Abstract
Background
For patients with ductal carcinoma in situ (DCIS), multiple national cancer organizations recommend that sentinel lymph node biopsy (SLNB) be offered when treated with mastectomy, but not when treated with breast-conserving surgery (BCS). This study analyzes national surgical trends of SLNB and axillary lymph node dissection (ALND) in DCIS patients undergoing breast surgery with the aim to quantify deviations from national guidelines.
Methods
A retrospective cohort analysis of the American College of Surgeons’ National Surgical Quality Improvement Program (ACS NSQIP) database from 2005 to 2017 identified patients with DCIS. Patients were categorized by their primary method of breast surgery, i.e. mastectomy or BCS, then further categorized by their axillary lymph node (ALN) management, i.e. no intervention, SLNB, or ALND. Data analysis was conducted via linear regression and a non-parametric Mann–Kendall test to assess a temporal trend and Sen’s slope.
Results
Overall, 43,448 patients with DCIS met the inclusion criteria: 20,504 underwent mastectomy and 22,944 underwent BCS. Analysis of DCIS patients from 2005 to 2017 revealed that ALND decreased and SLNB increased in every subgroup, regardless of surgical treatment modality. Evaluation in the mastectomy group increased overall: mastectomy alone increased from 57.1 to 65.8% (p < 0.01) and mastectomy with immediate reconstruction increased from 58.5 to 72.1% (p < 0.01). Increases also occurred in the total BCS population: partial mastectomy increased from 14.0 to 21.1% and oncoplastic surgery increased from 10.5 to 23.0% (both p < 0.01).
Conclusions
Despite national guideline recommendations for the management of ALN surgery in DCIS patients, approximately 20–30% of cases continue to not follow these guidelines. This warrants further education for surgeons and patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The axillary lymph node (ALN) basin serves as the first site of spread for many breast cancers. As such, surgical evaluation of nodes guided by ALN mapping has been a crucial component of breast cancer treatment since the early 1990s.1 ALN evaluation at the time of breast surgery has proven to decrease the rates of cancer metastasis and improve short- and long-term mortality;2,3 however, recent trends in lymph node management have favored more judicious axillary surgery, with the sentinel lymph node biopsy (SLNB) largely replacing ALN dissection (ALND) in women with invasive breast cancer.3,4,5,–6
Ductal carcinoma in situ (DCIS) is a cancerous lesion without invasion beyond the duct, therefore all lymph nodes should be negative for disease. Nonetheless, current literature reports high rates of ALN interventions in DCIS patients, with large variations in practice patterns nationwide.7,8 However, evaluation of ALNs is not without significant risk to the patient, and unnecessary surgery should be avoided when possible. Rates of postoperative hematoma, infection, lymphedema, scarring, and nerve palsy, among other complications, are not insignificant and are well reported in the literature.9,10,11,–12
Prominent national cancer organizations have consequently developed recommendations to guide surgeons’ treatment algorithms with respect to management of the breast and the axilla in cases of DCIS. In 2005, the American Society for Clinical Oncology (ASCO) first recommended that SLNB should be undertaken in cases of mastectomy but not in cases of lumpectomy. In 2014, they reiterated these guidelines.13 Similarly, the National Comprehensive Cancer Network (NCCN) does not recommend that patients with DCIS undergoing breast-conserving surgery (BCS) receive SLNB. However, the NCCN recommends that SLNB should be ‘strongly considered’ in patients with DCIS undergoing mastectomy or for those patients undergoing an “excision in an anatomic location compromising the performance of a future sentinel lymph node procedure”.14
The purpose of this study was to utilize the American College of Surgeons’ National Surgical Quality Improvement Program (ACS NSQIP) database to analyze national trends in surgical lymph node evaluation in DCIS patients. Until now, no recent literature has sought to analyze adherence trends or to quantify the deviation from these national recommendations.
