Abstract
Already in utero developing articular cartilage is exposed to, and is as well dependent of, a certain degree of mechanical stimulation (Brommer et al., Equine Vet J 37(2):148–154, 2005). Likewise, adult hyaline cartilage is strongly regulated by a frequent input of dynamic load. It is now clear that articular chondrocytes and mesenchymal stem cells clearly benefit from physical stimuli in vitro (Grad et al., Clin Orthop Relat Res 469(10):2764–2772, 2011). The term preconditioning has evolved in the field of cartilage tissue engineering, roughly describing an enhanced in vitro chondrogenesis by application of different stimuli which aims to generate more functional constructs for implantation. Physical stimulation is one way to precondition cells and is commonly realized by the use of bioreactors. Bioreactor systems can closely reproduce the in vivo environment, and can provoke a highly efficient chondrogenesis. They offer the possibility to evaluate novel therapeutic approaches while avoiding ethically challenging animal models. Mechanical load can be applied by tension, hydrostatic pressure, compression, shear, and any combination of these stimuli. In particular, the combination of compression and shear very closely resembles a human joint situation (Grad et al., Tissue Eng 12(11):3171–3179, 2006). Physical stimulation of articular chondrocytes and mesenchymal stem cells can result in an upregulation of the classical chondrogenic markers such as collagen 2, proteoglycan-4 and aggrecan. Furthermore it has been shown that cell-matrix constructs that have been subjected to physical loading highlighted an organized cell-matrix alignment in the direction of the mechanical stimulation, when compared to free-swelling cell-matrix constructs (Salzmann et al., Tissue Eng Part A 15(9):2513–2524, 2009). Significantly increased mechanical properties have also been reported following mechanical stimulation in vitro. However, an effective chondrogenesis can only be generated when the stimulus is correctly applied in terms of modulus, frequency, duration and force. Furthermore, subjected cells have to be embedded within a 3-D environment which provides a sufficient mechanical backbone to withstand and transmit mechanical loads while in parallel still permitting effective chondrogenesis. Novel bioreactor tissue engineering approaches aiming for articular cartilage repair may focus on stem cell chondrogenesis combining physical with chemical stimuli, which have been shown to be very efficient in promoting in vitro chondrogenesis (Li et al., J Cell Physiol 227(5):2003–2012, 2011).
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Bioreactor
- Tissue Engineering
- Cartilage
- Chondrocyte
- Stem cells
- Knee
- Osteoarthritis
- Biomechanical Stimuli
- Preconditioning
-
Articular cartilage serves a predominantly biomechanical function.
-
Hyaline cartilage is dependent on mechanical input to maintain function and integrity.
-
Dynamic shear, compression, fluid flow and hydrostatic pressure are the biomechanical hallmarks within articulating joints.
-
During gait the human walking cadence is normally at the range of 1 Hz by which articular cartilage experiences stresses between 3 and 10 Mpa with a strain of 10–15 %.
-
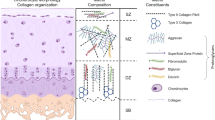
Articular chondrocytes mainly increase collagen type 2, 6, 9, 11, aggrecan, COMP, PRG-4 and glycosaminoglycan expression as a physiological response to mechanical load.
-
Bioreactors are devices to culture tissue by provision of a controllable, mechanically active environment.
-
Bioreactors can be operated in order to improve, but also study, the structure, properties and integration of tissue.
-
Bioreactors are capable of improving tissue construct size, cellularity and molecular composition of tissue such as cartilage by biomechanical modulation.
-
Flow perfusion, hydrostatic pressure, rotating wall, spinner flask, compression, shear or combined stimuli are the most common modes of bioreactor stimulation.
-
Bioreactor, functional tissue engineering is a rapidly increasing experimental and also early clinical field to study and precondition articular cartilage.
1 Introduction
Tissue Engineering is a field at the interface of engineering and biology which aims to repair or replace injured or diseased tissues and organs, such as articular cartilage. Since articular cartilage serves a predominantly biomechanical function, tailored tissue engineering principles are required. To meet this challenge a new paradigm termed functional tissue engineering is emphasizing biomechanical considerations during design and development of cell-scaffold constructs [1]. Bioreactors are devices to culture tissue by provision of a controllable, mechanically active environment which can be operated in order to improve, but also study, the structure, properties and integration of tissue. Bioreactors are capable of improving tissue construct size, cellularity and molecular composition.
It is becoming increasingly apparent that cartilage defects are frequently osteochondral lesions. The osteochondral junction represents the important backbone for the overlying hyaline cartilage. Effective articular cartilage repair can only be achieved when there is also healthy underlying bone [2]. Not only osteochondral lesions, but as well bone diseases such as infection, fractures, osteoarthritis or osteoporosis are becoming a major medical and socioeconomic problem. In this context, ex vivo tissue engineering strategies for de novo generation of bone tissue is also a major field of interest. The use of autologous bone-forming cells and three-dimensional porous scaffold materials are, comparable to cartilage tissue engineering, the two main components to realize bone tissue engineering [3]. Furthermore, different tissue engineering protocols have already realized tissue engineering production of osteochondral regenerates [4, 5]. However, this chapter is focussing on bioreactor tissue engineering with regard to articular cartilage. Nevertheless, there are many similarities concerning basic bioreactor principles, as well as typical bioreactor associated drawbacks such as insufficient nutrient and oxygen transport and removal of waste products from the cells at the interior of the scaffold.
An intra-articular environment can be regarded as harsh in terms of mechanical and chemical provocation. In particular, hyaline cartilage, which is covering the ends of long bones, is subjected to multiple repetitive load cycles and yet often produces a lifetime of pain free motion and weightbearing. It is a prerequisite for mammals to move and survive without lasting damage. Therefore, articular cartilage is a highly developed tissue in order to fulfil this task and withstand endless cycles of near frictionless motion during locomotion. The sacrifice associated with this specialization is a minimal ability to heal when damaged in post-puberty. Current cartilage repair procedures regularly fail to completely restore existing defects. Hyaline cartilage lesions are very frequent among adult subjects [6]. Furthermore, they often remain clinically silent initially, while morphologically progressing [7]. Synchronous to that, the well-balanced joint homeostasis can turn into an unstable equilibrium [8]. Both play a part in avicious circle, which may be initiated following blunt trauma, cruciate ligament ruptures, meniscal lesions or patella dislocation, but remain unnoticed over decades [9]. Only efficient cartilage repair techniques may arrest the progression of continuous degeneration before other means of osteoarthritis prevention become available [10, 11]. Although current cartilage repair procedures often struggle to result in a truly efficient repair, tissue engineering applications are on the threshold of clinical implementation in order to improve in vivo transplant performance. These applications may remain close to currently available cartilage repair techniques, such as chondrocyte transplantation.
