Abstract—A review of the composition and structure of deep-sea pelagic ichthyocenes in the Bransfield Strait and northern Weddell Sea (Powell Basin) in the Atlantic sector of the Southern Ocean is presented. The material was obtained in summer 2020 and 2022 at 26 stations at layers from 120–0 to 2200–0 m. Nine hundred one specimens of juveniles and adult fishes belonging to 19 species from 10 families were collected. Statistical analysis made it possible to identify four significantly different regions in which the dominant ichthyocenes were located in waters of different origin. These ichthyocenes are poor in the number of identified species, but often quite numerous. Relatively shallow-water representatives of the families Bathydraconidae, Channichthyidae, Liparidae, and Nototheniidae, endemic to the shelf and slope of Antarctica, dominated in the area of the Antarctic Strait and on the shelf of the Antarctic Peninsula. At the same time in the Bransfield Strait, the abyssal basin of the Powell Basin, and in the pelagic zone above deep-sea ridges and trenches bordering it from the north, meso- and bathypelagic fish from the families Mycthophidae, Bathylagidae, Gonostomatidae, Paralepididae, and Scopelarchidae prevailed. Near the northern boundary of the Powell Basin, the number of mesobatypelagic species and their average abundance were higher than in its central part (11 vs. 7 species and 63.5 vs. 84.1 sp./100 m2). In the central part of the basin, Bathylagus antarcticus dominated, with an average share of 40% in catches and 28.8 sp./100 m2, while in the northern part of the basin, Electrona antarctica dominated, with an average share of 33%, 27.4 sp./100 m2.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
INTRODUCTION
The modern appearance of ichthyofauna in the Southern Ocean formed about 30 million years ago, together with the appearance of the Drake Passage and subsequent formation of the Antarctic Circumpolar Current (ACC) system, which led to isolation and cooling of Antarctic water masses, as well as to the emergence of a massive ice sheet over the Antarctic continent [3, 22]. These processes caused the disappearance of many taxonomic groups of fish characteristic of other areas of the World Ocean and contributed to the evolution of a unique ichthyofauna with an extremely high level of endemism and dominance of representatives of the suborder Notothenioidei. The Antarctic ichthyofauna is less diverse than one would expect, given its considerable age and vast water volumes occupied by the ichthyocenes. According to the latest data, 374 species of Antarctic marine fish from 19 families have been described, which make up only slightly more than 1% of the world’s marine ichthyofauna. A quarter of all known fish species in this region inhabit the meso- and bathypelagial [25]. The deep-sea pelagic ichthyofauna of the Southern Ocean is also characterized by low species diversity and a high degree of species endemism [14, 16]. The most numerous taxonomic groups of deep-sea meso- and bathypelagic fish living here belong to the families Myctophidae, Bathylagidae, Paralepididae, Gonostomatidae, and Scopelarchidae [4, 17, 25, 26]. Species from these families make up more than 95% of the biomass of the entire mesopelagic ichthyofauna of the upper 1 km layer inhabiting the Weddell–Scotia seas region [29]. This also includes the Antarctic silverfish. Pleuragramma antarcticum, the only representative of the family Nototheniidae with a pelagic life cycle [11, 45].
When studying the composition and structure of Antarctic marine pelagic ecosystems, as a rule, the central role and special attention is given to euphausiids (Euphausiidae), and in particular, Antarctic krill Euphausia superba, as the main food source for Antarctic birds and marine mammals, as well as the most important object of industrial fishery. However, as knowledge accumulates, it grows increasingly obvious that the trophic relationships in this region are much more complex originally assumed by the concept of phytoplankton–krill–top predators. So in order to understand the holistic pattern of ecosystems functioning, it is also necessary to adequately take into account the contribution of the meso- and bathypelagic ichthyofauna as a key trophic component of marine communities [7]. Being consumers of small-sized herbivorous zooplankton, meso- and bathypelagic fish in turn play an important role as an intermediate link between primary consumers and predators of the highest trophic level [18, 42], thereby ensuring the stability of Antarctic ecosystems by creating alternative trophic pathways where krill play a less significant role. In areas of Antarctica where krill are low in abundance, pelagic fish communities are the main contributors to biomass in the upper 1000 m layer in both oceanic and deep coastal areas under pack ice [29, 30, 32]. Thus, the study of the species composition, distribution, and existence conditions of Antarctic ichthyofauna is necessary to predict the impact of a possible decrease in the number of krill caused by anthropogenic and/or climatic factors on the trophic structure of Southern Ocean communities.
The water area around the Antarctic Peninsula, as well as the boundary area between the Scotia and Weddell seas, is one of the most studied areas of Antarctica. Despite the large number of studies on various aspects of the functioning of deep-sea pelagic ecosystems in this region, only a small proportion directly concern the peculiarities in the distribution and species composition of pelagic fish communities [36, 40, 43]. Judging from the literature data, in studying the Antarctic ichthyocenes in these areas, in addition to adult fish, much attention was paid to the species composition and abundance of early pelagic stages of notothenias from by-catches of Antarctic krill trawls [6, 23, 24]. In this case, as a rule, the upper 200–400 m layer was catched, which led to significant underestimation of the contribution of mesopelagic species. Often, information on the composition of fish communities in the area is given together data on macrozooplankton [12, 29, 30, 35], which greatly complicates their interpretation and comparison. Earlier studies have established the composition and general patterns of distribution of species belonging to pelagic ichthyocenes [10]. It has been shown that the pelagic ichthyofauna above the shelf is represented mainly by juveniles of demersal notothenias and white-blooded pikes (Notothniidae, Channichtyidae) and, to a lesser extent, by various stages of development of mesopelagic species [21, 23, 24], while juveniles and adults of mesopelagic species with a clear dominance in terms of abundance and biomass of representatives of the families Myctophidae, Bathylagidae, and Paralepididae prevail above the shelf slope [12, 29, 30, 39]. Only in the area of the western Antarctic Peninsula, where circumpolar deep waters reach the shelf, this pattern is violated, contributing to the removal of mesopelagic fish to coastal areas [13]. In addition to bottom topography, another important factor governing the structure of communities is latitudinal zoning. With distance from the mainland to the north, closer to the central part of the Antarctic Circumpolar Current (ACC), the number and species composition of mesopelagic fishes significantly increase [42].