Methods
Data Source
The NSQIP database was utilized to conduct a retrospective cohort analysis. This study follows similar methodology, inclusion/exclusion criteria, data collection, and surgical categorization used by Jonczyk et al., with the addition of NSQIP data from 2017.15 All participant user files were obtained and approved by the NSQIP. Our Institutional Review Board deemed this study exempt from institutional review, given the NSQIP database is a de-identified data set. The NSQIP database has grown to over 700 participating hospitals nationwide, collecting over 300 perioperative and demographic variables for patients undergoing surgery in inpatient or outpatient settings. The NSQIP collects data randomly and selectively (for rare operations), representing 10% of nationwide operations annually.15,16
Patient and Procedure Selection
Inclusion criteria for this study included women of all ages who underwent either mastectomy or BCS for DCIS. We excluded men, as well as women undergoing surgery for benign disease. We also excluded patients with invasive breast cancer. International Classification of Diseases (ICD) codes were used to stratify all patients diagnosed with DCIS from 2005 to 2017. From these records, we used current procedural terminology (CPT) codes to categorize patients into two overall groups depending on surgery modality, i.e. mastectomy or BCS. These two groups were further subcategorized based on whether or not each underwent breast reconstruction at the time of their oncologic surgery: mastectomy included women who underwent mastectomy alone as well as those who received mastectomy followed by immediate reconstruction, while BCS included women undergoing partial mastectomy alone, as well as those who received oncoplastic surgery, a form of BCS using reconstructive techniques.17,18
CPT codes were used to categorize the four patient groups based on ALN management: SLNB, ALND, or no surgical intervention on the axillary nodes. A patient was considered to have undergone ALN intervention if the primary breast surgery code included surgery involving ALNs (e.g. radical mastectomy), or if there was a concurrent CPT code for SLNB/ALND in addition to the primary breast surgery. All CPT codes used for our case selection and categorization are included in Table 1.
Statistical Analysis
The primary outcome measured was the annual proportion of patients diagnosed with DCIS who underwent any ALN intervention in each breast surgery modality. A secondary outcome involved a subgroup analysis to determine the annual proportions of patients who underwent SLNB, ALND, or no axillary evaluation. Demographic variables collected included age, body mass index (BMI), race, smoking status, and presence of diabetes. These variables were compared across treatment groups and are shown in Table 2. Rates of specific annual ALN interventions for each treatment group were analyzed using a smoothed linear regression model and a non-parametric Mann–Kendall trend test and Sen’s slope. Results were considered significant for p < 0.05. All analysis was performed using R-Studio software (R Foundation for Statistical Computing, Vienna, Austria).
Results
A total of 43,448 patients from 2005 to 2017 met the inclusion and exclusion criteria: 9460 patients underwent mastectomy alone; 11,044 patients received mastectomy with immediate reconstruction; 20,895 received partial mastectomy; and 2049 patients received oncoplastic surgery. Demographic data are shown in Table 2.
Our primary analysis of 2005–2017 trends revealed the following changes in ALN evaluation in DCIS patients: ALN surgery (SLNB or ALND) in the mastectomy group increased overall: the mastectomy-alone group experienced increases from 57.1 to 65.8% (Sen’s slope 1.46%, R2 0.75, p < 0.01) and the mastectomy with immediate reconstruction group experienced increases from 58.5 to 72.1% (Sen’s slope 1.15%, R2 0.83, p < 0.01). Smaller but significant increases were also seen in the BCS population: the partial mastectomy group increased from 14.0 to 21.1% (Sen’s slope 0.75%, R2 0.81, p < 0.01) and the oncoplastic surgery group increased from 10.5 to 23.0% (Sen’s slope 1.08%, R2 0.82, p < 0.01). The changes in the BCS population were attributed to increasing rates of SLNB, despite decreasing rates of ALND.
