11C-PiB cortical retention in AD or DLB primarily reflects Αβ-related cerebral amyloidosis and not binding to Lewy bodies (LB) or NFT (Klunk et al. 2003; Fodero-Tavoletti et al. 2007; Lockhart et al. 2007; Ikonomovic et al. 2008). 11C-PiB has consistently provided quantitative information on Αβ burden in vivo, contributing new insights into Αβ deposition in the brain, allowing earlier detection of AD pathology (Klunk et al. 2004; Mintun et al. 2006; Rowe et al. 2007; Cohen et al. 2012) and accurate differential diagnosis of the dementias (Ng et al. 2007a; b; Rabinovici et al. 2007; Rowe et al. 2007).
2.1.2 11C-BF227
11C-BF-227 (BF227), a benzoxazole derivative, is also a promising tool as an Aβ imaging tracer (Okamura et al. 2004). In vitro binding studies with BF-227 demonstrated a Ki value of 4.3 nM for the compound to Aβ1–42 fibrils (Kudo 2006). A PET study using BF-227 showed AD patients were clearly distinguishable from age-matched controls with AD patients displaying significantly higher tracer retention in the cerebral cortex than controls (Kudo et al. 2007). Regional parametric analysis of the images further demonstrated a higher BF-227 retention in the posterior association cortex in AD patients. Unlike PiB, BF-227 presents with similar binding affinities for Aβ and α-synuclein (Fodero-Tavoletti et al. 2009a; b).
A few other C-11 Aβ radiotracers have been tested in humans. The evaluation of the stilbene derivative, 11C-SB13, showed that it could differentiate between five AD and six controls but had a lower effect size values when compared with 11C-PiB (Verhoeff et al. 2004). A preliminary study with the anti-Aβ agent 11C-ST1859 revealed small differences in radiotracer retention between nine AD patients and three healthy controls, but showed that the biodistribution and specificity of therapeutic agents can be assessed with PET (Bauer et al. 2006). Another Aβ radiotracer, 11C-AZD2138, not only showed reversible binding but also displayed very low non-specific binding to white matter (Johnson et al. 2009; Nyberg et al. 2009).
2.2 18F-Labeled Radiotracers
Unfortunately, the 20-min radioactive decay half-life of carbon-11 (11C) limits the use of 11C-PiB to centers with an on-site cyclotron and 11C radiochemistry expertise, making the cost of studies prohibitive for routine clinical use. To overcome these limitations, several Aβ tracers labeled with fluorine-18 (18F; half-life of 110 min) that permits centralized production and regional distribution have been developed and tested. Recent studies with the newly introduced 8F-labeled Aβ-specific radiotracers, 18F-florbetapir (Wong et al. 2010; Clark et al. 2011), 18F-florbetaben (Rowe et al. 2008; Barthel et al. 2011; Villemagne et al. 2011a; b), 18F-flutemetamol (Nelissen et al. 2009; Vandenberghe et al. 2010), and 18F-NAV4694 (Rowe et al. 2013a; b), have been successfully replicating the results obtained with 11C-PiB. The latest entry into the list of fluorinated Aβ radiotracers is 18F-MK-3328, developed by Merck, that has the disadvantage that >17% of its cortical signal was attributed to binding to monoamine oxidase-B (MAO-B) (Sur et al. 2010).
2.2.1 18F-FDDNP
A marked progression in the development of Aβ imaging tracers was the synthesis and characterization by Barrio and colleagues of a very lipophilic radiofluorinated 6-dialkylamino-2-naphthyethylidene derivative that presents nanomolar affinity to Aβ fibrils (Barrio et al. 1999; Agdeppa et al. 2001a; b). 18F-FDDNP is reported to bind both the extracellular Aβ plaques and the intracellular NFT in AD (Shoghi-Jadid et al. 2002) while also binding to prion plaques in Creutzfeldt-Jakob disease (CJD) brain tissue (Bresjanac et al. 2003). However, in vitro evaluation of FDDNP in concentrations similar to those achieved during a PET scan showed limited binding to both NFT and Aβ plaques (Thompson et al. 2009). 18F-FDDNP was used to obtain the first human PET images of Aβ in an 82-year-old woman with AD. AD patients present with higher accumulation and slower clearance of 18F-FDDNP than controls in brain areas such as the hippocampus (Shoghi-Jadid et al. 2002). Retention time of 18F-FDDNP in these brain regions was correlated with lower memory performance scores, regional glucose hypometabolism, and brain atrophy (Small et al. 2002; Small et al. 2006). However, the dynamic range of 18F-FDDNP cortical uptake values between HC and AD patients is small (9%). Direct comparison of 18F-FDDNP with 11C-PIB in monkeys (Noda et al. 2008) and in human subjects showed very limited dynamic range of 18F-FDDNP (Shih et al. 1987; Tolboom et al. 2009a; b) and in a longitudinal study was found to be less useful than 11C-PiB and 18F-fluorodeoxyglucose (FDG) for examining disease progression (Ossenkoppele et al. 2012). 18F-FDDNP has also been shown to bind to pure tauopathies, such as patients with progressive supranuclear palsy (Kepe et al. 2013) and in individuals suspected of chronic traumatic encephalopathy (Barrio et al. 2015; Chen et al. 2018; Omalu et al. 2018). Interestingly, 18F-FDDNP still remains the only Aβ tracer showing retention in the medial temporal cortex of AD patients (Shih et al. 1987) and correlation with cerebrospinal fluid (CSF) tau (Tolboom et al. 2009a; b).
2.2.2 18F-NAV4694
Developed by AstraZeneca, the most salient features of 18F-NAV4694 are, as with 11C-AZD2138, fast tracer kinetics and low non-specific binding to white matter, similar to that one observed with 11C-PiB, which is helpful for the detection of small Aβ cortical deposits at very early stages of the disease process (Sundgren-Andersson et al. 2009; Jureus et al. 2010). Head-to-head comparison with PIB suggests 18F-NAV4694 has a better binding profile than PiB (Rowe et al. 2013a; b; Rowe et al. 2016). Clinical studies with 18F-NAV4694 showed a clear distinction in tracer retention between healthy controls and AD patients (Rowe et al. 2013a; b; Rowe et al. 2016). To this date, 18F-NAV4694 remains the only main F-18 Aβ radiotracer that has not yet been presented to the Food and Drug Administration (FDA) for approval.
2.2.3 18F-Florbetaben
18F-florbetaben (a.k.a. Neuraceq™; AV1; BAY94–9172), synthesized by Kung and colleagues (Zhang et al. 2005a; b), developed by Bayer Healthcare, and nowadays commercialized by Life Molecular Imaging Inc., has been shown to bind with high affinity to Aβ in brain homogenates and selectively labeled Aβ plaques and CAA in AD tissue sections (Zhang et al. 2005a; b). After injection into Tg2576 transgenic mice, ex vivo brain sections showed localization of 18F-florbetaben in regions with Aβ plaques as confirmed by thioflavin binding (Zhang et al. 2005a, b). At tracer concentrations achieved during human PET studies, florbetaben did not show binding to LB or NFT-tau in post-mortem cortices from DLB or FTLD patients (Fodero-Tavoletti et al. 2012). In human studies, cortical retention of 18F-florbetaben was higher at 90 min post-injection in all AD subjects compared to age-matched controls and FTLD patients, with binding matching the reported post-mortem distribution of Aβ plaques (Rowe et al. 2008). Recently completed phase II clinical studies further confirmed these results (Barthel et al. 2011). A longitudinal study in MCI subjects established the usefulness of FBB PET in predicting progression to AD (Ong et al. 2013; Ong et al. 2015). 18F-Florbetaben is highly correlated with 11C-PiB (Villemagne et al. 2012a; b; c; d; Rowe et al. 2017) and was used to detect the presence or absence of AD pathology in the brain in subjects from a wide spectrum of neurodegenerative diseases (Villemagne et al. 2011a; b) aiding in the differential diagnosis of dementia (Ceccaldi et al. 2018) and clarifying diagnosis in patients with ambiguous CSF results (Manca et al. 2019). It has been shown that 18F-florbetaben can make accurate ante-mortem differential diagnosis of brain amyloidosis (Sabbagh et al. 2017), and phase III confirmed 18F-florbetaben is highly associated with Aβ plaque pathology (Sabri et al. 2015), leading to its approval for clinical use by the FDA. 18F-Florbetaben has been recently incorporated as an Aβ radiotracer to ADNI-3.
2.2.4 18F-Florbetapir
As 18F-florbetaben, 18F-florbetapir is a stilbene derivative that was also synthesized by Kung and colleagues at the University of Pennsylvania (Zhang et al. 2005a, b) and has been developed by Avid Radiopharmaceuticals. Initial in vitro evaluation showed binding to Aβ plaques in AD brain sections (Choi et al. 2009; Lin et al. 2009). The most salient feature of this tracer is its rapid reversible binding characteristics allowing scanning only at 45–50 min after injection, similar to 11C-PiB (Wong et al. 2010). 18F-Florbetapir (Lister-James et al. 2011) has become the most widely used Aβ radiotracer after 11C-PiB and has been adopted as the Aβ radiotracer for ADNI 2 and also used in patient selection and to evaluate treatment response in a large number of multicenter therapeutic trials around the world. Several multicenter phase I and II studies in AD, MCI, and HC showed the ability of 18F-florbetapir to discriminate between AD and age-matched controls, with ~50% of the MCI subjects presenting with high 18F-florbetapir retention (Fleisher et al. 2011). Multicenter studies using 18F-florbetapir to assess the relationship between Aβ burden and cognition showed that in clinically normal older individuals, Aβ burden in the brain is associated with poorer memory performance (Sperling et al. 2012), while MCI subjects with high Aβ burden in the brain are at a significantly higher risk of cognitive decline over 18 months (Doraiswamy et al. 2012). An initial phase III study in 35 volunteers demonstrated a 96% agreement between 18F-florbetapir and neuropathology for the detection of brain Aβ in vivo and no retention in young controls (Clark et al. 2011). An extension phase III in 59 volunteers established that 18F-florbetapir has a sensitivity of 92% and a specificity of 100% for the detection of Aβ pathology (Clark et al. 2012). The use of 18F-florbetapir changes patient management (Pontecorvo et al. 2017a; b), and using a combination of visual inspection with a semiquantitative approach improves diagnostic accuracy (Pontecorvo et al. 2017a; b). No significant difference in the rates of Aβ accumulation was observed when compared against PiB in longitudinal studies (Su et al. 2019), where a slow but steady increase in florbetapir signal was observed in young controls (Gonneaud et al. 2017), which was associated with incipient cognitive decline (Bischof et al. 2016) even at low Aβ levels (Landau et al. 2018). 18F-Florbetapir (as Amyvid™) was the first radiotracer approved by the FDA for detection of Aβ in vivo and the first 18F-labeled radiotracer approved by the FDA since FDG.
2.2.5 18F-Flutemetamol
Another fluorinated tracer, developed by GE Healthcare, that also completed a phase III study (Curtis et al. 2015) is 18F-flutemetamol (a.k.a. GE067; Vizamyl™) (Serdons et al. 2009a; b). Phase I and II studies demonstrated that 18F-flutemetamol can clearly differentiate between AD and HC (Nelissen et al. 2009; Vandenberghe et al. 2010) and that when combined with brain atrophy, it could be predictive of disease progression in MCI subjects (Thurfjell et al. 2012). 18F-Flutemetamol brain retention is highly correlated with 11C-PIB (Vandenberghe et al. 2010; Mountz et al. 2015; Lowe et al. 2017) and with neuropathology (Wolk et al. 2011; Wong et al. 2012; Thal et al. 2015; Thal et al. 2018) where no difference was found in the 18F-flutemetamol regional brain distribution between MCI subjects and end-stage AD patients (Farrar et al. 2019). 18F-Flutemetamol has been shown to aid in the clinical diagnosis and management of individuals presenting with memory decline (Zwan et al. 2017; Leuzy et al. 2019). 18F-Flutemetamol has been approved for clinical use by the FDA.
All Aβ imaging radiotracers present a similar pattern of tracer retention in AD, with the highest retention in frontal, temporal, and posterior cingulate cortices, reflecting Aβ plaque burden (Fig. 10.1). While the cortical retention of 18F-florbetapir, 18F-florbetaben, and 18F-flutemetamol provides a clear separation of AD patients from HC subjects, the degree of cortical retention with 18F-florbetapir and 18F-florbetaben is lower than with 11C-PiB (Villemagne et al. 2012a; b; c; d; Wolk et al. 2012a; b), showing a narrower dynamic range of SUVR values that visually appears as a relatively higher degree of non-specific binding to white matter. While the cortical retention of 18F-flutemetamol is similar to that of 11C-PiB, the non-specific retention in white matter is much higher (Vandenberghe et al. 2010). Due to this lower signal-to-noise ratio, visual read-outs of the images are more challenging than with 11C-PiB (Rowe and Villemagne 2011). These issues are not observed when using 18F-NAV4694, which is an Aβ tracer that yields high contrast images with relatively low tracer retention in white matter and has proven to perform slightly better than 11C-PiB (Rowe et al. 2016). While 11C-PiB PET and 18F-NAV4694 images in subjects with AD pathology usually clearly show high radiotracer retention in the gray matter in excess of that in subjacent white matter, these FDA-approved 18F tracers frequently show loss of the normal gray-white matter demarcation as the predominant evidence of cortical Aβ deposition (Rowe and Villemagne 2011).
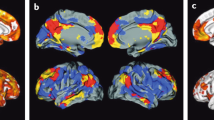
Representative sagittal (top row), transaxial (middle row), and coronal (bottom row) PET images overlaid on MRI from Alzheimer’s disease patients obtained with different Aβ imaging radiotracers. From left to right, 11C-PiB, 18F-florbetaben, 18F-flutemetamol, 18F-florbetapir, and 18F-NAV4694. The images show the typical pattern of tracer retention in AD, with the highest retention in frontal, temporal, and posterior cingulate cortices, reflecting Aβ plaque burden
3 Aβ Imaging in Alzheimer’s Disease
On visual inspection, cortical retention of Aβ tracers is higher in AD, with a regional brain distribution that is highest in frontal, cingulate, precuneus, striatum, parietal, and lateral temporal cortices, while occipital, sensorimotor, and mesial temporal cortices are much less affected (Villemagne et al. 2018). Both quantitative and visual assessment of PET images present a pattern of radiotracer retention that seems to replicate the sequence of Αβ deposition found at autopsy (Braak and Braak 1997), with initial deposition in the orbitofrontal cortex, inferior temporal, cingulate gyrus, and precuneus, followed by the remaining prefrontal cortex, lateral temporal, and parietal cortices (Fig. 10.2). Multimodality studies in early AD have shown that the regional pattern of Αβ deposition is similar to the anatomy of the “default network” (Buckner et al. 2005; Sperling et al. 2009), a specific, anatomically defined brain system responsible for internal modes of cognition, such as self-reflection processes, conscious resting state, or episodic memory retrieval (Buckner et al. 2008; Wermke et al. 2008), an association that is present even in non-demented subjects (Drzezga et al. 2011). The regional distribution of Αβ tracers in the brain sometimes varies with the specific Αβ distribution characteristic of the underlying pathology, for example, carriers of mutations associated with familial AD (Klunk et al. 2007; Koivunen et al. 2008a; b; Villemagne et al. 2009a; b) and subjects with posterior cortical atrophy (Ng et al. 2007a; b; Tenovuo et al. 2008; Kambe et al. 2010; Formaglio et al. 2011) or CAA (Johnson et al. 2007; Dierksen et al. 2010) each presenting a different regional 11C-PiB distribution of retention to the one usually observed in sporadic AD (Klunk et al. 2004; Rowe et al. 2007).
Top row. Schematics showing the stages of Aβ deposition in the human brain as proposed by Braak and Braak (Braak and Braak 1997). Bottom row. Representative sagittal PET images showing the regional brain distribution of 11C-PIB in two asymptomatic healthy age-matched controls, one with low (PiB- HC) and one with high Aβ burden (PiB+ HC), a subject classified as mild cognitive impairment (MCI) with significant Aβ deposition in the brain, and an Alzheimer’s disease (AD) patient with even a higher Aβ burden in the brain. The pattern of PiB retention replicates the sequence of Αβ deposition described from post-mortem studies (Braak and Braak 1997)
Cross-sectional semiquantitative PET studies using diverse Αβ radiotracers have shown a robust difference in retention between AD patients and age-matched controls (Klunk et al. 2004; Price et al. 2005; Mintun et al. 2006; Kudo et al. 2007; Pike et al. 2007; Rabinovici et al. 2007; Rowe et al. 2007; Jack Jr. et al. 2008a; b; Morris et al. 2009; Furst et al. 2010; Rowe et al. 2010; Barthel et al. 2011; Fleisher et al. 2011; Villemagne et al. 2011a; b; Camus et al. 2012; Cselenyi et al. 2012). It should be noted that about 20–30% of clinically diagnosed probable AD patients have been shown to have a low Aβ imaging scan, which effectively rules out AD pathology as the etiology of the dementia.
Longitudinal studies have shown that significant, albeit small, increases in Αβ deposition can be measured but also that these increases in Αβ deposition are present across the whole spectrum of cognitive stages, from cognitively unimpaired individuals to AD dementia patients (Engler et al. 2006; Jack Jr. et al. 2009; Okello et al. 2009a; b; Resnick et al. 2010; Rinne et al. 2010; Sojkova et al. 2011a; b; Villemagne et al. 2011a; b; Villain et al. 2012; Jack Jr. et al. 2013; Villemagne et al. 2013; Landau et al. 2015). Αβ accumulation is observed in individuals considered to have low Αβ loads with about 7% of them progressing above the threshold in about 2.5 years (Vlassenko et al. 2011), and subtle cognitive decline is observed in those subthreshold individuals that accumulate Αβ over time (Landau et al. 2018). Αβ accumulation is a slow and protracted process that precedes by about two decades the onset of the typical phenotype of AD dementia (Jack Jr. et al. 2013; Villemagne et al. 2013; Landau et al. 2015). In individuals with the highest Aβ burdens independent of clinical classification and also as Alzheimer’s dementia progresses, the rates of Aβ deposition start to slow down (Villain et al. 2012; Jack Jr. et al. 2013; Villemagne et al. 2013; Landau et al. 2015). This slower rate of Αβ deposition observed at the late stages of the disease might be attributed to a saturation of the process or, much more likely, extensive neuronal death that precludes production of Αβ.
4 Ante-mortem Post-mortem Correlations
While clinicopathological studies have shown that the accuracy of clinical assessments for the diagnosis of AD ranges between 70 and 90%, depending on the specialization of the center, the regional retention of radiotracers is highly correlated with regional Αβ plaques as reported at autopsy or biopsy (Bacskai et al. 2007; Ikonomovic et al. 2008; Leinonen et al. 2008a; b; Villemagne et al. 2009a; b; Burack et al. 2010; Clark et al. 2011; Kadir et al. 2011; Sabbagh et al. 2011; Sojkova et al. 2011a; b; Wolk et al. 2011; Clark et al. 2012; Wong et al. 2012; Curtis et al. 2015; Sabri et al. 2015), with a higher Aβ concentration in the frontal cortex than in the hippocampus, consistent with previous reports (Arnold et al. 2000; Naslund et al. 2000). As mentioned before, and in contrast with CSF studies, Αβ imaging studies can assess both the quantity and regional brain distribution of Αβ, as observed in the different patterns of regional distribution of Αβ observed in sporadic AD (Klunk et al. 2004; Nordberg 2007), familial AD (Klunk et al. 2007; Koivunen et al. 2008a; b; Villemagne et al. 2009a; b), familial British dementia (Villemagne et al. 2010), and CAA subjects (Johnson et al. 2007; Dierksen et al. 2010). The results from Αβ imaging might be considered for the brain regions to be sampled and stained for the neuropathological diagnosis of AD (Montine et al. 2012).
There have been a handful of cases reported with a discrepancy between a positive neuropathological finding or a strong biomarker profile of AD and a low PET signal (Leinonen et al. 2008a; b; Tomiyama et al. 2008; Cairns et al. 2009; Schöll et al. 2011; Sojkova et al. 2011a; b; Ikonomovic et al. 2012). For example, Tomiyama and colleagues reported a case of a novel APP mutation (E693Delta) where the mutant Αβ does not fibrillize in the same way as wild-type Aβ (Inayathullah and Teplow 2011) in which the 11C-PiB study showed low cortical retention (Tomiyama et al. 2008). Schöll and colleagues report on two subject carriers of the Artic APP mutation (Schöll et al. 2011). These two mutation carriers, while presenting with low 11C-PiB retention, had a clear “AD pattern” of brain hypometabolism as measured by FDG, low Aβ1–42, and elevated t-tau and p-tau in CSF (Schöll et al. 2011). On the one hand, these cases illustrate the possibility that different conformations of Aβ deposits (Levine 3rd and Walker 2010) may affect the binding of tracers and that Aβ imaging may not recognize all types of Aβ pathologies with equal sensitivity (Walker et al. 2008; Rosen et al. 2010). On the other, it raises issues not only in regard to the sensitivity of Aβ imaging (e.g., what is the minimal amount of Aβ detected by PET?) but also in regard to the evaluation of brain tissue (e.g., type of tissue dye, type of antibodies used for immunohistochemistry and ELISA, diagnostic criteria, etc.).
Second-generation radiofluorinated Aβ imaging radiotracers underwent phase III studies comparing ante-mortem PET with post-mortem findings. All these tracers showed a very high accuracy against neuropathology, with a sensitivity of 92% and a specificity of 100% for florbetapir (Clark et al. 2012), a sensitivity of 86% and a specificity of 92% for flutemetamol (Curtis et al. 2015), and a sensitivity of 98% and a specificity of 89% for florbetaben (Sabri et al. 2015). Based on the results of these phase III studies, all three tracers received Food and Drug Administration (FDA) and European Medicines Agency (EMA) approval for clinical use, specifically to rule out the presence of Aβ-amyloid in the brain.
5 Aβ Deposition in Non-demented Individuals and Its Relation with Cognition
About 25–35% of elderly subjects performing within normal limits on cognitive tests present with high cortical 11C-PiB retention, predominantly in the prefrontal and posterior cingulate/precuneus regions (Mintun et al. 2006; Rowe et al. 2007; Aizenstein et al. 2008; Villemagne et al. 2008a; b; Mormino et al. 2009; Reiman et al. 2009a; b; Rowe et al. 2010). These findings are in perfect agreement with post-mortem reports that ~25% of non-demented older individuals over the age of 75 have Aβ plaques in the brain (Davies et al. 1988; Morris and Price 2001; Forman et al. 2007). Aβ deposition in these non-demented individuals (Mintun et al. 2006; Rowe et al. 2007; Aizenstein et al. 2008; Villemagne et al. 2008a; b) might reflect preclinical AD (Price and Morris 1999). Furthermore, the prevalence of subjects with high 11C-PiB retention increases each decade at the same rate as the reported prevalence of plaques in non-demented subjects in autopsy studies (Rowe et al. 2010). While cross-sectional examination of these controls essentially show no significant differences in cognition (Mintun et al. 2006; Aizenstein et al. 2008; Jack Jr. et al. 2008a; b; Villemagne et al. 2008a; b; Rowe et al. 2010), significant cognitive differences in females were reported (Pike et al. 2011). More recently, Sperling and colleagues reported that high Aβ deposition in the brain was correlated with lower immediate and delayed memory scores in otherwise healthy controls (Sperling et al. 2012). Interestingly, 23% of healthy elderly controls showed signs of neurodegeneration but no signs of Aβ pathology: This group of healthy elderly controls was defined by the presence of neurodegeneration in the absence of Aβ pathology and was proposed to have suspected non-Alzheimer’s pathophysiology (SNAP) (Jack Jr. et al. 2012). While Aβ is a strong predictor of cognitive decline, neurodegeneration alone did not differ from those controls without neurodegeneration or Aβ in the brain (Burnham et al. 2016). When this basic approach was applied to the general population, it was found that almost all 50-year-old cases had no signs of either Aβ or neurodegeneration, while by 80 years old ~75% of the cohort presented signs of one or both markers (Jack Jr. et al. 2014). The detection of Aβ pathology at the presymptomatic stage is also of crucial importance because if this group truly represents preclinical AD (Price and Morris 1999; Thal et al. 2004; Backman et al. 2005; Small et al. 2007), it is precisely the group that may benefit the most from therapies aimed at reducing or eliminating Αβ from the brain before irreversible neuronal or synaptic loss occurs (Sperling et al. 2011a; b; c). Moreover, some individuals remain cognitively intact, while others develop cognitive impairment, despite carrying the same pathological burden, so factors that convey resilience or susceptibility to Αβ should also be considered.
Individuals fulfilling criteria for MCI are a heterogeneous group with a wide spectrum of underlying pathologies (Petersen 2000; Winblad et al. 2004). About 40–60% of carefully characterized subjects with MCI will subsequently progress to meet criteria for AD over a 3–4-year period (Petersen et al. 1995; Petersen et al. 1999; Petersen 2000). Αβ imaging has proven useful in identifying MCI individuals with and without AD pathology. Approximately 50–70% of the subjects classified as MCI present with high cortical 11C-PiB retention (Price et al. 2005; Kemppainen et al. 2007; Pike et al. 2007; Forsberg et al. 2008; Lowe et al. 2009; Mormino et al. 2009). While most studies found that subjects classified as non-amnestic MCI show low Αβ levels in the brain (Pike et al. 2007; Lowe et al. 2009), other reports did not (Wolk et al. 2009). More specifically, subjects classified as non-amnestic single domain MCI usually do not show Αβ deposition, consistent with a non-AD underlying pathological process (Pike et al. 2007; Villemagne et al. 2011a; b).
Subjective cognitive impairment (SCI) or subjective cognitive decline (SCD) is believed to be an early behavioral manifestation along the AD spectrum that may precede objective MCI and is associated with up to a sixfold increased risk of developing AD (Jessen et al. 2010). In order to better characterize those SCI/SCD individuals that might have confounding personality or affective factors unrelated to AD, several studies evaluated the relationship between SCI/SCD and specific markers of AD pathology. While only a few individuals with SCI/SCD develop AD within a few years (Slot et al. 2019), these individuals are likely to already harbor Aβ in the brain (Buckley et al. 2016; Perrotin et al. 2017; Hu et al. 2019), but Aβ burden has a stronger association to worries than to the severity of the cognitive complaints (Verfaillie et al. 2019).
To date, the relationship between Αβ deposition and the development of clinical symptoms is not fully understood. Although Aβ burden as assessed by PET does not correlate with measures of memory impairment in established clinical AD, it does correlate with memory impairment in MCI and healthy older subjects (Pike et al. 2007; Villemagne et al. 2008a; b). In subjects with MCI, this Aβ-related impairment is thought to be mediated through tau and hippocampal atrophy (Mormino et al. 2009; Sperling et al. 2019) while modulated by cognitive reserve (Roe et al. 2008; Rentz et al. 2010) and glucose metabolism (Cohen et al. 2009; Hanseeuw et al. 2017), although at the AD stage there is no longer correlation between glucose metabolism, cognition, and Αβ burden (Furst et al. 2010).
Longitudinal studies are helping clarify the relationship between Aβ burden and cognitive decline in healthy individuals and SCI/SCD and MCI subjects. High 11C-PiB cortical retention in non-demented elderly subjects is associated with a greater risk of cognitive decline (Villemagne et al. 2008a; b; Resnick et al. 2010; Villemagne et al. 2011a; b). For example, the evaluation of either the cognitive trajectories (Villemagne et al. 2008a; b; Resnick et al. 2010) or changes in cognition prospectively (Morris et al. 2009; Villemagne et al. 2011a; b; Doraiswamy et al. 2012) shows that individuals with substantial Αβ deposition are more likely to present with cognitive decline. Aggregating the results from recently reported longitudinal studies reveals that 65% of MCI subjects with marked 11C-PiB retention progressed to AD over 2–3 years, showing that MCI subjects with high Aβ deposition in the brain are >10 times more likely to progress to AD than those with low Aβ deposition, highlighting the clinical relevance and potential impact in patient management of Aβ imaging (Forsberg et al. 2008; Okello et al. 2009a; b; Wolk et al. 2009; Villemagne et al. 2011a; b; Rowe et al. 2013a; b). The observations that 11C-PiB retention in non-demented individuals relates to episodic memory impairment, one of the earliest clinical symptoms of AD, and that extensive Aβ deposition is associated with a significant higher risk of cognitive decline in HC and with progression from MCI to AD emphasize the non-benign nature of Aβ deposition and support the hypothesis that Aβ deposition occurs well before the onset of symptoms, further suggesting that early disease-specific therapeutic intervention at the presymptomatic stage might be the most promising approach to either delay onset or halt disease progression (Sperling et al. 2011a; b; c).