Our main objective was to study the biodiversity of deep-sea ichthyocenes and patterns in the distribution of mass species of pelagic fish in summer in the Bransfield Strait and little-studied northern part of Weddell Sea (Powell Basin) depending on water mass structure.
MATERIALS AND METHODS
The material for this study was collected on cruises 79 and 87 of the R/V Akademik Mstislav Keldysh, carried out on January 24–22, 2020, and January 22–February 12, 2022. The area of work on both cruises covered Bransfield Strait, the northern part of the Weddell Sea (Powell Basin), as well as the area of the South Orkney Islands (waters above the Orkney Trench and the Laurie Trench) (Fig. 1). To collect ichthyological materials, an Samyshev–Aseev modified Isaacs–Kydd nonclosing midwater pelagic trawl (SAMT) equipped with a 25-m-long double bag was used; the outer bag was made of knotted nylon thread of 1-mm-diameter and a mesh size of 50 mm; the inner bag was made of a nodal mesh with a size of 5 mm and had a end insert from a no. 15 nylon sieve. The mouth area of the trawl was 6 m2. In total, 16 trawl stations were completed in 2020, at which 17 samples were taken, and in 2022, 10 trawl stations, at which 10 samples were taken (Table 1). The catches were done both in coastal and offshore areas, from 120–0 to 2212–0 m layers at a vessel speed of 3 knots without long exposure of the trawl at any of the passable depths. The lower limit of the fishing layer was determined by readings from a Senti DT temperature and pressure sensor manufactured by Star-Oddyi. The distance traveled by the vessel between the start and end trawl points was considered the great circle distance [48] and was calculated with an Internet calculator [38].
The distance covered by the trawl at each station was calculated with previously published equations [46]. The volume of filtered water was determined by multiplying the mouth area of the trawl by the distance between the start and end points of the trawling. The abudance of fish caught in each trawl was calculated based on the volume of filtered water. The biomass of caught fish is given in the text for stations where the corresponding measurements were obtained in 2022, but it was not taken into account for comparative purposes, since when sampling with nonclosing fishing gear, its underestimation is inevitable, because the largest and/or most mobile pelagic fish specimens evade the trawl [15], such as, e.g., Paralepididae species in Antarctic waters. While having a small effect on the total abundance of fish in the catch, this can lead to significantly underestimation of the total biomass of the sample. Initially, the samples were fixed with 2–4% formalin neutralized by seawater, and then transferred to 75% ethanol.
Statistical analysis was performed with the software package PRIMER v. 6 [8]. Data processing was based on the matrix of species similarity of samples, calculated with the Bray–Curtis index; the percentage of species and abundance of fish in the sample were used as the initial data. To reduce the influence of dominant species, before construction of the similarity matrix, the data were transformed by extracting the square root. When statistically analyzing the results, clustering was done by unweighted pair group method with arithmetic mean (UPGMA) [41] and ordination by nonmetric multidimensional scaling (MDS) [28]. To assess the reliability of the clustering results, the SIMPROF permutation test was performed (999 iterations, p = 0.05). Standardization and data transformation was not applied.
Information about the hydrophysical parameters of the medium (temperature and salinity in the water column) was obtained in the course of vertical soundings. In 2020, an SBE 19plus hydrophysical probe was used to obtain temperature and salinity profiles; data from 11 soundings were used. In 2022, hydrological measurements were carried out using an Idronaut OCEAN SEVEN 320Plus probe, lowered on a cable-rope from the surface to the bottom. The data of 34 soundings were used to construct the sections. A total of five hydrophysical profiles were selected to describe the structure of waters in 2020 and 2022 (Fig. 2).
The layout of hydrophysical transects in 2020 (a) and 2022 (b) ( , hydrophysical stations; (1) transect through Powell Basin; (2) transect above the underwater rises of Powell Basin; (3) transect through shelf of Antarctic Peninsula, deep-water part of Powell Basin, and South Orkney Plateau; (4) Bransfield Strait; (5) Antarctic Strait).
, hydrophysical stations; (1) transect through Powell Basin; (2) transect above the underwater rises of Powell Basin; (3) transect through shelf of Antarctic Peninsula, deep-water part of Powell Basin, and South Orkney Plateau; (4) Bransfield Strait; (5) Antarctic Strait).
RESULTS
Features of the water structure in the study areas. To describe the hydrophysical conditions in the 2020 survey area, sections were chosen that passed through the shelf and central part Powell Basin, as well as the area of the Philip and South Scotia ridges (Figs. 3, 4).
