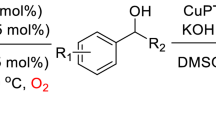
Abstract
For the last few years, we have been involved in hydrocarbon oxidation using tert-butylhydroperoxide and catalytic quantities of chromium(VI).1,2 we have precedently observed that an excess of oxygen source is required to reach high or full conversion of the organic substrate owing to the concomittant unproductive decomposition of t-BuOOH by chromium.3
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Muzart J. Tetrahedron Lett. 1986, 27 ,3139; 1987, 28 ,2131.
Muzart J. Chem. Rev. 1992, 92, 113.
Muzart J., N’ait Ajjou A. J. Mol. Catal. 1991, 66 ,155.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1993 Springer Science+Business Media New York
About this chapter
Cite this chapter
Muzart, J., N’Ait Ajjou, A. (1993). Chromium(VI) Oxide Catalyzed Oxidations by Tert-Butylhydroperoxide: Benzylic Oxidation Versus Tert-Butylhydroperoxide Decomposition. In: Barton, D.H.R., Martell, A.E., Sawyer, D.T. (eds) The Activation of Dioxygen and Homogeneous Catalytic Oxidation. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-3000-8_62
Download citation
DOI: https://doi.org/10.1007/978-1-4615-3000-8_62
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-6307-1
Online ISBN: 978-1-4615-3000-8
eBook Packages: Springer Book Archive