Abstract
Schooling makes it easier for fish to find food objects, increases the intensity of feeding and, as a result, improves food supply. The effect is achieved due to well-developed imitative behavior and stress reduction in schooling fish. Staying in a school increases the duration and frequency of feeding, expands the range of available food items, allows the development of new spatial and food resources, reduces the time spent on restoring feeding behavior after stressful loads due to a change in biotope, threat or attack by a predator, and in other cases. Forming a school makes it possible to overcome the resistance of territorial fish that protect food resources in their home areas. Higher feeding efficiency in a school equally affects both non-predatory and predatory schooling fish. However, being in a school can presumably also lead to increased intra-school food competition.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The main significance of schooling in the feeding behavior of fish lies, first of all, in significantly facilitating the search for prey and accelerating the rate of food consumption. The effect is achieved due to the fact that when moving in a school, it is enough that not everyone, but only a part of the fish, who are the first to start feeding, are the first to notice the patch of forage organisms. The view of feeding fish, as well as, possibly, the spreading food stimuli of other modalities, for example, chemical or acoustic, attract the rest of the school here, and soon all the fish gather in the discovered feeding area and begin to intensively feed until the availability of feeding organisms decreases below the critical level due to a decrease in the density of food objects or until the fish stop feeding for some other reason—due to the decrease in feeding motivation, the emergence of danger, changes in illumination or other abiotic conditions, etc. The concentration of all the fish in the school in the feeding area is achieved not only due to the direct attractive action of stimuli of various nature, but also due to well-developed imitative behavior in fish. The fish that are the first to find food and show a directed behavioral reaction to its source, drag their closest partners with them, and after them all the other fish of the school (Radakov, 1973; Gerasimov, 1983). It is precisely because of imitative behavior that individuals of different ages in mixed schools often do not differ from each other in terms of the probability and speed of finding feeding areas (Peuhkuri, 1998a).
Rapid detection of food objects and, as a result, a higher rate of feeding of fish in a school is achieved not only due to imitative behavior, but also due to the fact that the mere presence of partners in a school or simply individuals of their own species is already enough to increase the intensity of food consumption. Single individuals of the pristella Pristella maxillaris (=riddlei) and the European bitterling Rhodeus amarus eat much more food if they see even non-feeding individuals of their own species (Radakov and Mocheck, 1972). When setting up this experiment, the influence of imitative behavior on the intensity of food intake by single fish was completely excluded, since the fish-actors were pre-fed to saturation, and the type of food did not cause them to display any elements of feeding behavior. The presence effect, i.e., the presence of other individuals of the same species nearby or the presence of visual or other sensory contact with them is undoubtedly associated with the suppression of stress that develops in isolated schooling fish (Lebedeva et al., 2000). One of the manifestations of stress caused by this factor is a sharp increase in the intensity of respiration in schooling fish during isolation (Malyukina, 1966; Parker, 1973; Ross and Backman, 1992; Nadler et al., 2016). Being in an unfamiliar environment causes less stress, as measured by an increase in blood cortisol levels, if the fish (zebrafish Danio rerio) are in a shoal rather than in isolation (Pagnussat et al., 2013).
