Abstract
Agencies reporting on disease outbreaks face many choices about what to report and the scale of its dissemination. Reporting impacts an epidemic by influencing individual decisions directly, and the social network in which they are made. We simulated a dynamic multiplex network model—with coupled infection and communication layers—to examine behavioral impacts from the nature and scale of epidemiological information reporting. We explored how adherence to protective behaviors (social distancing) can be facilitated through epidemiological reporting, social construction of perceived risk, and local monitoring of direct connections, but eroded via social reassurance. We varied reported information (total active cases, daily new cases, hospitalizations, hospital capacity exceeded, or deaths) at one of two scales (population level or community level). Total active and new case reporting at the population level were the most effective approaches, relative to the other reporting approaches. Case reporting, which synergizes with test-trace-and-isolate and vaccination policies, should remain a priority throughout an epidemic.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Key messages
-
How disease outbreaks are reported can alter public responses to an epidemic by influencing individual decisions, and alter the public’s social network
-
We modeled reporting with five distinct types of outbreak data at two different scales to simulate how reporting can alter behavior in a coupled dynamic multiplex network
-
Case reporting at the population level outperformed reporting of deaths, hospital capacity, and hospitalizations at the community level; we discuss important caveats.
Introduction
Epidemic mitigation is often contingent on individual adherence to public control measures (for example, for SARS-CoV or COVID-19, H5N1, H1N1, HIV) [1, 2]. Control measures often rely on dissemination of reliable information via reporting. Thus, reporting about risk and control measures has a critical role, especially in the early stages of a pandemic [1]. Priority should be placed on how to most effectively communicate information to mitigate an epidemic [3], especially by improving social adherence to protective behaviors. Agencies reporting on a disease outbreak face many choices regarding what to report, or what statistics to emphasize, when providing information to the general public [4, 5]. For example, should a reporting agency focus on the ultimate outcomes of infection (such as the number of people who have died or are hospitalized), or on the current case burden (such as the number of new cases each day, or the number of current active cases)? The scale of reporting is another key component. Should an agency focus on reporting local information for a specific community or instead place emphasis on trends over a wider region?
Public-facing health media outlets should tailor communicated information for risk reporting [6], as risk assessment is influenced by individual perception [7] and different forms of information can vary in evoking perceived risk [6, 8]. The importance of the information (and misinformation) available to people regarding their own personal risk of infection has been demonstrated to have the potential, at the very least, of influencing epidemic outcomes [3, 4, 9]. For reporting agencies, priority should be given to the data most likely to promote responsible and appropriate concern as well as the subsequent adoption of protective behaviors, rather than either incite panic or inadvertently encourage complacency [3, 4, 10]. Reporting agencies have the responsibility [11] and capacity to balance generating concern while lessening irrational fear, which was evident for COVID-19 via panic buying [12] and across social media [13]—although the risk of COVID-19 likely greatly outweighed potential damage due to panic [14].
People do not process information received from reports in isolation. Individuals vary in their adherence to interventions contingent on personal health risk factors [15], but may additionally modulate their own behavior based on that of friends [16, 17]. Each person’s perceived risk of infection depends on the health and behavior of those within their social community, and individuals often underestimate self-perceived risk [18] relative to others in their neighborhoods [15]. Humans exhibit biological avoidance responses to contagion such as emotional disgust, as well as innate abilities for detecting early signs of contagious sickness in others [19, 20]. Physiological responses to sickness—facial expressions, altered walking gait, and other visible signals—may, however, signal to others the need for help [21, 22]. These evolved person-to-person behavioral responses would not necessarily be elicited by mass media or abstract population health statistics, but perceived disease risk might be. Consequently, there are complex and non-linear impacts of reported information in the real world.
Reported case numbers may also inspire individuals to alter their behaviors, and observation of those behavioral changes may then influence others within their social network, especially those who might be less attentive to the direct reporting. For instance, Rosa and Jade may receive the same reports, but only Jade adheres to protective behavior based on the report. Upon observing this change, Rosa may be socially influenced to engage in more protective behaviors. Individuals may broadly perceive a behavioral pattern in their social groups, resulting in a perceived social norm [16]. The nature and scale of reporting can, therefore, have important consequences for both the adoption of beneficial behaviors, and then for a cascading spread through a social network, meaningfully impacting the course of an epidemic.
Researchers have indirectly quantified the spread of information seeking for influenza-like illnesses [23, 24] and protective behaviors [25] by following internet search trends. Recent work combined such an approach with measures of reporting, providing evidence of the beneficial effects of reporting [26]. Within districts of Germany, growing internet searches of ‘corona’ predicted subsequent local increases in infections, while reported case numbers predicted a local decrease in infections [26]. One interpretation might be that information seeking via internet searches is indicative of growing concern generated via awareness of increases in local exposures. Steiger et al. acknowledged the difficulty in interpreting what internet searches measure [26]. For instance, search results can overestimate disease prevalence [27] because searches, themselves, spread through social learning, algorithms that uprank popular searches, or both processes [28]. Even so, awareness and reporting are likely to be closely coupled [26].
