Abstract
Free radicals are highly reactive chemical species generated during normal metabolic processes, which in excess can lead to membrane damage. Elaborate anti-oxidant defence systems exist to protect against oxidative stress.
There is accumulating evidence of altered antioxidant capacity in schizophrenia. Membrane dysfunction can be secondary to free radical-mediated pathology, and may contribute to specific aspects of schizophrenic symptomatology and complications of its treatment. Specifically, free radical-mediated abnormalities may contribute to the development of a number of clinically significant consequences, including prominent negative symptoms, tardive dyskinesia, neurological ‘soft’ signs and parkinsonian symptoms. Our previous results showing altered membrane dynamics and antioxidant enzyme activities in schizophrenia, and findings from other investigators, are consistent with the notion of free radical-mediated neurotoxicity in schizophrenia. These findings provide a theoretical basis from which the development of novel therapeutic strategies such as fatty acid and antioxidant supplementation can occur in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Schizophrenia is one of the major mental disorders and is of unknown aetiology. It is associated with a great deal of morbidity and annual economic costs that total billions of dollars. The illness has a variable course, with a substantial proportion of patients having poor outcome in spite of the use of the best currently available treatments. Thus, the development of novel strategies to improve outcome, even for a modest number of patients, will be of great benefit.
Many theories abound conceptualising the pathophysiology of schizophrenia, including neuronal maldevelopment, impaired neurotransmission, viral infections, autoimmune dysfunction and others. Further, there are a broad variety of apparently disparate biological findings in schizophrenia,[1] possibly due to aetiological heterogeneity; however, it is possible that a final common pathogenetic pathway(s) may mediate the recognisable syndromes of schizophrenia. The scientific challenge is to identify the pathological process(es) that may account for the diverse clinical and biological features of schizophrenia.
The neuronal membrane can serve as a point of convergence for the above-mentioned theoretical models. The neuronal membrane is the structural and functional site of neurotransmitter receptors, ion channels, signal transduction and drug effects. The membrane is also a point where there is a natural intersection between genetic and environmental factors.[2] Membrane defects, such as those induced by decreased amounts of polyunsaturated fatty acids (PUFAs) in phospholipids,[3,4] can significantly alter a broad range of membrane functions, and presumably modify behaviour through multiple ‘downstream’ biological effects.
Previous results from our laboratories[5–7] and those of other investigators[8–11] show decreased levels of PUFAs in both peripheral and central membranes of individuals with schizophrenia. It is well known that oxidative stress can lead to the type of membrane abnormalities that have been observed in schizophrenia.[12] Thus, free radical-mediated pathological processes may provide an explanatory model for the specific membrane alterations in schizophrenia, indicating that a role for oxidative stress might exist for some domains of schizophrenic pathophysiology as has been proposed by several investigators.[12–16] This review details the intimate connections between oxidative stress and membrane pathology, with findings specific to schizophrenia.
1. Free Radicals and the Antioxidant Defence System (AODS)
1.1 Free Radicals and Membrane Perturbations
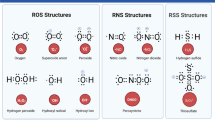
Free radicals are generated in vivo during many normal biochemical reactions involving oxygen and nitric oxide (fig. 1), including the mitochondrial electron transfer chain, NADPH-dependent oxidation, and auto-oxidation of PUFAs and catecholamines.[17–19] These free radicals (primarily the reactive oxygen species, superoxide and hydroxy radicals) play an important role in membrane lipid peroxidation.
Production of reactive oxygen species and the defence mechanism against damage by reactive oxygen. Superoxide dismutase (SOD) catalyses the conversion of superoxide radicals (O2 -) to hydrogen peroxide. Catalase (CAT) and glutathione peroxidase (GSH-Px) convert hydrogen peroxide to water. Glutathione (GSH) is utilised by GSH-Px to yield the oxidised form of glutathione (GSSG), which is converted back to GSH by glutathione reductase (GR). Hydrogen peroxide is susceptible to auto-oxidation to form hydroxyl radicals (OH), particularly in the presence of metal catalysts such as iron. In addition, nitric oxide, which is the product of a 5-electron oxidation of the amino acid L-arginine, can also produce hydroxyl radicals as well as nitrogen dioxide radical (NO2).
PUFAs are major components of membrane phospholipids. They are highly susceptible to free radical insult and auto-oxidation, which result in the formation of peroxyradicals and lipid peroxide intermediates. The existence of these products within cell membranes results in an unstable membrane structure, altered membrane fluidity and permeability, and impaired signal transduction.[20] Hydroperoxides can further decompose to other toxic species (aldehydes, including malonyldialdehyde), which can damage adjacent cells, membrane-bound enzymes and receptors, cause cross-linking between various types of molecules, and result in membrane breakdown, cytotoxicity, mutagenicity and enzyme modification.[20–22] Aldehydes can react with lipids and proteins forming lipofuscin, which accumulates in neuronal cells, particularly in regions of active free radical metabolism.
Thus, the unchecked effects of free radicals can result in cellular dysfunction, loss of membrane integrity and even cell death. The brain, which is rich in PUFAs, is particularly vulnerable to free radical-mediated damage.
1.2 The AODS and the Pathological Significance of Its Disruption
Under physiological conditions the potential for free radical-mediated damage is kept in check by the antioxidant defence system (AODS), which is comprised of a series of enzymatic and non-enzymatic components.
The critical antioxidant enzymes include superoxide dismutase (SOD; E.C.1.15.1.6), catalase (CAT; E.C.1.11.1.6) and glutathione (GSH) peroxidase (GSH-Px; E.C.1.11.1.9). These enzymes act cooperatively at different sites in the metabolic pathway of free radicals (see fig. 1). Hydrogen peroxide produced by SOD is decomposed to water and oxygen by the haeme protein CAT, thereby preventing the formation of hydroxy radicals. Failure of this first line of antioxidant defence may lead to an initiation of lipid peroxidation. Selenium-dependent GSH-Px protects against lipid peroxidation by converting hydrogen peroxide to water, or more critically by converting toxic hydroperoxides to less toxic alcohols. Since SOD, CAT and GSH-Px are critical to different stages of free radical metabolism, altered activity of one enzyme without compensatory changes in the other enzymes may leave the membranes vulnerable to damage. Thus, the differential patterning of the antioxidant enzyme activities may provide important clues to the pathogenetic mechanisms of abnormal free radical metabolism.[23]
The non-enzymatic antioxidant components that may be equally important in the overall AODS consist of molecules that react with activated oxygen species and thereby prevent the propagation of free radical chain reactions. The most common non-enzymatic antioxidant molecules are albumin, uric acid, bilirubin, GSH, α-tocopherol (vitamin E), ascorbic acid (vitamin C) and β-carotene.
Oxidative stress is a state when there is a disequilibrium between pro-oxidant processes and the AODS in favour of the former. There are potentially multiple pathological consequences of increased oxidative stress, due either to increased free radical production and/or inefficient antioxidant systems (e.g. increased SOD and/or decreased CAT activity), which lead to lipid peroxidation.
Such changes in free radical metabolism have implications for the pathophysiology of schizophrenia:
-
lipid peroxidation can alter the PUFA content of cell membranes, and such changes in PUFA have been reported in patients with schizophrenia[5–7,9,10,24]
-
high levels of hydrogen peroxide and lipid peroxides lead to decreased synthesis of prostaglandins,[25] which also has been reported in patients with schizophrenia[26,27]
-
lipid peroxidation is associated with increased dopamine and decreased γ-aminobutyric acid (GABA) uptake by synaptosomes[28]
-
hydroxyl radicals decrease synaptic efficiency and impair action potential generation in hippocampal pyramidal cells[29]
-
hydrogen peroxide can inhibit dopamine β-hydroxylase[30]
-
free radicals may decrease brain GABA receptor function.[31]
It has been suggested that increased scavenging activity of SOD in the absence of increased superoxide production can depress free radical-dependent reactions, such as those catalysed by oxygenases,[32] thus resulting in decreased catecholamine production.
2. Evidence for Alterations in the AODS in Schizophrenia
Free radicals are involved in brain membrane pathology,[33] and may play an important role in neuropsychiatric disorders.
Hoffer et al.[34] first proposed a role for toxic radicals in the aetiology of schizophrenia in 1954. The first study evaluating any indices of the AODS, however, was reported over 20 years later.[35] Subsequent studies have generally examined indirect measures of free radical activity, since direct measures of free radicals in vivo are difficult and cumbersome. The majority of studies have examined the levels of nonenzymatic antioxidants as well as activities of key antioxidant enzymes in plasma and red blood cells (RBCs), based on the notion that changes in the levels of antioxidant molecules and enzyme activities may lead to oxidative stress. A few studies, however, have provided more direct evidence of oxidative membrane damage by examining levels of lipid peroxidation products. A brief review of recent findings in schizophrenia is presented in sections 2.1. to 2.3.
2.1 Non-Enzymatic Antioxidant Molecules
2.1.1 Plasma Antioxidant Capacity
A major contribution to the total antioxidant capacity comes from antioxidant molecules in plasma. The relative contribution of each plasma antioxidant in vivo depends not only the its efficacy but also its level in biological fluid. It is known that albumin, uric acid and ascorbic acid are the major contributors (>85%) to the total antioxidant capacity in human plasma.[36,37] This predominance is largely due to their high levels relative to those of other antioxidants in blood, e.g. bilirubin, α-tocopherol and β-carotene. Although individual antioxidants play a specific role in the AODS, these antioxidants may act cooperatively in vivo to provide synergistic protection to the organs against oxidative damage. Therefore, it is more meaningful to evaluate the AODS by measuring both the individual levels of antioxidants and the overall antioxidant status.
We have recently established procedures to monitor plasma total antioxidant status (TAS). TAS and levels of individual antioxidants (i.e. albumin, bilirubin and uric acid) were measured in plasma of male patients with schizophrenia, using a within-patient, repeated measures, on-off haloperidol treatment design. Plasma TAS was significantly lower in drug-free and haloperidol-treated patients with schizophrenia compared with that in healthy controls (table I).[38] Plasma TAS in patients was not significantly correlated with smoking status, as assessed by plasma levels of cotinine (the major metabolite of nicotine). Individual plasma levels of antioxidants (albumin, bilirubin[39] and uric acid[40]) were also significantly lower in patients with schizophrenia than in healthy controls (table I). In summary, the observed decreases in plasma TAS, as well as levels of individual antioxidants, in patients with schizophrenia suggest an increased risk for oxidative damage, and lend additional support to our hypothesis that oxidative stress may have a pathophysiological role in schizophrenia.
