Abstract
Purpose
The study aims to search for potent cellulase producer from the gut of Gryllotalpa africana as well as to characterize and determine the effect of the purified enzyme on the cellulosic waste materials.
Methods
The potent cellulolytic strain was identified through morphological, biochemical, physiological, and molecular characterization like 16S rRNA and fatty acid methyl ester profile. After the optimization of cellulase production, the enzyme was purified through DEAE-Sepharose column chromatographic separation. The molecular weight of the purified enzyme was determined by SDS-PAGE analysis. The purified enzyme was characterized in terms of its activity and its effect on cotton fiber, and sawdust was also studied.
Result
The selected potent strain GAC 16.2 was identified as Acinetobacter junii that was capable to produce enhanced cellulase (112.38 ± 0.87 U/ml) at standardized optimum fermentation conditions. The molecular weight of the purified enzyme was determined as 55 kDa. The utmost activity of the purified enzyme was detected pH 7.0, temperature 50 °C, and in the presence of metal ions like Mg2+ and Mn2+. The substantive degradation of cotton fiber and sawdust has been observed in a reasonably short period.
Conclusion
Purified cellulase from the selected isolate A. junii GAC16.2, a gut isolate of G. africana, has the potentiality to degrade cellulosic substances. This property can make the isolate a potent candidature for industrial application, as well as an effective biotechnological tool for environmental monitoring through cellulosic waste management.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Cellulose is the most abundant renewable organic molecule on the earth (Klemm et al. 2005). Its enzymatic degradation by cellulase is a promising approach for its effective utilization. The resultant reducing sugars could be used for the production of desired value-added byproducts (Sukumaran et al. 2005; Octave and Thomas 2009). This enzyme is extensively consumed in paper and pulp industries, in textile industry, for the production of biofuel, as an anti-biofilm agent, for waste management in pharmaceutical industries, for pollution control, and also in food as well as in feed industries (Sreena et al. 2016; Aarti et al. 2018; Thomas et al. 2018; Sreena and Sebastian 2018).
Insects are the most species-rich and diverse group of arthropods (Moran and Telang 1998). They harbor a rich and complex microbial community in their guts as symbionts that differ in taxa due to their altered feeding habits. In the course of evolution, these symbiotic associations between insects and different microorganisms have been deeply rooted (Moran and Telang 1998). These symbionts exhibit beneficial activities for their hosts. One such activity is the production of various important hydrolytic enzymes including cellulase. Thus, they play an indispensable role in the digestion of ingested food and are a “hot spot” for useful genes and proteins (Dillon and Dillon 2004; Dharne et al. 2006). Though only a few detail studies have been made concerning the enzyme-producing insect gut bacteria, it requires further observations (Dillon and Dillon 2004).
Gryllotalpa africana (order: Orthoptera) is an important soil-inhabiting agricultural pest. Their natural habitat is moistened humus-enriched biotopes. In summer, they make galleries near the ground surface, and overwinter they reside at underground holes having a depth of 0.8–1.2 meter. These insects normally feed on a wide range of economically important plants, particularly potato, wheat, maize, oats, sugar beet, barley, rice, soya, vegetable crops, and grasses (Heinrichs and Barrion 2004; James et al. 2010). It is one of the important major soil pests in eastern Gangetic plains of West Bengal, India. They cause 5–6% plant damage along with 10–15% tuber damage to potato plants (Konar et al. 2005).
Studies regarding the microbial cellulase production have been concerted mostly on fungi, but there is an increasing interest in bacteria (Sukumaran et al. 2005). Further, the report on cellulase production of proteobacteria is very scanty. Members under the genus Acinetobacterare common free-living saprophytic archaea. Owning to their metabolic versatility, they are capable to adapt in varied habitat including the gut of insects (Sand et al. 2011; Malhotra et al. 2012; Vilanova et al. 2012).
Keeping in view the essentiality of the facts, the present investigations were carried out to search for new potent cellulase producers from the gut of G. africana and assess the fermentation parameters for optimization of enzyme production by the potent isolate in submerged condition. The characterization of the purified cellulase and its efficiency on cotton fiber and sawdust was also studied.
Materials and methods
Collection of insects
To check the presence of the cellulase-producing gut bacteria of Gryllotalpa africana (order: Orthoptera), the insects were collected from the different potato fields and agro-waste dumps located at the Gangetic plain of West Bengal, India. The sites from where the insects were collected are Hatgobindapur (23.2578° N, 87.9788° E), Memari (23.1745° N, 88.1034° E) Polempur (23.2044° N, 87.8435° E), Sagrai (23.1514° N, 87.8169° E), Debipur (23.1422° N, 88.1585° N) of Purba-Bardhaman district and Arambag (22.8728° N, 87.7911° E), Gurap (23.0348° N, 88.1218° E), Singur (22.8150° N, 88.2345° E), Talandu (23.0182° N, 88.3567° E), and Chinchura (22.9012° N, 88.3899° E) of Hooghly district of West Bengal, India. A total of 20 adult insects were collected, two from each location.
Isolation of gut bacteria
The insects were retained in starvation for 2 days to exclude allochthonous microbial communities from their gut. Then, they were used as a source of screening for cellulolytic microorganisms (Zhang and Jackson 2008). The wings of the insects were excised and then the body surfaces were washed thoroughly with 70% ethanol. The insects were aseptically dissected within the laminar airflow cabinet. The guts were removed immediately after dissection. Portions of the gut were cleaned, cut into fragments, split open by longitudinal incision, and transferred to sterile Petri plates. It was then thoroughly flushed with sterilized chilled phosphate-buffered saline solution (0.89% NaCl, pH, 7.0). Subsequently, the gut fragments were homogenized with the same buffer solution in the ratio of 10:1 (volume:weight). The homogenates were used as inoculum for microbial culture.
Microbial culture
The gut homogenate (0.5 g) of each of the test insects was used after 10 serial dilutions. Carboxymethyl cellulose (CMC)-agar medium (CMC 0.2%, peptone 0.5%, beef extract 0.5% NaCl 0.5%, agar 2%, Merck, pH 7.0) was used for screening of the cellulolytic bacteria. Samples (0.1 ml) were taken from each dilution and within a laminar airflow cabinet and poured aseptically on sterile CMC-agar plate in duplicate. The inoculated CMC-agar plates were incubated at 35 °C for 48 h. The well-grown colonies with a different morphology from the incubated CMC-agar plates were isolated. Thereafter, they were streaked several times on the same medium to obtain pure cultures. The pure cultures were maintained on CMC-agar slants at 4 °C for further use.
Screening of isolates for qualitative enzyme production
For the screening of potent cellulase producers, the isolates were grown in the CMC-agar plate at 35 °C for 48 h. After that, the plates were flooded with 1% Congo red dye and destained with 1 M NaCl (Teather and Wood 1982). Finally, the promising isolates were screened based on their clear zone (halo) forming ability.