Here, we explore how reporting may facilitate adherence to non-pharmaceutical interventions, not just through direct dissemination of information, but also through accelerating the social construction of perceived risk. This change occurs when reporting, or another social process, ‘tips’ some individuals into being supportive of protective behaviors and enables concern to spread dynamically through direct connections in their social network. Here, we incorporate parameters representing both social and biological contagion [29] using a simulated, coupled dynamic, multiplex network model to examine how the nature and scale of reporting potentially generate concern and a subsequent adherence to protective measures. Our model is an advance on earlier models, which have considered how precautionary behavior can spread ahead of the disease itself through social learning [30, 31], but which have not included a separable communication network and reporting function as we present here. Such an approach importantly unites behavioral responses to outbreaks [30,31,32] under a dynamic disease transmission network [33, 34], which includes a coupled information layer [35, 36], and reporting of outbreak data.
Methods
Base model
We extended a previously published stochastic model by adding a ‘reporter’ function. Specifics of the complete model’s construction are fully detailed elsewhere [37, 38] (also included in Supplementary Material). Briefly, we used multiplex social networks that we dynamically modeled through coupled infection and communication layers, which incrementally inform each other as time progressed [37, 38]. We simulated a closed population of 2000 individuals, broken into three age groups (children [24%], younger adults [63%], and older adults [13%]) within 10 communities of equal size (200 individuals). We randomly generated network structure and rewired into 9 distinct network structures with a 3 × 3 design for levels of homophily (absence of homophily, homophily in the communication layer, homophily in both layers) and levels of relative modularity [39] for the constructed communities (0.4 in both layers, 0.6 in both layers, 0.4 in the infection layer with 0.6 in the communication layer).
We used an epidemiological model identical to that of Silk et al. [38], also with parameter values adapted from recent publications documenting the COVID-19 pandemic (Supplementary Methods). Though we rely on disease parameters based on COVID-19, we intend the underlying principles of the coupled dynamics model design to extend to other respiratory diseases that can be effectively managed in similar ways. We included seven epidemiological compartments: susceptible (S), exposed (E), pre-symptomatic (I1), symptomatic (I2), hospitalized (I3), recovered (R), and dead (D).
We determined adherence to protective behaviors by a Bernoulli draw, contingent on a probability determined by an underlying concern trait. An individual’s concern responded, at each time point, to multiple features of their environment. As further detailed in previous work [37, 38], concern could be modified via social construction (individuals gain concern with a higher proportion of adherent connections), awareness (individuals gain concern with a higher proportion of direct connections that were symptomatic), and reassurance (individuals reduce in concern if none of their connections are infected). We selected values for these parameters based on previously published analyses [37, 38] that were quantitative in the model (Table 1). Here, we co-varied social construction and awareness, and set them as either strong (0.4) or weak (0.1). Reassurance, similarly, we defined as either strong (− 0.075) or weak (− 0.025).
Due to the synthesis of underlying theoretical frameworks necessary for this modeling approach, we made several assumptions. The justifications for these assumptions are detailed elsewhere [37, 38] (Supplementary Methods), but we also explore potential modifications in the Discussion. Given our prior work with this model, we did not dramatically alter the mechanics of the underlying model as this would require reanalysis and understanding of the base model (that included varying network structure, awareness, social construction, and reassurance), which now has a developed knowledge set that facilitates an accrued and informed understanding of how the system functions.
Reporting of epidemiological information
We added a ‘reporter’ function to the previous model specifications and summed, at each time-step, outputs of one of five types of reporting, either: total (active) cases, new cases since the previous time-step, current hospitalizations, hospital capacity exceeded, or deaths. We set hospital capacity at a value of 20, which per capita is between that of Japan and Germany, but higher than the United States [40]. We included a parameter for a ‘daily’ probability of a positive COVID test per time-step that varied for symptomatic individuals (0.02, 0.05, 0.10, 0.25, 0.50, 0.75, 1.00) (Supplementary Fig. 1, for cumulative probabilities). For asymptomatic testing, the probability was a constant function (0.2) of the probability of daily testing for symptomatic testing within each run. We included two probabilities for death being attributed to COVID (0.75, 1.00), while we always identified hospitalized patients as COVID positive (1.00).
The strength of response parameter represents the magnitude of risk perception invoked by reporting on the disease [6, 8], by either attenuating or amplifying the effect of the number of positive tests. To integrate strength of response, we aggregated positive tests at the two levels, community level or population level, and then multiplied those respective results by this scalar parameter. These modified numbers were then fed back into the model at the respective scale, either as community specific values or one population value aggregated from all 2000 individuals. Note that, for population-level reporting, we divided strength of response by the number of communities, effectively diffusing reporting among the communities. In each case, the result was then added to each individuals’ current concern within each community alone or within the entire population (see Supplementary Methods). To provide one example, a concern of 0.000 would result in a probability of being adherent of 0.500, a reporting value increase of 0.55 would then result in this probability changing to 0.634 at the subsequent time-step. We varied strength of response discontinuously between 0.001 and 10.0. Finally, we included a delay function to account for variation in latency of the collection and dissemination of information for reporting (1, 4, 7 time-steps).