Temperature and salinity distribution on transect above underwater rises in northern Powell Basin in 2020 (abbreviations same as in Fig. 3).
The hydrological structure of waters at stations in the southeastern part of the section (stations 6651–6653, 6656) (Fig. 3) was characterized by the presence of a 300- to 200-m supercooled upper layer with temperatures of –1.5 to –2°С and relatively low salinity 33.2–34.4 PSU, which was a consequence of summer melting of the ice cover, which prevented heating of the surface water layer. On the shelf, this supercooled layer was located from the surface to the bottom, while at stations in the central Powell Basin, a layer of relatively warm (0–0.6°C, 34.6 PSU) deep water (WDW) was located below it, which occupied depths from about 300 to 1500–1700 m. At stations in the northeastern part of the section (station 6607–6610), the water structure was typical of the Weddell Sea in summer [2]. In the upper 100–150 m, there was a heated summer surface layer with a strong vertical temperature gradient. Below it, at a depth of about 200 m, there was a summer cold intermediate layer (CIL) with a subsurface temperature minimum with values of about –0.5°С (Fig. 3). Then, there was a layer of warm deep water with a temperature of up to 0.6°C at a depth of about 500 m; farther towards the bottom, the temperature decreased. At stations in the northwestern part of the study area (stations 6601, 6602) (Fig. 4), a slightly different hydrophysical pattern was observed: water coming from the western part of Weddell Sea together with drift ice and a large number of icebergs (Weddell Sea shelf water) underwent mixing on underwater rises due to internal waves generated by tidal currents. As a result, a weakly stratified vertical structure formed, where the deep temperature maximum was not pronounced.
In January 2022, the distribution of the main hydrophysical characteristics in the Powell Basin also generally corresponded to the long-term average [34] and differed from 2020 only by more pronounced heating of the surface layer, facilitated by the absence of melting ice and retreat of the ice edge south of the 2020 boundary. Three layers can be distinguished in the section: the upper layer, 20 to 100 m, was occupied by Antarctic surface water that formed during summer heating. The temperature increased from south to north, from 0.5 to 2°C. The salinity distribution contained local minima, probably associated with the melting of ice; the values were mostly within 34.40–34.50 PSU. Below was a cold intermediate layer (CIL), the result of winter convection; its thickness varied from 50 to 150 m (Fig. 5). The minimum potential temperatures were observed at slope stations (stations 7345–7348) and reached –1.69°C, while salinity gradually increased with depth without significant gradients. Starting from a depth of 200 m, in the central deep part of the Powell Basin, warm deep water was observed, well defined by the zero isotherm [5, 31], corresponding to a salinity of 34.70 PSU. The rise of the temperature and salinity contours in the central part of the Powell Basin was facilitated by cyclonic circulation there.
Temperature and salinity distribution on transect through Powell Basin (1), South Orkney Plateau (2), and Orkney Trench (3) in 2022. (abbreviations same as in Fig. 3; SW, shelf water).
In 2022, in the Bransfield Strait, a typical thermohaline water structure was observed for this area [20, 27, 44]: most of the strait was occupied by cold, saline, with a negative temperature, modified Weddell Sea water (TWW). At the same time, warm and desalinated modified Bellingshausen Sea water (TBW) also spread in a narrow jet along the South Shetland Islands with the Bransfield Current, which also entered the middle of the strait in the upper 50 m layer (Fig. 6). The lower part of the Bransfield Current jet was modified circumpolar deep water (mCDW), warmer and saltier, with a core at a depth of 300 m.
The section through the Antarctic Sound (Fig. 7.) was characterized by a decrease in temperature and salinity from north to south. The northern shallow part of the strait (stations 7324, 7325) was probably filled with water from the Bransfield Strait, which had a higher potential temperature, with a maximum of –0.10°C in the upper 60 m layer. Increased water salinity was also observed here, only, in contrast to the temperature, its maximum was in the bottom layer, amounting to 34.55 PSU at depths of about 150 m. The deep-water central part of the strait was affected by water from the Weddell Sea with a lower temperature and relative desalination due to ice melting in the upper 100 m layer.
SAMT catches in different operations sites. The collection of juvenile and adult fish caught by SAMT in 2020 and 2022 included 901 specimens, in total belonging to 19 species from 10 families (Table 2).
As a result of cluster analysis of the collected materials, including the SIMPROF permutation test, four main groups of samples (A, B, C, and D) were identified at a similarity level of 4.7 (π = 7.0, p = 0.001), 29.3 (π = 3.3, p = 0.009) and 41.6% (π = 2.5, p = 0.032); at a higher level, no statistically significant data structure was observed (Fig. 8).
Results of cluster analysis of SAMT samples in operations area during cruises 79 and 87 of R/V Akademik Mstislav Keldysh in summer 2020 and 2022. A, B, C, D are significant (p = 0.05) groups of samples identified by the SIMPROF test. (A, Powell Basin and waters above trenches and underwater rises bordering its northern part; B, Bransfield Strait and adjacent waters in zone of influence of Bransfield Current; C, Antarctic Strait; D, shelf water in western Powell Basin
Graphically, all selected groups on the MDS diagram are represented as a cluster of points, or individual points, which, nevertheless, are well separated from each other (Fig. 9).
MDS diagram with results of station ordination based on species composition and abundance of deep-sea fish caught by SAMT in study area during cruises 79 and 87 of R/V Akademik Mstislav Keldysh in summer 2020 and 2022: ovals, main ichthyocenes of meso- and bathypelagial, identified at similarity level of 10%; 2D stress, 0.1; for the rest, see notation in Fig. 8.
