Abstract
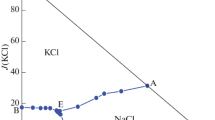
The phase equilibria in the system NaBr–KBr–CaBr2–H2O at 323 K were studied using the isothermal dissolution equilibrium method. Using the experimental solubilities of salts data, phase diagram was constructed. The phase diagram have two invariant points, five univariant curves, and four crystallization fields. The equilibrium solid phases in the system are NaBr, NaBr · 2H2O, KBr, and CaBr2 · 4H2O. The solubilities of salts in the system at 323 K were calculated by Pitzer’s equation. There is shown that the calculated solubilities agree well with experimental data.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Y. T. Lin, J. Salt Lake Res. 9, 56 (2001).
Y. T. Lin, J. Salt Lake Res. 14, 1 (2006).
S. H. Sang, H. A. Yin, S. J. Ni, and C. J. Zhang, J. Chengdu Univ. Tech. (Sci. Technol. Ed., China) 33, 414 (2006).
X. Y. Zhao, S. H. Sang, and M. L. Sun, J. Salt Lake Res. 19, 35 (2011).
S. H. Sang, T. R. Li, and Z. Cui, J. Salt Lake Res. 12, 29 (2013).
R. Z. Cui, S. H. Sang, Y. X. Hu, and J. W. Hu, Acta Geol. Sin. 87, 1668 (2014).
Z. L. Zhang, S. H. Sang, M. Li, and C. H. Hou, Chem. Eng. (China) 37, 45 (2009).
F. M. Hu, S. H. Sang, Y. G. Zhang, and J. J. Zhang, J. Salt Chem. Ind. 41, 12 (2012).
R. Z. Cui, S. H. Sang, D. W. Li, and Q. Z. Liu, CALPHAD 49, 120 (2015).
R. Z. Cui, S. H. Sang, and Q. Liu, J. Chem. Eng. Data 61, 444 (2016).
C. E. Harvie and J. H. Weare, Geochim. Cosmochim. Acta 44, 981 (1980).
C. E. Harvie, H. P. Eugster, and J. H. Weare, Geochim. Cosmochim. Acta 46, 1603 (1982).
C. E. Harvie, N. Moller, and J. H. Weare, Geochim. Cosmochim. Acta 48, 723 (1984).
N. Moller, Geochim. Cosmochim. Acta 52, 821 (1988).
J. P. Greenberg and N. Moller, Geochim. Cosmochim. Acta 53, 2503 (1989).
C. Christov and N. Moller, Geochim. Cosmochim. Acta 68, 1309 (2004).
C. Christov and N. Moller, Geochim. Cosmochim. Acta 68, 3717 (2004).
R. J. Speneer, N. J. Moller, and H. Weare, Geochim. Cosmochim. Acta 54, 575 (1990).
C. Balarew and C. Christov, C.R. Acad. Bulg. Sci. 45, 49 (1992).
C. Balarew, C. Christov, S. Petrenko, and V. Valyashko, J. Solution Chem. 22, 173 (1993).
C. Christov, C. Balarew, V. Valyashko, and S. Petrenko, J. Solution Chem. 23, 595 (1994).
C. Christov, S. Petrenko, C. Balarew, and V. Valyashko, Monatsh. Chem. 125, 1371 (1994).
C. Christov, J. Chem. Thermodyn. 27, 1267 (1995).
C. Christov and C. Balarew, J. Solution Chem. 24, 1171 (1995).
C. Christov, CALPHAD 20, 501 (1996).
C. Christov, Coll. Czech. Chem. Commun. 61, 1585 (1996).
C. Christov, J. Chem. Thermodyn. 37, 1036 (2005).
C. Christov, S. Velikova, and K. Ivanova, J. Chem. Thermodyn. 32, 1505 (2000).
C. Christov, Geochim. Cosmochim. Acta 71, 3557 (2007).
C. Christov, CALPHAD 35, 42 (2011).
C. Christov, J. Chem. Thermodyn. 43, 344 (2011).
C. Christov, CALPHAD 36, 71 (2012).
C. Christov, J. Chem. Thermodyn. 47, 335 (2012).
C. Christov, J. Chem. Thermodyn. 55, 7 (2012).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cui, RZ., Wang, W., Yang, L. et al. Phase Diagram of Quaternary System NaBr–KBr–CaBr2–H2O at 323 K. Russ. J. Phys. Chem. 92, 475–481 (2018). https://doi.org/10.1134/S003602441803024X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003602441803024X