Screening of isolates for quantitative enzyme production
For the quantitative assay, the selective isolates were grown in M2 medium broth (Table 1) of initial pH 7.0. In a 100 ml Erlenmeyer flask, a 25 ml medium was taken, and it was inoculated with 4% inoculum (1.2 × 107 CFU/ml), v/v. The inoculated flask was incubated at 35 °C for 3 days at shaking condition (100 rpm). After incubation, the content was centrifuged at 8000 rpm for 10 min at 4 °C. The cell-free supernatant was used for enzyme assay.
Enzyme assay
The enzymatic activity of cellulase was measured according to the method of Denison and Koehn (1977) using 1% CMC in phosphate buffer (pH 7.0) as a substrate. The hydrolyzed end product from the CMC substrate due to the cellulolytic activity was measured at 540 nm by the dinitrosalicylic method using glucose as the standard and expressed as U/ml. A mixture of pre-inoculated production medium and the reaction mixture was used as a control. One enzyme unit (U) is defined as the amount of enzyme capable to release 1 mM of glucose from the substrate in 1 min at 38 °C. The concentration of soluble protein was measured by the method of Lowry et al. (1951).
Characterization and identification of the selected isolate
The selected isolate was grown on the CMC-agar plate after 48 h of incubation. The colony characterizations were made through visual observation. Micro-morphological studies were done using a compound microscope and a scanning electron microscope (SEM), Model Hitachi-530, Japan (Dietz and Mathews 1969). Physiological and biochemical characterization of the selected isolate was made according to the Society of American Microbiologists (1957) and Bergey’s Manual of Systematic Bacteriology (Garrity 2005).
The DNA of the selected isolate was isolated (Rainey et al. 1996). The 16S rRNA gene was amplified with primers 8-27f (5′-AGAGTTTGATCCTGGCTCAG-3′) and 1500r (5′-AGAAAGGAGGTGATCCAGCCA-3′), separated on 1% agarose gel, eluted, and purified using a QIA quick gel extraction kit (Qiagen, USA). The purified PCR product was sequenced with four forward and three reverse primers, namely, 8-27f, 357f (5′-CTCCTACGGGAGGCAGCAG-3′), 704f (5′-TAGCGGTGAAATGCGTAGA-3′), 1114f (5′-GCAACGAGCGCAACC-3′), 685r (5′-TCTACGCATTTCACCGCTAC-3′), 1110r (5′-GGGTTGCGCTCGTTG-3′), and 1500r (E. coli numbering system). The 16S rDNA gene sequence was determined by the dideoxy chain termination method using the Big-Dye terminator kit at an ABI 310 Genetic Analyzer (Applied Biosystems, USA).
A sequence similarity search was done using GenBank BLASTN (Altschul et al. 1997). Sequences of closely related taxa were retrieved and aligned using the program CLUSTAL_X (Thompson et al. 1997), and the alignment was adjusted manually. For neighbor-joining analysis (Saitou and Nei 1987), distances between the sequences were calculated using Kimura’s two-parameter model (Kimura 1980). Bootstrap analysis was performed to assess the confidence limits of the branching (Felsenstein 1985).
Fatty acid composition of the cell membrane was determined by fatty acid methyl ester (FAME) analysis according to MIDI version 6.1, Method RTSBA6, Matches Library RTSBA6 6.10 (Morey et al. 2013).
Submerged fermentation
Four days old culture of the selected isolate was used as a source of inoculum for performing various experiments. Batch experiments were carried out in a 100-ml Erlenmeyer flask containing 25 ml medium at 35 °C for 3 days at shaking (100 rpm) condition using 4% inoculum (1.2 × 107CFU/ml) and an initial pH of 7.0. After fermentation, the cell-free culture broth was used as a crude enzyme for assay. The strategy adopted to optimize one parameter at a time. The optimized parameter of an experiment was considered for the designing of subsequent experiments keeping all other conditions constant. All the experiments were done in triplicate.
To obtain maximum cellulase production from the selected isolate, the effects of various fermentation factors were optimized. To perform medium suitability tests, different media (M1, M2, M3, and M4) were taken into consideration (Table 1). Among the various physical factors, incubation time (24–84 h) with an interval of 12 h, inoculum volume (1–9%) with an interval of 1%, pH (5.0–8.5) with an interval of 0.25, temperature (30–44 °C) with an interval of 2 °C, and aeration (static to 200 rpm) with an interval of 50 rpm, were optimized. For the nutritional factors, the suitability of the various supplementary carbon sources (sucrose (Sac), fructose (Fru), maltose (Mal), lactose (Lac), dextrose (Dex), mannitol (Man), and malt extract (Mal ex)) were trialed. The effects of several concentrations (0.4–2%, w/v) of selected supplementary carbon sources were also evaluated. To determine the most effective substrate concentration for enzyme production, CMC was tested at different concentrations (0.2–2.8%, w/v). Different inorganic nitrogen sources (ammonium sulfate (AS), potassium nitrate (PN) and ammonium chloride (AC), sodium nitrate (SN), and urea) were also taken into consideration. The effective suitable concentration of selected inorganic nitrogen sources was also checked within a range of 0.4–2%, w/v. Vitamins such as ascorbic acid (ASC), pyridoxine (PYR), thiamine (THI), biotin (BIO), niacin (NIA), and riboflavin (RIB) were also trialed at different concentrations (up to 60 μg/ml). The effect of supplementation of casein hydrolysate and molasses (up to 0.01 %, w/v) was also tested. The effect of different surfactants like SDS, EDTA, Tween 80 and Triton X-100, and their effective concentration (up to 0.2 μg/ml) was also determined.
Purification
After centrifugation (8000 rpm for 10 min), the supernatant was subjected to ammonium sulfate precipitation with uninterrupted stirring at 4 °C. The precipitated proteins were recovered by centrifugation (10000 rpm for 20 min at 4 °C) and dialyzed (using LA401, HiMedia) against 20 mM potassium phosphate buffer (pH − 6.75) at 4 °C with three changes during 24 h. The partially purified dialyzed sample was used for the DEAE-Sepharose column (Sigma). The dialyzed proteins were eluted using a linear gradient of 0.05–1.0 M NaCl (in phosphate buffer) with a flow rate of 1 ml/min. Using fraction collectors, the eluted fractions were collected. After collection, respective fractions were screened for protein estimation (Lowry et al. 1951) and measuring total activity by the dinitrosalicylic acid method (Denison and Koehn 1977).
Characterization of the purified enzyme
The molecular weight of the purified enzyme was characterized by carrying out 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) (Laemmli 1970). The molecular ladder (10–250 kDa) (Thermo Fisher Scientific) was used to estimate the molecular weight (M) of the purified enzyme.