The samples of group A encompassed the vast majority of ichthyological collections acquired in summer 2020 and 2022 in deep-water areas of the Powell Basin in the Weddell Sea, as well as above the trenches and underwater uplifts bordering this basin from the northwest and northeast. Materials were collected at 17 stations at layers from 634–0 (station 6687) to 2212–0 m (station 6617). In total, 718 specimens of juvenile and adult fish were caught here in the pelagic zone, belonging to 12 species from 6 families. The largest number of taxa were recorded in the mesopelagic family Myctophidae, where a total of 7 species were noted in the study area (Electrona antarctica, Gymnoscopelus braueri, G. opisthopterus, G. nicholsi, Krefftichthys andersoni, Protomyctophum bolini, and P. choriodon) (Fig. 10), or 58% of the total number of caught species. Each of the five other families recorded in deep water in the Powell Basin and adjacent waters (Bathylagidae, Gonostomatidae, Scopelarchidae, Paralepididae, Macrouridae) were represented by only one species in the collections (Table 2). Catch values at stations in the area of the Powell Basin and deep-water trenches and seamounts fringing its northern part varied from 41.8 (station 7362) to 123.5 (station 7385) sp./100 m2 (Fig. 11). All successful trawls were carried out inthe layers from 634–0 to 2143–0 m. It should be noted that in the catches of two shallow-water trawls at depths of 305–0 (station 6601) and 120–0 m (station 6614), juvenile and adult fish were completely absent. The number of deep-sea fish caught at stations in the central deep-water basin of the Powell Basin, as well as near its western and eastern slopes (stations 6607, 6617, 6619, 6653, 6655, 7362, 7367) was 41.8–84.4 (average 63.5) sp./100 m2, whereas in trawl catches at stations in the northern part of the Weddell Gyre, above deep-sea trenches and seamounts (stations 6601, 6684, 6685, 6686, 6687, 6688, 6690, 7378, 7383, 7385), their abundance was noticeably higher, between 49.4 and 123.5 sp./100 m2, 84.1 sp./100 m2, on average. Data for 2022 on the biomass of deep-sea fish from SAMT catches in the Powell Basin and adjacent waters showed that it varied between 369.3 (station 7378) and 942.1 (station 7383) g/100 m2 (on average, 684.7 g/100 m2). No significant differences were found in the biomass of deep-sea fishes in different parts of this study area. The most abundant in catches were two mesopelagic species, Electrona antarctica (Myctophidae) and Bathylagus antarcticus (Bathylagidae) (Fig. 12). The total share of the first and second species in SAMT catches in summer 2020 and 2022 was about 55% in terms of abundance and in terms of biomass - 53%. These species were encountered at almost all stations, except for the shallowest ones and/or those in the coastal shelf zone; they had a similar distribution pattern throughout the studied water area.
Ratio of the dominant pelagic fish taxa in SAMT catches in study area on cruises 79 and 87 of R/V Akademik Mstislav Keldysh in summer 2020 and 2022, in groups of samples identified by cluster analysis from different regions of the studied water area. See Fig. 11 for notation. ((1) Bathylagus antarcticus; (2) Electrona antarctica; (3) Notolepis coatsorum; (4) Gymnoscopelus braueri; (5) G. opisthopterus; (6) Pleuragramma antarctica; (7) Cyclothone microdon; (8) Champsocephalus gunnari; (9) other).
Distribution of total abundance of pelagic fish based on results of SAMT catches in different study areas during cruises 79 and 87 of R/V Akademik Mstislav Keldysh (colored areas are identified by cluster analysis of species composition of catch:  ,A;
,A;  , B;
, B;  , C;
, C;  , D; numbers, abundance: (1) 10; (2) 50; (3) 100 sp./100 m2).
, D; numbers, abundance: (1) 10; (2) 50; (3) 100 sp./100 m2).
Group B includes all stations from the Bransfield Strait and northwestern part of the shelf and slope of the Antarctic Peninsula. Trawl operations in the Bransfield Strait and adjacent waters were carried out in summer 2020 and 2022 at five stations encompassing catch depths between 400–0 and 1160–0 m. A total of 119 specimens of 10 species from 4 families were caught here. Lanternfish (Myctophidae) were represented by the largest number of taxa in the study area, which included six species, or 60% of the total number of species in catches, while caught specimens of all other families were represented by one (Paralepididae, Bathylagidae) or two species (Channichthyidae); their total share in catches was 40% at the study area. Deep-sea fish catches in the area of the Bransfield Strait varied at different stations from 13.0 (station 6597) to 87.5 sp./100 m2 (station 7358) (see Fig. 11); moreover, at the 440–550–0 m trawling layers, the number of fish in catches was slightly less than at the deeper water layers of 1160–1000–0 m: 13.0–33.8 (21.4 on average) and 46.2–87.5 (66.9 on average) sp./100 m2 respectively. The available data for 2022 on the biomass of deep-sea fish from SAMT catches in this study area also demonstrated similar trends: in shallow-water trawls spanning the 440–550–0 m layers, the catch biomass was 131.5–386.7 g/100 m2, and in deep-sea trawls at layers 1160–1000–0 m, it was 455.6–1703.0 g/100 m2. The most common deep-sea fish species in group B trawls were two species of lanternfish—Electrona antarctica and Gymnoscoplus braueri—found in this study area in each sample. The abundance of these species was 8.2–35.2 (average 22.0) and 1.6–7.5 (average 4.4) sp./100 m2, respectively
Group C included trawl collections in 2020 and 2022, made in the Antarctic Strait separating the Joinville Islands from the northeastern tip of the Antarctic Peninsula, at two stations (6648 and 7331) with trawling layers of 650–0 and 590–0 m. The species composition of fish in the catches was somewhat poorer than in the Bransfield Strait. In total, 55 specimens of juveniles and adult fish of seven species from five families were caught here; mesopelagic fish were virtually absent in catches and only one specimen was represented: Electrona antarctica (Myctophidae). All other fish from the catches (86%) belonged to species endemic to Antarctic waters from the families Bathydraconidae, Channichthyidae, and Nototheniidae, or to highly specialized species (family Liparidae) well adapted to the existence conditions at low and even negative temperatures. Deep-sea fish catches in the Antarctic Strait varied between 32.3 and 64.8 sp./100 m2, and the biomass at the only station where measurements were taken (station 7331) was 2120.6 g/100 m2. The dominant position in catches in the Antarctic Strait was occupied by the Antarctic silverfish Pleuragramma antarcticum (Nototheniidae), whose shares in abundance and biomass were 72–79 and 78%, respectively.