2 Cartilage Repair
Already in the 20th century a great variety of surgical techniques have been proposed in order to address existing articular cartilage lesions. Until today, three basically different surgical options have evolved and are in frequent worldwide use. However, there remains controversy on how and when which surgical technique should be applied. A worldwide accepted standard guideline does not exist [12]. Arthroscopic microfracturing aims for in situ repair of the cartilage defect. Bone marrow stem cells (BMSCs) migrate into the defect and settle within the debrided lesion. It is anticipated that these cells differentiate into chondrocytes leading to a phenotypically correct repair of the lesion. Osteochondral transplantation, which can be achieved using open, mini-open or arthroscopic techniques, is aiming to replace damaged tissue immediately. Not only the chondral surface, but the underlying bone is also extracted using hollow cutters and consecutively replaced by autologous or allogenous osteochondral cylinders, which have the same dimensions. Defective cartilage is immediately replaced by hyaline cartilage, while at the same time addressing the underlying bone. Autologous chondrocyte implantation (ACI) aims to regenerate the cartilage defect by using isolated autologous chondrocytes, which have been previously obtained from the joint and expanded in vitro. The surgical techniques all have their specific advantages and disadvantages. On any account, they have in common that restitutio ad integrum commonly does not occur following the post-operative intervention [13]. While early randomized controlled trials comparing operative techniques against each other remained mostly inconclusive [14–16], current evidence has shown superiority of cell-based methods, autologous chondrocyte transplantation, in comparison to microfracturing [17] as well when comparing with osteochondral transplantation [18]. These aspects become particularly true when horizontally large (above 3–4 cm2) defects are concerned. Furthermore, a correlation between the quality of the repair tissue and clinical symptoms have been described. When mostly hyaline and hyaline-like tissue evolves at the defective site, the likelihood of a satisfying clinical outcome is clearly increased [19]. It was shown during clinical ACI that initial strong collagen type 2 and CD-44 expression within the chondrocytes is significantly correlated with an improved clinical outcome [20]. Tissue quality can be regarded as one major aspect when articular cartilage repair is concerned. Morphologically and thus functionally well-developed tissue following cartilage repair procedures is more likely correlated with a satisfying long-term clinical outcome than the opposite [21, 22]. The mechanical properties of articular cartilage are clearly related to its well-balanced composition of collagens and proteoglycans. Those are maintained by the only co-existing compound within articular cartilage, the chondrocyte. This simple two-component structure was initially considered to be easy to reproduce and has been considered a perfect target for tissue engineering applications. However, hyaline cartilage is constantly dependent on a wide array of biomechanical und biochemical input in order to maintain its structure and integrity. It has been shown in an ankle fracture model that, when knee joint cartilage is not mechanically stimulated, it is suffering from atrophy when compared to loaded control [23]. It also has been shown that certain cartilage specific growth factors are upregulated following the postoperative course of surgically induced cartilage repair [24]. Proinflammatory cytokines, such as interleukin beta 1 and tumor necrosis factor alpha, play a major role within a functioning intra-articular environment [9]. Maintenance of correctly operating hyaline cartilage is owing to a required constant physicobiochemical input in order to provide a well-balanced joint homeostasis. Bearing this in mind, the postoperative rehabilitation following articular cartilage intervention is critical. However, it is required that the respective transplanttraverses certain stages of maturation [25]. It may take up 2–3 years post-implantation until a transplant can be regarded morphologically mature and fully ready to use [26]. It was shown during laboratory analysis of different clinical ACI products that the collagen 2/collagen 1 ratio was fardistant from that of native tissue at the time of transplantation [27]. This may be expected since the biopsies that are taken prior to ACI are usually small and contain few cells. Therefore, in vitro expansion procedures are required to increase cell numbers and it is known that proliferation is antagonistic to differentiation. Significant chondrocyte dedifferentiation occurs, with a concomitant increase in collagen I expression, as cell numbers and time in 2D monolayer, increase [28]. Progressive rehabilitation schemes have shown an improved outcome when comparing with traditional schemes following Matrix-assisted Chondrocyte Implantation (m-ACI) [29], but currently there is not enough evidence to constitute exact time points when a patient/a transplant is completely recovered. Related to failed complete restitution of articular cartilage, the transplantation of very immature tissue and consequently long patient rehabilitation tissue engineering principles are required for future cartilage defect repair.
3 Tissue Engineering
Tissue engineering aims to overcome limitations of traditional therapies by repairing or replacing damaged tissue with a de novo tissue that resembles the native tissue. These principles clearly aim for improved tissue quality at the time of transplantation in order to enhance the respective performance in vivo. Furthermore, patient recovery can be accelerated when more mature constructs are being implanted which require less time until full maturation. The term preconditioning has emerged to describe in vitro procedures that better prepare transplants for natural in vivo environments.
Among the large number of different ways to fulfil such principles, bioreactor tissue engineering defines a major aspect when articular cartilage repair is concerned. Bioreactors follow the goal to mimic (mostly biomechanical) natural joint surroundings. In that way cells behave in vitro as if they were in vivo [30]. In response to a biomechanical input, chondrocytes produce matrix as a natural response leading to protection and preparation for future mechanical stimulation; in the way of “form follows function.” Following this concept, potential transplants can be trained/preconditioned prior to re-implantation. A major challenge to overcome is the inverse relationship between tissue maturation and its potential to integrate and adapt to the healthy tissue surrounding the defect.
Various bioreactors have been developed which can apply any combination of mechanical, chemical, electrical or magnetic stimulation to enhance mass transfer and nutrient transport within seeded cells, facilitating the correct tissue development. During musculoskeletal tissue engineering, they are applied for growth of three dimensional tissues, such as cartilage, prior to implantation. In vivo articular cartilage is affected by different biomechanical forces, such as direct compression, tensile and shear forces, the generation of hydrostatic pressure, cyclic osmotic changes, electric gradients as well as changes in the pH. There are a multitude of bioreactor systems available of varying complexity. However, an ideal system would allow a precise control of the physiological environment of the culture. Temperature, oxygen concentration, pH value, nutrients, media flow rate, metabolite concentration and eventually as well specific tissue markers have to be kept within close limits. The culture of tissue is a non-steady state process in which parameters constantly change. Bioreactor culture has to provide nutrients and gases as the respective tissue is accustomed to in vivo. A bioreactor mechanical stimulus should be of a dynamic and intermittent character rather than being static pressure in order to induce chondrogenesis. There should be an adequate fluid exchange within the cultured constructs to provide every cell with nutrients. Applied biomechanical load should be physiological. While low levels of stress has been shown to remain unanswered by the cells, too strong mechanical stimulation can even result in apoptotic processes being initiated. Furthermore the importance of scaffold binding sites to transmit mechanical signals to seeded chondrocytes during the initial moments of bioreactor culture has been reported.