To ascertain the optimum pH value of the purified cellulase from the selected isolate, for its activity, several buffers like acetate buffer (pH 4 and 5), citrate buffer (5 and 6), phosphate buffer (pH 6–8), Tris-HCL (pH 8 and 9), Glycine-NaOH buffer (pH 9 and 10), sodium bicarbonate-NaOH buffer (pH 10 and 11), and KCl-NaOH buffer (pH 12) were used. The activity assay was performed by using 2% CMC with these different pH buffers and incubated at 50 °C for 20 min according to Miller (1959).
The influence of temperature on the purified enzyme activity was determined at a wide range of temperatures (20–80 °C) in phosphate buffer (pH 7.0) for 20 min using 2% CMC as a substrate. The activity was determined by the DNS method (Miller 1959).
To check the stability of the purified enzyme upon variable pH conditions, various buffers like acetate buffer (pH 4 and 5), citrate buffer (5 and 6), phosphate buffer (pH 6–8), Tris-HCL (pH 8 and 9), Glycine-NaOH buffer (pH 9 and 10), sodium bicarbonate-NaOH buffer (pH 10 and 11), and KCl-NaOH buffer (pH 12) were tested (Miller 1959).
To test the thermal stability of the purified enzyme, residual activity was determined in different temperature setup (20–80 °C) in 100 mM phosphate buffer (pH 7.0) (Balasubramanian et al. 2012).
The purified enzyme from GAC 16.2, the most promising cellulolytic microbe isolate, was investigated against different substrates (1%) for the substrate specificity test. The substrates were CMC, xylan, avicel, filter paper, and chitin. Reaction mixtures were performed in phosphate buffers (pH 7.0) for 20 min at 50 °C followed by the DNS method (Miller 1959).
The effects of metal ions, inhibitors, and surfactants on the activity of the purified cellulase were also tested. The metal ions employed in this study were K+, Na+, Ca2+, Mn2+, Mg2+, Fe2+, Ni2+, Hg2+, and Co2+ with a concentration of 1 mM and 5 mM. Among the inhibitors, phenylmethanesulfonyl fluoride (PMSF), β-mercaptoethanol (βME), dithiothreitol (DTT), EDTA, iodoacetic acid (IAA), and urea were taken into account at the concentration of (5 mM). Surfactants like SDS, Tween 20, and Triton X-100 were employed at 1%, v/v. Purified enzyme with 20 mM of phosphate buffer (pH 7.0) mixed separately with metal ions, inhibitors, and surfactants to make different reaction mixtures and individually incubated at 37 °C for 30 min. Relative activity was estimated by the DNS method (Miller 1959).
Effect of cellulase on cotton fiber and sawdust
In one case, pieces of sterilized cotton fibers (CF) were dipped into 200 μl purified enzyme (in 20 mM potassium phosphate buffer, pH 6.75) at 37 °C for varied exposures (12–48 h). In another case, the sterilized sawdust (SD) was primarily washed and allowed to exposure (24–48 h) in the enzyme. The amount of reducing sugar was estimated for both of the enzyme-treated CF and SD (Denison and Koehn 1977). Control sets (without enzyme treatment) were also prepared simultaneously. The SEM studies were made after 10 min of gold metallization (Model Hitachi-530, Japan) following Dietz and Mathews (1969).
Statistical analysis
The standard error (±) of mean was calculated from three determinations using Microsoft Office Excel 2013. The significance level of the variables was determined by one-way ANOVA at the level of 95% (P < 0.05) using the software IBM-SPSS (V.23).
Results
Isolation and Screening for cellulase producers
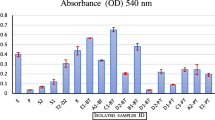
Out of 43 isolates, 5 isolates were primarily screened based on the halo zone forming capability in the CMC-agar plate treated with Congo-red and NaCl. Based on DNS assay, the isolate GAC 16.2 was chosen as a potent cellulase producer having production capability 0.26 ± 0.09 U/ml (Figs. 1 and 2).
Characterization and identification of the selected isolate
The colony of GAC 16.2 was grown on the CMC-agar plate. It was observed as off-white, circular with convex elevation, and having an entire margin. The cells were found singly or in aggregate, ellipsoidal 3–4 μm × 2–2.5 μm (Fig. 3), Gram-negative, confluent growth between 15–50 °C, and tolerance of pH between 4.0–10.0 and NaCl up to 5%, and utilization of citrate as sole carbon source, and acid production from xylose, glucose, fructose, sucrose and meso-inositol, and nitrate reduction were found positive (Data not shown).
16S rDNA sequence (1407 bases) of the isolate GAC 16.2 showed a 100% pairwise similarity with Acinetobacter junii LMG 998T (AM410704). The phylogenetic tree also suggested the closest relationship of GAC 16.2 with the type strain of Acinetobacter junii (Fig. 4). This strain (GAC 16.2) has also been deposited to Microbial Type Culture Collection (MTCC), India (Accession no. MTCC 11818), and the 16S rDNA sequence has been submitted to NCBI (GenBank Acc No. KJ174319).
From the FAME analysis of isolate GAC 16.2, contain of saturated and unsaturated fatty acids found to be 30.38% and 69.39% respectively (Fig. 5, Table 2). FAME similarity index of GAC 16.2 was very close to A. calcoaceticus (index 0.210) and A. haemolyticus (index 0.209).
Optimization for cellulase production
From the experimental result, medium M1 was found the most suitable for enzyme production (0.26 ± 0.09 U/ml), followed by M3, M2, and M4 with production 0.156 ± 0.013, 0.132 ± 0.012, and 0.114 ± 0.011 U/ml, respectively (Fig. 6a).
The enzyme production ability of the isolate increased with the fermentation period up to 72 h (0.35 ± 0.01 U/ml); thereafter, it declined (Fig. 6b). The experiment revealed (Fig. 6c) that the enzyme production increased with the increase of inoculum size up to 8% (0.626 ± 0.021 U/ml). The optimum initial pH of the production medium was found as 6.75 for cellulase production (0.65 ± 0.03 U/ml) by the isolate GAC 16.2 (Fig. 6d). The isolate showed optimum enzyme production (0.69 ± 0.015 U/ml) at 38 °C which decreased sharply on either side of that temperature (Fig. 6e). It was found that the isolate GAC 16.2 produced best (Fig. 6f) at 150 rpm (0.82 ± 0.041 U/ml).