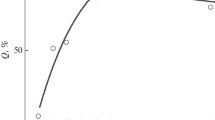
Fast detection of food, more efficient foraging, and increased feeding intensity in fish in a school compared to single fish have been experimentally demonstrated using a large number of examples. Such data were obtained for the Atlantic herring Clupea harengus, the flier Centrarchus macropterus, the three-spined stickleback Gasterosteus aculeatus, the Eurasian minnow Phoxinus phoxinus, the Arctic cisco Coregonus autumnalis, the yellow-finnes sculpin Cottocomephorus grewingkii, the flathead grey mullet Mugil cephalus, and many others (Nikol’skii and Kukushkin, 1943; Blaxter and Holliday, 1958; Parrish et al., 1958; Volkova and Kozhov, 1966; Volkova, 1969; Radakov and Mocheck, 1972; Olla and Samet, 1974; Mittelbach, 1984; Foster, 1985; Ranta and Lindström, 1990; Ryer and Olla, 1992; Steinegger et al., 2020). It has been established that, while in a school, the parrotfish Scarus rivulatus and other fish feed twice as intensively during the entire daytime as when they are outside the school (Fig. 1) (Welsh and Bellwood, 2012). Using the examples of the obligate schooling Eurasian minnow and the facultative schooling goldfish Carassius auratus, it was shown that with an increase in the school size, the speed of detection of hidden prey by fish increases (Pitcher et al., 1982), primarily due to a wider search for food objects and a greater “sensor equipment” of a large school. Using the example of the three-spined stickleback, it has been established that the greater the number of individuals in a shoal, the faster they begin to feed when transplanted into a new aquarium, the greater the percentage of feeding fish and the higher the feeding intensity—the number of food grasps per unit of time and the number of grasps made in one demonstration of the tilting upside down position, which is a specific feeding position in sticklebacks. These changes in the feeding behavior of fish occur most dynamically when the number of experimental fish increases from 1–2 to 5–10 individuals (Fig. 2) (Ranta and Kaitala, 1991). Forming small schools enabled juveniles of the pumpkinseed Lepomis gibbosus to successfully hunt cladocerans and other aquatic invertebrates (Mittelbach, 1984).
Average frequency of food grasping by the schooling ( ) and solitary (
) and solitary ( ) parrotfish Scarus rivulatus (a) and parrotfish of other species (b)—S. ghobban, S. schlegeli, S. niger, S. flavipectoralis and Hipposcarus longiceps when eating during the daytime in one of the bays of the Great Barrier Reef; (
) parrotfish Scarus rivulatus (a) and parrotfish of other species (b)—S. ghobban, S. schlegeli, S. niger, S. flavipectoralis and Hipposcarus longiceps when eating during the daytime in one of the bays of the Great Barrier Reef; ( )—mean value error (according to: Welsh and Bellwood, 2012).
)—mean value error (according to: Welsh and Bellwood, 2012).
Compared to solitary individuals, staying in a school gives fish the opportunity to develop a larger number of feeding points, which was demonstrated in experiments on the walleye pollock Gadus chalcogrammus (= Theragra chalcogramma) (Fig. 3) (Baird et al., 1991). Of interest are the observations made on the behavior of the Barrier Reef chromis Chromis nitida, a small schooling plankton-eating fish that stays near coral colonies and hides in them during daylight hours in case of danger or at night for rest. To feed, these fish leave their shelters and move away from them for a short distance into the open ocean, where the conditions for feeding are much better, but the threat of being attacked by predators is also higher. It was found that the greater the distance at which a school moves away from the edge of the coral colony, the higher the number of the school and the larger the fish that make up it (Fig. 4). In turn, in larger schools, fish feeding is more intense. In other words, the development of dangerous, but profitable areas of the reservoir by schooling fish is achieved only if the level of protection (safety) of fish, their ability to resist predators, is simultaneously increased. Either this is achieved due to specific protective mechanisms of schooling behavior, the effectiveness of which is proportional to the size of a school, or due to better locomotor abilities in large fish, which are able to move faster to shelters (Sackley and Kaufman, 1996). During school feeding, food resources are more fully assimilated, the range of consumed objects is expanded (Ranta and Lindström, 1990), which contributes to a decrease in intra-school food competition. Being in a school, especially in a large one, prolongs the feeding time of fish when a danger (predator) appears (Milinski, 1976).
