Abstract
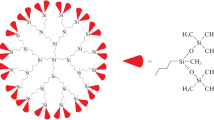
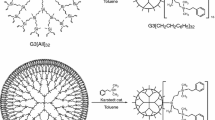
The heat capacities of first- and third-generation carbosilane dendrimers with terminal phenyldioxolane groups are studied as a function of temperature via vacuum and differential scanning calorimetry in the range of 6 to 520 K. Physical transformations that occur in the above temperature range are detected and their standard thermodynamic characteristics are determined and analyzed. Standard thermodynamic functions Cpο(T), [H°(T) − H°(0)], [S°(T) − S°(0)], and [G°(T) − H°(0)] in the temperature range of T → 0 to 520 K for different physical states and the standard entropies of formation of the studied dendrimers at T = 298.15 K are calculated, based on the obtained experimental data.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
D. A. Tomalia, A. M. Naylor, and W. A. Goddard, Angew. Chem., Int. Ed. Engl. 29, 138 (1990).
A. W. Bosman, H. M. Janssen, and E. W. Meijer, Chem. Rev. 99, 1665 (1999).
S. A. Ponomarenko, E. A. Tatarinova, A. M. Muzafarov, et al., Chem. Mater. 18, 4101 (2006).
A. M. Muzafarov, N. G. Vasilenko, E. A. Tatarinova, G. M. Ignat’eva, V. M. Myakushev, M. A. Obrezkova, I. B. Meshkov, N. V. Voronina, and O. V. Novozhilov, Polymer Sci., Ser. C 53, 48 (2011).
S. Svenson and D. A. Tomalia, Adv. Drug Deliv. Rev. 57, 2106 (2005).
R. K. Tekade, P. V. Kumar, and N. K. Jain, Chem. Rev. 109, 49 (2009).
A. I. Kuklin, A. N. Ozerin, A. Kh. Islamov, et al., J. Appl. Crystallogr. 36, 679 (2003).
B. V. Lebedev, M. V. Ryabkov, E. A. Tatarinova, E. A. Rebrov, and A. M. Muzafarov, Russ. Chem. Bull. 52, 545 (2003).
N. N. Smirnova, O. V. Stepanova, T. A. Bykova, et al., Thermochim. Acta 440, 188 (2006).
A. S. Tereshchenko, G. S. Tupitsyna, E. A. Tatarinova, A. V. Bystrova, A. M. Muzafarov, N. N. Smirnova, and A. V. Markin, Polymer Sci., Ser. B 52, 41 (2010).
A. V. Markin, Ya. S. Samosudova, N. N. Smirnova, et al., J. Therm. Anal. Calorim. 105, 663 (2011).
A. V. Markin, S. S. Sologubov, N. N. Smirnova, et al., Thermochim. Acta 617, 144 (2015).
S. S. Sologubov, A. V. Markin, N. N. Smirnova, et al., J. Phys. Chem. B 119, 14527 (2015).
S. S. Sologubov, A. V. Markin, N. N. Smirnova, et al., J. Therm. Anal. Calorim. 125, 595 (2016).
Ya. S. Samosudova, A. V. Markin, N. N. Smirnova, et al., J. Chem. Thermodyn. 98, 33 (2016).
N. A. Novozhilova, Cand. Sci. (Chem.) Dissertation (Enikolopov Inst. Synth. Polym. Mater. RAS, Moscow, 2013).
R. M. Varushchenko, A. I. Druzhinina, and E. L. Sorkin, J. Chem. Thermodyn. 29, 623 (1997).
V. M. Malyshev, G. A. Mil’ner, E. L. Sorkin, and V. F. Shibakin, Prib. Tekh. Eksp., No. 6, 195 (1985).
G. W. H. Höhne, W. F. Hemminger, and H. J. Flammersheim, Differential Scanning Calorimetry (Springer, Berlin, 2003).
V. A. Drebushchak, J. Therm. Anal. Calorim. 79, 213 (2005).
M. E. Wieser, N. Holden, T. B. Coplen, et al., Pure Appl. Chem. 85, 1047 (2013).
A. V. Markin, Doctoral (Chem.) Dissertation (Lobachevsky Nizhegor. State Univ., Nizh. Novgorod, 2013).
Solid State Physics and Chemistry of Organic Compounds, Ed. by Yu. A. Pentin (Mir, Moscow, 1967) [in Russian].
B. V. Lebedev, Thermochim. Acta 297, 143 (1997).
S. Alford and M. Dole, J. Am. Chem. Soc. 77, 4774 (1955).
G. Adam and J. H. Gibbs, J. Chem. Phys. 43, 139 (1965).
W. Kauzmann, Chem. Rev. 43, 219 (1948).
B. V. Lebedev and I. B. Rabinovich, Dokl. Akad. Nauk SSSR 237, 641 (1977).
J. P. McCullough and D. W. Scott, Calorimetry of Non-Reacting Systems (Butterworth, London, 1968).
J. D. Cox, D. D. Wagman, and V. A. Medvedev, CODATA Key Values for Thermodynamics (Hemisphere, New York, 1989).
M. W. Chase, Jr., J. Phys. Chem. Ref. Data, Monograph 9 1–2, 1 (1998).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.N. Smirnova, S.S. Sologubov, Yu.A. Sarmini, A.V. Markin, N.A. Novozhilova, E.A. Tatarinova, A.M. Muzafarov, 2017, published in Zhurnal Fizicheskoi Khimii, 2017, Vol. 91, No. 12, pp. 2030–2038.
Rights and permissions
About this article
Cite this article
Smirnova, N.N., Sologubov, S.S., Sarmini, Y.A. et al. Thermodynamic properties of first- and third-generation carbosilane dendrimers with terminal phenyldioxolane groups. Russ. J. Phys. Chem. 91, 2317–2325 (2017). https://doi.org/10.1134/S0036024417110279
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024417110279