The high prevalence of Αβ deposition in non-demented individuals raises questions in regard to Αβ toxicity and deposition. The evidence points to the fact that Αβ toxicity in the form of oligomers and Αβ deposition in the form of aggregates precede by many years the appearance of clinical symptoms (Price and Morris 1999). These neurotoxic processes eventually lead to synaptic and neuronal dysfunction, manifested as cognitive impairment, gray matter atrophy, and glucose hypometabolism (Eckert et al. 2003; Suo et al. 2004; Leuner et al. 2007). Therefore, it is highly likely that 11C-PiB retention in non-demented individuals reflects preclinical AD (Mintun et al. 2006; Rowe et al. 2007; Aizenstein et al. 2008; Villemagne et al. 2008a; b). This may be associated or attributed to a different susceptibility/vulnerability to Αβ, at either a cellular (frontal neurons seem to be more resistant to Αβ than hippocampal neurons) (Roder et al. 2003; Resende et al. 2007), a regional (compensatory upregulation of choline acetyltransferase (ChAT) activity in the frontal cortex and hippocampi of MCI and early AD subjects) (DeKosky et al. 2002), or an individual or personal level, either due to a particular cognitive reserve (Mortimer 1997; Stern 2002; Kemppainen et al. 2008; Roe et al. 2008), due to differences in Aβ conformation affecting toxicity and/or aggregation (Lockhart et al. 2005; Deshpande et al. 2006; Levine 3rd and Walker 2008; Walker et al. 2008), or because an idiosyncratic threshold must be exceeded for synaptic failure and neuronal death to ensue (Suo et al. 2004). These factors would help explain why some older individuals with a significant Αβ burden are cognitively unimpaired, while others with lower Αβ burden and no genetic predisposing factors have already developed the full clinical AD phenotype. Ongoing longitudinal studies will help identify those subjects that despite substantial Aβ deposition do not develop the AD phenotype, thus allowing to isolate genetic or environmental factors that may convey resistance to Aβ (Sojkova et al. 2011a; b).
The lack of a strong association between Aβ deposition and measures of cognition, synaptic activity, and neurodegeneration in AD, in addition to the evidence of Αβ deposition in a high percentage of MCI and asymptomatic HC, suggests that Αβ is an early and necessary, though not sufficient, cause for cognitive decline in AD (Villemagne et al. 2008a; b; Rabinovici et al. 2010; Sojkova et al. 2011a; b), indicating the involvement of other downstream mechanisms, likely triggered by Αβ such as NFT formation, synaptic failure, and eventually neuronal loss. Also at play are factors as age, years of education, occupational level, and brain volume among others that, under the umbrella of “cognitive reserve,” appear to have a modulatory role between cognition and Αβ deposition (Roe et al. 2008; Chetelat et al. 2010a; b; Tucker and Stern 2011). Selective tau imaging studies are helping elucidate the bridge between Aβ deposition and the persistent decline in cognitive function observed in AD (Fodero-Tavoletti et al. 2011, Villemagne et al. 2015, Villemagne et al. 2018).
6 Relation of Aβ Imaging with Other Biomarkers
Another rapidly growing area is the exploration of the potential association between biomarkers of Aβ deposition or neurodegeneration (Wahlund and Blennow 2003; Morris et al. 2005; Sunderland et al. 2005; de Leon et al. 2006; Thal et al. 2006; Blennow et al. 2007; Shaw et al. 2007; Clark et al. 2008; Jack Jr. et al. 2010; Albert et al. 2011; McKhann et al. 2011; Storandt et al. 2012) and Aβ burden as measured by PET (Jack Jr. et al. 2008a; b; Jack Jr. et al. 2010). The new National Institute on Aging and Alzheimer’s Association (NIA-AA) Research Framework—AT(N) (Jack Jr. et al. 2016; Jack et al. 2018)—is based on a biological definition of Alzheimer’s disease and uses three kinds of biomarkers, markers of Aβ (A) and tau (T) pathology and markers of neuronal injury/neurodegeneration (N), for characterizing the physiopathology of AD. The implementation of the AT(N) classification is helping to refine prognosis in clinical practice and identify those at-risk individuals more likely to clinically progress, for their inclusion in therapeutic trials (Cummings 2019).
6.1 FDG
While the typical pattern of Aβ deposition in sporadic AD is highest in frontal, cingulate, precuneus, striatum, parietal, and lateral temporal cortices (Klunk et al. 2004), in FDG studies, the typical “AD pattern” is of temporoparietal and posterior cingulate hypometabolism with sparing of the basal ganglia, thalamus, cerebellum, and primary sensorimotor cortex (Salmon et al. 1994; Devanand et al. 1997; Coleman 2005; Jagust et al. 2007). Due to its high sensitivity (>90%) for detecting temporoparietal and posterior cingulate hypometabolism, FDG-PET has improved diagnostic and prognostic accuracy in patients with probable AD (Salmon et al. 1994; Kennedy et al. 1995; Small et al. 1995; Silverman et al. 2001; Silverman et al. 2002). A similar pattern of hypometabolism has been reported in normal elderly ApoE ε4 carriers (Reiman et al. 1996), MCI subjects (Chetelat et al. 2003, Chetelat et al. 2005, Mosconi et al. 2006), asymptomatic subjects with mutations associated with familial AD (Kennedy et al. 1995; Rossor et al. 1996), and subjects with a strong family history of AD (Mosconi et al. 2006a; b). FDG hypometabolism is correlated with cognition (Landau et al. 2009; Furst et al. 2010) and is predictive of future cognitive decline (Drzezga et al. 2003; Mosconi et al. 2004; Drzezga et al. 2005).
Visual assessment of PET images by clinicians blinded to the clinical status has demonstrated that 11C-PiB was more accurate than FDG to distinguish AD from HC (Ng et al. 2007a; b) and slightly better than FDG in differentiating AD from FTLD (Rabinovici et al. 2011). Similarly, Aβ imaging outperforms FDG at identifying MCI subtypes (Lowe et al. 2009).
While some reports found no association between FDG and 11C-PiB in AD (Furst et al. 2010), others found an inverse correlation between them in temporal and parietal cortices (Cohen et al. 2009), but no correlation has been shown in the frontal lobe (Klunk et al. 2004; Edison et al. 2007). A possible explanation for this dissociation is that there are compensatory mechanisms such as upregulation of ChAT in the frontal lobe of MCI individuals and mild AD patients (DeKosky et al. 2002; Ikonomovic et al. 2007) that might delay the manifestation of synaptic dysfunction; therefore, no hypometabolism is observed on FDG in the frontal lobe. Glucose metabolism in the frontal lobe eventually decreases as the disease progresses, and the characteristic temporoparietal pattern of AD becomes less apparent (Mielke et al. 1992; Herholz 1995). Conversely to what is observed in the frontal area, in AD patients ChAT is significantly decreased in the posterior cingulated gyrus (Ikonomovic et al. 2011), which is characterized by being one of the regions where marked and early Aβ deposition is associated with substantial hypometabolism (Drzezga et al. 2011; Yokokura et al. 2011).
6.2 CSF
Analysis of CSF allows simultaneous measurement of the concentration of the two main hallmarks of AD, Aβ and tau. It has been reported to be highly accurate in the diagnosis of AD as well of predicting cognitive decline (Blennow et al. 2007; de Leon et al. 2007; Hansson et al. 2007; Mattsson et al. 2009; Mattsson et al. 2011). The typical CSF profile in AD is low Aβ1–42 and high total (t-tau) and phosphorylated tau (p-tau) (Strozyk et al. 2003; Hampel et al. 2010). Slower Aβ clearance from the brain (Bateman et al. 2006; Mawuenyega et al. 2010) is likely to lead to Aβ deposition in the brain and lower CSF Aβ1–42 concentrations. While there is no correlation between 11C-PiB retention and CSF t-tau or p-tau (Tolboom et al. 2009a; b; Forsberg et al. 2010), several studies have reported a strong inverse correlation between Aβ deposition in the brain and CSF Aβ1–42 (Fagan et al. 2006; Fagan et al. 2007; Forsberg et al. 2008; Koivunen et al. 2008a; b; Fagan et al. 2009; Grimmer et al. 2009; Landau et al. 2013; Mattsson et al. 2017). Both high Aβ burden and low CSF Aβ1–42 have been observed in cognitively unimpaired individuals probably reflecting Aβ deposition years before the manifestation of the AD phenotype (Mintun et al. 2006; Fagan et al. 2007; Rowe et al. 2007; Aizenstein et al. 2008; Fagan et al. 2009). Large population studies, such as ADNI, AIBL, and DIAN (Shaw et al. 2007; Apostolova et al. 2010; Li et al. 2015; McDade et al. 2018), have been crucial for the validation of Aβ imaging and CSF assessments as antecedent biomarkers of AD as well as determining the longitudinal trajectories of these fluid biomarkers (Toledo et al. 2013; Fagan et al. 2014; Wang et al. 2018), while the introduction of automated immunoassay platforms has allowed reproducibility of results across time and across centers (Rozga et al. 2017; Schindler et al. 2018; Shaw et al. 2019). Given that CSF and Aβ imaging measure different biochemical pools of Aβ (Roberts et al. 2017), the inverse relationship between them is not linear (Toledo et al. 2015). This is also reflected in how these biomarkers change over time. In contrast to the sigmoid shape curve observed for longitudinal Aβ imaging studies in the brain (Jack Jr. et al. 2013; Villemagne et al. 2013), CSF Aβ1–42 decreases over time tend to adopt a logarithmical shape (Toledo et al. 2013).
6.3 Plasma Aβ
In recent years, there has been a revolution in the ability to measure Aβ peptides and APP fragments in plasma. Several reports demonstrate that plasma Aβ levels can predict both CSF Aβ and brain Aβ with an accuracy ~90% (Fandos et al. 2017; Ovod et al. 2017; Nakamura et al. 2018; Palmqvist et al. 2019). The introduction of these tests will have immediate effects as an inexpensive way to identify individuals at risk of developing AD, lowering the costs of screening for therapeutic AD trials by reducing number of lumbar punctures and Aβ PET scans.
6.4 MRI Volumetrics
While Aβ imaging or CSF Aβ provides information in regard to Aβ pathology, CSF tau quantification, FDG, and structural MRI provide information related to the neurodegenerative process (Jack Jr. et al. 2010). Hippocampal and cortical gray matter atrophy and ventricular enlargement are typical, albeit not specific, MRI findings in AD and MCI (Frisoni et al. 2010; Drago et al. 2011). Furthermore, it has been shown that the rates of hippocampal atrophy might be predictive of conversion from MCI to AD (Jack Jr. et al. 2008a; b).
Aβ deposition is associated with regional cerebral atrophy as measured by MRI (Archer et al. 2006; Bourgeat et al. 2010; Chetelat et al. 2010a; b; Becker et al. 2011) and correlates with the rates of cerebral atrophy (Becker et al. 2011; Tosun et al. 2011). Moreover, the data suggest that the relationship between Aβ deposition and cortical atrophy is sequential, where Aβ deposition precedes synaptic dysfunction and neuronal loss (Chetelat et al. 2010a, b, Drzezga et al. 2011, Forster et al. 2012), and then manifested as structural changes (Becker et al. 2011). While some reports did not find a correlation between atrophy trajectories and Aβ deposition (Josephs et al. 2008; Driscoll et al. 2010), most reported that those with high Aβ deposition showed a significantly higher rate of cortical atrophy compared to those with low Aβ burden (Chetelat et al. 2012; Sarro et al. 2016; Fletcher et al. 2018; Dang et al. 2019). These discrepancies might be due to degree of tau deposition in the brain, which is believed to mediate the effects of Aβ on brain volumetrics (Ossenkoppele et al. 2019; La Joie et al. 2020).
6.5 Neuroinflammation
Microglia are the primary resident immune surveillance cells in the brain and are thought to play a significant role in the pathogenesis of several neurodegenerative disorders including AD (Venneti et al. 2006). Activated microglia can be measured using radioligands for the translocator protein (TSPO), formerly known as the peripheral benzodiazepine receptor. The TSPO is upregulated on activated macrophages and microglia and has been established as a biomarker of neuroinflammation in the central nervous system. Most reports found that in AD there is no association between Aβ deposition and microglial activation measured by [11C]PK11195 (Edison et al. 2008a; b; Okello et al. 2009a; b; Wiley et al. 2009) despite being an association between [11C]PK11195 binding and both glucose hypometabolism and severity of dementia (Edison et al. 2008a; b; Yokokura et al. 2011). Higher TSPO tracer binding is observed in AD patients compared to elderly controls (Kreisl et al. 2018), but TSPO polymorphism affects the binding of these tracers in the brain (Kreisl et al. 2013). New developed radiotracers for activated microglia are focusing on cell membrane markers like P2X7 receptors (Berdyyeva et al. 2019; Hagens et al. 2020), as well as renewed interest on tracers binding to COX-1 and COX-2 (Kim et al. 2018). Also, PET studies assessing the relationship between Aβ deposition and neuroinflammation have been conducted using the MAO-B inhibitor 11C-l-d-deprenyl as a marker for reactive astrocytosis (Carter et al. 2012; Rodriguez-Vieitez and Nordberg 2018). The most interesting issue in regard to imaging reactive astrocytosis is that, in contrast with activated microglia, it is an early phenomenon, suggesting it might be a better neuroinflammatory target at the preclinical and prodromal stages of AD (Scholl et al. 2015; Rodriguez-Vieitez et al. 2016).
7 Relation of Aβ Deposition with Genetic Risks and Predisposing Factors
Age is the strongest risk factor in sporadic AD, with the prevalence of the disease increasing exponentially with age. These risks are increased in the presence of the APOE ε4 allele (Farrer et al. 1997). Both risk factors have been directly associated with Aβ burden as measured by PET (Reiman et al. 2009a; b; Morris et al. 2010; Rowe et al. 2010).
To date, APOE ε4 is the most consistent genetic risk factor associated with sporadic AD, and its presence has been associated with an earlier age of onset and a dose-dependent—either one or two APOE ε4 alleles—higher risk of developing AD (Farrer et al. 1997). Examination of APOE ε4 allele status revealed that, independent of clinical classification, ε4 carriers present with significantly higher 11C-PiB retention than non-ε4 carriers, further emphasizing the crucial role that APOE plays in the metabolism of Αβ (Reiman et al. 2009a; b; Morris et al. 2010; Rowe et al. 2010). When specific clinical groups are examined, the results— from either autopsy or Αβ imaging studies— are not that clear-cut. While some studies found significant differences in Αβ burden between AD ε4 carriers and non-ε4 carriers (Gomez-Isla et al. 1996; Drzezga et al. 2008; Grimmer et al. 2010), others did not (Berg et al. 1998; Klunk et al. 2004; Rowe et al. 2007; Rabinovici et al. 2010).
Both symptomatic and asymptomatic individual carriers of autosomal mutations within the APP or presenilin 1 or 2 genes associated with familial AD present with very high and early 11C-PiB retention in the caudate nuclei, retention that seems to be independent of mutation type or disease severity (Klunk et al. 2007; Koivunen et al. 2008a; b; Remes et al. 2008; Villemagne et al. 2009a; b). Larger multicenter studies are helping elucidate the value of several biomarkers in these autosomal mutation carriers, assessment that might lead to better targeted disease-specific therapeutic strategies (Bateman et al. 2012; Benzinger et al. 2013).
While a similar pattern of Aβ deposition as seen in familial AD has been reported in adults with Down syndrome (Cohen et al. 2018), a completely different pattern of 11C-PiB retention was observed in a mutation carrier of familial British dementia (Villemagne et al. 2010).
There are several other factors that have been associated with AD. Among these, the most relevant are vascular risk factors (VRF), such as diabetes, hypercholesterolemia, hypertension, hyperhomocysteinemia, etc. The influence of these VRF on AD pathology is an area that, despite the long and intense scientific debate, has not been clearly established (Viswanathan et al. 2009). Studies using MRI white matter hyperintense lesions as surrogate markers of vascular disease in association with assessment of Aβ deposition and cognition (Kuczynski et al. 2008; Marchant et al. 2011) suggest that cerebrovascular lesions may accelerate the clinical presentation of AD (Riekse et al. 2004) by having an independent effect on cognition (Vemuri et al. 2017; DeCarli et al. 2019).
As with AD, VRF are strongly age-dependent factors that share similar pathogenic pathways, such as oxidative stress or inflammation, so it is not easy to extricate one from the other in order to ascertain a causal, casual, or comorbid association between them (Rosendorff et al. 2007). On the other hand, in light of the absence of disease-modifying therapies for AD, the relevance of these risk factors rests in the fact that early intervention, especially at middle age, might have an impact on delaying either the onset or progression of AD, as suggested by recent reports of a lowering incidence of AD (Satizabal et al. 2016; Pase et al. 2017).
8 Aβ Imaging in Other Neurodegenerative Conditions
Aβ imaging in a wide spectrum of neurodegenerative conditions has yielded diverse results. Although lower than in AD, similar patterns of 11C-PiB retention are usually observed in DLB (Rowe et al. 2007; Gomperts et al. 2008; Maetzler et al. 2009). Cortical 11C-PiB retention is also higher in subjects diagnosed with CAA (Johnson et al. 2007), while there is usually no cortical 11C-PiB retention in patients with FTLD (Rabinovici et al. 2007; Rowe et al. 2007; Drzezga et al. 2008; Engler et al. 2008). Aβ imaging has also facilitated differential diagnosis in cases of patients with atypical presentations of dementia (Ng et al. 2007a; b; Wolk et al. 2012a; b).
8.1 Cerebral Amyloid Angiopathy
CAA, characterized by Aβ deposits in and around the media of small arteries and arterioles of the cerebral cortex and leptomeninges, has been found to be present in most patients with AD (Jellinger and Attems 2005). To this date, neuropathologic examination of the brain remains the only definitive method for diagnostic confirmation of CAA. However, the combination of Aβ imaging with 11C-PiB PET and T2* susceptibility-weighted (SWI) MR imaging (Knudsen et al. 2001) allows for the concomitant assessment of molecular and structural changes in vivo. The combination of cerebral microhemorrhages (MH) and superficial siderosis (SS) has been suggested as a radiological marker for CAA (Feldman et al. 2008). Aβ imaging studies have shown both processes may be intimately associated with Aβ deposition (Dierksen et al. 2010; Dhollander et al. 2011), preferentially in posterior areas of the brain, showing a distinct pattern of 11C-PiB retention than the one observed in sporadic AD (Johnson et al. 2007). Furthermore, focal Aβ deposition might be useful in predicting new MH in CAA patients (Gurol et al. 2012; Raniga et al. 2017). While deep subcortical MH are generally associated with VRF, lobar MH (particularly posterior) (Pettersen et al. 2008) are usually attributed to CAA (Knudsen et al. 2001; Vernooij et al. 2008). Lobar MH are a frequent finding in AD patients or even in cognitively normal older individuals, and they are strongly associated with increasing age and Aβ deposition (Yates et al. 2011). This association between Aβ and vascular lesions has crucial implications not only for the selection and risk stratification of individuals undergoing anticoagulant therapies but also in those enrolled in anti-Aβ therapeutic trials (Weller et al. 2009).
8.2 Lewy Body Diseases
While AD is the most common cause of dementia in the elderly, DLB accounts for 20% of the cases (McKeith et al. 2005). The pathological hallmark of DLB is the presence of α-synuclein-containing LB within the neocortical and limbic regions (McKeith et al. 2005; Kantarci et al. 2020), as well as substantial loss of pigmented dopaminergic neurons in the substantia nigra, reflected in a marked dopaminergic terminal denervation in the striatum (McKeith and Mosimann 2004). More refined neuropathological techniques have revealed extensive synaptic deposits of α-synuclein, more concordant with the DLB clinical phenotype than the usually sparse cortical LB (Schulz-Schaeffer 2010). Overlap of cognitive symptoms early in the disease course offers a challenge for clinicians making it difficult to distinguish between patients that will develop AD and those that will ultimately develop DLB. Post-mortem histopathological studies have identified that 50–80% of DLB cases often have cortical Aβ deposits with characteristics and a distribution similar to AD patients (McKeith et al. 2005; Rowe et al. 2007; Edison et al. 2008a; b; Kantarci et al. 2020). This has also been described as “mixed” DLB/AD or “Lewy body variant of AD.” So-called “pure” DLB is much less common, and it is not clear if the clinical characteristics and prognosis differ from the mixed-pathology cases. On visual inspection, cortical Aβ tracer retention in DLB is generally lower and more variable than in AD, probably reflecting a larger spectrum of Αβ deposition (Rowe et al. 2007; Gomperts et al. 2008; Maetzler et al. 2009). Therefore Aβ imaging is not a useful tool in differentiating between AD and DLB, but might assist in the differential diagnosis of those cases (“pure” DLB) without Aβ deposition in the brain (Armstrong et al. 2000; Landau and Villemagne 2020).
Taking advantage of its high affinity for α-synuclein (Fodero-Tavoletti et al. 2009a; b), 11C-BF227 has been used for the assessment of multiple system atrophy (MSA), a disease characterized by the glial accumulation of α-synuclein in the absence of Aβ plaques, showing a higher retention in MSA patients compared to age-matched controls (Kikuchi et al. 2010). Interestingly, while 11C-BF227 retention in DLB patients was lower compared to AD in most cortical regions, DLB presented with significantly higher 11C-BF227 retention in the amygdala (Furukawa et al. 2011).
In contrast to Aβ imaging, assessing the integrity of dopaminergic terminals via dopamine transporter (DAT) or vesicular monoamine transporter type 2 (VMAT2) imaging can robustly detect reductions of dopaminergic nigrostriatal afferents in DLB patients, assisting in the differential diagnosis from AD (O'Brien et al. 2004; Koeppe et al. 2008; Villemagne et al. 2012a; b; c; d; Gomperts et al. 2016).
Cortical Aβ is not usually present in cognitively intact Parkinson’s disease (PD) patients, and no significant 11C-PiB cortical retention is usually observed in these patients (Johansson et al. 2008; Mashima et al. 2017). Aβ deposits, both vascular and parenchymal, are frequent, however, in PD patients who develop dementia (PDD) (Edison et al. 2008a; b; Gomperts et al. 2008; Maetzler et al. 2008; Maetzler et al. 2009; Burack et al. 2010; Foster et al. 2010; Kalaitzakis et al. 2011; Gomperts et al. 2016).
The contribution of Aβ to the development of Lewy body diseases remains unclear, but cortical Aβ deposits are associated with extensive α-synuclein lesions and higher levels of insoluble α-synuclein lesions (Pletnikova et al. 2005), as well as exacerbation of neuronal injury (Masliah et al. 2001). Furthermore, it has been shown that in PD, CSF Aβ is predictive of cognitive decline (Siderowf et al. 2010). Along these lines, while Aβ imaging cannot contribute to the differential diagnosis between AD and DLB, it may have prognostic relevance in these Lewy body diseases. For example, similar to what has been reported for PDD, where higher Αβ burden was associated with a shorter prodromal phase before dementia (Halliday et al. 2008), Αβ burden in DLB patients was inversely correlated with the interval from onset of cognitive impairment to the full development of the DLB phenotype, faster rates of cortical atrophy, and faster rates of clinical progression (Rowe et al. 2007; Sarro et al. 2016; Donaghy et al. 2019).
8.3 Frontotemporal Lobar Degeneration
FTLD is a syndrome that can also be clinically difficult to distinguish from early-onset AD, especially at the initial stages of the disease. Based on clinical phenotype, FTLD has been categorized mainly into two classes: behavioral variant of FTLD (bvFTLD) with distinct changes in behavior and personality with little effect on language functions and progressive aphasias (comprised by semantic dementia (svFTLD), progressive non-fluent aphasia (nfvFTLD), and logopenic aphasia (lvFTLD)) showing progressive language deficits, with less obvious personality and behavioral changes (Rabinovici & Miller 2010).
Aβ deposition is not a pathological trait of FTLD, and definitive differential diagnosis can only be established after post-mortem examination of the human brain. Neuropathological examination of the brain in FTLD shows variable frontal and temporal atrophy (Cairns et al. 2007; Hodges and Patterson 2007; Snowden et al. 2007), microvacuolation, and neuronal loss, with white matter myelin loss, astrocytic gliosis, as well as neuronal and glial inclusions (Cairns et al. 2007). Three intraneuronal inclusion types have been identified in FTLD: (1) hyperphosphorylated tau (FTLD-tau), (2) ubiquitin-hyperphosphorylated and proteolysed TAR-DNA binding protein 43 (FTLD-TDP-43), and (3) fused sarcoma positive (FUS) (Taniguchi et al. 2004; Mott et al. 2005; Neumann et al. 2006; Neumann et al. 2009). Tau-positive inclusions account for approximately 40% of all FTLD cases, the remainder being tau negative and TDP-43/ubiquitin positive (Josephs et al. 2004). In bvFTLD, both tau and TDP-43 are equally prevalent; and while 90% of the SD cases show TDP-43 pathology, PNFA presents predominantly (70%) tau pathology (Josephs et al. 2004; Mackenzie et al. 2008). In contrast, logopenic progressive aphasia (LPA) is thought to be a language presentation of AD, with typical Aβ and NFT (Leyton et al. 2011). Absence of Aβ and tau deposition is considered one of the features of limbic-predominant age-related TDP-43 encephalopathy (LATE) (Nelson et al. 2019), a neurodegenerative condition that presents with an amnestic dementia similar to AD, which also presents a typical signature on FDG studies (Botha et al. 2018).
Aβ imaging has been helpful in the differential diagnosis between FTLD and AD (Rabinovici et al. 2007; Rowe et al. 2007; Drzezga et al. 2008; Engler et al. 2008). Furthermore, Aβ imaging has been used to ascertain the absence of AD pathology in the different FTLD aphasias (Drzezga et al. 2008; Rabinovici et al. 2008; Leyton et al. 2011). Despite displaying similar specificities, the diagnostic performance of Aβ imaging proved to be more sensitive for the diagnosis of FTLD than FDG (Rabinovici et al. 2011).
8.4 Prion Diseases
No cortical Aβ deposition is usually observed in sporadic Creutzfeldt-Jakob disease (CJD) cases (Villemagne et al. 2009a; b; Dulamea and Solomon 2016; Matias-Guiu et al. 2017). Due to the rapid progression and relatively short duration of symptomatic illness, sporadic CJD patients do not demonstrate appreciable PrP-plaque deposition at autopsy (Liberski 2004). Although some sporadic CJD molecular subtypes can manifest larger PrP plaques (Hill et al. 2003), the most common subtype usually demonstrates only very fine “synaptic” and small perivacuolar deposits (Hill et al. 2003; Liberski 2004). A study in transmissible spongiform encephalopathies showed that Aβ imaging with 11C-BF227 might be able to distinguish between Gerstmann-Straussler-Scheinker disease (GSS) and sporadic CJD disease with clear tracer retention in brain regions of PrP deposition (Okamura et al. 2010).
8.5 Traumatic Brain Injury and Chronic Traumatic Encephalopathy
Traumatic brain injury (TBI) has been associated with a number of long-term sequelae, including an increased risk for dementia (Plassman et al. 2000; Barnes et al. 2014); however, the long-term effects of a single TBI event in regard to triggering AD-related proteinopathies remain controversial (Furst and Bigler 2016; LoBue et al. 2019). While some studies have reported higher cortical Aβ deposition in TBI (Malkki 2014; Gatson et al. 2016), others have found no evidence of it (Weiner et al. 2017). These discrepancies might be in part explained by slightly different definitions of TBI, as well as in the cause or severity of the event. A more careful selection of and better clinical and neuropsychological characterization of participants is needed to elucidate this issue.
Chronic traumatic encephalopathy (CTE) is a neurodegenerative 3R/4R tauopathy associated with a history of repetitive head trauma (McKee et al. 2018). However, in about 50% of CTE cases, other pathological protein deposits such as Aβ, α-synuclein, and TDP-43 are also present (McKee et al. 2013), which, added to a variable degree of cortical gray matter atrophy, make the interpretation of molecular imaging studies complicated.
9 Clinical and Research Applications of Amyloid Imaging
Aβ imaging has been widely used in research settings as well as in a host of disease-specific therapeutic trails while ascertaining its place as a diagnostic and prognostic tool in the clinical evaluation and management of dementia patients.