In our subgroup analysis of the mastectomy patients, SLNB increased from 27.5 to 48.6% (Sen’s slope 2.14%, R2 0.90, p < 0.01), while ALND decreased from 29.6 to 17.2% (Sen’s slope −0.61%, R2 0.09, p = 0.02). For patients undergoing mastectomy followed by immediate reconstruction, SLNB increased from 34.8% in 2005 to 64.0% in 2017 (Sen’s slope 2.09%, R2 0.78, p < 0.01), while ALND decreased from 23.7 to 8.1% (Sen’s slope − 0.90%, R2 0.83, p < 0.01). Patients undergoing partial mastectomy and oncoplastic surgery saw similar significant increases in SLNB, and a decrease in ALND in the partial mastectomy cohort, with insignificant change in the oncoplastic surgery cohort (see Table 3).
Discussion
Breast cancer affects one in eight women in the US,19 and 20% of diagnosed breast cancers are DCIS. Widespread adoption of breast cancer screening and improved imaging technology have led to an increase in the incidence of DCIS detection, from 1.8 per 100,000 to over 32 per 100,000 in over the last 30 years.7 Despite the rate of ALN metastasis in DCIS being very low,20,21 rates of ALN surgery for DCIS of between 18 and 63% have been reported in the literature depending on treatment modality;7,8 however, its benefit on cancer recurrence, metastasis, or survival has not been proven. It is for this reason that several national cancer organizations have made recommendations to guide surgeon’s treatment of ALNs in DCIS.
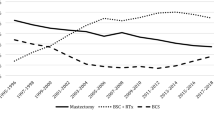
Over the last 13 years, we found that there have been significant increases in the rates of SLNB, as well as concomitant decreases in the rates of ALND, regardless of surgery modality, in patients undergoing breast surgery for DCIS. However, when considering total rates of sentinel lymph node evaluation, there have been increases in the rates of evaluation in patients undergoing both mastectomy and BCS for DCIS, despite guidelines that state that in most cases it is not necessary to evaluate ALNs when undergoing BCS (see Fig. 1).
The rates of ALN evaluation reported here for mastectomy and BCS are consistent with previous reports. Shah et al. investigated patients who underwent mastectomy for DCIS from 2000 to 2008 and found that the overall ALN evaluation rate was 51%.8 Similarly, Coromilas et al. reported that from 2006 to 2012, ALN evaluation for DCIS increased significantly in patients undergoing mastectomy, but stayed relatively stable in patients undergoing BCS.22
This analysis is unique in several ways. First, it is the most recent large-volume, multicenter analysis currently available. Specifically, it is the only analysis that accounts for trends over a time period that includes 2013–2017. Second, it specifically analyzes trends in SLNB and ALND for each of four possible surgical treatment modalities for DCIS. It objectively quantifies these trends over a 13-year period, providing insight into the changing annual practice patterns year over year, on average. We used Sen’s slope to analyze the temporal trend, which is advantageous when compared with the traditional least squares regression method because it uses the median of the slopes of all data as opposed to the mean, thus being more resistant to the effects of outliers.
This study not only analyzed the rate of SLNB and ALND at the time of mastectomy or partial mastectomy but also further delineated differences among patients undergoing immediate breast reconstruction or oncoplastic surgery. With high present-day interest in oncoplastic surgery,23 future organizational guidelines need to be specific as to when to perform ALN evaluation in oncoplastic surgery. NCCN guidelines suggest that SLN biopsy can be considered in patients undergoing excision in an anatomic location compromising the performance of a future sentinel lymph node procedure. This may be pertinent with Level 2 volume displacement oncoplastic operations using reduction mammaplasty or mastopexy techniques where quadrants of breast tissue may be removed,24 which may explain the 23% ALN evaluation being performed in the oncoplastic surgery arm. Additionally, we found that from 2005 to 2017, the increase in the rate of ALN evaluation was greatest in patients undergoing mastectomy with immediate breast reconstruction. Perhaps this was due to numerous reports in the literature about breast reconstruction obscuring future cancer surveillance,25,26,–27 but a limitation of using large databases such as the ACS NSQIP is that queries such as this cannot be made ad hoc.
