Overview
- Highlights the development by the emerging field of regenerative medicine of bioengineered tissue platforms that best mimic the structure and cellular heterogeneity of in vivo tissue
- Details how microengineering technologies have resulted in advanced methods for creating and culturing 3-D human tissue--resulting in viability for longer periods of time
- Explores the potential of bioengineering and microengineering to aid in overcoming limitations to genetic and histological tumor classification and the development of personalized therapies
Part of the book series: Cancer Drug Discovery and Development (CDD&D)
Buy print copy
About this book
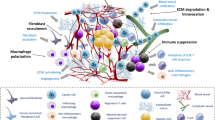
Cancer cell biology research in general, and anti-cancer drug development specifically, still relies on standard cell culture techniques that place the cells in an unnatural environment. As a consequence, growing tumor cells in plastic dishes places a selective pressure that substantially alters their original molecular and phenotypic properties.The emerging field of regenerative medicine has developed bioengineered tissue platforms that can better mimic the structure and cellular heterogeneity of in vivo tissue, and are suitable for tumor bioengineering research. Microengineering technologies have resulted in advanced methods for creating and culturing 3-D human tissue. By encapsulating the respective cell type or combining several cell types to form tissues, these model organs can be viable for longer periods of time and are cultured to develop functional properties similar to native tissues. This approach recapitulates the dynamic role of cell–cell, cell–ECM, and mechanical interactions inside the tumor. Further incorporation of cells representative of the tumor stroma, such as endothelial cells (EC) and tumor fibroblasts, can mimic the in vivo tumor microenvironment. Collectively, bioengineered tumors create an important resource for the in vitro study of tumor growth in 3D including tumor biomechanics and the effects of anti-cancer drugs on 3D tumor tissue. These technologies have the potential to overcome current limitations to genetic and histological tumor classification and development of personalized therapies.
Similar content being viewed by others
Keywords
Table of contents (10 chapters)
-
Front Matter
Editors and Affiliations
About the editors
Aleksander Skardal, PhD, is Assistant Professor, Regenerative Medicine and Biomedical Engineering, Wake Forest Institute for Regenerative Medicine, Wake Forest School of Medicine.
Bibliographic Information
Book Title: Tumor Organoids
Editors: Shay Soker, Aleksander Skardal
Series Title: Cancer Drug Discovery and Development
DOI: https://doi.org/10.1007/978-3-319-60511-1
Publisher: Humana Cham
eBook Packages: Medicine, Medicine (R0)
Copyright Information: Springer International Publishing AG, part of Springer Nature 2018
Hardcover ISBN: 978-3-319-60509-8Published: 27 October 2017
Softcover ISBN: 978-3-319-86874-5Published: 28 August 2018
eBook ISBN: 978-3-319-60511-1Published: 20 October 2017
Series ISSN: 2196-9906
Series E-ISSN: 2196-9914
Edition Number: 1
Number of Pages: XVI, 213
Number of Illustrations: 2 b/w illustrations, 50 illustrations in colour
Topics: Cancer Research