Group D included a single sample (station 6651) collected on a relatively shallow shelf in the western Powell Basin at depths of 505–0 m. The catch was very poor both in species composition and abundance. In total, at the station 9 specimens of fish were caught, belonging to 2 species from 2 families: mackeral whitefish Champsocephalus gunnari (Channichthyidae) and Antarctic lanternfish Electrona antarctica (Myctophidae). Moreover, only the latter species can be attributed to proper mesopelagic species. The total number of fish in the catch was 14.3 sp./100 m2, of which 67% were Champsocephalus gunnari. Initially, it seems that the considered station should instead be assigned to group B (based on the location, presence of myctophids, and absence of cyclothones and bathylagids). However, the small number of species (two in all) and dominance of mackeral whitefish forced the program’s algorithm to place it in a separate cluster supported by statistically significant differences.
DISCUSSION
The results of processing our materials show that the pelagic ichthyocenes in the waters south of the southern boundary of the ACC and Scotia Front, which separate the waters of the Scotia and Weddell seas, are rather poor in biodiversity, but often numerous (due to the high abundance and biomass of dominant species) communities, both relatively shallow-water, endemic to the Antarcti shelf and slope, representatives of the families Bathydraconidae, Channichthyidae, Liparidae, Nototheniidae, and deeper meso- and bathypelagic fishes, mostly consisting of species of the family Mycthophidae, Bathylagidae, Gonostomatidae, Paralepididae, and Scopelarchidae. Their habitats were mainly localized in the water column above trenches and deep-water basins in the northern Weddell Sea. The data obtained agree well with earlier studies of pelagic Antarctic ichthyofauna [9, 19, 42, 47]. The study of the taxonomic composition, abundance, and nature of the spatial and vertical distribution of pelagic fish communities in different regions of the studied water area, in our opinion, shows their close relationship with the hydrophysical characteristics and structure of the water masses in these areas. Indeed, the deep-water communities of meso- and bathypelagic fish, observed mainly in the central abyssal basin and surrounding periphery of the Powell Basin, as well as partly in the deep-water area of the Bransfield Strait, in summer are apparently closely associated with the warm layer (0–0.6° C, 34.6 PSU) of deep water (WDW) occupying layers from about 300 to 1500–1700 m. As well, mesobathipelagic fish apparently reach their maximum abundance at layers below 400 m; in the overlying layers, only their larval stages are present, while juveniles and sexually mature specimens, judging from our data, are either very few or completely absent. The natural limiting environmental factor in the Powell Basin and the deep parts of the Bransfield Strait, preventing the penetration of meso- and bathypelagic fish into shallower layers, is the presence here of a supercooled, formed as a result of winter convection, overlying water layer 200–300 m thick a with a temperature of –1.5 to –2°С and relatively low salinity of 33.2–34.4 PSU. Deep-sea fish may avoid waters with similar characteristics and/or are only randomly introduced into them. In the upper 20–100 m layer, occupied by warm and relatively desalinated Antarctic Surface Water (AASW) formed during summer heating, there are only a few larvae of mesopelagic fish, mainly of the family Paralepididae [1]. Moving within the Powell Basin from south to north to the area of the South Scotia Ridge and deep-sea trenches serving as a natural boundary separating waters of the Weddell and Scotia seas, the species composition and abundance of deep-sea fish communities becomes somewhat higher. At stations near the northern boundary of the Powell Basin, the number of caught species and average abundance of mesobathypelagic fish were higher than at stations above its central part and western and eastern slopes (respectively, on average, 11 vs. 7 species and 63.5 vs. 84.1 sp./100 m2). In addition, at stations in the central part of the basin, compared to its northern part, Bathylagus antarcticus noticeably dominated in catches(average 40 vs. 24% of the total number of fish in the catch, or 28.8 vs. 20.2 sp./100 m2), and the number Electrona antarctica, on the contrary, was noticeably smaller (average 25 vs. 33% or 12.9 vs. 27.4 sp./100 m2). The collections on the slopes and in the central part of the Powell Basin also lacked such mesobatypelagic fish species as Gymnoscopelus nicholsi, Krefftichthys andersoni, Protomyctophum choriodon, Protomyctophum bolini, and Benthalbella elongata. According to earlier data, the largest values of primary production, biomass and species composition, and phyto- and zooplankton were observed on the northern periphery of the Powell Basin, and the minimum values were observed in the area of its abyssal basin. The high abundance and diversity