Simulations
We conducted modeling in R3.6.3 [41] and provide all of the relevant code in the supplementary material, as well as on GitHub: https://github.com/matthewsilk/CoupledDynamics3_reporter. We present the general procedure of the model in Fig. 1. We simulated 768,960 uniquely independent runs with 4,608 parameter combinations across the 9 networks, with 10–20 replicates (Table 1). Replicates each started with a random selection of five individuals that were infected. Simulations continued for 300 time-steps or until epidemics died out, whichever came first. We assessed the mitigating effects of different parameters on the modeled epidemic curves visually by (a) a shift downward in the maximum infection peak and (b) a later peak. Both indicate a flattening in the epidemic peak. To facilitate discussion, we organized the values into relative categorical bins (Table 2).
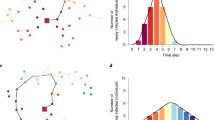
Visual summary of the model’s procedure. We simplified the network layers to 2 communities (400 nodes). The disease model [left] and reporting output [lower-right] show the results from a single exemplar run, averaged across communities. The concern plot [right] shows concern generation from a single run, averaged by community per time-step
Results
What is reported?
Even with high strength of response values, reporting deaths and dwindling hospital capacity was functionally ineffective at mitigating the epidemic for the population size investigated (Fig. 2; Supplementary Fig. 2). With reporting strength values ten times those of the maximum simulated for total active case and new case reporting, hospital capacity resulted in little discernible change in either the height of the infection peak or when it occurred, except for a small reduction at a very high strength of response. Similarly, reporting deaths resulted in a small shift at very high strengths of response. The reporting of hospitalizations had a small effect at moderate response strengths (Fig. 2; Supplementary Fig. 3). We observed the highest benefit of reporting, however, for that of total active and new cases. Notably, these benefits were only pronounced with weak awareness and social construction of concern. Consequently, Figs. 2, 3, and 4 have been constrained to presenting results with weak awareness and social construction.
Influence of reporting scale and type. We organize plots by row as the strength of response (strong [top], weak [bottom]). The x-axis shows a subset of strength of response values as a factor, while the y-axis is the peak infections per run. The boxes are filled according to type of reporting, while the outline corresponds to the scale of reporting. Delay and probability of testing are not fixed; awareness and social construction are fixed at 0.1
Total active case reporting proved most effective at mitigating the epidemic peak, even at low strengths of response. This was driven by the gross number of reported cases being much higher when reporting total active cases relative to new case reporting. The peak number for reported active cases were 6.64 (median, IQR 5.39–7.57) times higher than the peaks for reported new cases. New case reporting, however, required only a four-to-five times greater strength of response to gain an equivalent benefit in mitigating the epidemic peak (Fig. 3; Supplementary Fig. 4), indicating that the response to new cases was slightly more effective per case than the response to total active cases. We propose that this was caused by the peak in the number of new cases occurs earlier in the epidemic than the peak in the number of total active cases (Supplementary Fig. 5).
Impact of the reporting of total active cases relative to new cases. Active case reporting (both population and community level) is simplified as horizontal bars (median) and shading (interquartile range) for a low strength of response (0.05 top [orange online]) or moderate strength of response (0.20 bottom [red online]). We represent new case reporting by points at the median with whiskers for interquartile range. Points are colored by population- and community-level reporting. The x-axis shows strength of response as a factor. Awareness and social construction are set to 0.10; reassurance is set to strong; probability of testing was fixed at 0.25; delay was not fixed. See Supplementary 4 for weak reassurance
Scale of reporting
The best scale at which to report epidemiological information was contingent on what was being reported. For active case, death, and hospitalization reporting, a population-level scale was more effective than a community-level scale (Fig. 2; Supplementary Figs. 2 and 3). For new case reporting, however, there was a less pronounced distinction in the mitigating benefits of the two scales of reporting at low and negligible strengths of response. Finally, for hospital capacity, the community-level scale of reporting was more effective, relative to population-level reporting. Overall, population-level reporting was more, or equally, effective across the types of reporting except for reports of hospital capacities being exceeded, which had limited effectiveness anyway.