Because DCIS has not extended beyond the breast duct, patients diagnosed with DCIS theoretically have no risk for lymph node metastasis, which would therefore preclude the need for axillary surgery at all.28 However, DCIS is upstaged to invasive carcinoma in as many as 20% of cases on final pathology.29,30,31,–32 In addition, while the risk of nodal metastasis is reportedly low in DCIS, final pathology of invasive carcinoma after mastectomy would necessitate some degree of lymph node evaluation as per present-day standard of care. Mastectomy is known to cause distortion of normal lymphatic drainage,33 and therefore subsequent lymph node evaluation would, of necessity, likely require full axillary lymphadenectomy. This would no doubt be excessive in a majority of cases, and it is for this reason that major cancer organizations recommend to strongly consider sentinel node biopsy at the time of mastectomy for DCIS. Our study has appropriately shown the decline of ALND and rise of SLNB to over 70% in the mastectomy with reconstruction group, potentially due to this rationale. While approximately 30% of mastectomy patients with DCIS not undergoing any ALN evaluation may be high, the authors acknowledge that there may be situations where a patient’s demographic background (such as age or severe comorbidities) may not justify the diagnostic information that axillary surgical evaluation provides, especially if the patient is not eligible for adjuvant treatment.
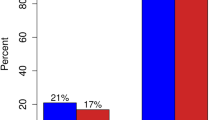
It is disturbing that 17% of patients undergoing mastectomy alone for DCIS are still receiving ALND, even though this rate has decreased dramatically over the course of the study period. It has been documented in the literature that histologic touch preparations for sentinel node biopsy sometimes give false positive results,34,35 which may be the reason for a completion ALND in some instances. However, the incidence of false positives is small and likely does not fully explain our finding. Several studies have identified risk factors for invasive carcinoma or SLN metastases despite a core biopsy that shows DCIS; these include high nuclear grade, size of the DCIS cluster, or comedonecrosis.36,37,–38 Unfortunately, the NSQIP database does not provide preoperative cancer-specific data such as pathology results, staging information, or clinical tumor characteristics, which may have driven surgical decision making leading to many of these completion dissections.
There is also no clear explanation for 21% of patients receiving axillary staging interventions while undergoing partial mastectomy for DCIS. Moreover, these rates actually increased between 2005 and 2017. One may hypothesize that the surgeons who performed sentinel lymph node biopsies in these situations justified their operative reasoning on the clinical presentations of the patient where they felt an axillary mass or were worried about breast tumor characteristics. The fact of the matter remains that if guidelines were followed, this 21% of patients would not have gotten unnecessary axillary surgery because the final pathology remained DCIS. Again, all patients whose final pathology showed invasive cancer were excluded from our analysis.
Numerous recent studies such as the ACOSOG Z0011 trial, NSABP B-32 trial, and International Breast Cancer Study Group (IBCSG) 23-01 trial support limited ALN surgery, even with invasive disease. These are undoubtedly the impetus behind the universally decreasing rates of ALND and concomitant increase in SLNB, and have laid the foundation for the ASCO and NCCN guidelines for ALN management for invasive carcinoma.39,40,–41 Similar principles have been extended to patients undergoing breast surgery for DCIS.
Given this, the results of our paper can be used to effect change. Societies such as the Society of Surgical Oncology or the American Society of Breast Surgeons can highlight education sessions specifically focused on axillary nodal surgery in the setting of DCIS. Certainly, a topic of interest would be how to address axillary surgery in the patient with DCIS who is receiving extensive oncoplastic surgery. Cancer accreditation organizations could request mandated practice patterns using the appropriateness of axillary nodal surgery in DCIS as a quality metric measure. As investigation on surgical indications for DCIS increases, and in the wake of new data that support minimizing axillary surgery, we should make every effort to adhere to established guidelines when considering axillary surgery on patients with DCIS.