There are currently different options to stimulate chondrogenic cells. Certain basic principles are by now familiar. The cellular response to load is specific to the type of load applied, and this has been shown to be true across the knee joint [31]. During the same loading cycle, the lateral tibial plateau has a greater cartilage contact deformation, but lower cartilage contact area when compared with the medial compartment. Both compartments demonstrate a cartilage contact deformation of between 10 and 15 %. The rotation of the femur with respect to the tibia also varies during gait [32]. A physiological response of articular chondrocytes can be identified by the production of typical markers of hyaline cartilage. Those are generally collagen type 2, 6, 9 and 11, aggrecan as well as the different glycosaminoglycans that are attached to the protein backbone. Furthermore, a healthy response to mechanical stimulation can be detected when the cells are expressing lubricin (proteoglycan 4, PRG-4) and cartilage oligomeric protein (COMP). Also, histological and mechanical properties have to resemble mature cartilage in order to achieve functioning transplants [33].
Human hyaline cartilage can be described as being viscoelastic, resulting from its structural and chemical properties. The dynamic equilibrium of articular cartilage is related to its biphasic system. The solid phase is represented by porous and permeable parts of the ECM consistent of a collagen mesh, non-collagenous proteins and the non-covalently bound proteoglycans. The other phase is represented by the interstitial fluid along with ions solubilized within. The fluid phase can be separated into water and ions to have three different phases within articular cartilage. Negatively charged proteoglycans are capable of binding positively charged ions and thus water (fluid phase) along the osmotic gradient. This fluid influx, and the resulting cartilage swelling, is limited by external compressive forces and the resisting tensile collagenous network to reach a steady state. The complex orchestration of the cartilaginous network only works when the ECM and chondrocytes are well-balanced. Accordingly, it is the general goal during cartilage tissue engineering procedures to simultaneously proliferate and correctly differentiate chondrocytes, which are commonly cultured three-dimensionally. Human articular cartilage is composed of 60–80 % water, 10–20 % collagen type 2, 5–7 % aggregating proteoglycans, the rest being chondrocytes. This structure develops slowly and is dependent of mechanical forces during embryonic organogenesis. The tissue constantly remodels during the lifetime of the organism, emphasising the constant need for the correct biomechanical signals to be applied.
In such, synovial joints are the constant subject of several combining physical factors resulting in reactive change of volume, pressure gradient and fluid flow. Articular cartilage is typically exposed to stresses between 3 and 10 MPa with potential peaks up to 20 MPa at the hip joint. The human walking cadence is normally at the range of 1 Hz, which increases or decreases depending on speed of locomotion. Deformation of human cartilage without pathology is commonly at around 10–15 % strain. These are the cornerstones to which bioreactor tissue engineering principles are adjusting to in order to provoke a physiological tissue response. Hence, those values are true for mature human cartilage which has previously undergone complex differentiation processes during development and therefore have to be adapted for early in vitro tissue engineering processes. During in vitro tissue engineering it has been shown that chondrocytes are capable of reacting on biomechanical stimulations and converting them into intracellular signals which are essential for the maintenance of the entire tissue. While there is still a lack of knowledge, it is known that deformation of the chondrocyte itself may take part in a mechanical signal transduction pathway. Chondrocytes can react on shifting of currents and resulting electrical fields induced by mechanical forces. Furthermore mechanosensors such as integrins have been reported to reside on the extracellular membrane which can provide direct contact with the intracellular ECM. Moreover, mechanical stimulation can result in the activation of ion channels via shifting of the membrane potential [34, 35]. The exact mechanism of load sensing is unknown, and may be dependent on the cell type used (chondrocyte versus MSC), the scaffold material in which the cells are embedded, whether the matrix permits cell attachments and through which membrane binding proteins. It has previously been shown that the mechanoregulation of chondrocytes in agarose gels requires the cells themselves to produce extracellular matrix prior to being responsive [1]. Whether this is hydrogel specific remains to be seen.
4 Bioreactor Systems
There are a variety of different bioreactor-induced ways to apply mechanical load. Following uniaxial compression the tissue hydrostatic pressure is increasing related to the resistance of the negative charges within the solid phase. Hydrostatic pressure, mostly related to the fixed charged density of the proteoglycans, is increasing to prevent tissue deformation. The collagenous network does not have major effects during these processes. However, when the compression is maintained as with static compression, more fluid is extravasated off the respective construct and pressure upon the collagens is constantly increased. In parallel, less fluid can pass through the construct resulting in a more rigid solid phase. These processes take place in order to protect rigid parts of the ECM from higher load peaks. In contrast to direct compression, hydrostatic pressure does not result in macroscopic deformation of cartilage. Since the solid matrix phase of cartilage is intrinsically incompressible, no tissue deformation will occur under an external hydrostatic load. However, hydrostatic pressure is considered as one of the most important forms of loading to act on cartilage in vivo. In contrast to direct compression, hydrostatic pressure is commonly not capable of harming the exposed tissue. Following the external application of hydrostatic pressure, hydrostatic pressure will increase inside a subjected cell-scaffold-construct. Pressurization results in only 10 % of the load remaining as direct compression on the solid phase. Hydrostatic pressure may result in only minimal strain at the cellular level. Thus, hydrostatic pressure may have direct effects on cell membrane ion channels with a pressure-dependent change in intracellular ion concentrations. Alterations in intracellular ion concentrations lead to changes in cellular gene expression, protein production and eventually biomechanical properties. During locomotion, and thus joint movement, intermittent shear stress is applied on to the tissue surface. In response, articular cartilage does not reduce volume, but deforms its structure. Shear stress is therefore mostly affecting the upper layers of cartilage or cell-scaffold constructs. Hence, chondrocytes within the upper layer of cartilage are commonly horizontally orientated, while those in deeper layers are usually found in vertical columns. But also cells within the less flexible deeper layers are thus strongly affected by shear stress, which results in an increased collagen content. There are bioreactors capable of exposing subjected constructs to isolated stimuli, while there are also devices that are capable of a combinatory mechanical input. The latter more closely resembles the human articulating joint and thus may generate a more tissue specific response. During bioreactor tissue engineering, one can adjust the magnitude, frequency, onset, and duration of load application. A physiological biomechanical stimulus closely resembles joint motion of a human knee joint. The interaction between subchondral bone and cartilage which is a important for load transmission and maintenance of both tissues. This interaction is hard to mimic in a bioreactor. Within which, cartilage is recognizing stimuli as a rolling movement of direct compression in concert with a generation of shear and tensile forces and high hydrostatic pressure [36].