Assuming active case reporting with a negligible reporting effect and strong reassurance, communities that had infections emerge later in the run were hit more severely (Fig. 4a), in line with prior findings [37]. With a moderate testing probability and strength of response, however, community-level reporting negated the increased severity observed in late-hit communities (Fig. 4b). Population-level reporting completely reversed this phenomenon, such that communities where infections emerged later were hit less severely. This latter reversal effect was only visible with a strong reassurance effect. Increasing the strength of response while retaining a high testing probability removed this reversal effect (Fig. 4c), likely due to overall high effectiveness in reducing cases irrespective of scale. This was also apparent from the population-level reversal effect being reproducible when we retained high strengths of response, but lowered the testing probability. Strength of response and probability of testing interacted with the scale of reporting to alter the severity of late-hit, relative to early-hit, communities. Probability of testing had a largely synergistic relationship with strength of response (Supplementary Results). We should note that with a weak reassurance effect, later hit communities did not experience more severe epidemics in the first place (Fig. 4d). Though, with weak reassurance, late-hit communities with community-level reporting benefited from a high strength of response, but only with a low probability of testing (Fig. 4e).
Changes in severity of epidemic peaks across time over the ten communities. Reporting scale is indicated by the outline color (population-level = gray outline; community-level = black outline). Probability of testing is indicated by shading (light = 0.02, dark = 0.25). Each box shows coefficients of linear regressions for each run with a model of when peak infections occur, as predicted by the infection peaks. Thus, positive y-values indicate lower infection peaks in early-hit communities, while negative values indicate lower infection peaks in late-hit communities. The top row of plots illustrates strong reassurance, the bottom row weak reassurance. Left plots are for a negligible strength of response (0.01), the middle plots are for a low strength of response (0.10), the right plots show a moderate strength of response (0.50). Awareness and social construction are set to 0.10; delay was not fixed
Awareness, social construction, and reassurance
When awareness and social construction were strong, the benefit of reporting was generally not observable across the different levels and types of reporting (Fig. 5). The combined effect of strong social construction and awareness overwhelmed the benefit afforded by concern generated via reporting. Awareness and social construction were, consequently, fixed at weak values throughout the previous results on reporting.
Influence of awareness, social construction, and reassurance relative to active case reporting. Plots are organized by row as the level of reporting (community [top], population [bottom]), while columns as the value of awareness and social construction (0.1 [left], 0.4 [right]). The x-axis treats strength of response as a factor, while the y-axis is the peak infections per run. The boxes are colored according to Reassurance (strong [gray or blue online], weak [black or navy online]); delay was not fixed. Boxes with a strength of response < 0.75 show the results of 3780 runs, while the box with 0.75 shows 540 runs
If all else was equal, strong reassurance resulted in heightened infection peaks relative to weak reassurance (Fig. 5), as explored more fully in prior models [37, 38]. We found the benefit of reporting to be more pronounced with strong reassurance, such that equal steps in the strength of response resulted in a greater absolute change in the magnitude of infection peaks, relative to runs with weak reassurance. Consequently, runs with a strong reassurance experienced consistently higher peaks relative to weak reassurance; though this difference was reduced when reporting strengths were moderate or high, for active and new case reporting, respectively. Reporting deaths, hospitalizations, and hospital capacity were not strong enough, even with moderate to very high strengths of response, to alter the relative difference between the infection peaks of runs with strong versus weak reassurance (Supplementary Figs. 6, 7).
Discussion
Nature of reporting
The reporting of epidemiological information to the public has the potential to alter the shape of an epidemic curve through the generation of concern, which can promote adherence to protective behaviors. Here, we demonstrate the efficacy of total active and total new case reporting over reports of hospitalizations, hospital capacity, and deaths. While case reporting is often used for global and national surveillance, public health agencies should implement policies with forethought as to how this might shape public concern. The real-world generation of concern has been associated with media reporting based on surveys of H1N1 risk in the United Kingdom [10], though the authors emphasized the importance of timing for media reporting. Indeed, the effectiveness of reporting hospitalizations, hospital capacity, and deaths may be impeded by a lack of timeliness [42, 43] and, thus, any resulting change in behavior occurs too late to shape the initial epidemic. Public-facing health media outlets should time reporting to coincide with imminent increases in the local risk of contagion, to avoid reassurance or a unintended public perceptions of sensationalism [10]. Furthermore, concern and adherence to protective behaviors may be most effectively generated during a behaviorally receptive phase [37].
Scale of reporting
Overall, reporting at the population level had greater efficacy than reporting at the community level. Timing was, in part, why population-level reporting was effective, with a disproportionate mitigation benefit in late-hit communities. Previous work suggests that the onset of reassurance can erode initially high levels of concern in communities that are not yet incubating infections—or where substantial infections exist but are not yet fully reported (e.g., [44,45,46])—and result in higher epidemic peaks [37]. Consequently, public health agencies should practice care in balancing public concern and reassurance in the absence of infection risk. As with timing of reporting, over-reporting may create a perception of sensationalism, as posited for public health responses to or media coverage of H1N1 [47, 48] and H5N1 [6]. Unfortunately, a formal analysis of United States newspaper coverage of H5N1 found that the media relied heavily on loaded terminology and worst-case scenarios [6]. This may erode trust [48], an intervening variable of interest [49, 50], which could skew responsiveness to case reporting. Public-facing health media outlets could effectively increase initial concern by synchronizing reports with impending disease risk based on rates in neighboring communities. Similarly, altering initial concern of our simulated population, without reporting [37], often facilitates a rapid mitigating response.