There are several limitations to this study. While the NSQIP database draws from over 700 hospitals across the country and contains data for millions of surgical cases, its primary purpose is to allow for the analysis of perioperative data in order to minimize perioperative complications and increase quality and safety control. The database is not comprehensive of all surgical cases performed, but rather includes approximately 10–20% of annual surgeries within the US. It also does not contain many of the patient-specific variables that drive surgeon decision making, especially in oncologic surgery. However, using the NSQIP database for the selection of cases for trend analysis is commonly accepted in the general body of literature.12,15,42 Data collection by the NSQIP is a trained position, in each hospital, that is frequently audited, and NSQIP audits have revealed a human error rate of < 3%.43 Additionally, the NSQIP database is a combination of academic and community hospital settings that give a reasonable representation of breast surgery being performed in the US. Next, oncoplastic surgery is a newer and growing practice, and as such does not yet have specific CPT codes for oncoplastic procedures. We selected oncoplastic cases from the database using our coding experience by comparing codes with the coding protocol we use for these cases at our institution. Lastly, this retrospective analysis is purely descriptive and does not seek to establish causation, which is an opportunity for further prospective research.
Conclusion
Despite national cancer organizations recommending SLNB when undergoing mastectomy for DCIS, and in most cases recommending to not offer SLNB for patients undergoing BCS for DCIS, approximately 20–30% of cases continue to not follow these guidelines. Further prospective investigation is needed to discover the factors associated with practices contrary to these guidelines. Opportunities to educate patients and surgeons regarding the appropriate use of SLNB in DCIS are warranted.
References
Tanis PJ, Nieweg OE, Valdes Olmos RA, Th Rutgers EJ, Kroon BB. History of sentinel node and validation of the technique. Breast Cancer Res. 2001;3(2):109–12.
Jacobson JA, Danforth DN, Cowan KH, d’Angelo T, Steinberg SM, Pierce L, et al. Ten-year results of a comparison of conservation with mastectomy in the treatment of stage I and II breast cancer. N Engl J Med. 1995;332(14):907–11.
Darby S, McGale P, Correa C, Taylor C, Arriagada R, Clarke M, et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet. 2011;378(9804):1707–16.
Fisher B, Anderson S, Bryant J, Margolese RG, Deutsch M, Fisher ER, et al. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. New Engl J Med. 2002;347(16):1233–41.
Kelsall JE, McCulley SJ, Brock L, Akerlund MTE, Macmillan RD. Comparing oncoplastic breast conserving surgery with mastectomy and immediate breast reconstruction: case-matched patient reported outcomes. J Plast Reconstr Aesthet Surg. 2017;70(10):1377–1385
Chatterjee A, Serniak N, Czerniecki BJ. Sentinel lymph node biopsy in breast cancer: a work in progress. Cancer J. 2015;21(1):7.
Mitchell KB, Lin H, Shen Y, Colfry A, Kuerer H, Shaitelman SF, et al. DCIS and axillary nodal evaluation: compliance with national guidelines. BMC Surg. 2017;17(1):12.
Shah DR, Canter RJ, Khatri VP, Bold RJ, Yang AD, Martinez SR. Utilization of sentinel lymph node biopsy in patients with ductal carcinoma in situ undergoing mastectomy. Ann Surg Oncol. 2013;20(1):24–30.
Killelea BK, Long JB, Dang W, Mougalian SS, Evans SB, Gross CP, et al. Associations between sentinel lymph node biopsy and complications for patients with ductal carcinoma in situ. Ann Surg Oncol. 2018;25(6):1521–9.
Pyfer B, Chatterjee A, Chen L, Nigriny J, Czerniecki B, Tchou J, et al. Early postoperative outcomes in breast conservation surgery versus simple mastectomy with implant reconstruction: a NSQIP analysis of 11,645 patients. Ann Surg Oncol. 2016;23(1):92–8.
Mattingly AE, Ma Z, Smith PD, Kiluk JV, Khakpour N, Hoover SJ, et al. Early postoperative complications after oncoplastic reduction. Southern Med J. 2017;110(10):660–6.
Jonczyk MM, Jean J, Graham R, Chatterjee A. Trending towards safer breast cancer surgeries? Examining acute complication rates from a 13-year NSQIP analysis. Cancers. 2019;11(2):253. https://doi.org/10.3390/cancers11020253.