2.1.2 Water- and Fat-Soluble Vitamins
Ascorbic acid is a biological antioxidant that acts as a chain-breaking scavenger for peroxy radicals and acts synergistically with α-tocopherol. In the brain, the basal ganglia, which is rich in dopamine and glutamate, contains high levels of ascorbic acid, ranking among the tissues with the highest levels of this vitamin.[41] Therefore, the antioxidant function of ascorbic acid may serve as an essential defence line against dopamine- or glutamate-induced neurodegenerative processes.[42]
Previous studies of the levels of ascorbic acid in patients with schizophrenia have reported variable results. More recently, in a carefully controlled study, Suboticanec et al.[43] demonstrated that both plasma and urinary ascorbic acid levels were decreased in patients with chronic schizophreniaFootnote 1 relative to healthy individuals, even after controlling for diet. After ascorbic acid supplementation for 1 month, group differences were no longer significant. It is thus suggested that ascorbic acid requirements for patients with schizophrenia may be higher than for healthy individuals.
It is well known that the fat-soluble vitamin α-tocopherol is involved in oxidative metabolism and associated with diseases linked to oxidative stress. McCreadie et al.[44] found lower ratios of α-tocopherol to cholesterol in patients with schizophrenia compared with that in healthy controls. Specifically, inpatients who were most seriously ill had the lowest ratios. More recently, Brown et al.[45] also reported decreased lipid-corrected α-tocopherol levels in patients with schizophrenia who had antipsychotic-induced tardive dyskinesia (TD), relative to healthy controls, but not in antipsychotic-treated patients without dyskinesia.
2.2 Scavenging Antioxidant Enzymes
Among the 3 key scavenging antioxidant enzymes, SOD has been the most frequently studied. Increased SOD activity has been reported in the RBCs of patients with schizophrenia by some investigators[35,46–50] but not by others.[51] Antipsychotic-naïve patients with first-episode schizophreniform disorder and schizophrenia show both increased[52] and decreased[53] SOD activity. In the latter study,[53] the mean duration of illness was 4.46 days, much shorter than in other studies, which may account for the discordant findings. It is possible that with progression of the illness, the SOD levels rise as a compensatory response to oxidative stress.[53]
In contrast, blood GSH-Px activity was found to be lower, relative to healthy control individuals, in antipsychotic-treated patients with chronic schizophrenia,[54] in drug-free female patients with schizophrenia[46] and in antipsychotic-naïve psychotic children.[55] Zhang et al.[56] have recently reported increased plasma GSH-Px activity in long term antipsychotic-free as well as antipsychotic-naïve patients with schizophrenia. However, our data demonstrated a significant and positive correlation between plasma GSH-Px activity and psychosis severity.[57] Furthermore, no differences were found in GSH-Px levels of skin fibroblasts from patients with schizophrenia and healthy control individuals,[56] suggesting that plasma GSH-Px elevation in patients may be a state-dependent change and not a consequence of the course of illness or treatment with antipsychotics. In platelets, Buckman et al.[58] found that GSH-Px activity was inversely correlated with computed tomographic (CT) scan measures of brain atrophy in patients with chronic schizophrenia, specifically in those with nonparanoid schizophrenia with a predominance of negative symptoms.
Although the above studies show abnormalities in individual antioxidant enzymes, the physiology of the AODS suggests that examining a single enzyme may have limited value for elucidating the role of abnormal free radical metabolism in disease processes.[49] Since SOD, CAT and GSH-Px are critical to different stages of free radical metabolism, as mentioned in section 1.2, alteration in the activity of one enzyme without compensatory changes in other enzymes may leave membranes vulnerable to damage. Thus, the differential patterning of the antioxidant enzyme activities may provide important clues to the pathogenetic mechanisms of abnormal free radical metabolism.[49]
Recently, we evaluated the 3 critical enzymes of the AODS (SOD, CAT and GSH-Px) in patients with schizophrenia using a within-subject, repeated measures, on-off haloperidol treatment design.[50,57] Among these enzymes in erythrocytes and plasma, only RBC SOD was found to be significantly higher in drug-free patients with schizophrenia than in age- and sex-matched healthy volunteers (table II). Our finding is consistent with most previous reports of increased SOD activity in erythrocytes of drug-free and drug-treated patients with schizophrenia.[12] On the other hand, the AODS enzyme activities were not significantly correlated with age, or the age at onset or the duration of illness. In addition, none of the major AODS enzymes showed significant differences between relapsed and clinically stable patients.
2.3 Factors Influencing Antioxidant Status
2.3.1 Age
Free radical production and the AODS are thought to play important roles in the mechanism of aging.[59] For example, SOD activity in the brain increases significantly with aging,[60–62] reflecting a self-protection mechanism against an increased production of superoxide radicals in the brain. On the other hand, a decreased level of GSH is found in the brain with aging, suggesting an increased susceptibility to oxidative damage with accelerated aging.[63] Therefore, the rate of membrane oxidative damage by free radicals may provide an index to determine life span.[64–67] Antioxidant supplementation has been used to prolong life span in experimental animals.[68–70] It is thus surmised that the aging process may be simply the sum of the deleterious radical reactions going on continuously throughout life in cells and tissue.[71]
We have previously examined the relationship between plasma antioxidant protein levels and clinically relevant demographic features, e.g. age, age at onset of illness, duration of illness, days on drug treatment, duration of drug-free period, and body mass index (BMI), in individuals with schizophrenia.[39] The reduction of plasma antioxidant protein levels in schizophrenia appears to be age-related. In healthy volunteers, plasma albumin levels were not correlated with age. By contrast, there was an inverse correlation between this parameter and age in patients with schizophrenia, although there was no significant difference in mean age between the two groups. A similar inverse correlation with age is seen in plasma uric acid levels in patients with schizophrenia.[40] Plasma total bilirubin levels were positively correlated with age in healthy volunteers, whereas an age-related decrease was observed in patients with schizophrenia. Furthermore, these reductions in plasma albumin and bilirubin level were not related to other clinically relevant demographic factors, particularly duration of illness.
Taken together, age appears to be an important factor modifying the AODS in schizophrenia.
2.3.2 Antipsychotic Treatment
It is not known whether antipsychotics have a similar effect on antioxidant enzyme activities in patients with first-episode and in those with chronic schizophrenia. It is interesting that low erythrocyte SOD activity has been found in never-treated patients experiencing first-episode psychosis and was associated with impaired premorbid school functioning.[72] Follow-up studies in this patient population could provide more robust evidence of antipsychotic effects. Further, the effects of conventional and atypical antipsychotic drugs, such as clozapine, olanzapine and risperidone, should be assessed as they may differ.
The controlled discontinuation of haloperidol in patients allows the examination of the effects of the drug on biochemical indices involved in the AODS. Recently, we have compared levels of antioxidants and AODS enzymes in patients during haloperidol treatment and during withdrawal from the drug.[38–40,50,57] In plasma, neither TAS nor individual antioxidant levels were significantly affected by haloperidol withdrawal or treatment. Moreover, the duration of the drug free period had no effect. Although a clearer picture of treatment effects can be best provided by prospective treatment studies of drug-naïve patients, the decreased plasma antioxidant capacity seen in chronic schizophrenia does not appear to be due to the effects of antipsychotics.
In erythrocytes, among the 3 major AODS enzymes, GSH-Px activities were shown to be significantly higher in patients undergoing haloperidol withdrawal compared with haloperidol-treated patients (table II). In addition, SOD activities showed a trend towards being higher. Abdalla et al.[46] found that SOD and GSH-Px activities were not significantly different between patients with schizophrenia on and off antipsychotic treatment. Buckman et al.[58] also found that platelet GSH-Px activity was not correlated with antipsychotic treatment. The above studies, however, did not utilise a within-patient repeated measures design, which is a more conservative way to address this issue.
It has been shown that haloperidol increases SOD activity in certain rat brain regions and is associated with decreases in lipid peroxidation.[73] Our findings are the opposite of that found in rat brains, suggesting that haloperidol may not have a direct regulatory effect on AODS enzymes in patients. Discontinuation of antipsychotics in patients with chronic schizophrenia is often associated with worsening of symptoms, probably a consequence of alterations in the dopaminergic system due to lifting the chronic antipsychotic-induced blockade of dopamine D2 receptors. Worsening symptoms and accompanying stress that may be due to a hyperdopaminergic state may lead to increased superoxide radical production and consequently increased SOD activity as observed in our study. The observed increase in GSH-Px activity could be compensatory, due to increased SOD-induced production of hydrogen peroxide. It is well recognised that nonspecific stress can induce oxidative stress.[74,75] Thus, alterations in SOD activity in our study may be a consequence of a changed behavioural state rather than direct effects of haloperidol, with secondary increases in GSH-Px activity.
2.3.3 Cigarette Smoking
The prevalence of cigarette smoking in patients with schizophrenia is between 70 to 90%, compared with that of 35 to 54% for all psychiatric patients and 30 to 35% for the general population.[76–78]
Cigarette smoke contains, in addition to nicotine, thousands of substances in the gas and tar phases, many of which are free radicals such as peroxyl radicals, oxides of nitrogen, hydroquinones and other radical species.[79–82] Chronic cigarette smoking is associated with oxidative stress due to the increased free radical burden, reflected in alterations of various oxidative stress indices,[83] and may contribute directly to decreases in the levels of antioxidants. In light of this, it is important to know whether cigarette smoking accounts for the AODS abnormalities seen in schizophrenia.
We examined plasma cotinine levels as an index of smoking. In our studies,[38–40] it appears that cigarette smoking does not significantly reduce the levels of plasma TAS and individual antioxidants, with the exception of bilirubin. Although bilirubin is a more efficient antioxidant than albumin, the overall contribution to plasma TAS from bilirubin is <5%, as compared with >40% from albumin.[37] Therefore, cigarette smoking plays an insignificant role in altering levels of plasma antioxidant proteins.
We further examined whether tobacco smoking may affect AODS enzyme activities via smoking-induced oxidative stress. In healthy control individuals, there was an inverse relationship between erythrocyte SOD activity and plasma cotinine levels.[50] In patients, there was no such relationship when patients were medicated with haloperidol. Thus, smoking cannot account for an increased erythrocyte SOD activity in patients with schizophrenia after haloperidol discontinuation, although we are unable to account for the absence of the relationship between cotinine level and SOD activity in patients.
2.3.4 Diet
Diet and alcohol are the major factors affecting both the antioxidant system and the production of free radicals and reactive oxygen-containing species.[84] Plasma levels of antioxidants of dietary origin (e.g. tocopherols, ascorbic acid, carotenoids, etc.) are influenced directly by nutritional supplements as well as food and alcohol consumption.[85–87]
Among the major plasma antioxidants, only uric acid levels were correlated significantly and positively with BMI in patients with schizophrenia (both on and off haloperidol treatment); no such correlation was seen in healthy volunteers.[40] No patients were receiving any uricosuric agents that potentially could decrease uric acid levels. If anything, they were at greater risk for increased uric acid levels because of their higher BMI. In our studies,[38–40,50,57] all patients were hospitalised and maintained on a control balanced diet without alcohol consumption. Therefore, it is unlikely that their decreased plasma antioxidant status resulted from a dietary deficiency or alcohol consumption as compared with their healthy volunteer counterparts.