The application of Aβ imaging in research and clinical settings raises issues referring to the quantitative or semiquantitative approaches used to determine Aβ burden. Given that the studies are performed in individuals or populations who might not tolerate long scanning sessions, the majority of studies use a short 20- or 30-min PET scan when Aβ tracers reach, at different periods after injection, apparent steady state (Lopresti et al. 2005; Rowe et al. 2007; Yee et al. 2007; Edison et al. 2008a; b). While these image acquisition intervals are more than enough to obtain clinically useful images, it is unlikely they adequately or reliably establish Aβ burden in therapeutic trials (Rinne et al. 2010; Ostrowitzki et al. 2011; Ossenkoppele et al. 2012), where drug-induced changes in tracer metabolism, blood-brain barrier permeability, cerebral blood flow, gray matter atrophy, target conformation, etc. might affect Aβ tracer binding (Lopresti et al. 2005; Price et al. 2005). Finally, the issue of partial volume correction (PVC) raises several other questions: How much of the intrinsic noise of the PET signal is augmented by PVC? Should longitudinal studies be evaluated without PVC (Jack Jr. et al. 2013; Villemagne et al. 2013; Landau et al. 2015)? How to correct for spillover radioactivity from white matter? All these issues remain contentious.
9.1 Aβ Imaging in Large Observational Cohorts
Since the launch of the Alzheimer’s Disease Neuroimaging Initiative (ADNI https://adni.loni.usc.edu) in the USA in 2003 ever-growing, similarly oriented consortia such as the Australian Imaging, Biomarker and Lifestyle (AIBL, https://aibl.csiro.au) Study of Ageing (Ellis et al. 2009) and Europe’s Amyloid Imaging to Prevent Alzheimer’s Disease (AMYPAD, http://amypad.eu) (Frisoni et al. 2019) have been organized and assembled around the world, setting the groundwork for biomarker discovery and the establishing of standards for early diagnosis of sporadic AD (Villemagne et al. 2014). A similar worldwide consortium, the Dominantly Inherited Alzheimer’s Network (DIAN, https://dian.wustl.edu) (Morris et al. 2012), focuses on autosomal mutations leading to AD. The various accomplishments of these large cohort observational studies, especially in regard to Aβ imaging, have contributed substantially to a better understanding of the underlying physiopathology of aging and AD. These accomplishments are basically predicated in the trinity of multimodality, standardization, and sharing. This multimodality approach can now better identify those subjects with AD-specific traits that are more likely to present cognitive decline in the near future and that might represent the best candidates for smaller but more efficient therapeutic trials, trials that, by the gained and shared knowledge of some of the pathophysiological mechanisms underpinning aging and AD pathology, can be more focused on a specific target or a specific stage of the disease process.
9.2 Aβ Imaging in Disease-Specific Therapeutic Trials
Aβ imaging with PET is also contributing to the development of more effective therapies by allowing better selection of patients for multicenter anti-Aβ therapy trials around the world (Long and Holtzman 2019) and providing a means to ascertain target engagement and measure their impact on Aβ burden (Rinne et al. 2010; Ostrowitzki et al. 2011; Liu et al. 2015) as well as outcome measure of their effectiveness (Doody et al. 2014; Salloway et al. 2014; Sevigny et al. 2016).
While these studies represent one of the principal applications of Aβ imaging today, a key challenge for these trials is that reductions in Aβ burden, as measured by either CSF or PET, have not been accompanied with cognitive stabilization or improvement (Doody et al. 2014; Salloway et al. 2014; Sevigny et al. 2016). Furthermore, some of these therapies were associated with side effects such as edema and microhemorrhages. Other Aβ-centered therapeutic mechanisms have been attempted, such as inhibition or modulation of β- and γ-secretases, but most have been discontinued as well (Egan et al. 2019, Long and Holtzman 2019). Since study participants were, while mild, symptomatic AD patients, these setbacks have been attributed to treatment occurring too late in the course of disease, so trials like A4 have enrolled cognitively normal elderly individuals with high Aβ, aiming at preventing the cognitive decline expected in this cohort (Sperling et al. 2014). Others have argued that these failures are clear evidence that Aβ reduction will not be an effective strategy for treating AD and that Aβ as a target for treatment or preventing AD should be abandoned.
The contribution of Aβ imaging to the selection of patients for therapeutic trials needs to be complemented with adequate MRI sequences such as SWI or GRE for the detection of vascular pathology to rule out the possibility of vascular complications such as Aβ imaging-related abnormalities (ARIA), either as “vasogenic edema” and/or sulcal effusion (ARIA-E) or as hemosiderin deposits including MH and SS (ARIA-H) (Cordonnier 2010; Sperling et al. 2011a; b; c). The presence of MH is impacting on patient selection in therapeutic trials, and some anti-Aβ immunotherapy trials are already excluding individuals with MH. To make matters worse, about one third of non-demented individuals with high Aβ burden and no prior history of cerebrovascular disease show MH (Yates et al. 2011). While focal Aβ deposition might be a better predictor of a vascular event (Gurol et al. 2012), only longitudinal follow-up of large cohorts of patients will allow elucidation of the relevant risk factors for lobar MH.
9.3 The Centiloid Scale
One of the obstacles for the widespread implementation of Aβ imaging in therapeutic trials or comparison of results in different cohorts across different centers is that Aβ tracers present with differing pharmacological and pharmacokinetic properties, yielding results in different dynamic ranges. These issues, summed to the use of a diverse arsenal of quantitative approaches and contrasting criteria for the selection of internal scaling region and different thresholds to separate high from low Aβ burden in the brain, pose a challenge when trying to compare results from different cohorts or different therapeutic trials that use different Aβ tracers as Aβ quantitative tools. Therefore, a method was developed to standardized all Aβ imaging results under a single common semiquantitative scale—called the Centiloid (Klunk et al. 2015)—to improve the clinical and research application of these Aβ tracers. Since then, all F-18-labeled radiotracers Aβ tracers currently in use have been cross-calibrated against PiB (Rowe et al. 2016; Rowe et al. 2017; Battle et al. 2018; Navitsky et al. 2018) to enable translation into Centiloids.
The other obstacle is to define a threshold. One of the issues is in regard to the utility of a cut-off given the continuous nature of Aβ deposition (Jagust 2011; Mormino et al. 2012). While thresholds are arbitrary, in order to adopt one, it needs to be shown that it is relevant and accurate from a diagnostic and/or prognostic point of view. From a clinical point of view, a visual binary read will help separate those with a significant Aβ burden in the brain from those with a null or low Aβ burden. Similar dilemmas arise in research settings. In order to avoid being trapped in this dichotomy, some researchers use tertiles (Resnick et al. 2010; Rowe et al. 2013a; b), while others are proposing to distinguish two thresholds for “positivity,” one more sensitive, to detect the early stages of Aβ deposition usually in still cognitively unimpaired elderly individuals, and another, a higher one, that can more specifically ascribe the clinical phenotype of dementia to Aβ pathology (Klunk et al. 2012).
The introduction of the Centiloid scale has tried to solve this problem, and while it transforms all Aβ tracers’ semiquantitative results into a single universal scale, the idiosyncratic properties of these Aβ tracers remain so they might be more or less sensitive or accurate to provide a similar statement to a similar Aβ burden in the brain. Most of the studies have used neuropathology to establish Centiloid thresholds. For example, a large multicenter study using neuropathology from pre-mortem 11C-PiB studies found that below a threshold of 12.2 Centiloids can distinguish between plaques and no plaques, while a threshold of 24.4 Centiloids identified intermediate-to-high AD neuropathological changes (La Joie et al. 2018). Similar neuropathology-based thresholds were determined with other Aβ tracers such as 18F-florbetaben yielding 21 Centiloids (Dore et al. 2019) or 18F-florbetapir yielding 24 Centiloids (Navitsky et al. 2018).
9.4 Appropriate Use Criteria and Clinical Impact of Aβ Imaging
Also, clinical criteria for the appropriate use of Aβ imaging has been established highlighting the need to integrate Aβ imaging with a comprehensive clinical and cognitive evaluation performed by a clinician experienced in the evaluation of dementia, ensuring a positive impact on patient management (Johnson et al. 2013). These criteria clearly stipulate the specific cases Aβ imaging should be used, such as patients with persistent or progressive unexplained cognitive impairment, progressive atypical or unclear clinical presentation of dementia, or dementia onset in individuals 65 years or younger (Johnson et al. 2013; Apostolova et al. 2016). It also outlines the inappropriate use of Aβ imaging, such as cases of probable AD with typical age of onset or to determine dementia severity, asymptomatic individuals or unconfirmed cognitive complaint, a family history of dementia or presence of the APOE ε4 allele, as well as non-medical use such as litigation for insurance purposes (Johnson et al. 2013; Apostolova et al. 2016).
These criteria are being tested in multicentre trials such as the Imaging Dementia—Evidence for Amyloid Scanning (IDEAS, https://www.ideas-study.org), assessing the clinical utility of Aβ imaging in the workup of patients with MCI or dementia of uncertain origin. The initial report from this trial showed that results from Aβ imaging were associated with more than a 60% change in the clinical management of these patients within 90 days, usually reflected in a change in acetylcholine esterase inhibitor medication (Rabinovici et al. 2019). Overall, Aβ imaging leads to change in diagnostic confidence and patient management in up to 60% of cases attending memory clinics (de Wilde et al. 2018; Rabinovici et al. 2019).
10 CODA
The clinical diagnosis of AD is typically based on progressive cognitive impairments while excluding other diseases. Clinical diagnosis of sporadic disease is challenging, however, often presenting mild and non-specific symptoms attributable to diverse and overlapping pathology presenting similar phenotypes. Overall, the accuracy of clinical diagnosis of AD compared to neuropathological examination ranges between 70% and 90% (Beach et al. 2012). While clinical criteria, together with current structural neuroimaging techniques (CT or MRI), are sufficiently sensitive and specific for the diagnosis of AD at the mid to late stages of the disease, they are focused on relatively non-specific features such as memory and functional activity decline or brain atrophy that are late features in the course of the disease. Confirmation of diagnosis of AD still relies on autopsy. But how reliable is pathology? How much of a “gold standard” is autopsy (Jellinger 2010; Scheltens and Rockwood 2011)? A more sensible paradigm, adopting a more fluid model that integrates all cognitive, biochemical, and imaging biomarkers that can provide a better predictive framework, has been proposed in the new diagnostic criteria for AD (Dubois et al. 2010; Sperling and Johnson 2010; McKhann et al. 2011), MCI (Albert et al. 2011), and preclinical AD (Sperling et al. 2011a; b; c). Along the same lines, Scheltens and Rockwood suggest that “a shift in focus from why people with AD have plaques and tangles to why not all people with plaques and tangles have AD would be welcome” (Scheltens and Rockwood 2011). While most people with AD pathology develop AD dementia, there are some that while having the pathology do not. What makes them different? What conveys resistance to some or, conversely, what makes others more vulnerable to the same pathological burden? Identifying these factors will allow a more discerning selection of individuals that will benefit from early intervention with anti-Aβ medication while also providing insights into potential preventive strategies or even new therapeutic approaches (Sojkova and Resnick 2011; Sperling et al. 2011a; b; c). While the new diagnostic criteria has blurred once again the boundaries between MCI and AD (Morris 2012), it also allows for earlier diagnosis and therapeutic intervention, arguing that when there is a clear history of progressive cognitive decline, objective evidence from psychometric tests of episodic memory impairment and characteristic abnormalities in the CSF, and/or neuroimaging studies such as Aβ imaging, dementia is no more required for the diagnosis of probable AD (Dubois et al. 2010; Sperling and Johnson 2010; McKhann et al. 2011; Jack Jr. et al. 2016; Jack et al. 2018). Thus, as the criteria for the diagnosis of AD changes, Aβ imaging is likely, given its impact on patient management (Rabinovici et al. 2019), to play an increasingly important role in clinical practice provided it is accessible and affordable (Villemagne et al. 2010).
At this stage Aβ imaging, in combination with other modalities, is probably the best diagnostic tool to rule out Aβ pathology in individuals being evaluated for cognitive decline. The data to date clearly show that Aβ production and deposition is not a benign process with a more prominent role at the early, mostly asymptomatic, stages of the disease (Hyman 2011; Karran et al. 2011); therefore, anti-Aβ therapy at these stages seems a logical approach (Sperling et al. 2011a, b, c). On the other hand, we still do not have the full grasp of the long-term cognitive consequences of Aβ deposition. Longitudinal Aβ imaging, especially of cognitively unimpaired elderly individuals, is allowing the assessment of pathology almost simultaneously with tau, cognition, and other biomarkers of neurodegeneration. This will provide not only a better picture of how these different factors interact but also the weight or relevance this complementary and/or supplementary information plays at different stages of the disease.
The pathological process usually begins decades before symptoms are evident, making early identification difficult. This in turn precludes early intervention with disease-modifying medications during the presymptomatic period, which by arresting neuronal loss would presumably achieve the maximum benefits of such therapies (Villemagne et al. 2008a; b; Sperling et al. 2011a; b; c). Therefore, a change in the diagnostic paradigm is warranted, where diagnosis moves away from identification of signs and symptoms of neuronal failure—indicating that central compensatory mechanisms have been exhausted and extensive synaptic and neuronal damage is present—to the non-invasive detection of specific biomarkers for particular traits underlying the pathological process (Clark et al. 2008). Given the complexity and sometimes overlapping characteristics of these disorders, and despite recent advances in molecular neurosciences, it is unlikely that a single biomarker will be able to provide the diagnostic certainty required for the early detection of neurodegenerative diseases like AD, especially for the identification of at-risk individuals before the development of the typical phenotype. Therefore, a multimodality approach combining biochemical and neuroimaging methods is warranted (Shaw et al. 2007).
Biomarkers are important as they can be used to identify the risk of developing a disease (antecedent biomarkers), aid in identifying disease (diagnostic biomarkers), assess disease evolution (progression biomarkers), predict future disease course (prognostic biomarkers), or evaluate and/or customize therapy (theranostic biomarkers). While CSF Aβ and tau, structural, and molecular neuroimaging (both FDG and Aβ imaging) are all good—specially if combined—diagnostic biomarkers for AD, evaluation of the data available to date suggests that Aβ imaging and CSF assessments of Aβ are good antecedent biomarkers at the preclinical and prodromal stages of the disease (Jack Jr. et al. 2010; Ossenkoppele et al. 2011; Villemagne et al. 2011a; b). Conversely, CSF assessments of tau and p-tau, structural neuroimaging, and FDG seem to be excellent biomarkers of disease progression (Jack Jr. et al. 2010). Despite the fact that Aβ accumulation is still ongoing at the AD stage (Villemagne et al. 2011a; b; Villain et al. 2012), and in contrast to what is observed at the preclinical and prodromal phases (Pike et al. 2007; Villemagne et al. 2008a; b; Cohen et al. 2012), Aβ burden at this stage is not correlated with markers of neurodegeneration, disease severity, or cognitive impairment (Hyman 2011; Karran et al. 2011). A review of the literature shows that quantification of Aβ1–42, total tau, and p-tau in CSF and FDG and a long list of assessments of global or regional brain atrophy as well as white matter hyperintensities with MRI have all been proposed as predictors of conversion to AD (Chang and Silverman 2004; Smith et al. 2008; Jack Jr. et al. 2010). While larger longitudinal studies will further clarify the role of Aβ imaging in predicting conversion from MCI to AD, one of the most important conclusions to be drawn from these studies is that the likelihood of developing AD in a cognitively unimpaired individual with a low Aβ burden scan is extremely small (Villemagne et al. 2011a; b).
Another aspect to be considered is prediction of therapeutic response. As new therapies enter clinical trials, the role of Aβ imaging in vivo is becoming increasingly crucial. Αβ imaging allows the in vivo assessment of brain Αβ pathology and its changes over time, providing highly accurate, reliable, and reproducible quantitative statements of regional or global Aβ burden in the brain. As mentioned before, therapies, especially those targeting irreversible neurodegenerative processes, have a better chance to succeed if applied early, making early detection of the underlying pathological process so important. Therefore, Aβ imaging is an ideal tool for the selection of adequate candidates for anti-Aβ therapeutic trials while also monitoring—and potentially predicting—treatment response, thus aiding in reducing sample size and minimizing cost while maximizing outcomes. Studies are already showing the utility of this technique (Rinne et al. 2010; Ostrowitzki et al. 2011; Liu et al. 2015). These results, taken together with the results from longitudinal studies, support the growing consensus that anti-Aβ therapy to be effective may need to be given early in the course of the disease, perhaps even before symptoms appear (Sperling et al. 2011a, b, c), and that downstream mechanisms may also need to be addressed to successfully prevent the development of AD. Furthermore, the slow rate of Aβ deposition indicates that the time window for altering Aβ accumulation prior to the full manifestation of the clinical phenotype may be very wide (Villemagne et al. 2011a, b).
The introduction of radiotracers for the non-invasive in vivo quantification of Αβ burden in the brain has revolutionized the approach to the evaluation of AD. Αβ imaging has extended our insight into Aβ deposition in the brain, where Aβ burden as measured by PET matches histopathological reports of Αβ distribution in aging and dementia, it appears more accurate than FDG for the diagnosis of AD, and it is an excellent aid in the differential diagnosis of AD from FTLD (Mathis et al. 2005; Johnson 2006; Mathis et al. 2007; Villemagne et al. 2008a; b; Jagust 2009; Rabinovici and Jagust 2009; Drzezga 2010; Klunk 2011; Villemagne et al. 2012a; b; c; d). APOE ε4 carriers, independent of diagnosis or disease severity, present with higher Aβ burden than non-ε4 carriers, and high retention of Aβ radiotracers closely corresponds with low CSF Aβ levels and increasing age (Reiman et al. 2009a; b; Fagan and Holtzman 2010; Morris et al. 2010; Rowe et al. 2010). Although Αβ burden as assessed by PET does not strongly correlate with cognitive impairment in AD, it does correlate with memory impairment and a higher risk for cognitive decline in the aging population and MCI subjects. This correlation with memory impairment, one of the earliest symptoms of AD, suggests that Αβ deposition is not part of normal aging, supporting the hypothesis that Αβ deposition occurs well before the onset of symptoms and it likely represents preclinical AD in asymptomatic individuals and prodromal AD in MCI. Further longitudinal observations, coupled with different disease-specific biomarkers to assess potential downstream effects of Aβ, are required to confirm this hypothesis and further elucidate the role of Αβ deposition in the course of Alzheimer’s disease.
References
Agdeppa ED, Kepe V, Liu J, Flores-Torres S, Satyamurthy N, Petric A, Cole GM, Small GW, Huang SC, Barrio JR (2001a) Binding characteristics of radiofluorinated 6-dialkylamino-2-naphthylethylidene derivatives as positron emission tomography imaging probes for beta-amyloid plaques in Alzheimer’s disease. J Neurosci 21(24):RC189
Agdeppa ED, Kepe V, Shoghi-Jadid K et al (2001b) In vivo and in vitro labeling of plaques and tangles in the brain of an Alzheimer’s disease patient: a case study. J Nucl Med 42(suppl 1):65
Aizenstein HJ, Nebes RD, Saxton JA, Price JC, Mathis CA, Tsopelas ND, Ziolko SK, James JA, Snitz BE, Houck PR, Bi W, Cohen AD, Lopresti BJ, Dekosky ST, Halligan EM, Klunk WE (2008) Frequent amyloid deposition without significant cognitive impairment among the elderly. Arch Neurol 65(11):1509–1517
Albert MS, Dekosky ST, Dickson D, Dubois B, Feldman HH, Fox NC, Gamst A, Holtzman DM, Jagust WJ, Petersen RC, Snyder PJ, Carrillo MC, Thies B, Phelps CH (2011) The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement 7(3):270–279
Apostolova LG, Haider JM, Goukasian N, Rabinovici GD, Chetelat G, Ringman JM, Kremen S, Grill JD, Restrepo L, Mendez MF, Silverman DH (2016) Critical review of the appropriate use criteria for amyloid imaging: effect on diagnosis and patient care. Alzheimers Dement (Amst) 5:15–22
Apostolova LG, Hwang KS, Andrawis JP, Green AE, Babakchanian S, Morra JH, Cummings JL, Toga AW, Trojanowski JQ, Shaw LM, Jack CR Jr, Petersen RC, Aisen PS, Jagust WJ, Koeppe RA, Mathis CA, Weiner MW, Thompson PM (2010) 3D PIB and CSF biomarker associations with hippocampal atrophy in ADNI subjects. Neurobiol Aging 31(8):1284–1303
Archer HA, Edison P, Brooks DJ, Barnes J, Frost C, Yeatman T, Fox NC, Rossor MN (2006) Amyloid load and cerebral atrophy in Alzheimer’s disease: an 11C-PIB positron emission tomography study. Ann Neurol 60(1):145–147
Armstrong RA, Cairns NJ, Lantos PL (2000) Beta-amyloid deposition in the temporal lobe of patients with dementia with Lewy bodies: comparison with non-demented cases and Alzheimer's disease. Dement Geriatr Cogn Disord 11(4):187–192
Arnold SE, Han LY, Clark CM, Grossman M, Trojanowski JQ (2000) Quantitative neurohistological features of frontotemporal degeneration. Neurobiol Aging 21(6):913–919
Backman L, Jones S, Berger AK, Laukka EJ, Small BJ (2005) Cognitive impairment in preclinical Alzheimer's disease: a meta-analysis. Neuropsychology 19(4):520–531
Bacskai BJ, Frosch MP, Freeman SH, Raymond SB, Augustinack JC, Johnson KA, Irizarry MC, Klunk WE, Mathis CA, Dekosky ST, Greenberg SM, Hyman BT, Growdon JH (2007) Molecular imaging with Pittsburgh compound B confirmed at autopsy: a case report. Arch Neurol 64(3):431–434