Relatively recently, it was found that individuals that are part of the same school can perform different functions when searching for and obtaining food, but due to cooperation, the efficiency of achieving the desired goal by fish is significantly increased. Long-term tracking of the behavior of individuals of the gold-saddle goatfish Parupeneus cyclostomus, living among coral reefs, found that some individuals in a shoal perform the function of beaters, chasing and forcing moving prey to hide in shelters among corals. Other fish from the same shoal surround such a shelter and, exploring it, find and seize the hidden prey. After some time, they themselves become beaters, providing other individuals of their shoal an opportunity to feed, i.e., there is a constant rotation of the functions performed by partners in the school. In the population of these fish, there are also individuals who prefer to stay alone and feed, looking for sedentary prey hidden in the ground (Strübin et al., 2011).
With an increase in the size of a school, the time spent by fish on the feeding spot increases progressively (Fig. 5), and the proportion of longer visits to the feeding area increases (Fig. 6). Thus, in schools consisting of 20 individuals, the proportion of visits by fish to a feeding area lasting ≥1 min was 25% for goldfish and 15% for Eurasian minnow, while in the smallest shoals consisting of only 2 fish, 74% of visits in goldfish and 88% in minnow had a duration <10 s (Magurran and Pitcher, 1983). It is possible that this effect is facilitated not only by the faster detection by a school of a patch of forage organisms, but also by the increased skittishness of individuals or fish in small schools and their slow adaptation to a new environment, the desire to stay longer in shelters and leave them less often. Fish, being in a larger school, more easily overcome the protective effect of dense swarms of planktonic crustaceans. For example, the blue-green chromis Ch. viridis feeds on such prey the faster the larger its school (Smith and Warburton, 1992).
Frequency distribution of the duration of visits by the Eurasian minnow Phoxinus phoxinus to feeding areas (a–e) and shelters (g–j) during 8 min of observations depending on the number of individuals in a school, ind.: a, f—2; b, g—4; c, h—6; d, i—12; e, j—20 (according to: Magurran and Pitcher, 1983).
Fish in small schools are characterized by frequent bursts and sharp turns to the sides, which indicates their restlessness (Fig. 7). High schooling fish, for example, Atlantic herring, when kept alone, stop feeding and may die from stress (Gerasimov, 1962). Under artificial conditions, single individuals of the yellowfin Baikal sculpin do not feed, while in a shoal they actively consume the food offered to them (Volkova, 1969). Another explanation for more efficient feeding in a school may be mutual stimulation of each other by fish. Thus, the visual perception of a feeding individual has a stronger attracting effect on the three-spined stickleback than the presentation of each of the stimuli separately—the appearance of food or individuals of its own species (Keenleyside, 1955). The sight of feeding conspecifics leads the high schooling Atlantic herring to such a strong excitation that individuals that are far from food reflexively make grasping movements (Gerasimov, 1962, 1983).
An interesting behavior called hiding behavior is shown by schooling fish in an unfamiliar environment or at wariness caused by other reasons (Milanovskii and Rekubratskii, 1960). It consists in the fact that one or more fish separate from the school, located at some distance from the food, which quickly approach and grasp the food and immediately return to the school. Group hiding can also take place, i.e., the whole school quickly makes a burst to food and moves away. Such behavior is interpreted as a manifestation of food excitation (Rekubratskii, 1967).
In some cases, the development of food resources by fish is facilitated by the formation of schools that are heterogeneous in terms of species. Thus, joint schools of the midnight parrotfish S. coelestinus and the blue tang surgeonfish Acanthurus coeruleus feed more successfully on young shoots of algae in areas of the reef protected by the territorial threespot damselfish Stegastes (=Eupomocentrus) planifrons, which is able to defend its area from single individuals, but is not able to successfully resist a fish school (Alevizon, 1976). This feature of school feeding of fish is well illustrated by the example of the herbivorous parrotfish S. iseri (=croicensis), in which some individuals lead a territorial lifestyle and constantly stay on their individual feeding area, while others move over a rather large territory and often unite. The ratio of “stray” schooling and territorial individuals of the parrotfish is related to the abundance of the euryphagous damselfish, which actively guards its territories and prevents other fish, including S. iseri, from feeding on them. The observations made showed that stray individuals of S. iseri are much more often attacked than schooling individuals of this species. Single individuals also have a higher food motivation. Thus, the formation of schools allows stray individuals of parrotfish, which do not have their own food area due to the high density of the damselfish, to successfully exist in conditions of high food competition. Schools of S. iseri often include other herbivorous fish, which also increases their food potential (Buckman and Ogden, 1973; Robertson et al., 1976).