3. Free Radical Pathology in Schizophrenia
3.1 Lipid Peroxidation
Changes in the AODS do not necessarily reflect increased oxidative stress and subsequent membrane lipid damage. While evidence of peroxidative damage is limited in patients with schizophrenia, the findings have been consistent. Increased blood levels of malondialdehyde[44,88,88–91] and a similar increase in the levels of lipid peroxides in CSF samples[92,93] have been reported. More recently, Mahadik et al.[94] reported elevated plasma lipid peroxides levels at the onset of psychosis in never-medicated, first-episode patients. Their findings, if replicated, may indicate the presence of oxidative stress very early in the course of illness, and independent of treatment.
Increased levels of pentane, another marker of lipid peroxidation, have also been reported in patients with schizophrenia relative to control individuals.[95,96] We are currently examining the issue of whether such a reduction of antioxidant level is associated with increased levels of lipid peroxides in plasma and postmortem brain tissues of patients who had had schizophrenia.
3.2 Mitochondria
Mitochondria process most of the cellular oxygen to provide energy that drives almost all metabolic processes, and also are the site of significant free radical production. About 3% of all oxygen consumed is converted to superoxide, and subsequently to hydrogen peroxide.[97] Thus, there is an enormous and continuous free radical burden associated with mitochondria. Antioxidant systems keep this in check. When the equilibrium between pro-oxidant and antioxidant systems are disturbed in favour of the former, mitochondrial damage can occur. Mitochondrial membranes, similar to neuronal membranes, are vulnerable to lipid peroxidation. Any impairment in mitochondrial oxidative phosphorylation can lead to a broad range of cellular disturbances, including decreased neurotransmission, decreased DNA repair and finally cell death.
Cytochrome-c oxidase (COX, Complex IV) is a key enzyme in the mitochondrial electron transport chain. Decreased activity of this enzyme has been reported in the frontal cortex and caudate nucleus of patients with schizophrenia.[98] Interestingly, COX activity is maintained by cardiolipin, a key lipid in the mitochondrial membrane, which is highly susceptible to peroxidation during oxidative stress.[99] Several lines of evidence suggest decreased oxidative metabolism in some brain areas in schizophrenia,[100] and may be explained in part by mitochondrial dysfunction. We propose that this is secondary to oxidative stress due either to decreased antioxidant capacity and/or increased free radical burden.
3.3 Findings in the Brain
There have been only 2 reports of postmortem studies involving investigation of SOD activity in schizophrenia. An early study by Wise et al.[101] found no difference in the diencephalons (thalamus, and epi-, sub- and hypothalamus) between patients with schizophrenia and control individuals. The recent study by Loven et al.,[102] however, demonstrated an increased activity of manganese-SOD, a key antioxidant enzyme in mitochondria, in the temporal and frontal cortices of antipsychotic-treated patients who had had schizophrenia.
Several electron microscopic studies of brains from patients with schizophrenia have found large amounts of lipofuscin-like material in oligodendrocytes,[103] abnormal pigment-laden neurons,[104] and axonal deposits of lipofuscin-like bodies.[105] As noted in section 1.1, lipofuscin is a by-product of lipid peroxidation. While some studies have reported gliosis, a potential response to neuronal loss, in patients with schizophrenia, others have not.[106] A general assumption but possible misconception is that cell death is a necessary consequence of oxidative stress. Nevertheless, these microscopic findings may provide indirect evidence of oxidative stress in schizophrenia.
3.4 Tardive Dyskinesia
There is general consensus that antipsychotics are a necessary, but not sufficient, factor for the development of TD,[107] although some have argued that TD is integral to the schizophrenic disease processes.[108] Free radical-mediated pathology has been implicated in the development of TD.[12,109,110]
Lohr et al.[93] found a significantly higher level of lipid peroxidation products (diene conjugates) in the CSF of patients with schizophrenia who had TD compared with those who did not have TD. They also demonstrated that the severity of TD was positively correlated with levels of diene conjugates, and, more critically, high levels of diene conjugates were associated with subsequent development of TD. Recently, Peet et al.[90] found a highly significant correlation between plasma levels of lipid peroxidation products and dyskinesia severity. Zubenko and Cohen[111] have shown that platelet membrane fluidity is altered in patients with TD, but not in similarly antipsychotic-treated patients without TD. In addition, treatment with α-tocopherol reduces the severity of TD in patients with schizophrenia.[112,113] If indeed free radicals play a role in the development of TD, patients with an inadequate AODS would be more likely to develop TD.
4. Evidence for Altered Membrane Dynamics in Schizophrenia
4.1 Phospholipids and Polyunsaturated Fatty Acids in Peripheral Tissues
Early findings, comprehensively reviewed by Rotrosen and Wolkin,[26] demonstrated variable alterations in the levels of phosphatidylcholine (PC), phosphatidylserine (PS) and phosphatidylinositol (PI), and consistent decreases of phosphatidylethanolamine (PE) levels in RBC membranes, in psychotic patients. These results are diverse and inconsistent due primarily to differences in patient groups and methodology. Such discrepancies, however, may be explained in part by the more recent finding of a bimodal distribution of RBC PUFAs in patients, in contrast to the unimodal distribution seen in healthy controls.[10] Phospholipid abnormalities have also been found in medication-free patients with schizophrenia,[114] and decreases in all 4 key membrane phospholipids were found in fibroblasts from antipsychotic-naïve patients with schizophrenia.[115] These latter findings suggest that phospholipid and fatty acid abnormalities may be disease-related. At the very least, they are present early in the course of illness, prior to the initiation of treatment.
Significant decreases in the levels of both arachidonic acid [AA; 20:4(n-6)] and linoleic acid [18:2(n-6); a precursor of AA], but an increase in total n-3 fatty acid levels, in the plasma of patients with schizophrenia who were from England, Scotland and Ireland has been reported.[116] Such decreases in the levels of PUFAs were also demonstrated in RBC membranes of patients with schizophrenia.[5,8,10,117] Moreover, the decreases of RBC PUFA levels were independent of haloperidol treatment.[6] There are initial data from fibroblasts of drug-naïve patients with first-episode schizophrenia of decreased AA levels.[118]
Studies to date, however, have focused on patients with chronic illness and poor outcome. Since outcome is often determined early, critical questions are whether low PUFA levels are seen early in the course of illness and are associated with later poor outcome. To address this issue, a longitudinal study of patients with first-episode schizophrenia was initiated. Preliminary findings from 17 patients and 15 control individuals showed a significant reduction in RBC AA levels at the antipsychotic-naïve baseline.[119] Moreover, a significant correlation was demonstrated between RBC phospholipid PUFA levels and 31P magnetic resonance spectroscopic (MRS) measures (for details see section 4.2.1) of phospholipid metabolites in the combined right and left frontal lobe, but not other brain regions including caudate, occipital, parietal and temporal areas.[120] Specifically, both total and individual PUFA (20:4, 22:5 and 22:6) levels were significantly (p < 0.02) and positively correlated with phosphomonoesters levels. These findings further support the notion that decreased membrane fatty acid levels in peripheral tissue may be associated with similar changes in the brain.
In light of findings of membrane defects in a variety of peripheral cell types (platelets, RBC and fibroblasts), it has been proposed that in schizophrenia, membrane compositional defects may occur in all cell membranes in the body, and are thus detectable in both extra-neural tissues and the brain.[121]
4.2 Membrane Lipids in the Brain
4.2.1 In Vivo Magnetic Resonance Spectroscopic Measurements
Using 31P-MRS, Pettegrew et al.[122,123] demonstrated significantly reduced levels of phosphomonoesters (phospholipid precursors) and significantly increased levels of phosphodiesters (phospholipid breakdown products) in the frontal cortices of antipsychotic-naïve first-episode patients as compared with controls. In addition, increased ATP and decreased inorganic orthophosphate levels were also found in the frontal cortex. The authors[123] suggested that changes in membrane phospholipids might be related to molecular changes that precede the onset of clinical symptoms and brain structural changes in schizophrenia, while changes in high energy phosphate metabolism may be state dependent.
Other groups[124–128] also reported similar findings of membrane phospholipid perturbations in both acutely and chronically ill patients. Also based on 31P-MRS findings, Keshavan et al.[129] suggested a possible familial basis for membrane phospholipid changes in schizophrenia.
Direct evidence of decreased fatty acid levels comes from postmortem studies (see section 4.2.2) of patients with schizophrenia relative to healthy control individuals, findings that may underlie the phospholipid abnormalities observed using 31P-MRS.
4.2.2 High Pressure Liquid Chromatographic Measurements in Postmortem Samples
We have recently developed a procedure to separate and quantify various major membrane phospholipids by high pressure liquid chromatography using an evaporative light scattering detector. Analysis of covariance was conducted to control for significant potential effects of the brain collection and storage variables on the group differences for the levels of membrane phospholipids in the caudate region of postmortem brain tissue.[7] The membrane phospholipid levels were significantly different between the schizophrenia and control groups (table III). Both PE and PC levels were significantly reduced in the schizophrenia group. Such changes were not associated with any of the brain collection and storage parameters, and were unlikely to be caused by the effects of antipsychotics. In addition, a small but marginally significant increase in PI level was also found in the schizophrenia group. There are no significant differences between the control groups with and without other mental disorders. The present findings lend further support to the concept of membrane phospholipid deficits in schizophrenia.
Comparison of phospholipid subclass levels in the caudate region of postmortem brain tissue in patients who had had schizophrenia and control groups with and without other psychiatric disorders (reproduced from Yao et al.,[7] with permission)
Furthermore, a robust reduction of total levels of PUFAs was found in the brains of those individuals who had had schizophrenia (table IV), relative to healthy controls and to those with other psychiatric disorders. This is consistent with the observed reduction of membrane levels of PE and PC. Specifically, the decrease of PUFA levels was largely attributable to reductions in AA and, to a lesser extent, its precursors linoleic acid and 20:2(n-6) in the caudate region. A similar decrease of AA was also found in the frontal cortex of patients who had had schizophrenia.[9] These data are in accordance with findings of plasma and RBC membrane fatty acid levels.
Thus, there is ample evidence for the existence of membrane phospholipid and fatty acid defects in early and chronic schizophrenia. As is known from membrane physiology, changes in the composition of membranes leads to a number of functional disturbances, and such pathological changes of relevance to schizophrenia are reviewed in section 5.
5. Oxidative Stress, Membrane Dysfunction and Neurotransmission
Much of the research focus in schizophrenia has been on neurotransmitter systems. Although the role of dopamine in the pathophysiology of schizophrenia remains preeminent, recent findings suggest that multiple neurotransmitter systems may be faulty. In many ways, schizophrenia can be conceptualised as being associated with ‘multi-neurotransmitter’ pathology. Whether these are primary or secondary to other pathological processes, such as oxidative stress and membrane dysfunction, will need to be determined in future studies. It is important to recognise, however, that alterations in the metabolism of several neurotransmitter systems can both contribute to, and be modified by, oxidative stress (or membrane dysfunction).