Barnes DE, Kaup A, Kirby KA, Byers AL, Diaz-Arrastia R, Yaffe K (2014) Traumatic brain injury and risk of dementia in older veterans. Neurology 83(4):312–319
Barrio JR, Huang SC, Cole G, Satyamurthy N, Petric A, Phelps ME, Small G (1999) PET imaging of tangles and plaques in Alzheimer disease with a highly hydrophobic probe. J Label Compd Radiopharm 42:S194–S195
Barrio JR, Small GW, Wong KP, Huang SC, Liu J, Merrill DA, Giza CC, Fitzsimmons RP, Omalu B, Bailes J, Kepe V (2015) In vivo characterization of chronic traumatic encephalopathy using [F-18]FDDNP PET brain imaging. Proc Natl Acad Sci U S A 112(16):E2039–E2047
Barthel H, Gertz HJ, Dresel S, Peters O, Bartenstein P, Buerger K, Hiemeyer F, Wittemer-Rump SM, Seibyl J, Reininger C, Sabri O (2011) Cerebral amyloid-beta PET with florbetaben ((18)F) in patients with Alzheimer's disease and healthy controls: a multicentre phase 2 diagnostic study. Lancet Neurol 10(5):424–435
Bateman RJ, Munsell LY, Morris JC, Swarm R, Yarasheski KE, Holtzman DM (2006) Human amyloid-beta synthesis and clearance rates as measured in cerebrospinal fluid in vivo. Nat Med 12(7):856–861
Bateman RJ, Xiong C, Benzinger TL, Fagan AM, Goate A, Fox NC, Marcus DS, Cairns NJ, Xie X, Blazey TM, Holtzman DM, Santacruz A, Buckles V, Oliver A, Moulder K, Aisen PS, Ghetti B, Klunk WE, McDade E, Martins RN, Masters CL, Mayeux R, Ringman JM, Rossor MN, Schofield PR, Sperling RA, Salloway S, Morris JC (2012) Clinical and biomarker changes in dominantly inherited Alzheimer's disease. N Engl J Med 367(9):795–804
Battle MR, Pillay LC, Lowe VJ, Knopman D, Kemp B, Rowe CC, Dore V, Villemagne VL, Buckley CJ (2018) Centiloid scaling for quantification of brain amyloid with [(18)F]flutemetamol using multiple processing methods. EJNMMI Res 8(1):107
Bauer M, Langer O, Dal-Bianco P, Karch R, Brunner M, Abrahim A, Lanzenberger R, Hofmann A, Joukhadar C, Carminati P, Ghirardi O, Piovesan P, Forloni G, Corrado ME, Lods N, Dudczak R, Auff E, Kletter K, Muller M (2006) A positron emission tomography microdosing study with a potential antiamyloid drug in healthy volunteers and patients with Alzheimer's disease. Clin Pharmacol Ther 80(3):216–227
Beach TG, Monsell SE, Phillips LE, Kukull W (2012) Accuracy of the clinical diagnosis of Alzheimer disease at National Institute on Aging Alzheimer disease centers, 2005–2010. J Neuropathol Exp Neurol 71(4):266–273
Becker JA, Hedden T, Carmasin J, Maye J, Rentz DM, Putcha D, Fischl B, Greve DN, Marshall GA, Salloway S, Marks D, Buckner RL, Sperling RA, Johnson KA (2011) Amyloid-beta associated cortical thinning in clinically normal elderly. Ann Neurol 69(6):1032–1042
Benzinger TL, Blazey T, Jack CR Jr, Koeppe RA, Su Y, Xiong C, Raichle ME, Snyder AZ, Ances BM, Bateman RJ, Cairns NJ, Fagan AM, Goate A, Marcus DS, Aisen PS, Christensen JJ, Ercole L, Hornbeck RC, Farrar AM, Aldea P, Jasielec MS, Owen CJ, Xie X, Mayeux R, Brickman A, McDade E, Klunk W, Mathis CA, Ringman J, Thompson PM, Ghetti B, Saykin AJ, Sperling RA, Johnson KA, Salloway S, Correia S, Schofield PR, Masters CL, Rowe C, Villemagne VL, Martins R, Ourselin S, Rossor MN, Fox NC, Cash DM, Weiner MW, Holtzman DM, Buckles VD, Moulder K, Morris JC (2013) Regional variability of imaging biomarkers in autosomal dominant Alzheimer's disease. Proc Natl Acad Sci U S A 110(47):E4502–E4509
Berdyyeva T, Xia C, Taylor N, He Y, Chen G, Huang C, Zhang W, Kolb H, Letavic M, Bhattacharya A, Szardenings AK (2019) PET imaging of the P2X7 Ion Channel with a novel tracer [(18)F]JNJ-64413739 in a Rat model of Neuroinflammation. Mol Imaging Biol 21(5):871–878
Berg L, McKeel DW Jr, Miller JP, Storandt M, Rubin EH, Morris JC, Baty J, Coats M, Norton J, Goate AM, Price JL, Gearing M, Mirra SS, Saunders AM (1998) Clinicopathologic studies in cognitively healthy aging and Alzheimer's disease: relation of histologic markers to dementia severity, age, sex, and apolipoprotein E genotype. Arch Neurol 55(3):326–335
Bischof GN, Rodrigue KM, Kennedy KM, Devous MD Sr, Park DC (2016) Amyloid deposition in younger adults is linked to episodic memory performance. Neurology 87(24):2562–2566
Blennow K, Zetterberg H, Minthon L, Lannfelt L, Strid S, Annas P, Basun H, Andreasen N (2007) Longitudinal stability of CSF biomarkers in Alzheimer's disease. Neurosci Lett 419(1):18–22
Botha H, Mantyh WG, Murray ME, Knopman DS, Przybelski SA, Wiste HJ, Graff-Radford J, Josephs KA, Schwarz CG, Kremers WK, Boeve BF, Petersen RC, Machulda MM, Parisi JE, Dickson DW, Lowe V, Jack CR Jr, Jones DT (2018) FDG-PET in tau-negative amnestic dementia resembles that of autopsy-proven hippocampal sclerosis. Brain 141(4):1201–1217
Bourgeat P, Chetelat G, Villemagne VL, Fripp J, Raniga P, Pike K, Acosta O, Szoeke C, Ourselin S, Ames D, Ellis KA, Martins RN, Masters CL, Rowe CC, Salvado O (2010) Beta-amyloid burden in the temporal neocortex is related to hippocampal atrophy in elderly subjects without dementia. Neurology 74(2):121–127
Braak H, Braak E (1997) Frequency of stages of Alzheimer-related lesions in different age categories. Neurobiol Aging 18(4):351–357
Bresjanac M, Smid LM, Vovko TD, Petric A, Barrio JR, Popovic M (2003) Molecular-imaging probe 2-(1-[6-[(2-fluoroethyl)(methyl) amino]-2-naphthyl]ethylidene) malononitrile labels prion plaques in vitro. J Neurosci 23(22):8029–8033
Buckley RF, Maruff P, Ames D, Bourgeat P, Martins RN, Masters CL, Rainey-Smith S, Lautenschlager N, Rowe CC, Savage G, Villemagne VL, Ellis KA, AIBL study (2016) Subjective memory decline predicts greater rates of clinical progression in preclinical Alzheimer's disease. Alzheimers Dement 12(7):796–804
Buckner RL, Andrews-Hanna JR, Schacter DL (2008) The brain's default network: anatomy, function, and relevance to disease. Ann N Y Acad Sci 1124:1–38
Buckner RL, Snyder AZ, Shannon BJ, LaRossa G, Sachs R, Fotenos AF, Sheline YI, Klunk WE, Mathis CA, Morris JC, Mintun MA (2005) Molecular, structural, and functional characterization of Alzheimer's disease: evidence for a relationship between default activity, amyloid, and memory. J Neurosci 25(34):7709–7717
Burack MA, Hartlein J, Flores HP, Taylor-Reinwald L, Perlmutter JS, Cairns NJ (2010) In vivo amyloid imaging in autopsy-confirmed Parkinson disease with dementia. Neurology 74(1):77–84
Burnham SC, Bourgeat P, Dore V, Savage G, Brown B, Laws S, Maruff P, Salvado O, Ames D, Martins RN, Masters CL, Rowe CC, Villemagne VL, AIBL Research Group (2016) Clinical and cognitive trajectories in cognitively healthy elderly individuals with suspected non-Alzheimer's disease pathophysiology (SNAP) or Alzheimer's disease pathology: a longitudinal study. Lancet Neurol 15(10):1044–1053
Cairns NJ, Bigio EH, Mackenzie IR, Neumann M, Lee VM, Hatanpaa KJ, White CL 3rd, Schneider JA, Grinberg LT, Halliday G, Duyckaerts C, Lowe JS, Holm IE, Tolnay M, Okamoto K, Yokoo H, Murayama S, Woulfe J, Munoz DG, Dickson DW, Ince PG, Trojanowski JQ, Mann DM (2007) Neuropathologic diagnostic and nosologic criteria for frontotemporal lobar degeneration: consensus of the Consortium for Frontotemporal Lobar Degeneration. Acta Neuropathol 114(1):5–22
Cairns NJ, Ikonomovic MD, Benzinger T, Storandt M, Fagan AM, Shah AR, Reinwald LT, Carter D, Felton A, Holtzman DM, Mintun MA, Klunk WE, Morris JC (2009) Absence of Pittsburgh compound B detection of cerebral amyloid beta in a patient with clinical, cognitive, and cerebrospinal fluid markers of Alzheimer disease: a case report. Arch Neurol 66(12):1557–1562
Camus V, Payoux P, Barre L, Desgranges B, Voisin T, Tauber C, La Joie R, Tafani M, Hommet C, Chetelat G, Mondon K, de La Sayette V, Cottier JP, Beaufils E, Ribeiro MJ, Gissot V, Vierron E, Vercouillie J, Vellas B, Eustache F, Guilloteau D (2012) Using PET with (18)F-AV-45 (florbetapir) to quantify brain amyloid load in a clinical environment. Eur J Nucl Med Mol Imaging 39(4):621–631
Cappai R, White AR (1999) Amyloid beta. Int J Biochem Cell Biol 31(9):885–889
Carter SF, Scholl M, Almkvist O, Wall A, Engler H, Langstrom B, Nordberg A (2012) Evidence for astrocytosis in prodromal Alzheimer disease provided by 11C-deuterium-L-deprenyl: a multitracer PET paradigm combining 11C-Pittsburgh compound B and 18F-FDG. J Nucl Med 53(1):37–46
Ceccaldi M, Jonveaux T, Verger A, Krolak-Salmon P, Houzard C, Godefroy O, Shields T, Perrotin A, Gismondi R, Bullich S, Jovalekic A, Raffa N, Pasquier F, Semah F, Dubois B, Habert MO, Wallon D, Chastan M, Payoux P, Guedj E, Stephens A, NEUUS in AD study group (2018) Added value of (18)F-florbetaben amyloid PET in the diagnostic workup of most complex patients with dementia in France: a naturalistic study. Alzheimers Dement 14(3):293–305
Chang CY, Silverman DH (2004) Accuracy of early diagnosis and its impact on the management and course of Alzheimer's disease. Expert Rev Mol Diagn 4:63–69
Chen ST, Siddarth P, Merrill DA, Martinez J, Emerson ND, Liu J, Wong KP, Satyamurthy N, Giza CC, Huang SC, Fitzsimmons RP, Bailes J, Omalu B, Barrio JR, Small GW (2018) FDDNP-PET tau brain protein binding patterns in military personnel with suspected chronic traumatic Encephalopathy1. J Alzheimers Dis 65(1):79–88
Chetelat G, Desgranges B, de la Sayette V, Viader F, Berkouk K, Landeau B, Lalevee C, Le Doze F, Dupuy B, Hannequin D, Baron JC, Eustache F (2003) Dissociating atrophy and hypometabolism impact on episodic memory in mild cognitive impairment. Brain 126(Pt 9):1955–1967
Chetelat G, Eustache F, Viader F, De La Sayette V, Pelerin A, Mezenge F, Hannequin D, Dupuy B, Baron JC, Desgranges B (2005) FDG-PET measurement is more accurate than neuropsychological assessments to predict global cognitive deterioration in patients with mild cognitive impairment. Neurocase 11(1):14–25
Chetelat G, Villemagne VL, Bourgeat P, Pike KE, Jones G, Ames D, Ellis KA, Szoeke C, Martins RN, O'Keefe GJ, Salvado O, Masters CL, Rowe CC (2010a) Relationship between atrophy and beta-amyloid deposition in Alzheimer disease. Ann Neurol 67(3):317–324
Chetelat G, Villemagne VL, Pike KE, Baron JC, Bourgeat P, Jones G, Faux NG, Ellis KA, Salvado O, Szoeke C, Martins RN, Ames D, Masters CL, Rowe CC (2010b) Larger temporal volume in elderly with high versus low beta-amyloid deposition. Brain 133(11):3349–3358
Chetelat G, Villemagne VL, Villain N, Jones G, Ellis KA, Ames D, Martins RN, Masters CL, Rowe CC (2012) Accelerated cortical atrophy in cognitively normal elderly with high beta-amyloid deposition. Neurology 78(7):477–484
Choi SR, Golding G, Zhuang Z, Zhang W, Lim N, Hefti F, Benedum TE, Kilbourn MR, Skovronsky D, Kung HF (2009) Preclinical properties of 18F-AV-45: a PET agent for Abeta plaques in the brain. J Nucl Med 50(11):1887–1894
Clark CM, Davatzikos C, Borthakur A, Newberg A, Leight S, Lee VM, Trojanowski JQ (2008) Biomarkers for early detection of Alzheimer pathology. Neurosignals 16(1):11–18
Clark CM, Pontecorvo MJ, Beach TG, Bedell BJ, Coleman RE, Doraiswamy PM, Fleisher AS, Reiman EM, Sabbagh MN, Sadowsky CH, Schneider JA, Arora A, Carpenter AP, Flitter ML, Joshi AD, Krautkramer MJ, Lu M, Mintun MA, Skovronsky DM (2012) Cerebral PET with florbetapir compared with neuropathology at autopsy for detection of neuritic amyloid-beta plaques: a prospective cohort study. Lancet Neurol 11(8):669–678
Clark CM, Schneider JA, Bedell BJ, Beach TG, Bilker WB, Mintun MA, Pontecorvo MJ, Hefti F, Carpenter AP, Flitter ML, Krautkramer MJ, Kung HF, Coleman RE, Doraiswamy PM, Fleisher AS, Sabbagh MN, Sadowsky CH, Reiman EP, Zehntner SP, Skovronsky DM (2011) Use of florbetapir-PET for imaging beta-amyloid pathology. JAMA 305(3):275–283
Cohen AD, McDade E, Christian B, Price J, Mathis C, Klunk W, Handen BL (2018) Early striatal amyloid deposition distinguishes down syndrome and autosomal dominant Alzheimer's disease from late-onset amyloid deposition. Alzheimers Dement 14(6):743–750
Cohen AD, Price JC, Weissfeld LA, James J, Rosario BL, Bi W, Nebes RD, Saxton JA, Snitz BE, Aizenstein HA, Wolk DA, Dekosky ST, Mathis CA, Klunk WE (2009) Basal cerebral metabolism may modulate the cognitive effects of Abeta in mild cognitive impairment: an example of brain reserve. J Neurosci 29(47):14770–14778
Cohen AD, Rabinovici GD, Mathis CA, Jagust WJ, Klunk WE, Ikonomovic MD (2012) Using Pittsburgh compound B for in vivo PET imaging of Fibrillar amyloid-Beta. Adv Pharmacol 64:27–81
Coleman RE (2005) Positron emission tomography diagnosis of Alzheimer's disease. Neuroimaging Clin N Am 15(4):837–846
Cordonnier C (2010) Brain microbleeds. Pract Neurol 10(2):94–100
Cselenyi Z, Jonhagen ME, Forsberg A, Halldin C, Julin P, Schou M, Johnstrom P, Varnas K, Svensson S, Farde L (2012) Clinical validation of 18F-AZD4694, an amyloid-beta-specific PET Radioligand. J Nucl Med 53(3):415–424
Cummings J (2019) The National Institute on Aging-Alzheimer's Association framework on Alzheimer's disease: application to clinical trials. Alzheimers Dement 15(1):172–178
Cummings JL, Vinters HV, Cole GM, Khachaturian ZS (1998) Alzheimer's disease: etiologies, pathophysiology, cognitive reserve, and treatment opportunities. Neurology 51(1):S2–S17; discussion S65–17
Curtis C, Gamez JE, Singh U, Sadowsky CH, Villena T, Sabbagh MN, Beach TG, Duara R, Fleisher AS, Frey KA, Walker Z, Hunjan A, Holmes C, Escovar YM, Vera CX, Agronin ME, Ross J, Bozoki A, Akinola M, Shi J, Vandenberghe R, Ikonomovic MD, Sherwin PF, Grachev ID, Farrar G, Smith AP, Buckley CJ, McLain R, Salloway S (2015) Phase 3 trial of flutemetamol labeled with radioactive fluorine 18 imaging and neuritic plaque density. JAMA Neurol 72(3):287–294
Dang C, Yassi N, Harrington KD, Xia Y, Lim YY, Ames D, Laws SM, Hickey M, Rainey-Smith S, Sohrabi HR, Doecke JD, Fripp J, Salvado O, Snyder PJ, Weinborn M, Villemagne VL, Rowe CC, Masters CL, Maruff P, A. R. Group (2019) Rates of age- and amyloid beta-associated cortical atrophy in older adults with superior memory performance. Alzheimers Dement (Amst) 11:566–575
Davies L, Wolska B, Hilbich C, Multhaup G, Martins R, Simms G, Beyreuther K, Masters CL (1988) A4 amyloid protein deposition and the diagnosis of Alzheimer's disease: prevalence in aged brains determined by immunocytochemistry compared with conventional neuropathologic techniques. Neurology 38(11):1688–1693
de Leon MJ, DeSanti S, Zinkowski R, Mehta PD, Pratico D, Segal S, Rusinek H, Li J, Tsui W, Saint Louis LA, Clark CM, Tarshish C, Li Y, Lair L, Javier E, Rich K, Lesbre P, Mosconi L, Reisberg B, Sadowski M, DeBernadis JF, Kerkman DJ, Hampel H, Wahlund LO, Davies P (2006) Longitudinal CSF and MRI biomarkers improve the diagnosis of mild cognitive impairment. Neurobiol Aging 27(3):394–401
de Leon MJ, Mosconi L, Blennow K, DeSanti S, Zinkowski R, Mehta PD, Pratico D, Tsui W, Saint Louis LA, Sobanska L, Brys M, Li Y, Rich K, Rinne J, Rusinek H (2007) Imaging and CSF studies in the preclinical diagnosis of Alzheimer's disease. Ann N Y Acad Sci 1097:114–145
de Wilde A, van der Flier WM, Pelkmans W, Bouwman F, Verwer J, Groot C, van Buchem MM, Zwan M, Ossenkoppele R, Yaqub M, Kunneman M, Smets EMA, Barkhof F, Lammertsma AA, Stephens A, van Lier E, Biessels GJ, van Berckel BN, Scheltens P (2018) Association of Amyloid Positron Emission Tomography with Changes in diagnosis and patient treatment in an unselected memory clinic cohort: the ABIDE project. JAMA Neurol 75(9):1062–1070
DeCarli C, Villeneuve S, Maillard P, Harvey D, Singh B, Carmichael O, Fletcher E, Olichney J, Farias S, Jagust W, Reed B, Mungas D (2019) Vascular burden score impacts cognition independent of amyloid PET and MRI measures of Alzheimer's disease and vascular brain injury. J Alzheimers Dis 68(1):187–196
DeKosky ST, Ikonomovic MD, Styren SD, Beckett L, Wisniewski S, Bennett DA, Cochran EJ, Kordower JH, Mufson EJ (2002) Upregulation of choline acetyltransferase activity in hippocampus and frontal cortex of elderly subjects with mild cognitive impairment. Ann Neurol 51(2):145–155
Deshpande A, Mina E, Glabe C, Busciglio J (2006) Different conformations of amyloid beta induce neurotoxicity by distinct mechanisms in human cortical neurons. J Neurosci 26(22):6011–6018
Devanand DP, Jacobs DM, Tang MX, Del Castillo-Castaneda C, Sano M, Marder K, Bell K, Bylsma FW, Brandt J, Albert M, Stern Y (1997) The course of psychopathologic features in mild to moderate Alzheimer disease. Arch Gen Psychiatry 54(3):257–263
Dhollander I, Nelissen N, Van Laere K, Peeters D, Demaerel P, Van Paesschen W, Thijs V, Vandenberghe R (2011) In vivo amyloid imaging in cortical superficial siderosis. J Neurol Neurosurg Psychiatry 82(4):469–471
Dierksen GA, Skehan ME, Khan MA, Jeng J, Nandigam RN, Becker JA, Kumar A, Neal KL, Betensky RA, Frosch MP, Rosand J, Johnson KA, Viswanathan A, Salat DH, Greenberg SM (2010) Spatial relation between microbleeds and amyloid deposits in amyloid angiopathy. Ann Neurol 68(4):545–548
Donaghy PC, Firbank MJ, Thomas AJ, Lloyd J, Petrides G, Barnett N, Olsen K, O'Brien JT (2019) Amyloid imaging and longitudinal clinical progression in dementia with Lewy bodies. Am J Geriatr Psychiatry 28(5):573–577
Doody RS, Thomas RG, Farlow M, Iwatsubo T, Vellas B, Joffe S, Kieburtz K, Raman R, Sun X, Aisen PS, Siemers E, Liu-Seifert H, Mohs R, Alzheimer's Disease Cooperative Study Steering Committee; Solanezumab Study Group (2014) Phase 3 trials of solanezumab for mild-to-moderate Alzheimer's disease. N Engl J Med 370(4):311–321
Doraiswamy PM, Sperling RA, Coleman RE, Johnson KA, Reiman EM, Davis MD, Grundman M, Sabbagh MN, Sadowsky CH, Fleisher AS, Carpenter A, Clark CM, Joshi AD, Mintun MA, Skovronsky DM, Pontecorvo MJ (2012) Amyloid-beta assessed by florbetapir F 18 PET and 18-month cognitive decline: A multicenter study. Neurology 79(16):1636–1644
Dore V, Bullich S, Rowe CC, Bourgeat P, Konate S, Sabri O, Stephens AW, Barthel H, Fripp J, Masters CL, Dinkelborg L, Salvado O, Villemagne VL, De Santi S (2019) Comparison of (18)F-florbetaben quantification results using the standard Centiloid, MR-based, and MR-less CapAIBL((R)) approaches: validation against histopathology. Alzheimers Dement 15(6):807–816
Drago V, Babiloni C, Bartres-Faz D, Caroli A, Bosch B, Hensch T, Didic M, Klafki HW, Pievani M, Jovicich J, Venturi L, Spitzer P, Vecchio F, Schoenknecht P, Wiltfang J, Redolfi A, Forloni G, Blin O, Irving E, Davis C, Hardemark HG, Frisoni GB (2011) Disease tracking markers for Alzheimer's disease at the prodromal (MCI) stage. J Alzheimers Dis 26(Suppl 3):159–199
Driscoll I, Zhou Y, An Y, Sojkova J, Davatzikos C, Kraut MA, Ye W, Ferrucci L, Mathis CA, Klunk WE, Wong DF, Resnick SM (2010) Lack of association between (11)C-PiB and longitudinal brain atrophy in non-demented older individuals. Neurobiol Aging 32(12):2123–2130
Drzezga A (2010) Amyloid-plaque imaging in early and differential diagnosis of dementia. Ann Nucl Med 24(2):55–66
Drzezga A, Becker JA, Van Dijk KR, Sreenivasan A, Talukdar T, Sullivan C, Schultz AP, Sepulcre J, Putcha D, Greve D, Johnson KA, Sperling RA (2011) Neuronal dysfunction and disconnection of cortical hubs in non-demented subjects with elevated amyloid burden. Brain 134(Pt 6):1635–1646
Drzezga A, Grimmer T, Henriksen G, Stangier I, Perneczky R, Diehl-Schmid J, Mathis CA, Klunk WE, Price J, DeKosky S, Wester HJ, Schwaiger M, Kurz A (2008) Imaging of amyloid plaques and cerebral glucose metabolism in semantic dementia and Alzheimer's disease. NeuroImage 39(2):619–633
Drzezga A, Grimmer T, Riemenschneider M, Lautenschlager N, Siebner H, Alexopoulus P, Minoshima S, Schwaiger M, Kurz A (2005) Prediction of individual clinical outcome in MCI by means of genetic assessment and (18)F-FDG PET. J Nucl Med 46(10):1625–1632
Drzezga A, Lautenschlager N, Siebner H, Riemenschneider M, Willoch F, Minoshima S, Schwaiger M, Kurz A (2003) Cerebral metabolic changes accompanying conversion of mild cognitive impairment into Alzheimer's disease: a PET follow-up study. Eur J Nucl Med Mol Imaging 30(8):1104–1113
Dubois B, Feldman HH, Jacova C, Cummings JL, Dekosky ST, Barberger-Gateau P, Delacourte A, Frisoni G, Fox NC, Galasko D, Gauthier S, Hampel H, Jicha GA, Meguro K, O'Brien J, Pasquier F, Robert P, Rossor M, Salloway S, Sarazin M, de Souza LC, Stern Y, Visser PJ, Scheltens P (2010) Revising the definition of Alzheimer's disease: a new lexicon. Lancet Neurol 9(11):1118–1127
Dulamea A, Solomon E (2016) Role of the biomarkers for the diagnosis of Creutzfeldt-Jakob disease. J Med Life 9(2):211–215
Eckert A, Keil U, Marques CA, Bonert A, Frey C, Schussel K, Muller WE (2003) Mitochondrial dysfunction, apoptotic cell death, and Alzheimer's disease. Biochem Pharmacol 66(8):1627–1634
Edison P, Archer HA, Gerhard A, Hinz R, Pavese N, Turkheimer FE, Hammers A, Tai YF, Fox N, Kennedy A, Rossor M, Brooks DJ (2008a) Microglia, amyloid, and cognition in Alzheimer's disease: An [11C](R)PK11195-PET and [11C]PIB-PET study. Neurobiol Dis 32(3):412–419
Edison P, Archer HA, Hinz R, Hammers A, Pavese N, Tai YF, Hotton G, Cutler D, Fox N, Kennedy A, Rossor M, Brooks DJ (2007) Amyloid, hypometabolism, and cognition in Alzheimer disease: an [11C]PIB and [18F]FDG PET study. Neurology 68(7):501–508
Edison P, Rowe CC, Rinne JO, Ng S, Ahmed I, Kemppainen N, Villemagne VL, O'Keefe G, Nagren K, Chaudhury KR, Masters CL, Brooks DJ (2008b) Amyloid load in Parkinson's disease dementia and Lewy body dementia measured with [11C]PIB positron emission tomography. J Neurol Neurosurg Psychiatry 79(12):1331–1338
Egan MF, Kost J, Voss T, Mukai Y, Aisen PS, Cummings JL, Tariot PN, Vellas B, van Dyck CH, Boada M, Zhang Y, Li W, Furtek C, Mahoney E, Harper Mozley L, Mo Y, Sur C, Michelson D (2019) Randomized trial of Verubecestat for prodromal Alzheimer's disease. N Engl J Med 380(15):1408–1420
Ellis KA, Bush AI, Darby D, De Fazio D, Foster J, Hudson P, Lautenschlager NT, Lenzo N, Martins RN, Maruff P, Masters C, Milner A, Pike K, Rowe C, Savage G, Szoeke C, Taddei K, Villemagne V, Woodward M, Ames D (2009) The Australian imaging, biomarkers and lifestyle (AIBL) study of aging: methodology and baseline characteristics of 1112 individuals recruited for a longitudinal study of Alzheimer's disease. Int Psychogeriatr 21(4):672–687
Engler H, Forsberg A, Almkvist O, Blomquist G, Larsson E, Savitcheva I, Wall A, Ringheim A, Langstrom B, Nordberg A (2006) Two-year follow-up of amyloid deposition in patients with Alzheimer's disease. Brain 129(Pt 11):2856–2866
Engler H, Santillo AF, Wang SX, Lindau M, Savitcheva I, Nordberg A, Lannfelt L, Langstrom B, Kilander L (2008) In vivo amyloid imaging with PET in frontotemporal dementia. Eur J Nucl Med Mol Imaging 35(1):100–106
Fagan AM, Holtzman DM (2010) Cerebrospinal fluid biomarkers of Alzheimer's disease. Biomark Med 4(1):51–63
Fagan AM, Mintun MA, Mach RH, Lee SY, Dence CS, Shah AR, Larossa GN, Spinner ML, Klunk WE, Mathis CA, Dekosky ST, Morris JC, Holtzman DM (2006) Inverse relation between in vivo amyloid imaging load and cerebrospinal fluid Abeta(42) in humans. Ann Neurol 59(3):512–519
Fagan AM, Mintun MA, Shah AR, Aldea P, Roe CM, Mach RH, Marcus D, Morris JC, Holtzman DM (2009) Cerebrospinal fluid tau and ptau(181) increase with cortical amyloid deposition in cognitively normal individuals: implications for future clinical trials of Alzheimer's disease. EMBO Mol Med 1(8–9):371–380
Fagan AM, Roe CM, Xiong C, Mintun MA, Morris JC, Holtzman DM (2007) Cerebrospinal fluid tau/beta-amyloid(42) ratio as a prediction of cognitive decline in nondemented older adults. Arch Neurol 64(3):343–349
Fagan AM, Xiong C, Jasielec MS, Bateman RJ, Goate AM, Benzinger TL, Ghetti B, Martins RN, Masters CL, Mayeux R, Ringman JM, Rossor MN, Salloway S, Schofield PR, Sperling RA, Marcus D, Cairns NJ, Buckles VD, Ladenson JH, Morris JC, Holtzman DM, Dominantly Inherited Alzheimer Network (2014) Longitudinal change in CSF biomarkers in autosomal-dominant Alzheimer's disease. Sci Transl Med 6(226):226ra230
Fandos N, Pérez-Grijalba V, Pesini P, Olmos S, Bossa M, Villemagne VL, Doecke J, Fowler C, Masters CL, Sarasa M (2017) Plasma amyloid β 42/40 ratios as biomarkers for amyloid β cerebral deposition in cognitively normal individuals. Alzheimers Dement (Amst) 8:179–187