Forming a school makes it possible to overcome the resistance of territorial fish, fiercely defending their territories, and other schooling fish. When a school of such fish moves along the reef, stop for feeding of even a few individuals exponentially involves all other fish in the food-procuring activity. This is a very effective technique for successful feeding in individual territories protected by solitary hosts, unable to restrain the simultaneous invasion of a large number of fish (Vine, 1974; Alevizon, 1976; Robertson et al., 1976; Mochek, 1987).
A theoretical model has been proposed to describe the rate of consumption of forage organisms by schooling planktophagous fish (Eggers, 1976). The model is a translation of the well-known Holling’s disk equation, which describes the rate of consumption of forage organisms (С) by single-feeding individuals: C = λN/(1 + λN × TH), where λ is the volume of the visual field (a cylinder with a radius equal to the maximum lateral visibility distance of prey), N is the density of prey, TH is the time from the beginning of the pursuit to the capture of prey. The change in the density of forage organisms during feeding of a school is described as: dNi/dt = −Ci/γ, where γ is the index of the probability of meeting the i-th individual with food, Ci is the rate of consumption of forage organisms by fish in the school in relation to the i-th individual, Ni is the density of forage organisms in the volume of the visual field of the i-th individual of the school. The expression of the average feeding values of a fish school and the density of forage organisms is achieved by approximating the model in the form of finite difference equations: Ci = γNi/(1 + γNi − 1TH) and Ni = Ni − 1 − (Ci − 1)/γ. The study of the proposed model showed that the probability of school formation decreases with high food density, large distance between individuals, and small school size. According to the model, the advantages of schooling behavior are especially pronounced with aggregated food distribution.
A schooling lifestyle helps to find and obtain food not only for non-predatory fish, but also for those predatory fish that, like predatory mammals, are able to use the so-called collective hunting tactics. This tactic allows them to overcome the resistance of small schooling fish that are prey items (Major, 1978; Potts, 1981; Landeau and Terborgh, 1986; Parrish, 1993; Hansen et al., 2022).
It should be noted that, along with the obvious advantages that arise, being together in a school can lead, as expected, to an increase in intra-school food competition between school members (Pitcher, 1986; Pitcher and Parrish, 1993). Such competition manifests itself in a reduction in the time spent by fish grasping and swallowing food objects (Street et al., 1984), or in a decrease in the frequency of hunting bursts made by predatory fish attacking prey in a group (Hansen et al., 2022). This effect becomes noticeable when the number of fish in a school reaches 10–12 individuals (Morgan and Colgan, 1987) and it is more pronounced, the more numerous the school is (Uematsu and Takamori, 1976). The intention of hungry fish to stay in smaller schools is also interpreted as one of the adaptations to a decrease in food competition between schooling partners (Van Havre and FitzGerald, 1988; Krause, 1993). It has been established that in heterogeneous schools, larger individuals, consuming the main part of the food, compete with smaller fish (Krause, 1994). If the proportion of large individuals in a school is low, their feeding activity is also reduced (Peuhkuri, 1997, 1998b). In some coral reef fish, such as the adult grunts Haemulon spp., the intensity of feeding significantly increases during solitary swimming compared to schooling: the average frequency of food grasping from the substrate increases from 2.3 to 12.5 per min (Pereira and Ferreira, 2013). Similar data were also obtained for another coral fish, the bluehead wrasse Thalassoma bifasciatum (White and Warner, 2007). It is believed that food competition is reduced in this way, while the schooling is required for these fish mainly for protection from predators. In general, the number of examples showing a decrease in the efficiency of feeding in fish forming schools is small, and they do not always concern obligate schooling species.
