Abstract
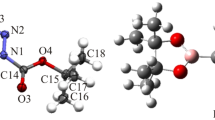
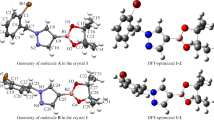
The title compound, C21H29NS2, has been synthesized and its crystal structure has been determined from single crystal X-ray diffraction data. Crystals are monoclinic, a = 11.4923(8), b = 13.1842(7), c = 14.6583(8) Å, β = 109.983(6)°, sp. gr. P21/c, Z = 4. Mesityl and thiazole groups are in cis positions with respect to the cyclobutane ring. The cyclobutane ring is puckered, with a dihedral angle of 26.6(2)° between the two three-atom planes. The crystal structure involves one weak intermolecular C–H···S hydrogen-bond. The molecular geometry was also optimized using density functional theory (DFT/B3LYP) method with the 6-311G(d, p) basis set in ground state. Geometric parameters (bond lengths, bond angles and torsion angles) and vibrational assignments have been calculated theoretically and compared with the experimental data.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Ahmedzade, C. Kirilmis, A. Cukurovali, et al., S. Afr. J. Chem. 56, 21 (2003).
J. Safaei-Ghomi, F. Salimi, and A. Ramazani, J. Chem. Sci. 125, 1087 (2013)
C. Kirilmis, M. Koca, C. Arici, et al., Acta Crystallogr. E 61, o555 (2005).
F. Yakuphanoglu, A. Cukurovali, and I. Yilmaz, Opt. Mater. 27, 1363 (2005).
CrysAlis Pro Software System, Version 1.171.35.11 (Agilent Technologies UK, 2011).
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, et al., J. Appl. Crystallogr. 42, 339 (2009).
G. M. Sheldrick, Acta Crystallogr. A 64, 112 (2008).
A. L. Spek, J. Appl. Crystallogr. 36, 7 (2003).
L. J. Farrugia, J. Appl. Crystallogr. 30, 565 (1997).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., Gaussian 09, revision B.01 (Gaussian, Wallingford, 2010).
A. D. Becke, Phys. Rev. A 38, 3098 (1988).
A. D. Becke, J. Chem. Phys. 98, 5648 (1993).
C. Lee, W. Yang, and R. D. Parr, Phys. Rev. B. 37, 785 (1998).
G. A. Petersson, A. Bennett, T. G. Tensfeldt, et al., J. Chem. Phys. 89, 2193 (1988).
K. K. Irikura, R. D. Johnson, and R. N. Kacker, J. Phys. Chem. A 109, 8430 (2005).
D. C. Swenson, M. Yamamoto, and D. J. Burton, Acta Crystallogr. C 53, 1445 (1997).
F. H. Allen, Acta Crystallogr. B 40, 64 (1984).
M. Dinçer, N. Özdemir, A. Çukurovali, et al., Acta Crystallogr. E 60, o1523 (2004).
F. Sen, M. Dinçer, A Çukurovali, et al., Acta Crystallogr. E 67, o958 (2011).
F. Sen, M. Dinçer and A. Cukurovali, J. Mol. Struct. 1076, 1 (2014).
M. Koca, C. Kirilmis, and C. Arici, Acta Crystallogr. E 66, o523 (2010).
C. Kirilmis, M. Koca, C. Arici, et al., Acta Crystallogr. E 61, o1176 (2005).
J. P. Foster and F. Weinhold, J. Am. Chem. Soc. 102, 7211 (1980).
A. E. Reed and F. Weinhold, J. Chem. Phys. 78, 4066 (1983).
A. E. Reed, R. B. Weinstock and F. Weinhold, J. Chem. Phys. 83, 735 (1985).
F. H. Allen, O. Kennard, D. G. Watson, et al., J. Chem. Soc. Perkin Trans. 2, S1 (1987).
J. Hartung, N. Schneiders, and Yu. Bergsträsser, Acta Crystallogr. E 62, o4713 (2006).
J. Hartung, N. Schneiders, and Yu. Bergsträsser, Acta Crystallogr. E 61, o421 (2005).
J. Hartung, M. Schwarz, E. F. Paulus, et al., Acta Crystallogr. C 62, o386 (2006).
J. Hartung, S. Altermann, I. Svoboda, et al., Acta Crystallogr. E 61, o1738 (2005).
J. Bernstein, R. E. Davis, L. Shimoni, et al., Angew. Chem. Int. Ed. Engl., 34, 1555 (1995).
T. Steiner, Crystallogr. Rev. 6, 1 (1996).
J. Hartung, M. Schwarz, I. Svoboda, et al., Acta Crystallogr. C 59, o682 (2003).
R. Dennington, II, T. Keith, and J, Millam, GaussView, Version 4.1.2 (SemichemInc, Shawnee Mission, KS, 2007).
G. Socrates, Infrared Characteristic Group Frequencies (Wiley, New York, 1980).
F. R. Dollish, W. G. Fateley, and F. F. Bentley, Characteristic Raman Frequencies of Organic Compounds (Wiley, New York, 1997).
G. Varsanyi, Vibrational Spectra of Benzene Derivatives (Academic, NewYork, 1969).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Şen, B., Barim, E., Kirilmis, C. et al. Synthesis, structural characterization, and DFT calculations of 3-buthyl-4-(3-methyl-3-mesitylcyclobut-1-yl)-1,3-thiazole-2(3H)-thione. Crystallogr. Rep. 61, 243–248 (2016). https://doi.org/10.1134/S1063774516020188
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063774516020188