Communication with the public
An intervening variable not modeled here is how people differ in understanding numeric measures. Generally, objective measures can be informative in understanding disease risk, and may reduce some types of media reporting bias [51], but such an effect is not guaranteed [3, 52]. Some demographics or cultures may respond more strongly to framings of economic or social risk [1, 53], rather than disease risk. Case numbers often elicit concern via negative framing [18], and may be challenging for less numerate members of the public to contextualize [18, 54]. This latter point is important as 7.8% of Americans cannot complete one-step numeracy problems in English, which includes assessments of counting ability [55]. Numeracy has been show to interact with risk magnitude, such that more numerate individuals are more likely to be concerned with a higher magnitude of risk (that is, disease developing in 500 of 1000, versus 50 of 1000 people) [56]. Research has also linked level of education to knowledge regarding the symptoms and transmission of respiratory disease [57]. Public-facing health media outlets should contextualize reporting in a relatable framework considerate of the diverse backgrounds and skills that different members of the public possess [3, 5]. Publishing untargeted or comprehensive information may not provide an accruing benefit for less numerate individuals and could instead alienate members of the public. Public-facing health media outlets should give priority to how information can be disseminated simply, but effectively, to encourage the adoption of protective behaviors [5].
Awareness, social construction, and reassurance
Our model emphasized the importance of awareness and social construction of concern for mitigating the epidemic. Here, strong awareness and social construction showed a greater contribution than even the strongest performing type of reporting. This observation parallels data on public perceptions of H1N1 in Canada [48], where 58.91% of polled respondents said they would be concerned ‘when one or more of their direct contacts became infected,’ while only 27.98% said ‘when infection was reported in ≥ 10% of the general populace’ [48]. More respondents anticipated that awareness of the disease in their direct network would have a greater influence on their concern than reported case numbers. Also of note is the role of reassurance, which we used, here, to simulate the erosion of concern over time within a social context. Such an effect has been documented via surveys [10, 58]. For example, interviews of 4047 Italian respondents indicated that their concern of H1N1, their perceived risk of infection, and their use of protective behaviors all declined over time along with the actual risk of infection [58]. Here, moderate reporting generated concern sufficient to counteract the effects of strong reassurance, such that epidemic peaks were similar to those in runs with weak reassurance. Future research would benefit from comparing the role of reporting in communities with disparate tendencies for reassurance.
Adapting to shifting conditions
We recognize that the intersection between social and health behavior has dramatically shifted throughout the course of the pandemic. For instance, we now have numerous highly effective vaccines [59], which have been released concurrent with a swell in public belief in and engagement with misinformation [60]. Although vaccine effectiveness is robust against hospitalizations and deaths, protection against infection and transmission is not complete [59]. This is further complicated by growing concerns about waning immunity [61], and varying vaccine uptake across demographic groups [62]. These dynamics result in complex interactions. For example, how does vaccine uptake or willingness to accept misinformation intersect with concern-generating processes, especially in the face of future pandemic waves? High vaccine uptake decreases transmission, but also might reduce awareness and, thus, prevent rapid adherence in response to local outbreaks. Individuals who do not practice social distancing might also be unwilling to vaccinate, but could gain natural immunity. Such dynamics, therefore, likely require competing information flow or network layers, with individual properties or identities altering the probability of information and vaccine acceptance. Future research would benefit from altering our model construction, or building similar models that utilize multiplex information and disease layers, either relying on simulations or by expanding to include data-based analyses for further inference of these complex dynamics.
Conclusion
Presently, some nations are transitioning from a surging COVID-19 pandemic, managed primarily by protective behaviors, to a percentage of vaccinated residents facing sustained low-case numbers and a continued risk of variants. In areas where vaccination rates are low, however, public health agencies and public-facing health media outlets are primarily responsible for recommending public practices to manage COVID-19 outbreaks through the use of protective behaviors. In the absence of reporting, public reassurance is much more likely to decrease concern, and so reduce the efficacy of protective behaviors prior to new increases in risk. An individual’s awareness of past pandemics has been associated with greater awareness and knowledge of newer viral risks [63]. However, subsequent increases in concern may be stunted [10] and the persistent use of preventative behaviors through subsequent waves may vary [64].
Case reporting can occur in tandem with test-trace-and-isolate policies which effectively synergize with vaccination efforts [65]. Sustained testing is vital for test-trace-isolate programs [66] and can facilitate continued reporting to mitigate future outbreaks. In a recent review of COVID-19 cases in the United States, Li et al. [67] emphasized that public information campaigns have a strong effect on reducing case growth rates relative to other policies, including closures, event cancellations, and movement restrictions [67]. Public information campaigns often extend beyond simply reporting infection status and numerical data, but we emphasize that reporting can have indirect benefits outside of direct policy health action. Thus, we recommend that all aspects of the multi-modal dissemination of information remain a research priority to amplify the effectiveness of reports, which may not necessarily be government run [68, 69].