Thus, the association of fish in schools in most cases facilitates the detection of food objects, increases the intensity of feeding, and, as a result, increases the supply of fish with food. Schooling not only leads to a decrease in the time spent on searching and detecting food objects, but also increases the duration and efficiency of feeding, and increases the time that fish spend on getting food. As a result, fish in schools consume food more food and do it more often, restore their feeding behavior much faster after stress loads caused, for example, by a predator attack or a change in biotope. Being in a school makes fish more competitive in obtaining food, expands the range of food objects available to them, and allows them to develop new spatial and food resources. The benefits of a schooling lifestyle equally affect both non-predatory and predatory schooling fish.
REFERENCES
Alevizon, W.S., Mixed schooling and its possible significance in a tropical Western Atlantic parrotfish and surgeonfish, Copeia, 1976, vol. 1976, no. 4, pp. 796–798. https://doi.org/10.2307/1443464
Baird, T.A., Ryer, C.H., and Olla, B.L., Social enhancement of foraging on an ephemeral food source in juvenile walleye pollock, Theragra chalcogramma, Environ. Biol. Fish., 1991, vol. 31, no. 3, pp. 307–311. https://doi.org/10.1007/BF00000697
Blaxter, J.H.S. and Holliday, F.G., Herring (Clupea harengus L.) in aquaria. 2. Feeding, Mar. Res., 1958, no. 6, pp. 1–22.
Buckman, N.S. and Ogden, J.C., Territorial behavior of the striped parrotfish Scarus croicensis Bloch (Scaridae), Ecology, 1973, vol. 54, no. 6, pp. 1377–1382. https://doi.org/10.2307/1934202
Eggers, D.M., Theoretical effect of schooling by planktivorous fish predators on rate of prey consumption, J. Fish. Res. Board Can., 1976, vol. 33, no. 9, pp. 1964–1971. https://doi.org/10.1139/f76-250
Foster, S.A., Group foraging by a coral reef fish: A mechanism for gaining access to defended resources, Anim. Behav., 1985, vol. 33, no. 3, pp. 782–792. https://doi.org/10.1016/S0003-3472(85)80011-7
Gerasimov, V.V., Feeding behavior of the Murmansk herring in and out of the school under aquatic conditions, Tr. Murmansk. Morsk. Biol. Inst. Akad. Nauk SSSR, 1962, no. 4, pp. 254–259
Gerasimov, V.V., Ekologo-fiziologicheskie zakonomernosti stainogo povedeniya ryb (Ecological and Physiological Patterns of Schooling Behavior of Fish) Moscow: Nauka, 1983.
Hansen, M.J., Krause, S., Dhellemmes, H., et al., Mechanisms of prey division in striped marlin, a marine group hunting predator, Commun. Biol., 2022, vol. 5, Article 1161. https://doi.org/10.1038/s42003-022-03951-3
Keenleyside, M.H.A., Some aspects of the schooling behaviour of fish, Behaviour, 1955, vol. 8, no. 1, pp. 183–247. https://doi.org/10.1163/156853955X00229
Krause, J., The influence of hunger on shoal size choice by three-spined sticklebacks, Gasterosteus aculeatus, J. Fish. Biol., 1993, vol. 43, no. 5, pp. 775–780. https://doi.org/10.1111/j.1095-8649.1993.tb01154.x
Krause, J., The influence of food competition and predation risk on size-assortative shoaling in juvenile chub (Leuciscus cephalus), Ethology, 1994, vol. 96, no. 2, pp. 105–116. https://doi.org/10.1111/j.1439-0310.1994.tb00886.x
Landeau, L. and Terborgh, J., Oddity and confusion effect in predation, Anim. Behav., 1986, vol. 34, no. 5, pp. 1372–1380. https://doi.org/10.1016/S0003-3472(86)80208-1
Lebedeva, N.E., Kasumyan, A.O., and Golovkina, T.V., The correction of the physiological status of carp Cyprinus carpio by natural chemical signals, J. Ichthyol., 2000, vol. 40, no. 3, pp. 258–265.