The multitude of bioreactor systems currently used is further complicated by a lack of standardisation and validation. Most systems are custom built, making comparisons between devices difficult. Additionally the various groups use different cells (age, species, origin, expanded versus non-expanded, different culture media) and the scaffolds in which the cells are embedded can also be radically different, leading to a varying degree of load transmission potentially through differing mechanisms. Even taking these differences into account, certain trends have become apparent.
4.1 Static Culture and Tension
When 3-D cultured chondrocytes are cultured in vitro under static free-swelling conditions, which is the current practice to realize matrix-assisted chondrocyte transplantation, it has been shown that little benefit is observed when chondrogenesis is concerned [27]. Static culture results in a non-homogenous cell distribution that does not resemble the native tissue [37]. Extracellular matrix production is not enhanced by isolated 3-D surroundings and chondrocytes have been shown to even downregulate typical markers for chondrogenic differentiation such as collagen type 2 or aggrecan under such growth conditions [38]. Furthermore the failure of static cultures to recreate the mechanical environment of in vivo tissue and to achieve mass transport of nutrients into large scaffolds result in the preferential growth of cells at the periphery of the scaffold which lacks the biomechanical and histological properties of native tissue- it has been previously termed as an edging effect [39].
Similar to static culture, tensile loading is not a typical stimulus within human joint surroundings and thus in isolation is not truly physiologically relevant for articular cartilage. Thereby, experimental studies mostly described detrimental effects following tension bioreactor tissue engineering. Yet, inhibitory effects such as down-regulation of proteoglycan production have been reported [40].
4.2 Bioreactors- Increasing Fluid Exchange
4.2.1 Flow Perfusion
Interstitial flow in articular cartilage is secondary to shear and compressive deformations during locomotion [41]. It is linked with the well-characterized heterogeneity in structure and composition of its extracellular matrix. During flow perfusion bioreactor tissue engineering, cell culture medium is pumped continuously through a cell-matrix construct without internal transport limitations. Hereby the local nutrient supply, mass transfer is higher when comparing to rotating wall or spinner flask systems [42]. Homogenous cell distribution and higher cell seeding effectiveness are resultant. Also biomechanical input is placed onto the cells, which has been shown to enhance mechanical properties but as well to enhance the expression of the osteoblastic phenotype [43]. Flow perfusion may serve to provide every construct-cultured chondrocyte with nutrients in order to avoid edging effects. However, simple flow perfusion does not propose a relevant and adequate stimulus for articular chondrocytes to establish a functioning ECM as under these conditions it is not associated with a concurrent change in osmolarity within the tissue.
4.2.2 Rotating Wall
The principle of a rotating wall bioreactor is following basic rules of gravity. Cell-seeded scaffolds are cultured within medium-filled culture flasks. These are constantly rotated and thus kept from descending to the bottom of the flask. Thereby a dynamic laminar flow with a definitive shearing force evolves at the construct surface which is provoking an even cellular distribution and enhanced biomechanical properties. The rotating wall bioreactor is kept within a standard CO2 incubator and to enable proper gaseous exchange, one side of the bioreactor chamber is a semi-permeable membrane. It was also shown that the rotating wall principle as well is applicable for isolated cells [44]. As with perfusion bioreactors, the rotating wall bioreactor allows for a larger construct to be cultured with a more even cell distribution, but does not apply mechanical stimulation more associated with an articulating joint.
4.2.3 Spinner Flask
During spinner flask bioreactor tissue engineering, cell-seeded scaffolds are attached to e.g. needles and are suspended in a flask of culture medium. A magnetic stir bar is constantly mixing the medium from the bottom of the flask and thus providing with a turbulent mixing of medium nutrients to the respective constructs. Thereby, the mechanical properties of the resulting tissue are enhanced. Drawbacks are to be found within the fact that related to the turbulent medium supply, application spikes appear that may result in mechanical cell death and unbalanced nutrient supply. Also, a fibrous capsule may generate around the constructs with biomechanically weak tissue within [37]. It was furthermore shown that alkaline phosphatase activity and osteocalcin secretion was higher in cells that were previously cultured within a spinner flask when comparing to static or rotating wall bioreactors indicating osteogenetic processes.
4.3 Bioreactors- Applying Mechanical Loads
4.3.1 Hydrostatic Pressure
During physiological locomotion, synovial joint fluid is kept within the cartilage ECM, which is increasing hydrostatic pressure. It is related to the negatively charged proteoglycans and limited by the collagenous network. When hydrostatic pressure is applied intermittently at physiological levels ECM production is promoted. The opposite is setting in when the stimulus is static. Parkkinen and colleagues already in 1993 reported an increased glycosaminoglycan synthesis during hydrostatic pressure bioreactor tissue engineering in cartilage explant culture. This effect could not be provoked within monolayer culture [45]. In contrast, when cartilage cells are cultured three-dimensionally within a scaffold and furthermore subjected to static hydrostatic pressure the production of external matrix is upregulated. One has to note that when the applied external artificial stimulus is non-physiological, e.g. too high, apoptosis may be induced within the cells. Generally hydrostatic pressure is one very attractive mechanical stimulus in order to increase chondrogenesis and thus ECM production and modulation. It works best when applied dynamically within physiological limits of 7–10 MPa. Within this range it has also been demonstrated that hydrostatic pressure has the potential to enhance chondrogenesis of both bone marrow [46] and infrapatellar fat pad derived MSCs [47].
4.3.2 Compression
Uniaxial compression as being observed within human joints is one of the most heavily studied modes of mechanical stimulation. If a compressive force is applied statically over time it is now accepted that in the majority of cases, a detrimental effect to the tissue will arise. Down-regulation of the typical markers collagen type 2 and 6, aggrecan and glycosaminoglycans are the result. Also, when cultured over short periods, dynamic loading of 15 % strain at 1 Hz did not result in a significantly upregulated hyaline-like ECM expression when comparing with static culturing conditions [48]. Numerous studies involving dynamic compression suggest a beneficial effect of load for chondrogenesis within chondrocytes, which is identified by the upregulation of collagen type 2 and aggrecan [1, 49]. Of studies involving compression alone, mostly a frequency of 1 Hz and either 10 % or 15 % compression has been applied [1, 49, 50]. Similar magnitudes led to the greatest increase in chondrogenic gene expression and GAG synthesis in MSCs [51–53]. Interestingly, it was also shown that dynamic modulation of chondrocytes can also to an extent counteract the usually detrimental expression of interleukin 1 beta [54, 55]. The effect of uniaxial compression appears to be dependent on the extent of matrix that is present around the cells [1]. It has also been proposed that it shows differing effects depending on whether chondrocytes or MSCs are stimulated. While there is little doubt that uniaxial load stimulates matrix synthesis in mature chondrocytes, uniaxial load alone does not appear to be able to induce chondrogenesis in BMSCs. Either a shear component is required [56] or a pre-stimulation with TGF-β is required to first induce chondrogenesis, resulting a in cell phenotype responsive to compression alone [57].