Lyman GH, Temin S, Edge SB, Newman LA, Turner RR, Weaver DL, et al. Sentinel lymph node biopsy for patients with early-stage breast cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2014;32(13):1365–83.
Gradishar WJ, Anderson BO, Balassanian R, Blair SL, Burstein HJ, Cyr A, et al. Breast Cancer, Version 4.2017, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw. 2018;16(3):310–20.
Jonczyk MM, Jean J, Graham R, Chatterjee A. Surgical trends in breast cancer: a rise in novel operative treatment options over a 12 year analysis. Breast Cancer Res Treat. 2019;173(2):267–274.
American College of Surgeons (ACS). ACS National Surgical Quality Improvement Program. Overview and description of ACS NSQIP database and participant user files. Chicago, IL: ACS; 2018.
Losken A, Hart AM, Chatterjee A. Updated evidence on the oncoplastic approach to breast conservation therapy. Plast Reconstr Surg. 2017;140(5S Advances in Breast Reconstruction):14S–22S.
Chatterjee A, Gass J, Patel K, Holmes D, Kopkash K, Peiris L, et al. A consensus definition and classification system of oncoplastic surgery developed by the American Society of Breast Surgeons. Ann Surg Oncol. 2019;26(11):3436–3444.
American Cancer Society. Breast Cancer Facts & Figures 2017–2018: Atlanta, GA: American Cancer Society, Inc.; 2017. Available at: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/breast-cancer-facts-and-figures-2017-2018.pdf.
Tada K, Ogiya A, Kimura K, Morizono H, Iijima K, Miyagi Y, et al. Ductal carcinoma in situ and sentinel lymph node metastasis in breast cancer. World J Surg Oncol. 2010;8:6.
Osako T, Iwase T, Ushijima M, Horii R, Fukami Y, Kimura K, et al. Incidence and prediction of invasive disease and nodal metastasis in preoperatively diagnosed ductal carcinoma in situ. Cancer Sci. 2014;105(5):576–82.
Coromilas EJ, Wright JD, Huang Y, Feldman S, Neugut AI, Chen L, et al. The influence of hospital and surgeon factors on the prevalence of axillary lymph node evaluation in ductal carcinoma in situ. JAMA Oncol. 2015;1(3):323–32.
Chatterjee A, Gass J, Burke MB, Kopkash K, El-Tamer MB, Holmes DR, et al. Results from the American Society of breast surgeons oncoplastic surgery committee 2017 survey: current practice and future directions. Ann Surg Oncol. 2018;25(10):2790–4.
Chatterjee A, Dayicioglu D, Khakpour N, Czerniecki BJ. Oncoplastic Surgery: Keeping it simple with 5 essential volume displacement techniques for breast conservation in a patient with moderate to large sized breasts. Cancer Control. 2017;24(4):1–11.
Skinner KA, Silberman H, Dougherty W, Gamagami P, Waisman J, Sposto R, et al. Breast cancer after augmentation mammoplasty. Ann Surg Oncol. 2001;8(2):138–44.
Miglioretti DL, Rutter CM, Geller BM, Cutter G, Barlow WE, Rosenberg R, et al. Effect of breast augmentation on the accuracy of mammography and cancer characteristics. JAMA. 2004;291(4):442–50.
Silverstein MJ, Handel N, Gamagami P, Gierson ED, Furmanski M, Collins AR, et al. Breast cancer diagnosis and prognosis in women following augmentation with silicone gel-filled prostheses. Eur J Cancer. 1992;28(2–3):635–40.
McCarten D, Gemignani ML. Current Management of the Axilla. Clin Obstet Gynecol. 2016;59(4):743–55.
Watanabe Y, Anan K, Saimura M, Koga K, Fujino M, Mine M, et al. Upstaging to invasive ductal carcinoma after mastectomy for ductal carcinoma in situ: predictive factors and role of sentinel lymph node biopsy. Breast Cancer. 2018;25(6):663–70.
Sakr R, Antoine M, Barranger E, Dubernard G, Salem C, Darai E, et al. Value of sentinel lymph node biopsy in breast ductal carcinoma in situ upstaged to invasive carcinoma. Breast J. 2008;14(1):55–60.