of zooplankton communities in this area, which exceed those in other studied areas by five to ten times, indicates that this water area is an important feeding ground for various Antarctic plankton-eating species, from fish to whales [1]. The increased biodiversity of deep-sea fish communities along the northern boundary of the Weddell Sea may also be due to the fact that only a minority of meso- and bathypelagic fish species, such as Electrona antarctica, Bathylagus antarcticus, and Notolepis coatsorum, completely passes through its life cycle in the Weddell Sea, while most species noted here exist only as expatriates in Antarctic waters, which for them, in this case, can be considered as a sterile removal zone, where adults cannot spawn and eggs and larvae not undergo development and metamorphosis [9, 11, 37, 39]. Such species usually do not achieve a high abundance, since the local population is replenished only by the introduction of juveniles or mature individuals from outside. Approaching the boundaries separating waters of different origin, the number of such expatriate species should increase, while their role in the ecosystem should increase. Judging from the results of our studies, the fish communities observed in the Antarctic Strait and on the relatively shallow shelf of the Antarctic Peninsula differ radically in their taxonomic composition and abundance from those of deeper water areas of the studied water area. Apparently, they are also determined by the hydrophysical parameters of the medium and structure of the water masses occupying these areas of the sea. In our opinion, the presence in these areas in the Southern Hemisphere’s summer period of supercooled waters with negative temperatures (–1.5 to –2°С) and relatively low salinity (33.2–34.4 PSU), occupying almost the entire space from surface to bottom, is the main reason for the predominance in the pelagial of species that, in the evolution of the Antarctic ichthyofauna, have developed adaptations to such existence conditions. These are primarily different species of crocodile icefishes (Channichthyidae), Antarctic dragonfishes (Bathydraconidae), cod icefish (Nototheniidae), and snail fishes (Liparidae). Some species from these families, such as the Antarctic silverfish Pleuragramma antarcticum, can occupy a dominant position in the ichthyocenes and attain a relatively high abundance in catches [33], while the deeper-water representatives of the “true” mesopelagic ichthyofauna appear in them only sporadically, as single individuals.
CONCLUSIONS
(1) In summer 2020 and 2022, 19 species of juvenile and adult pelagic fish belonging to 10 families were recorded in the Bransfield Strait, Antarctic Strait, and shelf and deep water parts of the Powell Basin.
(2) Analysis of the abundance and biodiversity of the deep-sea pelagic ichthyofauna made it possible to identify four main ichthyocenes (A, B, C, and D) in the studied water area, supported by statistically significant differences and confined, respectively, to the deep-water regions of the Powell Basin, the Bransfield Strait, and the northwestern part of the shelf and slope of the Antarctic Peninsula, to the Antarctic Strait, as well as to the deep-water shelf of the western part of the Powell Basin.
(3) Deep-sea pelagic ichthyocenes in the waters south of the southern boundary of the Antarctic Circumpolar Current and the Scotia Front, are quite poor in biodiversity, but often numerous (due to the high abundance and biomass of dominant species) communities. They include as relatively shallow, endemic for the shelf and slope of Antarctica, representativesof the families Bathydraconidae, Channichthyidae, Liparidae, Nototheniidae, as well as deeper meso- and bathypelagic fishes, mostly consisting of species of the families Mycthophidae, Bathylagidae, Gonostomatidae, Paralepididae, and Scopelarchidae.
(4) The study of the taxonomic composition, abundance, and nature of the horizontal and vertical distribution of the identified pelagic fish communities in different regions of the studied water area demonstrated their close relationship with the hydrophysical characteristics and structure of the water masses present in these areas.
REFERENCES
E. G. Morozov, D. I. Frey, A. A. Polukhin, et al., “Mesoscale variability of the ocean in the northern part of the Weddell Sea,” Oceanology 60 (5), 573–588 (2020). https://doi.org/10.1134/S0001437020050173
E. G. Morozov, V. A. Spiridonov, T. N. Molodtsova, et al., “Investigations of the ecosystem in the Atlantic Sector of Antarctica (cruise 79 of the R/V Akademik Mstislav Keldysh),” Oceanology 60 (5), 721–723 (2020). https://doi.org/10.1134/S0001437020050161
M. E. Anderson, “The origin and evolution of the Antarctic ichthyofauna,” in Fishes of the Southern Ocean, Ed. by O. Gon and P. C. Heemstra (Smith Inst. Ichthyol., Grahamstown, 1990), pp. 28–33.
A. P. Andriashev, “A general review of the Antarctic fish fauna,” in Biogeography and Ecology of Antarctica (Junk Publisher, The Hague, 1965), pp. 491–550.