4.4 Combined Stimuli
Bioreactors have been developed which are capable of applying shear superimposed over compression [30, 58]. An example of such a device can be seen in Fig. 5.1. Within this device, shear, compression or a combination of the two can be independently controlled and regulated. Related to the fact that human articular chondrocytes are the subject of combined mechanical stimuli [59] it has been shown during in vitro experiments that a combined mechanical stimulus can be more efficient in generating chondrogenesis when compared to isolated stimulation [60]. The implementation of motion patterns which approximate the kinematics of physiological joint motion can lead to the development of a tissue with properties similar to native articular cartilage. It has been shown that responses can be detected at the mRNA level within hours of the load being applied [58]. Studies have shown that dynamic compression and sliding surface motion, applied by a ceramic ball, improves the gene expression and the synthesis of cartilage specific matrix molecules in chondrocyte-scaffold constructs [61]. In such combined bioreactors one is capable to recognize differences in reacting ECM production. Sliding surface motion will more strongly result in the expression of Lubricin. Dynamic compression will more strongly evoke the expression of collagen type 2 and aggrecan. These different types of matrix expression are reminiscent of those appearing in vivo where chondrocytes are adapted to their biomechanical input [62]. It has been demonstrated that shear, superimposed over compression, is able to induce chondrogenesis of human MSCs in the absence of exogenous TGF-β [63] and as expected the response is dependent on the amplitude and frequency applied [64]. It has been proposed that shear is required for chondrogenic induction of MSCs [56].
Left: Cross sectional schematic of the scaffold with its holder. The ceramic hip ball (A) is pressed against the cell-seeded scaffold (B). The scaffold (B) is held in place within the main holder (C) by means of a circular PEEK (poly(ether ether ketone)) ring (D). A final ring of cell free fibrin/ PU scaffold (E) provides a final structural support to hold the sample in place. Right: Sample in holder. Both the sample and ball are housed in a glass bell to increase sterility (Reproduced from Schätti et al. [56] with kind permission from eCM journal)
4.5 Synergistic Processes
The synovial joint cavity is the host of a great variety of different growth factors, cytokines and other proteins. They interact heavily with mechanical stimuli in order to orchestrate an equilibrium within the articular cartilage. It is known that growth factors work synergistically with mechanical stimuli [65]. Such synergism was demonstrated when bovine articular chondrocytes overexpressing bone-morphogenetic protein-2 (BMP-2) were subjected to dynamic compression, shear and fluid flow within a bioreactor. When comparing to the isolated stimuli it was discovered that singular BMP-2 influence was more effective to induce the expression of typical chondrogenic markers when compared to a singular mechanical input. However, when those stimuli were combined clear synergistic effects were detected that were higher than the sum of the individual treatments for the expression GAG/DNA, collagen type 2, and cartilage oligomeric protein (COMP). Histology revealed a functional organization in combined groups including an intense safranin O staining. Also, immunostaining for collagen II and aggrecan was well detected with most intense expression within combined groups [38]. Paralleling growth factors or cytokines, as well hypoxia has been shown to result in an improved chondrogenesis over control in terms of stabilization of the chondrogenic phenotype [39].
Similar effects have been seen during the promotion of chondrogenesis in MSCs. Adipose derived cells transduced with IGF1 have been shown to lead to a chondrogenic response [66]. The chondrogenic response of human MSCs under multiaxial load can be further enhanced when the cells are transduced with adenoviral Sox9 [67] and such systems can be used to dissect the different regulation pathways of chondrogenic genes.
5 Conclusion
For the application of bioreactor tissue engineering, cells, either matrix-associated or condensed, are required for mechanical stimulation. When articular cartilage repair is concerned, chondrocytes are already involved with the maintenance of cartilage tissue and thus tailored for tissue engineering applications [68]. Hence, current clinical tissue engineering principles concentrate on the application of autologous chondrocytes. Yet, only one clinical bioreactor tissue engineering product is using autologous chondrocytes for knee joint cartilage repair, which have been mechanically stimulated within a bioreactor in beforehand, is currently in use [69]. Chondrocytes are expanded and seeded into a bovine type I collagen 3-dimensional honey- comb matrix. The seeded scaffold is then processed in a bioreactor in which culture conditions, including hydrostatic pressure, seeks to induce the chondrocytes to synthesize cartilage glycoproteins. On line, quality control is becoming a novel issue in cartilage repair. In particular tissue construct mechanical properties may be one significant part to support tissue quality and consecutive in vivo resilience. Notably, the Food and Drug Administration (FDA) requested mechanical data for all articular cartilage repair products in their guidance for “Repair or Replace Knee Cartilage”, which additionally emphasizes the importance of mechanical characterization of cartilage constructs.
Related to the fact that the realization of ACI still requires two full operations and is adjunctive with a potential harmful donor-site morbidity [70], alternative cell sources are being robustly investigated [71, 72]. Almqvist and colleagues have shown satisfying midterm results when applying allogenic chondrocytes for knee cartilage defect repair among 21 subjects [73]. However, allogenic material may be ethical challenging, has potential disease transmission and immunological rejection risks and moreover might not be accessible in every country. Mesenchymal stem cells are capable of differentiating into different tissues such as bone and cartilage. Complex differentiation processes are actually required when aiming for true and foremost lasting chondrogenic differentiation [74]. Though, clinical studies have shown a similar outcome when comparing the effects of autologous chondrocyte transplantation versus autologous stem cell transplantation for the treatment of knee cartilage defects [75]. However, chondrogenic differentiation of stem cells is a difficult task, while current methods tend to induce an inadequate, hypertrophic differentiation cascade reminiscent of endochondral bone formation [76]. While MSCs have been found in numerous tissues, the detection of a progenitor like cell within cartilage itself which does not appear to be hypertrophic [77] may lead to new potential therapies. Effective bioreactor tissue engineering, potentially combining mechanical with physical stimuli, may be very attractive for future cartilage repair procedures when using mesenchymal stem cells [56].