Magurran, A.E. and Pitcher, T.J., Foraging, timidity and shoal size in minnows and goldfish, Behav. Ecol. Sociobiol., 1983, vol. 12, no. 2, pp. 147–152. https://doi.org/10.1007/BF00343206
Major, P.F., Predator-prey interactions in two schooling fishes, Caranx ignobilis and Stolephorus purpureus, Anim. Behav., 1978, vol. 26, no. 3, pp. 760–777. https://doi.org/10.1016/0003-3472(78)90142-2
Malyukina, G.A., Some questions of the physiology of the schooling behavior of fish, Tr. VNIRO, 1966, vol. 60, pp. 201–213.
Milanovskii, Yu.E. and Rekurbatskii, V.A., On the methods of studying the schooling behavior of fish, Nauchn. Dokl. Vyssh. Shk. Biol. Nauki, 1960, no. 4, pp. 77–81.
Milinski, M., Constraints placed by predators on feeding behavior, in The Behaviour of Teleost Fishes, Boston: Springer, 1976, pp. 236–252. https://doi.org/10.1007/978-1-4684-8261-4_9
Mittelbach, G., Group size and feeding rate in bluegills, Copeia, 1984, vol. 1984, no. 4, pp. 998–1000. https://doi.org/10.2307/1445350
Mochek, A.D., Etologicheskaya organizatsiya pribrezhnykh soobshchestv morskikh ryb (Ethological Organization of Coastal Marine Fish Communities), Moscow: Nauka, 1987.
Morgan, M.J. and Colgan, P.W., The effect of predator presence on foraging in bluntnose minnows, Pimephales notatus, Environ. Fish Biol., 1987, vol. 20, no. 2, pp. 105–111. https://doi.org/10.1007/BF00005290
Nadler, L.E., Killen, S.S., McClure, E.C., et al., Shoaling reduces metabolic rate in a gregarious coral reef fish species, J. Exp. Biol., 2016, vol. 219, no. 18, pp. 2802–2805. https://doi.org/10.1242/jeb.139493
Nikol’skii, G.V. and Kukushkin, A.A., On the question of the influence of stocking density on the intensity of food consumption by fish, Zool. Zh., 1943, vol. 22, no. 2, pp. 73–76.
Olla, B.L. and Samet, C., Fish-to-fish attraction and the facilitation of feeding behavior as mediated by visual stimuli in striped mullet, Mugil cephalus, J. Fish. Res. Board Can., 1974, vol. 31, no. 10, pp. 1621–1630. https://doi.org/10.1139/f74-204
Pagnussat, N., Piato, A.L., Schaefer, I.C., et al., One for all and all for one: The importance of shoaling on behavioral and stress responses in zebrafish, Zebrafish, 2013, vol. 10, no. 3, pp. 338–342. https://doi.org/10.1089/zeb.2013.0867
Parker, F.R., Reduced metabolic rates in fishes as a result of induced schooling, Trans. Am. Fish. Soc., 1973, vol. 102, no. 1, pp. 125–131. https://doi.org/10.1577/1548-8659(1973)102<125:RMRIFA>2.0.CO;2
Parrish, B.B., Blaxter, J.H., and Holliday, F.G., Herring (Clupea harengus L.) in aquaria establishment, Mar. Res., 1958, no. 5, pp. 1–11.