E. C. Carmack and T. D. Foster, “On the flow of water out of the Weddell Sea,” Deep-Sea Res. 22, 711–724 (1975). https://doi.org/10.1016/0011-7471(75)90077-7
I. A. Catalán, B. Morales-Nin, J. B. Company, et al., “Environmental influences on zooplankton and micronekton distribution in the Bransfield Strait and adjacent waters,” Polar Biol. 31, 691–707 (2008). https://doi.org/10.1007/s00300-008-0408-1
L. Chunlin, Z. Chi, L. Yang, et al., “Age and growth of Antarctic deep-sea smelt (Bathylagus antarcticus), an important mesopelagic fish in the Southern Ocean,” Deep-Sea Res. II 201, 1–11 (2022). https://doi.org/10.1016/j.dsr2.2022.105122
K. R. Clarke and R. M. Warwick, Primer-6 computer program (Natural Environment Research Council, Plymouth, 2005). http://refhub.elsevier.com/S0079-6611(16)30145-8/h0110. Accessed January 11, 2020.
M. A. Collins, G. Stowasser, S. Fielding, et al., “Latitudinal and bathymetric patterns in the distribution and abundance of mesopelagic fish in the Scotia Sea,” Deep-Sea Res. II 59, 189–198 (2012). https://doi.org/10.1016/j.dsr2.2011.07.003
Biogeographic Atlas of the Southern Ocean, Ed. by C. De Broyer, P. Koubbi, H. J. Griffths, (Scientifc Committee on Antarctic Research, Cambridge, 2014).
H. H. Dewitt, P. C. Heemstra, and O. Gon, “Nototheniidae,” in Fishes of the Southern Ocean, Ed. by O. Gon and P. C. Heemstra (Smith Inst. Ichthyol., Grahamstown, 1990), pp. 279–331.
J. Donnelly, T. T. Sutton, and J. J. Torres, “Distribution and abundance of micronekton and macrozooplankton in the NW Weddell Sea: Relation to a spring ice-edge bloom,” Polar Biol. 29, 280–293 (2006). https://doi.org/10.1007/s00300-005-0051-z
J. Donnelly and J. J. Torres, “Pelagic fishes in the Marguerite Bay region of the West Antarctic Peninsula continental shelf,” Deep-Sea Res. II 55, 523–539 (2008). https://doi.org/10.1016/j.dsr2.2007.11.015
J. T. Eastman, “The nature of the diversity of Antarctic fishes,” Polar Biol. 28, 93–107 (2005). https://doi.org/10.1007/s00300-004-0667-4
J. Gjosaeter and K. Kawaguchi, “A review of the world resources of mesopelagic fish,” FAO Fish. Tech. Pap., No. 193, 151 (1980).
T. M. Greely, J. V. Gartner Jr., and J. J. Torres, “Age and growth of Electrona antarctica (Pisces: Myctophidae) the dominant mesopelagic fish of the southern ocean,” Mar. Biol. 133, 145–158 (1999).https://doi.org/10.1007/s002270050453
G. Hempel, Antarctic marine food webs, in Antarctic Nutrient Cycles (Springer, Berlin, 1985).
T. L. Hopkins and J. J. Torres, “Midwater food web in the vicinity of a marginal ice zone in the western Weddell Sea,” Deep Sea Res. Part A. Oceanogr. Res. Pap. 36 (4), 543–560 (1989). https://doi.org/10.1016/0198-0149(89)90005-8
P. A. Hulley, “Results of the research cruises of FRV “Walther Herwig” to South America. 58. Family Myctophidae (Osteichthyes, Myctophiformes),” Arch. Fischereiwiss 31, 1–300 (1981).
W. G. Huneke, O. Huhn, and M. Schroeder, “Water masses in the Bransfield Strait and adjacent seas, austral summer 2013,” Polar Biol. 39, 789–798 (2016). https://doi.org/10.1007/s00300-016-1936-8
C. D. Jones, P. Koubbi, B. Catalano, et al., “Mesopelagic and larval fish survey,” NOAA Technical Memorandum NMFS SWFSC 524, 28–40 (2014).
J. P. Kennett, “Paleoceanographic and biogeographic evolution of the Southern Ocean during the Cenozoic, and Cenozoic microfossil datums,” Palaeogeogr., Palaeoclimatol., Palaeoecol. 31, 123–152 (1980). https://doi.org/10.1016/0031-0182(80)90017-6
A. Kellermann and K. H. Kock, “Patterns of spatial and temporal distribution and their variation in early life stages of Antarctic fish in the Antarctic Peninsula region,” in Antarctic Ocean and Resources Variability (Springer, Berlin, 1988), pp. 147–159.
A. Kellermann and S. Schadwinkel, “Winter aspects of the ichthyoplankton community in Antarctic Peninsula waters,” Polar Biol. 11, 117–127 (1991).
K. H. Kock, Antarctic fish, in Key Environments (Pergamon Press, Oxford, 1985), pp. 173–192.
K. H. Kock, Antarctic Fish and Fisheries (University Press, Cambridge, 1992).
V. A. Krechik, D. I. Frey, and E. G. Morozov, “Peculiarities of water circulation in the central part of the Bransfield Strait in January 2020,” Dokl. Earth Sci. 496 Part 1, 92–95 (2021). https://doi.org/10.1134/S1028334X21010116
J. B. Kruskal and M. Wish, Multidimensional Scaling, Quantitative Application in the Social Sciences (Sage Univ. Press, Beverly Hills, 1978).