Among the several carbon sources, lactose supplementation effected a sharp increase in production (49.95 ± 1.72 U/ml) but no such boosting effect in the case of sucrose and mannitol that was recorded (Fig. 7a). The optimum concentration of lactose was further checked (Fig. 7b) and found as 1.4% (55.23 ± 1.86 U/ml). For assessing the effect of substrate (CMC) concentration on enzyme production, it was found that the production increased slowly with the increase of substrate concentration up to 2% (57.19 ± 1.54 U/ml) and declined sharply thereafter (Fig. 7c). In the experiment of nitrogen source variation, SN was found much effective (92.24 ± 1.11 U/ml) in comparison to AS, PN, and AC (Fig. 7d). Further, the optimum concentration of SN was ascertained at the level of 1.6% for cellulase production (96.38 ± 2.4 U/ml) (Fig. 7e).
Among the vitamins, ASC, PYR, THI, BIO, NIA, and RIB are found as positive for cellulase production (Fig. 7f). However, BIO boosted cellulase production maximally (108.61 ± 0.63 U/ml). Further, the medium supplementation with molasses and casein hydrolysate found to increase enzyme production by 109.55 ± 0.78 U/ml and 112.38 ± 0.87 U/ml respectively (Fig. 7g). In the present study, the surfactants did not show any positive role in enzyme production (Fig. 7h).
Purification
Ammonium sulfate precipitation of the enzyme was standardized, and maximum activity was observed at 40–80% saturation. Cell-free fermented broth exhibited a specific activity of 0.43 U/mg, whereas the dialyzed enzyme showed a specific activity of 2.59 U/mg with 6.02-fold enhancement. After ion-exchange chromatography (DEAE-Sepharose column), all the fractions were assayed for cellulase activity, and fraction F4 found best (3.95 U/mg). The specific activity was successively amplified at every stage of purification, and ultimate augmentation was 9.2-fold (Table 3).
Characterization of the purified enzyme
The purified cellulase from GAC 16.2 was found as a single band with a molecular weight (M) of about 55 kDa on SDS-PAGE gel (Fig. 8), and no subunit was observed, and therefore, the purified enzyme was considered to be homogenous. The optimum pH and temperature for the utmost activity of the purified enzyme were found to be 7 (Fig. 9a) and 50 °C (Fig. 9b) respectively. Cellulase from GAC 16.2 exhibited its stability more than 60% in response to pH ranges from 6 to 9, and temperature ranges from 40–60 °C (Fig. 9a, b). Beyond this range, stability decreased at the level of less than 40%.
In course of evaluation of substrate specificity, purified enzyme represented its utmost activity against CMC (100%) and moderate activity towards filter paper, xylan but least activity in contrast to avicel, and no reaction output against chitin (Table 4).
The effect of metal ions on purified cellulase reflected that at the 1 mM concentration of different metal ions, Mn2+, Co2+, and Mg2+ activated the cellulase activity; however, Ni2+ and Hg2+ remarkably inhibited the enzyme activity. However, at the level of 5 mM, only Mg2+ and Mn2+ exhibited their positive effects (Table 5). Among the inhibitors, cellulase activity was inhibited significantly by EDTA and moderately by IAA, PMSF, and urea accordingly (Table 6). All the tested surfactants have an inhibitory effect on enzyme activity (Table 7).
Effect of cellulase on CF and SD
In both cases, the effect of the enzyme treatment was pronounced with increased time (Figs. 10 and 11). The efficient enzyme activity has supported by the analysis of the end product (Table 8).
All sets of experiments found statistically significant at the level of P < 0.05.
Discussions
Cellulose exists as crystalline or amorphous microfibrils in plant cell walls. In the gut, the cellulose fibers first need to get fragmented into simpler sugar residues to make it accessible to insects, a process in which bacteria are typically involved through enzymatic digestion (Russell et al. 2009; Watanabe and Tokuda 2010). The important pest G. africana dwells in humus-enriched moistened soil and consumes a wide range of economically important plants including potato (James et al. 2010). The pest consumes invertebrates, including insects and earthworms as food alternatively (Banerjee et al. 2017). Literature regarding their gut digestive enzyme is not available. However, Banerjee et al. (2016, 2017) reported two potent enzyme-producing bacteria Rhodococcus opacus GAA 31.1 and Kitasatospora cheerisanensis GAP 12.4 from their gut capable to produce amylase and protease, respectively. So, the experiment was designed through conceiving the idea that the gut of this agriculturally important major pest may contain potential sources of cellulolytic microflora that support in the digestion of the consumed food enriched with cellulosic material.
A potent cellulolytic isolate, GAC 16.2, was ultimately screened from the gut content of G. africana. Through the study of morphological, physiological, biochemical, and molecular characterization and specific study of cell membrane fatty acid analysis of the selected isolate, GAC 16.2 was identified as Acintobacter junii. Dhayal et al. (2014) isolated a strain of A. junii from the larvae of Anopheles stephensi. There is also a report of isolation of Acintobacter spp. from the gut of Glossina palpalis palpalis (Geiger et al. 2009). There is also a report about the involvement of A. junii in fresh cattle manure composite culture (Ghosh et al. 2018). The A. junii F6-02, a soil isolate of Taiwan, is also capable to produce xylanase (Lo et al. 2010).
The isolated GAC 16.2 tolerates a long range temperature (15–50 °C) and pH (4.0–10.0). An endophytic bacterium, Lentibacillus populi, isolated from poplar, is also reported to grow within a wide range of temperatures (15–50 °C) and pH at a range of 6.0–9.0 (Sun et al. 2016). Besides this, Nakajo et al. (2006) reported that Enterococcus faecalis can grow on broad ranges of pH (4.0–11). The members within the genus are also habituated to grow at a wide range of temperatures and pH. Hrenovic et al. (2014) isolated a strain of A. baumannii which grows at low pH (3.37) and tolerates high temperature at (50 °C). Hrenovic et al. (2016) reported the growth of A. baumannii beyond 42 °C. The growth of A. seohaensis sp. nov. growth was observed at pH 5.5–8.0 and temperature10–40 °C (Yoon and Kim 2007).
Common phenomenon or regulation for the growth and production ability of microorganisms is not available (Roy and Sen 2013). Thus, optimization of fermentation conditions is of great significance in microbial production including the enzyme. The isolate GAC 16.2 showed maximum cellulase production (0.26 ± 0.09 U/ml) on the M1 in comparison to the other tested media. The increase in cellulase production on the M1 medium can be explained by the availability of the required nutrients than that of other tested media for the isolate. Among the cellulolytic bacterial community, Bacillus, Micrococcus, and Cellulomonas are common, and their enzyme production was observed up to 0.034 U/ml (Immanuel et al. 2006). Other cellulase producers like Brevibacillus and Geobacillus are found to produce maximally 0.02 U/ml and 0.058 U/ml, respectively (Rastogi et al. 2009). The enzyme production ability of the isolate GAC 16.2 increased with the fermentation period up to 72 h (0.35 ± 0.01 U/ml); thereafter, it declined. This pattern is observed in cellulase producers, viz., Bacillus pumilus EWBCM1 (Shankar and Isaiarasu 2011) and B. subtilis (Bai et al. 2012). The optimum incubation period depends primarily on nutrient availability and other cultural conditions (Wanderley et al. 2004). Nutrient availability decreases with the increase in the age of the culture. The production increased with the increase of inoculum size up to 8% (0.626 ± 0.021 U/ml), and this data is corroborated with the finding of Omojasola and Jilani (2009).