Parrish, J.K., Comparisons of the hunting behavior of four piscine predators attacking schooling prey, Ethology, 1993, vol. 95, no. 3, pp. 233–246. https://doi.org/10.1111/j.1439-0310.1993.tb00473.x
Pereira, P.H.C. and Ferreira, B.P., Effects of life phase and schooling patterns on the foraging behaviour of coral-reef fishes from the genus Haemulon, J. Fish. Biol., 2013, vol. 82, no. 4, pp. 1226–1238. https://doi.org/10.1111/jfb.12054
Peuhkuri, N., Size-assortative shoaling in fish: The effect of oddity on foraging behaviour, Anim. Behav., 1997, vol. 54, no. 2, pp. 271–278. https://doi.org/10.1006/anbe.1996.0453
Peuhkuri, N., Body size and food-patch finding in a stickleback shoal, J. Fish. Biol., 1998a, vol. 53, no. 3, pp. 687–689. https://doi.org/10.1111/j.1095-8649.1998.tb01011.x
Peuhkuri, N., Shoal composition, body size and foraging in sticklebacks, Behav. Ecol. Sociobiol., 1998b, vol. 43, nos. 4–5, pp. 333–337. https://doi.org/10.1007/s002650050499
Pitcher, T.J., Predators and food are the keys to understanding fish shoals: A review of recent experiments, Nat. Can., 1986, vol. 113, no. 3, pp. 225–233.
Pitcher, T.J. and Parrish, B.L., Functions of shoaling behavior in teleosts, in Behaviour of Teleost Fishes, London: Chapman and Hall, 1993, pp. 262–439.
Pitcher, T.J., Magurran, A.E., and Winfield, I.J., Fish in larger school find food faster, Behav. Ecol. Sociobiol., 1982, vol. 10, no. 2, pp. 149–151. https://doi.org/10.1007/BF00300175
Potts, G.W., Behavioural interactions between the Carangidae (Pisces) and their prey on the fore-reef slope of Aldabra, with notes on other predators, J. Zool., 1981, vol. 195, no. 3, pp. 385–404. https://doi.org/10.1111/j.1469-7998.1981.tb03472.x
Radakov, D.V., Schooling in the Ecology of Fish, New York: John Wiley, 1973.
Radakov, D.V. and Mocheck, A.D., Mutual stimulation in schools of Pristella riddlei Meek and Rhodeus sericeus (Pallas), J. Ichthyol., 1972, vol. 12, no. 3, pp. 538–540.
Ranta, E. and Kaitala, V., School size affects individual feeding success in three-spined sticklebacks (Gasterosteus aculeatus L.), J. Fish. Biol., 1991, vol. 39, no. 5, pp. 733–737. https://doi.org/10.1111/j.1095-8649.1991.tb04402.x
Ranta, E. and Lindström, K., Assortative schooling in three-spined sticklebacks?, Ann. Zool. Fennici, 1990, vol. 27, no. 2, pp. 67–75.
Rekubratskii, V.A., Ecological stereotypes of food-procuring and protective behavior of fish, in Povedenie i retseptsii ryb (Behavior and Reception of fish), Moscow: Nauka, 1967, pp. 121–125.
Robertson, D.R., Sweatman, H.P.A., Fletcher, E.A., and Cleland, M.G., Schooling as a mechanism for circumventing the territoriality of competitors, Ecology, 1976, vol. 57, no. 6, pp. 1208–1220. https://doi.org/10.2307/1935045
Ross, R.M., Backman, T.W.H., and Limburg, K.E., Group-size-mediated metabolic rate reduction in American shad, Trans. Am. Fish. Soc., 1992, vol. 121, no. 3, pp. 385–390. https://doi.org/10.1577/1548-8659(1992)121%3C0385:NG-MRRI%3E2.3.CO;2
Ryer, C.H. and Olla, B.L., Social mechanisms facilitating exploitation of spatially variable ephemeral food patches in a pelagic marine fish, Anim. Behav., 1992, vol. 44, no. 1, pp. 69–74. https://doi.org/10.1016/S0003-3472(05)80756-0
Sackley, P.G. and Kaufman, L.S., Effect of predation on foraging height in a planktivorous coral reef fish, Chromis nitida, Copeia, 1996, vol. 1996, no. 3, pp. 726–729. https://doi.org/10.2307/1447539