Wilkie C, White L, Dupont E, Cantor A, Cox CE. An update of sentinel lymph node mapping in patients with ductal carcinoma in situ. Am J Surg. 2005;190(4):563–6.
Jakub JW, Murphy BL, Gonzalez AB, Conners AL, Henrichsen TL, Maimone St, et al. A validated nomogram to predict upstaging of ductal carcinoma in situ to invasive disease. Ann Surg Oncol. 2017;24(10):2915–24.
Virnig BA, Tuttle TM, Shamliyan T, Kane RL. Ductal carcinoma in situ of the breast: a systematic review of incidence, treatment, and outcomes. J Natl Cancer Inst. 2010;102(3):170–8.
Valdes EK, Boolbol SK, Cohen JM, Feldman SM. Intra-operative touch preparation cytology; does it have a role in re-excision lumpectomy? Ann Surg Oncol. 2007;14(3):1045–50.
Guidroz JA, Johnson MT, Scott-Conner CEH, De Young BR, Weigel RJ. The use of touch preparation for the evaluation of sentinel lymph nodes in breast cancer. Am J Surg. 2010;199(6):792–6.
Meijnen P, Oldenburg HS, Loo CE, Nieweg OE, Peterse JL, Rutgers EJ. Risk of invasion and axillary lymph node metastasis in ductal carcinoma in situ diagnosed by core-needle biopsy. Br J Surg. 2007;94(8):952–6.
Yi M, Krishnamurthy S, Kuerer HM, Meric-Bernstam F, Bedrosian I, Ross MI, et al. Role of primary tumor characteristics in predicting positive sentinel lymph nodes in patients with ductal carcinoma in situ or microinvasive breast cancer. Am J Surg. 2008;196(1):81–7.
Renshaw AA. Predicting invasion in the excision specimen from breast core needle biopsy specimens with only ductal carcinoma in situ. Arch Pathol Lab Med. 2002;126(1):39–41.
Galimberti V, Cole BF, Zurrida S, Viale G, Luini A, Veronesi P, et al. Axillary dissection versus no axillary dissection in patients with sentinel-node micrometastases (IBCSG 23-01): a phase 3 randomised controlled trial. Lancet Oncol. 2013;14(4):297–305.
Giuliano AE, Hunt KK, Ballman KV, Beitsch PD, Whitworth PW, Blumencranz PW, et al. Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA. 2011;305(6):569–75.
Krag DN, Anderson SJ, Julian TB, Brown AM, Harlow SP, Costantino JP, et al. Sentinel-lymph-node resection compared with conventional axillary-lymph-node dissection in clinically node-negative patients with breast cancer: overall survival findings from the NSABP B-32 randomised phase 3 trial. Lancet Oncol. 2010;11(10):927–33.
Nocera NF, Pyfer BJ, De La Cruz LM, Chatterjee A, Thiruchelvam PT, Fisher CS. NSQIP analysis of axillary lymph node dissection rates for breast cancer: implications for resident and fellow participation. J Surg Educ. 2018;75(5):1281–6.
American College of Surgeons. User Guide for the 2017 ACS NSQIP participant use data file (PUF). October 2018.
Acknowledgments
The ACS NSQIP was the sole source of data used for this study; they have not verified and are not responsible for the statistical validity of the data analysis or the conclusions derived by the authors. This project does not represent the views or plans of the ACS or the ACS NSQIP. The described study was supported by the National Center for Advancing Translational Sciences, National Institutes of Health (NIH), and Award Number TL1TR002546. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Funding
Sources of Financial or Material Support None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pyfer, B.J., Jonczyk, M., Jean, J. et al. Analysis of Surgical Trends for Axillary Lymph Node Management in Patients with Ductal Carcinoma In Situ Using the NSQIP Database: Are We Following National Guidelines?. Ann Surg Oncol 27, 3448–3455 (2020). https://doi.org/10.1245/s10434-020-08374-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-08374-0