5.1 Neurotransmitters as Contributors to the Free Radical Burden
While schizophrenia is not necessarily viewed as a neurodegenerative disorder, findings from known neurodegenerative syndromes such as Parkinson’s disease can provide heuristic models applicable to schizophrenia. For example, in Parkinson’s disease the dopamine turnover rate may be a determinant in the severity of dopamine neuronal degeneration, because neurons with higher metabolic rates are subjected to greater oxidative stress from dopamine-derived free radicals, such as dopamine semiquinones. It is well known that the metabolism of dopamine yields free radicals under normal physiological conditions.[130] A number of dopamine metabolic pathways exist that lead to the generation of hydroxyl radicals (fig. 2). Dopamine is susceptible to auto-oxidation when the AODS is weak.[131] Moreover, the progressive loss of dopamine triggers an increase in turnover of the neurotransmitter in the remaining neurons and facilitates the accumulation of toxic byproducts of dopamine metabolism.
Interestingly, it has been recognised that dopamine-mediated toxicity is also mediated through the action of dopamine at N-methyl-D-aspartate (NMDA) glutamate receptors.[132–134] There is accumulating evidence that NMDA receptor-mediated excitotoxicity involves free radicals such as superoxide and nitric oxide.[135,136] In fact, antioxidants (e.g. ascorbate and α-tocopherol) protect neurons against glutamate neurotoxicity.[137,138]
Neurotransmitters, particularly glutamate, can induce other metabolic processes that increase free radical production. Activation of NMDA receptors by glutamate stimulates phospholipase A2 (PLA2) activity and results in the release of AA to act as a second messenger, which in turn can lead to the formation of free radicals.[139] Decreased availability of AA, due either to increased PLA2 activity or lipid peroxidation, can lead to impaired glutamatergic neurotransmission, which has been proposed as a pathogenetic mechanism in schizophrenia.[140] A dopamine-glutamate imbalance has also been implicated in schizophrenia.[141] Antipsychotics that block dopamine receptors may also enhance glutamatergic neurotransmission.
5.2 Membrane Dynamics and Neurotransmission
Changes in membrane dynamics can affect transmembrane processes.[142] Inhibition of transmembrane dopamine uptake by increasing the synaptic plasma membrane cholesterol to phospholipid ratio (C/PL) has previously been shown.[143] Thus, the function of the dopamine transporter receptor (DATR) is highly influenced by the lipid composition of membrane environment. Decreased DATR density has been found in cortical areas with high metabolic activity in the brains of individuals with schizophrenia.[144] Further, both n-6 and n-3 series of PUFAs may be involved in the presynaptic receptor control of dopamine release.[145]
Serotonin 5-HT2 receptors in the brain are thought to play a regulatory role in behaviour.[146] However, conflicting findings from brain imaging and autopsy studies had left unresolved the issue of whether serotonergic function is abnormal in schizophrenia. The recent development of serotonin-dopamine antagonists as antipsychotic drugs (i.e. atypical antipsychotics) that potently block 5-HT2 receptors has renewed interest in this area. Modification of 5-HT2A receptors in particular, and probably 5-HT1A and 5-HT2C and possibly 5-HT6, 5-HT7 and 5-HT3 receptors, may have important modulatory roles to play in schizophrenia, as the research with the next generation of antipsychotics suggests.
5-HT2 receptors stimulate the release of AA in hippocampal neurons through the activation of PLA2 that is independent of inositolphospholipid hydrolysis.[147] Thus, serotonin may potentially mediate some pathophysiological processes through receptor-stimulated AA or eicosanoids. We have recently demonstrated that drug-free patients with schizophrenia exhibited reduced physiological responsivity mediated through the platelet 5-HT2 receptor complex. This could be normalised by haloperidol treatment,[148] even though haloperidol is a less potent blocker of 5-HT2A receptors than clozapine, risperidone or olanzapine. Serotonin is also believed to have a role in excitotoxic neurotoxicity.[149] Future studies should explore further the interaction between serotonergic transmission and phospholipid metabolism.
Oxidative stress can both be caused by and lead to abnormal neurotransmitter metabolism. The subsequent membrane dysfunction can have important adverse effects on central neurotransmitter systems that contribute to development of some clinical features of schizophrenia.
6. Clinical Implications
Since the pathophysiology in schizophrenia still remains unclear, understanding of the clinical implications of oxidative stress and membrane deficits is also limited. Several intriguing associations have been demonstrated between biochemical indices and clinical assessments in schizophrenia. For example, there is evidence for both phospholipid abnormality[123] and impaired free radical metabolism[102] in the prefrontal cortex of individuals with schizophrenia, which is a key brain area implicated in the disorder. Since PUFAs are preferentially vulnerable to free radical insult, it is conceivable that membrane essential fatty acid (EFA) levels in these areas may also be decreased. Prominent negative symptoms have been associated with low levels of AA in RBCs (table V)[10] and low levels of platelet GSH-Px.[58] Positive symptoms, however, have been positively correlated with SOD activity.[52] Decreased SOD activity is associated with deteriorating premorbid school functioning[53] and TD.[150]
Comparison of red blood cell fatty acid levels in patients with schizophrenia who had predominantly negative and positive symptoms (reproduced from Glen et al.,[10] with permission)
To test whether defects in the AODS are related to the severity of psychopathology, we have systematically examined relations between the 3 key scavenging antioxidant enzymes (SOD, CAT and GSH-Px) and various psychosis rating scales in drug-free groups of patients with schizophrenia.[50,57,151] To reduce the likelihood of Type I errors, we adjusted the standard significance level (p = 0.05) using a Bonferroni correction for each biochemical index (adjusted α = 0.05/4; clinical measures = 0.013). Among the 3 major AODS enzymes, both RBC and plasma GSH-Px were found to be significantly and positively correlated with the 3-day mean Bunney-Hamburg Psychosis Rating (BHPR) scores (table VI). Such a correlation was present in patients both on and off haloperidol treatment. In addition, SOD activities were inversely and significantly correlated with the BHPR and SANS scores. We also showed a significant inverse correlation of plasma TAS to various psychopathology ratings in the patients (fig. 3). A similar inverse correlation between plasma level of uric acid (a major antioxidant in plasma) and BHPR scale was also demonstrated in patients both on and off haloperidol treatment.[40]
Relationships of plasma total antioxidant status (TAS) to (a) Brief Psychiatric Rating Scale (BPRS) score and (b) the Scale for the Assessment of Negative Symptoms (SANS) score in drug-free patients with schizophrenia(reproduced from Yao et al.,[38] with permission).
On the other hand, a positive correlation has been demonstrated between plasma bilirubin and the BPRS scores in drug-free patients with schizophrenia (table VI). As mentioned earlier (section 2.3.1), there is an increase in plasma total bilirubin level with aging.[39] Since bilirubin is one of the most efficient antioxidants in plasma,[37] such an increased production of bilirubin may reflect an increased antioxidant protection against aging-related oxidative stress. Likewise, an association between plasma total bilirubin and the psychosis severity also suggests a need for increased production of antioxidants to combat oxidative stress.
Taken together, these findings suggest that alterations in AODS enzymes and antioxidant status may be a consequence of changed symptom severity rather than a direct effect of antipsychotic drug treatment, and further suggest the possibility of therapeutic approaches using currently available treatments.[15,152,153]
7. Therapeutic Treatment
It is abundantly evident that there are abnormalities in AODSs in schizophrenia, predisposing to oxidative stress, which in turn is likely to contribute to the membrane deficits that have also been observed in individuals with the disorder. These observations offer an opportunity to develop novel adjunctive therapeutic strategies that take advantage of known and existing methods of reducing oxidative stress[154] and repairing membrane deficits.[155] The use of antioxidants in treating and preventing oxidative stress is a very active area of research, particularly for a variety of brain disorders.[156]
Oxidative stress, however, is an outcome of complex and multidetermined interconnected systems of free radical generation and antioxidant systems. There are likely to be many risk factors for oxidative stress in schizophrenia.[12,15] Exogenous risk factors, such as increased calorie intake, smoking and alcohol use are frequently seen in individuals with schizophrenia, and may offer targets for reducing oxidative stress.
7.1 Decreasing Free Radical Production
Oxidative stress can be reduced by lowering the substrates for free radical formation and by inhibiting enzyme activities that produce free radicals. There are several exogenous factors including diet, smoking, pollution, and possibly a sedentary lifestyle, which can be modified to reduce free radical formation and increase overall antioxidant capacity. Therefore, the regimens outlines in sections 7.1 to 7.3 may assist patients with schizophrenia to reduce their oxidative stress.
7.1.1 Reducing Caloric Intake
It has been shown that caloric intake is correlated with oxidative stress, particularly in the brain,[64] and high caloric intake is associated with decreased maximal longevity, presumably due to oxidative stress. By contrast, decreased caloric intake in animals is associated with increased maximal longevity, and maintenance of cognitive and motor skills for longer duration.[157] It is unclear whether total calorie intake or the origin of the calories makes a difference in the generation of free radicals. Diets high in fats and iron (e.g. from red meat) can increase free radical production. There are, however, no studies in schizophrenia that have examined caloric restriction as a means of reducing oxidative stress.
7.1.2 Cessation of Cigarette Smoking
It is well known that cigarette smoking is associated with reduced levels of plasma antioxidants of dietary origin.[80–82] One of the major compounds in the gas phase of tobacco smoke is nitric oxide. It has been suggested that nitric oxide reacts with smoke olefins to form carbon-centred radicals.[81] In addition, the tar phase contains a semiquinone radical that promotes hydrogen peroxide formation.[79] Moreover, tobacco smoke may increase free radical formation by activating neutrophils.[158]
We have demonstrated that higher levels of plasma cotinine did not account for the lower levels of plasma TAS seen in our group of patients with schizophrenia.[38] It is not clear, however, whether plasma TAS is affected by chronic tobacco consumption. Future investigations will need to determine whether plasma TAS can be improved by cessation of smoking.
7.1.3 Decreasing Alcohol Consumption
Peroxidation of membrane lipids has long been considered as a possible cause for ethanol-induced hepatic lesions.[159,160] In humans, ethanol is mainly derived from consumption of alcoholic beverages. Most of the absorbed ethanol is removed by oxidation to first acetaldehyde and then to acetate. Chronic alcohol consumption is likely to diminish the oxidising capacity of the liver. The resulting accumulation of acetaldehydes may stimulate lipid peroxidation, and subsequently cause mitochondrial damage. Thus, excessive and prolonged ethanol ingestion is associated with an increase in oxidative stress. Therefore, reducing alcohol intake could be of benefit in reducing oxidative stress in patients with schizophrenia.
7.2 Antioxidant Treatment
The use of antioxidants in schizophrenia has been almost exclusively limited to the use of α-tocopherol in the treatment of TD. α-Tocopherol is a highly efficient lipid-soluble ‘chain-breaking’ antioxidant that acts to stabilise plasma membranes. Membrane α-tocopherols are susceptible to oxidation, which converts them to tocopherolquinone. It is conceivable that such a metabolite may be increased under pathological conditions.[161] Numerous studies in animals and humans have demonstrated protective effects of α-tocopherol against a large variety of free radical-mediated pathological insults.[162] Supra-normal doses of α-tocopherol have been safely and effectively used to reduce the severity of TD. Several studies,[90,112,113,163,164] albeit with relatively small sample sizes, have reported decreases in the severity of dyskinesia with α-tocopherol treatment; however, others have not.[165–167] A shorter duration of TD was associated with better therapeutic response.