Farrar G, Molinuevo JL, Zanette M (2019) Is there a difference in regional read [(18)F]flutemetamol amyloid patterns between end-of-life subjects and those with amnestic mild cognitive impairment? Eur J Nucl Med Mol Imaging 46(6):1299–1308
Farrer LA, Cupples LA, Haines JL, Hyman B, Kukull WA, Mayeux R, Myers RH, Pericak-Vance MA, Risch N, van Duijn CM (1997) Effects of age, sex, and ethnicity on the association between apolipoprotein E genotype and Alzheimer disease. A meta-analysis. APOE and Alzheimer disease meta analysis consortium. JAMA 278(16):1349–1356
Feldman HH, Maia LF, Mackenzie IR, Forster BB, Martzke J, Woolfenden A (2008) Superficial siderosis: a potential diagnostic marker of cerebral amyloid angiopathy in Alzheimer disease. Stroke 39(10):2894–2897
Fleisher AS, Chen K, Liu X, Roontiva A, Thiyyagura P, Ayutyanont N, Joshi AD, Clark CM, Mintun MA, Pontecorvo MJ, Doraiswamy PM, Johnson KA, Skovronsky DM, Reiman EM (2011) Using positron emission tomography and florbetapir F18 to image cortical amyloid in patients with mild cognitive impairment or dementia due to Alzheimer disease. Arch Neurol 68(11):1404–1411
Fletcher E, Filshtein TJ, Harvey D, Renaud A, Mungas D, DeCarli C (2018) Staging of amyloid beta, t-tau, regional atrophy rates, and cognitive change in a nondemented cohort: results of serial mediation analyses. Alzheimers Dement (Amst) 10:382–393
Fodero-Tavoletti MT, Brockschnieder D, Villemagne VL, Martin L, Connor AR, Thiele A, Berndt A, McLean CA, Krause S, Rowe CC, Masters CL, Dinkelborg L, Dyrks T, Cappai R (2012) In vitro characterisation of [18F]–florbetaben, an Aβ imaging radiotracer. Nucl Med Biol 39(7):1042–1048. https://doi.org/10.1016/j.nucmedbio.2012.1003.1001
Fodero-Tavoletti MT, Mulligan RS, Okamura N, Furumoto S, Rowe CC, Kudo Y, Masters CL, Cappai R, Yanai K, Villemagne VL (2009a) In vitro characterisation of BF227 binding to alpha-synuclein/Lewy bodies. Eur J Pharmacol 617(1–3):54–58
Fodero-Tavoletti MT, Okamura N, Furumoto S, Mulligan RS, Connor AR, McLean CA, Cao D, Rigopoulos A, Cartwright GA, O'Keefe G, Gong S, Adlard PA, Barnham KJ, Rowe CC, Masters CL, Kudo Y, Cappai R, Yanai K, Villemagne VL (2011) 18F-THK523: a novel in vivo tau imaging ligand for Alzheimer's disease. Brain 134(Pt 4):1089–1100
Fodero-Tavoletti MT, Rowe CC, McLean CA, Leone L, Li QX, Masters CL, Cappai R, Villemagne VL (2009b) Characterization of PiB binding to white matter in Alzheimer disease and other dementias. J Nucl Med 50(2):198–204
Fodero-Tavoletti MT, Smith DP, McLean CA, Adlard PA, Barnham KJ, Foster LE, Leone L, Perez K, Cortes M, Culvenor JG, Li QX, Laughton KM, Rowe CC, Masters CL, Cappai R, Villemagne VL (2007) In vitro characterization of Pittsburgh compound-B binding to Lewy bodies. J Neurosci 27(39):10365–10371
Formaglio M, Costes N, Seguin J, Tholance Y, Le Bars D, Roullet-Solignac I, Mercier B, Krolak-Salmon P, Vighetto A (2011) In vivo demonstration of amyloid burden in posterior cortical atrophy: a case series with PET and CSF findings. J Neurol 258(10):1841–1851
Forman MS, Mufson EJ, Leurgans S, Pratico D, Joyce S, Leight S, Lee VM, Trojanowski JQ (2007) Cortical biochemistry in MCI and Alzheimer disease: lack of correlation with clinical diagnosis. Neurology 68(10):757–763
Forsberg A, Almkvist O, Engler H, Wall A, Langstrom B, Nordberg A (2010) High PIB retention in Alzheimer's disease is an early event with complex relationship with CSF biomarkers and functional parameters. Curr Alzheimer Res 7(1):56–66
Forsberg A, Engler H, Almkvist O, Blomquist G, Hagman G, Wall A, Ringheim A, Langstrom B, Nordberg A (2008) PET imaging of amyloid deposition in patients with mild cognitive impairment. Neurobiol Aging 29(10):1456–1465
Forster S, Grimmer T, Miederer I, Henriksen G, Yousefi BH, Graner P, Wester HJ, Forstl H, Kurz A, Dickerson BC, Bartenstein P, Drzezga A (2012) Regional expansion of Hypometabolism in Alzheimer's disease follows amyloid deposition with temporal delay. Biol Psychiatry 71(9):792–797
Foster ER, Campbell MC, Burack MA, Hartlein J, Flores HP, Cairns NJ, Hershey T, Perlmutter JS (2010) Amyloid imaging of Lewy body-associated disorders. Mov Disord 25(15):2516–2523
Frisoni GB, Barkhof F, Altomare D, Berkhof J, Boccardi M, Canzoneri E, Collij L, Drzezga A, Farrar G, Garibotto V, Gismondi R, Gispert JD, Jessen F, Kivipelto M, Lopes Alves I, Molinuevo JL, Nordberg A, Payoux P, Ritchie C, Savicheva I, Scheltens P, Schmidt ME, Schott JM, Stephens A, van Berckel B, Vellas B, Walker Z, Raffa N (2019) AMYPAD diagnostic and patient management study: rationale and design. Alzheimers Dement 15(3):388–399
Frisoni GB, Fox NC, Jack CR Jr, Scheltens P, Thompson PM (2010) The clinical use of structural MRI in Alzheimer disease. Nat Rev Neurol 6(2):67–77
Furst AJ, Bigler ED (2016) Amyloid plaques in TBI: incidental finding or precursor for what is to come? Neurology 86(9):798–799
Furst AJ, Rabinovici GD, Rostomian AH, Steed T, Alkalay A, Racine C, Miller BL, Jagust WJ (2010) Cognition, glucose metabolism and amyloid burden in Alzheimer's disease. Neurobiol Aging 33(2):215–225
Furukawa K, Okamura N, Tashiro K, Furumoto S, Tomita N, Yanai K, Kudo Y, Arai H (2011) PET imaging with BF-227 in dementia with Lewy bodies. Alzheimers Dement 7(Suppl 1):S12–S13
Gatson JW, Stebbins C, Mathews D, Harris TS, Madden C, Batjer H, Diaz-Arrastia R, Minei JP (2016) Evidence of increased brain amyloid in severe TBI survivors at 1, 12, and 24 months after injury: report of 2 cases. J Neurosurg 124(6):1646–1653
Gomez-Isla T, West HL, Rebeck GW, Harr SD, Growdon JH, Locascio JJ, Perls TT, Lipsitz LA, Hyman BT (1996) Clinical and pathological correlates of apolipoprotein E epsilon 4 in Alzheimer's disease. Ann Neurol 39(1):62–70
Gomperts SN, Marquie M, Locascio JJ, Bayer S, Johnson KA, Growdon JH (2016) PET Radioligands reveal the basis of dementia in Parkinson's disease and dementia with Lewy bodies. Neurodegener Dis 16(1–2):118–124
Gomperts SN, Rentz DM, Moran E, Becker JA, Locascio JJ, Klunk WE, Mathis CA, Elmaleh DR, Shoup T, Fischman AJ, Hyman BT, Growdon JH, Johnson KA (2008) Imaging amyloid deposition in Lewy body diseases. Neurology 71(12):903–910
Gonneaud J, Arenaza-Urquijo EM, Mezenge F, Landeau B, Gaubert M, Bejanin A, de Flores R, Wirth M, Tomadesso C, Poisnel G, Abbas A, Desgranges B, Chetelat G (2017) Increased florbetapir binding in the temporal neocortex from age 20 to 60 years. Neurology 89(24):2438–2446
Grimmer T, Riemenschneider M, Forstl H, Henriksen G, Klunk WE, Mathis CA, Shiga T, Wester HJ, Kurz A, Drzezga A (2009) Beta amyloid in Alzheimer's disease: increased deposition in brain is reflected in reduced concentration in cerebrospinal fluid. Biol Psychiatry 65(11):927–934
Grimmer T, Tholen S, Yousefi BH, Alexopoulos P, Forschler A, Forstl H, Henriksen G, Klunk WE, Mathis CA, Perneczky R, Sorg C, Kurz A, Drzezga A (2010) Progression of cerebral amyloid load is associated with the apolipoprotein E epsilon4 genotype in Alzheimer's disease. Biol Psychiatry 68(10):879–884
Gurol ME, Dierksen G, Betensky R, Gidicsin C, Halpin A, Becker A, Carmasin J, Ayres A, Schwab K, Viswanathan A, Salat D, Rosand J, Johnson KA, Greenberg SM (2012) Predicting sites of new hemorrhage with amyloid imaging in cerebral amyloid angiopathy. Neurology 79(4):320–326
Hagens MHJ, Golla SSV, Janssen B, Vugts DJ, Beaino W, Windhorst AD, O'Brien-Brown J, Kassiou M, Schuit RC, Schwarte LA, de Vries HE, Killestein J, Barkhof F, van Berckel BNM, Lammertsma AA (2020) The P2X7 receptor tracer [(11)C]SMW139 as an in vivo marker of neuroinflammation in multiple sclerosis: a first-in man study. Eur J Nucl Med Mol Imaging 47(2):379–389
Halliday G, Hely M, Reid W, Morris J (2008) The progression of pathology in longitudinally followed patients with Parkinson's disease. Acta Neuropathol 115(4):409–415
Hampel H, Blennow K, Shaw LM, Hoessler YC, Zetterberg H, Trojanowski JQ (2010) Total and phosphorylated tau protein as biological markers of Alzheimer's disease. Exp Gerontol 45(1):30–40
Hanseeuw BJ, Betensky RA, Schultz AP, Papp KV, Mormino EC, Sepulcre J, Bark JS, Cosio DM, LaPoint M, Chhatwal JP, Rentz DM, Sperling RA, Johnson KA (2017) Fluorodeoxyglucose metabolism associated with tau-amyloid interaction predicts memory decline. Ann Neurol 81(4):583–596
Hansson O, Zetterberg H, Buchhave P, Andreasson U, Londos E, Minthon L, Blennow K (2007) Prediction of Alzheimer's disease using the CSF Abeta42/Abeta40 ratio in patients with mild cognitive impairment. Dement Geriatr Cogn Disord 23(5):316–320
Hardy J (1997) Amyloid, the presenilins and Alzheimer's disease. Trends Neurosci 20(4):154–159
Herholz K (1995) FDG PET and differential diagnosis of dementia. Alzheimer Dis Assoc Disord 9(1):6–16
Hill AF, Joiner S, Wadsworth JD, Sidle KC, Bell JE, Budka H, Ironside JW, Collinge J (2003) Molecular classification of sporadic Creutzfeldt-Jakob disease. Brain 126(Pt 6):1333–1346
Hodges JR, Patterson K (2007) Semantic dementia: a unique clinicopathological syndrome. Lancet Neurol 6(11):1004–1014
Hu X, Teunissen CE, Spottke A, Heneka MT, Duzel E, Peters O, Li S, Priller J, Buerger K, Teipel S, Laske C, Verfaillie SCJ, Barkhof F, Coll-Padros N, Rami L, Molinuevo JL, van der Flier WM, Jessen F (2019) Smaller medial temporal lobe volumes in individuals with subjective cognitive decline and biomarker evidence of Alzheimer's disease-data from three memory clinic studies. Alzheimers Dement 15(2):185–193
Hyman BT (2011) Amyloid-dependent and amyloid-independent stages of Alzheimer disease. Arch Neurol 68(8):1062–1064
Ikonomovic MD, Abrahamson EE, Isanski BA, Wuu J, Mufson EJ, DeKosky ST (2007) Superior frontal cortex cholinergic axon density in mild cognitive impairment and early Alzheimer disease. Arch Neurol 64(9):1312–1317
Ikonomovic MD, Abrahamson EE, Price JC, Hamilton RL, Mathis CA, Paljug WR, Debnath ML, Cohen AD, Mizukami K, Dekosky ST, Lopez OL, Klunk WE (2012) Early AD pathology in a [C-11]PiB-negative case: a PiB-amyloid imaging, biochemical, and immunohistochemical study. Acta Neuropathol 123(3):433–447
Ikonomovic MD, Klunk WE, Abrahamson EE, Mathis CA, Price JC, Tsopelas ND, Lopresti BJ, Ziolko S, Bi W, Paljug WR, Debnath ML, Hope CE, Isanski BA, Hamilton RL, DeKosky ST (2008) Post-mortem correlates of in vivo PiB-PET amyloid imaging in a typical case of Alzheimer's disease. Brain 131(Pt 6):1630–1645
Ikonomovic MD, Klunk WE, Abrahamson EE, Wuu J, Mathis CA, Scheff SW, Mufson EJ, DeKosky ST (2011) Precuneus amyloid burden is associated with reduced cholinergic activity in Alzheimer disease. Neurology 77(1):39–47
Inayathullah M, Teplow DB (2011) Structural dynamics of the DeltaE22 (Osaka) familial Alzheimer's disease-linked amyloid beta-protein. Amyloid 18(3):98–107
Isacson O, Seo H, Lin L, Albeck D, Granholm AC (2002) Alzheimer's disease and Down's syndrome: roles of APP, trophic factors and ACh. Trends Neurosci 25(2):79–84
Jack CR Jr, Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB, Holtzman DM, Jagust W, Jessen F, Karlawish J, Liu E, Molinuevo JL, Montine T, Phelps C, Rankin KP, Rowe CC, Scheltens P, Siemers E, Snyder HM, Sperling R, Contributors (2018) NIA-AA research framework: toward a biological definition of Alzheimer's disease. Alzheimers Dement 14(4):535–562
Jack CR Jr, Bennett DA, Blennow K, Carrillo MC, Feldman HH, Frisoni GB, Hampel H, Jagust WJ, Johnson KA, Knopman DS, Petersen RC, Scheltens P, Sperling RA, Dubois B (2016) A/T/N: An unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology 87(5):539–547
Jack CR Jr, Knopman DS, Jagust WJ, Shaw LM, Aisen PS, Weiner MW, Petersen RC, Trojanowski JQ (2010) Hypothetical model of dynamic biomarkers of the Alzheimer's pathological cascade. Lancet Neurol 9(1):119–128
Jack CR Jr, Knopman DS, Weigand SD, Wiste HJ, Vemuri P, Lowe V, Kantarci K, Gunter JL, Senjem ML, Ivnik RJ, Roberts RO, Rocca WA, Boeve BF, Petersen RC (2012) An operational approach to National Institute on Aging-Alzheimer's Association criteria for preclinical Alzheimer disease. Ann Neurol 71(6):765–775
Jack CR Jr, Lowe VJ, Senjem ML, Weigand SD, Kemp BJ, Shiung MM, Knopman DS, Boeve BF, Klunk WE, Mathis CA, Petersen RC (2008a) 11C PiB and structural MRI provide complementary information in imaging of Alzheimer's disease and amnestic mild cognitive impairment. Brain 131(Pt 3):665–680
Jack CR Jr, Lowe VJ, Weigand SD, Wiste HJ, Senjem ML, Knopman DS, Shiung MM, Gunter JL, Boeve BF, Kemp BJ, Weiner M, Petersen RC (2009) Serial PIB and MRI in normal, mild cognitive impairment and Alzheimer's disease: implications for sequence of pathological events in Alzheimer's disease. Brain 132(Pt 5):1355–1365
Jack CR Jr, Weigand SD, Shiung MM, Przybelski SA, O'Brien PC, Gunter JL, Knopman DS, Boeve BF, Smith GE, Petersen RC (2008b) Atrophy rates accelerate in amnestic mild cognitive impairment. Neurology 70(19 Pt 2):1740–1752
Jack CR Jr, Wiste HJ, Lesnick TG, Weigand SD, Knopman DS, Vemuri P, Pankratz VS, Senjem ML, Gunter JL, Mielke MM, Lowe VJ, Boeve BF, Petersen RC (2013) Brain beta-amyloid load approaches a plateau. Neurology 80(10):890–896
Jack CR Jr, Wiste HJ, Weigand SD, Rocca WA, Knopman DS, Mielke MM, Lowe VJ, Senjem ML, Gunter JL, Preboske GM, Pankratz VS, Vemuri P, Petersen RC (2014) Age-specific population frequencies of cerebral beta-amyloidosis and neurodegeneration among people with normal cognitive function aged 50-89 years: a cross-sectional study. Lancet Neurol 13(10):997–1005
Jagust W (2009) Mapping brain beta-amyloid. Curr Opin Neurol 22(4):356–361
Jagust W, Reed B, Mungas D, Ellis W, Decarli C (2007) What does fluorodeoxyglucose PET imaging add to a clinical diagnosis of dementia? Neurology 69(9):871–877
Jagust WJ (2011) Amyloid imaging: liberal or conservative? Let the data decide. Arch Neurol 68(11):1377–1378
Jellinger, K. (1990). Morphology of Alzheimer disease and related disorders. Alzheimer disease: epidemiology, neuropathology, neurochemistry, and clinics. K. Maurer, P. Riederer and H. Beckmann. Berlin, Springer-Verlag: 61-77
Jellinger KA (2010) Con: can neuropathology really confirm the exact diagnosis? Alzheimers Res Ther 2(2):11
Jellinger KA, Attems J (2005) Prevalence and pathogenic role of cerebrovascular lesions in Alzheimer disease. J Neurol Sci 229–230:37–41
Jellinger KA, Bancher C (1998) Neuropathology of Alzheimer's disease: a critical update. J Neural Transm Suppl 54:77–95
Jessen F, Wiese B, Bachmann C, Eifflaender-Gorfer S, Haller F, Kolsch H, Luck T, Mosch E, van den Bussche H, Wagner M, Wollny A, Zimmermann T, Pentzek M, Riedel-Heller SG, Romberg HP, Weyerer S, Kaduszkiewicz H, Maier W, Bickel H, German Study on Aging, Cognition and Dementia in Primary Care Patients Study Group (2010) Prediction of dementia by subjective memory impairment: effects of severity and temporal association with cognitive impairment. Arch Gen Psychiatry 67(4):414–422
Johansson A, Savitcheva I, Forsberg A, Engler H, Langstrom B, Nordberg A, Askmark H (2008) [(11)C]-PIB imaging in patients with Parkinson's disease: preliminary results. Parkinsonism Relat Disord 14(4):345–347
Johnson AE, Jeppsson F, Sandell J, Wensbo D, Neelissen JA, Jureus A, Strom P, Norman H, Farde L, Svensson SP (2009) AZD2184: a radioligand for sensitive detection of beta-amyloid deposits. J Neurochem 108(5):1177–1186
Johnson KA (2006) Amyloid imaging of Alzheimer's disease using Pittsburgh compound B. Curr Neurol Neurosci Rep 6(6):496–503
Johnson KA, Gregas M, Becker JA, Kinnecom C, Salat DH, Moran EK, Smith EE, Rosand J, Rentz DM, Klunk WE, Mathis CA, Price JC, Dekosky ST, Fischman AJ, Greenberg SM (2007) Imaging of amyloid burden and distribution in cerebral amyloid angiopathy. Ann Neurol 62(3):229–234
Johnson KA, Minoshima S, Bohnen NI, Donohoe KJ, Foster NL, Herscovitch P, Karlawish JH, Rowe CC, Carrillo MC, Hartley DM, Hedrick S, Pappas V, Thies WH (2013) Appropriate use criteria for amyloid PET: a report of the Amyloid Imaging Task Force, the Society of Nuclear Medicine and Molecular Imaging, and the Alzheimer's Association. Alzheimers Dement 9(1):e1–e16
Johnson N, Davis T, Bosanquet N (2000) The epidemic of Alzheimer's disease. How can we manage the costs? PharmacoEconomics 18(3):215–223
Josephs KA, Holton JL, Rossor MN, Godbolt AK, Ozawa T, Strand K, Khan N, Al-Sarraj S, Revesz T (2004) Frontotemporal lobar degeneration and ubiquitin immunohistochemistry. Neuropathol Appl Neurobiol 30(4):369–373
Josephs KA, Whitwell JL, Ahmed Z, Shiung MM, Weigand SD, Knopman DS, Boeve BF, Parisi JE, Petersen RC, Dickson DW, Jack CR Jr (2008) Beta-amyloid burden is not associated with rates of brain atrophy. Ann Neurol 63(2):204–212
Jureus A, Swahn BM, Sandell J, Jeppsson F, Johnson AE, Johnstrom P, Neelissen JA, Sunnemark D, Farde L, Svensson SP (2010) Characterization of AZD4694, a novel fluorinated Abeta plaque neuroimaging PET radioligand. J Neurochem 114(3):784–794
Kadir A, Marutle A, Gonzalez D, Scholl M, Almkvist O, Mousavi M, Mustafiz T, Darreh-Shori T, Nennesmo I, Nordberg A (2011) Positron emission tomography imaging and clinical progression in relation to molecular pathology in the first Pittsburgh compound B positron emission tomography patient with Alzheimer's disease. Brain 134(Pt 1):301–317
Kalaitzakis ME, Walls AJ, Pearce RK, Gentleman SM (2011) Striatal Abeta peptide deposition mirrors dementia and differentiates DLB and PDD from other parkinsonian syndromes. Neurobiol Dis 41(2):377–384
Kambe T, Motoi Y, Ishii K, Hattori N (2010) Posterior cortical atrophy with [11C] Pittsburgh compound B accumulation in the primary visual cortex. J Neurol 257(3):469–471
Kantarci K, Lowe VJ, Chen Q, Przybelski SA, Lesnick TG, Schwarz CG, Senjem ML, Gunter JL, Jack CR Jr, Graff-Radford J, Jones DT, Knopman DS, Graff-Radford N, Ferman TJ, Parisi JE, Dickson DW, Petersen RC, Boeve BF, Murray ME (2020) Beta-amyloid PET and neuropathology in dementia with Lewy bodies. Neurology 94(3):e282–e291
Karran E, Mercken M, De Strooper B (2011) The amyloid cascade hypothesis for Alzheimer's disease: an appraisal for the development of therapeutics. Nat Rev Drug Discov 10(9):698–712
Kemppainen NM, Aalto S, Karrasch M, Nagren K, Savisto N, Oikonen V, Viitanen M, Parkkola R, Rinne JO (2008) Cognitive reserve hypothesis: Pittsburgh compound B and fluorodeoxyglucose positron emission tomography in relation to education in mild Alzheimer's disease. Ann Neurol 63(1):112–118
Kemppainen NM, Aalto S, Wilson IA, Nagren K, Helin S, Bruck A, Oikonen V, Kailajarvi M, Scheinin M, Viitanen M, Parkkola R, Rinne JO (2007) PET amyloid ligand [11C]PIB uptake is increased in mild cognitive impairment. Neurology 68(19):1603–1606
Kennedy AM, Frackowiak RS, Newman SK, Bloomfield P, Seaward J, Roques P, Lewington G, Cunningham VJ, Rossor MN (1995) Deficits in cerebral glucose metabolism demonstrated by positron emission tomography in individuals at risk of familial Alzheimer's disease. Neurosci Lett 186(1):17–20
Kepe V, Bordelon Y, Boxer A, Huang SC, Liu J, Thiede FC, Mazziotta JC, Mendez MF, Donoghue N, Small GW, Barrio JR (2013) PET imaging of neuropathology in tauopathies: progressive supranuclear palsy. J Alzheimers Dis 36(1):145–153
Khachaturian ZS (1985) Diagnosis of Alzheimer's disease. Arch Neurol 42(11):1097–1105
Kikuchi A, Takeda A, Okamura N, Tashiro M, Hasegawa T, Furumoto S, Kobayashi M, Sugeno N, Baba T, Miki Y, Mori F, Wakabayashi K, Funaki Y, Iwata R, Takahashi S, Fukuda H, Arai H, Kudo Y, Yanai K, Itoyama Y (2010) In vivo visualization of alpha-synuclein deposition by carbon-11-labelled 2-[2-(2-dimethylaminothiazol-5-yl)ethenyl]-6-[2-(fluoro)ethoxy]benzoxazole positron emission tomography in multiple system atrophy. Brain 133(Pt 6):1772–1778
Kim MJ, Shrestha SS, Cortes M, Singh P, Morse C, Liow JS, Gladding RL, Brouwer C, Henry K, Gallagher E, Tye GL, Zoghbi SS, Fujita M, Pike VW, Innis RB (2018) Evaluation of two potent and selective PET Radioligands to image COX-1 and COX-2 in rhesus monkeys. J Nucl Med 59(12):1907–1912
Klunk W, Cohen AD, Bi W, Weissfeld L, Aizenstein H, McDade E, Mountz J, Nebes R, Saxton J, Snitz B, Lopez O, Price J, Mathis C (2012) Why we need two cutoffs for amyloid imaging: early versus Alzheimer's-like amyloid-positivity. Alzheimers Dement 8(4):P453–P454
Klunk WE (2011) Amyloid imaging as a biomarker for cerebral beta-amyloidosis and risk prediction for Alzheimer dementia. Neurobiol Aging 32(Suppl 1):S20–S36
Klunk WE, Engler H, Nordberg A, Wang Y, Blomqvist G, Holt DP, Bergstrom M, Savitcheva I, Huang GF, Estrada S, Ausen B, Debnath ML, Barletta J, Price JC, Sandell J, Lopresti BJ, Wall A, Koivisto P, Antoni G, Mathis CA, Langstrom B (2004) Imaging brain amyloid in Alzheimer's disease with Pittsburgh compound-B. Ann Neurol 55(3):306–319
Klunk WE, Koeppe RA, Price JC, Benzinger TL, Devous MD Sr, Jagust WJ, Johnson KA, Mathis CA, Minhas D, Pontecorvo MJ, Rowe CC, Skovronsky DM, Mintun MA (2015) The Centiloid Project: standardizing quantitative amyloid plaque estimation by PET. Alzheimers Dement 11(1):1–15. e11–14
Klunk WE, Lopresti BJ, Ikonomovic MD, Lefterov IM, Koldamova RP, Abrahamson EE, Debnath ML, Holt DP, Huang GF, Shao L, DeKosky ST, Price JC, Mathis CA (2005) Binding of the positron emission tomography tracer Pittsburgh compound-B reflects the amount of amyloid-beta in Alzheimer's disease brain but not in transgenic mouse brain. J Neurosci 25(46):10598–10606
Klunk WE, Price JC, Mathis CA, Tsopelas ND, Lopresti BJ, Ziolko SK, Bi W, Hoge JA, Cohen AD, Ikonomovic MD, Saxton JA, Snitz BE, Pollen DA, Moonis M, Lippa CF, Swearer JM, Johnson KA, Rentz DM, Fischman AJ, Aizenstein HJ, DeKosky ST (2007) Amyloid deposition begins in the striatum of presenilin-1 mutation carriers from two unrelated pedigrees. J Neurosci 27(23):6174–6184
Klunk WE, Wang Y, Huang GF, Debnath ML, Holt DP, Mathis CA (2001) Uncharged thioflavin-T derivatives bind to amyloid-beta protein with high affinity and readily enter the brain. Life Sci 69(13):1471–1484
Klunk WE, Wang Y, Huang GF, Debnath ML, Holt DP, Shao L, Hamilton RL, Ikonomovic MD, DeKosky ST, Mathis CA (2003) The binding of 2-(4′-methylaminophenyl)benzothiazole to postmortem brain homogenates is dominated by the amyloid component. J Neurosci 23(6):2086–2092
Knudsen KA, Rosand J, Karluk D, Greenberg SM (2001) Clinical diagnosis of cerebral amyloid angiopathy: validation of the Boston criteria. Neurology 56(4):537–539
Koeppe RA, Gilman S, Junck L, Wernette K, Frey KA (2008) Differentiating Alzheimer's disease from dementia with Lewy bodies and Parkinson's disease with (+)-[11C]dihydrotetrabenazine positron emission tomography. Alzheimers Dement 4(1 Suppl 1):S67–S76
Koivunen J, Pirttila T, Kemppainen N, Aalto S, Herukka SK, Jauhianen AM, Hanninen T, Hallikainen M, Nagren K, Rinne JO, Soininen H (2008a) PET amyloid ligand [C]PIB uptake and cerebrospinal fluid beta-amyloid in mild cognitive impairment. Dement Geriatr Cogn Disord 26(4):378–383
Koivunen J, Verkkoniemi A, Aalto S, Paetau A, Ahonen JP, Viitanen M, Nagren K, Rokka J, Haaparanta M, Kalimo H, Rinne JO (2008b) PET amyloid ligand [11C]PIB uptake shows predominantly striatal increase in variant Alzheimer's disease. Brain 131(Pt 7):1845–1853
Kreisl WC, Henter ID, Innis RB (2018) Imaging translocator protein as a biomarker of Neuroinflammation in dementia. Adv Pharmacol 82:163–185
Kreisl WC, Jenko KJ, Hines CS, Lyoo CH, Corona W, Morse CL, Zoghbi SS, Hyde T, Kleinman JE, Pike VW, McMahon FJ, Innis RB, Biomarkers Consortium PET Radioligand Project Team (2013) A genetic polymorphism for translocator protein 18 kDa affects both in vitro and in vivo radioligand binding in human brain to this putative biomarker of neuroinflammation. J Cereb Blood Flow Metab 33(1):53–58
Kuczynski B, Reed B, Mungas D, Weiner M, Chui HC, Jagust W (2008) Cognitive and anatomic contributions of metabolic decline in Alzheimer disease and cerebrovascular disease. Arch Neurol 65(5):650–655
Kudo Y (2006) Development of amyloid imaging PET probes for an early diagnosis of Alzheimer's disease. Minim Invasive Ther Allied Technol 15(4):209–213