Data availability
The code has been made freely available online: https://github.com/matthewsilk/CoupledDynamics3_reporter
References
Leppin A, Aro AR. Risk perceptions related to SARS and avian influenza: theoretical foundations of current empirical research. Int J Behav Med. 2009;16(1):7–29.
Abrams JE. “Spitting is dangerous, indecent, and against the law!” legislating health behavior during the American tuberculosis crusade. J Hist Med Allied Sci. 2013;68(3):416–50.
Winograd DM, Fresquez CL, Egli M, Peterson EK, Lombardi AR, Megale A, et al. Rapid review of virus risk communication interventions: directions for COVID-19. Patient Educ Couns. 2021;104(8):1834–59.
Coppola V, Camus O. Preventing without stigmatizing: The complex stakes of information on AIDS. Patient Educ Couns. 2007;67(3):255–60.
Freeman ALJ, Kerr J, Recchia G, Schneider CR, Lawrence ACE, Finikarides L, et al. Communicating personalized risks from COVID-19: guidelines from an empirical study. R Soc Open Sci. 2021. https://doi.org/10.1098/rsos.201721.
Dudo AD, Dahlstrom MF, Brossard D. Reporting a potential pandemic: a risk-related assessment of Avian Influenza coverage in U.S. newspapers. Sci Commun. 2007;28(4):429–54.
Freudenburg WR. Perceived risk, real risk: Social science and the art of probabilistic risk assessment. Science. 1988;242(4875):44–9.
Niu C, Jiang Z, Liu H, Yang K, Song X, Li Z. The influence of media consumption on public risk perception: a meta-analysis. J Risk Res. 2020;13:1–27.
Zhong W. Simulating influenza pandemic dynamics with public risk communication and individual responsive behavior. Comput Math Organ Theory. 2017;23(4):475–95.
Rubin GJ, Potts HWW, Michie S. The impact of communications about swine flu (influenza A H1N1v) on public responses to the outbreak: results from 36 national telephone surveys in the UK. Health Technol Assess. 2010;14(34):183–266.
The LANCET. COVID-19: fighting panic with information. Lancet. 2020;395(10224):537.
Islam T, Pitafi AH, Arya V, Wang Y, Akhtar N, Mubarik S, et al. Panic buying in the COVID-19 pandemic: a multi-country examination. J Retail Consum Serv. 2021;59:102357.
Depoux A, Martin S, Karafillakis E, Preet R, Wilder-Smith A, Larson H. The pandemic of social media panic travels faster than the COVID-19 outbreak. J Trav Med. 2020. https://doi.org/10.1093/jtm/taaa031.
Drury J, Reicher S, Stott C. COVID-19 in context: why do people die in emergencies? It’s probably not because of collective psychology. Br J Soc Psychol. 2020;59(3):686–93.
Wise T, Zbozinek TD, Michelini G, Hagan CC, Mobbs D. Changes in risk perception and self-reported protective behaviour during the first week of the COVID-19 pandemic in the United States. R Soc Open Sci. 2020;7(9):200742.
Dickie R, Rasmussen S, Cain R, Williams L, MacKay W. The effects of perceived social norms on handwashing behaviour in students. Psychol Health Med. 2018;23(2):154–9.
Christakis NA, Fowler JH. Social contagion theory: examining dynamic social networks and human behavior. Stat Med. 2013;32(4):556–77.
Bavel JJV, Baicker K, Boggio PS, Capraro V, Cichocka A, Cikara M, et al. Using social and behavioural science to support COVID-19 pandemic response. Nat Hum Behav. 2020;4(5):460–71.
Axelsson J, Sundelin T, Olsson MJ, Sorjonen K, Axelsson C, Lasselin J, et al. Identification of acutely sick people and facial cues of sickness. Proc R Soc B Biol Sci. 1870;2018(285):20172430.
Sundelin T, Karshikoff B, Axelsson E, Höglund CO, Lekander M, Axelsson J. Sick man walking: Perception of health status from body motion. Brain Behav Immun. 2015;48:53–6.
Steinkopf L. The signaling theory of symptoms: an evolutionary explanation of the placebo effect. Evol Psychol. 2015;13(3):1474704915600559.
Schrock JM, Snodgrass JJ, Sugiyama LS. Lassitude: the emotion of being sick. Evol Hum Behav. 2020;41(1):44–57.
Kandula S, Shaman J. Reappraising the utility of Google flu trends. PLoS Comput Biol. 2019;15(8):e1007258.
Ginsberg J, Mohebbi MH, Patel RS, Brammer L, Smolinski MS, Brilliant L. Detecting influenza epidemics using search engine query data. Nature. 2009;457(7232):1012–4.