Most studies utilised a dosage range of α-tocopherol 1200 to 1600 IU/day. Peet et al.[90] found a clinically significant response with 1200 IU/day, with the therapeutic effect maintained for 7 to 13 months after discontinuation of α-tocopherol.
While most studies have been relatively brief (<12 weeks), Adler et al.[167] conducted a 36-week study that showed robust effects of α-tocopherol in reducing dyskinesia severity. By contrast, the largest (n = 158) and longest (12 months) study to date[168] has shown reductions in dyskinesia severity in both treatment and placebo groups, but there was not a significant different between the 2 groups.
In another study, significant decreases in psychiatric symptoms were also found in patients with schizophrenia following α-tocopherol treatment.[169]
The above findings, albeit indirectly, provide further evidence for a role of free radical-mediated pathology in some dimensions of schizophrenic illness and its complications. Furthermore, patients with an increased oxidative stress are more likely to develop TD, possibly antipsychotic-induced.
7.3 Essential Fatty Acid Supplementation
There is evidence of an improvement in clinical state, albeit modest, with EFA supplementation in schizophrenia.[152] This strategy is based on the fairly consistent evidence that there are a variety of membrane deficits in schizophrenia.[3,4,121]
It is not known whether patients with schizophrenia in Western countries have a low dietary intake of EFAs. There is an indication, however, that outcome is better in so-called underdeveloped countries (where the diet is characterised by a high intake of EFAs) than in Western countries.[170] Moreover, a highly significant correlation was found between low dietary fat intake and good schizophrenia outcome.[171] Thus, there is a close correlation between a good lifetime outcome for schizophrenia and a high ratio of unsaturated to saturated fat intake.[172]
Early studies of EFA supplementation were aimed at dyskinesia, using an animal model.[173,174] Subsequently, in a double-blind, placebo-controlled study of evening primrose oil supplementation in a group of patients with psychiatric disorders (primarily schizophrenia), no significant effects on TD were seen, although a significant improvement was noted on the Weschler memory scale and psychopathology scores.[8] Similarly negative results on the antidyskinetic effect of EFA supplementation were also obtained by other investigators.[175–177] Such a failure to improve the movement disorder may have resulted from one or a combination of factors such as low dosage, inadequate duration of dietary trial, as well as chronicity and irreversibility of TD.[178]
However, a recent open-labelled study by Mellor et al.[179] has shown that dietary intake of eicosapentaenoic acid (EPA) led to significant improvements of scores on rating scales for both schizophrenic symptomatology and TD. In a separate study conducted in India,[180] an improvement of total scores on the Positive and Negative Syndrome Scales (PANSS) was also noted in patients with schizophrenia following antipsychotic treatment in conjunction with EPA supplementation.
More recently, in a small double-blind trial of EFA supplementation, Peet[181] observed the greatest improvements in total PANSS, positive symptoms and general psychopathology in antipsychotic-treated patients who received EPA supplementation as compared with those given antipsychotics and a docosahexaenoic acid-enriched oil or a corn oil placebo. No significant differences in the improvement in negative symptoms were demonstrated among the 3 treatment groups. When EPA alone was used, Puri et al.[182] reported sustained remission of positive and negative symptoms in one drug-naïve patient with schizophrenia over a 1-year period. The improvement of schizophrenic symptomatology was further accompanied by a correction in RBC membrane phospholipid abnormalities.
Taken together, the above findings suggest further support for a potential therapeutic effect of EPA in the treatment of schizophrenia. Careful double-blind studies may now be indicated.
8. Conclusions
Extensive, albeit fragmentary, findings from neurochemical and neuroendocrine studies of schizophrenia have not provided conclusive evidence for any specific aetiological theory of the disorder,[1] perhaps due to aetiopathogenetic heterogeneity.[183] However, there exists a point of convergence for many of these theoretical models, one that occurs at the level of the neuronal membrane, which is the site of neurotransmitter receptors, ion channels, signal transduction and drug effects. Membrane deficits, specifically free radical-mediated, can significantly alter a broad range of membrane functions. Previous results showing altered membrane dynamics and antioxidant status in schizophrenia, and findings from other investigators (see section 2), are consistent with the notion of free radical-mediated neurotoxicity in schizophrenia.
The neurotoxicity can be a consequence of increased free radical production and/or an inadequate AODS. It is clear that there are multiple pathways that lead to excess free radical generation and subsequent oxidative stress (fig. 4). The pathological effects of oxidative stress are likely to be mediated through the well recognised effects of lipid peroxidation on neuronal and mitochondrial membrane components.[97,184]
If oxidative stress can be proven to have a key role in the pathophysiology of schizophrenia, then the next step is the development of novel, adjunctive antioxidant and membrane protective treatments. It is possible that adjunctive interventions can be ‘tailored’ to respond to specific pathological mechanisms that lead to excess free radical generation, whether it is mitochondrial dysfunction or calcium dysregulation. A major question remains about the timing and duration of such interventions. There is evidence, at least for TD, that treatment with α-tocopherol is more effective in patients with a shorter TD duration.[185] Encouragingly, the development of treatment trials with adjunctive antioxidants should be extremely short because there already exist many antioxidant compounds with relatively low known toxicity, and essential fatty acid supplementations.[12,153]
Caution, however, is indicated in utilising antioxidants in the treatment of schizophrenia in the absence of evidence for their efficacy in controlled studies, with the exception of α-tocopherol for treating TD. More importantly, antioxidants should not be utilised as a primary treatment for schizophrenia. Although there is a general assumption that administration of antioxidants is benign, this may not be necessarily true. Findings that the administration of the antioxidant β-carotene to smokers actually increased cancer rates, an effect opposite to what was expected, highlights this point.[186] Quenching free radicals in excess may have unintended consequences, since free radicals are also important in many biological reactions.[187] Therefore, future studies will need to carefully determine which antioxidants, at what dosages and in what combinations will have the greatest therapeutic benefit with the least risk.
Recently, dietary supplementation with EFAs has shown promise in ameliorating some of the clinical symptoms of schizophrenia[117,180,182] as well as cognitive impairments associated with dyslexia and attention deficit hyperactivity disorder.[188,189] Thus, investigating EFA metabolism has proved fruitful for generating and testing novel pathophysiological hypotheses and new therapeutic agents for schizophrenia.[190] A tremendous advantage to the clinical use of EPAs includes their low cost, lack of significant adverse effects and ease of use.
If indeed oxidative stress leads to membrane deficits, then there may be merit in combining membrane protective approaches, such as antioxidant and EFA supplementation. The efficacy of a combined antioxidant and EFA supplementation strategy may be greater that either approach alone. EFA supplementation alone may be sufficient to repair the loss of membrane fatty acids, but not in reducing or preventing ongoing oxidation of fatty acids by free radicals. Testing such a proposal is eminently feasible, utilising a double-blind, random-assignment approach. Further, the findings may have important ramifications in terms of preventive and early intervention approaches in populations at risk for schizophrenia, and may even shed light on the aetiological processes involved.
Notes
1In the studies referred to throughout the rest of this article, patients with chronic schizophrenia were being treated with antipsychotics, unless stated otherwise.
References
Liebennan JA, Koreen AR. Neurochemistry and neuroendocrinology of schizophrenia: a selective review. Schizophr Bull 1993; 19: 371–429
Horrobin DF, Glen AIM, Hudson CJ. Posssible relevance of phosplolipid abnormalities and genetic interactions in psychiatric disorders; the relationship between dyslexia and schizophrenia. Med Hypotheses 1995; 45: 605–13
Yao JK. Red blood cell and platelet fatty acid metabolism in schizophrenia. In: Peet M, Glen I, Horrobin DF, editors. Phospholipid spectrum disorder in psychiatry. Lancashire: Marius Press, 1999: 57–71
Yao JK, Reddy RD. Fatty acids and psychiatric disorders. In: Chow CK, editor. Fatty acid in foods and their health implications. 2nd ed. New York (NY): Marcel Dekker, 2000: 995–1012
Yao JK, van Kammen DP, Welker JA. Red blood cell membrane dynamics in schizophrenia. II. Fatty acid composition. Schizophr Res 1994; 13(3): 217–6
Yao JK, van Kammen DP, Welker JA, et al. Red blood cell membrane dynamics in schizophrenia. III Correlation of fatty acid abnormalities with clinical measures. Schizophr Res 1994; 13(3): 227–32
Yao JK, Leonard S, Reddy R. Membrane phospholipid abnormalities in portmortem brains from schizophrenic patients. Schizophr Res 2000; 42(1): 7–17