Kudo Y, Okamura N, Furumoto S, Tashiro M, Furukawa K, Maruyama M, Itoh M, Iwata R, Yanai K, Arai H (2007) 2-(2-[2-Dimethylaminothiazol-5-yl]Ethenyl)-6- (2-[Fluoro]Ethoxy)Benzoxazole: a novel PET agent for in vivo detection of dense amyloid plaques in Alzheimer's disease patients. J Nucl Med 48(4):553–561
La Joie R, Ayakta N, Borys E, Boxer AL, deCarli C, Dore V, Grinberg LT, Huang EJ, Jack JCR, Jin LW, Klunk WE, Ikonomovic M, Lockhart SM, Lowe VL, Masters CL, Miller BL, Mungas DM, Murray ME, O’Neil J, Olichney JM, Petersen RC, Reed BR, Rowe CC, Seeley WW, Vemuri P, Villemagne VL, Jagust WJ, Rabinovici GD (2018). Multi-site study of PiB-PET imaging using the Centiloid method: relationships to pathological measures of β-amyloid pathology. 12th human amyloid imaging. Miami Beach, FL. 12: 148–149
La Joie R, Visani AV, Baker SL, Brown JA, Bourakova V, Cha J, Chaudhary K, Edwards L, Iaccarino L, Janabi M, Lesman-Segev OH, Miller ZA, Perry DC, O'Neil JP, Pham J, Rojas JC, Rosen HJ, Seeley WW, Tsai RM, Miller BL, Jagust WJ, Rabinovici GD (2020) Prospective longitudinal atrophy in Alzheimer's disease correlates with the intensity and topography of baseline tau-PET. Sci Transl Med 12(524):eaau5732
Landau SM, Fero A, Baker SL, Koeppe R, Mintun M, Chen K, Reiman EM, Jagust WJ (2015) Measurement of longitudinal beta-amyloid change with 18F-Florbetapir PET and standardized uptake value ratios. J Nucl Med 56(4):567–574
Landau SM, Harvey D, Madison CM, Koeppe RA, Reiman EM, Foster NL, Weiner MW, Jagust WJ (2009) Associations between cognitive, functional, and FDG-PET measures of decline in AD and MCI. Neurobiol Aging 32(7):1207–1218
Landau SM, Horng A, Jagust WJ, Alzheimer's Disease Neuroimaging Initiative (2018) Memory decline accompanies subthreshold amyloid accumulation. Neurology 90(17):e1452–e1460
Landau SM, Lu M, Joshi AD, Pontecorvo M, Mintun MA, Trojanowski JQ, Shaw LM, Jagust WJ, Alzheimer's Disease Neuroimaging (2013) Comparing positron emission tomography imaging and cerebrospinal fluid measurements of beta-amyloid. Ann Neurol 74(6):826–836
Landau SM, Villemagne VL (2020) Can amyloid PET differentiate "pure" LBD from AD with or without LBD copathology? Neurology 94(3):103–104
Larson EB, Edwards JK, O'Meara E, Nochlin D, Sumi SM (1996) Neuropathologic diagnostic outcomes from a cohort of outpatients with suspected dementia. J Gerontol A Biol Sci Med Sci 51(suppl 6):M313–M318
Laruelle M, Slifstein M, Huang Y (2003) Relationships between radiotracer properties and image quality in molecular imaging of the brain with positron emission tomography. Mol Imaging Biol 5(6):363–375
Leinonen V, Alafuzoff I, Aalto S, Suotunen T, Savolainen S, Nagren K, Tapiola T, Pirttila T, Rinne J, Jaaskelainen JE, Soininen H, Rinne JO (2008a) Assessment of β-amyloid in a frontal cortical brain biopsy specimen and by positron emission tomography with carbon 11-labeled Pittsburgh compound B. Arch Neurol 65(10):1304–1309
Leinonen V, Alafuzoff I, Aalto S, Suotunen T, Savolainen S, Nagren K, Tapiola T, Pirttila T, Rinne J, Jaaskelainen JE, Soininen H, Rinne JO (2008b) Assessment of beta-amyloid in a frontal cortical brain biopsy specimen and by positron emission tomography with carbon 11-labeled Pittsburgh compound B. Arch Neurol 65(10):1304–1309
Leuner K, Hauptmann S, Abdel-Kader R, Scherping I, Keil U, Strosznajder JB, Eckert A, Muller WE (2007) Mitochondrial dysfunction: the first domino in brain aging and Alzheimer's disease? Antioxid Redox Signal 9(10):1659–1675
Leuzy A, Savitcheva I, Chiotis K, Lilja J, Andersen P, Bogdanovic N, Jelic V, Nordberg A (2019) Clinical impact of [(18)F]flutemetamol PET among memory clinic patients with an unclear diagnosis. Eur J Nucl Med Mol Imaging 46(6):1276–1286
LeVine H 3rd (1999) Quantification of beta-sheet amyloid fibril structures with thioflavin T. Methods Enzymol 309:274–284
Levine H 3rd, Walker LC (2008) Molecular polymorphism of Abeta in Alzheimer's disease. Neurobiol Aging 31(4):542–548
Levine H 3rd, Walker LC (2010) Molecular polymorphism of Abeta in Alzheimer's disease. Neurobiol Aging 31(4):542–548
Leyton CE, Villemagne VL, Savage S, Pike KE, Ballard KJ, Piguet O, Burrell JR, Rowe CC, Hodges JR (2011) Subtypes of progressive aphasia: application of the international consensus criteria and validation using {beta}-amyloid imaging. Brain 134(Pt 10):3030–3043
Li QX, Villemagne VL, Doecke JD, Rembach A, Sarros S, Varghese S, McGlade A, Laughton KM, Pertile KK, Fowler CJ, Rumble RL, Trounson BO, Taddei K, Rainey-Smith SR, Laws SM, Robertson JS, Evered LA, Silbert B, Ellis KA, Rowe CC, Macaulay SL, Darby D, Martins RN, Ames D, Masters CL, Collins S, A. R. Group (2015) Alzheimer's disease normative cerebrospinal fluid biomarkers validated in PET amyloid-beta characterized subjects from the Australian imaging, biomarkers and lifestyle (AIBL) study. J Alzheimers Dis 48(1):175–187
Liberski PP (2004) Amyloid plaques in transmissible spongiform encephalopathies (prion diseases). Folia Neuropathol 42(Suppl B):109–119
Lin KS, Debnath ML, Mathis CA, Klunk WE (2009) Synthesis and beta-amyloid binding properties of rhenium 2-phenylbenzothiazoles. Bioorg Med Chem Lett 19(8):2258–2262
Lister-James J, Pontecorvo MJ, Clark C, Joshi AD, Mintun MA, Zhang W, Lim N, Zhuang Z, Golding G, Choi SR, Benedum TE, Kennedy P, Hefti F, Carpenter AP, Kung HF, Skovronsky DM (2011) Florbetapir f-18: a histopathologically validated Beta-amyloid positron emission tomography imaging agent. Semin Nucl Med 41(4):300–304
Liu E, Schmidt ME, Margolin R, Sperling R, Koeppe R, Mason NS, Klunk WE, Mathis CA, Salloway S, Fox NC, Hill DL, Les AS, Collins P, Gregg KM, Di J, Lu Y, Tudor IC, Wyman BT, Booth K, Broome S, Yuen E, Grundman M, Brashear HR, Bapineuzumab 301 and 302 Clinical Trial Investigators (2015) Amyloid-beta 11C-PiB-PET imaging results from 2 randomized bapineuzumab phase 3 AD trials. Neurology 85(8):692–700
LoBue C, Munro C, Schaffert J, Didehbani N, Hart J, Batjer H, Cullum CM (2019) Traumatic brain injury and risk of Long-term brain changes, accumulation of pathological markers, and developing dementia: a review. J Alzheimers Dis 70(3):629–654
Lockhart A, Lamb JR, Osredkar T, Sue LI, Joyce JN, Ye L, Libri V, Leppert D, Beach TG (2007) PIB is a non-specific imaging marker of amyloid-beta (Abeta) peptide-related cerebral amyloidosis. Brain 130(Pt 10):2607–2615
Lockhart A, Ye L, Judd DB, Merritt AT, Lowe PN, Morgenstern JL, Hong G, Gee AD, Brown J (2005) Evidence for the presence of three distinct binding sites for the thioflavin T class of Alzheimer's disease PET imaging agents on beta-amyloid peptide fibrils. J Biol Chem 280(9):7677–7684
Long JM, Holtzman DM (2019) Alzheimer disease: An update on pathobiology and treatment strategies. Cell 179(2):312–339
Lopresti BJ, Klunk WE, Mathis CA, Hoge JA, Ziolko SK, Lu X, Meltzer CC, Schimmel K, Tsopelas ND, DeKosky ST, Price JC (2005) Simplified quantification of Pittsburgh compound B amyloid imaging PET studies: a comparative analysis. J Nucl Med 46(12):1959–1972
Lowe VJ, Kemp BJ, Jack CR Jr, Senjem M, Weigand S, Shiung M, Smith G, Knopman D, Boeve B, Mullan B, Petersen RC (2009) Comparison of 18F-FDG and PiB PET in cognitive impairment. J Nucl Med 50(6):878–886
Lowe VJ, Lundt E, Knopman D, Senjem ML, Gunter JL, Schwarz CG, Kemp BJ, Jack CR Jr, Petersen RC (2017) Comparison of [(18)F]Flutemetamol and [(11)C]Pittsburgh compound-B in cognitively normal young, cognitively normal elderly, and Alzheimer's disease dementia individuals. Neuroimage Clin 16:295–302
Mackenzie IR, Foti D, Woulfe J, Hurwitz TA (2008) Atypical frontotemporal lobar degeneration with ubiquitin-positive, TDP-43-negative neuronal inclusions. Brain 131(Pt 5):1282–1293
Maeda J, Ji B, Irie T, Tomiyama T, Maruyama M, Okauchi T, Staufenbiel M, Iwata N, Ono M, Saido TC, Suzuki K, Mori H, Higuchi M, Suhara T (2007) Longitudinal, quantitative assessment of amyloid, neuroinflammation, and anti-amyloid treatment in a living mouse model of Alzheimer's disease enabled by positron emission tomography. J Neurosci 27(41):10957–10968
Maetzler W, Liepelt I, Reimold M, Reischl G, Solbach C, Becker C, Schulte C, Leyhe T, Keller S, Melms A, Gasser T, Berg D (2009) Cortical PIB binding in Lewy body disease is associated with Alzheimer-like characteristics. Neurobiol Dis 34(1):107–112
Maetzler W, Reimold M, Liepelt I, Solbach C, Leyhe T, Schweitzer K, Eschweiler GW, Mittelbronn M, Gaenslen A, Uebele M, Reischl G, Gasser T, Machulla HJ, Bares R, Berg D (2008) [11C]PIB binding in Parkinson's disease dementia. NeuroImage 39(3):1027–1033
Maezawa I, Hong HS, Liu R, Wu CY, Cheng RH, Kung MP, Kung HF, Lam KS, Oddo S, Laferla FM, Jin LW (2008) Congo red and thioflavin-T analogs detect Abeta oligomers. J Neurochem 104(2):457–468
Majocha RE, Reno JM, Friedland RP, Van Haight C, Lyle LR, Marotta CA (1992) Development of a monoclonal antibody specific for ß/A4 amyloid in Alzheimer's disease brain for application to in vivo imaging of amyloid angiopathy. J Nucl Med 33(12):2184–2189
Malkki H (2014) Traumatic brain injury: PET imaging detects amyloid deposits after TBI. Nat Rev Neurol 10(1):3
Manca C, Hopes L, Kearney-Schwartz A, Roch V, Karcher G, Baumann C, Marie PY, Malaplate-Armand C, Jonveaux TR, Verger A (2019) Assessment of 18F-Florbetaben amyloid PET imaging in patients with suspected Alzheimer's disease and isolated increase in cerebrospinal fluid tau proteins. J Alzheimers Dis 68(3):1061–1069
Marchant NL, Reed BR, Decarli CS, Madison CM, Weiner MW, Chui HC, Jagust WJ (2011) Cerebrovascular disease, beta-amyloid, and cognition in aging. Neurobiol Aging 33(5):1006.e25-36
Maruyama M, Maeda J, Ji B, Zhang MY, Okauchi T, Ono M, Hattori S, Trojanoswki JQ, Lee VM, Fukumura T, Higuchi M, Suhara T (2009) In-vivo optical and PET detections of fibrillar tau lesions in a mouse model of tauopathies. Alzheimers Dement 55(4, Supplement 1):P209–P210. [abstract]
Mashima K, Ito D, Kameyama M, Osada T, Tabuchi H, Nihei Y, Yoshizaki T, Noguchi E, Tanikawa M, Iizuka T, Date Y, Ogata Y, Nakahara T, Iwabuchi Y, Jinzaki M, Murakami K, Suzuki N (2017) Extremely low prevalence of amyloid positron emission tomography positivity in Parkinson's disease without dementia. Eur Neurol 77(5–6):231–237
Masliah E, Rockenstein E, Veinbergs I, Sagara Y, Mallory M, Hashimoto M, Mucke L (2001) Beta-amyloid peptides enhance alpha-synuclein accumulation and neuronal deficits in a transgenic mouse model linking Alzheimer's disease and Parkinson's disease. Proc Natl Acad Sci U S A 98(21):12245–12250
Masters CL (2005) Neuropathology of Alzheimer's disease. In: Burns A, O'Brien J, Ames D (eds) Dementia, 3rd edn. Hodder Arnold, London, pp 393–407
Masters CL, Beyreuther K (2005) The neuropathology of Alzheimer's disease in the year 2005. In: Beal MF, Lang AE, Ludolph AC (eds) Neurodegenerative diseases: neurobiology, pathogenesis and therapeutics. Cambridge University Press, Cambridge, pp 433–440
Masters CL, Beyreuther K (2006) Alzheimer's centennial legacy: prospects for rational therapeutic intervention targeting the Abeta amyloid pathway. Brain 129(Pt 11):2823–2839
Masters CL, Cappai R, Barnham KJ, Villemagne VL (2006) Molecular mechanisms for Alzheimer's disease: implications for neuroimaging and therapeutics. J Neurochem 97(6):1700–1725
Masters CL, Simms G, Weinman NA, Multhaup G, McDonald BL, Beyreuther K (1985) Amyloid plaque core protein in Alzheimer disease and down syndrome. Proc Natl Acad Sci U S A 82(12):4245–4249
Mathis CA, Bacskai BJ, Kajdasz ST, McLellan ME, Frosch MP, Hyman BT, Holt DP, Wang Y, Huang GF, Debnath ML, Klunk WE (2002) A lipophilic thioflavin-T derivative for positron emission tomography (PET) imaging of amyloid in brain. Bioorg Med Chem Lett 12(3):295–298
Mathis CA, Klunk WE, Price JC, DeKosky ST (2005) Imaging technology for neurodegenerative diseases: progress toward detection of specific pathologies. Arch Neurol 62(2):196–200
Mathis CA, Lopresti BJ, Klunk WE (2007) Impact of amyloid imaging on drug development in Alzheimer's disease. Nucl Med Biol 34(7):809–822
Matias-Guiu JA, Guerrero-Marquez C, Cabrera-Martin MN, Gomez-Pinedo U, Romeral M, Mayo D, Porta-Etessam J, Moreno-Ramos T, Carreras JL, Matias-Guiu J (2017) Amyloid- and FDG-PET in sporadic Creutzfeldt-Jakob disease: correlation with pathological prion protein in neuropathology. Prion 11(3):205–213
Mattsson N, Andreasson U, Persson S, Arai H, Batish SD, Bernardini S, Bocchio-Chiavetto L, Blankenstein MA, Carrillo MC, Chalbot S, Coart E, Chiasserini D, Cutler N, Dahlfors G, Duller S, Fagan AM, Forlenza O, Frisoni GB, Galasko D, Galimberti D, Hampel H, Handberg A, Heneka MT, Herskovits AZ, Herukka SK, Holtzman DM, Humpel C, Hyman BT, Iqbal K, Jucker M, Kaeser SA, Kaiser E, Kapaki E, Kidd D, Klivenyi P, Knudsen CS, Kummer MP, Lui J, Llado A, Lewczuk P, Li QX, Martins R, Masters C, McAuliffe J, Mercken M, Moghekar A, Molinuevo JL, Montine TJ, Nowatzke W, O'Brien R, Otto M, Paraskevas GP, Parnetti L, Petersen RC, Prvulovic D, de Reus HP, Rissman RA, Scarpini E, Stefani A, Soininen H, Schroder J, Shaw LM, Skinningsrud A, Skrogstad B, Spreer A, Talib L, Teunissen C, Trojanowski JQ, Tumani H, Umek RM, Van Broeck B, Vanderstichele H, Vecsei L, Verbeek MM, Windisch M, Zhang J, Zetterberg H, Blennow K (2011) The Alzheimer's Association external quality control program for cerebrospinal fluid biomarkers. Alzheimers Dement 7(4):386–395. e386
Mattsson N, Lonneborg A, Boccardi M, Blennow K, Hansson O, Geneva Task Force for the Roadmap of Alzheimer's Biomarkers (2017) Clinical validity of cerebrospinal fluid Abeta42, tau, and phospho-tau as biomarkers for Alzheimer's disease in the context of a structured 5-phase development framework. Neurobiol Aging 52:196–213
Mattsson N, Zetterberg H, Hansson O, Andreasen N, Parnetti L, Jonsson M, Herukka SK, van der Flier WM, Blankenstein MA, Ewers M, Rich K, Kaiser E, Verbeek M, Tsolaki M, Mulugeta E, Rosen E, Aarsland D, Visser PJ, Schroder J, Marcusson J, de Leon M, Hampel H, Scheltens P, Pirttila T, Wallin A, Jonhagen ME, Minthon L, Winblad B, Blennow K (2009) CSF biomarkers and incipient Alzheimer disease in patients with mild cognitive impairment. JAMA 302(4):385–393
Mawuenyega KG, Sigurdson W, Ovod V, Munsell L, Kasten T, Morris JC, Yarasheski KE, Bateman RJ (2010) Decreased clearance of CNS beta-amyloid in Alzheimer's disease. Science 330(6012):1774
McDade E, Wang G, Gordon BA, Hassenstab J, Benzinger TLS, Buckles V, Fagan AM, Holtzman DM, Cairns NJ, Goate AM, Marcus DS, Morris JC, Paumier K, Xiong C, Allegri R, Berman SB, Klunk W, Noble J, Ringman J, Ghetti B, Farlow M, Sperling RA, Chhatwal J, Salloway S, Graff-Radford NR, Schofield PR, Masters C, Rossor MN, Fox NC, Levin J, Jucker M, Bateman RJ, Dominantly Inherited Alzheimer Network (2018) Longitudinal cognitive and biomarker changes in dominantly inherited Alzheimer disease. Neurology 91(14):e1295–e1306
McKee AC, Abdolmohammadi B, Stein TD (2018) The neuropathology of chronic traumatic encephalopathy. Handb Clin Neurol 158:297–307
McKee AC, Stern RA, Nowinski CJ, Stein TD, Alvarez VE, Daneshvar DH, Lee HS, Wojtowicz SM, Hall G, Baugh CM, Riley DO, Kubilus CA, Cormier KA, Jacobs MA, Martin BR, Abraham CR, Ikezu T, Reichard RR, Wolozin BL, Budson AE, Goldstein LE, Kowall NW, Cantu RC (2013) The spectrum of disease in chronic traumatic encephalopathy. Brain 136(Pt 1):43–64
McKeith IG, Dickson DW, Lowe J, Emre M, O'Brien JT, Feldman H, Cummings J, Duda JE, Lippa C, Perry EK, Aarsland D, Arai H, Ballard CG, Boeve B, Burn DJ, Costa D, Del Ser T, Dubois B, Galasko D, Gauthier S, Goetz CG, Gomez-Tortosa E, Halliday G, Hansen LA, Hardy J, Iwatsubo T, Kalaria RN, Kaufer D, Kenny RA, Korczyn A, Kosaka K, Lee VM, Lees A, Litvan I, Londos E, Lopez OL, Minoshima S, Mizuno Y, Molina JA, Mukaetova-Ladinska EB, Pasquier F, Perry RH, Schulz JB, Trojanowski JQ, Yamada M (2005) Diagnosis and management of dementia with Lewy bodies: third report of the DLB consortium. Neurology 65(12):1863–1872
McKeith IG, Mosimann UP (2004) Dementia with Lewy bodies and Parkinson's disease. Parkinsonism Relat Disord 10(Suppl 1):S15–S18
McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH, Klunk WE, Koroshetz WJ, Manly JJ, Mayeux R, Mohs RC, Morris JC, Rossor MN, Scheltens P, Carrillo MC, Thies B, Weintraub S, Phelps CH (2011) The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement 7(3):263–269
McLean CA, Cherny RA, Fraser FW, Fuller SJ, Smith MJ, Beyreuther K, Bush AI, Masters CL (1999) Soluble pool of Aß amyloid as a determinant of severity of neurodegeneration in Alzheimer's disease. Ann Neurol 46(6):860–866
Michaelis ML, Dobrowsky RT, Li G (2002) Tau neurofibrillary pathology and microtubule stability. J Mol Neurosci 19(3):289–293
Mielke R, Herholz K, Grond M, Kessler J, Heiss WD (1992) Differences of regional cerebral glucose metabolism between presenile and senile dementia of Alzheimer type. Neurobiol Aging 13(1):93–98
Mintun MA, Larossa GN, Sheline YI, Dence CS, Lee SY, Mach RH, Klunk WE, Mathis CA, DeKosky ST, Morris JC (2006) [11C]PIB in a nondemented population: potential antecedent marker of Alzheimer disease. Neurology 67(3):446–452
Montine TJ, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Dickson DW, Duyckaerts C, Frosch MP, Masliah E, Mirra SS, Nelson PT, Schneider JA, Thal DR, Trojanowski JQ, Vinters HV, Hyman BT (2012) National Institute on Aging-Alzheimer's Association guidelines for the neuropathologic assessment of Alzheimer's disease: a practical approach. Acta Neuropathol 123(1):1–11
Mormino EC, Brandel MG, Madison CM, Rabinovici GD, Marks S, Baker SL, Jagust WJ (2012) Not quite PIB-positive, not quite PIB-negative: slight PIB elevations in elderly normal control subjects are biologically relevant. NeuroImage 59(2):1152–1160
Mormino EC, Kluth JT, Madison CM, Rabinovici GD, Baker SL, Miller BL, Koeppe RA, Mathis CA, Weiner MW, Jagust WJ (2009) Episodic memory loss is related to hippocampal-mediated beta-amyloid deposition in elderly subjects. Brain 132(Pt 5):1310–1323
Morris JC (2012) Revised criteria for mild cognitive impairment may compromise the diagnosis of Alzheimer disease dementia. Arch Neurol 69(6):700–708. https://doi.org/10.1001/archneurol.2011.3152
Morris JC, Aisen PS, Bateman RJ, Benzinger TL, Cairns NJ, Fagan AM, Ghetti B, Goate AM, Holtzman DM, Klunk WE, McDade E, Marcus DS, Martins RN, Masters CL, Mayeux R, Oliver A, Quaid K, Ringman JM, Rossor MN, Salloway S, Schofield PR, Selsor NJ, Sperling RA, Weiner MW, Xiong C, Moulder KL, Buckles VD (2012) Developing an international network for Alzheimer research: the dominantly inherited Alzheimer network. Clin Investig (Lond) 2(10):975–984
Morris JC, Kimberly A, Quaid K, Holtzman DM, Kantarci K, Kaye J, Reiman EM, Klunk WE, Siemers ER (2005) Role of biomarkers in studies of presymptomatic Alzheimer's disease. Alzheimers Dement 1(2):145–151
Morris JC, Price AL (2001) Pathologic correlates of nondemented aging, mild cognitive impairment, and early-stage Alzheimer's disease. J Mol Neurosci 17(2):101–118
Morris JC, Roe CM, Grant EA, Head D, Storandt M, Goate AM, Fagan AM, Holtzman DM, Mintun MA (2009) Pittsburgh compound B imaging and prediction of progression from cognitive normality to symptomatic Alzheimer disease. Arch Neurol 66(12):1469–1475
Morris JC, Roe CM, Xiong C, Fagan AM, Goate AM, Holtzman DM, Mintun MA (2010) APOE predicts amyloid-beta but not tau Alzheimer pathology in cognitively normal aging. Ann Neurol 67(1):122–131
Mortimer JA (1997) Brain reserve and the clinical expression of Alzheimer's disease. Geriatrics 52(Suppl 2):S50–S53
Mosconi L, Sorbi S, de Leon MJ, Li Y, Nacmias B, Myoung PS, Tsui W, Ginestroni A, Bessi V, Fayyazz M, Caffarra P, Pupi A. (2006) Hypometabolism exceeds atrophy in presymptomatic early-onset familial Alzheimer’sdisease. J Nucl Med 47(11):1778–1786
Mosconi L, De Santi S, Li Y, Li J, Zhan J, Tsui WH, Boppana M, Pupi A, de Leon MJ (2006a) Visual rating of medial temporal lobe metabolism in mild cognitive impairment and Alzheimer's disease using FDG-PET. Eur J Nucl Med Mol Imaging 33(2):210–221
Mosconi L, Perani D, Sorbi S, Herholz K, Nacmias B, Holthoff V, Salmon E, Baron JC, De Cristofaro MT, Padovani A, Borroni B, Franceschi M, Bracco L, Pupi A (2004) MCI conversion to dementia and the APOE genotype: a prediction study with FDG-PET. Neurology 63(12):2332–2340
Mosconi L, Sorbi S, de Leon MJ, Li Y, Nacmias B, Myoung PS, Tsui W, Ginestroni A, Bessi V, Fayyazz M, Caffarra P, Pupi A (2006b) Hypometabolism exceeds atrophy in presymptomatic early-onset familial Alzheimer's disease. J Nucl Med 47(11):1778–1786
Mott RT, Dickson DW, Trojanowski JQ, Zhukareva V, Lee VM, Forman M, Van Deerlin V, Ervin JF, Wang DS, Schmechel DE, Hulette CM (2005) Neuropathologic, biochemical, and molecular characterization of the frontotemporal dementias. J Neuropathol Exp Neurol 64(5):420–428
Mountz JM, Laymon CM, Cohen AD, Zhang Z, Price JC, Boudhar S, McDade E, Aizenstein HJ, Klunk WE, Mathis CA (2015) Comparison of qualitative and quantitative imaging characteristics of [11C]PiB and [18F]flutemetamol in normal control and Alzheimer's subjects. Neuroimage Clin 9:592–598
Nakamura A, Kaneko N, Villemagne VL, Kato T, Doecke J, Doré V, Fowler C, Li Q-X, Martins R, Rowe C (2018) High performance plasma amyloid-β biomarkers for Alzheimer’s disease. Nature 554(7691):249
Naslund J, Haroutunian V, Mohs R, Davis KL, Davies P, Greengard P, Buxbaum JD (2000) Correlation between elevated levels of amyloid beta-peptide in the brain and cognitive decline. JAMA 283(12):1571–1577
Navitsky M, Joshi AD, Kennedy I, Klunk WE, Rowe CC, Wong DF, Pontecorvo MJ, Mintun MA, Devous MD Sr (2018) Standardization of amyloid quantitation with florbetapir standardized uptake value ratios to the Centiloid scale. Alzheimers Dement 14(12):1565–1571
Nelissen N, Van Laere K, Thurfjell L, Owenius R, Vandenbulcke M, Koole M, Bormans G, Brooks DJ, Vandenberghe R (2009) Phase 1 study of the Pittsburgh compound B derivative 18F-flutemetamol in healthy volunteers and patients with probable Alzheimer disease. J Nucl Med 50(8):1251–1259
Nelson PT, Dickson DW, Trojanowski JQ, Jack CR, Boyle PA, Arfanakis K, Rademakers R, Alafuzoff I, Attems J, Brayne C, Coyle-Gilchrist ITS, Chui HC, Fardo DW, Flanagan ME, Halliday G, Hokkanen SRK, Hunter S, Jicha GA, Katsumata Y, Kawas CH, Keene CD, Kovacs GG, Kukull WA, Levey AI, Makkinejad N, Montine TJ, Murayama S, Murray ME, Nag S, Rissman RA, Seeley WW, Sperling RA, White Iii CL, Yu L, Schneider JA (2019) Limbic-predominant age-related TDP-43 encephalopathy (LATE): consensus working group report. Brain 142(6):1503–1527
Neumann M, Rademakers R, Roeber S, Baker M, Kretzschmar HA, Mackenzie IR (2009) A new subtype of frontotemporal lobar degeneration with FUS pathology. Brain 132(Pt 11):2922–2931
Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT, Bruce J, Schuck T, Grossman M, Clark CM, McCluskey LF, Miller BL, Masliah E, Mackenzie IR, Feldman H, Feiden W, Kretzschmar HA, Trojanowski JQ, Lee VM (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314(5796):130–133
Newberg AB, Wintering NA, Plossl K, Hochold J, Stabin MG, Watson M, Skovronsky D, Clark CM, Kung MP, Kung HF (2006) Safety, biodistribution, and dosimetry of 123I-IMPY: a novel amyloid plaque-imaging agent for the diagnosis of Alzheimer's disease. J Nucl Med 47(5):748–754
Ng S, Villemagne VL, Berlangieri S, Lee ST, Cherk M, Gong SJ, Ackermann U, Saunder T, Tochon-Danguy H, Jones G, Smith C, O'Keefe G, Masters CL, Rowe CC (2007a) Visual assessment versus quantitative assessment of 11C-PIB PET and 18F-FDG PET for detection of Alzheimer's disease. J Nucl Med 48(4):547–552
Ng SY, Villemagne VL, Masters CL, Rowe CC (2007b) Evaluating atypical dementia syndromes using positron emission tomography with carbon 11 labeled Pittsburgh compound B. Arch Neurol 64(8):1140–1144
Ni R, Gillberg PG, Bergfors A, Marutle A, Nordberg A (2013) Amyloid tracers detect multiple binding sites in Alzheimer's disease brain tissue. Brain 136(Pt 7):2217–2227