Smith, M.F.L. and Warburton, K., Predator shoaling moderates the confusion effect in blue-green chromis, Chromis viridis, Behav. Ecol. Sociobiol., 1992, vol. 30, no. 2, pp. 103–107. https://doi.org/10.1007/BF00173946
Steinegger, M., Sarhan, H., and Bshary, R., Laboratory experiments reveal effects of group size on hunting performance in yellow saddle goatfish, Parupeneus cyclostomus, Anim. Behav., 2020, vol. 168, pp. 159–167. https://doi.org/10.1016/j.anbehav.2020.08.018
Street, N.E., Magurran, A.E., and Pitcher, T.J., The effects of increasing shoal size on handling time in goldfish, Carassius auratus L., J. Fish. Biol., 1984, vol. 25, no. 5, pp. 561–566. https://doi.org/10.1111/j.1095-8649.1984.tb04902.x
Strübin, C., Steinegger, M., and Bshary, R., On group living and collaborative hunting in the yellow saddle goatfish (Parupeneus cyclostomus), Ethology, 2011, vol. 117, no. 11, pp. 961–969. https://doi.org/10.1111/j.1439-0310.2011.01966.x
Uematsu, T. and Takamori, J., Social facilitation in feeding behaviour of the medaka, Oryzias latipes. I. Continuous observation during a short period, Jpn. J. Ecol., 1976, vol. 26, no. 3, pp. 135–140. https://doi.org/10.18960/seitai.26.3_135
Van Havre, N. and FitzGerald, G.J., Shoaling and kin recognition in the threespine stickleback (Gasterosteus aculeatus L.), Biol. Behav., 1988, vol. 13, no. 4, pp. 190–201.
Vine, P.J., Effects of algal grazing and aggressive behavior of the fishes Pomacentrus lividus and Acanthurus sohal on coral reef ecology, Mar. Biol., 1974, vol. 24, no. 2, pp. 131–136. https://doi.org/10.1007/BF00389347
Volkova, L.A., Feeding intensity of some Baikal fish as part of a school and solitary ones, in Biologicheskaya produktivnost’ vodoemov Sibiri (Biological Productivity of Water Bodies of Siberia), Moscow: Nauka, 1969, pp. 53–57.
Volkova, L.A. and Kozhov, M.M., On the behavior of some fish from Lake Baikal, Vopr. Ikhtiol., 1966, vol. 6, no. 1, pp. 120–126.
Welsh, J.Q. and Bellwood, D.R., How far do schools of roving herbivores rove? A case study using Scarus rivulatus, Coral Reefs, 2012, vol. 31, no. 4, pp. 991–1003. https://doi.org/10.1007/s00338-012-0922-z
White, J.W. and Warner, R.R., Behavioral and energetic costs of group membership in a coral reef fish, Oecologia, 2007, vol. 154, no. 2, pp. 423–433. https://doi.org/10.1007/s00442-007-0838-4
ACKNOWLEDGMENTS
The authors express their sincere gratitude to A.A. Kazhlaev, A.S. Patseva, and L.S. Alekseeva (Moscow State University), who provided great assistance in preparing the article for publication. The authors are sincerely grateful to P.I. Kirillov (Institute of Ecology and Evolution, Russian Academy of Sciences) for careful and constructive editing of the text and illustrations, which improved the quality of the article.
Funding
The article was prepared within the framework of scientific projects of the state assignment of the Moscow State University No. 121032300100-5 and the Institute of Ecology and Evolution, Russian Academy of Sciences No. 121122300056-3 in the Unified State Information System for Accounting the Results of Civil Research, Development and Technological Works.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
CONFLICT OF INTEREST
The author of this work declares that he has no conflicts of interest.
Additional information
Translated by S. Avodkova
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kasumyan, A.O., Pavlov, D.S. Schooling Behavior and Feeding of Fish. J. Ichthyol. 63, 1320–1327 (2023). https://doi.org/10.1134/S003294522307010X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003294522307010X