Vaddadi KS, Courtney P, Gilleard CS, et al. A double blind trial of essential fatty acid supplementation in patients with tardive dyskinesia. Psychiatry Res 1989; 27: 313–23
Horrobin DF, Manku MS, Hillman H, et al. Fatty acid levels in the brains of schizophrenics and normal controls. Biol Psychiatry 1991; 30: 795–805
Glen AIM, Glen EMT, Horrobin DF, et al. A red cell membrane abnormality in a sub-group of schizophrenic patients: Evidence for two diseases. Schizophr Res 1994; 12; 53–61
Peet M, Laugharne JDE, Rangarajan N, et al. Depleted red cell membrane essential fatty acids in drug-treated schizophrenic patients. J Psychiatr Res 1995; 29: 227–32
Reddy RD, Yao JK. Membrane protective strategies in schizophrenia: conceptual and treatment issues. In: Peet M, Glen I, Horrobin DF, editors. Phospholipid spectrum disorder in psychiatry. Lancashire UK: Marius Press, 1999: 75–88
Cadet JL, Lohr JB. Free radicals and the developmental pathology of schizophrenic burnout. Integr Psychiatry 1987; 5: 40–8
Lohr JB. Oxygen radicals and neuropsychiatric illness: some speculations. Arch Gen Psychiatry 1991; 48: 1097–6
Mahadik SP, Mukherjee S. Free radical pathology and anti-oxidant defense in schizophrenia: a review. Schizophr Res 1996; 19: 1–17
Smythies J. Oxidative reactions and schizophrenia: a review-discussion. Schizophr Res 1997; 24(3): 357–64
Halliwell B, Gutteridge JMC. Oxygen radicals and the central nervous system. Trends Neurosci 1984; 8: 22–66
Kalyanaraman B. Free radicals from catecholamine hormones, neuromelanins, and neurotoxins. In: Miquel J, Quintanilha AT, Weber H, editors. Handbook of free 107 radicals and antioxidants in biomedicine. Vol. I. Boca Raton: CRC Press, 1989: 147–59
Rice-Evans CA. Formation of free radicals and mechanisms of action in normal biochemical processes and pathological states. In: Rice-Evans CA, Burdon RH, editors. Free radical damage and its control. Amsterdam: Elsevier, 1994: 131–53
Ernster L. Lipid peroxidation in biological membranes mechanisms and implications. In: Yagi K, editor. Active oxygens, lipid peroxides, and antioxidants. Tokyo: CRC Press 1993: 11–38
Chan PH, Fishman RA. Transient formation of superoxide radicals in polyunsaturated fatty acid-induced brain swelling. J Neurochem 1980; 35: 1004–7
Chan PH, Fishman RA, Longar S, et al. Cellular and molecular effects of polyunsaturated fatty acids in brain ischemia and injury. Prog Brain Res 1985; 63: 227–35
Smith C. Free radical mechanisms of tissue injury. In: Moslen M, Smith C, editors. Free radical mechanisms of tissue injury. Boca Raton: CRC Press, 1992; 1: 1–22
Kaiya H, Horrobin DF, Manku MS, et al. Essential and other fatty acids in plasma in schizophrenic and normal individual from Japan. Biol Psychiatry 1991; 30: 357–62
Deby C, Deby-Dupont G. Oxygen species in prostaglandin biosynthesis in vitro and in vivo. In: Bannister WH, Bannister JV, editors. Development in biochemistry. Vol. 11B: biological and clinical aspects of superoxide and superoxide dismutase. New York (NY): Elsevier, 1980: 84–97
Rotrosen J, Wolkin A. Phospholipid and prostaglandin hypotheses of schizophrenia. In: Meltzer NY, editor. Psychopharmacology: the third generation on progress. New York (NY): Raven Press, 1987: 759–64
van Kammen DP, Yao JK, Goetz K. Polyunsaturated fatty acids, prostaglandins, and schizophrenia. Ann N Y Acad Sci 1989; 559: 411–23
Rafalowska U, Liu GJ, Floyd RA. Peroxidation induced changes in synaptosomal transport of dopamine and g-aminobutyric acid. Free Radic Biol Med 1989; 6: 485–92
Pellmar T. Electrophysiological correlates of peroxide damage in guinea pig hippocampus in vitro. Brain Res 1986; 364: 377–81
Levin EY, Kaufman S. Studies on the enzyme catalyzing the conversion of 3,4-dihydroxyphenylethylamine to norepinephrine. J Biol Chem 1961; 236: 2043–4
Schwartz RD, Skolnick P, Paul SM. Regulation of gamma-aminobutyric acid/barbiturate receptor-gated chloride ion flux in brain vesicles by phospholipase A2: possible role of oxygen radicals. J Neurochem 1988; 50: 565–71
Bartosz G. Free radicals and the developmental pathology of schizophrenic burnout [commentary]. Integr Psychiatry 1987 5: 43–4
Halliwell B. Reactive oxygen species and the central nervous system. J Neurochem 1992; 59: 1609–23
Hoffer A, Osmond H, Smythies J. Schizophrenia: a new approach. J Ment Sci 1954; 100: 29–5
Michelson AM, Puget K, Durosay P, et al. Clinical aspects of the dosage of erythrocuprein. In: Michelson AM, McCord JM, Fridovich I, editors. Superoxide and superoxide dismutase. London: Academic Press, 1977; 467–99
Wayner DDM, Burton GW, Ingold KU, et al. The relative contribution of vitamine E, urate, ascorbate and proteins to the total peroxyl radical-trapping antioxidant activity of human blood plasma. Biochim Biophys Acta 1987; 924: 408–19
Miller NJ, Rice-Evans C, Davies MJ, et al. A novel method for measuring antioxidant capacity and its application to monitoring the antioxidant status in premature neonates. Clin Sci 1993; 84: 407–12
Yao JK, Reddy R, McElhinny LG, et al. Reduced status of plasma total antioxidant capacity in schizophrenia. Schizophr Res 1998; 32: 1–8
Yao JK, Reddy RD, van Kammen DP. Abnormal age-related changes of plasma antioxidant proteins in schizophrenia. Psychiatry Res 2000; 97: 137–51
Yao JK, Reddy R, van Kammen DP. Reduced level of plasma antioxidant uric acid in schizophrenia. Psychiatry Res 1998; 80: 29–39
Oke AF, May L, Adams RN. Ascorbic acid distribution patterns in human brain. In: Burns JJ, Rivers JM, Machlin LJ, editors. Third Conference on Vitamin C. Ann N Y Acad Sci 1987; 498: 1–12
Rebec GV. Ascorbate: an antioxidant neuroprotectant and extracellular neuromodulator. In: Connor JR, editor. Metals and oxidative damage in neurological disorders. New York (NY): Plenum Press, 1997: 149–73
Suboticanec K, Folnegovic-Smalc V, Korbar M, et al. Vitamin C status in chronic schizophrenia. Biol Psychiatry 1990; 28: 959–66
McCreadie RG, MacDonald E, Wiles D, et al. The nithsdale schizophrenia surveys. XIV: plasma lipid peroxide and serum vitamin E levels in patients with and without tardive dyskinesia, and normal subjects. Br J Psychiatry 1995; 167: 610–17
Brown K, Reid A, White T, et al. Vitamin E, lipids, lipid peroxidation products and tardive dyskinesia. Biol Psychiatry 1998; 43: 863–67
Abdalla DSP, Manteiro HP, Olivera JAC. et al. Activities of Superoxide dismutase and glutathione peroxidase in schizophrenic and manic depressive patients. Clin Chem 1986; 32: 805–7
Golse B, Debray-Ritzen P, Puget K, et al. Dosages érythrocytaires et plaquettes des superoxyde dismutases (1 et 2) et de la glutathion peroxydase dans les pychoses infantiles de développement. Nouv Presse Med 1978; 7: 1952
Golse B, Debray Q, Puget K, et al. Dosages érythrocytaires de la superoxyde dismutases 1 et de la glutathion peroxydase dans les schizophrénies de l’adulte. Nouv Presse Med 1978; 7: 2070–1
Reddy R, Mahadik SP, Mukherjee M, et al. Enzymes of the antioxidant system in chronic schizophrenic patients. Biol Psychiatry 1991; 30: 409–12
Yao JK, Reddy R, McElhinny LG, et al. Effect of haloperidol on antioxidant defense system enzymes in schizophrenia. J Psychiatr Res 1998; 32: 385–91
Sinet PM, Debray Q, Carmagnol F, et al. Normal erythrocyte SOD values in two human diseases: schizophrenia and cystic fibrosis. In: Greenwald RA, Cohen G, editors. Oxy radicals and their scavenger systems. Vol. II. Cellular and medical aspects. New York (NY): Elsevier, 1983: 302–4
Khan NS, Das I. Oxidative stress and superoxide dismutase in schizophrenia. Biochem Soc Trans 1997; 25(3): 418S
Mukherjee S, Mahadik SP, Scheffer R, et al. Impaired antioxidant defense at the onset of psychosis. Schizophr Res 1996; 19: 19–26
Stoklasova A, Zapletalek M, Kudrnova K, et al. Glutathione peroxidase activity of blood in chronic schizophrenics [in Czech]. Sb Ved Pr Lek Fak Karlovy University Hradci Kralove Suppl. 1986; 29(1–2): 103–8
Golse B, Debray-Ritzen P, Puget K, et al. Dosages de la superoxyde dismutases 1 plaquettaire dans les pychoses infantiles de développement. Nouv Presse Med 1977; 6: 2449
Zhang ZJ, Ramchand CN, Ramchand R, et al. Glutathione peroxidase (GSHPx) activity in plasma and fibroblasts from schizophrenics and control. Biol Psychiatry 1998; 29: 103–4
Yao JK, Reddy R, van Kammen DP. Human plasma glutathione peroxidase and symptom severity in schizophrenia. Biol Psychiatry 1999; 45: 1512–5
Buckman TD, Kling AS, Eiduson S, et al. Platelet glutathione peroxidase and monoamine oxidase activity in schizophrenics with CT scan abnormalities: Relation to psychosocial variables. Psychiatry Res 1990; 31: 1–14
Harman D. Aging prospects for future increases in the functional life span. Age 1994; 17: 119–46
Vanella A, Goremia E, D’Urso G, et al. Superoxide dismutase activity in aging rat brian. Gerontology 1982; 228: 108–3
Marvilli L, Mondov B, Federicop R, et al. Superoxide dismutase activity in developing rat brian. J Neurochem 1978; 31: 363–4
Hiramastsu M, Kohno M, Edamatsu R, et al. Increased superoxide dismutase activity in aged human cerebrospinal fluid and rat brain determined by electron spin resonance spectrometry using spin trap method. J Neurochem 1992; 58: 116–4
Mori A, Packer L. Disorders of the oxidative stress defense system in senescence-accelerated mice (SAM, and prolongation of life span by supplementation with antioxidants and free radical spin traps. In: Packer L, Ong A, editors. Biological oxidants and antioixidants: molecular mechanisms and health effects. Champaign (IL): AOCS Press, 1998: 327–4
Sohal RS, Ku HH, Agarwal S, et al. Oxidative damage, mitochondrial oxidant generation and antioxidant defenses during aging and in response to food restriction in the mouse. Mech Ageing Dev 1994; 74: 121–33
Shigenaga MK, Hagen T, Ames BN. Oxidative damage and mitochondrial decay in aging. Proc Natl Acad Sci U S A 1994; 91: 10771–8
Harman D. Aging and Disease: Extending functional life span. Ann N Y Acad Sci 1996; 786: 321–6
Miquel J, Economos AC, Fleming J, et al. Mitochondrial role in cell aging. Exp Gerontol 1998; 15: 575–91