Lin YH, Liu CH, Chiu YC. Google searches for the keywords of “wash hands” predict the speed of national spread of COVID-19 outbreak among 21 countries. Brain Behav Immun. 2020;1(87):30–2.
Steiger E, Mussgnug T, Kroll LE. Causal graph analysis of COVID-19 observational data in German districts reveals effects of determining factors on reported case numbers. PLoS ONE. 2021;16(5):e0237277.
Lazer D, Kennedy R, King G, Vespignani A. The parable of google flu: traps in big data analysis. Science. 2014;343(6176):1203–5.
Bentley RA, Ormerod P. A rapid method for assessing social versus independent interest in health issues: a case study of ‘bird flu’ and ‘swine flu.’ Soc Sci Med. 2010;71(3):482–5.
Bauch CT, Galvani AP. Social and biological contagions. Science. 2013;342(6154):47–9.
Funk S, Gilad E, Watkins C, Jansen VA. The spread of awareness and its impact on epidemic outbreaks. Proc Natl Acad Sci. 2009;106(16):6872–7.
Funk S, Salathé M, Jansen VA. Modelling the influence of human behaviour on the spread of infectious diseases: a review. J R Soc Interface. 2010;7(50):1247–56.
Campbell E, Salathé M. Complex social contagion makes networks more vulnerable to disease outbreaks. Sci Rep. 2013;3(1):1905.
Zhang J, Yang C, Jin Z, Li J. Dynamics analysis of SIR epidemic model with correlation coefficients and clustering coefficient in networks. J Theor Biol. 2018;14(449):1–13.
Valdez LD, Braunstein LA, Havlin S. Epidemic spreading on modular networks: the fear to declare a pandemic. Phys Rev E. 2020;101(3): 032309.
Lynn CW, Papadopoulos L, Kahn AE, Bassett DS. Human information processing in complex networks. Nat Phys. 2020;16(9):965–73.
Bakshy E, Rosenn I, Marlow C, Adamic L. The role of social networks in information diffusion. In: Proceedings of the 21st international conference on World Wide Web. New York, NY: Association for Computing Machinery; 2012. p. 519–28. (WWW ’12). https://doi.org/10.1145/2187836.2187907
Silk MJ, Carrignon S, Bentley RA, Fefferman NH. Improving pandemic mitigation policies across communities through coupled dynamics of risk perception and infection. Proc R Soc B Biol Sci. 1955;2021(288):20210834.
Silk MJ, Carrignon S, Bentley RA, Fefferman NH. Observations and conversations: how communities learn about infection risk can impact the success of non-pharmaceutical interventions against epidemics. BMC Public Health. 2022;22(1):13.
Sah P, Leu ST, Cross PC, Hudson PJ, Bansal S. Unraveling the disease consequences and mechanisms of modular structure in animal social networks. Proc Natl Acad Sci. 2017;114(16):4165–70.
World Bank. Hospital beds (per 1000 people) [Data Set]. 2021. Report No.: SH.MED.BEDS.ZS. World Bank. https://data.worldbank.org/indicator/SH.MED.BEDS.ZS. Accessed 15 Jun 2021
R Core Team. R: a language and environment for statistical computing. Vienna, Austria: (Version 3.6.3) [Computer software] R Foundation for Statistical Computing; 2019. https://www.R-project.org/
Donovan S, Brett-Major D, Lawler JV. Lag between COVID-19 cases and subsequent deaths (preprint). Research Square. 2021. https://www.researchsquare.com/article/rs-434947/v2. Accessed 14 Sep 2021
Lipsitch M, Donnelly CA, Fraser C, Blake IM, Cori A, Dorigatti I, et al. Potential biases in estimating absolute and relative case-fatality risks during outbreaks. PLoS Neglect Trop Dis. 2015;9(7):e0003846.
Lau H, Khosrawipour T, Kocbach P, Ichii H, Bania J, Khosrawipour V. Evaluating the massive underreporting and undertesting of COVID-19 cases in multiple global epicenters. Pulmonology. 2021;27(2):110–5.
Li R, Pei S, Chen B, Song Y, Zhang T, Yang W, et al. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV-2). Science. 2020;368(6490):489–93.
Ruck DJ, Bentley RA, Borycz J. Early warning of vulnerable counties in a pandemic using socio-economic variables. Econ Hum Biol. 2021;1(41): 100988.
Briand S, Mounts A, Chamberland M. Challenges of global surveillance during an influenza pandemic. Public Health. 2011;125(5):247–56.
Taha SA, Matheson K, Anisman H. The 2009 H1N1 influenza pandemic: the role of threat, coping, and media trust on vaccination intentions in Canada. J Health Commun. 2013;18(3):278–90.
Freimuth VS, Musa D, Hilyard K, Quinn SC, Kim K. Trust during the early stages of the 2009 H1N1 pandemic. J Health Commun. 2014;19(3):321–39.