T. M. Lancraft, J. J. Torres, and T. L. Hopkins, “Micronekton and macrozooplankton in the open waters near Antarctic ice edge zones (AMERIEZ 1983 and 1986),” Polar Biol. 9 (4), 225–233 (1989). https://doi.org/10.1007/BF00263770
T. M. Lancraft, T. L. Hopkins, J. Torres, and J. Donnelly, “Oceanic micronektonic/macrozooplanktonic community structure and feeding in ice covered Antarctic waters during the winter (AMERIEZ 1988),” Polar Biol. 11, 157–167 (1991). https://doi.org/10.1007/BF00240204
E. G. Morozov, V. A. Krechik, D. I. Frey, et al., “Frontal zone between relatively warm and cold waters in the northern Weddell Sea,” in Antarctic Peninsula Region of the Southern Ocean (Springer Nature, 2021), pp. 31–53. https://doi.org/10.1007/978-3-030-78927-5_3
E. J. Murphy, J. L. Watkins, P. N. Trathan, et al., “Spatial and temporal operation of the Scotia Sea ecosystem: A review of large-scale links in a krill centred food web,” Philos. Trans. R. Soc. London B 362, 113–148 (2007). https://doi.org/10.1098/rstb.2006.1957
R. L. O’Driscolla, G. J. Macaulaya, S. Gauthier, et al., “Distribution, abundance and acoustic properties of Antarctic silverfish (Pleuragramma antarcticum) in the Ross Sea,” Deep Sea Res. II 58, 181–195 (2011). https://doi.org/10.1016/j.dsr2.2010.05.018
A. H. Orsi, W. D. Nowlin, and IIIT. Whitworth, “On the circulation and stratification of the Weddell Gyre,” Deep-Sea Res. I 40, 169–203 (1993). https://doi.org/10.1016/0967-0637(93)90060-G
M. L. Parker, W. R. Fraser, J. Ashford, et al., “Assemblages of Micronektonic fishes and invertebrates in a gradient of regional warming along the Western Antarctic Peninsula,” J. Mar. Syst. 152, 18–41 (2015). https://doi.org/10.1016/j.jmarsys.2015.07.005
U. Piatkowski, M. White, and W. Dimmler, “Micronekton of the Weddell Sea: Distribution and abundance,” Berichte zur Polarforschung. Alfred-Wegener-Institut fur Polar-und Meeresforschung, 73–81 (1990).
U. Piatkowski, P. G. Rodhouse, M. G. White, et al., “Nekton community of the Scotia Sea as sampled by the RMT 25 during austral summer,” Mar. Ecol.: Proc. Ser. 112, 13–28 (1994). https://doi.org/10.3354/meps112013
Planetcalc. Catalog of online calculators. https://planetcalc. Version 3.0.4170.0. November 2022.
C. Pusch, P. A. Hulley, and K. H. Kock, “Community structure and feeding ecology of mesopelagic fishes in the slope waters of King George Island (South Shetland Islands, Antarctica),” Deep-Sea Res. I 51, 1685–1708 (2004).
J. M. Rembiszewski, M. Krzeptowski, and T. B. Linkowski, “Fishes (Pisces) as by-catch in fisheries for krill Euphausia superba Dana (Euphausiacea, Crustacea),” Pol. Arch. Hydrobiol. 25, 677–693 (1978).
H. C. Romesburg, Cluster Analysis for Researchers (Lulu.com, Morrisville, NC, 2004).
R. A. Saunders, M. A. Collins, G. Stowasser, and G. A. Tarling, “Southern Ocean mesopelagic fish communities in the Scotia Sea are sustained by mass immigration,” Mar. Ecol.: Proc. Ser. 569, 173–185 (2017). https://doi.org/10.3354/meps12093
K. E. Skóra and J. Sosiński, “Observations on the ichthyofauna distribution in the regions of the Scotia Sea and Antarctic Peninsula,” Polish Polar Research 4 (1–4), 49–55 (1983).
R. Tokarczyk, “Classification of water masses in the Bransfield Strait and southern part of the Drake Passage using a method of statistical multidimensional analysis,” Polish Polar Research 8, 333–336 (1987).
The Antarctic Silverfish: A Keystone Species in a Changing Ecosystem, Ed. by M. Vacchi, E. Pisano, and L. Ghigliotti (Springer, Cham, 2017).
T. Wenneck, T. Falkenhaug, and O. A. Bergstad, “Strategies, methods, and technologies adopted on the R.V. G.O. Sars MAR-ECO expedition to the Mid-Atlantic Ridge in 2004,” Deep-Sea Res. II 55, 6–28 (2008). https://doi.org/10.1016/j.dsr2.2007.09.017
M. G. White and U. Piatkowski, “Abundance, horizontal and vertical distribution of fish in eastern Weddell Sea micronekton,” Polar Biol. 13, 41–53 (1993).
Wikipedia. Grtat-circle distance. https://en.wikipedia.org/ w/index.php?title=Great-circle_dist.
Funding
The study was carried out within state task no. FMWE-2022-0001.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kobyliansky, S.G., Mishin, A.V., Orlov, A.M. et al. Spatial Structure of Deep-Sea Pelagial Ichthyocenes in the Northern Weddell Sea (Powell Basin) and Brunsfield Strait in Summer 2020 and 2022. Oceanology 63, 574–588 (2023). https://doi.org/10.1134/S0001437023040070
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0001437023040070



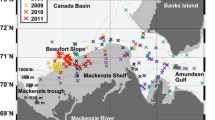

 , 2020; ♦, 2022; catch layers shown in color:
, 2020; ♦, 2022; catch layers shown in color:  , 400–0,
, 400–0,  , 500–0,
, 500–0,  , 1000–0,
, 1000–0,  , 1500–0,
, 1500–0,  , 2000–0 m.
, 2000–0 m.