Clapp NK, Satterfield LC, Bowler ND. Effects of the antioxidant butyrated hydroxyltoluence(BHT) on mortality in BALB/C mice. J Gerontol 1979; 34: 497–50
Comfort A. Effect of ethoxyquin on longevity of c3h mice. Nature 1991; 229: 254–5
Heidrick ML, Heidrick LC, Cook DE. Effect of dietary 2-mercaptoethanol on the life span, immune system, tumor incidence and lipid peroxidation damage in spleen lymphocytes of aging bc3f mice. Mech Aging 1984; 27: 341–58
Harman D. The aging process. Proc Natl Acad Science U S A 1981; 78: 7124–8
Mukherjee S, Mahadik SP, Correnti EE. The antioxidant defense system at the onset of psychosis. Biol Psychiatry 1994; 35: 701
Szabo L, Lajko K, Barabas K, et al. Effects of neuroleptics on lipid peroxidation and peroxide metabolism enzyme activities in various discrete areas of the rat brain. Gen Pharmacol 1983; 14: 537–39
Ohtsuka Y, Yabunaka N, Fujisawa H, et al. Effect of thermal stress on glutathione metabolism in human erythrocytes. Euro J Appl Physiol Occup Physiol 1994; 68: 87–91
Scarpellini F, Sbracia M, Scarpellini L. Psychological stress and lipoperoxidation in miscarriage. Ann N Y Acad Sci 1994; 709: 210–3
Hughes R, Hatsukami K, Mitchell E, et al. Prevalence of smoking among psychiatric outpatients. Am J Psychiatry 1986; 143: 993–7
Lohr JB, Flynn K. Smoking and schizophrenia. Schizophr Res 1992; 8: 93–102
Olincy A, Young DA, Freedman R. Increased levels of the nicotine metabolite continine in schizophrenic smokers compared to other smokers. Biol Psychiatry 1997; 42: 1–5
Church DF, Pryor WA. Free radical chemistry of cigarette smoke and its toxicological implications. Environ Health Perpect 1985; 64: 111–26
Chow KC.Vitamin E and cigarette smoking-induced oxidative damage. In: Packer L, Fuchs J, editors. Vitamin E in health and disease. New York: Marcel Decker, 1992: 683–97
Pryor WA, Stone K. Oxidants in cigarette smoke. Ann N Y Acad Sci 1992; 686: 29
Stegmayr B, Johansson I, Huhtasaari F, et al. Use of smokeless tobacco and cigarettes: effects on plasma levels of antioxidant vitamins. Int J Vitam Nutr Res 1993; 63: 195–200
Halliwell B. Cigarette smoking and health: a radical view. J R Soc Health 1993; 113: 91–6
Papas AM. Determinants of antioxidant status in humans. Lipids 1996; 31 Suppl.: 77S–82S
Becker K, Boetticher D, Leichsenring M. Antioxidant vitamins in malnourished Nigerian children. Int J Vitam Nutr Res 1994; 64: 306–10
Parfitt VJ, Rubba P, Bolton C, et al. A comparison of antioxidant status and free radical peroxidation on plasma lipoproteins in healthy young persons from Naples and Bristol. Eur Heart J 1994; 15: 871–6
Lecomte E, Herbeth B, Pirollet P et al. Effect of alcohol consumption on blood antioxidant nutrients and oxidative stress indicators. Am J Clin Nutr 1994; 60: 255–61
Evans DR, Puczkovski PY, Brandsma MJ, et al. Elevated plasma lipid peroxides in schizophrenic patients without dementia. Biol Psychiatry 1996; 39: 588
Guliaeva NV, Levshina IP, Obidin AM. Indices of lipid free radical oxidation and the antiradical protection of the brain: the neurochemical correlates of the development of the general adaptation syndrome. Zh Vyssh Nerv Deiat Im I P Paviova 1988; 38: 731–37
Peet M, Laugharne J, Rangarajan N et al. Tardive dyskinesia, lipid peroxidation, and sustained amelioration with vitamin E treatment. Int Clin Psychopharmacol 1993; 8: 151–53
Prilipko LL. The possible role of lipid peroxidation in the pathophysiology of mental disorders. In: Packer L, Prilipko L, Christen Y, editors. Free radicals in the brain. Berlin: Spinger-Verlag, 1992: 146–52
Pall HS, Williams AC, Blake DR, et al. Evidence of enhanced lipid peroxidation in the cerebrospinal fluid of patients taking phenothiazines. Lancet 1987; II: 596–7
Lohr JB, Kuczenski R, Bracha HS, et al. Increased indices of free radical activity in the cerebrospinal fluid of patients with tardive dyskinesia. Biol Psychiatry 1990; 28: 535–9
Mahadik SP, Mukherjee S, Scheffer R, et al. Elevated plasma lipid peroxides at the onset of nonaffective psychosis. Biol Psychiatry 1998; 43: 674–79
Kovaleva ES, Orlov ON, Tsutsul’kovskia MIA, et al. Lipid peroxidation processes in patients with schizophrenia. Zh Nevropatol Psikiatr 1989; 89: 108–10
Phillips M, Sabas M, Greenberg J. Increased pentane and carbon disulfide in the breath of patients with schizophrenia. J Clin Pathol 1993; 46: 861–4
Floyd RA. Mitochondrial damage in neurodegenerative disease. In: Packer L, Hiramatsu M, Yoshikawa T, editors. Free radicals in brain physiology and disorders. San Diego (CA): Academic Press, 1996: 313–29
Cavelier L, Jazin EE, Eriksson I, et al. Decreased cytochrome-c oxidase activity and lack of age-related accumulation of mitochondrial DNA deletions in the brains of schizophrenics. Genomics 1995; 29: 217–24
Radi R, Castro L, Rodriguez M, et al. Free radical damage to mitochondria. In: Beal MF, Howell N, Bodis-Wellner I, editors. Mitochondria and free radicals in neurodegenerative diseases. New York (NY): Wiley-Liss, 1997: 57–89
Buchsbaum MS, Neuchterlein KH, Haier RJ, et al. Glucose metabolic rate in normals and schizophrenics during the continuous performance test assessed by Positron Emission Tomography. Br J Psychiatry 1990; 156: 217–7
Wise CD, Baden MM, Stein L. Post-mortem measurement of enzymes in human brain: evidence of a central noradrenergic deficit in schizophrenia. J Psychiatr Res 1974; 11: 185–98
Loven DP, James JF, Biggs L, et al. Increased manganese-superoxide dismutase activity in postmortem brain from neuroleptic-treated psychotic patients. Biol Psychiatry 1996; 40: 230–32
Miyakawa T, Sumiyoshi Deshimaru M. Electron microscopic study on schizophrenia: mechanism of pathological changes. Acta Neuropathol (Berlin) 1972; 20: 67–77
Averback P. Structural lesions of the brain of young schizophrenics. Can J Neurol Sci 1981; 8: 73–6
Senitz D, Winkelmann E. Über morphologische befunde in der orbitofrontalen rinde bei menschen mit schizophrenen psychosen. Eine golgi-und eine elektonenoptische Studie. Psychiatr Neurol Med Psychol Beih 1981; 33: 1–9
Roberts GW. Schizophrenia: A neuropathological perspective. Br J Psychiatry 1991; 158: 8–17
Kane J, Lieberman J, Woerner M, et al. Tardive dyskinesia: New Research. In: Schulz SC, Tamminga CA, editors. Schizophrenia. New York (NY): Scientific Progress Oxford University Press 1989: 381–86
Crow TJ, Cross AJ, Johnstone EC, et al. Abnormal involuntary movements in schizophrenia: Are they related to the disease process or its treatment? Are they associated with changes in the dopamine receptors? J Clin Psychopharmacol 1982; 2: 336–40
Cadet JL, Lohr JB, Jeste DV. Free radicals and tardive dyskinesia. Trends Neurosci 1986; 9: 107–8
Cadet JL, Lohr JB. Possible involvement of free radicals in neuroleptic-induced movement disorders. Evidence from treatment of tardive dyskinesia with vitamin E. Ann N Y Acad Sci 1989; 570: 176–85
Zubenko GS, Cohen BM. A cell membrane correlate of tardive dyskinesia in patients treated with phenothiazines. Psychopharmacology 1986; 88: 230–36
Lohr JB, Cadet JL, Lohr MA, et al. Alpha-tocopherol in tardive dyskinesia. Lancet 1987; I: 913–4
Elkashef AM, Ruskin PE, Bacher N, et al. Vitamin E in the treatment of tardive dyskinesia. Am J Psychiatry 1990; 147: 505–6
Keshaven MS, Mallinger AG, Pettegrew JW, et al. Erythrocyte membrane phospholipids in psychotic patients. Psychiatry Res 1993; 49: 89–95
Mahadik SP, Mukherjee S, Correnti E, et al. Distribution of plasma membrane phospholipids and cholesterol in skin fibroblasts from drug-naive patients at the onset of psychosis. Schizophr Res 1994; 13: 239–47
Horrobin DF, Manku MS, Morse-Fisher N, et al. Essential fatty acids in plasma phospholipids in schizophrenics. Biol Psychiatry 1989; 25: 562–8
Peet M, Laugharne JDE, Mellor J, et al. Essential fatty acid deficiency in eyrthrocyte membranes from chronic schizophrenic patients, and the clinical effects of dietary supplementation. Prostaglandins Leukot Essent Fatty Acids 1996; 55: 71–5
Mahadik SP, Mukherjee S, Correnti EE, et al. Plasma membrane phospholipid fatty acid composition of cultured skin fibroblasts from schizophrenic patients: comparison with bipolar and normal controls. Psychiatry Res 1996; 63: 133–42
Reddy RD, Yao JK, Zeigler M, et al. Essential fatty acids and outcome in first-episode schizophrenia. Schizophr Res 1999; 36: 108
Yao JK, Stanley JA, Reddy RD, et al. Correlations between RBC fatty acids and 31P MRS brain measures in schizophrenia. Biol Psychiatry 2000; 47: 41S
Horrobin DF. Schizophrenia as a membrane lipid disorder which is expressed throughout the body. Prostaglandins Leukotr Essent Fatty Acids 1996; 55: 3–7
Pettegrew JW, Keshavan MS, Panchalingam K, et al. Alterations in brain high energy phosphate and membrane phospholipid metabolism in first episode drug naive schizophrenics. Arch Gen Psychiatry 1991; 48: 563–8
Pettegrew JW, Keshaven MS, Minshew NJ. 31P nuclear magnetic resonance spectroscopy: Neuro-development and schizophrenia. Schizophr Bull 1993; 19: 335–53
Deicken RF, Merrin E, Calabrese G, et al. 31Phosphorus MRSI in the frontal and parietal lobes in schizophrenia. Biol Psychiatry 1993; 33: 46A
Fukazako H, Takeuchi K, Fujimoto T, et al. 31P magnetic resonance spectroscopy of schizophrenic patients with neuroleptic resistant positive and negative symptoms. Biol Psychiatry 1992;31: 204A-5A
Fukuzako H, Fukuzako T, Takeuchi K, et al. Phosphorus magnetic resonance spectroscopy in schizophrenia: correlation between membrane phospholipid metabolism in temporal lobe and positive symptoms. Prog Neuropsychopharmacol Biol Psychiatry 1996; 20: 629–40
Stanley JA, Williamson PC, Drost DJ, et al. The study of schizophrenia via in vivo 31P and 1H MRS. Schizophr Res 1993; 9: 210
Williamson P, Drost D, Stanley J, et al. Localized phosphorus 31 magnetic resonance spectroscopy in chronic schizophrenic patients and normal controls [letter]. Arch Gen Psychiatry 1991; 48: 578
Keshaven MS, Pettegrew JW, Ward R. Are membrane phospholipid changes in schizophrenia familial? Biol Psychiatry 1993; 33: 37A-162A