Elledge BL, Brand M, Regens JL, Boatright DT. Implications of public understanding of Avian influenza for fostering effective risk communication. Health Promot Pract. 2008;9(Suppl. 4):54S-59S.
Young ME, Norman GR, Humphreys KR. Medicine in the popular press: the influence of the media on perceptions of disease. PLoS ONE. 2008;3(10):e3552.
Wachinger G, Renn O, Begg C, Kuhlicke C. The risk perception paradox: implications for governance and communication of natural hazards. Risk Anal. 2013;33(6):1049–65.
Dryhurst S, Schneider CR, Kerr J, Freeman ALJ, Recchia G, van der Bles AM, et al. Risk perceptions of COVID-19 around the world. J Risk Res. 2020;23(7–8):994–1006.
Peters E, Västfjäll D, Slovic P, Mertz CK, Mazzocco K, Dickert S. Numeracy and decision making. Psychol Sci. 2006;17(5):407–13.
Mamedova S, Pawlowski E. Adult Numeracy in the United States. Program for the International Assessment of Adult Competencies (PIAAC); 2020. Report No. NCES 2020025. https://nces.ed.gov/pubsearch/pubsinfo.asp?pubid=2020025
Wright AJ, Whitwell SCL, Takeichi C, Hankins M, Marteau TM. The impact of numeracy on reactions to different graphic risk presentation formats: an experimental analogue study. Br J Health Psychol. 2009;14(1):107–25.
Savoia E, Testa MA, Viswanath K. Predictors of knowledge of H1N1 infection and transmission in the U.S. population. BMC Public Health. 2012;12(1):328.
Ferrante G, Baldissera S, Moghadam PF, Carrozzi G, Trinito MO, Salmaso S. Surveillance of perceptions, knowledge, attitudes and behaviors of the Italian adult population (18–69 years) during the 2009–2010 A/H1N1 influenza pandemic. Eur J Epidemiol. 2011;26(3):211–9.
Zheng C, Shao W, Chen X, Zhang B, Wang G, Zhang W. Real-world effectiveness of COVID-19 vaccines: a literature review and meta-analysis. Int J Infect Dis. 2022;1(114):252–60.
Agley J, Xiao Y. Misinformation about COVID-19: evidence for differential latent profiles and a strong association with trust in science. BMC Public Health. 2021;21(1):89.
Pérez-Alós L, Armenteros JJA, Madsen JR, Hansen CB, Jarlhelt I, Hamm SR, et al. Modeling of waning immunity after SARS-CoV-2 vaccination and influencing factors. Nat Commun. 2022;13(1):1614.
Viswanath K, Bekalu M, Dhawan D, Pinnamaneni R, Lang J, McLoud R. Individual and social determinants of COVID-19 vaccine uptake. BMC Public Health. 2021;21(1):818.
Lin L, McCloud RF, Bigman CA, Viswanath K. Tuning in and catching on? Examining the relationship between pandemic communication and awareness and knowledge of MERS in the USA. J Public Health. 2017;39(2):282–9.
Lau JTF, Yang X, Tsui H, Kim JH. Monitoring community responses to the SARS epidemic in Hong Kong: from day 10 to day 62. J Epidemiol Commun Health. 2003;57(11):864–70.
Contreras S, Dehning J, Mohr SB, Bauer S, Spitzner FP, Priesemann V. Low case numbers enable long-term stable pandemic control without lockdowns. 15 Apr 2021. http://arxiv.org/abs/2011.11413. Accessed 9 Jun 2021
Grantz KH, Lee EC, D’Agostino McGowan L, Lee KH, Metcalf CJE, Gurley ES, et al. Maximizing and evaluating the impact of test-trace-isolate programs: a modeling study. PLoS Med. 2021;18(4):e1003585.
Li Y, Li M, Rice M, Zhang H, Sha D, Li M, et al. The impact of policy measures on human mobility, COVID-19 cases, and mortality in the US: a spatiotemporal perspective. Int J Environ Res Public Health. 2021;18(3):996.
Field E, Dyda A, Lau CL. COVID-19 real-time information system for preparedness and epidemic response (CRISPER). Med J Aust. 2021;214(8):386–386.
Karimuribo ED, Mutagahywa E, Sindato C, Mboera L, Mwabukusi M, Njenga MK, et al. A smartphone app (AfyaData) for innovative One Health disease surveillance from community to national levels in Africa: intervention in disease surveillance. JMIR Public Health Surveill. 2017;3(4):e94.
Acknowledgements
We would like to acknowledge Dave Hodgson for his support of MJS’ contributions.
Funding
This work was supported by the National Science Foundation under DEB Grant #2028710.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pritchard, A.J., Silk, M.J., Carrignon, S. et al. How reported outbreak data can shape individual behavior in a social world. J Public Health Pol 43, 360–378 (2022). https://doi.org/10.1057/s41271-022-00357-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1057/s41271-022-00357-7