Although progress has been achieved, there are still some significant hurdles to overcome before preconditioned tissue engineered cartilage repair can become a clinical reality. Not only do the optimal culture conditions need to be found, but they need to be implemented into systems which are good manufacturing process (GMP) compliant. This requires that each culture vessel is independent and there is no potential for cross contamination between patients. The tracking and record keeping required for GMP also increases costs, meaning that the treatment must be demonstrably an improvement on current treatments. Also the logistics of the system need to be considered. If a central manufacturing plant is established then reliable transportation to and from the hospital may be required and this might involve shipment of live human products across international borders. Otherwise the facilities need to be on-site and economies of scale need to be considered. In either case, suitable quality control and tracking is required.
Once overcome, the economic advantages of a reliable treatment for articular defects are vast. In the future, cartilage defects may be treated by use of bioreactor preconditioned de novo cell-scaffold constructs, which are able to still integrate, provide with high quality repair tissue and severely reduce the time required for patient rehabilitation. When efficient, the high initial tissue engineering cost may be offset by highly effective osteoarthritis prevention which is all for the benefit of the patient.
References
Mauck RL, Soltz MA, Wang CC, Wong DD, Chao PH, Valhmu WB, et al. Functional tissue engineering of articular cartilage through dynamic loading of chondrocyte-seeded agarose gels. J Biomech Eng. 2000;122(3):252–60.
Madry H. The subchondral bone: a new frontier in articular cartilage repair. Knee Surg Sports Traumatol Arthrosc. 2010;18(4):417–8.
Rauh J, Milan F, Gunther KP, Stiehler M. Bioreactor systems for bone tissue engineering. Tissue Eng Part B Rev. 2011;17(4):263–80.
Deng T, Lv J, Pang J, Liu B, Ke J. Construction of tissue-engineered osteochondral composites and repair of large joint defects in rabbit. J Tissue Eng Regen Med. 2012 Jul 9. doi:10.1002/term.1556. [Epub ahead of print].
Schaefer D, Martin I, Jundt G, Seidel J, Heberer M, Grodzinsky A, et al. Tissue-engineered composites for the repair of large osteochondral defects. Arthritis Rheum. 2002;46(9):2524–34.
Curl WW, Krome J, Gordon ES, Rushing J, Smith BP, Poehling GG. Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy. 1997;13(4):456–60.
Buckwalter JA, Mankin HJ, Grodzinsky AJ. Articular cartilage and osteoarthritis. Instr Course Lect. 2005;54:465–80.
Lotz MK, Carames B. Autophagy and cartilage homeostasis mechanisms in joint health, aging and OA. Nat Rev Rheumatol. 2011;7(10):579–87.
Kapoor M, Martel-Pelletier J, Lajeunesse D, Pelletier JP, Fahmi H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011;7(1):33–42.
Ding C, Cicuttini F, Blizzard L, Jones G. Genetic mechanisms of knee osteoarthritis: a population-based longitudinal study. Arthritis Res Ther. 2006;8(1):R8.
Ding C, Cicuttini F, Scott F, Cooley H, Boon C, Jones G. Natural history of knee cartilage defects and factors affecting change. Arch Intern Med. 2006;166(6):651–8.
Farr J, Cole B, Dhawan A, Kercher J, Sherman S. Clinical cartilage restoration: evolution and overview. Clin Orthop Relat Res. 2011;469(10):2696–705.
Loken S, Heir S, Holme I, Engebretsen L, Aroen A. 6-year follow-up of 84 patients with cartilage defects in the knee. Knee scores improved but recovery was incomplete. Acta Orthop. 2010;81(5):611–8.
Vasiliadis HS, Wasiak J. Autologous chondrocyte implantation for full thickness articular cartilage defects of the knee. Cochrane Database Syst Rev. 2010;10:CD003323.
Knutsen G, Drogset JO, Engebretsen L, Grontvedt T, Isaksen V, Ludvigsen TC, et al. A randomized trial comparing autologous chondrocyte implantation with microfracture. Findings at five years. J Bone Joint Surg Am. 2007;89(10):2105–12.
Knutsen G, Engebretsen L, Ludvigsen TC, Drogset JO, Grontvedt T, Solheim E, et al. Autologous chondrocyte implantation compared with microfracture in the knee. A randomized trial. J Bone Joint Surg Am. 2004;86-A(3):455–64.
Saris DB, Vanlauwe J, Victor J, Almqvist KF, Verdonk R, Bellemans J, et al. Treatment of symptomatic cartilage defects of the knee: characterized chondrocyte implantation results in better clinical outcome at 36 months in a randomized trial compared to microfracture. Am J Sports Med. 2009;37 Suppl 1:10S–9.
Bentley G, Biant LC, Vijayan S, Macmull S, Skinner JA, Carrington RW. Minimum ten-year results of a prospective randomised study of autologous chondrocyte implantation versus mosaicplasty for symptomatic articular cartilage lesions of the knee. J Bone Joint Surg Br. 2012;94(4):504–9.
Brun P, Dickinson SC, Zavan B, Cortivo R, Hollander AP, Abatangelo G. Characteristics of repair tissue in second-look and third-look biopsies from patients treated with engineered cartilage: relationship to symptomatology and time after implantation. Arthritis Res Ther. 2008;10(6):R132.
Niemeyer P, Pestka JM, Salzmann GM, Sudkamp NP, Schmal H. Influence of cell quality on clinical outcome after autologous chondrocyte implantation. Am J Sports Med. 2012;40(3):556–61.
Vasiliadis HS, Danielson B, Ljungberg M, McKeon B, Lindahl A, Peterson L. Autologous chondrocyte implantation in cartilage lesions of the knee: long-term evaluation with magnetic resonance imaging and delayed gadolinium-enhanced magnetic resonance imaging technique. Am J Sports Med. 2010;38(5):943–9.
Vavken P, Samartzis D. Effectiveness of autologous chondrocyte implantation in cartilage repair of the knee: a systematic review of controlled trials. Osteoarthritis Cartilage. 2010;18(6):857–63.
Hinterwimmer S, Krammer M, Krotz M, Glaser C, Baumgart R, Reiser M, et al. Cartilage atrophy in the knees of patients after seven weeks of partial load bearing. Arthritis Rheum. 2004;50(8):2516–20.
Schmal H, Niemeyer P, Zwingmann J, Stoffel F, Sudkamp NP, Mehlhorn AT. Association between expression of the bone morphogenetic proteins 2 and 7 in the repair of circumscribed cartilage lesions with clinical outcome. BMC Musculoskelet Disord. 2010;11:170.