Cohen G. Enzymatic/nonenzymatic sources of oxyradicals and regulation of antiodant defences. Ann N Y Acad Sci 1994; 738: 8–14
Zhang F, Dryhurst G. Effects of 1-cysteine on the oxidative chemistry of dopamine: new reaction pathways of potential relevance to ideopathic Parkinson’s disease. J Med Chem 1994; 37: 1084–98
Ben-Shacher D, Zuk B, Glinka Y. Dopamine neurotoxicity: Inhibition of mitochondrial respiration. J Neurochem 1995; 64: 718–23
Cadet JL, Kahler LA. Free radical mechanisms in schizophrenia and tardive dyskinesia. Neurosci Biobehav Rev 1994; 18:457–67
Michel PP, Hefti F. Toxicity of 6-hydroxydopamine and dopamine for dopaminergic neurons in culture. J Neurosci Res 1990; 76: 428–35
Coyle JT, Puttfarcken P. Oxidative stress, glutamate, and neuro-degenerative disorders. Science 1993; 262: 689–95
Patel M, Day BJ, Crapo JD, et al. Requirement for superoxide in excitotoxic cell death. Neuron 1996; 16: 345
Ciani E, Groneng L, Voltattorni M, et al. Inhibition of free radical production or free radical scavenging protects from excitatoxic cell death mediated by glutamate in cultures of cerebellar granule cells. Brain Res 1996; 728: 1–6
MacGregor DG, Higgins MJ, Jones PA, et al. Ascorbate attenuates the systemic kainate-induced neurotoxicity in the rat hippocampus. Brain Res 1996; 727: 133–44
Iuliano L, Pedersen JZ, Pratico D, et al. Role of hydroxyl radicals in the activation of human platelets. Eur J Biochem 1994; 221: 695–704
Olney JW, Farber NB. Glutamate receptor dysfunction and schizophrenia. Arch Gen Psychiatry 1995; 52: 998–1007
Carlsson M, Carlsson A. Schizophrenia: a subcortical neuro-transmitter imbalance syndrome? Schizophr Bull 1990; 16: 425–32
Hitri A, Hurd YL, Wyatt RJ, et al. Molecular, functional and biochemical characteristics of the dopamine transporter: Regional differences and clinical relevance. Clin Neuropharm 1994; 17: 1–22
Maguire PA, Druse MJ. The influence of cholesterol on synap-tic fluidity and dopamine uptake. Brain Res Bull 1989; 22: 431–37
Hitri A, Casanova MF, Kleinman JE, et al. Age-related changes in [3H]GBR 12935 binding site density in the prefrontal cortex of controls and schizphrenics. Biol Psychiatry 1995; 37: 175–82
Davidson B, Kurstjens NP, Patton J, et al. Essential fatty acids modulate apomorphine activity at receptors in cat caudate slices. Eur J Pharmacol 1988; 149: 317–22
Leysen JE, Pauwels PJ. 5-HT2 receptors, roles and regulation. Ann N Y Acad Sci 1990; 600: 183–91
Felder CC, Kanterman RY, Ma AL, et al. Serotonin stimulates phospholipase A2 and the release of arachidonic acid in hippocampal neurons by a type 2 serotonin receptor that is independent of inositolphospholipid hydrolysis. Proc Nat Acad Sci U S A 1990; 87: 2187–91
Yao JK, van Kammen DP, Moss HB, et al. A decreased serotonergic responsivity in platelets of unmedicated patients with Schizophrenia. Psychiatry Res 1996; 63: 123–32
Ohmori T, Abekawa T, Koyama T. The role of glutamate in the behavioral and neurotoxic effects of methamphetamine. Neurochem Int 1996; 29: 301–7
Yamada K, Kanba S, Anamizu S, et al. Low superoxide dismutase activity in schizophrenic patients with tardive dyskinesia. Psychol Med 1997; 27: 1223–5
Yao JK, van Kammen DP, Reddy RD, et al. Superoxide dismutase and negative symptoms in schizophrenia. Biol Psychiatry 1998; 43: 123S-4S
Mahadik SP, Evans DR. Essential fatty acids in the treatment of schizophrenia. Drugs of Today 1997; 33: 5–17
Reddy R, Yao JK. Schizophrenia: role of oxidative stress and essential fatty acids. In: Basu, Tk, Temple NJ, Garg ML, editors. Antioxidants in human health. Wallingford, Oxon: Cab International 1999; 27: 351–66
Mahadik SP, Scheffer RE. Oxidative injury and potential use of antioxidants in schizophrenia. Prostaglandins Leukotr Essent Fatty Acids 1996; 55: 45–54
Mahadik SP, Gowda S. Antioxidants in the treatment of schizophrenia. Drugs of Today 1996; 32: 553–65
Vatassery GT. Vitamin E: Neurochemical aspects and relevance to nervous system disorders. In: Connor JR. editor. Metals and oxidative damage in neurological disorders. New York (NY): Plenum Press, 1997: 175–88
Duffy PH, Leakey JE, Pipkin JL, et al. The physiologic, neurologic and behavioral effects of caloric restriction related to aging, disease, and environmental factors. Environ Res 1997; 73: 242–8
Ryder MI, Fujitaki R, Johnson G, et al. Alterations of neutrophil oxidative burst by in vitro smoke exposure: implications for oral and systemic diseases. Ann Peridont 1998; 3: 76–87
DiLuzio NR, Hartman AD. Role of lipid peroxidation on the pathogenesis of the ethanol-induced fatty liver. Fed Proc 1967; 26: 1436
Shaw S, Javatilleke E, Ross WA, et al. Ethanol-induced lipid peroxidation: Potentiation by long-term alcohol feeding and attenuation by methionine. J Lab Clin Med 1981; 98: 417
Vatassery GT, Smith WE, Quach HT. A liquid chromatographic method for the simultaneous determination of α-tocopherol and tocopherolquinone in human red blood cells aqnd other biological samples where tocopherol is easily oxidized during sample treatment. Anal Biochem 1993; 214: 426–30
Sokol RJ. Vitamin E and neurologic function in man. Free Radie Biol Med 1989 6: 189–207
Adler LA, Peselow E, Rotrosen J, et al. Vitamin E treatment of tardive dyskinesia. Am J Psychiatry 1993; 50: 1405–7
Egan MF, Hyde TM, Albers GW, et al. Treatment of tardive dyskinesia with vitamin E. Am J Psychiatry 1992; 149: 773–7
Corrigan FM, Van Rhijn AG, Mackay AV, et al. Vitamin E treatment of tardive dyskinesia [letter]. Am J Psychiatry 1993; 150: 991–2
Shriqui CL, Bradwejn J, Annable L, et al. Vitamin E in the treatment of tardive dyskinesia: a double-blind placebo-controlled study. Am J Psychiatry 1992; 149: 391–93
Adler LA, Edson R, Lavori P, et al. Long-term treatment effects of vitamin E for tardive dyskinesia. Biol Psychiatry 1998; 43: 868–72
Lohr JB, Lavori P. Whither vitamin E and tardive dyskinesia? Biol Psychiatry 1998; 43: 861–2
Sram RJ, Binkova B. Side effects of psychotropic therapy. In: Packer L, Prilipko L, Christen Y, editors. Free radicals in the brain. Berlin: Spinger-Verlag, 1992: 153–66
World Health Organization. Schizophrenia: an international follow-up study. New York: Wiley 1979
Christensen O, Christensen E. Fat consumption and schizophrenia. Acta Psychiat Scand 1988; 78: 587–91
Horrobin DF. The relationship between schizophrenia and essential fatty acids and eicosanoid production. Prostaglandins Leukotr Essent Fatty Acids 1992; 46: 71–7
Nohria V, Vaddadi KS. Tardive dyskinesia and essential fatty acids: an animal model study. In: Horrobin DF, editor. Clinical Uses of Essential Fatty Acids. Montreal: Eden Press 1982: 199–204
Costall B, Kelly M, Naylor RJ. The antidyskinetic action of dihomo-gamma-linolenic acid in the rodent. Br J Pharmacol 1984; 83: 733–40
Bourguignon A. Trial of evening primrose oil in the treatment of schizophrenia. L’Encephale 1984; 10: 241–50
Soulairac A, Lambinet H, Neuman JC. Schizophrenia and PGs: Therapeutic effects of PG precursors in the form of evening primrose oil. Ann Med Psychol (Paris) 1983; 8: 883–90
Wolkin A, Jordan B, Peselow E, et al. Essential fatty acid supplementation in tardive dyskinesia. Am J Psychiatry 1986; 143: 912–14
Vaddadi KS. Dyskinesias and their treatment with essential fatty acids: A review. Prostaglandins, Leukotr Essent Fatty Acids 1996; 55: 89–94
Mellor JE, Laugharne JDE, Peet M. Omega-3 fatty acid supplementation in schizophrenia patients. Hum Psychopharmacol 1996; 11: 39–46
Shah S, Vankar GK, Telang SD, et al. Eicosapentaenoic acid (EPA) as an adjunct in the treatment of schizophrenia. Schizophr Res 1998; 20: 158
Peet M. New Strategies for the treatment of schizophrenia: ω-3 polyunsaturated fatty acids. In: Peet M, Glen I, Horrobin D, editors. Phospholipid spectrum disorder in psychiatry. Lancashire: Marius Press 1999: 18: 189–92
Puri BK, Richardson AJ, Horrobin DF, et al. Eicosapentaenoic acid treatment in schizophrenia associated with symptom remission, normalisation of blood fatty acids, reduced neuronal membrane phospholipid turnover and structural brain changes. Int J Clin Pract 2000; 54: 57–63
Meltzer HY. Biological studies in schizophrenia. Schizophr Bull 1987; 13: 77–111
Jesberger JA, Richardson JS. Oxygen free radicals and brain dysfunction. Int J Neurosci 1991; 57: 1–17
Elkashef AM, Wyatt RJ. Tardive dyskinesia: possible involvement of free radicals and treatment with vitamin E. Schizophr Bull 1999; 25: 731–40
The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group. N Engl J Med 1994; 330: 1029–35
Halliwell B. The antioxidant paradox. Lancet 2000; 355: 1179–80
Richardson A, Easton T, McDaid AM, et al. Essential fatty acids in dyslexia: theory, evidence and clinical trials. In: Peet M, Glen I, Horrobin DF, editors. Phospholipid spectrum disorder in psychiatry. Lancashire: Marius Press. 1999: 21: 225–41
Stordy B. Long-chain fatty acids in the management of dyslexia and dyspraxia. In: Peet M, Glen I, Horrobin DF, editors. Phospholipid Spectrum disorder in psychiatry. Lancashire: Marius Press 1999; 23: 251–60
Fenton W, Hibbeln J, Knable M. Essential fatty acids, lipid membrane abnormalities, and the diagnosis and treatment of schizophrenia. Biol Psychiatry 2000; 47: 8–21
Acknowledgements
This study was supported in part by the Office of Research and Development (Merit Review), Department of Veterans Affairs, the Highland Drive VA Pittsburgh Healthcare System, and research grants from the National Institute of Mental Health (MH43742, MH44841 and MH58141) and National Alliance for Research on Schizophrenia and Depression. The authors are grateful to L. McElhinny and C. Korbanic for their technical assistance. Appreciation is also owed to the patients and nursing staff of the Schizophrenia Research Unit for their participation and collaboration.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yao, J.K., Reddy, R.D. & van Kammen, D.P. Oxidative Damage and Schizophrenia. Mol Diag Ther 15, 287–310 (2001). https://doi.org/10.2165/00023210-200115040-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00023210-200115040-00004