Noda A, Murakami Y, Nishiyama S, Fukumoto D, Miyoshi S, Tsukada H, Nishimura S (2008) Amyloid imaging in aged and young macaques with [11C]PIB and [18F]FDDNP. Synapse 62(6):472–475
Nordberg A (2004) PET imaging of amyloid in Alzheimer's disease. Lancet Neurol 3(9):519–527
Nordberg A (2007) Amyloid imaging in Alzheimer's disease. Curr Opin Neurol 20(4):398–402
Nyberg S, Jonhagen ME, Cselenyi Z, Halldin C, Julin P, Olsson H, Freund-Levi Y, Andersson J, Varnas K, Svensson S, Farde L (2009) Detection of amyloid in Alzheimer's disease with positron emission tomography using [11C]AZD2184. Eur J Nucl Med Mol Imaging 36(11):1859–1863
O'Brien J, Ames D, Burns A (2000) Dementia, 2nd edn. Arnold, London
O'Brien JT, Colloby S, Fenwick J, Williams ED, Firbank M, Burn D, Aarsland D, McKeith IG (2004) Dopamine transporter loss visualized with FP-CIT SPECT in the differential diagnosis of dementia with Lewy bodies. Arch Neurol 61(6):919–925
Ojida A, Sakamoto T, Inoue MA, Fujishima SH, Lippens G, Hamachi I (2009) Fluorescent BODIPY-based Zn(II) complex as a molecular probe for selective detection of neurofibrillary tangles in the brains of Alzheimer's disease patients. J Am Chem Soc 131(18):6543–6548
Okamura N, Shiga Y, Furumoto S, Tashiro M, Tsuboi Y, Furukawa K, Yanai K, Iwata R, Arai H, Kudo Y, Itoyama Y, Doh-ura K (2010) In vivo detection of prion amyloid plaques using [(11)C]BF-227 PET. Eur J Nucl Med Mol Imaging 37(5):934–941
Okamura N, Suemoto T, Furumoto S, Suzuki M, Shimadzu H, Akatsu H, Yamamoto T, Fujiwara H, Nemoto M, Maruyama M, Arai H, Yanai K, Sawada T, Kudo Y (2005) Quinoline and benzimidazole derivatives: candidate probes for in vivo imaging of tau pathology in Alzheimer's disease. J Neurosci 25(47):10857–10862
Okamura N, Suemoto T, Shimadzu H, Suzuki M, Shiomitsu T, Akatsu H, Yamamoto T, Staufenbiel M, Yanai K, Arai H, Sasaki H, Kudo Y, Sawada T (2004) Styrylbenzoxazole derivatives for in vivo imaging of amyloid plaques in the brain. J Neurosci 24(10):2535–2541
Okello A, Edison P, Archer HA, Turkheimer FE, Kennedy J, Bullock R, Walker Z, Kennedy A, Fox N, Rossor M, Brooks DJ (2009a) Microglial activation and amyloid deposition in mild cognitive impairment: a PET study. Neurology 72(1):56–62
Okello A, Koivunen J, Edison P, Archer HA, Turkheimer FE, Nagren K, Bullock R, Walker Z, Kennedy A, Fox NC, Rossor MN, Rinne JO, Brooks DJ (2009b) Conversion of amyloid positive and negative MCI to AD over 3 years: an 11C-PIB PET study. Neurology 73(10):754–760
Omalu B, Small GW, Bailes J, Ercoli LM, Merrill DA, Wong KP, Huang SC, Satyamurthy N, Hammers JL, Lee J, Fitzsimmons RP, Barrio JR (2018) Postmortem autopsy-confirmation of Antemortem [F-18]FDDNP-PET scans in a football player with chronic traumatic encephalopathy. Neurosurgery 82(2):237–246
Ong K, Villemagne VL, Bahar-Fuchs A, Lamb F, Chetelat G, Raniga P, Mulligan RS, Salvado O, Putz B, Roth K, Masters CL, Reininger CB, Rowe CC (2013) (18)F-florbetaben Abeta imaging in mild cognitive impairment. Alzheimers Res Ther 5(1):4
Ong KT, Villemagne VL, Bahar-Fuchs A, Lamb F, Langdon N, Catafau AM, Stephens AW, Seibyl J, Dinkelborg LM, Reininger CB, Putz B, Rohde B, Masters CL, Rowe CC (2015) Abeta imaging with 18F-florbetaben in prodromal Alzheimer's disease: a prospective outcome study. J Neurol Neurosurg Psychiatry 86(4):431–436
Opazo C, Luza S, Villemagne VL, Volitakis I, Rowe C, Barnham KJ, Strozyk D, Masters CL, Cherny RA, Bush AI (2006) Radioiodinated clioquinol as a biomarker for beta-amyloid: Zn complexes in Alzheimer's disease. Aging Cell 5(1):69–79
Ossenkoppele R, Lyoo CH, Sudre CH, van Westen D, Cho H, Ryu YH, Choi JY, Smith R, Strandberg O, Palmqvist S, Westman E, Tsai R, Kramer J, Boxer AL, Gorno-Tempini ML, La Joie R, Miller BL, Rabinovici GD, Hansson O (2019) Distinct tau PET patterns in atrophy-defined subtypes of Alzheimer's disease. Alzheimers Dement 16(2):335–344
Ossenkoppele R, Tolboom N, Foster-Dingley JC, Adriaanse SF, Boellaard R, Yaqub M, Windhorst AD, Barkhof F, Lammertsma AA, Scheltens P, van der Flier WM, van Berckel BN (2012) Longitudinal imaging of Alzheimer pathology using [11C]PIB, [18F]FDDNP and [18F]FDG PET. Eur J Nucl Med Mol Imaging 39(6):990–1000
Ossenkoppele R, van Berckel BN, Prins ND (2011) Amyloid imaging in prodromal Alzheimer's disease. Alzheimers Res Ther 3(5):26
Ostrowitzki S, Deptula D, Thurfjell L, Barkhof F, Bohrmann B, Brooks DJ, Klunk WE, Ashford E, Yoo K, Xu ZX, Loetscher H, Santarelli L (2011) Mechanism of amyloid removal in patients with Alzheimer disease treated with Gantenerumab. Arch Neurol 69(2):198–207
Ovod V, Ramsey KN, Mawuenyega KG, Bollinger JG, Hicks T, Schneider T, Sullivan M, Paumier K, Holtzman DM, Morris JC (2017) Amyloid β concentrations and stable isotope labeling kinetics of human plasma specific to central nervous system amyloidosis. Alzheimers Dement 13(8):841–849
Palmqvist S, Insel PS, Stomrud E, Janelidze S, Zetterberg H, Brix B, Eichenlaub U, Dage JL, Chai X, Blennow K, Mattsson N, Hansson O (2019) Cerebrospinal fluid and plasma biomarker trajectories with increasing amyloid deposition in Alzheimer's disease. EMBO Mol Med 11(12):e11170
Pase MP, Satizabal CL, Seshadri S (2017) Role of improved vascular health in the declining incidence of dementia. Stroke 48(7):2013–2020
Perrotin A, La Joie R, de La Sayette V, Barre L, Mezenge F, Mutlu J, Guilloteau D, Egret S, Eustache F, Chetelat G (2017) Subjective cognitive decline in cognitively normal elders from the community or from a memory clinic: differential affective and imaging correlates. Alzheimers Dement 13(5):550–560
Petersen RC (2000) Mild cognitive impairment: transition between aging and Alzheimer's disease. Neurologia 15(3):93–101
Petersen RC, Smith GE, Ivnik RJ, Tangalos EG, Schaid DJ, Thibodeau SN, Kokmen E, Waring SC, Kurland LT (1995) Apolipoprotein E status as a predictor of the development of Alzheimer's disease in memory-impaired individuals. JAMA 273:1274–1278
Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E (1999) Mild cognitive impairment: clinical characterization and outcome. Arch Neurol 56:303–308
Pettersen JA, Sathiyamoorthy G, Gao FQ, Szilagyi G, Nadkarni NK, St George-Hyslop P, Rogaeva E, Black SE (2008) Microbleed topography, leukoaraiosis, and cognition in probable Alzheimer disease from the Sunnybrook dementia study. Arch Neurol 65(6):790–795
Phelps ME (2000) PET: the merging of biology and imaging into molecular imaging. J Nucl Med 41(4):661–681
Pike KE, Ellis KA, Villemagne VL, Good N, Chetelat G, Ames D, Szoeke C, Laws SM, Verdile G, Martins RN, Masters CL, Rowe CC (2011) Cognition and beta-amyloid in preclinical Alzheimer's disease: data from the AIBL study. Neuropsychologia 49(9):2384–2390
Pike KE, Savage G, Villemagne VL, Ng S, Moss SA, Maruff P, Mathis CA, Klunk WE, Masters CL, Rowe CC (2007) Beta-amyloid imaging and memory in non-demented individuals: evidence for preclinical Alzheimer's disease. Brain 130(Pt 11):2837–2844
Pike VW (2009) PET radiotracers: crossing the blood-brain barrier and surviving metabolism. Trends Pharmacol Sci 30(8):431–440
Plassman BL, Havlik RJ, Steffens DC, Helms MJ, Newman TN, Drosdick D, Phillips C, Gau BA, Welsh-Bohmer KA, Burke JR, Guralnik JM, Breitner JC (2000) Documented head injury in early adulthood and risk of Alzheimer's disease and other dementias. Neurology 55(8):1158–1166
Pletnikova O, West N, Lee MK, Rudow GL, Skolasky RL, Dawson TM, Marsh L, Troncoso JC (2005) Abeta deposition is associated with enhanced cortical a-synuclein lesions in Lewy body diseases. Neurobiol Aging 26:1183–1192
Pontecorvo MJ, Arora AK, Devine M, Lu M, Galante N, Siderowf A, Devadanam C, Joshi AD, Heun SL, Teske BF, Truocchio SP, Krautkramer M, Devous MD Sr, Mintun MA (2017a) Quantitation of PET signal as an adjunct to visual interpretation of florbetapir imaging. Eur J Nucl Med Mol Imaging 44(5):825–837
Pontecorvo MJ, Siderowf A, Dubois B, Doraiswamy PM, Frisoni GB, Grundman M, Nobili F, Sadowsky CH, Salloway S, Arora AK, Chevrette A, Deberdt W, Dell'Agnello G, Flitter M, Galante N, Lowrey MJ, Lu M, McGeehan A, Devous MD Sr, Mintun MA (2017b) Effectiveness of Florbetapir PET imaging in changing patient management. Dement Geriatr Cogn Disord 44(3–4):129–143
Price JC, Klunk WE, Lopresti BJ, Lu X, Hoge JA, Ziolko SK, Holt DP, Meltzer CC, DeKosky ST, Mathis CA (2005) Kinetic modeling of amyloid binding in humans using PET imaging and Pittsburgh compound-B. J Cereb Blood Flow Metab 25(11):1528–1547
Price JL, Morris JC (1999) Tangles and plaques in nondemented aging and "preclinical" Alzheimer's disease. Ann Neurol 45(3):358–368
Qu W, Kung MP, Hou C, Benedum TE, Kung HF (2007a) Novel styrylpyridines as probes for SPECT imaging of amyloid plaques. J Med Chem 50(9):2157–2165
Qu W, Kung MP, Hou C, Jin LW, Kung HF (2007b) Radioiodinated aza-diphenylacetylenes as potential SPECT imaging agents for beta-amyloid plaque detection. Bioorg Med Chem Lett 17(13):3581–3584
Rabinovici GD, Furst AJ, Alkalay A, Racine CA, O'Neil JP, Janabi M, Baker SL, Agarwal N, Bonasera SJ, Mormino EC, Weiner MW, Gorno-Tempini ML, Rosen HJ, Miller BL, Jagust WJ (2010) Increased metabolic vulnerability in early-onset Alzheimer's disease is not related to amyloid burden. Brain 133(Pt 2):512–528
Rabinovici GD, Furst AJ, O'Neil JP, Racine CA, Mormino EC, Baker SL, Chetty S, Patel P, Pagliaro TA, Klunk WE, Mathis CA, Rosen HJ, Miller BL, Jagust WJ (2007) 11C-PIB PET imaging in Alzheimer disease and frontotemporal lobar degeneration. Neurology 68(15):1205–1212
Rabinovici GD, Gatsonis C, Apgar C, Chaudhary K, Gareen I, Hanna L, Hendrix J, Hillner BE, Olson C, Lesman-Segev OH, Romanoff J, Siegel BA, Whitmer RA, Carrillo MC (2019) Association of Amyloid Positron Emission Tomography with Subsequent Change in clinical management among Medicare beneficiaries with mild cognitive impairment or dementia. JAMA 321(13):1286–1294
Rabinovici GD, Jagust WJ (2009) Amyloid imaging in aging and dementia: testing the amyloid hypothesis in vivo. Behav Neurol 21(1):117–128
Rabinovici GD, Jagust WJ, Furst AJ, Ogar JM, Racine CA, Mormino EC, O'Neil JP, Lal RA, Dronkers NF, Miller BL, Gorno-Tempini ML (2008) Abeta amyloid and glucose metabolism in three variants of primary progressive aphasia. Ann Neurol 64(4):388–401
Rabinovici GD, Miller BL (2010) Frontotemporal lobar degeneration: epidemiology, pathophysiology, diagnosis and management. CNS Drugs 24(5):375–398
Rabinovici GD, Rosen HJ, Alkalay A, Kornak J, Furst AJ, Agarwal N, Mormino EC, O'Neil JP, Janabi M, Karydas A, Growdon ME, Jang JY, Huang EJ, Dearmond SJ, Trojanowski JQ, Grinberg LT, Gorno-Tempini ML, Seeley WW, Miller BL, Jagust WJ (2011) Amyloid vs FDG-PET in the differential diagnosis of AD and FTLD. Neurology 77(23):2034–2042
Raniga P, Desmond P, Yates P, Salvado O, Bourgeat P, Fripp J, Pejoska S, Woodward M, Masters CL, Rowe CC, Villemagne VL (2017) Focal Ab-amyloid deposition precedes cerebral microbleeds and Superficial siderosis: a case report. J Neurosci Neurol Disord 1:039–044
Reiman EM, Caselli RJ, Yun LS, Chen K, Bandy D, Minoshima S, Thibodeau SN, Osborne D (1996) Preclinical evidence of Alzheimer's disease in persons homozygous for the epsilon 4 allele for apolipoprotein E. N Engl J Med 334(12):752–758
Reiman EM, Chen K, Liu X, Bandy D, Yu M, Lee W, Ayutyanont N, Keppler J, Reeder SA, Langbaum JB, Alexander GE, Klunk WE, Mathis CA, Price JC, Aizenstein HJ, Dekosky ST, Caselli RJ (2009a) Fibrillar amyloid-β burden in cognitively normal people at 3 levels of genetic risk for Alzheimer's disease. Proc Natl Acad Sci U S A 106:6820–6825
Reiman EM, Chen K, Liu X, Bandy D, Yu M, Lee W, Ayutyanont N, Keppler J, Reeder SA, Langbaum JB, Alexander GE, Klunk WE, Mathis CA, Price JC, Aizenstein HJ, DeKosky ST, Caselli RJ (2009b) Fibrillar amyloid-beta burden in cognitively normal people at 3 levels of genetic risk for Alzheimer's disease. Proc Natl Acad Sci U S A 106(16):6820–6825
Remes AM, Laru L, Tuominen H, Aalto S, Kemppainen N, Mononen H, Nagren K, Parkkola R, Rinne JO (2008) Carbon 11-labeled Pittsburgh compound B positron emission tomographic amyloid imaging in patients with APP locus duplication. Arch Neurol 65(4):540–544
Rentz DM, Locascio JJ, Becker JA, Moran EK, Eng E, Buckner RL, Sperling RA, Johnson KA (2010) Cognition, reserve, and amyloid deposition in normal aging. Ann Neurol 67(3):353–364
Resende R, Pereira C, Agostinho P, Vieira AP, Malva JO, Oliveira CR (2007) Susceptibility of hippocampal neurons to Abeta peptide toxicity is associated with perturbation of Ca2+ homeostasis. Brain Res 1143:11–21
Resnick SM, Sojkova J, Zhou Y, An Y, Ye W, Holt DP, Dannals RF, Mathis CA, Klunk WE, Ferrucci L, Kraut MA, Wong DF (2010) Longitudinal cognitive decline is associated with fibrillar amyloid-beta measured by [11C]PiB. Neurology 74(10):807–815
Riekse RG, Leverenz JB, McCormick W, Bowen JD, Teri L, Nochlin D, Simpson K, Eugenio C, Larson EB, Tsuang D (2004) Effect of vascular lesions on cognition in Alzheimer's disease: a community-based study. J Am Geriatr Soc 52(9):1442–1448
Rinne JO, Brooks DJ, Rossor MN, Fox NC, Bullock R, Klunk WE, Mathis CA, Blennow K, Barakos J, Okello AA, de Liano SRM, Liu E, Koller M, Gregg KM, Schenk D, Black R, Grundman M (2010) 11C-PiB PET assessment of change in fibrillar amyloid-beta load in patients with Alzheimer's disease treated with bapineuzumab: a phase 2, double-blind, placebo-controlled, ascending-dose study. Lancet Neurol 9(4):363–372
Roberts BR, Lind M, Wagen AZ, Rembach A, Frugier T, Li QX, Ryan TM, McLean CA, Doecke JD, Rowe CC, Villemagne VL, Masters CL (2017) Biochemically-defined pools of amyloid-beta in sporadic Alzheimer's disease: correlation with amyloid PET. Brain 140(5):1486–1498
Roder S, Danober L, Pozza MF, Lingenhoehl K, Wiederhold KH, Olpe HR (2003) Electrophysiological studies on the hippocampus and prefrontal cortex assessing the effects of amyloidosis in amyloid precursor protein 23 transgenic mice. Neuroscience 120(3):705–720
Rodriguez-Vieitez E, Nordberg A (2018) Imaging Neuroinflammation: quantification of Astrocytosis in a multitracer PET approach. Methods Mol Biol 1750:231–251
Rodriguez-Vieitez E, Saint-Aubert L, Carter SF, Almkvist O, Farid K, Scholl M, Chiotis K, Thordardottir S, Graff C, Wall A, Langstrom B, Nordberg A (2016) Diverging longitudinal changes in astrocytosis and amyloid PET in autosomal dominant Alzheimer's disease. Brain 139(Pt 3):922–936
Roe CM, Mintun MA, D'Angelo G, Xiong C, Grant EA, Morris JC (2008) Alzheimer disease and cognitive reserve: variation of education effect with carbon 11-labeled Pittsburgh compound B uptake. Arch Neurol 65(11):1467–1471
Rosen RF, Ciliax BJ, Wingo TS, Gearing M, Dooyema J, Lah JJ, Ghiso JA, LeVine H 3rd, Walker LC (2010) Deficient high-affinity binding of Pittsburgh compound B in a case of Alzheimer's disease. Acta Neuropathol 119(2):221–233
Rosendorff C, Beeri MS, Silverman JM (2007) Cardiovascular risk factors for Alzheimer's disease. Am J Geriatr Cardiol 16(3):143–149
Rossor MN, Kennedy AM, Frackowiak RS (1996) Clinical and neuroimaging features of familial Alzheimer's disease. Ann N Y Acad Sci 777:49–56
Rowe CC, Ackerman U, Browne W, Mulligan R, Pike KL, O'Keefe G, Tochon-Danguy H, Chan G, Berlangieri SU, Jones G, Dickinson-Rowe KL, Kung HP, Zhang W, Kung MP, Skovronsky D, Dyrks T, Holl G, Krause S, Friebe M, Lehman L, Lindemann S, Dinkelborg LM, Masters CL, Villemagne VL (2008) Imaging of amyloid beta in Alzheimer's disease with (18)F-BAY94-9172, a novel PET tracer: proof of mechanism. Lancet Neurol 7(2):129–135
Rowe CC, Bourgeat P, Ellis KA, Brown B, Lim YY, Mulligan R, Jones G, Maruff P, Woodward M, Price R, Robins P, Tochon-Danguy H, O'Keefe G, Pike KE, Yates P, Szoeke C, Salvado O, Macaulay SL, O'Meara T, Head R, Cobiac L, Savage G, Martins R, Masters CL, Ames D, Villemagne VL (2013a) Predicting Alzheimer disease with beta-amyloid imaging: results from the Australian imaging, biomarkers, and lifestyle study of ageing. Ann Neurol 74(6):905–913
Rowe CC, Dore V, Jones G, Baxendale D, Mulligan RS, Bullich S, Stephens AW, De Santi S, Masters CL, Dinkelborg L, Villemagne VL (2017) (18)F-Florbetaben PET beta-amyloid binding expressed in Centiloids. Eur J Nucl Med Mol Imaging 44(12):2053–2059
Rowe CC, Ellis KA, Rimajova M, Bourgeat P, Pike KE, Jones G, Fripp J, Tochon-Danguy H, Morandeau L, O'Keefe G, Price R, Raniga P, Robins P, Acosta O, Lenzo N, Szoeke C, Salvado O, Head R, Martins R, Masters CL, Ames D, Villemagne VL (2010) Amyloid imaging results from the Australian imaging, biomarkers and lifestyle (AIBL) study of aging. Neurobiol Aging 31(8):1275–1283
Rowe CC, Jones G, Dore V, Pejoska S, Margison L, Mulligan RS, Chan JG, Young K, Villemagne VL (2016) Standardized expression of 18F-NAV4694 and 11C-PiB beta-amyloid PET results with the Centiloid scale. J Nucl Med 57(8):1233–1237
Rowe CC, Ng S, Ackermann U, Gong SJ, Pike K, Savage G, Cowie TF, Dickinson KL, Maruff P, Darby D, Smith C, Woodward M, Merory J, Tochon-Danguy H, O'Keefe G, Klunk WE, Mathis CA, Price JC, Masters CL, Villemagne VL (2007) Imaging beta-amyloid burden in aging and dementia. Neurology 68(20):1718–1725
Rowe CC, Pejoska S, Mulligan RS, Jones G, Chan JG, Svensson S, Cselenyi Z, Masters CL, Villemagne VL (2013b) Head-to-head comparison of 11C-PiB and 18F-AZD4694 (NAV4694) for beta-amyloid imaging in aging and dementia. J Nucl Med 54(6):880–886
Rowe CC, Villemagne VL (2011) Brain amyloid imaging. J Nucl Med 52(11):1733–1740
Rozga M, Bittner T, Hoglund K, Blennow K (2017) Accuracy of cerebrospinal fluid Abeta1-42 measurements: evaluation of pre-analytical factors using a novel Elecsys immunoassay. Clin Chem Lab Med 55(10):1545–1554
Sabbagh MN, Fleisher A, Chen K, Rogers J, Berk C, Reiman E, Pontecorvo M, Mintun M, Skovronsky D, Jacobson SA, Sue LI, Liebsack C, Charney AS, Cole L, Belden C, Beach TG (2011) Positron emission tomography and neuropathologic estimates of fibrillar amyloid-beta in a patient with down syndrome and Alzheimer disease. Arch Neurol 68(11):1461–1466
Sabbagh MN, Schauble B, Anand K, Richards D, Murayama S, Akatsu H, Takao M, Rowe CC, Masters CL, Barthel H, Gertz HJ, Peters O, Rasgon N, Jovalekic A, Sabri O, Schulz-Schaeffer WJ, Seibyl J (2017) Histopathology and Florbetaben PET in patients incorrectly diagnosed with Alzheimer's disease. J Alzheimers Dis 56(2):441–446
Sabri O, Sabbagh MN, Seibyl J, Barthel H, Akatsu H, Ouchi Y, Senda K, Murayama S, Ishii K, Takao M, Beach TG, Rowe CC, Leverenz JB, Ghetti B, Ironside JW, Catafau AM, Stephens AW, Mueller A, Koglin N, Hoffmann A, Roth K, Reininger C, Schulz-Schaeffer WJ, Florbetaben Phase 3 Study Group (2015) Florbetaben PET imaging to detect amyloid beta plaques in Alzheimer's disease: phase 3 study. Alzheimers Dement 11(8):964–974
Sair HI, Doraiswamy PM, Petrella JR (2004) In vivo amyloid imaging in Alzheimer's disease. Neuroradiology 46(2):93–104
Salloway S, Sperling R, Fox NC, Blennow K, Klunk W, Raskind M, Sabbagh M, Honig LS, Porsteinsson AP, Ferris S, Reichert M, Ketter N, Nejadnik B, Guenzler V, Miloslavsky M, Wang D, Lu Y, Lull J, Tudor IC, Liu E, Grundman M, Yuen E, Black R, Brashear HR, Bapineuzumab 301 and 302 Clinical Trial Investigators (2014) Two phase 3 trials of bapineuzumab in mild-to-moderate Alzheimer's disease. N Engl J Med 370(4):322–333
Salmon E, Sadzot B, Maquet P, Degueldre C, Lemaire C, Rigo P, Comar D, Franck G (1994) Differential diagnosis of Alzheimer's disease with PET. J Nucl Med 35(3):391–398
Sarro L, Senjem ML, Lundt ES, Przybelski SA, Lesnick TG, Graff-Radford J, Boeve BF, Lowe VJ, Ferman TJ, Knopman DS, Comi G, Filippi M, Petersen RC, Jack CR Jr, Kantarci K (2016) Amyloid-beta deposition and regional grey matter atrophy rates in dementia with Lewy bodies. Brain 139(Pt 10):2740–2750
Satizabal CL, Beiser AS, Chouraki V, Chene G, Dufouil C, Seshadri S (2016) Incidence of dementia over three decades in the Framingham heart study. N Engl J Med 374(6):523–532
Scheltens P, Rockwood K (2011) How golden is the gold standard of neuropathology in dementia? Alzheimers Dement 7(4):486–489
Schindler SE, Gray JD, Gordon BA, Xiong C, Batrla-Utermann R, Quan M, Wahl S, Benzinger TLS, Holtzman DM, Morris JC, Fagan AM (2018) Cerebrospinal fluid biomarkers measured by Elecsys assays compared to amyloid imaging. Alzheimers Dement 14(11):1460–1469
Schöll M, Almqvist O, Graff C, Nordberg A (2011) Amyloid imaging in members of a family harbouring the Arctic mutation. Alzheimers Dement 7(Suppl 1):303
Scholl M, Carter SF, Westman E, Rodriguez-Vieitez E, Almkvist O, Thordardottir S, Wall A, Graff C, Langstrom B, Nordberg A (2015) Early astrocytosis in autosomal dominant Alzheimer's disease measured in vivo by multi-tracer positron emission tomography. Sci Rep 5:16404
Schulz-Schaeffer WJ (2010) The synaptic pathology of alpha-synuclein aggregation in dementia with Lewy bodies, Parkinson's disease and Parkinson's disease dementia. Acta Neuropathol 120(2):131–143
Serdons, K., C. Terwinghe, P. Vermaelen, K. Van Laere, H. Kung, L. Mortelmans, G. Bormans and A. Verbruggen (2009a). "Synthesis and evaluation of (18)F-labeled 2-phenylbenzothiazoles as positron emission tomography imaging agents for amyloid plaques in Alzheimer's Disease." J Med Chem 52(5):1428-1437
Serdons K, Verduyckt T, Cleynhens J, Terwinghe C, Mortelmans L, Bormans G, Verbruggen A (2007) Synthesis and evaluation of a (99m)Tc-BAT-phenylbenzothiazole conjugate as a potential in vivo tracer for visualization of amyloid beta. Bioorg Med Chem Lett 17(22):6086–6090
Serdons K, Verduyckt T, Vanderghinste D, Cleynhens J, Borghgraef P, Vermaelen P, Terwinghe C, Van Leuven F, Van Laere K, Kung H, Bormans G, Verbruggen A (2009b) Synthesis of 18F-labelled 2-(4′-fluorophenyl)-1,3-benzothiazole and evaluation as amyloid imaging agent in comparison with [11C]PIB. Bioorg Med Chem Lett 19(3):602–605
Sevigny J, Chiao P, Bussiere T, Weinreb PH, Williams L, Maier M, Dunstan R, Salloway S, Chen T, Ling Y, O'Gorman J, Qian F, Arastu M, Li M, Chollate S, Brennan MS, Quintero-Monzon O, Scannevin RH, Arnold HM, Engber T, Rhodes K, Ferrero J, Hang Y, Mikulskis A, Grimm J, Hock C, Nitsch RM, Sandrock A (2016) The antibody aducanumab reduces Abeta plaques in Alzheimer's disease. Nature 537(7618):50–56
Shaw LM, Hansson O, Manuilova E, Masters CL, Doecke JD, Li QX, Rutz S, Widmann M, Leinenbach A, Blennow K (2019) Method comparison study of the Elecsys(R) beta-amyloid (1-42) CSF assay versus comparator assays and LC-MS/MS. Clin Biochem 72:7–14
Shaw LM, Korecka M, Clark CM, Lee VM, Trojanowski JQ (2007) Biomarkers of neurodegeneration for diagnosis and monitoring therapeutics. Nat Rev Drug Discov 6(4):295–303
Shih WJ, Markesbery WR, Clark DB, Goldstein S, Domstad PA, Coupal JJ, Kung H, DeKosky ST, DeLand FH (1987) Iodine-123 HIPDM brain imaging findings in subacute spongiform encephalopathy (Creutzfeldt-Jakob disease). J Nucl Med 28(9):1484–1487
Shoghi-Jadid K, Small GW, Agdeppa ED, Kepe V, Ercoli LM, Siddarth P, Read S, Satyamurthy N, Petric A, Huang SC, Barrio JR (2002) Localization of neurofibrillary tangles and beta-amyloid plaques in the brains of living patients with Alzheimer disease. Am J Geriatr Psychiatry 10(1):24–35
Siderowf A, Xie SX, Hurtig H, Weintraub D, Duda J, Chen-Plotkin A, Shaw LM, Van Deerlin V, Trojanowski JQ, Clark C (2010) CSF amyloid {beta} 1-42 predicts cognitive decline in Parkinson disease. Neurology 75(12):1055–1061
Silverman DH, Cummings JL, Small GW, Gambhir SS, Chen W, Czernin J, Phelps ME (2002) Added clinical benefit of incorporating 2-deoxy-2-[18F]fluoro-D-glucose with positron emission tomography into the clinical evaluation of patients with cognitive impairment. Mol Imaging Biol 4(4):283–293
Silverman DH, Small GW, Chang CY, Lu CS, Kung De Aburto MA, Chen W, Czernin J, Rapoport SI, Pietrini P, Alexander GE, Schapiro MB, Jagust WJ, Hoffman JM, Welsh-Bohmer KA, Alavi A, Clark CM, Salmon E, de Leon MJ, Mielke R, Cummings JL, Kowell AP, Gambhir SS, Hoh CK, Phelps ME (2001) Positron emission tomography in evaluation of dementia: regional brain metabolism and long-term outcome. JAMA 286(17):2120–2127