The pH of the medium influences the fermentation capability of an organism. The isolate GAC 16.2 produced cellulase maximally (0.65 ± 0.03 U/ml) at the production medium having an initial pH of 6.75. Like GAC 16.2, some cellulase producers preferred pH near the neutral for enzyme production (Younis et al. 2010; Bai et al. 2012). The selected isolate showed optimum enzyme production (0.69 ± 0.015 U/ml) at 38 °C which decreased sharply on either side of that temperature. At lower temperatures, the transport of substrate across the cells is limited and that affects the yield. Moreover, at elevated temperature, the energy requirement is high for growth due to the thermal denaturation of metabolic enzymes resulting in the inhibition in product formation (Aiba et al. 1973). But, the isolate does not require extra energy because it grows almost in ambient temperature.
Under agitation, the oxygen from the air becomes soluble in the fermentation broth. Thus, to provide sufficient oxygen to the fermenting organism, it requires agitation to accelerate the dissolution of oxygen into the medium. Aeration also increases nutrient availability as well as decreases loads of products from the vicinity of the producers (Brock and Madigan 1988). At increased agitation, the enzyme may get inactivated, but agitation at 150 rpm is suitable for its optimum activity (Singh and Kumar 1998). However, the working isolate could produce maximally at 150 rpm which is much less than the majority of the producers, indicating energy conservation. When a medium culture is shaken or stirred, the cellulose fibrils do not form a well-organized pellicle which is produced under static conditions. Cellulose mass will decrease with an increase in shaking speed which may reach up to 160 rpm (Pa’e et al. 2007). This may also be due to A. junii that is difficult to adapt to the harsh environment. Also, the original habitat of cellulase producing bacteria within insect gut is not at the much shaken condition.
Among various supplementary carbon sources, lactose presents a notable elevation in production (49.95 ± 1.72 U/ml). The optimum concentration of lactose was further checked and found as 1.4% (55.23 ± 1.86 U/ml). Cellulase production in Acremonium cellulolyticus and Microbacterium sp. was also enhanced by lactose (Fang et al. 2008; Sadhu et al. 2011). The exact cause(s) behind this synergistic effect of lactose with CMC on cellulase production is still unknown. However, the mechanism of lactose induction possibly lies with the intracellular level of galactose-1-phosphate that triggers the signaling for enzyme production. Since cellulose is insoluble, the enzyme itself cannot trigger the induction. It can be induced in the presence of soluble saccharide. Further cellulase production is intricately regulated by the activation and repression mechanisms of soluble sugars (Ilmen et al. 1997). Lactose is the only known economically feasible additive inducers of the cellulase gene (Pandey et al. 2011). It was also found that the production increased slowly with the increase of substrate (CMC) concentration up to 2% (57.19 ± 1.54 U/ml) and declined sharply thereafter. High substrate concentration can cause product inhibition, resulting in a substantial reduction in enzyme formation (Irshad et al. 2013). As a source of nitrogen, sodium nitrate was found much effective (92.24 ± 1.11 U/ml) compared to ammonium sulfate, potassium nitrate, ammonium chloride, and urea. This finding corroborates with the enzyme production of Cellulomonas flavigena (Rajoka 2004).
The metabolic activities of microorganisms are much influenced by vitamins as it acts as a prosthetic group for many enzymes. The vitamins pyridoxine, thiamine, niacin, and riboflavin were found to play a positive role for cellulase production. Mostly the vitamins are found optimally between 40 and 50 μg/ml (Banerjee et al. 2016; Banerjee et al. 2017). However, biotin enhances cellulase production maximally (108.61 ± 0.63 U/ml). It can be substantiated through biotin requirement by major cellulolytic gut bacteria for their growth (Baldwin and Allison 1983). There is no available report about the effect of ascorbic acid, thiamine, niacin, and riboflavin on cellulase production.
Supplementation of the medium with molasses and casein hydrolysate is found to increase enzyme production. Since the supplemented compounds are crude, such additions may persuade the necessity of certain imprecise features of the producer, resulting in enhanced enzyme production. Investigation in relation to improved cellulase production by the supplementation of molasses and casein hydrolysate till now has not been elucidated.
Commonly, surfactant alters the cell membrane permeability while facilitating the release of extracellular enzymes (Ahuja et al. 2004). In the present study, no enhancement of enzyme production was found due to the surfactant. It might have happened that the cellulase adsorbed by the cellulose form an inactive complex involved in blocking the release of the enzyme (Howell and Mangat 1978). There are reports on such nonproductive bindings (Steiner et al. 1988; Shankar and Isaiarasu 2011). Converse et al. (1988) attributed to the reduction in enzyme production by cellulase adsorption and steric inactivation of the enzyme. Such postulations help to explain the release of a higher amount of enzyme but with less activity.
The molecular weight of the purified cellulase produced by GAC 16.2 was found to be 55 kDa. This is higher than the cellulase of 45 kDa from Acinetobacter indicus KTCV2 (Karlapudi et al. 2018) and 48 kDa from Bacillus sp. (Sriariyanun et al. 2016). It was found to be lower than the cellulase of 58.6 kDa from Paenibacillus barcinonensis (Asha et al. 2012), 62 kDa from Bacillus mycoides (Balasubramanian et al. 2012), and 67 kDa from Trichoderma longibrachiatum (Pachauri et al. 2017). The most optimum pH of the enzyme produced by GAC 16.2 for its utmost activity was found at 7.0. Similar behavior of cellulase was also reported by Yin et al. (2010) and Balasubramanian et al. (2012). The purified cellulase performed best at 50 °C, and such character is treated as an industrial enzyme. Cellulase from B. mycoides S122C also exhibited its highest activity at 50 °C (Balasubramanian et al. 2012).
Metal ions can form complexes in association with proteins and other molecules related to enzymes. They may act as donors or acceptors of the electron as structural regulators (Riordan 1977). The activity of purified cellulase from GAC 16.2 is stimulated in the presence of metal ions such as Mg2+, Mn2+, and Co2+. Such stimulation in the activity has also been reported by Asha et al. (2012) that the support of Co2+ on the activity of cellulase has been documented by Yin et al. (2010). Inhibition in the cellulase activity of GAC 16.2 by Ni2+ and Hg2+ metal ions also substantiates with the findings of Yin et al. (2010). The inhibitors, mainly EDTA, IAA, and PMSF, display a significant inhibition in the enzyme activity which is related to the observation of Yin et al. (2010).