Hirschmuller A, Baur H, Braun S, Kreuz PC, Sudkamp NP, Niemeyer P. Rehabilitation after autologous chondrocyte implantation for isolated cartilage defects of the knee. Am J Sports Med. 2011;39(12):2686–96.
Cerynik DL, Lewullis GE, Joves BC, Palmer MP, Tom JA. Outcomes of microfracture in professional basketball players. Knee Surg Sports Traumatol Arthrosc. 2009;17(9):1135–9.
Albrecht C, Tichy B, Nurnberger S, Hosiner S, Zak L, Aldrian S, et al. Gene expression and cell differentiation in matrix-associated chondrocyte transplantation grafts: a comparative study. Osteoarthritis Cartilage. 2011;19(10):1219–27.
Schnabel M, Marlovits S, Eckhoff G, Fichtel I, Gotzen L, Vecsei V, et al. Dedifferentiation-associated changes in morphology and gene expression in primary human articular chondrocytes in cell culture. Osteoarthritis Cartilage. 2002;10(1):62–70.
Ebert JR, Robertson WB, Lloyd DG, Zheng MH, Wood DJ, Ackland T. Traditional vs accelerated approaches to post-operative rehabilitation following matrix-induced autologous chondrocyte implantation (MACI): comparison of clinical, biomechanical and radiographic outcomes. Osteoarthritis Cartilage. 2008;16(10):1131–40.
Wimmer MA, Grad S, Kaup T, Hanni M, Schneider E, Gogolewski S, et al. Tribology approach to the engineering and study of articular cartilage. Tissue Eng. 2004;10(9–10):1436–45.
Salzmann GM, Buchberger MS, Stoddart MJ, Grad S, Milz S, Niemyer P, et al. Varying regional topology within knee articular chondrocytes under simulated in vivo conditions. Tissue Eng Part A. 2011;17(3–4):451–61.
Hosseini A, Van de Velde SK, Kozanek M, Gill TJ, Grodzinsky AJ, Rubash HE, et al. In-vivo time-dependent articular cartilage contact behavior of the tibiofemoral joint. Osteoarthritis Cartilage. 2010;18(7):909–16.
Bian L, Fong JV, Lima EG, Stoker AM, Ateshian GA, Cook JL, et al. Dynamic mechanical loading enhances functional properties of tissue-engineered cartilage using mature canine chondrocytes. Tissue Eng Part A. 2010;16(5):1781–90.
Millward-Sadler SJ, Wright MO, Davies LW, Nuki G, Salter DM. Mechanotransduction via integrins and interleukin-4 results in altered aggrecan and matrix metalloproteinase 3 gene expression in normal, but not osteoarthritic, human articular chondrocytes. Arthritis Rheum. 2000;43(9):2091–9.
Millward-Sadler SJ, Wright MO, Flatman PW, Salter DM. ATP in the mechanotransduction pathway of normal human chondrocytes. Biorheology. 2004;41(3–4):567–75.
Heath CA, Magari SR. Mini-review: mechanical factors affecting cartilage regeneration in vitro. Biotechnol Bioeng. 1996;50(4):430–7.
Vunjak-Novakovic G, Martin I, Obradovic B, Treppo S, Grodzinsky AJ, Langer R, et al. Bioreactor cultivation conditions modulate the composition and mechanical properties of tissue-engineered cartilage. J Orthop Res. 1999;17(1):130–8.
Salzmann GM, Nuernberger B, Schmitz P, Anton M, Stoddart MJ, Grad S, et al. Physicobiochemical synergism through gene therapy and functional tissue engineering for in vitro chondrogenesis. Tissue Eng Part A. 2009;15(9):2513–24.
Wernike E, Li Z, Alini M, Grad S. Effect of reduced oxygen tension and long-term mechanical stimulation on chondrocyte-polymer constructs. Cell Tissue Res. 2008;331(2):473–83.
Connelly JT, Vanderploeg EJ, Levenston ME. The influence of cyclic tension amplitude on chondrocyte matrix synthesis: experimental and finite element analyses. Biorheology. 2004;41(3–4):377–87.
Olivares AL, Marsal E, Planell JA, Lacroix D. Finite element study of scaffold architecture design and culture conditions for tissue engineering. Biomaterials. 2009;30(30):6142–9.
Elder BD, Athanasiou KA. Hydrostatic pressure in articular cartilage tissue engineering: from chondrocytes to tissue regeneration. Tissue Eng Part B Rev. 2009;15(1):43–53.
Yeatts AB, Fisher JP. Bone tissue engineering bioreactors: dynamic culture and the influence of shear stress. Bone. 2011;48(2):171–81.
Marlovits S, Tichy B, Truppe M, Gruber D, Vecsei V. Chondrogenesis of aged human articular cartilage in a scaffold-free bioreactor. Tissue Eng. 2003;9(6):1215–26.
Parkkinen JJ, Ikonen J, Lammi MJ, Laakkonen J, Tammi M, Helminen HJ. Effects of cyclic hydrostatic pressure on proteoglycan synthesis in cultured chondrocytes and articular cartilage explants. Arch Biochem Biophys. 1993;300(1):458–65.
Miyanishi K, Trindade MC, Lindsey DP, Beaupre GS, Carter DR, Goodman SB, et al. Dose- and time-dependent effects of cyclic hydrostatic pressure on transforming growth factor-beta3-induced chondrogenesis by adult human mesenchymal stem cells in vitro. Tissue Eng. 2006;12(8):2253–62.
Vinardell T, Rolfe RA, Buckley CT, Meyer EG, Ahearne M, Murphy P, et al. Hydrostatic pressure acts to stabilise a chondrogenic phenotype in porcine joint tissue derived stem cells. Eur Cell Mater. 2012;23:121–34.
Nicodemus GD, Bryant SJ. The role of hydrogel structure and dynamic loading on chondrocyte gene expression and matrix formation. J Biomech. 2008;41(7):1528–36.
Chowdhury TT, Bader DL, Shelton JC, Lee DA. Temporal regulation of chondrocyte metabolism in agarose constructs subjected to dynamic compression. Arch Biochem Biophys. 2003;417(1):105–11.
Mauck RL, Wang CC, Oswald ES, Ateshian GA, Hung CT. The role of cell seeding density and nutrient supply for articular cartilage tissue engineering with deformational loading. Osteoarthritis Cartilage. 2003;11(12):879–90.
Campbell JJ, Lee DA, Bader DL. Dynamic compressive strain influences chondrogenic gene expression in human mesenchymal stem cells. Biorheology. 2006;43(3–4):455–70.