Slot RER, Sikkes SAM, Berkhof J, Brodaty H, Buckley R, Cavedo E, Dardiotis E, Guillo-Benarous F, Hampel H, Kochan NA, Lista S, Luck T, Maruff P, Molinuevo JL, Kornhuber J, Reisberg B, Riedel-Heller SG, Risacher SL, Roehr S, Sachdev PS, Scarmeas N, Scheltens P, Shulman MB, Saykin AJ, Verfaillie SCJ, Visser PJ, Vos SJB, Wagner M, Wolfsgruber S, Jessen F, Alzheimer's Disease Neuroimaging Initiative; DESCRIPA working group; INSIGHT-preAD study group; SCD-I working group; Wiesje M van der Flier (2019) Subjective cognitive decline and rates of incident Alzheimer's disease and non-Alzheimer's disease dementia. Alzheimers Dement 15(3):465–476
Small BJ, Gagnon E, Robinson B (2007) Early identification of cognitive deficits: preclinical Alzheimer's disease and mild cognitive impairment. Geriatrics 62(4):19–23
Small GW, Agdeppa ED, Kepe V, Satyamurthy N, Huang SC, Barrio JR (2002) In vivo brain imaging of tangle burden in humans. J Mol Neurosci 19(3):323–327
Small GW, Kepe V, Ercoli LM, Siddarth P, Bookheimer SY, Miller KJ, Lavretsky H, Burggren AC, Cole GM, Vinters HV, Thompson PM, Huang SC, Satyamurthy N, Phelps ME, Barrio JR (2006) PET of brain amyloid and tau in mild cognitive impairment. N Engl J Med 355(25):2652–2663
Small GW, Mazziotta JC, Collins MT, Baxter LR, Phelps ME, Mandelkern MA, Kaplan A, La Rue A, Adamson CF, Chang L et al (1995) Apolipoprotein E type 4 allele and cerebral glucose metabolism in relatives at risk for familial Alzheimer disease. JAMA 273(12):942–947
Smith EE, Egorova S, Blacker D, Killiany RJ, Muzikansky A, Dickerson BC, Tanzi RE, Albert MS, Greenberg SM, Guttmann CR (2008) Magnetic resonance imaging white matter hyperintensities and brain volume in the prediction of mild cognitive impairment and dementia. Arch Neurol 65(1):94–100
Snowden J, Neary D, Mann D (2007) Frontotemporal lobar degeneration: clinical and pathological relationships. Acta Neuropathol 114(1):31–38
Sojkova J, Driscoll I, Iacono D, Zhou Y, Codispoti KE, Kraut MA, Ferrucci L, Pletnikova O, Mathis CA, Klunk WE, O'Brien RJ, Wong DF, Troncoso JC, Resnick SM (2011a) In vivo fibrillar beta-amyloid detected using [11C]PiB positron emission tomography and neuropathologic assessment in older adults. Arch Neurol 68(2):232–240
Sojkova J, Resnick SM (2011) In vivo human amyloid imaging. Curr Alzheimer Res 8(4):366–372
Sojkova J, Zhou Y, An Y, Kraut MA, Ferrucci L, Wong DF, Resnick SM (2011b) Longitudinal patterns of beta-amyloid deposition in nondemented older adults. Arch Neurol 68(5):644–649
Song PJ, Bernard S, Sarradin P, Vergote J, Barc C, Chalon S, Kung MP, Kung HF, Guilloteau D (2008) IMPY, a potential beta-amyloid imaging probe for detection of prion deposits in scrapie-infected mice. Nucl Med Biol 35(2):197–201
Sperling R, Johnson K (2010) Pro: can biomarkers be gold standards in Alzheimer's disease? Alzheimers Res Ther 2(3):17
Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, Iwatsubo T, Jack CR Jr, Kaye J, Montine TJ, Park DC, Reiman EM, Rowe CC, Siemers E, Stern Y, Yaffe K, Carrillo MC, Thies B, Morrison-Bogorad M, Wagster MV, Phelps CH (2011a) Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement 7(3):280–292
Sperling RA, Jack CR Jr, Aisen PS (2011b) Testing the right target and right drug at the right stage. Sci Transl Med 3(111):111cm133
Sperling RA, Jack CR Jr, Black SE, Frosch MP, Greenberg SM, Hyman BT, Scheltens P, Carrillo MC, Thies W, Bednar MM, Black RS, Brashear HR, Grundman M, Siemers ER, Feldman HH, Schindler RJ (2011c) Amyloid-related imaging abnormalities in amyloid-modifying therapeutic trials: recommendations from the Alzheimer's Association research roundtable workgroup. Alzheimers Dement 7(4):367–385
Sperling RA, Johnson KA, Doraiswamy PM, Reiman EM, Fleisher AS, Sabbagh MN, Sadowsky CH, Carpenter A, Davis MD, Lu M, Flitter M, Joshi AD, Clark CM, Grundman M, Mintun MA, Skovronsky DM, Pontecorvo MJ (2012) Amyloid deposition detected with florbetapir F 18 ((18)F-AV-45) is related to lower episodic memory performance in clinically normal older individuals. Neurobiol Aging 34(3):822–831
Sperling RA, Laviolette PS, O'Keefe K, O'Brien J, Rentz DM, Pihlajamaki M, Marshall G, Hyman BT, Selkoe DJ, Hedden T, Buckner RL, Becker JA, Johnson KA (2009) Amyloid deposition is associated with impaired default network function in older persons without dementia. Neuron 63(2):178–188
Sperling RA, Mormino EC, Schultz AP, Betensky RA, Papp KV, Amariglio RE, Hanseeuw BJ, Buckley R, Chhatwal J, Hedden T, Marshall GA, Quiroz YT, Donovan NJ, Jackson J, Gatchel JR, Rabin JS, Jacobs H, Yang HS, Properzi M, Kirn DR, Rentz DM, Johnson KA (2019) The impact of amyloid-beta and tau on prospective cognitive decline in older individuals. Ann Neurol 85(2):181–193
Sperling RA, Rentz DM, Johnson KA, Karlawish J, Donohue M, Salmon DP, Aisen P (2014) The A4 study: stopping AD before symptoms begin? Sci Transl Med 6(228):228fs213
Stern Y (2002) What is cognitive reserve? Theory and research application of the reserve concept. J Int Neuropsychol Soc 8(3):448–460
Storandt M, Head D, Fagan AM, Holtzman DM, Morris JC (2012) Toward a multifactorial model of Alzheimer disease. Aging, Neurobiol
Strozyk D, Blennow K, White LR, Launer LJ (2003) CSF Abeta 42 levels correlate with amyloid-neuropathology in a population-based autopsy study. Neurology 60(4):652–656
Su Y, Flores S, Wang G, Hornbeck RC, Speidel B, Joseph-Mathurin N, Vlassenko AG, Gordon BA, Koeppe RA, Klunk WE, Jack CR Jr, Farlow MR, Salloway S, Snider BJ, Berman SB, Roberson ED, Brosch J, Jimenez-Velazques I, van Dyck CH, Galasko D, Yuan SH, Jayadev S, Honig LS, Gauthier S, Hsiung GR, Masellis M, Brooks WS, Fulham M, Clarnette R, Masters CL, Wallon D, Hannequin D, Dubois B, Pariente J, Sanchez-Valle R, Mummery C, Ringman JM, Bottlaender M, Klein G, Milosavljevic-Ristic S, McDade E, Xiong C, Morris JC, Bateman RJ, Benzinger TLS (2019) Comparison of Pittsburgh compound B and florbetapir in cross-sectional and longitudinal studies. Alzheimers Dement (Amst) 11:180–190
Sunderland T, Gur RE, Arnold SE (2005) The use of biomarkers in the elderly: current and future challenges. Biol Psychiatry 58(4):272–276
Sundgren-Andersson AK, Svensson SPS, Swahn BM, Juréus A, Sandell J, Johnson AE, Jeppsson F, Neelissen JAM, Halldin C, Johnström P, Schou M, Cselényi Z, Farde L (2009) AZD4694: Fluorinated Positron Emission Tomography (PET) radioligand for detection of beta-amyloid deposits. Alzheimers Dement 5(4, Supplement 1):P267–P268. [abstract]
Suo Z, Wu M, Citron BA, Wong GT, Festoff BW (2004) Abnormality of G-protein-coupled receptor kinases at prodromal and early stages of Alzheimer's disease: an association with early beta-amyloid accumulation. J Neurosci 24(13):3444–3452
Sur C, Zeng Z, Hostetler E, Connolly BM, Miller PJ, O’Malley S, Chen T, Culberson C, Harrison S, Mulhearn J, Wolkenberg S, Barrow J, Sanabria S, Cook JJ, Hargreaves R, Williams DL (2010) In vitro characterization of MK-3328: a novel fluorinated positron emission tomography tracer for beta-amyloid plaques. Alzheimers Dement 6(4):S40
Taniguchi S, McDonagh AM, Pickering-Brown SM, Umeda Y, Iwatsubo T, Hasegawa M, Mann DM (2004) The neuropathology of frontotemporal lobar degeneration with respect to the cytological and biochemical characteristics of tau protein. Neuropathol Appl Neurobiol 30(1):1–18
Tenovuo O, Kemppainen N, Aalto S, Nagren K, Rinne JO (2008) Posterior cortical atrophy: a rare form of dementia with in vivo evidence of amyloid-beta accumulation. J Alzheimers Dis 15(3):351–355
Thal DR, Beach TG, Zanette M, Heurling K, Chakrabarty A, Ismail A, Smith AP, Buckley C (2015) [(18)F]flutemetamol amyloid positron emission tomography in preclinical and symptomatic Alzheimer's disease: specific detection of advanced phases of amyloid-beta pathology. Alzheimers Dement 11(8):975–985
Thal DR, Beach TG, Zanette M, Lilja J, Heurling K, Chakrabarty A, Ismail A, Farrar G, Buckley C, Smith APL (2018) Estimation of amyloid distribution by [(18)F]flutemetamol PET predicts the neuropathological phase of amyloid beta-protein deposition. Acta Neuropathol 136(4):557–567
Thal DR, Del Tredici K, Braak H (2004) Neurodegeneration in normal brain aging and disease. Sci Aging Knowledge Environ 2004(23):pe26
Thal LJ, Kantarci K, Reiman EM, Klunk WE, Weiner MW, Zetterberg H, Galasko D, Pratico D, Griffin S, Schenk D, Siemers E (2006) The role of biomarkers in clinical trials for Alzheimer disease. Alzheimer Dis Assoc Disord 20(1):6–15
Thompson PW, Ye L, Morgenstern JL, Sue L, Beach TG, Judd DJ, Shipley NJ, Libri V, Lockhart A (2009) Interaction of the amyloid imaging tracer FDDNP with hallmark Alzheimer's disease pathologies. J Neurochem 109(2):623–630
Thurfjell L, Lotjonen J, Lundqvist R, Koikkalainen J, Soininen H, Waldemar G, Brooks DJ, Vandenberghe R (2012) Combination of biomarkers: PET [F]Flutemetamol imaging and structural MRI in dementia and mild cognitive impairment. Neurodegener Dis 10(1-4):246–249
Tolboom N, van der Flier WM, Yaqub M, Boellaard R, Verwey NA, Blankenstein MA, Windhorst AD, Scheltens P, Lammertsma AA, van Berckel BN (2009a) Relationship of cerebrospinal fluid markers to 11C-PiB and 18F-FDDNP binding. J Nucl Med 50(9):1464–1470
Tolboom N, Yaqub M, van der Flier WM, Boellaard R, Luurtsema G, Windhorst AD, Barkhof F, Scheltens P, Lammertsma AA, van Berckel BN (2009b) Detection of Alzheimer pathology in vivo using both 11C-PIB and 18F-FDDNP PET. J Nucl Med 50(2):191–197
Toledo JB, Bjerke M, Da X, Landau SM, Foster NL, Jagust W, Jack C Jr, Weiner M, Davatzikos C, Shaw LM, Trojanowski JQ, Alzheimer’s Disease Neuroimaging Initiative Investigators (2015) Nonlinear association between cerebrospinal fluid and florbetapir F-18 beta-amyloid measures across the spectrum of Alzheimer disease. JAMA Neurol 72(5):571–581
Toledo JB, Xie SX, Trojanowski JQ, Shaw LM (2013) Longitudinal change in CSF tau and Abeta biomarkers for up to 48 months in ADNI. Acta Neuropathol 126(5):659–670
Tomiyama T, Nagata T, Shimada H, Teraoka R, Fukushima A, Kanemitsu H, Takuma H, Kuwano R, Imagawa M, Ataka S, Wada Y, Yoshioka E, Nishizaki T, Watanabe Y, Mori H (2008) A new amyloid beta variant favoring oligomerization in Alzheimer's-type dementia. Ann Neurol 63(3):377–387
Tosun D, Schuff N, Mathis CA, Jagust W, Weiner MW (2011) Spatial patterns of brain amyloid-beta burden and atrophy rate associations in mild cognitive impairment. Brain 134(Pt 4):1077–1088
Trojanowski JQ (2002) Emerging Alzheimer's disease therapies: focusing on the future. Neurobiol Aging 23(6):985–990
Tucker AM, Stern Y (2011) Cognitive reserve in aging. Curr Alzheimer Res 8(4):354–360
Vandenberghe R, Van Laere K, Ivanoiu A, Salmon E, Bastin C, Triau E, Hasselbalch S, Law I, Andersen A, Korner A, Minthon L, Garraux G, Nelissen N, Bormans G, Buckley C, Owenius R, Thurfjell L, Farrar G, Brooks DJ (2010) 18F-flutemetamol amyloid imaging in Alzheimer disease and mild cognitive impairment: a phase 2 trial. Ann Neurol 68(3):319–329
Vemuri P, Lesnick TG, Przybelski SA, Knopman DS, Lowe VJ, Graff-Radford J, Roberts RO, Mielke MM, Machulda MM, Petersen RC, Jack CR Jr (2017) Age, vascular health, and Alzheimer disease biomarkers in an elderly sample. Ann Neurol 82(5):706–718
Venneti S, Lopresti BJ, Wiley CA (2006) The peripheral benzodiazepine receptor (translocator protein 18kDa) in microglia: from pathology to imaging. Prog Neurobiol 80(6):308–322
Verfaillie SCJ, Timmers T, Slot RER, van der Weijden CWJ, Wesselman LMP, Prins ND, Sikkes SAM, Yaqub M, Dols A, Lammertsma AA, Scheltens P, Ossenkoppele R, van Berckel BNM, van der Flier WM (2019) Amyloid-beta load is related to worries, but not to severity of cognitive complaints in individuals with subjective cognitive decline: the SCIENCe project. Front Aging Neurosci 11:7
Verhoeff NP, Wilson AA, Takeshita S, Trop L, Hussey D, Singh K, Kung HF, Kung MP, Houle S (2004) In-vivo imaging of Alzheimer disease beta-amyloid with [11C]SB-13 PET. Am J Geriatr Psychiatry 12(6):584–595
Vernooij MW, van der Lugt A, Ikram MA, Wielopolski PA, Niessen WJ, Hofman A, Krestin GP, Breteler MM (2008) Prevalence and risk factors of cerebral microbleeds: the Rotterdam scan study. Neurology 70(14):1208–1214
Villain N, Chetelat G, Grassiot B, Bourgeat P, Jones G, Ellis KA, Ames D, Martins RN, Eustache F, Salvado O, Masters CL, Rowe CC, Villemagne VL (2012) Regional dynamics of amyloid-beta deposition in healthy elderly, mild cognitive impairment and Alzheimer's disease: a voxelwise PiB-PET longitudinal study. Brain 135(Pt 7):2126–2139
Villemagne VL, Ataka S, Mizuno T, Brooks WS, Wada Y, Kondo M, Jones G, Watanabe Y, Mulligan R, Nakagawa M, Miki T, Shimada H, O'Keefe GJ, Masters CL, Mori H, Rowe CC (2009a) High striatal amyloid beta-peptide deposition across different autosomal Alzheimer disease mutation types. Arch Neurol 66(12):1537–1544
Villemagne VL, Burnham S, Bourgeat P, Brown B, Ellis KA, Salvado O, Szoeke C, Macaulay SL, Martins R, Maruff P, Ames D, Rowe CC, Masters CL (2013) Amyloid beta deposition, neurodegeneration, and cognitive decline in sporadic Alzheimer's disease: a prospective cohort study. Lancet Neurol 12(4):357–367
Villemagne VL, Cappai R, Barnham KJ, Cherny R, Opazo C, Novakovic KE, Rowe CC, Masters CL (2006) The Aβ centric pathway of Alzheimer’s disease. In: Barrow CJ, Small BJ (eds) Abeta Peptide and Alzheimer’s Disease. Springer-Verlag, London, pp 5–32
Villemagne VL, Dore V, Burnham SC, Masters CL, Rowe CC (2018) Imaging tau and amyloid-beta proteinopathies in Alzheimer disease and other conditions. Nat Rev Neurol 14(4):225–236
Villemagne VL, Fodero-Tavoletti MT, Masters CL, Rowe CC (2015) Tau imaging: early progress and future directions. Lancet Neurol 14(1):114–124
Villemagne VL, Fodero-Tavoletti MT, Pike KE, Cappai R, Masters CL, Rowe CC (2008a) The ART of loss: Aβ imaging in the evaluation of Alzheimer's disease and other dementias. Mol Neurobiol 38(1):1–15
Villemagne VL, Furumoto S, Fodero-Tavoletti MT, Mulligan RS, Hodges J, Piguet O, Pejoska S, Kudo Y, Masters CL, Yanai K, Rowe CC, Okamura N (2012a) In vivo tau imaging in Alzheimer's disease and other dementias. Alzheimers Dement 8(4):P9
Villemagne VL, Kim S, Rowe CC, Iwatsubo T (2014) Imago Mundi, Imago AD, Imago ADNI. Alzheimers Res Ther 6:62
Villemagne VL, Klunk WE, Mathis CA, Rowe CC, Brooks DJ, Hyman BT, Ikonomovic MD, Ishii K, Jack CR, Jagust WJ, Johnson KA, Koeppe RA, Lowe VJ, Masters CL, Montine TJ, Morris JC, Nordberg A, Petersen RC, Reiman EM, Selkoe DJ, Sperling RA, Van Laere K, Weiner MW, Drzezga A (2012b) Abeta imaging: feasible, pertinent, and vital to progress in Alzheimer's disease. Eur J Nucl Med Mol Imaging 39(2):209–219
Villemagne VL, McLean CA, Reardon K, Boyd A, Lewis V, Klug G, Jones G, Baxendale D, Masters CL, Rowe CC, Collins SJ (2009b) 11C-PiB PET studies in typical sporadic Creutzfeldt-Jakob disease. J Neurol Neurosurg Psychiatry 80(9):998–1001
Villemagne VL, Mulligan RS, Pejoska S, Ong K, Jones G, O'Keefe G, Chan JG, Young K, Tochon-Danguy H, Masters CL, Rowe CC (2012c) Comparison of (11)C-PiB and (18)F-florbetaben for Aβ imaging in ageing and Alzheimer's disease. Eur J Nucl Med Mol Imaging 39(6):983–989
Villemagne VL, Okamura N, Pejoska S, Drago J, Mulligan RS, Chetelat G, O'Keefe G, Jones G, Kung HF, Pontecorvo M, Masters CL, Skovronsky DM, Rowe CC (2012d) Differential diagnosis in Alzheimer's disease and dementia with Lewy bodies via VMAT2 and amyloid imaging. Neurodegener Dis 10(1–4):161–165
Villemagne VL, Ong K, Mulligan RS, Holl G, Pejoska S, Jones G, O'Keefe G, Ackerman U, Tochon-Danguy H, Chan JG, Reininger CB, Fels L, Putz B, Rohde B, Masters CL, Rowe CC (2011a) Amyloid imaging with 18F-Florbetaben in Alzheimer disease and other dementias. J Nucl Med 52(8):1210–1217
Villemagne VL, Pike K, Pejoska S, Boyd A, Power M, Jones G, Masters CL, Rowe CC (2010) 11C-PiB PET ABri imaging in Worster-drought syndrome (familial British dementia): a case report. J Alzheimers Dis 19(2):423–428
Villemagne VL, Pike KE, Chetelat G, Ellis KA, Mulligan RS, Bourgeat P, Ackermann U, Jones G, Szoeke C, Salvado O, Martins R, O'Keefe G, Mathis CA, Klunk WE, Ames D, Masters CL, Rowe CC (2011b) Longitudinal assessment of Aβ and cognition in aging and Alzheimer disease. Ann Neurol 69(1):181–192
Villemagne VL, Pike KE, Darby D, Maruff P, Savage G, Ng S, Ackermann U, Cowie TF, Currie J, Chan SG, Jones G, Tochon-Danguy H, O'Keefe G, Masters CL, Rowe CC (2008b) Aβ deposits in older non-demented individuals with cognitive decline are indicative of preclinical Alzheimer's disease. Neuropsychologia 46(6):1688–1697
Villemagne VL, Rowe CC (2010) Amyloid ligands for dementia. PET Clin 5:33–53
Villemagne VL, Rowe CC, Macfarlane S, Novakovic KE, Masters CL (2005) Imaginem oblivionis: the prospects of neuroimaging for early detection of Alzheimer's disease. J Clin Neurosci 12(3):221–230
Viswanathan A, Rocca WA, Tzourio C (2009) Vascular risk factors and dementia: how to move forward? Neurology 72(4):368–374
Villemagne VL, Furumoto S., Fodero-Tavoletti MT, Harada R, Mulligan RS, Kudo T, Masters CL, Rowe CC, Okamura N (2012) The Challenges of Tau Imaging. Future Neurol 7(4) 409–421
Vlassenko AG, Mintun MA, Xiong C, Sheline YI, Goate AM, Benzinger TL, Morris JC (2011) Amyloid-beta plaque growth in cognitively normal adults: longitudinal [11C]Pittsburgh compound B data. Ann Neurol 70(5):857–861
Wahlund LO, Blennow K (2003) Cerebrospinal fluid biomarkers for disease stage and intensity in cognitively impaired patients. Neurosci Lett 339(2):99–102
Walker LC, Rosen RF, Levine H 3rd (2008) Diversity of Abeta deposits in the aged brain: a window on molecular heterogeneity? Romanian J Morphol Embryol 49(1):5–11
Wang G, Xiong C, McDade EM, Hassenstab J, Aschenbrenner AJ, Fagan AM, Benzinger TLS, Gordon BA, Morris JC, Li Y, Bateman RJ, Dominantly Inherited Alzheimer Network (DIAN) (2018) Simultaneously evaluating the effect of baseline levels and longitudinal changes in disease biomarkers on cognition in dominantly inherited Alzheimer's disease. Alzheimers Dement (N Y) 4:669–676
Weiner MW, Harvey D, Hayes J, Landau SM, Aisen PS, Petersen RC, Tosun D, Veitch DP, Jack CR Jr, Decarli C, Saykin AJ, Grafman J, Neylan TC, Department of Defense Alzheimer's Disease Neuroimaging Initiative (2017) Effects of traumatic brain injury and posttraumatic stress disorder on development of Alzheimer's disease in Vietnam veterans using the Alzheimer's disease neuroimaging initiative: preliminary report. Alzheimers Dement (N Y) 3(2):177–188
Weller RO, Preston SD, Subash M, Carare RO (2009) Cerebral amyloid angiopathy in the aetiology and immunotherapy of Alzheimer disease. Alzheimers Res Ther 1(2):6
Wermke M, Sorg C, Wohlschlager AM, Drzezga A (2008) A new integrative model of cerebral activation, deactivation and default mode function in Alzheimer's disease. Eur J Nucl Med Mol Imaging 35(Suppl 1):S12–S24
Wiley CA, Lopresti BJ, Venneti S, Price J, Klunk WE, DeKosky ST, Mathis CA (2009) Carbon 11-labeled Pittsburgh compound B and carbon 11-labeled (R)-PK11195 positron emission tomographic imaging in Alzheimer disease. Arch Neurol 66(1):60–67
Winblad B, Palmer K, Kivipelto M, Jelic V, Fratiglioni L, Wahlund LO, Nordberg A, Backman L, Albert M, Almkvist O, Arai H, Basun H, Blennow K, de Leon M, DeCarli C, Erkinjuntti T, Giacobini E, Graff C, Hardy J, Jack C, Jorm A, Ritchie K, van Duijn C, Visser P, Petersen RC (2004) Mild cognitive impairment--beyond controversies, towards a consensus: report of the international working group on mild cognitive impairment. J Intern Med 256(3):240–246
Wolk DA, Grachev ID, Buckley C, Kazi H, Grady MS, Trojanowski JQ, Hamilton RH, Sherwin P, McLain R, Arnold SE (2011) Association between in vivo fluorine 18-labeled flutemetamol amyloid positron emission tomography imaging and in vivo cerebral cortical histopathology. Arch Neurol 68(11):1398–1403
Wolk DA, Price JC, Madeira C, Saxton JA, Snitz BE, Lopez OL, Mathis CA, Klunk WE, Dekosky ST (2012a) Amyloid imaging in dementias with atypical presentation. Alzheimers Dement 8(5):389–398
Wolk DA, Price JC, Saxton JA, Snitz BE, James JA, Lopez OL, Aizenstein HJ, Cohen AD, Weissfeld LA, Mathis CA, Klunk WE, DeKosky ST (2009) Amyloid imaging in mild cognitive impairment subtypes. Ann Neurol 65(5):557–568
Wolk DA, Zhang Z, Boudhar S, Clark CM, Pontecorvo MJ, Arnold SE (2012b) Amyloid imaging in Alzheimer's disease: comparison of florbetapir and Pittsburgh compound-B positron emission tomography. J Neurol Neurosurg Psychiatry 83(9):923–926
Wong DF, Moghekar AR, Rigamonti D, Brasic JR, Rousset O, Willis W, Buckley C, Smith A, Gok B, Sherwin P, Grachev ID (2012) An in vivo evaluation of cerebral cortical amyloid with [(18)F]Flutemetamol using positron emission tomography compared with parietal biopsy samples in living Normal pressure hydrocephalus patients. Mol Imaging Biol 15(2):230–237
Wong DF, Rosenberg PB, Zhou Y, Kumar A, Raymont V, Ravert HT, Dannals RF, Nandi A, Brasic JR, Ye W, Hilton J, Lyketsos C, Kung HF, Joshi AD, Skovronsky DM, Pontecorvo MJ (2010) In vivo imaging of amyloid deposition in Alzheimer disease using the radioligand 18F-AV-45 (florbetapir [corrected] F 18). J Nucl Med 51(6):913–920
Yates PA, Sirisriro R, Villemagne VL, Farquharson S, Masters CL, Rowe CC (2011) Cerebral microhemorrhage and brain {beta}-amyloid in aging and Alzheimer disease. Neurology 77(1):48–54
Ye L, Morgenstern JL, Gee AD, Hong G, Brown J, Lockhart A (2005) Delineation of positron emission tomography imaging agent binding sites on beta-amyloid peptide fibrils. J Biol Chem 280(25):23599–23604
Ye L, Velasco A, Fraser G, Beach TG, Sue L, Osredkar T, Libri V, Spillantini MG, Goedert M, Lockhart A (2008) In vitro high affinity alpha-synuclein binding sites for the amyloid imaging agent PIB are not matched by binding to Lewy bodies in postmortem human brain. J Neurochem 105(4):1428–1437
Yee S, Mathis C, Klunk W, Weissfeld L, Lopresti B, Bi W, Ziolko S, Berginc M, DeKosky S, Price J (2007) Optimal time window for standardized uptake ratio as a simplified measure of PIB retention. J Nucl Med 48:404
Yokokura M, Mori N, Yagi S, Yoshikawa E, Kikuchi M, Yoshihara Y, Wakuda T, Sugihara G, Takebayashi K, Suda S, Iwata Y, Ueki T, Tsuchiya KJ, Suzuki K, Nakamura K, Ouchi Y (2011) In vivo changes in microglial activation and amyloid deposits in brain regions with hypometabolism in Alzheimer's disease. Eur J Nucl Med Mol Imaging 38(2):343–351
Zhang W, Oya S, Kung MP, Hou C, Maier DL, Kung HF (2005a) F-18 Polyethyleneglycol stilbenes as PET imaging agents targeting Abeta aggregates in the brain. Nucl Med Biol 32(8):799–809
Zhang W, Oya S, Kung MP, Hou C, Maier DL, Kung HF (2005b) F-18 stilbenes as PET imaging agents for detecting beta-amyloid plaques in the brain. J Med Chem 48(19):5980–5988
Zwan MD, Bouwman FH, Konijnenberg E, van der Flier WM, Lammertsma AA, Verhey FR, Aalten P, van Berckel BN, Scheltens P (2017) Diagnostic impact of [(18)F]flutemetamol PET in early-onset dementia. Alzheimers Res Ther 9(1):2
Acknowledgments
We would like to thank Prof. Michael Woodward, Dr. Rachel Mulligan, Dr. Greg Savage, Dr. Joanne Robertson, Dr. Gordon Chan, Dr. Kenneth Young, Dr. Sylvia Gong, Mr. Ignatius Reilly, Ms. Fiona Lamb, Mrs. Svetlana Pejoska, and the Brain Research Institute for their assistance with this study.
This work was supported in part by Program Grant 1127007, Project Grant 1132604, and Research Fellowship 1046471 of the National Health and Medical Research Council of Australia and the Austin Hospital Medical Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Villemagne, V.L., Doré, V., Burnham, S., Rowe, C.C. (2021). Aβ Imaging in Aging, Alzheimer’s Disease, and Other Neurodegenerative Conditions. In: Dierckx, R.A.J.O., Otte, A., de Vries, E.F.J., van Waarde, A., Leenders, K.L. (eds) PET and SPECT in Neurology. Springer, Cham. https://doi.org/10.1007/978-3-030-53168-3_10
Download citation
DOI: https://doi.org/10.1007/978-3-030-53168-3_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-53167-6
Online ISBN: 978-3-030-53168-3
eBook Packages: MedicineMedicine (R0)