Insect gut symbionts, the less explored natural resources, may be the true pathfinders to meet the tremendous demand for biotechnological tools, the enzymes. The cellulase produced by the selected isolate GAC 16.2 can efficiently degrade the cellulosic cotton fiber and sawdust within a reasonably short duration. The detailed study of cellulase production by Acinetobacter is not available. Though, Selvam et al. (2014) optimized the production of cellulase from alkali-treated coffee pulp and pineapple waste using Acinetobacter sp. TSK-MASC at solid-state fermentation.
In conclusion, the isolate Acinetobacter junii GAC 16.2 has been found as a potent cellulase producer. The isolate could enhance cellulase production by 400-fold at standardized fermentation conditions. The enzyme may be considered as an effective tool for industrial degradation of the cellulosic waste material particularly cotton fiber and sawdust. However, the isolate has the potentiality to act as an efficient agent for environmental monitoring through organic waste management. Moreover, this study reveals the scope for utilization of the potential microbial wealth of the gut of G. africana in biotechnological applications.
Availability of data and materials
All data generated or analyzed during this study are included in this article.
References
Aarti C, Khusro A, Agastian P (2018) Carboxymethyl cellulase production optimization from Glutamicibacter arilaitensis strain ALA4 and its application in lignocellulosic waste biomass saccharification. Prep Biochem Biotechnol 48:853–866. https://doi.org/10.1080/10826068.2018.1514513
Ahuja SK, Ferreira GM, Moreira AR (2004) Production of an endoglucanase by the shipworm bacterium Teredinobacter turnirae. J Ind Microbiol Biotechnol 31:41–47. https://doi.org/10.1007/s10295-004-0113-1
Aiba S, Humphrey AE, Millis NF (1973) Biochemical Engineering. Academic Press, New York
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nuc Acids Res 25:3389–3402. https://doi.org/10.1093/nar/25.17.3389
Asha BM, Revathi M, Yadav A, Sakthivel N (2012) Purification and characterization of a thermophilic cellulase from a novel cellulolytic strain, Paenibacillus barcinonensis. J Microbiol Biotechnol 22:1501–1509. https://doi.org/10.4014/jmb.1202.02013
Bai S, Ravi KM, Mukesh KDJ, Balashanmugam P, Balakumaran MD, Kalaichelvan PT (2012) Cellulase production by Bacillus subtilis isolated from cow dung. Archives Appl Sci Res 4:269–279
Balasubramanian N, Toubarro D, Teixeira M, Simos N (2012) Purification and biochemical characterization of a novel thermo-stable carboxymethyl cellulase from azorean isolate Bacillus mycoides S122. Appl Biochem Biotechnol 168:2191–2204. https://doi.org/10.1007/s12010-012-9929-8
Baldwin RL, Allison MJ (1983) Rumen metabolism. J Anim Sci 57:461–477. https://doi.org/10.2527/animalsci1983.57Supplement_2461x
Banerjee S, Maiti TK, Roy RN (2016) Identification and product optimization of amylolytic Rhodococcus opacus GAA 31.1 isolated from gut of Gryllotalpa africana 14:133-141. https://doi.org/10.1016/j.jgeb.2016.05.005
Banerjee S, Maiti TK, Roy RN (2017) Protease production by thermo-alkaliphilic novel gut isolate Kitasatospora cheerisanensis GAP 12.4 from Gryllotalpa africana. Biocatal Biotransformation 35:168–176. https://doi.org/10.1080/10242422.2017.1306739
Brock TD, Madigan MT (1988) Biology of microorganisms. Prentice-Hall, New Jersey
Converse AO, Matsuno R, Tanaka M, Taniguchi M (1988) A model of enzyme adsorption and hydrolysis of microcrystalline cellulose with slow deactivation of the adsorbed enzyme. Biotechnol Bioeng 32:38–45. https://doi.org/10.1002/bit.260320107
Coral G, Arikan B, Unaldi MN, Guvenmes H (2002) Some properties of crude carboxymethyl cellulase of Aspergillus niger Z10 wild-type strain. Turkish J Biol 26:209–213
Denison DA, Koehn RD (1977) Cellulase activity of Poronia oedipus. Mycologia 69:592–603
Dharne M, Patole M, Shouche YS (2006) Microbiology of the insect gut: tales from mosquito and bees. J Biosci 6:293–295. https://doi.org/10.1007/BF02704100
Dhayal D, Parasher H, Sharma A, Kumar P, Adak T, Jaiwal R (2014) Diversity of culturable midgut bacteria of indian malarial vector Anopheles stephensi. J Int Acad Res Multidiscipl 2:305–311
Dietz A, Mathews J (1969) Scanning electron microscopy of selected members of the Streptomyces hygroscopicus Group. J Appl Micribiol 18:694–696
Dillon RJ, Dillon VM (2004) The gut bacteria of insects: nonpathogenic interactions. Ann Rev Entomol 49:71–92. https://doi.org/10.1146/annurev.ento.49.061802.123416
Fang X, Yano S, Inoue H, Sawayama S (2008) Lactose enhances cellulase production by the filamentous fungus Acremonium cellulolyticus. J Biosci Bioeng 106:115–120. https://doi.org/10.1263/jbb.106.115
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Garrity G (2005) Bergey’s manual of systematic bacteriology vol: 2 The proteobacteria. Springer, New York
Geiger A, Fardeau ML, Grebaut P, Vatunga G, Josénando T, Herder S, Cuny G, Truc P, Ollivier B (2009) First isolation of Enterobacter, Enterococcus, and Acinetobacter spp. as inhabitants of the tsetse fly (Glossina palpalis palpalis) midgut. Infect Genet Evol 9:1364–1370. https://doi.org/10.1016/j.meegid.2009.09.013
Ghosh D, Jana BB, Lahiri S, Bhakta J, Bhattacharjee A (2018) Assessing the cellulase enzyme heterogeneity of bacterial strains and their feedback to cattle manure degradation in a greenhouse model of in vivo pond ecosystem. Environ Monit Assess 190:452. https://doi.org/10.1007/s10661-018-6821-1
Heinrichs EA, Barrion AT (2004) Rice-feeding insects and selected natural enemies in West Africa: biology, ecology, identification. International Rice Research Institute and Abidjan, Los Baños
Howell JA, Mangat M (1978) Enzyme deactivation during cellulose hydrolysis. Biotechnol Bioeng 20:847–863. https://doi.org/10.1002/bit.260200607
Hrenovic J, Durn G, Goic-Barisic I, Kovacic A (2014) Occurrence of an environmental Acinetobacter baumannii strain similar to a clinical isolate in paleosol from Croatia. Appl Environ Microbiol 80:2860–2286. https://doi.org/10.1128/AEM.00312-14
Hrenovic J, Goic-Barisic I, Kazazic S, Kovacic A, Ganjto M, Tonkic M (2016) Carbapenem-resistant isolates of Acinetobacter baumannii in a municipal wastewater treatment plant, Croatia, 2014. Euro Surveill 21:pii = 30195. https://doi.org/10.2807/1560-7917.ES.2016.21.15.30195
Ilmen M, Saloheimo A, Onnela ML, Penttila ME (1997) Regulation of cellulase gene expression in the filamentous fungus Trichoderma reesei. Appl Environ Microbiol 63:1298–1306
Immanuel G, Dhanusha R, Prema P, Palavesam A (2006) Effect of different growth parameters on endoglucanase enzyme activity by bacteria isolated from coir retting effluents of estuarine environment. Int J Environ Sci Technol 3:25–34. https://doi.org/10.1007/BF03325904
Irshad M, Anwar Z, But HI, Afroz A, Ikram N, Rashid U (2013) The industrial applicability of purified cellulase complex indigenously produced by Trichoderma viride through solid-state bio-processing of agro industrial and municipal paper wastes. BioResources 8:145–157.