Huang CY, Hagar KL, Frost LE, Sun Y, Cheung HS. Effects of cyclic compressive loading on chondrogenesis of rabbit bone-marrow derived mesenchymal stem cells. Stem Cells. 2004;22(3):313–23.
Terraciano V, Hwang N, Moroni L, Park HB, Zhang Z, Mizrahi J, et al. Differential response of adult and embryonic mesenchymal progenitor cells to mechanical compression in hydrogels. Stem Cells. 2007;25(11):2730–8.
Chowdhury TT, Bader DL, Lee DA. Dynamic compression counteracts IL-1 beta-induced release of nitric oxide and PGE2 by superficial zone chondrocytes cultured in agarose constructs. Osteoarthritis Cartilage. 2003;11(9):688–96.
Chowdhury TT, Bader DL, Lee DA. Dynamic compression counteracts IL-1beta induced iNOS and COX-2 activity by human chondrocytes cultured in agarose constructs. Biorheology. 2006;43(3–4):413–29.
Schatti O, Grad S, Goldhahn J, Salzmann G, Li Z, Alini M, et al. A combination of shear and dynamic compression leads to mechanically induced chondrogenesis of human mesenchymal stem cells. Eur Cell Mater. 2011;22:214–25.
Thorpe SD, Buckley CT, Vinardell T, O’Brien FJ, Campbell VA, Kelly DJ. The response of bone marrow-derived mesenchymal stem cells to dynamic compression following TGF-beta3 induced chondrogenic differentiation. Ann Biomed Eng. 2010;38(9):2896–909.
Stoddart MJ, Ettinger L, Hauselmann HJ. Enhanced matrix synthesis in de novo, scaffold free cartilage-like tissue subjected to compression and shear. Biotechnol Bioeng. 2006;95(6):1043–51.
Brommer H, Brama PA, Laasanen MS, Helminen HJ, van Weeren PR, Jurvelin JS. Functional adaptation of articular cartilage from birth to maturity under the influence of loading: a biomechanical analysis. Equine Vet J. 2005;37(2):148–54.
Grad S, Gogolewski S, Alini M, Wimmer MA. Effects of simple and complex motion patterns on gene expression of chondrocytes seeded in 3D scaffolds. Tissue Eng. 2006;12(11):3171–9.
Grad S, Loparic M, Peter R, Stolz M, Aebi U, Alini M. Sliding motion modulates stiffness and friction coefficient at the surface of tissue engineered cartilage. Osteoarthritis Cartilage. 2012;20(4):288–95.
Li Z, Yao S, Alini M, Grad S. Different response of articular chondrocyte subpopulations to surface motion. Osteoarthritis Cartilage. 2007;15(9):1034–41.
Li Z, Kupcsik L, Yao SJ, Alini M, Stoddart MJ. Mechanical load modulates chondrogenesis of human mesenchymal stem cells through the TGF-beta pathway. J Cell Mol Med. 2010;14(6A):1338–46.
Li Z, Yao SJ, Alini M, Stoddart MJ. Chondrogenesis of human bone marrow mesenchymal stem cells in fibrin-polyurethane composites is modulated by frequency and amplitude of dynamic compression and shear stress. Tissue Eng Part A. 2010;16(2):575–84.
Mauck RL, Nicoll SB, Seyhan SL, Ateshian GA, Hung CT. Synergistic action of growth factors and dynamic loading for articular cartilage tissue engineering. Tissue Eng. 2003;9(4):597–611.
Li J, Zhao Q, Wang E, Zhang C, Wang G, Yuan Q. Dynamic compression of rabbit adipose-derived stem cells transfected with insulin-like growth factor 1 in chitosan/gelatin scaffolds induces chondrogenesis and matrix biosynthesis. J Cell Physiol. 2012;227(5):2003–12.
Kupcsik L, Stoddart MJ, Li Z, Benneker LM, Alini M. Improving chondrogenesis: potential and limitations of SOX9 gene transfer and mechanical stimulation for cartilage tissue engineering. Tissue Eng Part A. 2010;16(6):1845–55.
Grad S, Eglin D, Alini M, Stoddart MJ. Physical stimulation of chondrogenic cells in vitro: a review. Clin Orthop Relat Res. 2011;469(10):2764–72.
Crawford DC, Heveran CM, Cannon Jr WD, Foo LF, Potter HG. An autologous cartilage tissue implant NeoCart for treatment of grade III chondral injury to the distal femur: prospective clinical safety trial at 2 years. Am J Sports Med. 2009;37(7):1334–43.
Matricali GA, Dereymaeker GP, Luyten FP. Donor site morbidity after articular cartilage repair procedures: a review. Acta Orthop Belg. 2010;76(5):669–74.
Stoddart MJ, Grad S, Eglin D, Alini M. Cells and biomaterials in cartilage tissue engineering. Regen Med. 2009;4(1):81–98.
Caplan AI. Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physiol. 2007;213(2):341–7.
Dhollander AA, Verdonk PC, Lambrecht S, Verdonk R, Elewaut D, Verbruggen G, et al. Midterm results of the treatment of cartilage defects in the knee using alginate beads containing human mature allogenic chondrocytes. Am J Sports Med. 2012;40(1):75–82.
Goldring MB, Tsuchimochi K, Ijiri K. The control of chondrogenesis. J Cell Biochem. 2006;97(1):33–44.
Nejadnik H, Hui JH, Feng Choong EP, Tai BC, Lee EH. Autologous bone marrow-derived mesenchymal stem cells versus autologous chondrocyte implantation: an observational cohort study. Am J Sports Med. 2010;38(6):1110–6.
Weiss S, Hennig T, Bock R, Steck E, Richter W. Impact of growth factors and PTHrP on early and late chondrogenic differentiation of human mesenchymal stem cells. J Cell Physiol. 2010;223(1):84–93.
Williams R, Khan IM, Richardson K, Nelson L, McCarthy HE, Analbelsi T, et al. Identification and clonal characterisation of a progenitor cell sub-population in normal human articular cartilage. PLoS One. 2010;5(10):e13246.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag London
About this chapter
Cite this chapter
Salzmann, G.M., Stoddart, M.J. (2014). Bioreactor Tissue Engineering for Cartilage Repair. In: Emans, P., Peterson, L. (eds) Developing Insights in Cartilage Repair. Springer, London. https://doi.org/10.1007/978-1-4471-5385-6_5
Download citation
DOI: https://doi.org/10.1007/978-1-4471-5385-6_5
Published:
Publisher Name: Springer, London
Print ISBN: 978-1-4471-5384-9
Online ISBN: 978-1-4471-5385-6
eBook Packages: MedicineMedicine (R0)