James B, Atcha-Ahowé C, Godonou I, Baimey H, Goergen H, Sikirou R, Toko M (2010) Integrated pest management in vegetable production: a guide for extension workers in West Africa, Ibadan Oyo State.
Kar N, Roy RN, Sen SK, Ghosh K (2008) Isolation and characterization of extracellular enzyme producing Bacilli in the digestive tracts of rohu, Labeo rohita (hamilton) and Murrel Channa punctatus (Bloch). Asian Fish Sci 21:421–434
Karlapudi AP, Venkateswarulu TC, Srirama K, Dirisala VR, Kamarajugadda BP, Kota RK, Kodali VP (2018) Purification and lignocellulolytic potential of cellulase from newly isolated Acinetobacter indicus KTCV2 strain. Iran J Sci Technol Trans Sci 43:755–761. https://doi.org/10.1007/s40995-018-0600-2
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/BF01731581
Klemm D, Schmauder HP, Heinze T (2005) Biopolymers. In: Vandamme E, Beats S, Steinb Chel A (eds) Cellulose. Wiley, Weinheim. https://doi.org/10.1002/3527600035.bpol6010
Kocher G, Karla M, Banta G (2008) Optimization of cellulase production by submerged fermentation of rice straw by Trichoderma harzianum Rut-C 8230. Internet J Micbiol 5:1–5
Konar A, More KA, Mondal P (2005) Integrated management of mole cricket attacking potato in eastern Gangetic plains of West Bengal. J Plant Prot Sci 3:37–42
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of the bacteriophage T4. Nature 227:680–685
Lo YC, Lu WC, Chen CY, Chen WM, Chang JS (2010) Characterization and high-level production of xylanase from an indigenous cellulolytic bacterium Acinetobacter junii F6-02 from southern Taiwan soil. Biochem Eng J 53:77–84. https://doi.org/10.1016/j.bej.2010.09.011
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Malhotra J, Anand S, Jindal S, Rajagopal R, Lal R (2012) Acinetobacter indicus sp. nov., isolated from a hexachlorocyclohexane dump site. Int J Syst Evol Micribiol 62:2883–2890. https://doi.org/10.1099/ijs.0.037721-0
Miller GL (1959) Use of dinitrosaiicyiic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Moran NA, Telang A (1998) Bacteriocyte-associated symbionts of insects. Bio Science 48:295–304. https://doi.org/10.2307/1313356
Morey A, Oliveira ACM, Himelbloom BH (2013) Cellular fatty acid analysis for seafood bacteria identification. J Biol Life Sci 4:139–153. https://doi.org/10.5296/jbls.v4i2.3272
Nakajo K, Iwami Y, Komori R, Ishikawa S, Ueno T, Suzuki Y, Takahashi N (2006) The resistance to acidic and alkaline environments of endodontic pathogen Enterococcus faecalis. Oral Microbiol Immunol 21:283–288. https://doi.org/10.1111/j.1399-302X.2006.00289.x
Octave S, Thomas D (2009) Biorefinery: Toward an industrial metabolism. Biochimie 91:659–664. https://doi.org/10.1016/j.biochi.2009.03.015
Omojasola PF, Jilani OP (2009) Cellulase production by Trichoderma longi, Aspergillus niger and Saccharomyces cerevisae cultured on plantain peel. Res J Microbiol 4:67–74. https://doi.org/10.3923/JM.2009.67.74
Pa’e, N., Hui, C.C. and Muhamad, I.I (2007) Shaken culture fermentation for production of microbial cellulose from pineapple waste. International conference on waste to wealth, Malaysia.
Pachauri P, Aranganathan V, Sunil M, Sullia SB, Sudha D (2017) Purification and characterization of cellulase from a novel isolate of Trichoderma longibrachiatum. Biofuels:1–7. https://doi.org/10.1080/17597269.2017.1345357
Pandey A, Laorroche C, Ricke SC, Dussap C, Gnangsounou E (2011) Biofuels: alternative feed stocks and conversion process. Academic Press, New York
Rainey FA, Ward-Rainey N, Kroppenstedt RM, Stackebrandt E (1996) The genus Norcardiopsis represents a phylogenetically coherent taxon and a distinct actinomycete lineage: proposal of Norcardiopsaceae fam. Nov. Int J Syst Bacteriol 46:1088–1092. https://doi.org/10.1099/00207713-46-4-1088
Rajoka MI (2004) Influence of various fermentation variables on exo-glucanase production in Cellulomonas flavigena. Electron J Biotechnol 7:256–263. https://doi.org/10.2225/vol7-issue3-fulltext-2
Rastogi G, Muppidi LG, Gurram NR (2009) Isolation and characterization of cellulose-degrading bacteria from the deep subsurface of the Homestake gold mine, lead, South Dakota, USA. J Ind Microbiol Biot 36:585–598. https://doi.org/10.1007/s10295-009-0528-9
Riordan JF (1977) The role of metals in enzyme activity. Ann Clin Lab Sci 7:119–129
Roy RN, Sen SK (2013) Fermentation studies for the production of dibutyl phthalate, an ester bioactive compound from Streptomyces albidoflavus MTCC 3662 using low-priced substrates. Jordan J Biol Sci 6:177–181
Russell JB, Muck RE, Weimer PJ (2009) Quantitative analysis of cellulose degradation and growth of cellulolytic bacteria in the rumen. FEMS Microbiol Ecol 67:183–197. https://doi.org/10.1111/j.1574-6941.2008.00633.x
Sadhu S, Saha P, Mayilraj S, Maiti TK (2011) Lactose-enhanced cellulase production by Microbacterium sp. isolated from fecal matter of zebra (Equus zebra). Curr Microbiol 62:1050–1055. https://doi.org/10.1007/s00284-010-9816-x
Saha S, Roy RN, Sen SK, Roy AK (2006) Characterization of cellulase producing bacteria from the digestive tract of tilapia, Oreochromis mossambica (Peters) and grass carp, Ctenopharyngodon idella (Valenciennes). Aquacult Res 37:380–388. https://doi.org/10.1111/j.1365-2109.2006.01442.x
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Sand M, de Berardinis V, Mingote A, Santos H, Gottig S, Muller V, Averhoff B (2011) Salt adaptation in Acinetobacter baylyi: identification and characterization of a secondary glycine betaine transporter. Arch Microbiol 193:723–730. https://doi.org/10.1007/s00203-011-0713-x
Selvam K, Muthusamy G, Seralathan K-K, Govindharaju M, Senthilkuma B, Selvankumar T (2014) Process optimization of cellulase production from alkali-treated coffee pulp and pineapple waste using Acinetobacter sp. TSK-MASC 2. RSC Advances 4:10345–10351. https://doi.org/10.1039/C4RA00066H
Shankar T, Isaiarasu L (2011) Cellulase production by Bacillus pumilus EWBCM1 under varying cultural conditions. Middle-East J Sci Res 8:40–45
Singh VK, Kumar A (1998) Production and purification of an extracellular cellulase from Bacillus brevis VS-1. Biochem Mol Biol Int 45:443–452
Society of American Microbiologists (1957) Manual of Microbiological Methods. New York.
Sreena CP, Sebastian D (2018) Augmented cellulase production by Bacillus subtilis strain MU S1 using different statistical experimental designs. J Genet Eng Biotechnol 16:09–16. https://doi.org/10.1016/j.jgeb.2017.12.005
Sreena CP, Vimal KP, Sebastian D (2016) Production of cellulases and xylanase from Bacillus subtilis MU S1 isolated from protected areas of Munnar wildlife division. J Microbiol Biotechnol Food Sci 5:500–504. https://doi.org/10.15414/jmbfs.2016.5.6.500-504
Sriariyanun M, Tantayotai P, Yasurin P, Pornwongthong P, Cheenkachorn K (2016) Production, purification and characterization of an ionic liquid tolerant cellulase from Bacillus sp. isolated from rice paddy field soil. Electron J Biotechnol 19:23–28. https://doi.org/10.1016/j.ejbt.2015.11.002
Steiner W, Sattler W, Esterbauer H (1988) Adsorption of Trichoderma reesei cellulase on cellulose: experimental data and their analysis by different equations. Biotechnol Bioeng 32:853–865. https://doi.org/10.1002/bit.260320703
Sukumaran RK, Singhania RR, Pandey A (2005) Microbial cellulases-production, application and challenges. J Sci Ind Res 64:832–844
Sun P, Gao JL, Mao XJ, Zhao XH, Sun JG, Mengzhu LUM (2016) Lentibacillus populi sp. nov. a moderately halophilic, endophytic bacterium isolated from a poplar tree, and emended description of the genus Lentibacillus. Int J Syst Evol Microbiol 66:5281–5287. https://doi.org/10.1099/ijsem.0.001508
Teather RM, Wood PJ (1982) Use of Congo red-polysaccharide interactions in enumeration and characterization of cellulolytic bacteria from the bovine rumen. Appl Environ Microbiol 43:777–780
Thomas L, Ram H, Singh VP (2018) Inducible cellulase production from an organic solvent tolerant Bacillus sp. SV1 and evolutionary divergence of endoglucanase in different species of the genus Bacillus. Braz J Microbiol 49:429–442. https://doi.org/10.1016/j.bjm.2017.05.010
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882. https://doi.org/10.1093/nar/25.24.4876
Vilanova C, Marco G, Domı´nguez-Escriba L, Genove´s S, Sentandreu V, Bataller E, Ramo´n D, Porcar M (2012) Bacteria from acidic to strongly alkaline insect midguts: potential sources of extreme cellulolytic enzymes. Biomass Bioenerg 45:288–294. https://doi.org/10.1016/j.biombioe.2012.06.017
Wanderley KJ, Torres FAG, Moraes LMP, Ulhoa CJ (2004) Biochemical characterization of a-amylase from the yeast Cryptococcus flavus. FEMS Microbiol Lett 231:165–169. https://doi.org/10.1016/S0378-1097(03)00955-8
Watanabe H, Tokuda G (2010) Cellulolytic systems in insects. Annu Rev Entomol 55:609–632. https://doi.org/10.1146/annurev-ento-112408-085319
Yin LJ, Huang PS, Lin HH (2010) Isolation of cellulase-producing bacteria and characterization of the cellulase from the isolated bacterium Cellulomonas sp. YJ5. J Agric Food Chem 58:9833–9837. https://doi.org/10.1021/jf1019104
Yoon, JH, Kim IG, OH TKT (2007) Acinetobacter marinus sp. nov. and Acinetobacter seohaensis sp. nov., isolated from sea water of the yellow sea in Korea. J Microbiol Biotechnol 17: 1743-1750.
Younis MAM, Hezayen FF, Nour-Eldein MA, Shabeb MSA (2010) Optimization of cultivation medium and growth conditions for Bacillus subtilis KO strain isolated from sugar cane molasses. Am-Euras J Agric Environ Sci 7:31–37
Zhang HY, Jackson TA (2008) Autochthonous bacterial flora indicated by PCR-DGGE of 16S rRNA gene fragments from the alimentary tract of Costelytra zealandic (Coleoptera: Scarabaeidae). J Appl Microbiol 105:1277–1285. https://doi.org/10.1111/j.1365-2672.2008.03867.x
Acknowledgements
The authors S Banerjee and R N Roy thankfully acknowledge the funding support received from the University Grants Commission (F.No. 39-391/2010 SR), India. The authors also thankfully acknowledge the academic support of Prof. (Dr) Abhijit Mazumdar, Entomology research laboratory, Department of Zoology, The University of Burdwan, Rajbati, West Bengal, India.
Funding
This work was supported by the University Grants Commission, India under Grant (F.No. 39-391/2010 SR).
Author information
Authors and Affiliations
Contributions
SB performed and analyzed the experimental research work and primarily drafted the manuscript. RNR designed and supervised this work and also configured the manuscript. TKM helped to finalize the manuscript. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Banerjee, S., Maiti, T.K. & Roy, R.N. Production, purification, and characterization of cellulase from Acinetobacter junii GAC 16.2, a novel cellulolytic gut isolate of Gryllotalpa africana, and its effects on cotton fiber and sawdust. Ann Microbiol 70, 28 (2020). https://doi.org/10.1186/s13213-020-01569-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13213-020-01569-6